Semi-Automatic Algorithms for Estimation and Tracking of AP-Diameter of the IVC in Ultrasound Images
Abstract
1. Introduction
2. Background and Related Work
3. Proposed Algorithms
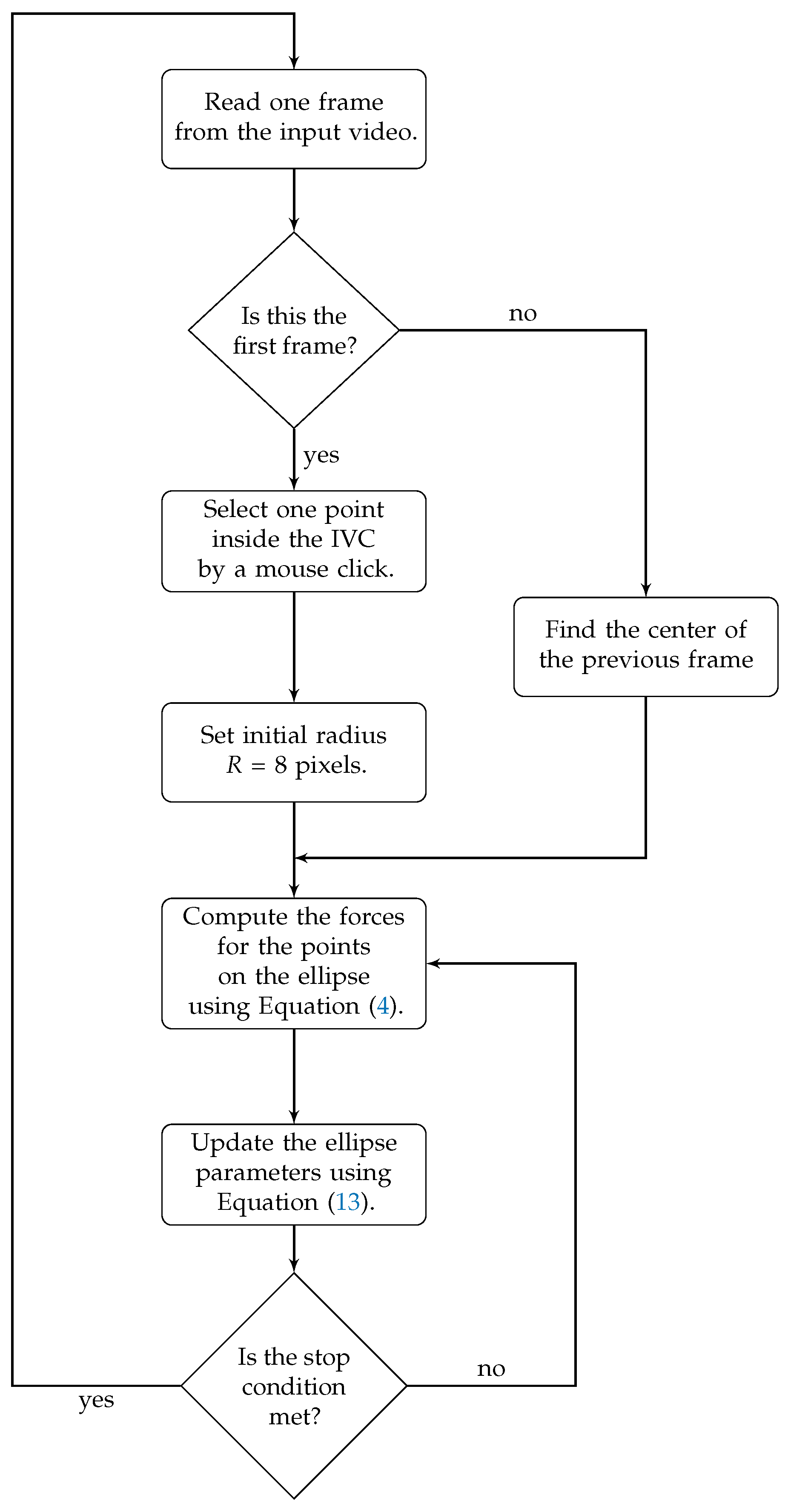
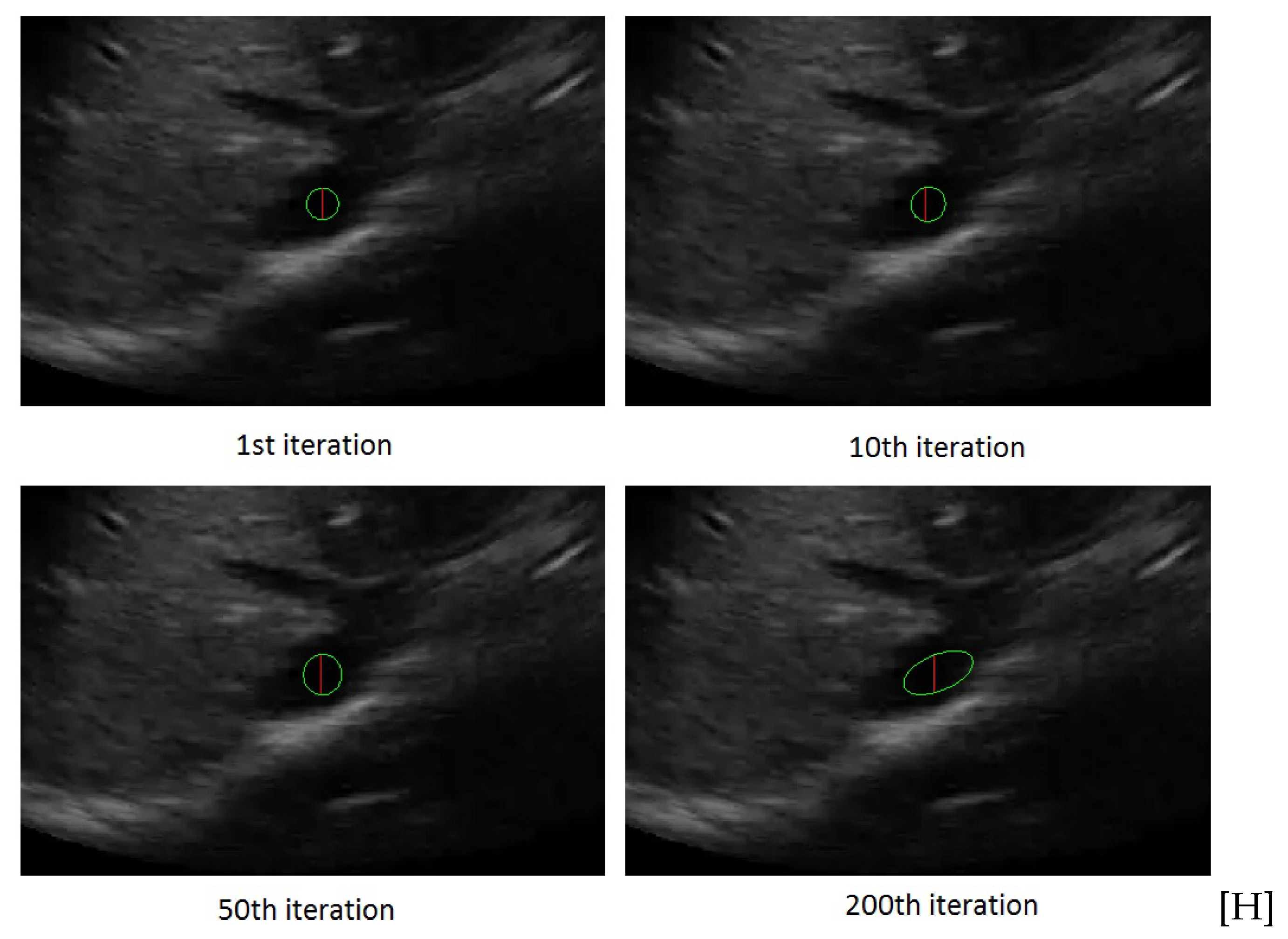
3.1. Active Ellipse Model
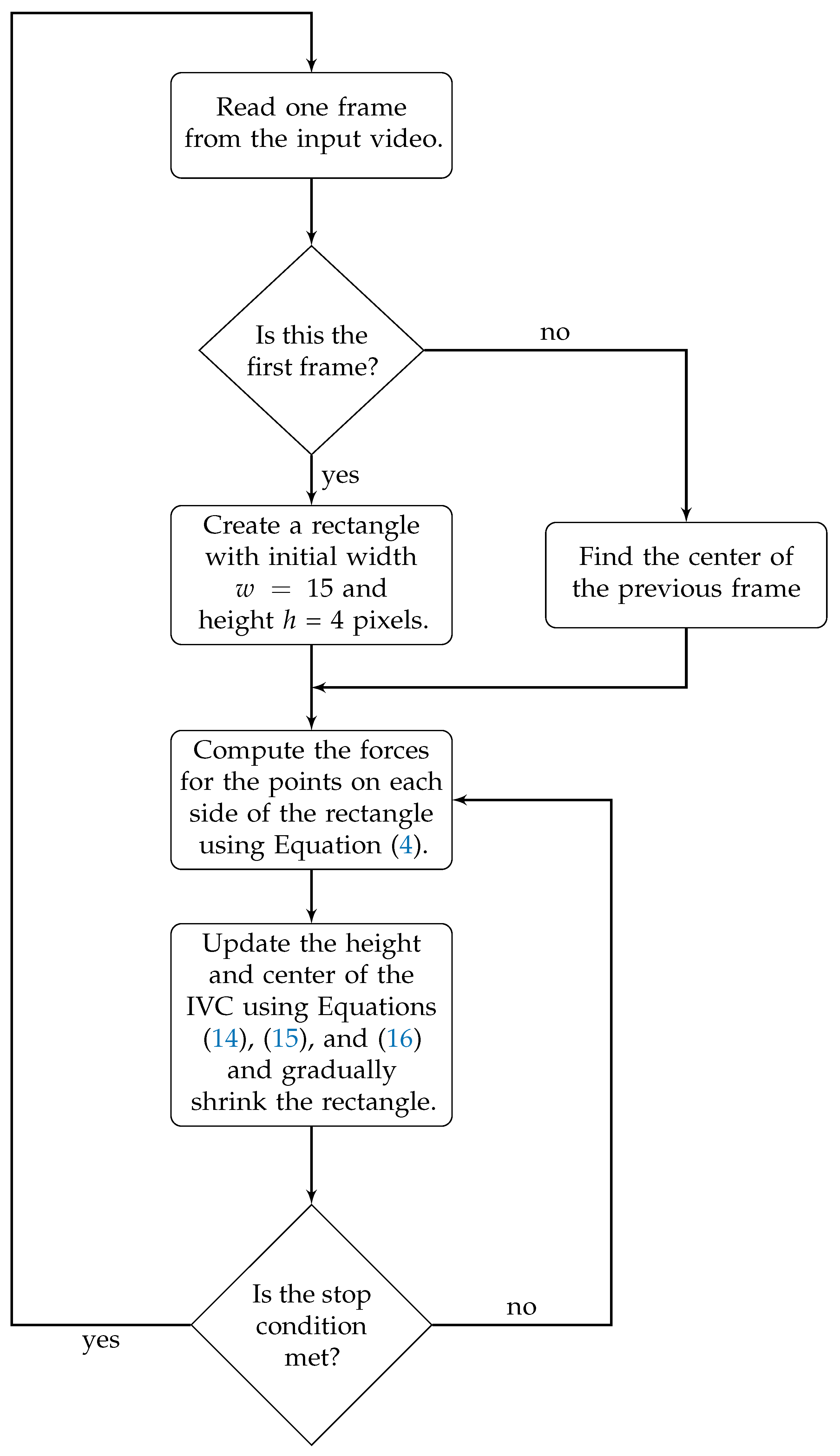
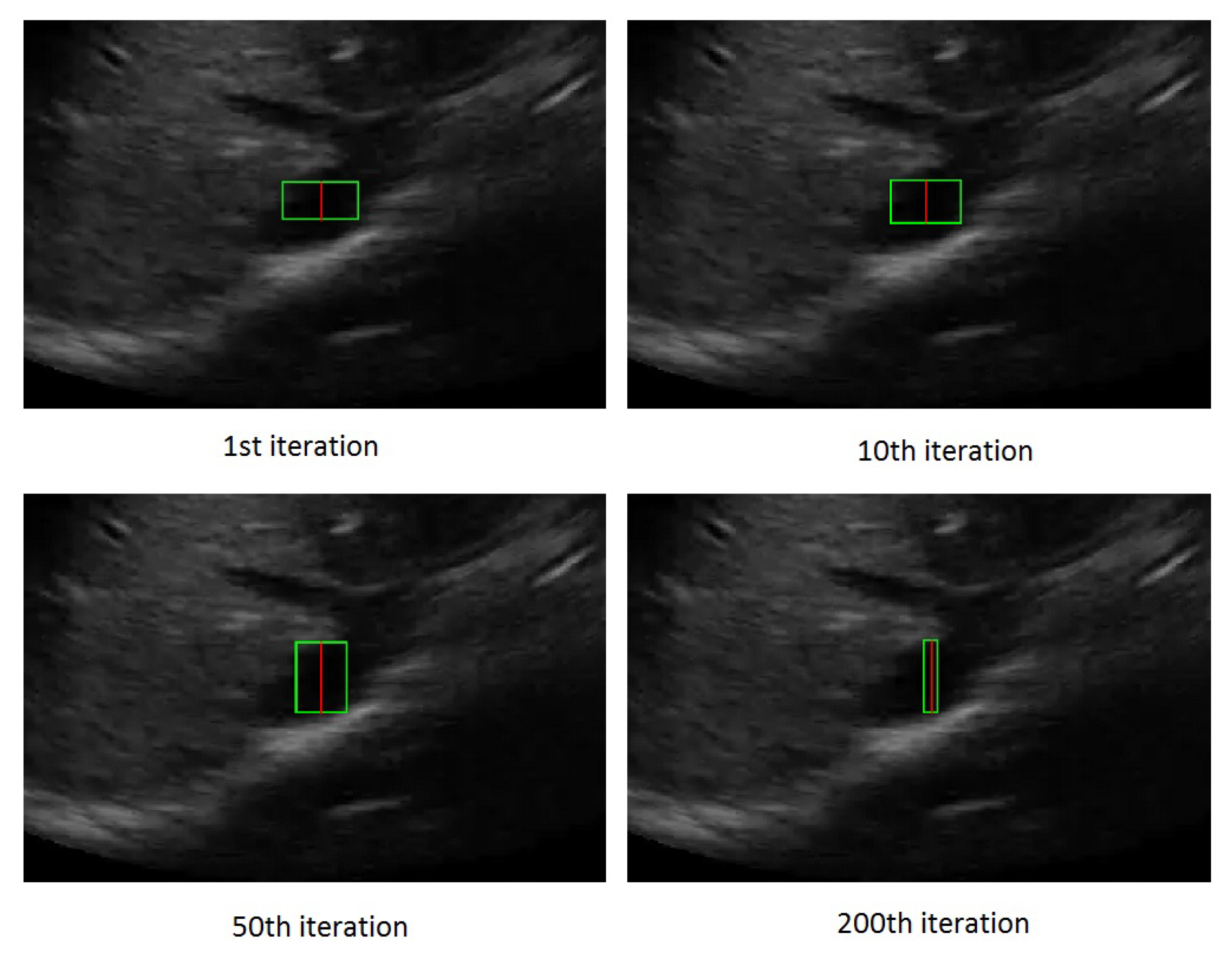
3.2. Active Rectangle Model
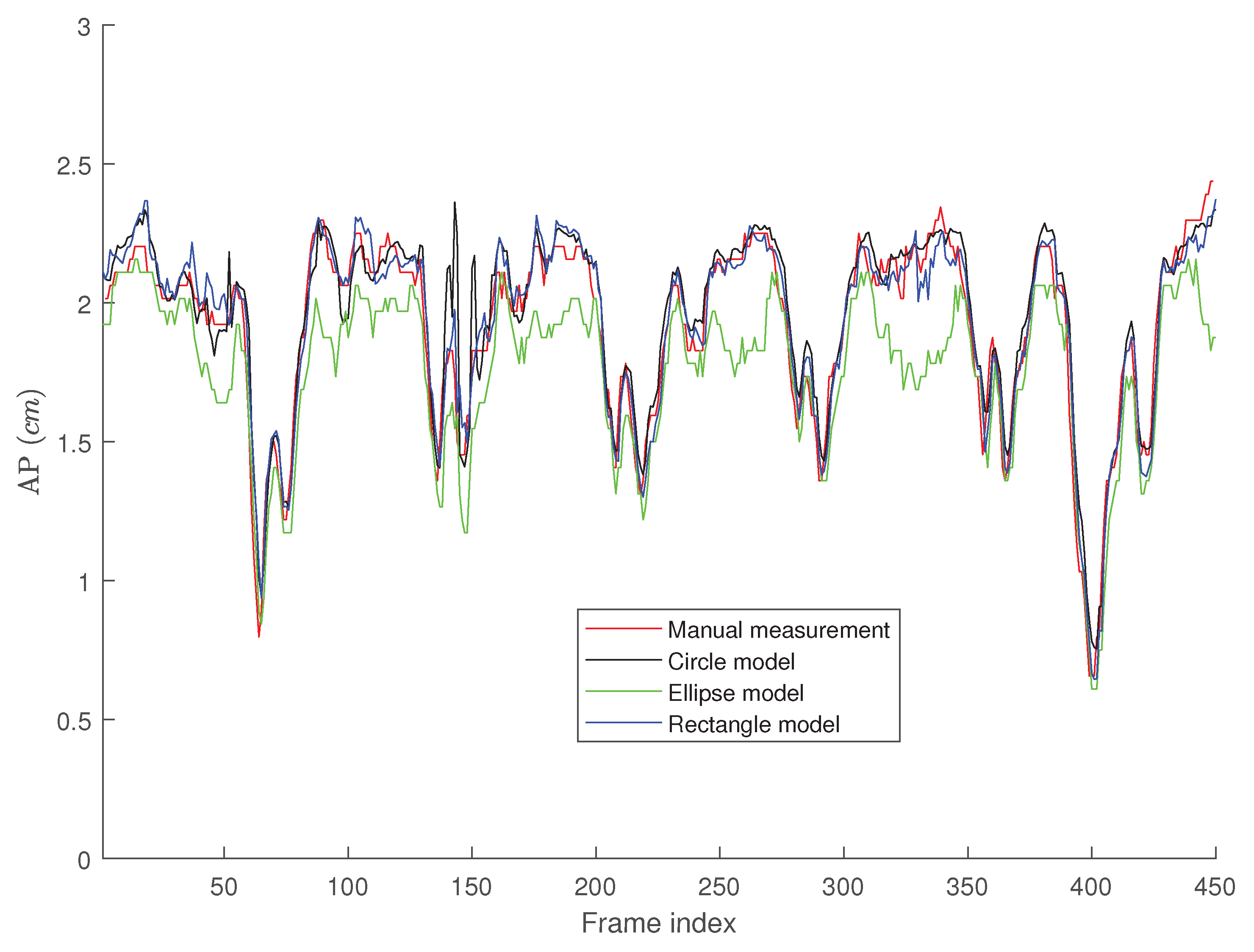
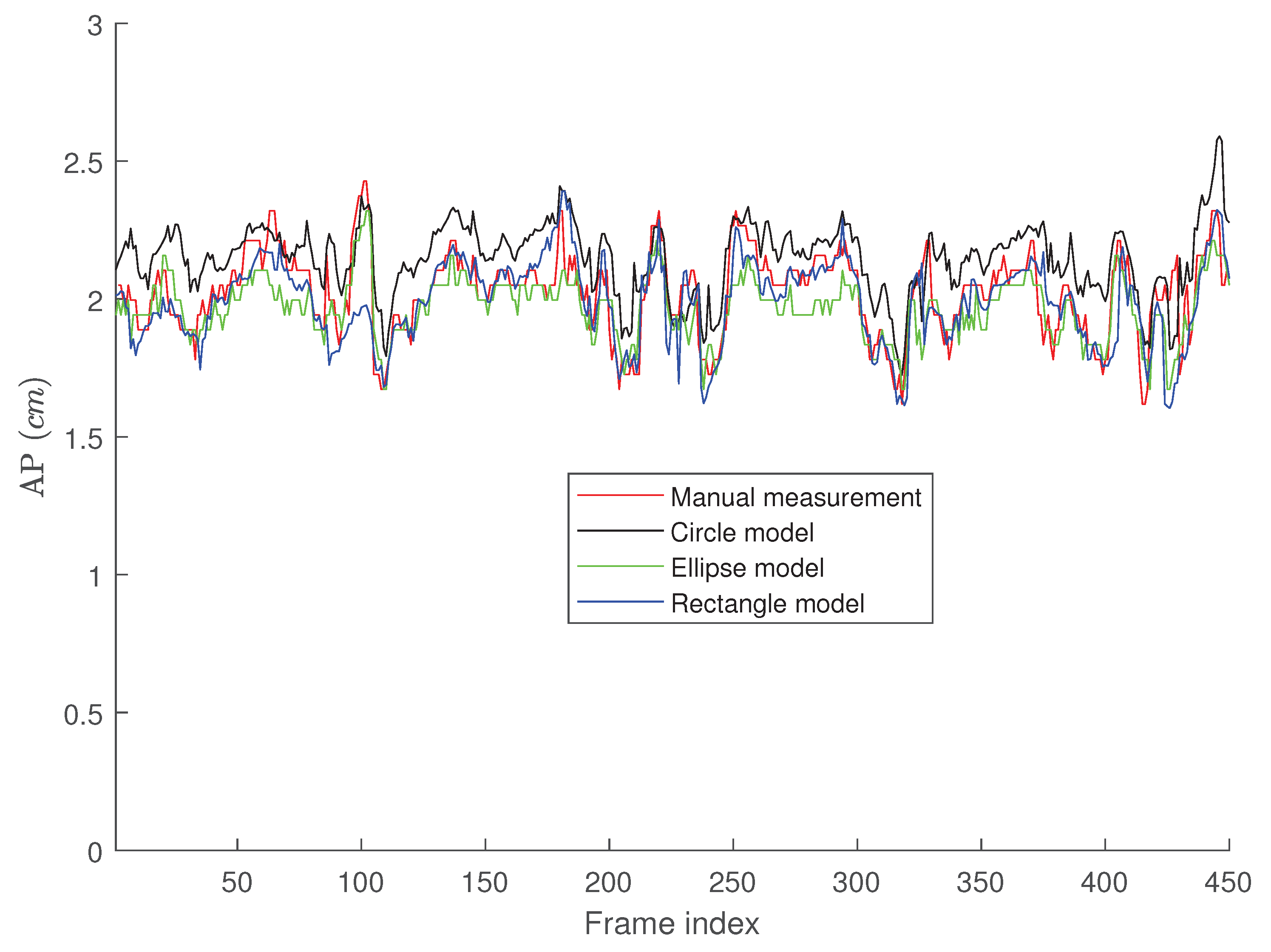
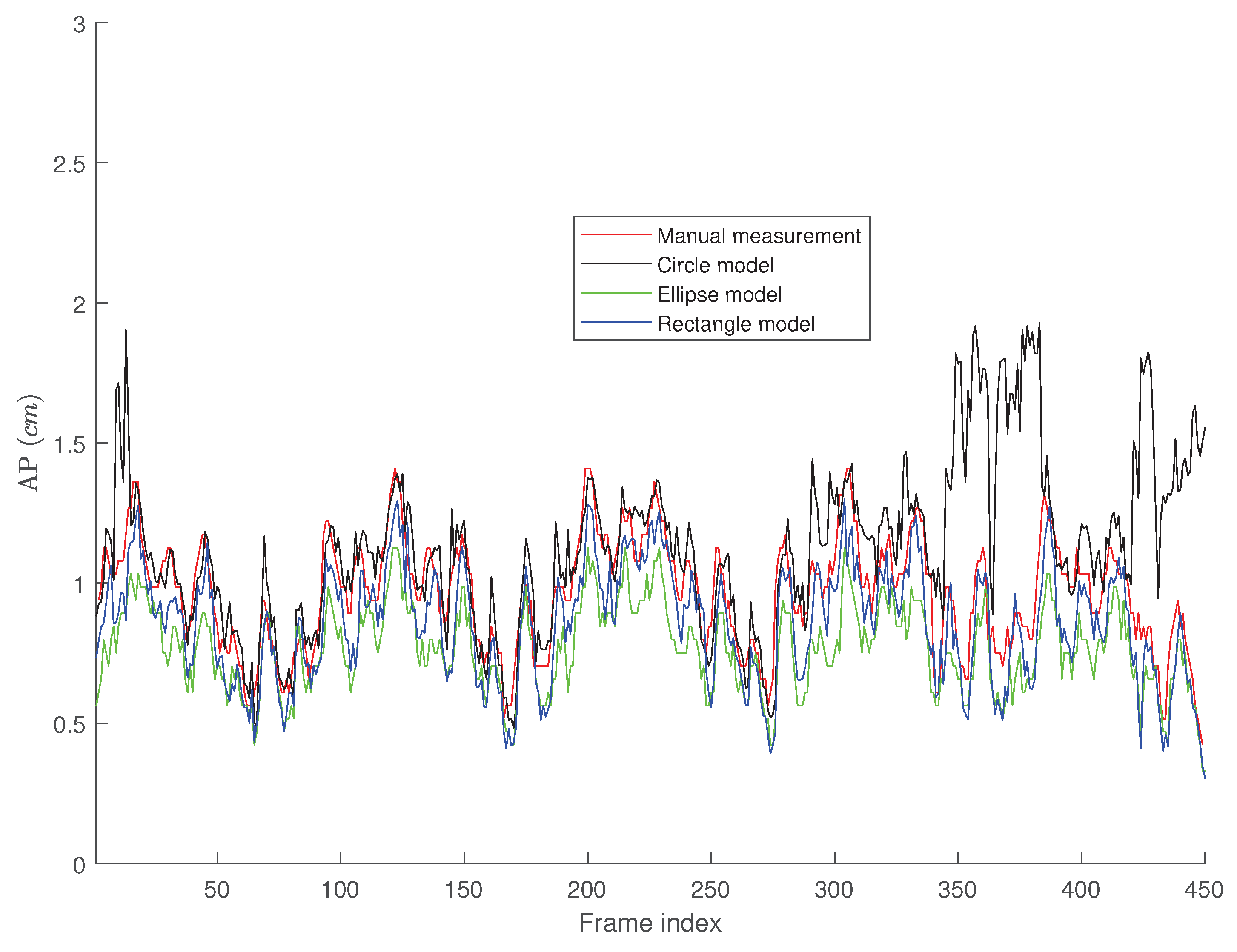
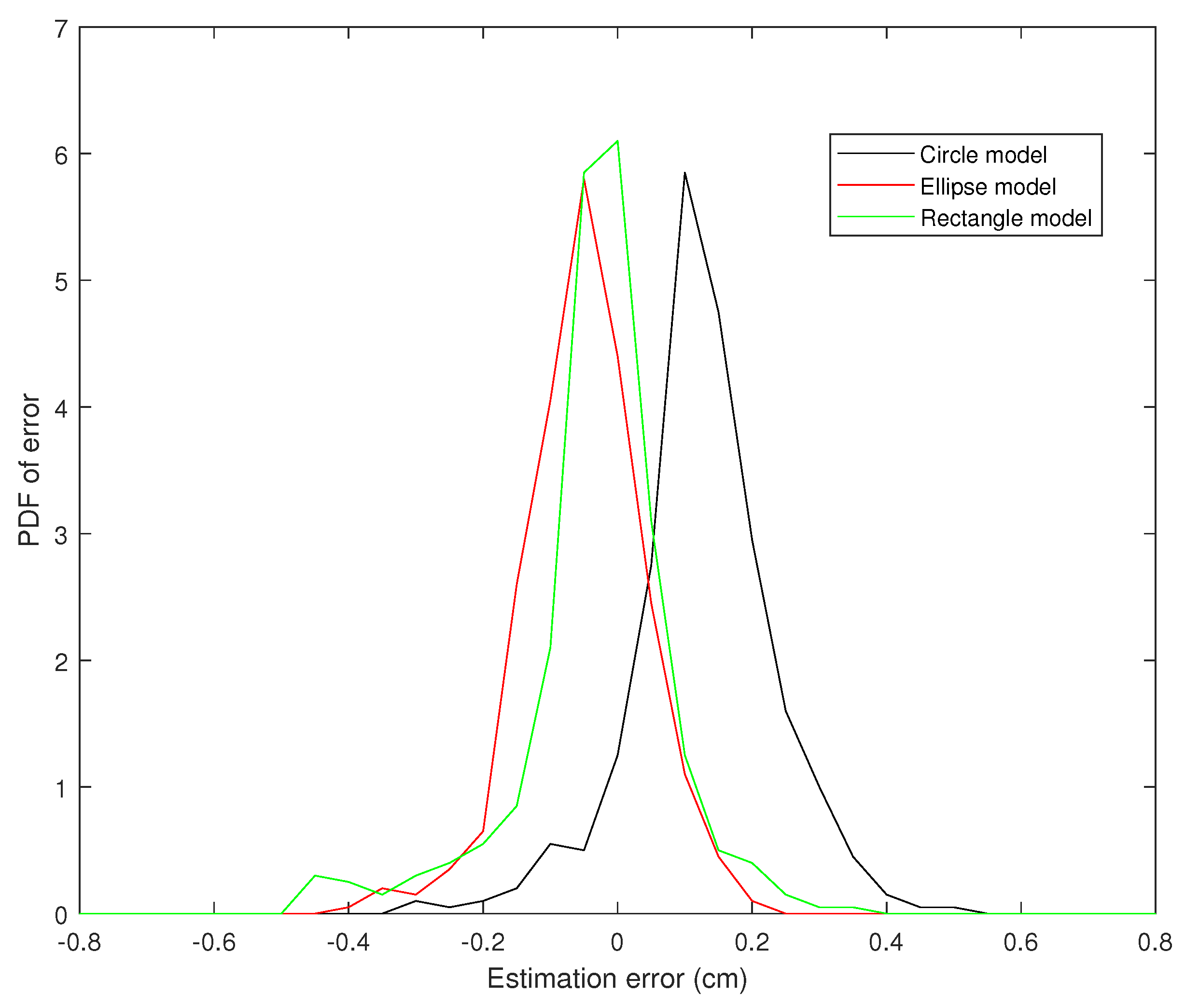
4. Results
Tracking Performance
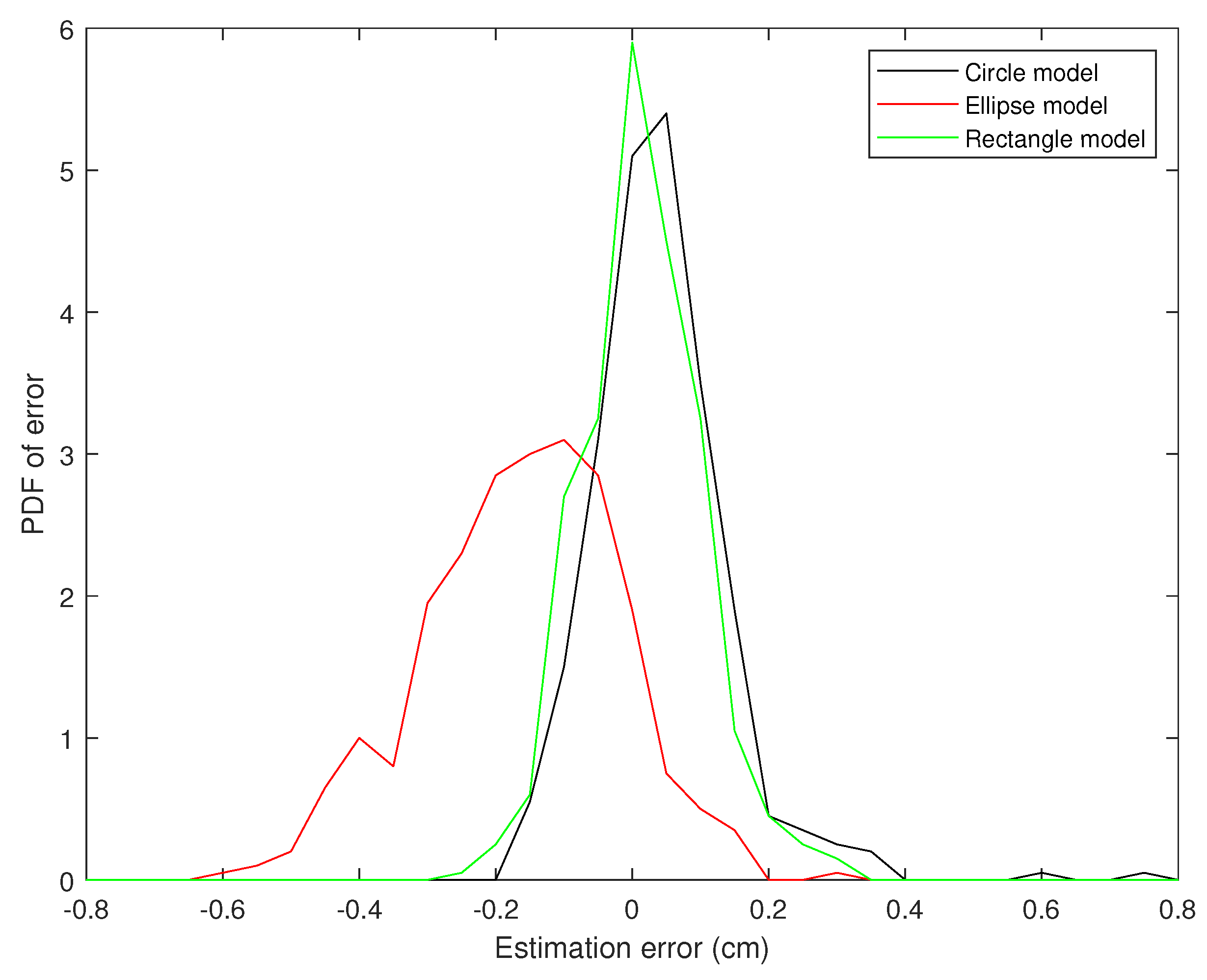
5. Discussion
5.1. The Performance of the Proposed Algorithms
5.2. Complexity Comparison
6. Conclusions
Author Contributions
Conflicts of Interest
Abbreviations
| AP | Anterior–posterior |
| IVC | Inferior vena cava |
| CSA | Cross sectional area. |
References
- Smyrniotis, V.; Kostopanagiotou, G.; Theodoraki, K.; Tsantoulas, D.; Contis, J.C. The role of central venous pressure and type of vascular control in blood loss during major liver resections. Am. J. Surg. 2004, 187, 398–402. [Google Scholar] [CrossRef] [PubMed]
- Rivers, E.; Nguyen, B.; Havstad, S.; Ressler, J.; Muzzin, A.; Knoblich, B.; Peterson, E.; Tomlanovich, M. Early goal-directed therapy in the treatment of severe sepsis and septic shock. N. Engl. J. Med. 2001, 345, 1368–1377. [Google Scholar] [CrossRef] [PubMed]
- Ikhsan, M.; Tan, K.K.; Putra, A.S.; Chew, T.H.S.; Kong, C.F. Automatic identification of blood vessel cross-section for central venous catheter placement using a cascading classifier. In Proceedings of the 2017 39th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Jeju Island, Korea, 11–15 July 2017; pp. 1489–1492. [Google Scholar]
- Ogum, C.; Hariharan, S.; Chen, D. Non-invasive central venous pressure estimation by ultrasound-guided internal jugular vein cross-sectional area measurement. Biomed. Phys. Eng. Express 2016, 2, 025004. [Google Scholar] [CrossRef]
- Charron, C.; Caille, V.; Jardin, F.; Vieillard-Baron, A. Echocardiographic measurement of fluid responsiveness. Curr. Opin. Crit. Care 2006, 12, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Durairaj, L.; Schmidt, G.A. Fluid therapy in resuscitated sepsis: Less is more. Chest J. 2008, 133, 252–263. [Google Scholar] [CrossRef] [PubMed]
- Barbier, C.; Loubières, Y.; Schmit, C.; Hayon, J.; Ricôme, J.L.; Jardin, F.; Vieillard-Baron, A. Respiratory changes in inferior vena cava diameter are helpful in predicting fluid responsiveness in ventilated septic patients. Intensive Care Med. 2004, 30, 1740–1746. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Zhu, L.; Qin, J.; Chui, Y.P.; Li, B.N.; Heng, P.A. Multiscale geodesic active contours for ultrasound image segmentation using speckle reducing anisotropic diffusion. Opt. Lasers Eng. 2014, 54, 105–116. [Google Scholar] [CrossRef]
- Sudha, S.; Suresh, G.; Sukanesh, R. Speckle noise reduction in ultrasound images by wavelet thresholding based on weighted variance. Int. J. Comput. Theory Eng. 2009, 1, 7–12. [Google Scholar]
- Wagner, R.F.; Smith, S.W.; Sandrik, J.M.; Lopez, H. Statistics of speckle in ultrasound B-scans. IEEE Trans. Sonics Ultrason. 1983, 30, 156–163. [Google Scholar] [CrossRef]
- Seabra, J.C.; Ciompi, F.; Pujol, O.; Mauri, J.; Radeva, P.; Sanches, J. Rayleigh mixture model for plaque characterization in intravascular ultrasound. IEEE Trans. Biomed. Eng. 2011, 58, 1314–1324. [Google Scholar] [CrossRef]
- Pereyra, M.; Dobigeon, N.; Batatia, H.; Tourneret, J.Y. Segmentation of skin lesions in 2-D and 3-D ultrasound images using a spatially coherent generalized Rayleigh mixture model. IEEE Trans. Med. Imaging 2012, 31, 1509–1520. [Google Scholar] [CrossRef] [PubMed]
- Tuthill, T.; Sperry, R.; Parker, K. Deviations from Rayleigh statistics in ultrasonic speckle. Ultrason. Imaging 1988, 10, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Karami, E.; Shehata, M.S.; Smith, A. Tracking of the Internal Jugular Vein in Ultrasound Images Using Optical Flow. In Proceedings of the 30th Annual IEEE Canadian Conference on Electrical and Computer Engineering (CCECE), Windsor, ON, Canada, 30 April–3 May 2017; pp. 1–4. [Google Scholar]
- Karami, E.; Shehata, M.; Smith, A. Ultrasound Image Segmentation Techniques for Tracking and Measurement of the Internal Jugular Vein. In Proceedings of the 24th Annual Newfoundland Electrical and Computer Engineering Conference (NECEC), St. John’s, NL, Canada, 5–6 November 2015. [Google Scholar]
- Karami, E.; Shehata, M.; McGuire, P.; Smith, A. A Semi-automated Technique for Internal Jugular Vein Segmentation in Ultrasound Images Using Active Contours. In Proceedings of the 2016 IEEE-EMBS International Conference on Biomedical and Health Informatics (BHI), Las Vegas, NV, USA, 24–27 February 2016; pp. 184–187. [Google Scholar]
- Liu, B.; Cheng, H.; Huang, J.; Tian, J.; Tang, X.; Liu, J. Probability density difference-based active contour for ultrasound image segmentation. Pattern Recognit. 2010, 43, 2028–2042. [Google Scholar] [CrossRef]
- Talebi, M.; Ayatollahi, A.; Kermani, A. Medical ultrasound image segmentation using genetic active contour. J. Biomed. Sci. Eng. 2011, 4, 105. [Google Scholar] [CrossRef]
- Noble, J.A. Ultrasound image segmentation and tissue characterization. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 2010, 224, 307–316. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Kiyomatsu, H.; Nakagawa, K.; Wang, J.; Kobayashi, E.; Sakuma, I. Accurate vessel segmentation in ultrasound images using a local-phase-based snake. Biomed. Signal Process. Control 2018, 43, 236–243. [Google Scholar] [CrossRef]
- Yim, P.J.; Foran, D.J. Volumetry of hepatic metastases in computed tomography using the watershed and active contour algorithms. In Proceedings of the IEEE 16th IEEE Symposium on Computer-Based Medical Systems, New York, NY, USA, 26–27 June 2003; pp. 329–335. [Google Scholar]
- Ali, S.; Madabhushi, A. An integrated region-, boundary-, shape-based active contour for multiple object overlap resolution in histological imagery. IEEE Trans. Med. Imaging 2012, 31, 1448–1460. [Google Scholar] [CrossRef]
- Nakamura, K.; Tomida, M.; Ando, T.; Sen, K.; Inokuchi, R.; Kobayashi, E.; Nakajima, S.; Sakuma, I.; Yahagi, N. Cardiac variation of inferior vena cava: New concept in the evaluation of intravascular blood volume. J. Med. Ultrason. 2013, 40, 205–209. [Google Scholar] [CrossRef]
- Baust, M. Polar Active Contours for Medical Applications. Ph.D. Thesis, Technische Universität München, München, Germany, 2012. [Google Scholar]
- Karami, E.; Shehata, M.; Smith, A. Segmentation and Tracking of Inferior Vena Cava in Ultrasound Images Using a Novel Polar Active Contour Algorithm. In Proceedings of the 5th IEEE Global Conference on Signal and Information Processing (GlobalSIP2017), Montreal, QC, Canada, 14–16 November 2017; pp. 1–5. [Google Scholar]
- Karami, E.; Shehata, M.; Smith, A. Estimation and Tracking of AP-diameter of the Inferior Vena Cava in Ultrasound Images Using a Novel Active Circle Algorithm. Comput. Biol. Med. 2018, 98, 16–25. [Google Scholar] [CrossRef]
- Karami, E.; Shehata, M.S.; Smith, A. Adaptive Polar Active Contour for Segmentation and Tracking in Ultrasound Videos. IEEE Trans. Circ. Syst. Video Technol. 2018. [Google Scholar] [CrossRef]
- Blehar, D.J.; Resop, D.; Chin, B.; Dayno, M.; Gaspari, R. Inferior vena cava displacement during respirophasic ultrasound imaging. Crit. Ultrasound J. 2012, 4, 18. [Google Scholar] [CrossRef] [PubMed]
- Guerrero, J.; Salcudean, S.E.; McEwen, J.A.; Masri, B.A.; Nicolaou, S. Real-time vessel segmentation and tracking for ultrasound imaging applications. IEEE Trans. Med. Imaging 2007, 26, 1079–1090. [Google Scholar] [CrossRef] [PubMed]


| Subject No. | (1) | (2) | (3) | (4) | (5) | (6) | (7) | (8) | Ave. | |
|---|---|---|---|---|---|---|---|---|---|---|
| Method | ||||||||||
| Circle model | 0.10 | 0.16 | 0.16 | 0.25 | 0.25 | 0.10 | 0.11 | 0.26 | 0.17 | |
| Ellipse model | 0.21 | 0.19 | 0.14 | 0.26 | 0.18 | 0.11 | 0.11 | 0.20 | 0.35 | |
| Rectangle model | 0.08 | 0.11 | 0.12 | 0.23 | 0.14 | 0.10 | 0.10 | 0.18 | 0.12 | |
| Subject No. | (1) | (2) | (3) | (4) | (5) | (6) | (7) | (8) | |
|---|---|---|---|---|---|---|---|---|---|
| Method | |||||||||
| Circle model | 0.06 | 0.29 | 0.48 | 0.57 | 0.75 | 0.41 | 0.44 | 0.43 | |
| Ellipse model | 0.11 | 0.32 | 0.35 | 0.59 | 0.48 | 0.54 | 0.47 | 0.38 | |
| Rectangle model | 0.05 | 0.18 | 0.19 | 0.42 | 0.28 | 0.37 | 0.39 | 0.29 | |
| Subject No. | (1) | (2) | (3) | (4) | (5) | (6) | (7) | (8) | |
|---|---|---|---|---|---|---|---|---|---|
| Method | |||||||||
| Circle model | 0.9987 | 0.9987 | 0.9652 | 0.7971 | 0.9985 | 0.9986 | 0.9988 | 0.9958 | |
| Ellipse model | 0.9974 | 0.9985 | 0.9945 | 0.9981 | 0.9986 | 0.9985 | 0.9991 | 0.9850 | |
| Rectangle model | 0.9993 | 0.9994 | 0.9949 | 0.9991 | 0.9992 | 0.9991 | 0.9994 | 0.9985 | |
| Subject No. | (1) | (2) | (3) | (4) | (5) | (6) | (7) | (8) | Ave. | |
|---|---|---|---|---|---|---|---|---|---|---|
| Method | ||||||||||
| Circle model | 0.41 | 0.39 | 0.63 | 0.65 | 0.36 | 0.49 | 0.55 | 0.64 | 0.51 | |
| Ellipse model | 0.42 | 0.43 | 0.31 | 0.47 | 0.44 | 0.49 | 0.54 | 0.56 | 0.45 | |
| Rectangle model | 0.37 | 0.33 | 0.27 | 0.28 | 0.29 | 0.47 | 0.54 | 0.55 | 0.39 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karami, E.; Shehata, M.S.; Smith, A. Semi-Automatic Algorithms for Estimation and Tracking of AP-Diameter of the IVC in Ultrasound Images. J. Imaging 2019, 5, 12. https://doi.org/10.3390/jimaging5010012
Karami E, Shehata MS, Smith A. Semi-Automatic Algorithms for Estimation and Tracking of AP-Diameter of the IVC in Ultrasound Images. Journal of Imaging. 2019; 5(1):12. https://doi.org/10.3390/jimaging5010012
Chicago/Turabian StyleKarami, Ebrahim, Mohamed S. Shehata, and Andrew Smith. 2019. "Semi-Automatic Algorithms for Estimation and Tracking of AP-Diameter of the IVC in Ultrasound Images" Journal of Imaging 5, no. 1: 12. https://doi.org/10.3390/jimaging5010012
APA StyleKarami, E., Shehata, M. S., & Smith, A. (2019). Semi-Automatic Algorithms for Estimation and Tracking of AP-Diameter of the IVC in Ultrasound Images. Journal of Imaging, 5(1), 12. https://doi.org/10.3390/jimaging5010012






