1. Introduction
In certain areas of application, tailor-made plastics now have properties which render them superior to natural materials and irreplaceable for modern technical applications. Composite materials containing plastic and metal parts are often used as electrical conductors, for example, in the automotive industry [
1]. These plastic-metal hybrid components require complex manufacturing processes to shape the metal parts and introduce the polymer by over-moulding the metal with plastic melt [
2]. Such processes often generate large amounts of production waste due to the narrow tolerances during quality control, and the resulting waste streams usually have a high material value due to the presence of both expensive metals and high-performance plastics characterized by high temperature and chemical resistance. A non-destructive process for the recovery of both components would therefore be highly beneficial.
Established methods for the recycling of metal-plastic composites are usually based on waste comminution followed by separation into different fractions [
3]. The utilization of waste from electrical and electronic equipment (WEEE) is an important area of research, motivated by the recovery value of precious metals from electronic components such as cables and printed circuit boards (PCBs) [
4,
5]. However, the recovery of the plastic matrix is also increasingly important [
6]. In conventional composite decomposition processes for cable scrap, composite waste comprising copper and polyvinyl chloride (PVC) is crushed in a shredder and then ground using bar, ball, roller, or hammer mills. Density separation methods, windings, and magnetic or electrostatic separators can then be used to separate the ground material, with the aim of minimizing the cross-contamination of the end products and recovering each component as pure as possible [
7,
8,
9,
10,
11,
12]. The best results for the separation of a metal-plastic composite (a parallel wire cable) were achieved by reducing the particle size to ~3 mm in a cage mill followed by air-jet screening, resulting in <1% cross-contamination [
7]. The separation of ground cable waste with a particle size <8 mm was carried out with the aid of an electrostatic precipitator, and 99.95% of the plastic and 98.8% of the copper fraction was recovered after optimizing the apparatus geometry, field strength, and humidity [
10].
Mechanical pulping has also been investigated for the recovery of PCBs. For example, grinding to a particle size of 0.5 mm in a ball mill followed by a complex separation system involving various dry and wet processes achieved the recovery of 95% of the metal with 66% purity [
13]. Furthermore, ground PCB material was concentrated from a hammer mill to 80% metal content using a circulating air classifier [
14]. Because the plastic matrix must be recovered in addition to the metal, many of the well-known thermal-chemical recycling processes are unsuitable. Chemical recycling has been investigated for the recycling of PCBs, for example, in the form of pyrolysis [
12,
15,
16,
17], de-polymerization in supercritical liquids [
18], or by gasification [
19].
In order to obtain pure metal and polymer fractions, solvent-based recovery processes could also be applied. Present techniques use solvents like toluene, n-hexane, methyl-ethyl-ketone, or xylene, for instance, which are highly flammable and dangerous to health [
20,
21,
22]. The process is applied for many different polymers (polypropylene, polyamides, polyolefin) and reveals adequate dissolution kinetics and polymer qualities; however, the recycling of metal-plastic composites and larger scale trials are not reported.
The CreaSolv
® Process (CreaSolv
® is a registered trademark of CreaCycle GmbH), developed and patented by the Fraunhofer Institute for Process Engineering and Packaging IVV, uses special formulations with a low risk for both users and the environment, ideally not to be classified according to Globally Harmonised System (GHS) criteria. These selective solvents-mixtures extract the target polymers from heterogeneous waste input streams, allowing the polymer solution to be separated from undissolved components or harmful and/or prohibited pollutants, such as plasticizers or halogenic flame retardants. At the end of the process, the solvent is recovered by drying in order to ensure closed-loop circulation and to keep the process economically viable. The recycled polymer (product) is solvent free, meets the properties of virgin material, and is suitable as a substitute material in production processes [
23,
24,
25].
Here, we modified the CreaSolv® Process to achieve the recycling of composite material comprising several copper panels over-moulded with a polymeric matrix of polybutylene terephthalate (PBT). Because the investigated waste streams are pure polymers (with the exception of the metal feeders), the cleaning and precipitation steps do not need to be integrated into this process. The metal parts are separated directly during the dissolution step by detaching the polymer matrix from the mechanically fixed conductor elements. The solvent recovered during the drying step can thus be recycled directly into the process. In particular, we investigated the influence of temperature on the kinetics of the dissolution and drying steps, as well as the influence of dissolution on the quality of the recovered recyclates. Due to the complexity of solvent-based recycling plants, the investment costs for the required equipment are fairly high in comparison to those for conventional mechanical recycling. For that reason, we identified the so-called debinding systems as state-of-the-art large-scale technologies for implementing our solvent-based PBT-copper recycling in a profitable way.
2. Materials and Methods
2.1. Samples
The composite material samples (
Figure 1) were hybrid components (190 × 75 × 30 mm, 74.5 g) comprising PBT GF30, a polyester matrix (polybutylene terephthalate) with 30% (
w/
w) glass fibre reinforcement. The trade name is BASF Ultradur
® B 4300 G6 (kindly provided by psm protech GmbH & Co. KG, Marktschellenberg). Flat copper elements (comprising 0.1% (
w/
w) of tin) had been partly fixed to the surface by contact points either made of plastic or over-moulded with the PBT polymer, and represented 48.3% (
w/
w) of the total weight.
The samples had been rejected from the production plant during manufacturing, but with few exceptions the reasons for rejection could not be determined by eye. This presumably reflected deviations from the working temperature of the injection mould, resulting in the incomplete filling of the cavity. More parts with bent copper contacts were also observed.
2.2. Solvents
As stated above, all six of the solvent mixtures tested were developed by CreaCycle GmbH, Grevenbroich. The selection of these so-called CreaSolv
® Formulations was based on the solubility parameters according to Hansen [
26]. Other factors included the commercial availability of the ingredients, the risk to user and environment, and the biological degradability [
27]. The exact chemical compositions of the formulations are proprietary and so they are named herein using the terms “CreaSolv
®1”, “CreaSolv
®2”, …, “CreaSolv
®6”. The mixtures 1–6 were composed of different types of solvents in order to examine their individual functionality and effectiveness.
Table 1 shows the properties of the applied CreaSolv
® Formulations.
Sample preparation for analytical issues was done using tetrahydrofuran and acetone, purchased from Merck KgaA, Darmstadt.
2.3. Laboratory-Scale Methods
The solvents were tested by placing ~1-cm polymer fragments in a 100-mL beaker and stirring with each solvent on a heating plate until the solvent became cloudy and the polymer showed signs of swelling. The solvents were tested at a minimum solution temperature (Td,min), causing a complete dissolution within a maximum time of 60 min, and at 5-K increments to determine the exact temperature-dependent dissolution time (td). Based on our experiences regarding polymer concentration and its influence on dissolution time, viscosity, and solvent recovery, the mass balance was calculated to determine a final dry matter of the solution at 7.5% (w/w). Solvent loss during this process was ignored.
Dissolution was also monitored after excluding the oxygen using a stirred three-neck round-bottom flask combined with a mushroom heater. Nitrogen was introduced into the flask via one opening and a return condenser at another opening was used to prevent liquid evaporation by stripping the solvent. The exhaust gas stream was passed via a dip tube into a water trap. A gastight thermocouple at the third opening was used to measure the product temperature.
The parameters of the thermal drying process were investigated using an oil-heated vacuum rotary evaporator. The dry matter value of the polymer solution was determined by continuous gravimetric measurements of the evaporator flask. Wherever necessary (e.g., sample preparation for analysis), small amounts of the polymer solution were dried by repeated solvent extraction using acetone.
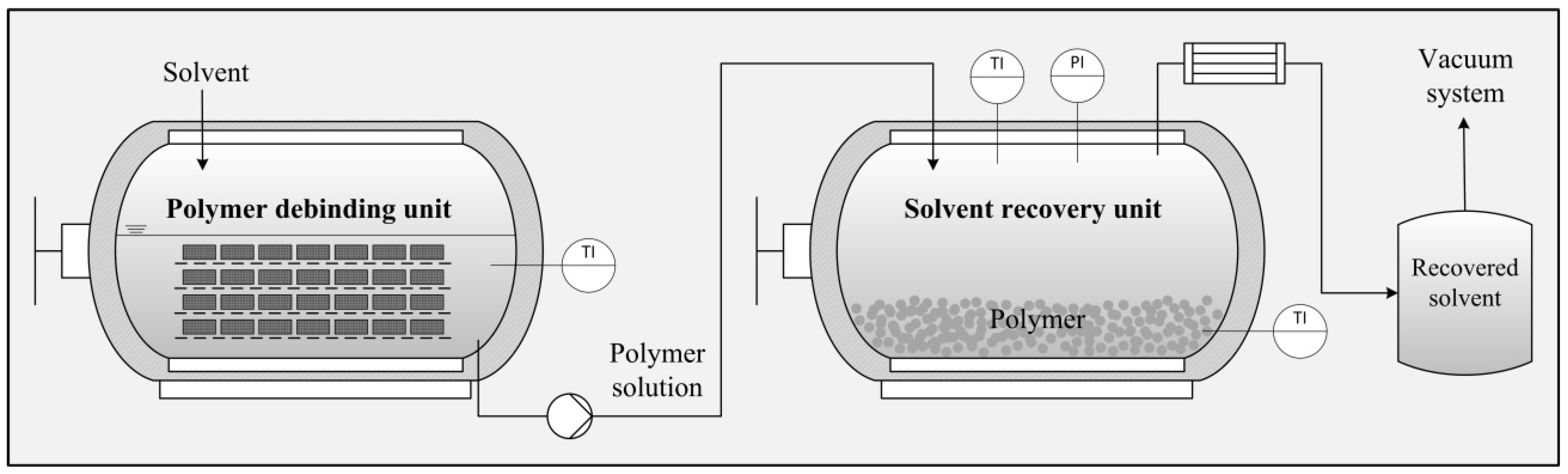
2.4. Technical-Scale Methods
Figure 2 shows the technical-scale process as a flow diagram. We used the debinding unit EDA-50 and the distillation unit LRA-50 (50-litre-scale in both cases) provided by Lömi GmbH, Großostheim. The entire test rig was operated in batch mode (i.e., the dissolution and drying processes were independent, time-separated partial steps). The debinding system (
Figure 2 left) was a horizontally oriented oil-heated stainless steel container that could be opened and closed at the front using a hatch provided with a screw cap. Within the vessel, the PBT-copper parts were applied to a removable, roll-mounted frame (500 × 220 mm) of perforated metal plates with spacers (40 mm). Therefore, the sample material did not have to undergo any previous cutting or sorting; we maintained its original dimensions, as described in
Section 2.1. The heated solvent was introduced and circulated using a centrifugal pump at 20 L/min and the remaining gas space was purged with nitrogen. After complete dissolution, the PBT-solution was conveyed to the distillation unit (
Figure 2 right) containing a water-cooled solvent trap and an integrated water ring pump for vacuum generation. Because of the pressure decay, the polymer solution was sucked into the distillation unit, resulting in the partial flash evaporation of the solvent. The remaining solvent then was evaporated during the residence time of the non-stirred polymer solution. The copper parts and vessel surface within the debinding unit were then washed with uncharged solvent. The resulting dilute polymer solution with a dry matter value of <1% (
w/
w) was available for the next dissolution batch. The residual solvent within the pre-dried PBT samples then was removed using a state-of-the-art melt degassing extruder at a throughput capacity of 1.5 kg/h.
2.5. Analytical Methods
The influence of the dissolution and drying processes on the molecular weight and polydispersity (PD) of the polymer were analysed by gel permeation chromatography (GPC) to measure qualitative changes in the PBT [
28,
29,
30].
A gravimetric infrared moisture analyser served to determine the residual solvent content or the dry matter value (DM), respectively, of polymer-solvent mixtures. For smaller residual solvent amounts of the polymer, gas chromatography (HS-GC) based on multiple head-space injection was applied [
31].
In order to estimate the glass fibre content of the PBT qualitatively, we used X-ray fluorescence analysis.
Melt flow rate (MFR) values of the solvent-free PBT samples were measured using a laboratory melt flow tester according to ISO 1133-1 [
32] at 250 °C and 2.16 kg.
Differential scanning calorimetry (DSC) according to ISO 11357-1 was used to measure the melting temperature of the polymer samples [
33,
34].
Typical mechanical tests of the polymer, including vicat softening temperature (ISO 306), tensile modulus, breaking stress, elongation at break (ISO 527-1/-2), charpy impact strength (ISO 179-1eU at 23 °C), and charpy notched impact strength (ISO 179-1eA at 23 °C), were carried out together with Sysplast GmbH & Co. KG, Nürnberg and Sitraplas GmbH, Bünde [
35,
36,
37].
In order to evaluate the quality of the recovered copper elements, we applied a metallographic analysis. Therefore, a micro-section of the metal was measured using an incident light microscope.
3. Results and Discussion
3.1. Dissolution Kinetics and Molecular Weight
In order to investigate the influence of temperature on the dissolution time, single fragments of the PBT samples were mixed with each of the six CreaSolv
® Formulations, but they only dissolved in CreaSolv
® 1 and 2. In both cases, the dissolution time was measured at four temperatures, as described above.
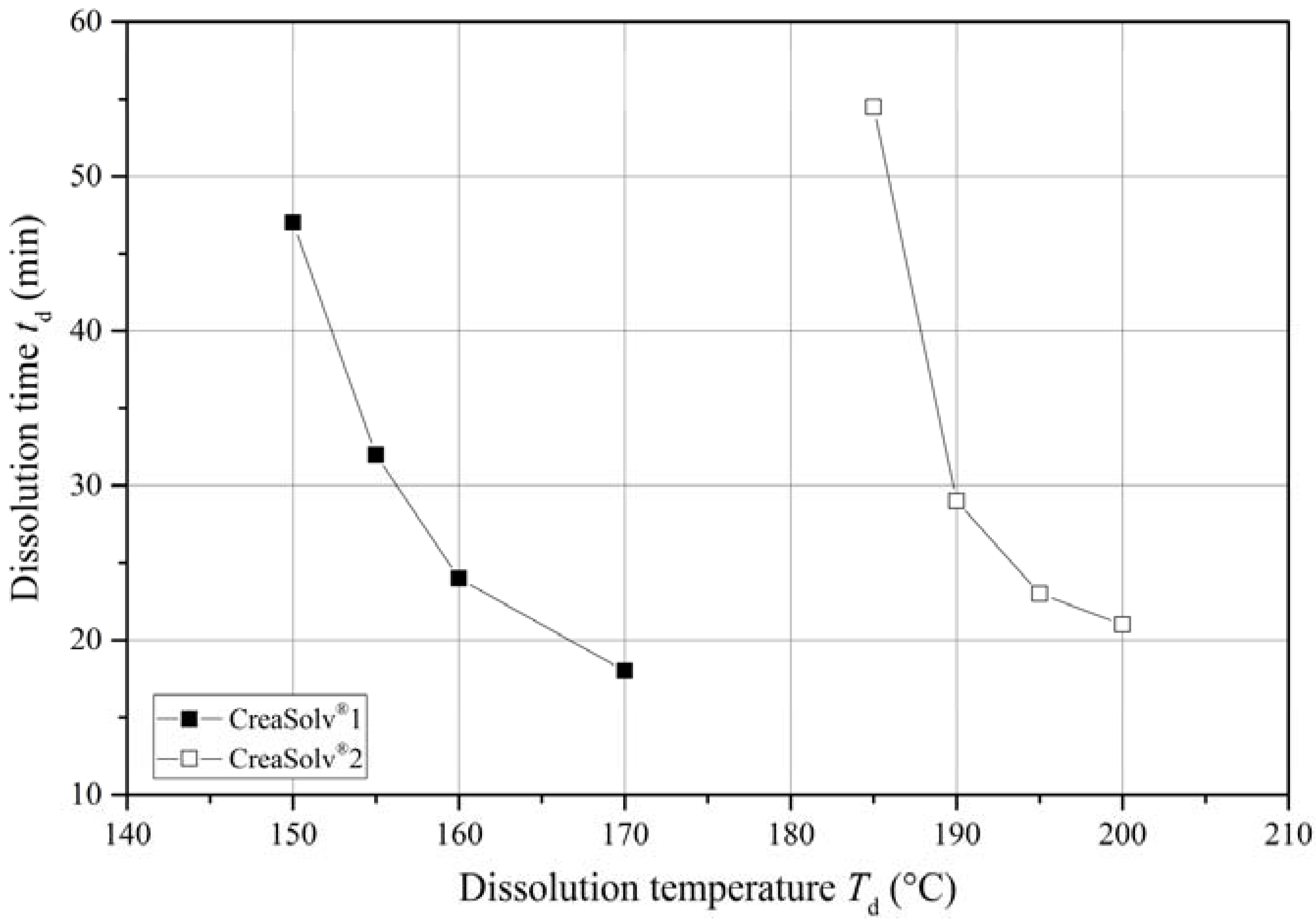
Figure 3 shows the dissolution times at a final polymer concentration of 7.5% (
w/
w) as a function of temperature. As expected, the dissolution time
td declined at higher temperatures because of an increased rate of diffusion/permeation and larger solubility parameter spheres [
26]. The greatest reduction occurred within a 5 K interval above the minimum dissolution temperature
Td,min. In both solvents,
td decreased by ~40% and fell slightly below the reported dissolution times of conventional solvents [
20]. However, further increasing the temperature had little additional impact. The flattening curve at more than 10 K above
Td,min indicates no significant changes in the
td. We therefore selected a temperature that is 5 K above
Td,min as the optimum range for potential technical implementation.
Furthermore, the Mw of PBT (Mw,PBT) was measured by gel permeation chromatography, as a function of the dissolution temperature for the two solvents, and compared to the input material. CreaSolv®1 substantially reduced the polymer chain length, whereas CreaSolv®2 had only a limited effect, even at a higher temperature. Using a dissolution temperature of 190°C, in the latter system, the Mw,PBT declined by only 11% from 4.7 × 104 Da (PD = 2.27) to 4.2 × 104 Da (PD = 2.10). Oxygen may be responsible for the decreasing Mw,PBT because solvent oxidation products can cleave the polymer by solvolysis. The dissolution process was therefore investigated in the absence of oxygen. For both solvents, the reduction in Mw was less severe under a nitrogen atmosphere. In the case of CreaSolv®2, the Mw,PBT decreased by only 4% to 4.5 × 104 Da (PD = 2.13). In the case of CreaSolv®1, at 155°C, the Mw,PBT decreased to 1.5 × 104 Da (PD = 2.33), which is still far below the initial chain length. CreaSolv®1 is therefore not recommended as a solvent for PBT under the selected parameters because the material qualities of the recycled polymer would be completely lost by a reduction in Mw,PBT of this magnitude. In contrast, CreaSolv®2 caused only a moderate decrease in the Mw,PBT, and the quality of the recycled polymer was therefore retained when CreaSolv®2 was used, despite the higher dissolution temperature.
In a further experimental series, the impact of the residence time
tr of CreaSolv
®2 on PBT was investigated more closely. The dissolution again took place in a nitrogen atmosphere.
Figure 4 shows the
Mw,PBT as a function of the temperature-time-load. For comparison, the values for dissolution at 190 and 200 °C but in oxygen atmosphere are also presented. We found that the rate of polymer degradation was initially slow, but increased with longer residence times. After the first 30 min, the
Mw,PBT had fallen by only 10%, but after a further 20 min it had fallen by 50%. Nevertheless, CreaSolv
®2 appears to be suitable for the successful recovery of PBT if oxygen is excluded, the temperature is maintained at 190 °C, and the residence time does not exceed 30 min.
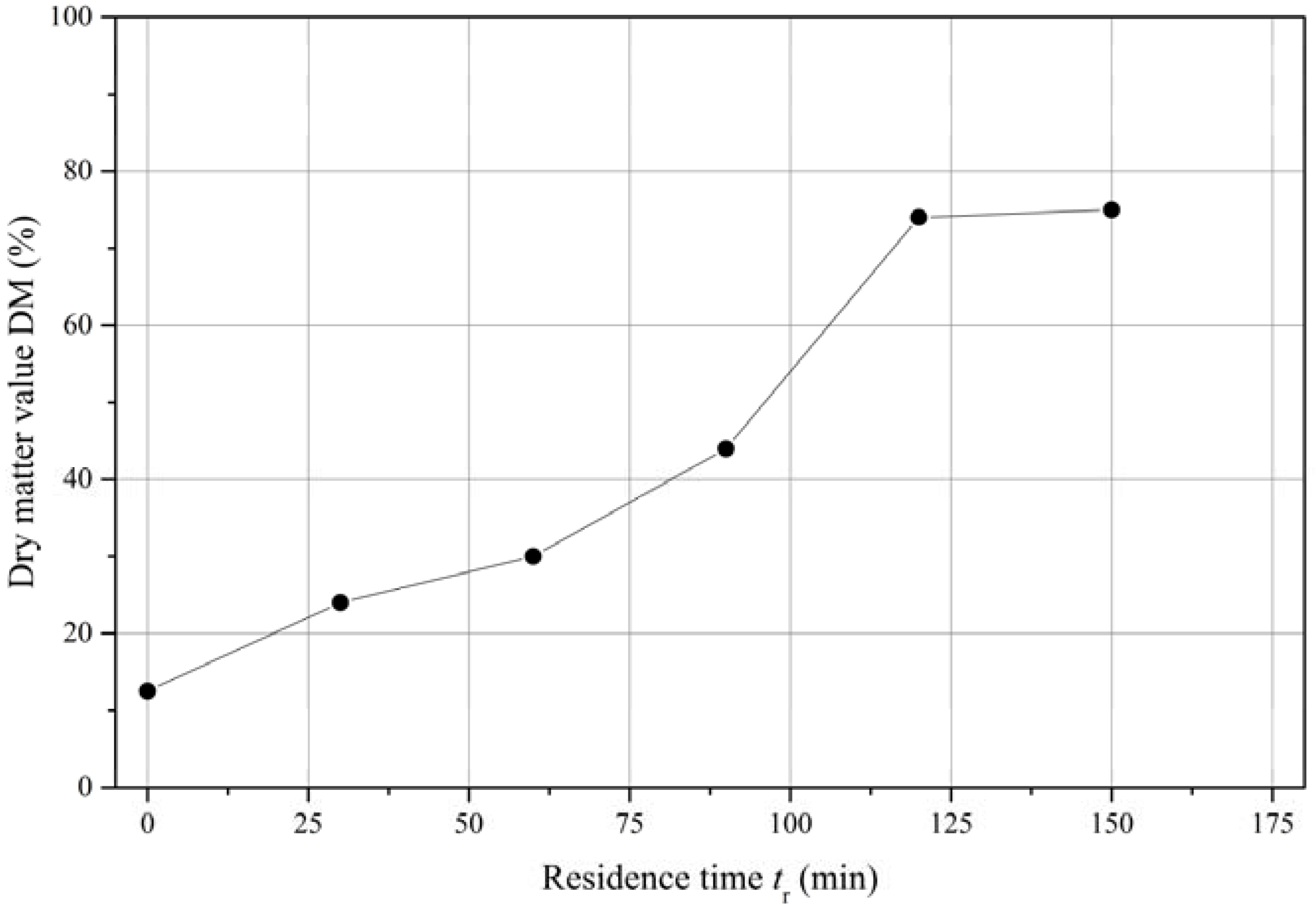
3.2. Drying Kinetics
Having shown that the quality of the PBT polymer suffers at elevated temperatures, the drying of the polymer solution (dissolved in a nitrogen atmosphere using CreaSolv
®2) was tested at 150 °C and 40 mbar using a rotary evaporator. This vacuum level approximately matches the typical lowest pressure levels that can be achieved using large-scale equipment.
Figure 5 shows the dry matter value (DM) as a function of the
tr.
The measurements were stopped when there was no further indication of condensate flow. The curve of the drying velocity shows a near constant slope until a critical DM value of ~75% (w/w) is achieved. Therefore, the process becomes inefficient at a residual solvent content of ~25% (w/w), and the drying method was changed to that described for solvent extraction using acetone. As already stated, due to the small amount of sample material at this scale, the final drying using a melt degassing extruder was not applicable.
3.3. Scale Up
We found that it was feasible to transfer the process from the laboratory scale to the small technical scale using state-of-the-art equipment from the German project partner Lömi GmbH, Großostheim. The parameters were chosen based on the results described above (i.e., CreaSolv®2, a dissolution temperature of 190 °C, a residence time of 30 min, and the exclusion of oxygen). Due to the absence of stirring (limited heat transfer) in both vessels, the dissolution time had to be adjusted to 60 min and the drying process was carried out using the same vacuum level but a higher temperature (170 °C) than the laboratory-scale process until reaching a DM of 75% (w/w). As described above, the pre-dried polymer powder then was fed into a melt degassing extruder, dried completely, and compounded into granules with an output mass flow of 1.5 kg/h. The recovered solvent was collected and recycled while maintaining its original properties.
3.4. Metal Quality
The copper parts remain inert in the polymer solution; first their surface properties were evaluated. The samples obtained after dissolution in CreaSolv
®1 and 2 both showed minor changes compared to the input material, in that the metal had a shiny surface. After recovery from the process, the surface of the metal material was dull. As indicated above, CreaSolv
®1 appears to create a highly reactive milieu which not only degraded the polymer but also attacked the metallic surface. However, the copper parts recovered from the process using CreaSolv
®2 in a nitrogen atmosphere showed no significant changes—visually or analytically (
Figure 6). The metallographic analysis of the metals by comparing their micro-sections with those of virgin material showed no changes in structure. CreaSolv
®2 therefore achieves a favourable outcome in regard to the recovery of the copper as well as the polymer.
3.5. Polymer Quality
The quality of the polymer was evaluated by comparing the post-process recycled material from the small-technical-scale trials with the input PBT for the injection moulding. In order to evaluate a suitable application of the recycled PBT, we extruded a polymer blend with 90% (
w/
w) virgin material and also compared its properties to those of the pure cases (
Figure 7). It was renounced to consider intermediates and/or products of the lab-scale trials, as they are unlikely to give a suitable indication for an industrial implementation of the process.
As shown in
Table 2, the recycled PBT deviated substantially from the virgin material in terms of MFR and mechanical properties. The GPC results suggest this is caused by a reduction in polymer chain length [
38]. We measured a drop of
Mw from 4.6 × 10
4 Da to 2.1 × 10
4 (PDI = 2.00) during dissolution and solvent recovery process, but no impact on the
Mw coming from residual degassing within the extruder. The reduction seems to be due to the higher residence time in the debinding unit and the increased temperature in the solvent recovery unit. In addition, it is likely that a small number of enclosures containing residual solvent remain within the granules and cannot be removed completely, thus reducing the melt viscosity. The head-space gas chromatography revealed a residual content of 996 ppm, which is below the critical solvent threshold but might be still high enough to have an impact. As another reason for the decrease in viscosity, the content of the inert glass fibres and their orientation might be responsible [
39]. Therefore, the virgin and recyclate samples were measured with X-ray fluorescence analysis. The silicon as well as the calcium content decreased by 55% and 38%, respectively, which indicates a strong depletion of the glass fibres within the PBT matrix. This corresponds to the observation of sedimented fibres that remained at the bottom of the debinding unit after polymer extraction. The blend sample showed just the slightest decline in quality, which indicates a high possibility for a suitable reuse—at least as substitution for start-up or purge material. In any case, a detailed inspection of the extruded strands of the samples revealed no visible differences between the original PBT and the blend material in terms of surface texture or colour.
4. Conclusions and Economic Evaluation
We have demonstrated that the CreaSolv
® Process is suitable for the recovery of both copper and PBT components from a polymer–metal composite material. Nevertheless, the implementation of an industrial-scale process remains an economic challenge because large quantities of waste would be required. Under certain conditions, this technology instead offers a good opportunity for the economical recycling of composites already at smaller scales because both the metal and (high-priced) polymer components can be recovered, thereby achieving higher redemption prices. The annual turnover of such a process therefore does not need to match breakeven volume associated with mass-produced materials such as PVC. Even if the recovered polymer is lower in quality than the input material, it can nevertheless be mixed with virgin material and/or (glass fibre) master batches in order to reinforce the PBT matrix. Based on our experiences, for such a pilot plant we would estimate investment costs of €750,000. This would include a debinding unit with a capacity of 1 t/d. In a worst-case scenario, we would reduce the redemption price of the recycled PBT from 1.70 (virgin PBT) to 1.10 €/kg and the recovered metallic components from 5.45 €/kg (virgin copper) to 4.30 €/kg. The processing energy for the (mechanical) pre-treatment of the copper parts which would be saved is not even included in the calculation. The material properties of the PBT blend (
Table 2) indicate an in-house usage of the recyclate as substitute material for virgin polymer as the most useful application. The operating costs of such a plant are estimated with a lump sum of 750 €/d for facilities, manpower, and maintenance in addition to the energy consumption by electrical power (0.15 €/kWh), which depends on the throughput of each case. Based on these assumptions, the annual input would only need to increase to 100 t to reach the breakeven point, even at moderate plant utilization (
Table 3). In a very conservative scenario (case 4; no use of polymer, only metal recovery), the input quantity would have to increase to 170 t to achieve a positive business case.
The global consumption of PBT in the year 2012 was 830,000 t, 32% of which was used for automotive applications including automotive electronics [
40]. Assuming that at least this 32% is applied in polymer-metal hybrids comprising 50% (
w/
w) of PBT, 531,000 t of composites left the production line in 2012. Assuming a reject rate of 10% of a company’s total production volume, at least 53,000 t/a of industrial waste would be available. Based on the results of the presented work, there is plenty of opportunity with this output for an efficient, environmentally sustainable, and profitable recycling system based on the CreaSolv
® Process, though the bundling of this highly dissipative waste generation might present a major challenge.