Current Concepts and Methods in Tissue Interface Scaffold Fabrication
Abstract
1. Introduction
2. Tissue Types
2.1. Bone
2.2. Cartilage
2.3. Ligament
2.4. Muscle
2.5. Skin
2.6. Tendons
3. Scaffold Fabrication Techniques
3.1. 3D-Printing
3.2. Bioprinting
3.3. Electrospinning
3.4. Gas Foaming
3.5. Hydrogels
3.6. Cryogels
3.7. Solvent Casting and Particulate Leaching
4. Materials and Additives
5. Combined Scaffold Fabrication Techniques
5.1. Drugs and Biomedical Tools
5.2. Multiple Scaffold Fabrication Methods for a Single Tissue
5.3. Combined Scaffolds Targeting Multiple Tissue Types
6. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Kim, Y.S.; Smoak, M.M.; Melchiorri, A.J.; Mikos, A.G. An Overview of the Tissue Engineering Market in the United States from 2011 to 2018. Tissue Eng. Part A 2019, 25, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Planat-Benard, V.; Varin, A.; Casteilla, L. MSCs and Inflammatory Cells Crosstalk in Regenerative Medicine: Concerted Actions for Optimized Resolution Driven by Energy Metabolism. Front. Immunol. 2021, 12, 626755. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Wang, L.; Pan, W.; Yang, F.; Jiang, W.; Wu, X.; Kong, X.; Dai, K.; Hao, Y. In vitro and in vivo study of additive manufactured porous Ti6Al4V scaffolds for repairing bone defects. Sci. Rep. 2016, 6, 34072. [Google Scholar] [CrossRef] [PubMed]
- Bose, S.; Roy, M.; Bandyopadhyay, A. Recent advances in bone tissue engineering scaffolds. Trends Biotechnol. 2012, 30, 546–554. [Google Scholar] [CrossRef]
- Valdoz, J.C.; Johnson, B.C.; Jacobs, D.J.; Franks, N.A.; Dodson, E.L.; Sanders, C.; Cribbs, C.G.; Van Ry, P.M. The ECM: To Scaffold, or Not to Scaffold, That Is the Question. Int. J. Mol. Sci. 2021, 22, 12690. [Google Scholar] [CrossRef]
- O’Brien, F.J. Biomaterials & scaffolds for tissue engineering. Mater. Today 2011, 14, 88–95. [Google Scholar] [CrossRef]
- Zhang, H.; Zhou, L.; Zhang, W. Control of scaffold degradation in tissue engineering: A review. Tissue Eng. Part. B Rev. 2014, 20, 492–502. [Google Scholar] [CrossRef]
- Woodruff, M.A.; Lange, C.; Reichert, J.; Berner, A.; Chen, F.; Fratzl, P.; Schantz, J.-T.; Hutmacher, D.W. Bone tissue engineering: From bench to bedside. Mater. Today 2012, 15, 430–435. [Google Scholar] [CrossRef]
- Liu, Y.; Luo, D.; Wang, T. Hierarchical Structures of Bone and Bioinspired Bone Tissue Engineering. Small 2016, 12, 4611–4632. [Google Scholar] [CrossRef]
- Manolagas, S.C. Birth and Death of Bone Cells: Basic Regulatory Mechanisms and Implications for the Pathogenesis and Treatment of Osteoporosis. Endocr. Rev. 2000, 21, 115–137. [Google Scholar] [CrossRef]
- Dempster, D.W. Exploiting and Bypassing the Bone Remodeling Cycle to Optimize the Treatment of Osteoporosis. J. Bone Mineral Res. 1997, 12, 1152–1154. [Google Scholar] [CrossRef]
- Langdahl, B.; Ferrari, S.; Dempster, D.W. Bone modeling and remodeling: Potential as therapeutic targets for the treatment of osteoporosis. Ther. Adv. Musculoskelet. Dis. 2016, 8, 225–235. [Google Scholar] [CrossRef]
- Dempster, D.W.; Lindsay, R. Pathogenesis of osteoporosis. Lancet 1993, 341, 797–801. [Google Scholar] [CrossRef]
- Qu, H.; Fu, H.; Han, Z.; Sun, Y. Biomaterials for bone tissue engineering scaffolds: A review. RSC Adv. 2019, 9, 26252–26262. [Google Scholar] [CrossRef]
- Stevens, M.M. Biomaterials for bone tissue engineering. Mater. Today 2008, 11, 18–25. [Google Scholar] [CrossRef]
- Koons, G.L.; Diba, M.; Mikos, A.G. Materials design for bone-tissue engineering. Nat. Rev. Mater. 2020, 5, 584–603. [Google Scholar] [CrossRef]
- Rouwkema, J.; Rivron, N.C.; van Blitterswijk, C.A. Vascularization in tissue engineering. Trends Biotechnol. 2008, 26, 434–441. [Google Scholar] [CrossRef]
- Murphy, C.M.; Haugh, M.G.; O’Brien, F.J. The effect of mean pore size on cell attachment, proliferation and migration in collagen–glycosaminoglycan scaffolds for bone tissue engineering. Biomaterials 2010, 31, 461–466. [Google Scholar] [CrossRef]
- Woodard, J.R.; Hilldore, A.J.; Lan, S.K.; Park, C.J.; Morgan, A.W.; Eurell, J.A.C.; Clark, S.G.; Wheeler, M.B.; Jamison, R.D.; Wagoner Johnson, A.J. The mechanical properties and osteoconductivity of hydroxyapatite bone scaffolds with multi-scale porosity. Biomaterials 2007, 28, 45–54. [Google Scholar] [CrossRef]
- Hixon, K.R.; Eberlin, C.T.; Lu, T.; Neal, S.M.; Case, N.D.; McBride-Gagyi, S.H.; Sell, S.A. The calcification potential of cryogel scaffolds incorporated with various forms of hydroxyapatite for bone regeneration. Biomed. Mater. 2017, 12, 025005. [Google Scholar] [CrossRef]
- Tatari, H. The structure, physiology, and biomechanics of articular cartilage: Injury and repair. Acta Orthop. Traumatol. Turc. 2007, 41 (Suppl. S2), 1–5. [Google Scholar]
- Neufeld, D.A. Bone healing after amputation of mouse digits and newt limbs: Implications for induced regeneration in mammals. Anat. Rec. 1985, 211, 156–165. [Google Scholar] [CrossRef]
- Mansour, J.M. Biomechanics of Cartilage. In Kinesiology: The Mechanics and Pathomechanics of Human Movement, 2nd ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2013; pp. 66–75. [Google Scholar]
- Chang, L.R.; Marston, G.; Martin, A. Anatomy, Cartilage. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2022. [Google Scholar]
- Loy, B.N.; Zimel, M.; Gowda, A.L.; Tooley, T.R.; Maerz, T.; Bicos, J.; Guettler, J. A Biomechanical and Structural Comparison of Articular Cartilage and Subchondral Bone of the Glenoid and Humeral Head. Orthop. J. Sports Med. 2018, 6, 2325967118785854. [Google Scholar] [CrossRef]
- Żylińska, B.; Silmanowicz, P.; Sobczyńska-Rak, A.; Jarosz, Ł.; Szponder, T. Treatment of Articular Cartilage Defects: Focus on Tissue Engineering. In Vivo 2018, 32, 1289–1300. [Google Scholar] [CrossRef]
- Richardson, J.B.; Caterson, B.; Evans, E.H.; Ashton, B.A.; Roberts, S. Repair of human articular cartilage after implantation of autologous chondrocytes. J. Bone Jt. Surg. Br. Vol. 1999, 81-B, 1064–1068. [Google Scholar] [CrossRef]
- Zellner, J.; Krutsch, W.; Pfeifer, C.G.; Koch, M.; Nerlich, M.; Angele, P. Autologous chondrocyte implantation for cartilage repair: Current perspectives. Orthop. Res. Rev. 2015, 7, 149–158. [Google Scholar] [CrossRef]
- Bexkens, R.; Ogink, P.T.; Doornberg, J.N.; Kerkhoffs, G.; Eygendaal, D.; Oh, L.S.; van den Bekerom, M.P.J. Donor-site morbidity after osteochondral autologous transplantation for osteochondritis dissecans of the capitellum: A systematic review and meta-analysis. Knee Surg. Sports Traumatol. Arthrosc. 2017, 25, 2237–2246. [Google Scholar] [CrossRef]
- Lien, S.-M.; Ko, L.-Y.; Huang, T.-J. Effect of pore size on ECM secretion and cell growth in gelatin scaffold for articular cartilage tissue engineering. Acta Biomater. 2009, 5, 670–679. [Google Scholar] [CrossRef]
- Musahl, V.; Karlsson, J. Anterior Cruciate Ligament Tear. N. Engl. J. Med. 2019, 380, 2341–2348. [Google Scholar] [CrossRef]
- Laurencin, C.T.; Freeman, J.W. Ligament tissue engineering: An evolutionary materials science approach. Biomaterials 2005, 26, 7530–7536. [Google Scholar] [CrossRef]
- Ballock, R.T.; Woo, S.L.-Y.; Lyon, R.M.; Hollis, J.M.; Akeson, W.H. Use of patellar tendon autograft for anterior cruciate ligament reconstruction in the rabbit: A long-term histologic and biomechanical study. J. Orthop. Res. 1989, 7, 474–485. [Google Scholar] [CrossRef] [PubMed]
- Krause, M.; Freudenthaler, F.; Frosch, K.H.; Achtnich, A.; Petersen, W.; Akoto, R. Operative Versus Conservative Treatment of Anterior Cruciate Ligament Rupture. Dtsch. Arztebl. Int. 2018, 115, 855–862. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Smietana, M.J.; Kostrominova, T.Y.; Wojtys, E.M.; Larkin, L.M.; Arruda, E.M. Three-Dimensional Engineered Bone–Ligament–Bone Constructs for Anterior Cruciate Ligament Replacement. Tissue Eng. Part A 2012, 18, 103–116. [Google Scholar] [CrossRef] [PubMed]
- Jackson, D.W.; Grood, E.S.; Goldstein, J.D.; Rosen, M.A.; Kurzweil, P.R.; Cummings, J.F.; Simon, T.M. A comparison of patellar tendon autograft and allograft used for anterior cruciate ligament reconstruction in the goat model. Am. J. Sports Med. 1993, 21, 176–185. [Google Scholar] [CrossRef]
- Chen, J.; Altman, G.H.; Karageorgiou, V.; Horan, R.; Collette, A.; Volloch, V.; Colabro, T.; Kaplan, D.L. Human bone marrow stromal cell and ligament fibroblast responses on RGD-modified silk fibers. J. Biomed. Mater. Res. Part A 2003, 67A, 559–570. [Google Scholar] [CrossRef]
- Vunjak-Novakovic, G.; Altman, G.; Horan, R.; Kaplan, D.L. Tissue engineering of ligaments. Annu. Rev. Biomed. Eng. 2004, 6, 131–156. [Google Scholar] [CrossRef]
- Konikoff, J.J. Development of a single stage active tendon prosthesis, I: Distal end attachment. J. Bone Joint Surg Am. 1974, 56, 848. [Google Scholar]
- Noyes, F.R.; Grood, E.S. The strength of the anterior cruciate ligament in humans and Rhesus monkeys. J. Bone Joint Surg Am. 1976, 58, 1074–1082. [Google Scholar] [CrossRef]
- Woo, S.L.; Hollis, J.M.; Adams, D.J.; Lyon, R.M.; Takai, S. Tensile properties of the human femur-anterior cruciate ligament-tibia complex. The effects of specimen age and orientation. Am. J. Sports Med. 1991, 19, 217–225. [Google Scholar] [CrossRef]
- Silva, M.; Ferreira, F.N.; Alves, N.M.; Paiva, M.C. Biodegradable polymer nanocomposites for ligament/tendon tissue engineering. J. Nanobiotechnol. 2020, 18, 23. [Google Scholar] [CrossRef]
- Kwee, B.J.; Mooney, D.J. Biomaterials for skeletal muscle tissue engineering. Curr. Opin. Biotechnol. 2017, 47, 16–22. [Google Scholar] [CrossRef]
- Mukund, K.; Subramaniam, S. Skeletal muscle: A review of molecular structure and function, in health and disease. Wiley Interdiscip. Rev. Syst. Biol. Med. 2020, 12, e1462. [Google Scholar] [CrossRef]
- Campbell, N.J.; Maani, C.V. Histology, Muscle. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2022. [Google Scholar]
- Langridge, B.; Griffin, M.; Butler, P.E. Regenerative medicine for skeletal muscle loss: A review of current tissue engineering approaches. J. Mater. Sci. Mater. Med. 2021, 32, 15. [Google Scholar] [CrossRef]
- Dave, H.D.; Shook, M.; Varacallo, M. Anatomy, Skeletal Muscle. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2022. [Google Scholar]
- Urciuolo, A.; Quarta, M.; Morbidoni, V.; Gattazzo, F.; Molon, S.; Grumati, P.; Montemurro, F.; Tedesco, F.S.; Blaauw, B.; Cossu, G.; et al. Collagen VI regulates satellite cell self-renewal and muscle regeneration. Nat. Commun. 2013, 4, 1964. [Google Scholar] [CrossRef]
- Bružauskaitė, I.; Bironaitė, D.; Bagdonas, E.; Bernotienė, E. Scaffolds and cells for tissue regeneration: Different scaffold pore sizes-different cell effects. Cytotechnology 2016, 68, 355–369. [Google Scholar] [CrossRef]
- Hofemeier, A.D.; Limon, T.; Muenker, T.M.; Wallmeyer, B.; Jurado, A.; Afshar, M.E.; Ebrahimi, M.; Tsukanov, R.; Oleksiievets, N.; Enderlein, J.; et al. Global and local tension measurements in biomimetic skeletal muscle tissues reveals early mechanical homeostasis. eLife 2021, 10, e60145. [Google Scholar] [CrossRef]
- Loreti, M.; Sacco, A. The jam session between muscle stem cells and the extracellular matrix in the tissue microenvironment. NPJ Regen. Med. 2022, 7, 16. [Google Scholar] [CrossRef]
- Piipponen, M.; Li, D.; Landén, N.X. The Immune Functions of Keratinocytes in Skin Wound Healing. Int. J. Mol. Sci. 2020, 21, 8790. [Google Scholar] [CrossRef]
- Sierra-Sánchez, Á.; Kim, K.H.; Blasco-Morente, G.; Arias-Santiago, S. Cellular human tissue-engineered skin substitutes investigated for deep and difficult to heal injuries. NPJ Regen. Med. 2021, 6, 35. [Google Scholar] [CrossRef]
- Dick, M.K.; Miao, J.H.; Limaiem, F. Histology, Fibroblast. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2022. [Google Scholar]
- Tottoli, E.M.; Dorati, R.; Genta, I.; Chiesa, E.; Pisani, S.; Conti, B. Skin Wound Healing Process and New Emerging Technologies for Skin Wound Care and Regeneration. Pharmaceutics 2020, 12, 735. [Google Scholar] [CrossRef]
- Fuchs, C.; Pham, L.; Henderson, J.; Stalnaker, K.J.; Anderson, R.R.; Tam, J. Multi-faceted enhancement of full-thickness skin wound healing by treatment with autologous micro skin tissue columns. Sci. Rep. 2021, 11, 1688. [Google Scholar] [CrossRef]
- Simman, R.; Phavixay, L. Split-thickness skin grafts remain the gold standard for the closure of large acute and chronic wounds. J. Am. Col. Certif. Wound Spec. 2011, 3, 55–59. [Google Scholar] [CrossRef][Green Version]
- Mison, M.B. Skin Flaps and Grafts. In Small Animal Surgical Emergencies; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2022; pp. 645–651. [Google Scholar] [CrossRef]
- Prohaska, J.; Cook, C. Skin Grafting. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2022. [Google Scholar]
- Zhong, S.P.; Zhang, Y.Z.; Lim, C.T. Tissue scaffolds for skin wound healing and dermal reconstruction. WIREs Nanomed. Nanobiotechnol. 2010, 2, 510–525. [Google Scholar] [CrossRef]
- Rho, K.S.; Jeong, L.; Lee, G.; Seo, B.-M.; Park, Y.J.; Hong, S.-D.; Roh, S.; Cho, J.J.; Park, W.H.; Min, B.-M. Electrospinning of collagen nanofibers: Effects on the behavior of normal human keratinocytes and early-stage wound healing. Biomaterials 2006, 27, 1452–1461. [Google Scholar] [CrossRef]
- Sell, S.A.; Wolfe, P.S.; Garg, K.; McCool, J.M.; Rodriguez, I.A.; Bowlin, G.L. The Use of Natural Polymers in Tissue Engineering: A Focus on Electrospun Extracellular Matrix Analogues. Polymers 2010, 2, 522–553. [Google Scholar] [CrossRef]
- Biggs, L.C.; Kim, C.S.; Miroshnikova, Y.A.; Wickström, S.A. Mechanical Forces in the Skin: Roles in Tissue Architecture, Stability, and Function. J. Investig. Dermatol. 2020, 140, 284–290. [Google Scholar] [CrossRef]
- Griffin, M.F.; Leung, B.C.; Premakumar, Y.; Szarko, M.; Butler, P.E. Comparison of the mechanical properties of different skin sites for auricular and nasal reconstruction. J. Otolaryngol.-Head Neck Surg. 2017, 46, 33. [Google Scholar] [CrossRef] [PubMed]
- Peixoto, T.; Carneiro, S.; Fangueiro, R.; Guedes, R.M.; Paiva, M.C.; Lopes, M.A. Engineering hybrid textile braids for tendon and ligament repair application. J. Appl. Polym. Sci. 2022, 139, 52013. [Google Scholar] [CrossRef]
- Burgio, V.; Civera, M.; Rodriguez Reinoso, M.; Pizzolante, E.; Prezioso, S.; Bertuglia, A.; Surace, C. Mechanical Properties of Animal Tendons: A Review and Comparative Study for the Identification of the Most Suitable Human Tendon Surrogates. Processes 2022, 10, 485. [Google Scholar] [CrossRef]
- Jaiswal, D.; Yousman, L.; Neary, M.; Fernschild, E.; Zolnoski, B.; Katebifar, S.; Rudraiah, S.; Mazzocca, A.D.; Kumbar, S.G. Tendon tissue engineering: Biomechanical considerations. Biomed. Mater. 2020, 15, 052001. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, E.; Ascenzi, M.G.; Farnum, C. Primary cilia are highly oriented with respect to collagen direction and long axis of extensor tendon. J. Orthop. Res. 2010, 28, 77–82. [Google Scholar] [CrossRef]
- Kuo, C.K.; Marturano, J.E.; Tuan, R.S. Novel strategies in tendon and ligament tissue engineering: Advanced biomaterials and regeneration motifs. BMC Sports Sci. Med. Rehabil. 2010, 2, 20. [Google Scholar] [CrossRef]
- Han, Y.; Lian, M.; Wu, Q.; Qiao, Z.; Sun, B.; Dai, K. Effect of Pore Size on Cell Behavior Using Melt Electrowritten Scaffolds. Front. Bioeng. Biotechnol. 2021, 9, 629270. [Google Scholar] [CrossRef]
- Wang, J.H.C.; Guo, Q.; Li, B. Tendon biomechanics and mechanobiology—A minireview of basic concepts and recent advancements. J. Hand Ther. 2012, 25, 133–141. [Google Scholar] [CrossRef]
- Pitta Kruize, C.; Panahkhahi, S.; Putra, N.E.; Diaz-Payno, P.; van Osch, G.; Zadpoor, A.A.; Mirzaali, M.J. Biomimetic Approaches for the Design and Fabrication of Bone-to-Soft Tissue Interfaces. ACS Biomater. Sci. Eng. 2021. [Google Scholar] [CrossRef]
- Aghaei, A.; Bochud, N.; Rosi, G.; Naili, S. Assessing the effective elastic properties of the tendon-to-bone insertion: A multiscale modeling approach. Biomech. Modeling Mechanobiol. 2021, 20, 433–448. [Google Scholar] [CrossRef]
- Chimerad, M.; Barazesh, A.; Zandi, M.; Zarkesh, I.; Moghaddam, A.; Borjian, P.; Chimehrad, R.; Asghari, A.; Akbarnejad, Z.; Khonakdar, H.A.; et al. Tissue engineered scaffold fabrication methods for medical applications. Int. J. Polym. Mater. Polym. Biomater. 2022, 1–25. [Google Scholar] [CrossRef]
- Yoon, J.-Y. 3D Scaffold Fabrication. In Tissue Engineering: A Primer with Laboratory Demonstrations; Yoon, J.-Y., Ed.; Springer International Publishing: Cham, Switzerland, 2022; pp. 155–174. [Google Scholar]
- Kumar, A.; Jacob, A. Techniques in scaffold fabrication process for tissue engineering applications: A review. J. Appl. Biol. Biotechnol. 2022, 10, 163–176. [Google Scholar] [CrossRef]
- Swathy Krishna, B.; Vandana, K. Recent Advances in Scaffold Fabrication Techniques for Tissue Engineering. In A Holistic and Integrated Approach to Lifestyle Diseases, 1st ed.; Apple Academic Press: Palm Bay, FL, USA, 2022; p. 29. [Google Scholar]
- Lu, T.; Li, Y.; Chen, T. Techniques for fabrication and construction of three-dimensional scaffolds for tissue engineering. Int. J. Nanomed. 2013, 8, 337–350. [Google Scholar] [CrossRef]
- Eltom, A.; Zhong, G.; Muhammad, A. Scaffold Techniques and Designs in Tissue Engineering Functions and Purposes: A Review. Adv. Mater. Sci. Eng. 2019, 2019, 3429527. [Google Scholar] [CrossRef]
- Zaszczyńska, A.; Moczulska-Heljak, M.; Gradys, A.; Sajkiewicz, P. Advances in 3D Printing for Tissue Engineering. Materials 2021, 14, 3149. [Google Scholar] [CrossRef]
- Tamay, D.G.; Dursun Usal, T.; Alagoz, A.S.; Yucel, D.; Hasirci, N.; Hasirci, V. 3D and 4D Printing of Polymers for Tissue Engineering Applications. Front. Bioeng. Biotechnol. 2019, 7, 164. [Google Scholar] [CrossRef]
- Advincula, R.C.; Dizon, J.R.C.; Caldona, E.B.; Viers, R.A.; Siacor, F.D.C.; Maalihan, R.D.; Espera, A.H. On the progress of 3D-printed hydrogels for tissue engineering. MRS Commun. 2021, 11, 539–553. [Google Scholar] [CrossRef]
- Kalyan, B.G.P.; Kumar, L. 3D Printing: Applications in Tissue Engineering, Medical Devices, and Drug Delivery. AAPS PharmSciTech 2022, 23, 92. [Google Scholar] [CrossRef]
- Chung, J.J.; Im, H.; Kim, S.H.; Park, J.W.; Jung, Y. Toward Biomimetic Scaffolds for Tissue Engineering: 3D Printing Techniques in Regenerative Medicine. Front. Bioeng. Biotechnol. 2020, 8, 586406. [Google Scholar] [CrossRef]
- Mankovich, N.J.; Cheeseman, A.M.; Stoker, N.G. The display of three-dimensional anatomy with stereolithographic models. J. Digit. Imaging 1990, 3, 200–203. [Google Scholar] [CrossRef]
- Hull, C.W. Apparatus for Production of Three-Dimensional Objects by Stereolithography. U.S. Patent No. 638905, 19 December 1989. [Google Scholar]
- Hwang, H.H.; Zhu, W.; Victorine, G.; Lawrence, N.; Chen, S. 3D-Printing of Functional Biomedical Microdevices via Light- and Extrusion-Based Approaches. Small Methods 2018, 2, 1700277. [Google Scholar] [CrossRef]
- Cima, M.J.; Sachs, E.M.; Cima, L.G.; Yoo, J.; Khanuja, S.; Borland, S.W.; Wu, B.M.; Giordano, R.A. Computer-Derived Microstructures by 3D Printing: Sio- and Structural Materials; The University of Texas at Austin: Austin, TX, USA, 1994. [Google Scholar]
- Levy, R.A.; Guduri, S.; Crawford, R.H. Preliminary experience with selective laser sintering models of the human temporal bone. AJNR Am. J. Neuroradiol. 1994, 15, 473–477. [Google Scholar]
- Zein, I.; Hutmacher, D.W.; Tan, K.C.; Teoh, S.H. Fused deposition modeling of novel scaffold architectures for tissue engineering applications. Biomaterials 2002, 23, 1169–1185. [Google Scholar] [CrossRef]
- Crump, S.S. Apparatus and Method for Creating Three-Dimensional Objects. U.S. Patent 5,121,329, 9 June 1992. [Google Scholar]
- Ozbolat, I.T. Scaffold-Based or Scaffold-Free Bioprinting: Competing or Complementing Approaches? J. Nanotechnol. Eng. Med. 2015, 6, 024701. [Google Scholar] [CrossRef]
- Ozbolat, I.T. 3—The Bioink. In 3D Bioprinting; Ozbolat, I.T., Ed.; Academic Press: Oxford, UK, 2017; pp. 41–92. [Google Scholar] [CrossRef]
- Ozbolat, I.T. 2—Design for Bioprinting. In 3D Bioprinting; Ozbolat, I.T., Ed.; Academic Press: Oxford, UK, 2017; pp. 13–39. [Google Scholar] [CrossRef]
- Gungor-Ozkerim, P.S.; Inci, I.; Zhang, Y.S.; Khademhosseini, A.; Dokmeci, M.R. Bioinks for 3D bioprinting: An overview. Biomater. Sci. 2018, 6, 915–946. [Google Scholar] [CrossRef] [PubMed]
- Mironov, V.; Reis, N.; Derby, B. Review: Bioprinting: A beginning. Tissue Eng. 2006, 12, 631–634. [Google Scholar] [CrossRef] [PubMed]
- Van Houten, S.K.; Bramson, M.T.K.; Corr, D.T. A Bioreactor for Controlled Electrical and Mechanical Stimulation of Developing Scaffold-Free Constructs. J. Biomech. Eng. 2022, 144, 094501. [Google Scholar] [CrossRef] [PubMed]
- Brassard, J.A.; Nikolaev, M.; Hübscher, T.; Hofer, M.; Lutolf, M.P. Recapitulating macro-scale tissue self-organization through organoid bioprinting. Nat. Mater. 2021, 20, 22–29. [Google Scholar] [CrossRef]
- Liu, X.; Hao, M.; Chen, Z.; Zhang, T.; Huang, J.; Dai, J.; Zhang, Z. 3D bioprinted neural tissue constructs for spinal cord injury repair. Biomaterials 2021, 272, 120771. [Google Scholar] [CrossRef]
- Yang, H.; Sun, L.; Pang, Y.; Hu, D.; Xu, H.; Mao, S.; Peng, W.; Wang, Y.; Xu, Y.; Zheng, Y.C.; et al. Three-dimensional bioprinted hepatorganoids prolong survival of mice with liver failure. Gut 2021, 70, 567–574. [Google Scholar] [CrossRef]
- Quílez, C.; de Aranda Izuzquiza, G.; García, M.; López, V.; Montero, A.; Valencia, L.; Velasco, D. Bioprinting for Skin. Methods Mol. Biol 2020, 2140, 217–228. [Google Scholar] [CrossRef]
- Liu, Y.; Peng, L.; Li, L.; Huang, C.; Shi, K.; Meng, X.; Wang, P.; Wu, M.; Li, L.; Cao, H.; et al. 3D-bioprinted BMSC-laden biomimetic multiphasic scaffolds for efficient repair of osteochondral defects in an osteoarthritic rat model. Biomaterials 2021, 279, 121216. [Google Scholar] [CrossRef]
- Di Martino, A.; Liverani, L.; Rainer, A.; Salvatore, G.; Trombetta, M.; Denaro, V. Electrospun scaffolds for bone tissue engineering. Musculoskelet. Surg. 2011, 95, 69–80. [Google Scholar] [CrossRef]
- Wright, L.D.; Young, R.T.; Andric, T.; Freeman, J.W. Fabrication and mechanical characterization of 3D electrospun scaffolds for tissue engineering. Biomed. Mater. 2010, 5, 055006. [Google Scholar] [CrossRef]
- Isaac, B.; Taylor, R.M.; Reifsnider, K. Mechanical and Dielectric Properties of Aligned Electrospun Fibers. Fibers 2021, 9, 4. [Google Scholar] [CrossRef]
- Lannutti, J.; Reneker, D.; Ma, T.; Tomasko, D.; Farson, D. Electrospinning for tissue engineering scaffolds. Mater. Sci. Eng. C 2007, 27, 504–509. [Google Scholar] [CrossRef]
- Maimouni, I.; Cejas, C.M.; Cossy, J.; Tabeling, P.; Russo, M. Microfluidics Mediated Production of Foams for Biomedical Applications. Micromachines 2020, 11, 83. [Google Scholar] [CrossRef]
- Dehghani, F.; Annabi, N. Engineering porous scaffolds using gas-based techniques. Curr. Opin. Biotechnol. 2011, 22, 661–666. [Google Scholar] [CrossRef]
- Garg, T.; Singh, O.; Arora, S.; Murthy, R. Scaffold: A novel carrier for cell and drug delivery. Crit Rev. Ther. Drug Carr. Syst. 2012, 29, 1–63. [Google Scholar] [CrossRef]
- Costantini, M.; Barbetta, A. Gas foaming technologies for 3D scaffold engineering. In Functional 3D Tissue Engineering Scaffolds; Elsevier: Amsterdam, The Netherlands, 2018; pp. 127–149. [Google Scholar] [CrossRef]
- Guarino, V.; Causa, F.; Salerno, A.; Ambrosio, L.; Netti, P.A. Design and Manufacture of Microporous Polymeric Materials with Hierarchal Complex Structure for Biomedical Application. Mater. Sci. Technol. 2008, 24, 1111–1117. [Google Scholar] [CrossRef]
- Toong, D.W.Y.; Toh, H.W.; Ng, J.C.K.; Wong, P.E.H.; Leo, H.L.; Venkatraman, S.; Tan, L.P.; Ang, H.Y.; Huang, Y. Bioresorbable Polymeric Scaffold in Cardiovascular Applications. Int. J. Mol. Sci. 2020, 21, 3444. [Google Scholar] [CrossRef]
- Barbetta, A.; Barigelli, E.; Dentini, M. Porous alginate hydrogels: Synthetic methods for tailoring the porous texture. Biomacromolecules 2009, 10, 2328–2337. [Google Scholar] [CrossRef]
- Salerno, A.; Iannace, S.; Netti, P.A. Open-Pore Biodegradable Foams Prepared via Gas Foaming and Microparticulate Templating. Macromol. Biosci. 2008, 8, 655–664. [Google Scholar] [CrossRef]
- Turnbull, G.; Clarke, J.; Picard, F.; Riches, P.; Jia, L.; Han, F.; Li, B.; Shu, W. 3D bioactive composite scaffolds for bone tissue engineering. Bioact. Mater. 2018, 3, 278–314. [Google Scholar] [CrossRef]
- El-Sherbiny, I.M.; Yacoub, M.H. Hydrogel scaffolds for tissue engineering: Progress and challenges. Glob. Cardiol. Sci. Pract. 2013, 2013, 316–342. [Google Scholar] [CrossRef]
- Shantha, K.L.; Harding, D.R.K. Synthesis and evaluation of sucrose-containing polymeric hydrogels for oral drug delivery. J. Appl. Polym. Sci. 2002, 84, 2597–2604. [Google Scholar] [CrossRef]
- El-Sherbiny, I.M.; Lins, R.J.; Abdel-Bary, E.M.; Harding, D.R.K. Preparation, characterization, swelling and in vitro drug release behaviour of poly[N-acryloylglycine-chitosan] interpolymeric pH and thermally-responsive hydrogels. Eur. Polym. J. 2005, 41, 2584–2591. [Google Scholar] [CrossRef]
- El-Sherbiny, I.M.; Harding, D.R.K.; Abdel-Bary, E.M. Preparation and Swelling Study of a pH-Dependent Interpolymeric Hydrogel Based on Chitosan for Controlled Drug Release. Int. J. Polym. Mater. Polym. Biomater. 2006, 55, 789–802. [Google Scholar] [CrossRef]
- Bhat, G.; Kandagor, V. 1-Synthetic polymer fibers and their processing requirements. In Advances in Filament Yarn Spinning of Textiles and Polymers; Zhang, D., Ed.; Woodhead Publishing: Sawston, UK, 2014; pp. 3–30. [Google Scholar] [CrossRef]
- Jabbari, E.; Nozari, S. Swelling behavior of acrylic acid hydrogels prepared by γ-radiation crosslinking of polyacrylic acid in aqueous solution. Eur. Polym. J. 2000, 36, 2685–2692. [Google Scholar] [CrossRef]
- Yamada, K.; Tabata, Y.; Yamamoto, K.; Miyamoto, S.; Nagata, I.; Kikuchi, H.; Ikada, Y. Potential efficacy of basic fibroblast growth factor incorporated in biodegradable hydrogels for skull bone regeneration. J. Neurosurg. 1997, 86, 871–875. [Google Scholar] [CrossRef]
- Anal, A.K.; Stevens, W.F. Chitosan–alginate multilayer beads for controlled release of ampicillin. Int. J. Pharm. 2005, 290, 45–54. [Google Scholar] [CrossRef]
- Lee, K.Y.; Mooney, D.J. Hydrogels for Tissue Engineering. Chem. Rev. 2001, 101, 1869–1880. [Google Scholar] [CrossRef] [PubMed]
- Shin, H.; Jo, S.; Mikos, A.G. Biomimetic materials for tissue engineering. Biomaterials 2003, 24, 4353–4364. [Google Scholar] [CrossRef]
- Nguyen, K.T.; West, J.L. Photopolymerizable hydrogels for tissue engineering applications. Biomaterials 2002, 23, 4307–4314. [Google Scholar] [CrossRef]
- Gutowska, A.; Bae, Y.H.; Feijen, J.; Kim, S.W. Heparin release from thermosensitive hydrogels. J. Control. Release 1992, 22, 95–104. [Google Scholar] [CrossRef]
- Ferreira, L.; Vidal, M.M.; Gil, M.H. Evaluation of poly(2-hydroxyethyl methacrylate) gels as drug delivery systems at different pH values. Int. J. Pharm. 2000, 194, 169–180. [Google Scholar] [CrossRef]
- D’Emanuele, A.; Staniforth, J.N. An Electrically Modulated Drug Delivery Device: I. Pharm. Res. 1991, 8, 913–918. [Google Scholar] [CrossRef]
- Sarkanen, J.R.; Kaila, V.; Mannerström, B.; Räty, S.; Kuokkanen, H.; Miettinen, S.; Ylikomi, T. Human adipose tissue extract induces angiogenesis and adipogenesis in vitro. Tissue Eng. Part A 2012, 18, 17–25. [Google Scholar] [CrossRef]
- Ekaputra, A.K.; Prestwich, G.D.; Cool, S.M.; Hutmacher, D.W. The three-dimensional vascularization of growth factor-releasing hybrid scaffold of poly (epsilon-caprolactone)/collagen fibers and hyaluronic acid hydrogel. Biomaterials 2011, 32, 8108–8117. [Google Scholar] [CrossRef]
- Phelps, E.A.; García, A.J. Engineering more than a cell: Vascularization strategies in tissue engineering. Curr. Opin. Biotechnol. 2010, 21, 704–709. [Google Scholar] [CrossRef]
- Okay, O.; Petrov, P. Polymeric Cryogels Macroporous Gels with Remarkable Properties—Preface; Springer: Berlin/Heidelberg, Germany, 2014; Volume 263. [Google Scholar]
- Lozinsky, V.I.; Galaev, I.Y.; Plieva, F.M.; Savina, I.N.; Jungvid, H.; Mattiasson, B. Polymeric cryogels as promising materials of biotechnological interest. Trends Biotechnol. 2003, 21, 445–451. [Google Scholar] [CrossRef]
- Hixon, K.R.; Lu, T.; Sell, S.A. A comprehensive review of cryogels and their roles in tissue engineering applications. Acta Biomater. 2017, 62, 29–41. [Google Scholar] [CrossRef]
- Kumar, A. (Ed.) Supermacroporous Cryogels: Biomedical and Biotechnological Applications, 1st ed.; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar] [CrossRef]
- Henderson, T.M.A.; Ladewig, K.; Haylock, D.N.; McLean, K.M.; O’Connor, A.J. Cryogels for biomedical applications. J. Mater. Chem. B 2013, 1, 2682–2695. [Google Scholar] [CrossRef]
- Kumari, J.; Karande, A.A.; Kumar, A. Combined Effect of Cryogel Matrix and Temperature-Reversible Soluble–Insoluble Polymer for the Development of in Vitro Human Liver Tissue. ACS Appl. Mater. Interfaces 2016, 8, 264–277. [Google Scholar] [CrossRef]
- Lozinsky, V.I.; Plieva, F.M. Poly(vinyl alcohol) cryogels employed as matrices for cell immobilization. 3. Overview of recent research and developments. Enzym. Microb. Technol. 1998, 23, 227–242. [Google Scholar] [CrossRef]
- Qi, C.; Yan, X.; Huang, C.; Melerzanov, A.; Du, Y. Biomaterials as carrier, barrier and reactor for cell-based regenerative medicine. Protein Cell 2015, 6, 638–653. [Google Scholar] [CrossRef]
- Hassan, C.; Peppas, N. Structure and Applications of Poly(vinyl alcohol) Hydrogels Produced by Conventional Crosslinking or by Freezing/Thawing Methods. In PVA Hydrogels Anionic Polymerisation Nanocomposites; Springer: Berlin/Heidelberg, Germany, 2000; Volume 153, pp. 37–65. [Google Scholar]
- Lozinsky, V.I. Polymeric cryogels as a new family of macroporous and supermacroporous materials for biotechnological purposes. Russ. Chem. Bull. 2008, 57, 1015–1032. [Google Scholar] [CrossRef]
- Liu, Y.; Vrana, N.E.; Cahill, P.A.; McGuinness, G.B. Physically crosslinked composite hydrogels of PVA with natural macromolecules: Structure, mechanical properties, and endothelial cell compatibility. J. Biomed. Mater. Res. B Appl. Biomater. 2009, 90, 492–502. [Google Scholar] [CrossRef]
- Ivanov, R.V.; Lozinsky, V.I.; Noh, S.K.; Lee, Y.R.; Han, S.S.; Lyoo, W.S. Preparation and characterization of polyacrylamide cryogels produced from a high-molecular-weight precursor. II. The influence of the molecular weight of the polymeric precursor. J. Appl. Polym. Sci. 2008, 107, 382–390. [Google Scholar] [CrossRef]
- Lozinsky, V.I.; Vainerman, E.S.; Ivanova, S.A.; Titova, E.F.; Shtil’man, M.I.; Belavtseva, E.M.; Rogozhin, S.V. Study of cryostructurization of polymer systems. VI. The influence of the process temperature on the dynamics of formation and structure of cross-linked polyacrylamide cryogels. Acta Polym. 1986, 37, 142–146. [Google Scholar] [CrossRef]
- Lozinsky, V.I.; Plieva, F.M.; Galaev, I.Y.; Mattiasson, B. The potential of polymeric cryogels in bioseparation. Bioseparation 2001, 10, 163–188. [Google Scholar] [CrossRef]
- Dinu, M.; Ozmen, M.; Dragan, E.S.; Okay, O. Freezing as a path to build macroporous structures: Superfast responsive polyacrylamide hydrogels. Polymer 2007, 48, 195–204. [Google Scholar] [CrossRef]
- Hwang, Y.; Zhang, C.; Varghese, S. Poly(ethylene glycol) cryogels as potential cell scaffolds: Effect of polymerization conditions on cryogel microstructure and properties. J. Mater. Chem. 2010, 20, 345–351. [Google Scholar] [CrossRef]
- Ozmen, M.M.; Dinu, M.V.; Dragan, E.S.; Okay, O. Preparation of Macroporous Acrylamide-based Hydrogels: Cryogelation under Isothermal Conditions. J. Macromol. Sci. Part A 2007, 44, 1195–1202. [Google Scholar] [CrossRef]
- Kathuria, N.; Tripathi, A.; Kar, K.K.; Kumar, A. Synthesis and characterization of elastic and macroporous chitosan–gelatin cryogels for tissue engineering. Acta Biomater. 2009, 5, 406–418. [Google Scholar] [CrossRef] [PubMed]
- Gang, E.J.; Jeong, J.A.; Hong, S.H.; Hwang, S.H.; Kim, S.W.; Yang, I.H.; Ahn, C.; Han, H.; Kim, H. Skeletal myogenic differentiation of mesenchymal stem cells isolated from human umbilical cord blood. Stem Cells 2004, 22, 617–624. [Google Scholar] [CrossRef] [PubMed]
- Carletti, E.; Motta, A.; Migliaresi, C. Scaffolds for tissue engineering and 3D cell culture. Methods Mol. Biol. 2011, 695, 17–39. [Google Scholar] [CrossRef] [PubMed]
- Siva, A.; Ansari, M.N.M. A Review on Bone Scaffold Fabrication Methods. Int. Res. J. Eng. Technol. 2015, 2, 1232–1238. [Google Scholar]
- Mikos, A.G.; Thorsen, A.J.; Czerwonka, L.A.; Bao, Y.; Langer, R.; Winslow, D.N.; Vacanti, J.P. Preparation and characterization of poly(l-lactic acid) foams. Polymer 1994, 35, 1068–1077. [Google Scholar] [CrossRef]
- Prasad, A.; Sankar, M.R.; Katiyar, V. State of Art on Solvent Casting Particulate Leaching Method for Orthopedic ScaffoldsFabrication. Mater. Today Proc. 2017, 4, 898–907. [Google Scholar] [CrossRef]
- Kim, H.J.; Kim, U.-J.; Leisk, G.G.; Bayan, C.; Georgakoudi, I.; Kaplan, D.L. Bone Regeneration on Macroporous Aqueous-Derived Silk 3-D Scaffolds. Macromol. Biosci. 2007, 7, 643–655. [Google Scholar] [CrossRef]
- Pattison, M.A.; Wurster, S.; Webster, T.J.; Haberstroh, K.M. Three-dimensional, nano-structured PLGA scaffolds for bladder tissue replacement applications. Biomaterials 2005, 26, 2491–2500. [Google Scholar] [CrossRef]
- Jin Yoon, J.; Ho Song, S.; Sung Lee, D.; Park, T.G. Immobilization of cell adhesive RGD peptide onto the surface of highly porous biodegradable polymer scaffolds fabricated by a gas foaming/salt leaching method. Biomaterials 2004, 25, 5613–5620. [Google Scholar] [CrossRef]
- Shin, M.; Abukawa, H.; Troulis, M.J.; Vacanti, J.P. Development of a biodegradable scaffold with interconnected pores by heat fusion and its application to bone tissue engineering. J. Biomed. Mater. Res. Part A 2008, 84A, 702–709. [Google Scholar] [CrossRef]
- Martins, P.M.; Nunes-Pereira, J.; Lanceros-Méndez, S.; Costa, C.M. Chapter 17—Synthetic polymer-based membranes for lithium-ion batteries. In Synthetic Polymeric Membranes for Advanced Water Treatment, Gas Separation, and Energy Sustainability; Ismail, A.F., Salleh, W.N.W., Yusof, N., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 383–415. [Google Scholar] [CrossRef]
- Sampath, U.G.T.M.; Ching, Y.C.; Chuah, C.H.; Sabariah, J.J.; Lin, P.-C. Fabrication of Porous Materials from Natural/Synthetic Biopolymers and Their Composites. Materials 2016, 9, 991. [Google Scholar] [CrossRef]
- Hutmacher, D.W. Scaffolds in tissue engineering bone and cartilage. Biomaterials 2000, 21, 2529–2543. [Google Scholar] [CrossRef]
- Siemann, U. Solvent cast technology—A versatile tool for thin film production. In Scattering Methods and Properties of Polymer Materials; Stribeck, N., Smarsly, B., Eds.; Springer: Berlin/Heidelberg, Germany, 2005; pp. 1–14. [Google Scholar]
- Hosseinkhani, M.; Mehrabani, D.; Karimfar, M.H.; Bakhtiyari, S.; Manafi, A.; Shirazi, R. Tissue engineered scaffolds in regenerative medicine. World J. Plast. Surg. 2014, 3, 3–7. [Google Scholar]
- Reddy, M.S.B.; Ponnamma, D.; Choudhary, R.; Sadasivuni, K.K. A Comparative Review of Natural and Synthetic Biopolymer Composite Scaffolds. Polymers 2021, 13, 1105. [Google Scholar] [CrossRef]
- Drury, J.L.; Dennis, R.G.; Mooney, D.J. The tensile properties of alginate hydrogels. Biomaterials 2004, 25, 3187–3199. [Google Scholar] [CrossRef]
- Sun, J.; Tan, H. Alginate-Based Biomaterials for Regenerative Medicine Applications. Materials 2013, 6, 1285–1309. [Google Scholar] [CrossRef]
- Lu, T.; Hixon, K.R.; Ona, W.J.; Carletta, M.N.; Garg, K.; Sell, S.A. An in vitro analysis of injectable methacrylated alginate cryogels incorporated with PRP targeting minimally invasive treatment of bone nonunion. Biomed. Phys. Amp Eng. Express 2018, 4, 055001. [Google Scholar] [CrossRef]
- Leong, N.L.; Petrigliano, F.A.; McAllister, D.R. Current tissue engineering strategies in anterior cruciate ligament reconstruction. J. Biomed. Mater. Res. Part A 2014, 102, 1614–1624. [Google Scholar] [CrossRef]
- Masuko, T.; Iwasaki, N.; Yamane, S.; Funakoshi, T.; Majima, T.; Minami, A.; Ohsuga, N.; Ohta, T.; Nishimura, S. Chitosan-RGDSGGC conjugate as a scaffold material for musculoskeletal tissue engineering. Biomaterials 2005, 26, 5339–5347. [Google Scholar] [CrossRef]
- Majima, T.; Funakosi, T.; Iwasaki, N.; Yamane, S.T.; Harada, K.; Nonaka, S.; Minami, A.; Nishimura, S. Alginate and chitosan polyion complex hybrid fibers for scaffolds in ligament and tendon tissue engineering. J. Orthop. Sci 2005, 10, 302–307. [Google Scholar] [CrossRef]
- Yamane, S.; Iwasaki, N.; Majima, T.; Funakoshi, T.; Masuko, T.; Harada, K.; Minami, A.; Monde, K.; Nishimura, S. Feasibility of chitosan-based hyaluronic acid hybrid biomaterial for a novel scaffold in cartilage tissue engineering. Biomaterials 2005, 26, 611–619. [Google Scholar] [CrossRef]
- Lee, K.Y.; Mooney, D.J. Alginate: Properties and biomedical applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef]
- Manoukian, O.S.; Sardashti, N.; Stedman, T.; Gailiunas, K.; Ojha, A.; Penalosa, A.; Mancuso, C.; Hobert, M.; Kumbar, S.G. Biomaterials for Tissue Engineering and Regenerative Medicine. In Encyclopedia of Biomedical Engineering; Narayan, R., Ed.; Elsevier: Oxford, UK, 2019; pp. 462–482. [Google Scholar] [CrossRef]
- Kim, C.H.; Park, S.J.; Yang, D.H.; Chun, H.J. Chitosan for Tissue Engineering. Adv. Exp. Med. Biol. 2018, 1077, 475–485. [Google Scholar] [CrossRef]
- Shao, H.J.; Chen, C.S.; Lee, Y.T.; Wang, J.H.; Young, T.H. The phenotypic responses of human anterior cruciate ligament cells cultured on poly(epsilon-caprolactone) and chitosan. J. Biomed. Mater. Res. A 2010, 93, 1297–1305. [Google Scholar] [CrossRef]
- Shao, H.J.; Lee, Y.T.; Chen, C.S.; Wang, J.H.; Young, T.H. Modulation of gene expression and collagen production of anterior cruciate ligament cells through cell shape changes on polycaprolactone/chitosan blends. Biomaterials 2010, 31, 4695–4705. [Google Scholar] [CrossRef]
- Hansson, A.; Hashom, N.; Falson, F.; Rousselle, P.; Jordan, O.; Borchard, G. In vitro evaluation of an RGD-functionalized chitosan derivative for enhanced cell adhesion. Carbohydr. Polym. 2012, 90, 1494–1500. [Google Scholar] [CrossRef]
- Muzzarelli, R.A.; Zucchini, C.; Ilari, P.; Pugnaloni, A.; Mattioli Belmonte, M.; Biagini, G.; Castaldini, C. Osteoconductive properties of methylpyrrolidinone chitosan in an animal model. Biomaterials 1993, 14, 925–929. [Google Scholar] [CrossRef]
- Goonoo, N.; Jeetah, R.; Bhaw-Luximon, A.; Jhurry, D. Polydioxanone-based bio-materials for tissue engineering and drug/gene delivery applications. Eur. J. Pharm. Biopharm. 2015, 97, 371–391. [Google Scholar] [CrossRef] [PubMed]
- Dong, C.; Lv, Y. Application of Collagen Scaffold in Tissue Engineering: Recent Advances and New Perspectives. Polymers 2016, 8, 42. [Google Scholar] [CrossRef] [PubMed]
- Kontturi, L.-S.; Järvinen, E.; Muhonen, V.; Collin, E.C.; Pandit, A.S.; Kiviranta, I.; Yliperttula, M.; Urtti, A. An injectable, in situ forming type II collagen/hyaluronic acid hydrogel vehicle for chondrocyte delivery in cartilage tissue engineering. Drug Deliv. Transl. Res. 2014, 4, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Jeon, O.; Song, S.J.; Lee, K.-J.; Park, M.H.; Lee, S.-H.; Hahn, S.K.; Kim, S.; Kim, B.-S. Mechanical properties and degradation behaviors of hyaluronic acid hydrogels cross-linked at various cross-linking densities. Carbohydr. Polym. 2007, 70, 251–257. [Google Scholar] [CrossRef]
- Czerner, M.; Fellay, L.S.; Suárez, M.P.; Frontini, P.M.; Fasce, L.A. Determination of Elastic Modulus of Gelatin Gels by Indentation Experiments. Procedia Mater. Sci. 2015, 8, 287–296. [Google Scholar] [CrossRef]
- Karimi, A.; Navidbakhsh, M. Material properties in unconfined compression of gelatin hydrogel for skin tissue engineering applications. Biomedizinische Technik 2014, 59, 479–486. [Google Scholar] [CrossRef]
- Tan, J.J.Y.; Lee, C.P.; Hashimoto, M. Preheating of Gelatin Improves its Printability with Transglutaminase in Direct Ink Writing 3D Printing. Int. J. Bioprint. 2020, 6, 296. [Google Scholar]
- Hixon, K.R.; Dunn, A.J.; Flores, R.; Minden-Birkenmaier, B.A.; Kalaf, E.A.G.; Shornick, L.P.; Sell, S.A. Using Electrospun Scaffolds to Promote Macrophage Phenotypic Modulation and Support Wound Healing. Electrospinning 2017, 1, 31–45. [Google Scholar] [CrossRef]
- Bello, A.B.; Kim, D.; Kim, D.; Park, H.; Lee, S.-H. Engineering and Functionalization of Gelatin Biomaterials: From Cell Culture to Medical Applications. Tissue Eng. Part B Rev. 2020, 26, 164–180. [Google Scholar] [CrossRef]
- Lukin, I.; Erezuma, I.; Maeso, L.; Zarate, J.; Desimone, M.F.; Al-Tel, T.H.; Dolatshahi-Pirouz, A.; Orive, G. Progress in Gelatin as Biomaterial for Tissue Engineering. Pharmaceutics 2022, 14, 1177. [Google Scholar] [CrossRef]
- Cao, Y.; Wang, B. Biodegradation of silk biomaterials. Int. J. Mol. Sci. 2009, 10, 1514–1524. [Google Scholar] [CrossRef]
- Hajiabbas, M.; Alemzadeh, I.; Vossoughi, M. A porous hydrogel-electrospun composite scaffold made of oxidized alginate/gelatin/silk fibroin for tissue engineering application. Carbohydr. Polym. 2020, 245, 116465. [Google Scholar] [CrossRef]
- Kadakia, P.U.; Jain, E.; Hixon, K.R.; Eberlin, C.T.; Sell, S.A. Sonication induced silk fibroin cryogels for tissue engineering applications. Mater. Res. Express 2016, 3, 055401. [Google Scholar] [CrossRef]
- Hixon, K.R.; Lu, T.; McBride-Gagyi, S.H.; Janowiak, B.E.; Sell, S.A. A Comparison of Tissue Engineering Scaffolds Incorporated with Manuka Honey of Varying UMF. BioMed Res. Int. 2017, 2017, 4843065. [Google Scholar] [CrossRef]
- Migonney, V. History of Biomaterials. In Biomaterials; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2014; pp. 1–10. [Google Scholar]
- Kunz, R.I.; Brancalhão, R.M.; Ribeiro, L.F.; Natali, M.R. Silkworm Sericin: Properties and Biomedical Applications. Biomed. Res. Int. 2016, 2016, 8175701. [Google Scholar] [CrossRef]
- Hasirci, V.; Hasirci, N. Polymers as Biomaterials. In Fundamentals of Biomaterials; Hasirci, V., Hasirci, N., Eds.; Springer: New York, NY, USA, 2018; pp. 65–82. [Google Scholar]
- Bazgir, M.; Zhang, W.; Zhang, X.; Elies, J.; Saeinasab, M.; Coates, P.; Youseffi, M.; Sefat, F. Fabrication and Characterization of PCL/PLGA Coaxial and Bilayer Fibrous Scaffolds for Tissue Engineering. Materials 2021, 14, 6295. [Google Scholar] [CrossRef]
- Touré, A.B.R.; Mele, E.; Christie, J.K. Multi-layer Scaffolds of Poly(caprolactone), Poly(glycerol sebacate) and Bioactive Glasses Manufactured by Combined 3D Printing and Electrospinning. Nanomaterials 2020, 10, 626. [Google Scholar] [CrossRef]
- Chen, S.; Carlson, M.A.; Zhang, Y.S.; Hu, Y.; Xie, J. Fabrication of injectable and superelastic nanofiber rectangle matrices ("peanuts") and their potential applications in hemostasis. Biomaterials 2018, 179, 46–59. [Google Scholar] [CrossRef]
- Vyas, C.; Zhang, J.; Øvrebø, Ø.; Huang, B.; Roberts, I.; Setty, M.; Allardyce, B.; Haugen, H.; Rajkhowa, R.; Bartolo, P. 3D printing of silk microparticle reinforced polycaprolactone scaffolds for tissue engineering applications. Mater. Sci. Eng. C 2021, 118, 111433. [Google Scholar] [CrossRef]
- Luginina, M.; Schuhladen, K.; Orrú, R.; Cao, G.; Boccaccini, A.R.; Liverani, L. Electrospun PCL/PGS Composite Fibers Incorporating Bioactive Glass Particles for Soft Tissue Engineering Applications. Nanomaterials 2020, 10, 978. [Google Scholar] [CrossRef]
- Rai, B.; Lin, J.; Lim, Z.; Guldberg, R.; Hutmacher, D.; Cool, S. Differences between in vitro viability and differentiation and in vivo bone-forming efficacy of human mesenchymal stem cells cultured on PCL-TCP scaffolds. Biomaterials 2010, 31, 7960–7970. [Google Scholar] [CrossRef]
- Greenwald, D.; Shumway, S.; Albear, P.; Gottlieb, L. Mechanical comparison of 10 suture materials before and after in vivo incubation. J. Surg. Res. 1994, 56, 372–377. [Google Scholar] [CrossRef]
- Madurantakam, P.A.; Cost, C.P.; Simpson, D.G.; Bowlin, G.L. Science of nanofibrous scaffold fabrication: Strategies for next generation tissue-engineering scaffolds. Nanomedicine 2009, 4, 193–206. [Google Scholar] [CrossRef]
- Horvath, T.; Kalman, M.; Szabo, T.; Roman, K.; Zsoldos, G.; Szabone Kollar, M. The mechanical properties of polyethylene-terephthalate (PET) and polylactic-acid (PDLLA and PLLA), the influence of material structure on forming. IOP Conf. Ser. Mater. Sci. Eng. 2018, 426, 012018. [Google Scholar] [CrossRef]
- Ma, Z.; Kotaki, M.; Yong, T.; He, W.; Ramakrishna, S. Surface engineering of electrospun polyethylene terephthalate (PET) nanofibers towards development of a new material for blood vessel engineering. Biomaterials 2005, 26, 2527–2536. [Google Scholar] [CrossRef] [PubMed]
- Gu, H.; AlFayez, F.; Ahmed, T.; Bashir, Z. Poly(ethylene terephthalate) Powder-A Versatile Material for Additive Manufacturing. Polymers 2019, 11, 2041. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Xu, J.; Kang, Y.; Li, Y.; Wang, L.; Yan, X.; Jiang, J.; Zhao, J. Acceleration of ligamentization and osseointegration processes after anterior cruciate ligament reconstruction with autologous tissue-engineered polyethylene terephthalate graft. Ann. Transl. Med. 2021, 9, 770. [Google Scholar] [CrossRef]
- Cai, J.; Yang, Y.; Ai, C.; Jin, W.; Sheng, D.; Chen, J.; Chen, S. Bone Marrow Stem Cells-Seeded Polyethylene Terephthalate Scaffold in Repair and Regeneration of Rabbit Achilles Tendon. Artif. Organs 2018, 42, 1086–1094. [Google Scholar] [CrossRef]
- Hassan, M.H.; Omar, A.M.; Daskalakis, E.; Hou, Y.; Huang, B.; Strashnov, I.; Grieve, B.D.; Bártolo, P. The Potential of Polyethylene Terephthalate Glycol as Biomaterial for Bone Tissue Engineering. Polymers 2020, 12, 3045. [Google Scholar] [CrossRef]
- Whelove, O.E.; Cozad, M.J.; Lee, B.D.; Sengupta, S.; Bachman, S.L.; Ramshaw, B.J.; Grant, S.A. Development and in vitro studies of a polyethylene terephthalate-gold nanoparticle scaffold for improved biocompatibility. J. Biomed. Mater. Res. B Appl. Biomater. 2011, 99, 142–149. [Google Scholar] [CrossRef]
- Cohen, D.J.; Ferrara, L.; Stone, M.B.; Schwartz, Z.; Boyan, B.D. Cell and Tissue Response to Polyethylene Terephthalate Mesh Containing Bone Allograft in Vitro and in Vivo. Int. J. Spine Surg. 2020, 14, S121–S132. [Google Scholar] [CrossRef]
- Rai, R.; Tallawi, M.; Grigore, A.; Boccaccini, A.R. Synthesis, properties and biomedical applications of poly(glycerol sebacate) (PGS): A review. Prog. Polym. Sci. 2012, 37, 1051–1078. [Google Scholar] [CrossRef]
- Hajiali, H.; Shahgasempour, S.; Naimi-Jamal, M.R.; Peirovi, H. Electrospun PGA/gelatin nanofibrous scaffolds and their potential application in vascular tissue engineering. Int. J. Nanomed. 2011, 6, 2133–2141. [Google Scholar] [CrossRef]
- Harris, L.D.; Kim, B.-S.; Mooney, D.J. Open pore biodegradable matrices formed with gas foaming. J. Biomed. Mater. Res. 1998, 42, 396–402. [Google Scholar] [CrossRef]
- Mahmoudifar, N.; Doran, P.M. Tissue engineering of human cartilage and osteochondral composites using recirculation bioreactors. Biomaterials 2005, 26, 7012–7024. [Google Scholar] [CrossRef]
- Sekiya, N.; Ichioka, S.; Terada, D.; Tsuchiya, S.; Kobayashi, H. Efficacy of a poly glycolic acid (PGA)/collagen composite nanofibre scaffold on cell migration and neovascularisation in vivo skin defect model. J. Plast. Surg. Hand Surg. 2013, 47, 498–502. [Google Scholar] [CrossRef]
- Lin, X.; Wang, W.; Zhang, W.; Zhang, Z.; Zhou, G.; Cao, Y.; Liu, W. Hyaluronic Acid Coating Enhances Biocompatibility of Nonwoven PGA Scaffold and Cartilage Formation. Tissue Eng. Part C Methods 2017, 23, 86–97. [Google Scholar] [CrossRef]
- El-Sayed, N.M.; El-Bakary, M.A.; Ibrahim, M.A.; Elgamal, M.A.; Hamza, A.A. Characterization of the mechanical and structural properties of PGA/TMC copolymer for cardiac tissue engineering. Microsc. Res. Tech. 2021, 84, 1596–1606. [Google Scholar] [CrossRef]
- Kim, M.J.; Hwang, M.Y.; Kim, J.; Chung, D.J. Biodegradable and Elastomeric Poly(glycerol sebacate) as a Coating Material for Nitinol Bare Stent. BioMed Res. Int. 2014, 2014, 956952. [Google Scholar] [CrossRef]
- Jeffries, E.M.; Allen, R.A.; Gao, J.; Pesce, M.; Wang, Y. Highly elastic and suturable electrospun poly(glycerol sebacate) fibrous scaffolds. Acta Biomater. 2015, 18, 30–39. [Google Scholar] [CrossRef]
- Pachekoski, W.; Belem, L. Thermal, Mechanical and Morphological Properties of Poly (Hydroxybutyrate) and Polypropylene Blends After Processing. Mater. Res. 2009, 12, 159–164. [Google Scholar] [CrossRef]
- Soleymani Eil Bakhtiari, S.; Karbasi, S.; Toloue, E.B. Modified poly(3-hydroxybutyrate)-based scaffolds in tissue engineering applications: A review. Int. J. Biol. Macromol. 2021, 166, 986–998. [Google Scholar] [CrossRef]
- Mohan, A.; Girdhar, M.; Kumar, R.; Chaturvedi, H.S.; Vadhel, A.; Solanki, P.R.; Kumar, A.; Kumar, D.; Mamidi, N. Polyhydroxybutyrate-Based Nanocomposites for Bone Tissue Engineering. Pharmaceuticals 2021, 14, 1163. [Google Scholar] [CrossRef]
- Kaniuk, Ł.; Stachewicz, U. Development and Advantages of Biodegradable PHA Polymers Based on Electrospun PHBV Fibers for Tissue Engineering and Other Biomedical Applications. ACS Biomater. Sci. Eng. 2021, 7, 5339–5362. [Google Scholar] [CrossRef]
- Rivera-Briso, A.L.; Serrano-Aroca, Á. Poly(3-Hydroxybutyrate-co-3-Hydroxyvalerate): Enhancement Strategies for Advanced Applications. Polymers 2018, 10, 732. [Google Scholar] [CrossRef]
- Amaro, L.; Correia, D.M.; Martins, P.M.; Botelho, G.; Carabineiro, S.A.C.; Ribeiro, C.; Lanceros-Mendez, S. Morphology Dependence Degradation of Electro-and Magnetoactive Poly(3-hydroxybutyrate-co-hydroxyvalerate) for Tissue Engineering Applications. Polymers 2020, 12, 953. [Google Scholar] [CrossRef]
- Righetti, M.C.; Cinelli, P.; Mallegni, N.; Stäbler, A.; Lazzeri, A. Thermal and Mechanical Properties of Biocomposites Made of Poly(3-hydroxybutyrate-co-3-hydroxyvalerate) and Potato Pulp Powder. Polymers 2019, 11, 308. [Google Scholar] [CrossRef]
- Romero-Araya, P.; Pino, V.; Nenen, A.; Cárdenas, V.; Pavicic, F.; Ehrenfeld, P.; Serandour, G.; Lisoni, J.G.; Moreno-Villoslada, I.; Flores, M.E. Combining Materials Obtained by 3D-Printing and Electrospinning from Commercial Polylactide Filament to Produce Biocompatible Composites. Polymers 2021, 13, 3806. [Google Scholar] [CrossRef]
- Barroca, N.; Marote, A.; Vieira, S.I.; Almeida, A.; Fernandes, M.H.V.; Vilarinho, P.M.; da Cruz, E.S.O.A.B. Electrically polarized PLLA nanofibers as neural tissue engineering scaffolds with improved neuritogenesis. Colloids Surf. B Biointerfaces 2018, 167, 93–103. [Google Scholar] [CrossRef]
- Gaaz, T.S.; Sulong, A.B.; Akhtar, M.N.; Kadhum, A.A.; Mohamad, A.B.; Al-Amiery, A.A. Properties and Applications of Polyvinyl Alcohol, Halloysite Nanotubes and Their Nanocomposites. Molecules 2015, 20, 22833–22847. [Google Scholar] [CrossRef]
- Sa’adon, S.; Ansari, M.N.M.; Razak, S.I.A.; Anand, J.S.; Nayan, N.H.M.; Ismail, A.E.; Khan, M.U.A.; Haider, A. Preparation and Physicochemical Characterization of a Diclofenac Sodium-Dual Layer Polyvinyl Alcohol Patch. Polymers 2021, 13, 2459. [Google Scholar] [CrossRef]
- Nagakawa, Y.; Kato, M.; Suye, S.I.; Fujita, S. Fabrication of tough, anisotropic, chemical-crosslinker-free poly(vinyl alcohol) nanofibrous cryogels via electrospinning. RSC Adv. 2020, 10, 38045–38054. [Google Scholar] [CrossRef]
- Rathinavel, S.; Ekambaram, S.; Korrapati, P.S.; Sangeetha, D. Design and fabrication of electrospun SBA-15-incorporated PVA with curcumin: A biomimetic nanoscaffold for skin tissue engineering. Biomed. Mater. 2020, 15, 035009. [Google Scholar] [CrossRef]
- Xu, M.; Qin, M.; Zhang, X.; Zhang, X.; Li, J.; Hu, Y.; Chen, W.; Huang, D. Porous PVA/SA/HA hydrogels fabricated by dual-crosslinking method for bone tissue engineering. J. Biomater. Sci. Polym. Ed. 2020, 31, 816–831. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.; Zhou, Y.; Lu, W.; Zhu, W.; Li, Y.; Chen, K.; Zhang, G.; Xu, J.; Deng, Z.; Wang, D. Characterization of a novel polyvinyl alcohol/chitosan porous hydrogel combined with bone marrow mesenchymal stem cells and its application in articular cartilage repair. BMC Musculoskelet. Disord. 2019, 20, 257. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.; Kang, E.H.; Choi, S.; Jeon, E.J.; Cho, J.H.; Kang, D.; Lee, H.; Yun, I.S.; Cho, S.-W. Tissue-Adhesive Chondroitin Sulfate Hydrogel for Cartilage Reconstruction. ACS Biomater. Sci. Eng. 2021, 7, 4230–4243. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Xu, Q.; Johnson, M.; Wang, X.; Lyu, J.; Li, Y.; McMahon, S.; Greiser, U.A.S.; Wang, W. A chondroitin sulfate based injectable hydrogel for delivery of stem cells in cartilage regeneration. Biomater. Sci. 2021, 9, 4139–4148. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Chen, W.; Chen, Y.; Mo, X.; Fan, C. Chondroitin sulfate modified 3D porous electrospun nanofiber scaffolds promote cartilage regeneration. Mater. Sci. Eng. C 2021, 118, 111312. [Google Scholar] [CrossRef] [PubMed]
- Kwon, H.J.; Han, Y. Chondroitin sulfate-based biomaterials for tissue engineering. Turk. J. Biol. 2016, 40, 290–299. [Google Scholar] [CrossRef]
- Andrews, S.; Cheng, A.; Stevens, H.; Logun, M.T.; Webb, R.; Jordan, E.; Xia, B.; Karumbaiah, L.; Guldberg, R.E.; Stice, S. Chondroitin Sulfate Glycosaminoglycan Scaffolds for Cell and Recombinant Protein-Based Bone Regeneration. Stem Cells Transl. Med. 2019, 8, 575–585. [Google Scholar] [CrossRef]
- Wang, T.; Yang, F. A comparative study of chondroitin sulfate and heparan sulfate for directing three-dimensional chondrogenesis of mesenchymal stem cells. Stem. Cell Res. Ther. 2017, 8, 284. [Google Scholar] [CrossRef]
- Akhshabi, S.; Biazar, E.; Singh, V.; Keshel, S.H.; Geetha, N. The effect of the carbodiimide cross-linker on the structural and biocompatibility properties of collagen-chondroitin sulfate electrospun mat. Int. J. Nanomed. 2018, 13, 4405–4416. [Google Scholar] [CrossRef]
- Sundaresan, G.; Abraham, R.J.J.; Appa Rao, V.; Narendra Babu, R.; Govind, V.; Meti, M.F. Established method of chondroitin sulphate extraction from buffalo (Bubalus bubalis) cartilages and its identification by FTIR. J. Food Sci. Technol. 2018, 55, 3439–3445. [Google Scholar] [CrossRef]
- Guthold, M.; Liu, W.; Sparks, E.A.; Jawerth, L.M.; Peng, L.; Falvo, M.; Superfine, R.; Hantgan, R.R.; Lord, S.T. A comparison of the mechanical and structural properties of fibrin fibers with other protein fibers. Cell Biochem. Biophys. 2007, 49, 165–181. [Google Scholar] [CrossRef]
- Bandiera, A.; Passamonti, S.; Dolci, L.S.; Focarete, M.L. Composite of Elastin-Based Matrix and Electrospun Poly(L-Lactic Acid) Fibers: A Potential Smart Drug Delivery System. Front. Bioeng. Biotechnol. 2018, 6, 127. [Google Scholar] [CrossRef]
- Klimek, K.; Ginalska, G. Proteins and Peptides as Important Modifiers of the Polymer Scaffolds for Tissue Engineering Applications-A Review. Polymers 2020, 12, 844. [Google Scholar] [CrossRef]
- Daamen, W.F.; Veerkamp, J.H.; van Hest, J.C.M.; van Kuppevelt, T.H. Elastin as a biomaterial for tissue engineering. Biomaterials 2007, 28, 4378–4398. [Google Scholar] [CrossRef]
- Xing, H.; Lee, H.; Luo, L.; Kyriakides, T.R. Extracellular matrix-derived biomaterials in engineering cell function. Biotechnol. Adv. 2020, 42, 107421. [Google Scholar] [CrossRef]
- Holst, J.; Watson, S.; Lord, M.S.; Eamegdool, S.S.; Bax, D.V.; Nivison-Smith, L.B.; Kondyurin, A.; Ma, L.; Oberhauser, A.F.; Weiss, A.S.; et al. Substrate elasticity provides mechanical signals for the expansion of hemopoietic stem and progenitor cells. Nat. Biotechnol. 2010, 28, 1123–1128. [Google Scholar] [CrossRef]
- Klotzsch, E.; Smith, M.L.; Kubow, K.E.; Muntwyler, S.; Little, W.C.; Beyeler, F.; Gourdon, D.; Nelson, B.J.; Vogel, V. Fibronectin forms the most extensible biological fibers displaying switchable force-exposed cryptic binding sites. Proc. Natl. Acad. Sci. USA 2009, 106, 18267–18272. [Google Scholar] [CrossRef]
- Palomino-Durand, C.; Pauthe, E.; Gand, A. Fibronectin-Enriched Biomaterials, Biofunctionalization, and Proactivity: A Review. Appl. Sci. 2021, 11, 12111. [Google Scholar] [CrossRef]
- Parisi, L.; Toffoli, A.; Ghezzi, B.; Mozzoni, B.; Lumetti, S.; Macaluso, G.M. A glance on the role of fibronectin in controlling cell response at biomaterial interface. Jpn. Dent. Sci. Rev. 2020, 56, 50–55. [Google Scholar] [CrossRef]
- Klingberg, F.; Chau, G.; Walraven, M.; Boo, S.; Koehler, A.; Chow, M.L.; Olsen, A.L.; Im, M.; Lodyga, M.; Wells, R.G.; et al. The fibronectin ED-A domain enhances recruitment of latent TGF-β-binding protein-1 to the fibroblast matrix. J. Cell Sci. 2018, 131, jcs201293. [Google Scholar] [CrossRef]
- Pankov, R.; Yamada, K.M. Fibronectin at a glance. J. Cell Sci. 2002, 115, 3861–3863. [Google Scholar] [CrossRef]
- Liverani, L.; Killian, M.S.; Boccaccini, A.R. Fibronectin Functionalized Electrospun Fibers by Using Benign Solvents: Best Way to Achieve Effective Functionalization. Front. Bioeng. Biotechnol. 2019, 7, 68. [Google Scholar] [CrossRef]
- Floren, M.; Tan, W. Three-dimensional, soft neotissue arrays as high throughput platforms for the interrogation of engineered tissue environments. Biomaterials 2015, 59, 39–52. [Google Scholar] [CrossRef]
- Kim, M.; Kim, S.E.; Kang, S.S.; Kim, Y.H.; Tae, G. The use of de-differentiated chondrocytes delivered by a heparin-based hydrogel to regenerate cartilage in partial-thickness defects. Biomaterials 2011, 32, 7883–7896. [Google Scholar] [CrossRef]
- Wang, J.; Xiao, L.; Wang, W.; Zhang, D.; Ma, Y.; Zhang, Y.; Wang, X. The Auxiliary Role of Heparin in Bone Regeneration and its Application in Bone Substitute Materials. Front. Bioeng. Biotechnol. 2022, 10, 837172. [Google Scholar] [CrossRef]
- Sakiyama, S.E.; Schense, J.C.; Hubbell, J.A. Incorporation of heparin-binding peptides into fibrin gels enhances neurite extension: An example of designer matrices in tissue engineering. FASEB J. 1999, 13, 2214–2224. [Google Scholar] [CrossRef]
- Kimicata, M.; Mahadik, B.; Fisher, J.P. Long-Term Sustained Drug Delivery via 3D Printed Masks for the Development of a Heparin-Loaded Interlayer in Vascular Tissue Engineering Applications. ACS Appl. Mater. Interfaces 2021, 13, 50812–50822. [Google Scholar] [CrossRef] [PubMed]
- Sakiyama-Elbert, S.E.; Hubbell, J.A. Development of fibrin derivatives for controlled release of heparin-binding growth factors. J. Control. Release 2000, 65, 389–402. [Google Scholar] [CrossRef]
- Wissink, M.J.; Beernink, R.; Scharenborg, N.M.; Poot, A.A.; Engbers, G.H.M.; Beugeling, T.; Van Aken, W.G.; Feijen, J. Endothelial cell seeding of (heparinized) collagen matrices: Effects of bFGF pre-loading on proliferation (after low density seeding) and pro-coagulant factors. J. Control. Release 2000, 67, 141–155. [Google Scholar] [CrossRef]
- Weatherford, D.A.; Sackman, J.E.; Reddick, T.T.; Freeman, M.B.; Stevens, S.L.; Goldman, M.H. Vascular endothelial growth factor and heparin in a biologic glue promotes human aortic endothelial cell proliferation with aortic smooth muscle cell inhibition. Surgery 1996, 120, 433–439. [Google Scholar] [CrossRef]
- Tavsanli, B.; Okay, O. Mechanically strong hyaluronic acid hydrogels with an interpenetrating network structure. Eur. Polym. J. 2017, 94, 185–195. [Google Scholar] [CrossRef]
- Chircov, C.; Grumezescu, A.M.; Bejenaru, L.E. Hyaluronic acid-based scaffolds for tissue engineering. Rom. J. Morphol. Embryol. 2018, 59, 71–76. [Google Scholar]
- Venugopal, J.; Low, S.; Choon, A.T.; Ramakrishna, S. Interaction of cells and nanofiber scaffolds in tissue engineering. J. Biomed. Mater. Res. Part B Appl. Biomater. 2008, 84B, 34–48. [Google Scholar] [CrossRef]
- Chun, C.; Lee, D.Y.; Kim, J.-T.; Kwon, M.-K.; Kim, Y.-Z.; Kim, S.-S. Effect of molecular weight of hyaluronic acid (HA) on viscoelasticity and particle texturing feel of HA dermal biphasic fillers. Biomater. Res. 2016, 20, 24. [Google Scholar] [CrossRef]
- Martínez-Sanz, E.; Ossipov, D.A.; Hilborn, J.; Larsson, S.; Jonsson, K.B.; Varghese, O.P. Bone reservoir: Injectable hyaluronic acid hydrogel for minimal invasive bone augmentation. J. Control. Release 2011, 152, 232–240. [Google Scholar] [CrossRef]
- Paidikondala, M.; Wang, S.; Hilborn, J.; Larsson, S.; Varghese, O.P. Impact of Hydrogel Cross-Linking Chemistry on the in Vitro and in Vivo Bioactivity of Recombinant Human Bone Morphogenetic Protein-2. ACS Appl. Bio Mater. 2019, 2, 2006–2012. [Google Scholar] [CrossRef]
- Tsanaktsidou, E.; Kammona, O.; Kiparissides, C. Recent Developments in Hyaluronic Acid-Based Hydrogels for Cartilage Tissue Engineering Applications. Polymers 2022, 14, 839. [Google Scholar] [CrossRef]
- Pek, Y.S.; Wan, A.C.; Ying, J.Y. The effect of matrix stiffness on mesenchymal stem cell differentiation in a 3D thixotropic gel. Biomaterials 2010, 31, 385–391. [Google Scholar] [CrossRef]
- Chen, J.D.; Wang, Y.; Chen, X. In Situ Fabrication of Nano-hydroxyapatite in a Macroporous Chitosan Scaffold for Tissue Engineering. J. Biomater. Sci. Polym. Ed. 2009, 20, 1555–1565. [Google Scholar] [CrossRef]
- Kim, T.J.; Lee, Y.J.; Kim, D.J. Separation of Hyaluronic Acid from Plant and Animal Tissues. In Biochemical Engineering for 2001; Springer: Tokyo, Japan, 1992. [Google Scholar]
- Neal, R.A.; McClugage, S.G.; Link, M.C.; Sefcik, L.S.; Ogle, R.C.; Botchwey, E.A. Laminin nanofiber meshes that mimic morphological properties and bioactivity of basement membranes. Tissue Eng. Part C Methods 2009, 15, 11–21. [Google Scholar] [CrossRef]
- Talovic, M.; Marcinczyk, M.; Ziemkiewicz, N.; Garg, K. Laminin Enriched Scaffolds for Tissue Engineering Applications. Adv. Tissue Eng. Regen. Med. Open Access 2017, 2, 00033. [Google Scholar]
- Kikkawa, Y.; Hozumi, K.; Katagiri, F.; Nomizu, M.; Kleinman, H.K.; Koblinski, J.E. Laminin-111-derived peptides and cancer. Cell Adh. Migr. 2013, 7, 150–256. [Google Scholar] [CrossRef]
- Li, Q.; Chau, Y. Neural differentiation directed by self-assembling peptide scaffolds presenting laminin-derived epitopes. J. Biomed. Mater. Res. Part A 2010, 94, 688–699. [Google Scholar] [CrossRef]
- Nicosia, R.F.; Bonanno, E.; Smith, M.; Yurchenco, P. Modulation of Angiogenesis In Vitro by Laminin-Entactin Complex. Dev. Biol. 1994, 164, 197–206. [Google Scholar] [CrossRef]
- Chan, B.P.; Leong, K.W. Scaffolding in tissue engineering: General approaches and tissue-specific considerations. Eur. Spine J. 2008, 17 (Suppl. S4), 467–479. [Google Scholar] [CrossRef]
- Akkouch, A.; Zhang, Z.; Rouabhia, M. A novel collagen/hydroxyapatite/poly(lactide-co-ε-caprolactone) biodegradable and bioactive 3D porous scaffold for bone regeneration. J. Biomed. Mater. Res. Part A 2011, 96A, 693–704. [Google Scholar] [CrossRef]
- Ma, Z.; Mao, Z.; Gao, C. Surface modification and property analysis of biomedical polymers used for tissue engineering. Colloids Surf. B Biointerfaces 2007, 60, 137–157. [Google Scholar] [CrossRef]
- Yang, Y.; Kang, Y.; Sen, M.; Park, S. Bioceramics in Tissue Engineering. In Biomaterials for Tissue Engineering Applications: A Review of the Past and Future Trends; Burdick, J.A., Mauck, R.L., Eds.; Springer: Vienna, Austria, 2011; pp. 179–207. [Google Scholar]
- Baino, F.; Novajra, G.; Vitale-Brovarone, C. Bioceramics and Scaffolds: A Winning Combination for Tissue Engineering. Front. Bioeng. Biotechnol. 2015, 3, 202. [Google Scholar] [CrossRef]
- Yunos, D.M.; Bretcanu, O.; Boccaccini, A.R. Polymer-bioceramic composites for tissue engineering scaffolds. J. Mater. Sci. 2008, 43, 4433–4442. [Google Scholar] [CrossRef]
- Wang, M. Developing bioactive composite materials for tissue replacement. Biomaterials 2003, 24, 2133–2151. [Google Scholar] [CrossRef]
- Hench, L.L. Bioceramics, a clinical success. Am. Ceram. Soc. Bull. 1998, 77, 67–74. [Google Scholar]
- Lu, J.; Yu, H.; Chen, C. Biological properties of calcium phosphate biomaterials for bone repair: A review. RSC Adv. 2018, 8, 2015–2033. [Google Scholar] [CrossRef] [PubMed]
- Amini, A.R.; Laurencin, C.T.; Nukavarapu, S.P. Bone tissue engineering: Recent advances and challenges. Crit. Rev. Biomed. Eng. 2012, 40, 363–408. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.Z.; Leung, V.Y.; Dong, Q.R.; Cheung, K.M.; Chan, D.; Lu, W.W. Mesenchymal Stem Cell-Based Repair of Articular Cartilage with Polyglycolic Acid-Hydroxyapatite Biphasic Scaffold. Int. J. Artif. Organs 2008, 31, 480–489. [Google Scholar] [CrossRef]
- Sun, W.; Gregory, D.A.; Tomeh, M.A.; Zhao, X. Silk Fibroin as a Functional Biomaterial for Tissue Engineering. Int. J. Mol. Sci. 2021, 22, 1499. [Google Scholar] [CrossRef]
- Day, R.M.; Boccaccini, A.R.; Shurey, S.; Roether, J.A.; Forbes, A.; Hench, L.L.; Gabe, S.M. Assessment of polyglycolic acid mesh and bioactive glass for soft-tissue engineering scaffolds. Biomaterials 2004, 25, 5857–5866. [Google Scholar] [CrossRef]
- Ahmed, I.; Collins, C.A.; Lewis, M.P.; Olsen, I.; Knowles, J.C. Processing, characterisation and biocompatibility of iron-phosphate glass fibres for tissue engineering. Biomaterials 2004, 25, 3223–3232. [Google Scholar] [CrossRef]
- Qian, Y.; Han, Q.; Chen, W.; Song, J.; Zhao, X.; Ouyang, Y.; Yuan, W.; Fan, C. Platelet-Rich Plasma Derived Growth Factors Contribute to Stem Cell Differentiation in Musculoskeletal Regeneration. Front. Chem. 2017, 5, 89. [Google Scholar] [CrossRef]
- Gallego-Muñoz, P.; Ibares-Frías, L.; Valsero-Blanco, M.C.; Cantalapiedra-Rodriguez, R.; Merayo-Lloves, J.; Martínez-García, M.C. Effects of TGFβ1, PDGF-BB, and bFGF, on human corneal fibroblasts proliferation and differentiation during stromal repair. Cytokine 2017, 96, 94–101. [Google Scholar] [CrossRef]
- Surmacz, E. Growth factor receptors as therapeutic targets: Strategies to inhibit the insulin-like growth factor I receptor. Oncogene 2003, 22, 6589–6597. [Google Scholar] [CrossRef]
- Davydova, N.; Harris, N.C.; Roufail, S.; Paquet-Fifield, S.; Ishaq, M.; Streltsov, V.A.; Williams, S.P.; Karnezis, T.; Stacker, S.A.; Achen, M.G. Differential Receptor Binding and Regulatory Mechanisms for the Lymphangiogenic Growth Factors Vascular Endothelial Growth Factor (VEGF)-C and -D. J. Biol. Chem. 2016, 291, 27265–27278. [Google Scholar] [CrossRef]
- Guan, N.; Liu, Z.; Zhao, Y.; Li, Q.; Wang, Y. Engineered biomaterial strategies for controlling growth factors in tissue engineering. Drug Deliv. 2020, 27, 1438–1451. [Google Scholar] [CrossRef]
- Whitaker, M.; Quirk, R.; Howdle, S.; Shakesheff, K. Growth factor release from tissue engineering scaffolds. J. Pharm. Pharmacol. 2001, 53, 1427–1437. [Google Scholar] [CrossRef]
- Hollinger, J.O.; Leong, K. Poly(α-hydroxy acids): Carriers for bone morphogenetic proteins. Biomaterials 1996, 17, 187–194. [Google Scholar] [CrossRef]
- Boden, S.D. Bioactive factors for bone tissue engineering. Clin. Orthop. Relat. Res. 1999, 367, S84–S94. [Google Scholar] [CrossRef]
- Whang, K.; Goldstick, T.K.; Healy, K.E. A biodegradable polymer scaffold for delivery of osteotropic factors. Biomaterials 2000, 21, 2545–2551. [Google Scholar] [CrossRef]
- Park, J.Y.; Shim, J.-H.; Choi, S.-A.; Jang, J.; Kim, M.; Lee, S.H.; Cho, D.-W. 3D printing technology to control BMP-2 and VEGF delivery spatially and temporally to promote large-volume bone regeneration. J. Mater. Chem. B 2015, 3, 5415–5425. [Google Scholar] [CrossRef]
- Lee, S.S.; Kim, J.H.; Jeong, J.; Kim, S.H.L.; Koh, R.H.; Kim, I.; Bae, S.; Lee, H.; Hwang, N.S. Sequential growth factor releasing double cryogel system for enhanced bone regeneration. Biomaterials 2020, 257, 120223. [Google Scholar] [CrossRef]
- Wu, R.; Gao, G.; Xu, Y. Electrospun Fibers Immobilized with BMP-2 Mediated by Polydopamine Combined with Autogenous Tendon to Repair Developmental Dysplasia of the Hip in a Porcine Model. Int. J. Nanomed. 2020, 15, 6563–6577. [Google Scholar] [CrossRef]
- Poldervaart, M.T.; Wang, H.; van der Stok, J.; Weinans, H.; Leeuwenburgh, S.C.G.; Öner, F.C.; Dhert, W.J.A.; Alblas, J. Sustained Release of BMP-2 in Bioprinted Alginate for Osteogenicity in Mice and Rats. PLoS ONE 2013, 8, e72610. [Google Scholar] [CrossRef]
- Chung, Y.I.; Ahn, K.M.; Jeon, S.H.; Lee, S.Y.; Lee, J.H.; Tae, G. Enhanced bone regeneration with BMP-2 loaded functional nanoparticle-hydrogel complex. J. Control. Release 2007, 121, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.W.; Kang, K.S.; Lee, S.H.; Kim, J.Y.; Lee, B.K.; Cho, D.W. Bone regeneration using a microstereolithography-produced customized poly(propylene fumarate)/diethyl fumarate photopolymer 3D scaffold incorporating BMP-2 loaded PLGA microspheres. Biomaterials 2011, 32, 744–752. [Google Scholar] [CrossRef] [PubMed]
- Mooney, D.J.; Kaufmann, P.M.; Sano, K.; Schwendeman, S.P.; Majahod, K.; Schloo, B.; Vacanti, J.P.; Langer, R. Localized delivery of epidermal growth factor improves the survival of transplanted hepatocytes. Biotechnol. Bioeng. 1996, 50, 422–429. [Google Scholar] [CrossRef]
- Chen, G.; Ito, Y.; Imanishi, Y. Photo-immobilization of epidermal growth factor enhances its mitogenic effect by artificial juxtacrine signaling. Biochim. Biophys. Acta 1997, 1358, 200–208. [Google Scholar] [CrossRef]
- Haller, M.F.; Saltzman, W.M. Localized Delivery of Proteins in the Brain: Can Transport Be Customized? Pharm. Res. 1998, 15, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, Y.; Ajioka, I.; Akaike, T. Gene transfection of multicellular spheroid of hepatocytes on an artificial substrate. Cytotechnology 1998, 26, 65–78. [Google Scholar] [CrossRef] [PubMed]
- von Recum, H.; Kikuchi, A.; Yamato, M.; Sakurai, Y.; Okano, T.; Kim, S.W. Growth factor and matrix molecules preserve cell function on thermally responsive culture surfaces. Tissue Eng. 1999, 5, 251–265. [Google Scholar] [CrossRef]
- Asiri, A.; Saidin, S.; Sani, M.H.; Al-Ashwal, R.H. Epidermal and fibroblast growth factors incorporated polyvinyl alcohol electrospun nanofibers as biological dressing scaffold. Sci. Rep. 2021, 11, 5634. [Google Scholar] [CrossRef]
- Hori, K.; Sotozono, C.; Hamuro, J.; Yamasaki, K.; Kimura, Y.; Ozeki, M.; Tabata, Y.; Kinoshita, S. Controlled-release of epidermal growth factor from cationized gelatin hydrogel enhances corneal epithelial wound healing. J. Control. Release 2007, 118, 169–176. [Google Scholar] [CrossRef]
- Han, L.; Li, P.; Tang, P.; Wang, X.; Zhou, T.; Wang, K.; Ren, F.; Guo, T.; Lu, X. Mussel-inspired cryogels for promoting wound regeneration through photobiostimulation, modulating inflammatory responses and suppressing bacterial invasion. Nanoscale 2019, 11, 15846–15861. [Google Scholar] [CrossRef]
- Yoon, J.J.; Kim, J.H.; Park, T.G. Dexamethasone-releasing biodegradable polymer scaffolds fabricated by a gas-foaming/salt-leaching method. Biomaterials 2003, 24, 2323–2329. [Google Scholar] [CrossRef]
- Weng, T.; Zhang, W.; Xia, Y.; Wu, P.; Yang, M.; Jin, R.; Xia, S.; Wang, J.; You, C.; Han, C.; et al. 3D bioprinting for skin tissue engineering: Current status and perspectives. J. Tissue Eng. 2021, 12, 20417314211028574. [Google Scholar] [CrossRef]
- Kang, S.S.; Gosselin, C.; Ren, D.; Greisler, H.P. Selective stimulation of endothelial cell proliferation with inhibition of smooth muscle cell proliferation by fibroblast growth factor-1 plus heparin delivered from fibrin glue suspensions. Surgery 1995, 118, 280–286, discussion 286–287. [Google Scholar] [CrossRef]
- Fujisato, T.; Sajiki, T.; Liu, Q.; Ikada, Y. Effect of basic fibroblast growth factor on cartilage regeneration in chondrocyte-seeded collagen sponge scaffold. Biomaterials 1996, 17, 155–162. [Google Scholar] [CrossRef]
- Martin, I.; Vunjak-Novakovic, G.; Yang, J.; Langer, R.; Freed, L.E. Mammalian chondrocytes expanded in the presence of fibroblast growth factor 2 maintain the ability to differentiate and regenerate three-dimensional cartilaginous tissue. Exp. Cell Res. 1999, 253, 681–688. [Google Scholar] [CrossRef]
- Shireman, P.K.; Hampton, B.; Burgess, W.H.; Greisler, H.P. Modulation of vascular cell growth kinetics by local cytokine delivery from fibrin glue suspensions. J. Vasc. Surg. 1999, 29, 852–861, discussion 862. [Google Scholar] [CrossRef][Green Version]
- Chandler, L.A.; Doukas, J.; Gonzalez, A.M.; Hoganson, D.K.; Gu, D.-L.; Ma, C.; Nesbit, M.; Crombleholme, T.M.; Herlyn, M.; Sosnowski, B.A.; et al. FGF2-Targeted Adenovirus Encoding Platelet-Derived Growth Factor-B Enhances de Novo Tissue Formation. Mol. Ther. 2000, 2, 153–160. [Google Scholar] [CrossRef]
- Hile, D.D.; Amirpour, M.L.; Akgerman, A.; Pishko, M.V. Active growth factor delivery from poly(D,L-lactide-co-glycolide) foams prepared in supercritical CO(2). J. Control. Release 2000, 66, 177–185. [Google Scholar] [CrossRef]
- Lee, K.; Silva, E.A.; Mooney, D.J. Growth factor delivery-based tissue engineering: General approaches and a review of recent developments. J. R. Soc. Interface 2011, 8, 153–170. [Google Scholar] [CrossRef]
- Zhao, X.; Liu, L.; Wang, J.; Xu, Y.; Zhang, W.; Khang, G.; Wang, X. In vitro vascularization of a combined system based on a 3D printing technique. J. Tissue Eng. Regen. Med. 2016, 10, 833–842. [Google Scholar] [CrossRef]
- Tabata, Y.; Miyao, M.; Inamoto, T.; Ishii, T.; Hirano, Y.; Yamaoki, Y.; Ikada, Y. De Novo Formation of Adipose Tissue by Controlled Release of Basic Fibroblast Growth Factor. Tissue Eng. 2000, 6, 279–289. [Google Scholar] [CrossRef]
- Krewson, C.E.; Saltzman, W.M. Transport and elimination of recombinant human NGF during long-term delivery to the brain. Brain Res. 1996, 727, 169–181. [Google Scholar] [CrossRef]
- Cao, X.; Shoichet, M.S. Delivering neuroactive molecules from biodegradable microspheres for application in central nervous system disorders. Biomaterials 1999, 20, 329–339. [Google Scholar] [CrossRef]
- Saltzman, W.M.; Mak, M.W.; Mahoney, M.J.; Duenas, E.T.; Cleland, J.L. Intracranial delivery of recombinant nerve growth factor: Release kinetics and protein distribution for three delivery systems. Pharm. Res. 1999, 16, 232–240. [Google Scholar] [CrossRef]
- Benoit, J.P.; Faisant, N.; Venier-Julienne, M.C.; Menei, P. Development of microspheres for neurological disorders: From basics to clinical applications. J. Control. Release 2000, 65, 285–296. [Google Scholar] [CrossRef]
- Cho, Y.I.; Choi, J.S.; Jeong, S.Y.; Yoo, H.S. Nerve growth factor (NGF)-conjugated electrospun nanostructures with topographical cues for neuronal differentiation of mesenchymal stem cells. Acta Biomater. 2010, 6, 4725–4733. [Google Scholar] [CrossRef]
- Xu, H.; Yu, Y.; Zhang, L.; Zheng, F.; Yin, Y.; Gao, Y.; Li, K.; Xu, J.; Wen, J.; Chen, H.; et al. Sustainable release of nerve growth factor for peripheral nerve regeneration using nerve conduits laden with Bioconjugated hyaluronic acid-chitosan hydrogel. Compos. Part B Eng. 2022, 230, 109509. [Google Scholar] [CrossRef]
- Newland, B.; Newland, H.; Lorenzi, F.; Eigel, D.; Welzel, P.; Fischer, D.; Wang, W.; Freudenberg, U.; Rosser, A.; Werner, C. Injectable Glycosaminoglycan-Based Cryogels from Well-Defined Microscale Templates for Local Growth Factor Delivery. ACS Chem. Neurosci. 2021, 12, 1178–1188. [Google Scholar] [CrossRef]
- Huang, Y.; Wu, W.; Liu, H.; Chen, Y.; Li, B.; Gou, Z.; Li, X.; Gou, M. 3D printing of functional nerve guide conduits. Burns Trauma 2021, 9, tkab011. [Google Scholar] [CrossRef]
- Li, X.; Wang, X.; Wang, X.; Chen, H.; Zhang, X.; Zhou, L.; Xu, T. 3D bioprinted rat Schwann cell-laden structures with shape flexibility and enhanced nerve growth factor expression. 3 Biotech 2018, 8, 342. [Google Scholar] [CrossRef]
- Hafeman, A.E.; Li, B.; Yoshii, T.; Zienkiewicz, K.; Davidson, J.M.; Guelcher, S.A. Injectable Biodegradable Polyurethane Scaffolds with Release of Platelet-derived Growth Factor for Tissue Repair and Regeneration. Pharm. Res. 2008, 25, 2387. [Google Scholar] [CrossRef] [PubMed]
- Jian, K.; Yang, C.; Li, T.; Wu, X.; Shen, J.; Wei, J.; Yang, Z.; Yuan, D.; Zhao, M.; Shi, J. PDGF-BB-derived supramolecular hydrogel for promoting skin wound healing. J. Nanobiotechnol. 2022, 20, 201. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Hadjiargyrou, M. Induction of cell migration in vitro by an electrospun PDGF-BB/PLGA/PEG-PLA nanofibrous scaffold. J. Biomed. Nanotechnol. 2011, 7, 823–829. [Google Scholar] [CrossRef] [PubMed]
- Welsh, W.R.; Kim, H.D.; Jong, Y.S.; Valentini, R.F. Controlled release of platelet-derived growth factor using ethylene vinyl acetate copolymer (EVAc) coated on stainless-steel wires. Biomaterials 1995, 16, 1319–1325. [Google Scholar] [CrossRef]
- Kirn, H.D.; Valentini, R.F. Human osteoblast response in vitro to platelet-derived growth factor and transforming growth factor-β delivered from controlled-release polymer rods. Biomaterials 1997, 18, 1175–1184. [Google Scholar] [CrossRef]
- Lohmann, C.H.; Schwartz, Z.; Niederauer, G.G.; Carnes, D.L., Jr.; Dean, D.D.; Boyan, B.D. Pretreatment with platelet derived growth factor-BB modulates the ability of costochondral resting zone chondrocytes incorporated into PLA/PGA scaffolds to form new cartilage in vivo. Biomaterials 2000, 21, 49–61. [Google Scholar] [CrossRef]
- Park, Y.J.; Lee, Y.M.; Lee, J.Y.; Seol, Y.J.; Chung, C.P.; Lee, S.J. Controlled release of platelet-derived growth factor-BB from chondroitin sulfate-chitosan sponge for guided bone regeneration. J. Control. Release 2000, 67, 385–394. [Google Scholar] [CrossRef]
- Chenite, A.; Chaput, C.; Wang, D.; Combes, C.; Buschmann, M.D.; Hoemann, C.D.; Leroux, J.C.; Atkinson, B.L.; Binette, F.; Selmani, A. Novel injectable neutral solutions of chitosan form biodegradable gels in situ. Biomaterials 2000, 21, 2155–2161. [Google Scholar] [CrossRef]
- Lin, M.; Overgaard, S.; Glerup, H.; Søballe, K.; Bünger, C. Transforming growth factor-beta1 adsorbed to tricalciumphosphate coated implants increases peri-implant bone remodeling. Biomaterials 2001, 22, 189–193. [Google Scholar] [CrossRef]
- Mann, B.K.; Schmedlen, R.H.; West, J.L. Tethered-TGF-beta increases extracellular matrix production of vascular smooth muscle cells. Biomaterials 2001, 22, 439–444. [Google Scholar] [CrossRef]
- Hauptstein, J.; Forster, L.; Nadernezhad, A.; Groll, J.; Teßmar, J.; Blunk, T. Tethered TGF-β1 in a Hyaluronic Acid-Based Bioink for Bioprinting Cartilaginous Tissues. Int. J. Mol. Sci. 2022, 23, 924. [Google Scholar] [CrossRef]
- Jimi, S.; Jaguparov, A.; Nurkesh, A.; Sultankulov, B.; Saparov, A. Sequential Delivery of Cryogel Released Growth Factors and Cytokines Accelerates Wound Healing and Improves Tissue Regeneration. Front. Bioeng. Biotechnol. 2020, 8, 345. [Google Scholar] [CrossRef]
- Cui, X.; Liu, M.; Wang, J.; Zhou, Y.; Xiang, Q. Electrospun scaffold containing TGF-β1 promotes human mesenchymal stem cell differentiation towards a nucleus pulposus-like phenotype under hypoxia. IET Nanobiotechnol. 2015, 9, 76–84. [Google Scholar] [CrossRef]
- Nicoll, S.B.; Denker, A.E.; Tuan, R.S. In Vitro Characterization of Transforming Growth Factor-ß1-Loaded Composites of Biodegradable Polymer and Mesenchymal Cells. Cells Mater. 1995, 5, 231–244. [Google Scholar]
- King, T.W.; Patrick, C.W., Jr. Development and in vitro characterization of vascular endothelial growth factor (VEGF)-loaded poly(DL-lactic-co-glycolic acid)/poly(ethylene glycol) microspheres using a solid encapsulation/single emulsion/solvent extraction technique. J. Biomed. Mater. Res. 2000, 51, 383–390. [Google Scholar] [CrossRef]
- Lee, K.Y.; Peters, M.C.; Anderson, K.W.; Mooney, D.J. Controlled growth factor release from synthetic extracellular matrices. Nature 2000, 408, 998–1000. [Google Scholar] [CrossRef]
- Murphy, W.L.; Peters, M.C.; Kohn, D.H.; Mooney, D.J. Sustained release of vascular endothelial growth factor from mineralized poly(lactide-co-glycolide) scaffolds for tissue engineering. Biomaterials 2000, 21, 2521–2527. [Google Scholar] [CrossRef]
- Sheridan, M.H.; Shea, L.D.; Peters, M.C.; Mooney, D.J. Bioabsorbable polymer scaffolds for tissue engineering capable of sustained growth factor delivery. J. Control. Release 2000, 64, 91–102. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, Z.; Lu, W.W.; Zhen, W.; Yang, D.; Peng, S. Novel biomaterial strategies for controlled growth factor delivery for biomedical applications. NPG Asia Mater. 2017, 9, e435. [Google Scholar] [CrossRef]
- Gnavi, S.; di Blasio, L.; Tonda-Turo, C.; Mancardi, A.; Primo, L.; Ciardelli, G.; Gambarotta, G.; Geuna, S.; Perroteau, I. Gelatin-based hydrogel for vascular endothelial growth factor release in peripheral nerve tissue engineering. J. Tissue Eng. Regen. Med. 2017, 11, 459–470. [Google Scholar] [CrossRef]
- Kai, D.; Prabhakaran, M.P.; Jin, G.; Tian, L.; Ramakrishna, S. Potential of VEGF-encapsulated electrospun nanofibers for in vitro cardiomyogenic differentiation of human mesenchymal stem cells. J. Tissue Eng. Regen. Med. 2017, 11, 1002–1010. [Google Scholar] [CrossRef]
- Miraftab, M.; Saifullah, A.N.; Çay, A. Physical stabilisation of electrospun poly(vinyl alcohol) nanofibres: Comparative study on methanol and heat-based crosslinking. J. Mater. Sci. 2015, 50, 1943–1957. [Google Scholar] [CrossRef]
- Dos Santos, D.M.; de Annunzio, S.R.; Carmello, J.C.; Pavarina, A.C.; Fontana, C.R.; Correa, D.S. Combining Coaxial Electrospinning and 3D Printing: Design of Biodegradable Bilayered Membranes with Dual Drug Delivery Capability for Periodontitis Treatment. ACS Appl. Bio. Mater. 2022, 5, 146–159. [Google Scholar] [CrossRef]
- Liu, J.; Yao, X.; Wang, Z.; Ye, J.; Luan, C.; He, Y.; Lin, H.; Fu, J. A flexible porous chiral auxetic tracheal stent with ciliated epithelium. Acta Biomater. 2021, 124, 153–165. [Google Scholar] [CrossRef]
- John, J.V.; McCarthy, A.; Su, Y.; Ackerman, D.N.; Shahriar, S.M.S.; Carlson, M.A.; Reid, S.P.; Santarpia, J.L.; Zhu, W.; Xie, J. Nanofiber capsules for minimally invasive sampling of biological specimens from gastrointestinal tract. Acta Biomater. 2022, 146, 211–221. [Google Scholar] [CrossRef]
- Vyas, C.; Ates, G.; Aslan, E.; Hart, J.; Huang, B.; Bartolo, P. Three-Dimensional Printing and Electrospinning Dual-Scale Polycaprolactone Scaffolds with Low-Density and Oriented Fibers to Promote Cell Alignment. 3D Print Addit. Manuf. 2020, 7, 105–113. [Google Scholar] [CrossRef]
- Kim, T.G.; Chung, H.J.; Park, T.G. Macroporous and nanofibrous hyaluronic acid/collagen hybrid scaffold fabricated by concurrent electrospinning and deposition/leaching of salt particles. Acta Biomater. 2008, 4, 1611–1619. [Google Scholar] [CrossRef]
- Rao, F.; Yuan, Z.; Li, M.; Yu, F.; Fang, X.; Jiang, B.; Wen, Y.; Zhang, P. Expanded 3D nanofibre sponge scaffolds by gas-foaming technique enhance peripheral nerve regeneration. Artif. Cells Nanomed. Biotechnol. 2019, 47, 491–500. [Google Scholar] [CrossRef]
- Chen, S.; Carlson, M.A.; Li, X.; Siddique, A.; Zhu, W.; Xie, J. Minimally Invasive Delivery of 3D Shape Recoverable Constructs with Ordered Structures for Tissue Repair. ACS Biomater. Sci. Eng. 2021, 7, 2204–2211. [Google Scholar] [CrossRef]
- Zhu, M.; Tan, J.; Liu, L.; Tian, J.; Li, L.; Luo, B.; Zhou, C.; Lu, L. Construction of biomimetic artificial intervertebral disc scaffold via 3D printing and electrospinning. Mater. Sci. Eng. C 2021, 128, 112310. [Google Scholar] [CrossRef]
- Kamali, A.; Shamloo, A. Fabrication and evaluation of a bilayer hydrogel-electrospinning scaffold prepared by the freeze-gelation method. J. Biomech. 2020, 98, 109466. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.; Gao, X.; Wang, J.; Chen, H.; Tong, C.; Tan, Y.; Tan, Z. Triple-Layer Vascular Grafts Fabricated by Combined E-Jet 3D Printing and Electrospinning. Ann. Biomed. Eng. 2018, 46, 1254–1266. [Google Scholar] [CrossRef] [PubMed]
- Freystetter, B.; Grab, M.; Grefen, L.; Bischof, L.; Isert, L.; Mela, P.; Bezuidenhout, D.; Hagl, C.; Thierfelder, N. Combining 3D-Printing and Electrospinning to Manufacture Biomimetic Heart Valve Leaflets. J. Vis. Exp. 2022, 181, e63604. [Google Scholar] [CrossRef] [PubMed]
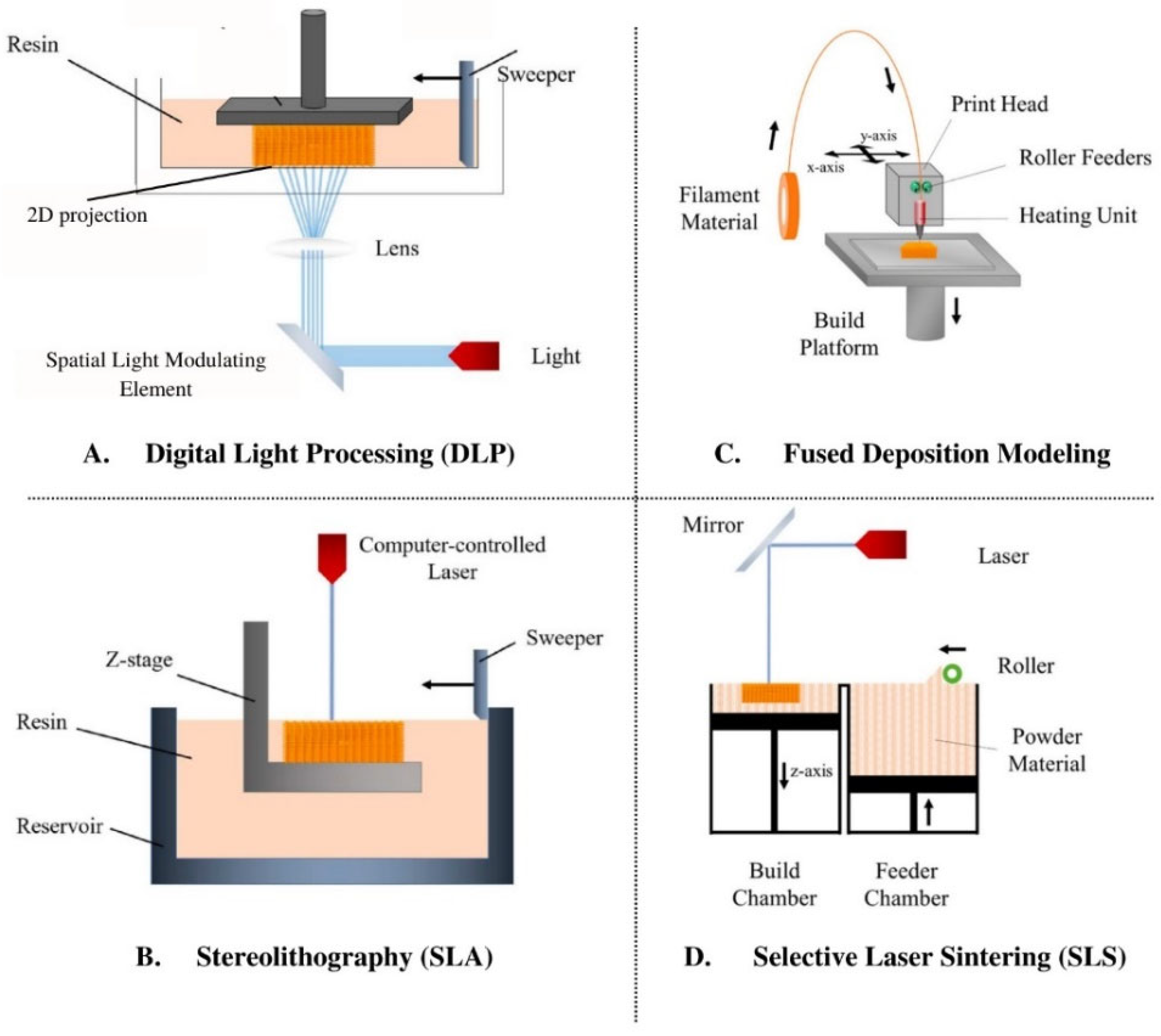
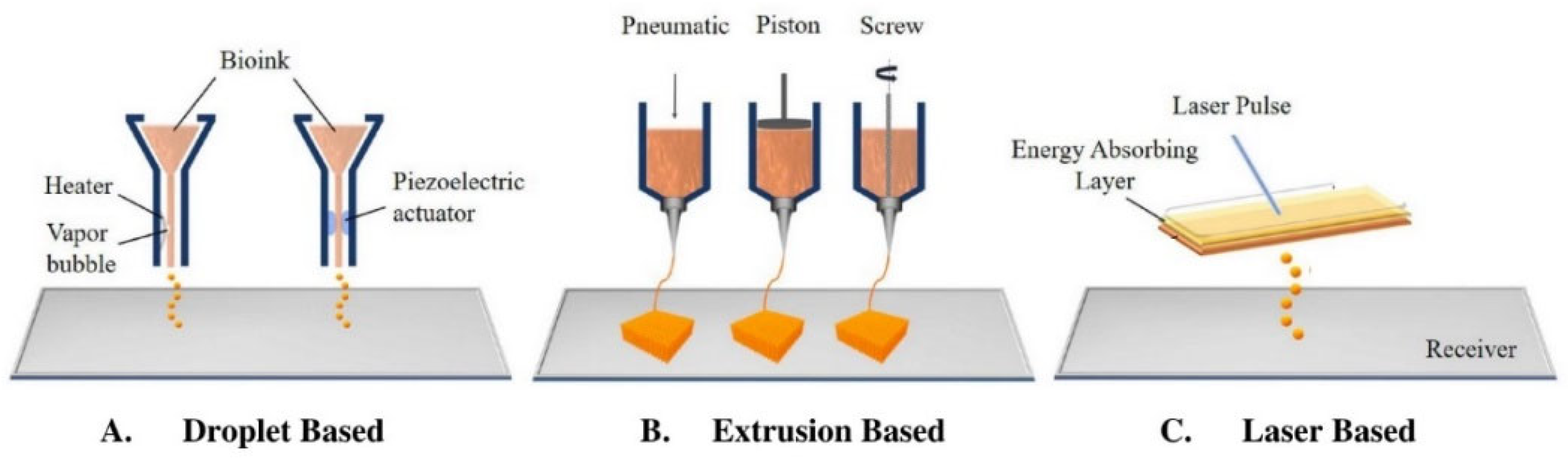
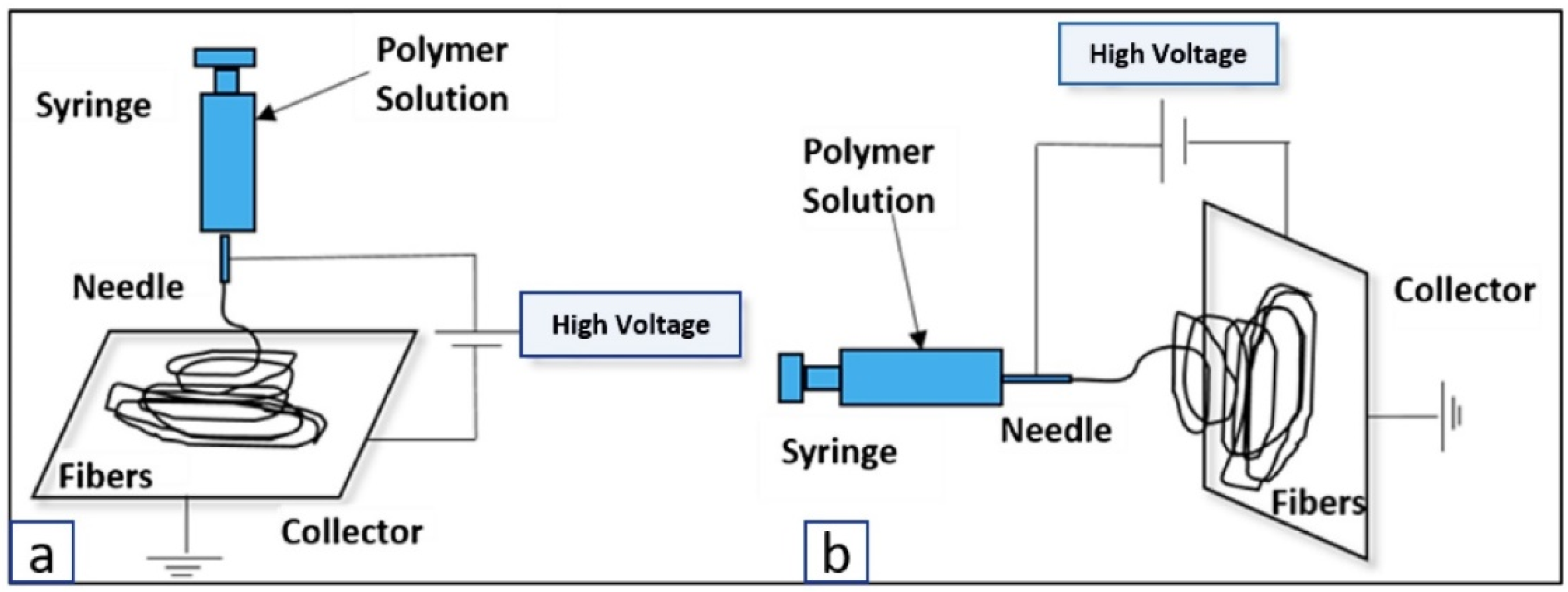
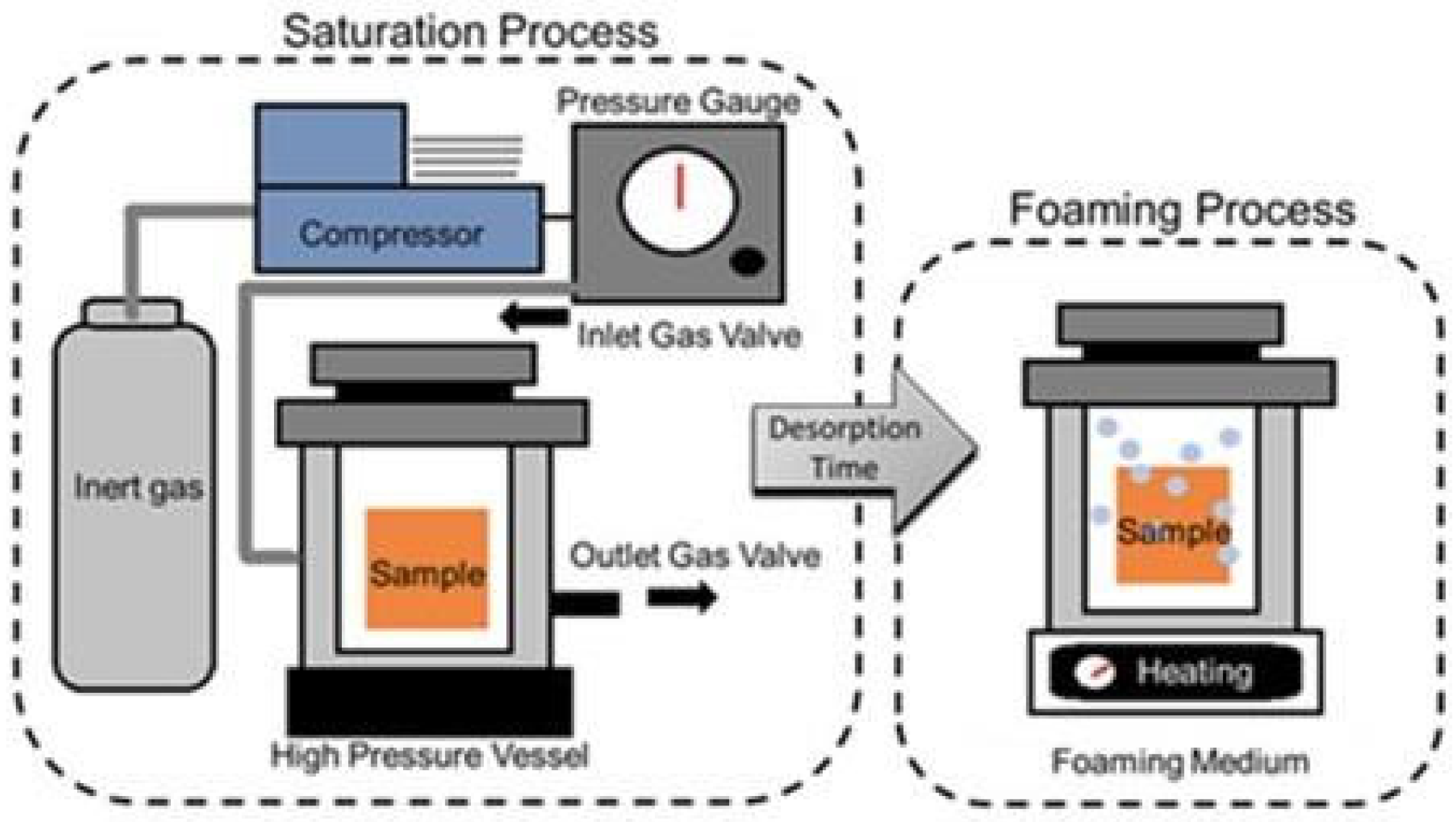


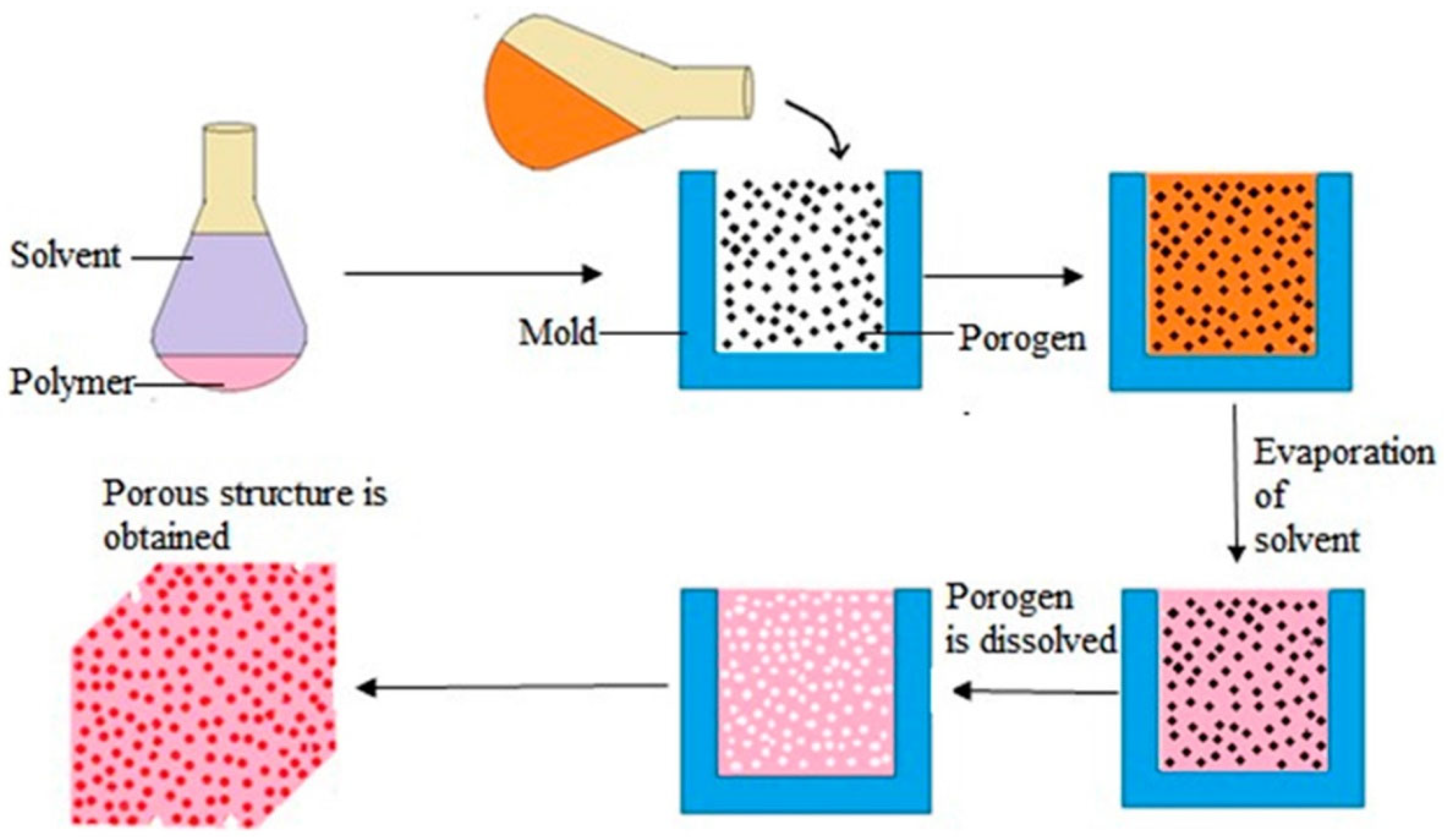
| Polymer | Strength/Elastic Modulus | Fabrication Method | Target Tissue | Advantage(s) | Disadvantage(s) | Notes: |
|---|---|---|---|---|---|---|
| Natural Polymers | ||||||
| Alginate | Hydrogel: <1 kPa to 1000 kPa [166] | Electrospinning; Hydrogels; Cryogels [167,168] | Bone; Cartilage; Ligament [167] | Biocompatible; Encapsulate cells [167,169,170,171,172]. | Lacks mechanical strength; Nondegradable unless ionically crosslinked; Slow degradation [166,167,169,171]. | Alginate gels are typically nanoporous (pore size ~5 nm), allowing for rapid diffusion of small molecules through the gel; Potential for drug delivery [167,173]. |
| Chitosan | Fibers: 3500 +/− 780 kPa Scaffold: 70 +/− 10 kPa [174] | Hydrogel; Cryogel [150,175] | Bone; Cartilage; Ligament; Nerves [169,174] | Antimicrobial properties; Biocompatible; Chemically modifiable [169,176,177,178]. | Limited cell adhesion; Low mechanical strength [169,176,177,178]. | Has been reported to promote bone formation [179]. |
| Collagen I fiber | 100–2900 MPa [180] | 3D-printing; Electrospinning [181] | Bone; Cartilage; Ligament; Skin; Tendon [181] | Biocompatible; Major component of native ACL [169]. | Lacks mechanical strength; Immunogenic [169]. | The addition of hyaluronic acid can prolong degradation and serve as a delivery system for chondrocytes in cartilage tissue engineering [182,183]. |
| Gelatin | Scaffold: 10–100 kPa [184,185] | 3D-printing; Electrospinning; Hydrogels; Cryogels [150,186,187] | Bone; Cardiac; Ligament; Muscle; Skin; Tendon [188] | Biocompatible; Biodegradable; Cost-effective; Cell compatibility; Low toxicity [189]. | No thermal stability; Poor mechanical properties; Short degradation rate [189]. | Gelatin is derived from collagen and can be used as a cost-effective substitute [188]. |
| Silk | 5–12 GPa for Bombyx Mori with sericin, 15–17 GPa without sericin [190] | Electrospinning; Hydrogel; Cryogel [191,192,193] | Bone; Cartilage; Ligament; Skin [169,174,194] | Good tensile strength [37,169]. | Limited cell adhesion; Sericin coating is immunogenic [37,169]. | Two main constituents: fibroins and sericin. Sericin, absent in spider silk, acts as a glue for the fibroin fibers and elicits an immune response. These constituents contain (varying) amounts of alanine, glycine, and serine [195]. |
| Synthetic Materials | ||||||
| Poly(caprolactone) (PCL) | 0.4 GPa; 3.2 MPa [169,180,196] | 3D-printing; Electrospinning; Gas Foaming [197,198,199] | Bone; Ligament; Soft and Hard Tissues [169,200,201,202] | Common FDA-approved suture material; Easily manufactured [169]. | Biologically inert; Slow degradation rate (years) [14,169]. | Elongation at break 80%, Tg = −60 C, Tm = 60 C [196]. |
| Poly(diaxonane) (PDX) | 100,000,000 N/m2 = 0.1 GPa, 2–46 MPa [180,203] | Electrospinning [180] | Bone; Cartilage; Ligament [180] | Common FDA-approved suture material; Easily manufactured; Shape memory [169]. | Rapid loss of mechanical strength [169]. | PDX/50% Hydroxyapatite scaffolds allow for excellent scaffold mineralization for bone tissue engineering [204]. |
| Polyethylene terephthalate (PET) | 1.57–5.2 GPa [205] | 3D-printing; Electrospinning [206,207] | Bone; Ligament; Tendon [208,209,210] | Biocompatible; Biodegradable; High tensile strength; Stiffness [194,210]. | High crystallinity makes it difficult to print [210]. | Frequently made into meshes containing allografts/autografts, or meshes for hernia repair [211,212]. |
| Poly(glycolic acid) (PGA) | 7.0–10 GPa [213] | Electrospinning; Gas Foaming [214,215] | Cartilage; Skin [216,217] | Common FDA-approved suture material; Easily manufactured [169]. | Rapid degradation and loss of mechanical strength; Biologically inert; Acidic degradation byproduct [169]. | Frequently used in combination with other materials as coatings (ex. hyaluronic acid) [218,219]. |
| Poly(glycerol sebacate) (PGS) | 0.04–1.2 MPa [213,220] | 3D-printing; Electrospinning [198,213] | Soft Tissue [201,213] | Biocompatible; Biodegradable; Cost effective; Flexible [213]. | Differences between in vivo and in vitro degradation [221]. | Fast degradation (6 mo in vitro) [213]. |
| Poly(3-hydroxybutyrate) (PHB) | 3 GPa [222] | Electrospinning; Salt Leaching; Solvent Casting [223] | Bone; Cartilage; Skin; Tendon; Nerves [194,223,224] | Biocompatible; Piezoelectric [223]. | Brittleness; Hydrophobicity; Low degradation rate [223]. | Often combined with 3 hydroxyvaleric acid (HV) to increase degradation rate and reduce crystallinity; elongation at break 2%; Tg = 1–2 C, Tm = 170 [194,196]. |
| Poly(3-hydroxybutyrate-co-3-hydroxy valerate) (PHBV) | Tensile modulus = 1100 MPa [196] | Electrospinning [225] | Cardiac; Cartilage; Liver; Nerve [225] | Biocompatible; Biodegradable; Low toxicity; Piezoelectric; Thermoplasticity [226,227]. | Hydrophobic; Low mechanical strength; Often requires additives to promote cell adhesion; Poor mechanical properties [226]. | Elongation at break 17%; Tg = 2 C; Tm = 145 C; Tensile strength = 20 MPa [196,225,228]. |
| Poly(lactic-co-glycolic acid) (PLGA) | 40.4–134.5 MPa [213] | Electrospinning [197] | Ligament; Vascular [169,197] | Degradation rate can be tailored by changing the ratio of PLA:PGA [14,169]. | Acidic degradation byproducts; Biologically inert; Reduce cell adhesion; Non-hydrophobic [14,169]. | Degradation rate of 32% weight loss observed at 5 weeks in vitro [213]. |
| Poly(L-lactic acid) (PLLA) | 1–4 GPa [213] | 3D-printing; Electrospinning [229] | Ligament; Neural; Hard and Soft Tissues [169,230] | Easily manufactured; Improved cell adhesion; Slow degradation rate [169]. | Acidic degradation byproduct; Biologically inert [169]. | Viscoelastic properties can be improved by using braid-twist method [169]. |
| Polyvinyl alcohol (PVA) | 48 +/− 3 GPa [231] | Electrospinning; Cryogels [232,233] | Bone; Skin [234,235] | Biocompatible; Good mechanical properties; Non-toxic [169,231]. | Low thermal stability [169,231]. | Young’s modulus and compressive strength increases with PVA concentration [236]. |
| ECM-Derived Polymers | ||||||
| Chondroitin sulfate | Hydrogel: 1.2–11.3 kPa [237,238] | Electrospinning; Hydrogel [237,239] | Bone; Cartilage; Neural; Skin [240] | Biocompatible; Biodegradable; Readily available; Water soluble [240]. | Differences in material quality; Fast degradation; Low thermal resistance; Tunability Weak mechanical properties [240]. | Source: Joint, Nasal, and Tracheal cartilage. Support osteogenesis and suppress bone resorption [241,242,243,244]. |
| Elastin | Bovine ligament: 1 MPa [245] | Electrospinning [246] | Ligament; Skin [247,248] | Provides elasticity, resiliency, cell adhesion and growth; Artificial forms are available [247]. | Difficult to purify; May stimulate an immune response [249,250]. | Source: Aorta. Used in scaffolding for cell generation therapy; Elasticity provides the necessary mechanical cues for maintaining and expanding hematopoietic stem cells [249,250]. |
| Fibronectin | Fibers: 1–15 MPa [251] | Electrospinning; Hydrogel [249,252] | Bone; Dental Tissue [253] | Promotes cell adhesion, migration, spreading, and proliferation; Wound healing [247,253]. | Decreased cell viability; Insufficient cell-anchorage cues [252]. | Source: Plasma; Promote cell adhesion and control cellular function via peptide domains. Angiogenic differentiation [249,254,255,256,257]. |
| Heparin/Heparan Sulfate-Derived | Hydrogel: 2.3 kPa [258] | Hydrogel [258] | Bone; Cartilage; Neural; Vascular [258,259,260,261] | Anticoagulant; Anti-inflammatory [127,259,262,263]. | Human umbilical vein endothelial cells (HUVEC) metabolic activity is sensitive to heparin (negatively impacted at 10 and 1000 μg/mL) [261]. | Excellent matrix for in vitro culture of articular chondrocytes; Facilitate interactions at cell surface receptors [258,264]. |
| Hyaluronic acid | Hydrogel: 200 kPa [265] | Bioprinting; Electrospinning; Hydrogel; Cryogel; Salt Leaching [266,267] | Bone; Cartilage; Skin [268,269,270,271] | Chemically modified (crosslinked) to improve viscoelastic properties [268]. | Poor biomechanical properties (viscoelasticity and half-life); Pure hydroxyapatite (HA) gel does not permit adhesion [268,272]. | Source: Rooster comb, Umbilical cords, Vitreous humor. The HA-based hydrogel can maintain morphology of chondrocytes [266,267,273,274]. |
| Laminin | Fiber: (blended with PCL) 21.83 kPa | Electrospinning [275] | Nervous System; Skeletal Muscle; Vascular [276] | Biomaterial enrichment; Cell adhesion, differentiation, migration, and wound healing [247]. | Difficult to synthesize long peptides and mimic structure [277]. | Source: Heart, Placenta. Provide basement membrane-like scaffold. Participate in biological process (angiogenesis and neural differentiation) [249,275,278,279]. |
| Growth Factor | Target & Application in Tissue Engineering | Fabrication Methods | References |
|---|---|---|---|
| Bone morphogenetic protein-2 (BMP-2) | Bone regeneration; Cell proliferation | 3D-printing; Bioprinting; Electrospinning; Gas foaming; Hydrogel; Cryogel; Solvent Casting/Particulate Leaching | [300,301,302,303,304,305,306,307,308] |
| Epidermal growth factor (EGF) | Cell proliferation; Neural Stem Cell Differentiation; Wound healing | Bioprinting; Electrospinning; Gas foaming; Hydrogel; Cryogel; Solvent Casting/Particulate Leaching | [309,310,311,312,313,314,315,316,317,318] |
| Fibroblast growth factor (FGF) | Angiogenesis; Bone/Cartilage Regeneration; Cell Proliferation; Nerve Growth | 3D-printing; Electrospinning; Gas Foaming; Solvent Casting/Particulate Leaching | [262,263,314,319,320,321,322,323,324,325,326,327] |
| Nerve growth factor (NGF) | Axonal Growth; Neurite Extension in Central and Peripheral Nervous Systems | 3D-printing; Bioprinting; Electrospinning; Hydrogel; Cryogel | [260,328,329,330,331,332,333,334,335,336] |
| Platelet-derived growth factor (PDGF) | Angiogenesis; Endothelial Cell Proliferation; Wound Healing | 3D-printing; Electrospinning; Gas foaming; Hydrogel | [326,337,338,339,340,341,342,343] |
| Transforming growth factor-β (TGF-β) | Bone/Cartilage Regeneration; ECM Production | 3D-printing; Bioprinting; Electrospinning; Hydrogel; Cryogel; Solvent Casting/Particulate Leaching | [325,326,341,344,345,346,347,348,349,350] |
| Vascular endothelial growth factor (VEGF) | Angiogenesis; Endothelial Cell Proliferation | 3D-printing; Bioprinting; Electrospinning; Gas foaming; Hydrogel; Cryogel; Solvent Casting/Particulate Leaching | [264,303,304,318,351,352,353,354,355,356,357] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vesvoranan, O.; Anup, A.; Hixon, K.R. Current Concepts and Methods in Tissue Interface Scaffold Fabrication. Biomimetics 2022, 7, 151. https://doi.org/10.3390/biomimetics7040151
Vesvoranan O, Anup A, Hixon KR. Current Concepts and Methods in Tissue Interface Scaffold Fabrication. Biomimetics. 2022; 7(4):151. https://doi.org/10.3390/biomimetics7040151
Chicago/Turabian StyleVesvoranan, Oraya, Amritha Anup, and Katherine R. Hixon. 2022. "Current Concepts and Methods in Tissue Interface Scaffold Fabrication" Biomimetics 7, no. 4: 151. https://doi.org/10.3390/biomimetics7040151
APA StyleVesvoranan, O., Anup, A., & Hixon, K. R. (2022). Current Concepts and Methods in Tissue Interface Scaffold Fabrication. Biomimetics, 7(4), 151. https://doi.org/10.3390/biomimetics7040151






