Abstract
We report the analysis of comb-like polymers by solid-state NMR. The polymers were previously evaluated as solid-polymer-electrolytes (SPE) for lithium-polymer-metal batteries that have suitable ionic conductivity at 60 °C. We propose to develop a correlation between 13C solid-state NMR measurements and phase segregation. 13C solid-state NMR is a perfect tool for differentiating polymer phases with fast or slow motions. 7Li was used to monitor the motion of lithium ions in the polymer, and activation energies were calculated.
1. Introduction
Global warming is a major challenge of the twenty-first century, and finding solutions is critical for the future of humanity. The widespread implementation of a fully electric transportation network must be a part of the solution. Electrical vehicles (EV) using lithium-ion batteries remain one of the most promising avenues in the short term. At present, Li-ion batteries containing liquid electrolytes are the most prevalent cells used as the electrical propulsion power in EV; however, their performance and safety are not sufficient to fully compete with gasoline-powered motors [1,2]. Considering the safety issue, liquid electrolytes are unavoidably problematic due to their flammability and the toxic fumes released during overcharging/abuse or over heating [3]. Solid electrolytes are currently getting much attention from the scientific community [4]. Based on the discovery of ionic conduction in poly(ethylene oxide) (PEO) by Wright in 1975 [5] and its first application to batteries by Armand and co-workers in 1979 [6], solid polymer electrolytes (SPE) have been investigated by many groups as a safety-improving solution. An acceptable SPE needs excellent lithium conductivity in a wide temperature range [7] and good mechanical strength [8] in order to limit dendrite growth from the lithium metal anode, which is critical for the production of efficient Li-polymer batteries. More importantly, simple and economically viable processing as a thin film is a major advantage. In 2007, Balsara and co-workers introduced a new SPE, poly(ethylene oxide-b-polystyrene). Poly(styrene) acts as a reinforcement block in SPE [8] to prevent the formation of dendrites by presenting an impenetrable wall [9,10].
Recently, Bouchet et al. developed single-ion triblock copolymers that demonstrated excellent ionic and mechanical performance at 80 °C [11]. Using elegant polymer architectures with rigid and soft blocks to create phase separation is now a trend in the development of a new SPE [12,13,14,15]. The development of SPEs involves complex architectures that influence the interaction between the soft and the hard blocks. The investigation of Li+ ion mobility by different techniques appears to be a fascinating field of research. Several articles were published on the investigation of SPE by solid-state NMR [16,17,18,19], especially scrutinizing the mobility of lithium coordinated with the polymer chains; however, the examination of the mobility of the organic part has not been extensively studied. In this work, we describe the use of a combination of 13C and 7Li high-resolution solid-state NMR results to establish a complete portrait of high-performance comb-like copolymers.
2. Results and Discussion
2.1. Description of Polymers Investigated by Solid-State NMR
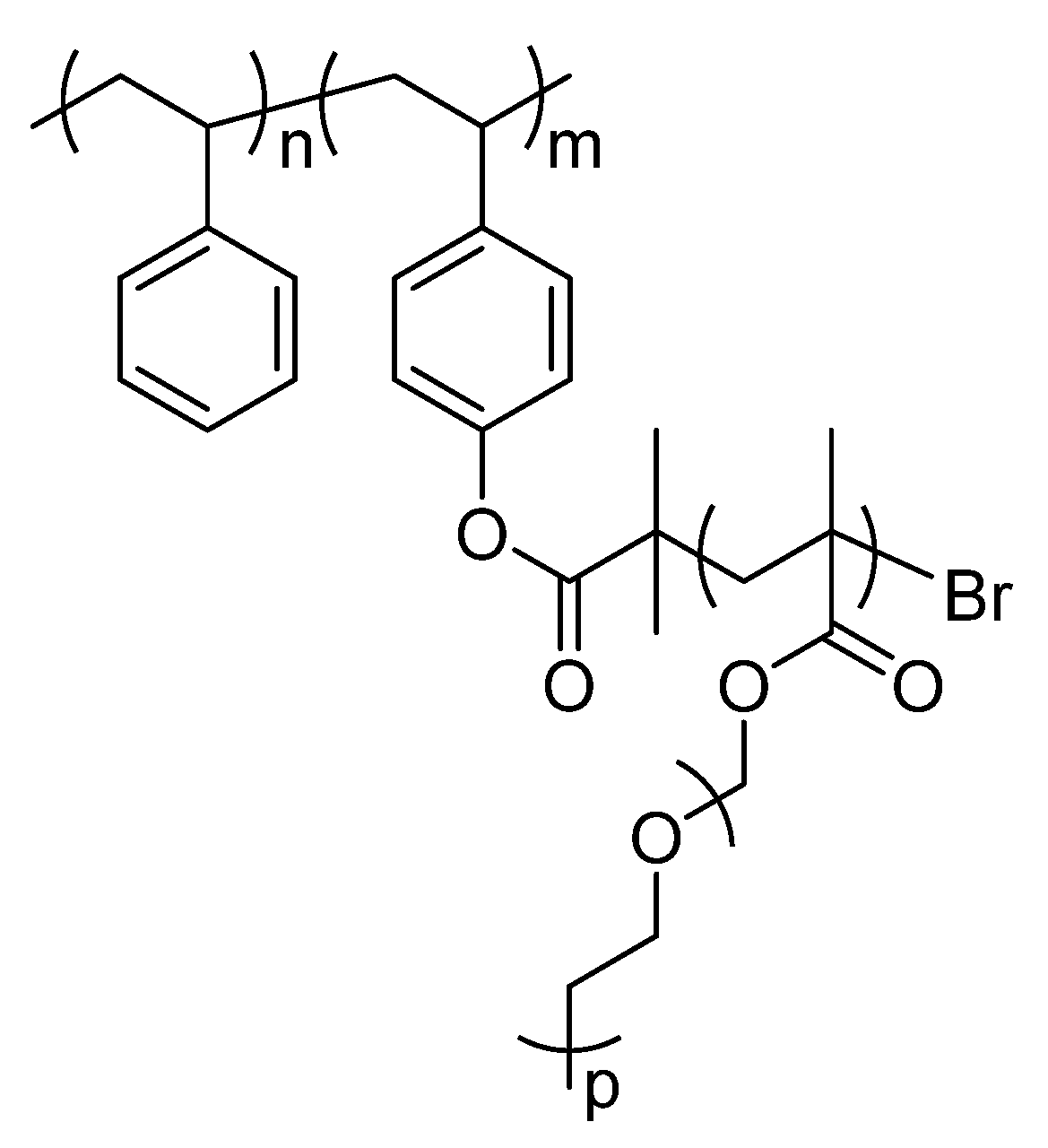
Comb-like copolymers with soft and hard blocks, i.e., with low and high Tgs, display phase segregation when prepared as films [12]. This behaviour is key to promote good ionic conductivity and to prevent dendrite growth [20]. Comb-like copolymers are based on a poly(styrene) (PS) backbone obtained by anionic polymerization. PS backbone is used as a reinforcement block for preventing dendrite growth during cell operation, phenyl groups are very rigid structures. The backbone is a hard block with a reported Young Modulus of 3 GPa [8]. Grafting poly(ethylene glycol) methyl ether methacrylate (PEGMA) with a Mn = 500 was done by atom-transfer radical polymerization (ATRP). Poly(ethylene glycol) is known to be a very good polymer for Li+ transportation, moreover short chains have a highest conductivity due by the lack of crystallinity. PEGMA is polymerized by soft radical polymerization while ethylene oxide is polymerized by a less safe process only. This monomer enables the formation of highly branched structures as a polymer graft. Both techniques allow narrow poly(dispersity), which facilitates the structure-properties relationship, and therefore better control of the desired properties. Scheme 1 shows the structure of the polymers investigated in this article.
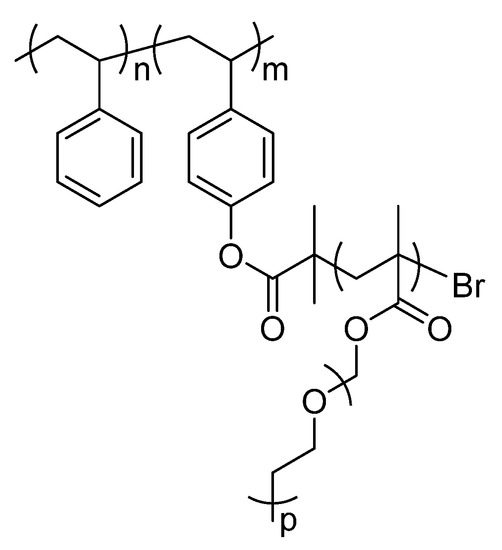
Scheme 1.
Structure of the polymers investigated by Solid-state NMR.
In order to make a comprehensive comparison between the structures, different ratios of soft/hard blocks were selected. Table 1 reports the different characteristics of the polymers that are reported in ref. [12]. The PEGMA/PS ratio is important because it has been directly related to the electrochemical performance of the cells [12]. Polymer 3 was not used as SPE because the high molecular weight of the PEGMA block made it impossible to dissolve in a reasonable amount of solvent. All the polymers were doped with bis(trifluoromethane)sulfonimide lithium salt (LiTFISI) to promote ionic conductivity. Consequently, the study of this polymer by solid-state NMR is an interesting tool for elucidating the lithium motion. The rigidity of the structure is assessed by solid-state 13C NMR, and an improved understanding of the polymer micro-structure is obtained, as reported in the case of cross-linked polymers [21]. The purpose of this study is to develop a relationship between the lithium mobility and the electrochemical results.

Table 1.
Polymer Characteristics.a
2.2. Characterization of Copolymers by 13C Solid-State NMR
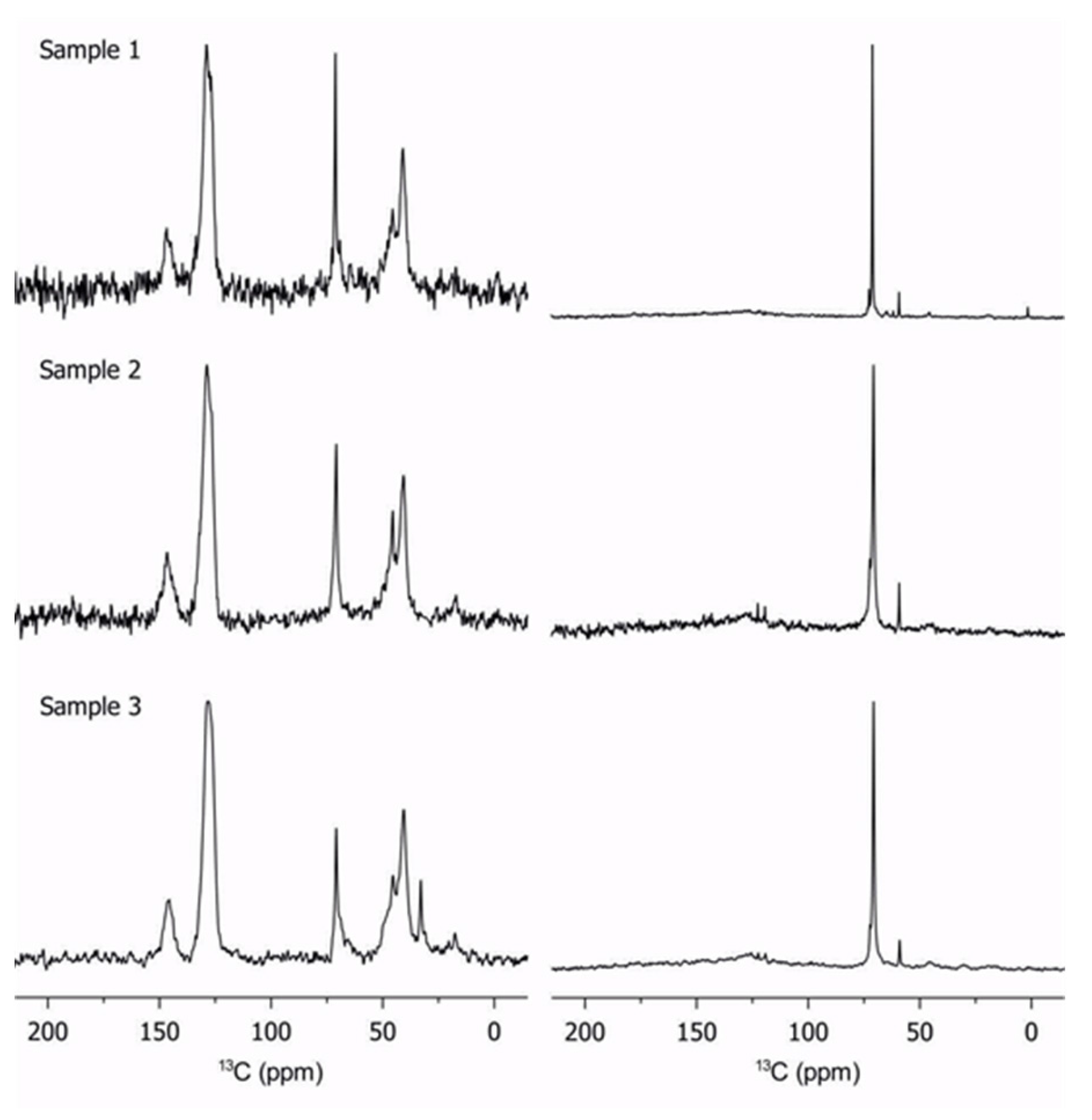
Solid-state 13C NMR is invaluable for the characterization of polymers [22,23,24,25]. Qualitative information is obtained for the dynamics in heterogeneous systems. The signal is obtained using different polarization schemes, which preferentially excite rigid or dynamic molecular segments. In this work, cross-polarization is used to excite the rigid regions, while a simple 90° pulse excites the rigid and mobile segments. Experimental details are reported in Appendix A. Figure 1 shows the results for three different polymers with differing PEGMA to PS ratios.
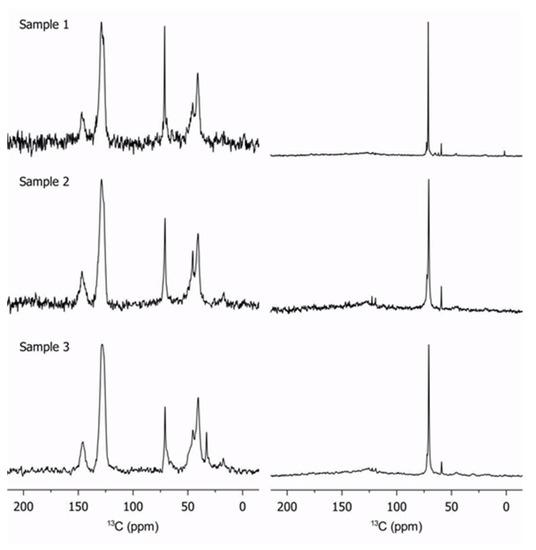
Figure 1.
Cross-polarization solid-state 13C NMR (left column) and direct pulse solid-state 13C NMR excitation (right column) of samples 1 to 3 at 298 K. All spectra are normalized by maximum peak intensity; the change in noise level thus reflects the amount of rigid segments in the polymer.
The cross-polarization spectra (left column in Figure 1) are dominated by the PS main chain (peaks at 150 and 125 ppm) while the direct-pulse with low-power decoupling spectra (right column in Figure 1) are dominated by the poly(ethylene glycol) chains at 70 ppm. A fraction of PEGMA between 25–75 ppm is observed on the cross-polarization spectra, which indicates that the PEGMA chains display considerable rigidity. More specifically, the PEGMA backbones (25–50 ppm) appear to be less mobile than the pendant groups (70 ppm), which have lower intensity. However, it should be noted that the pendant groups are still rigid enough to show up in the cross-polarization spectra. This partial rigidity of pendant groups possibly results from their coordination with lithium salts. Furthermore, the 13C NMR results show that the phase segregation reported by Daigle et al. [12] is not complete and that a model consisting of two perfectly separated blocks has to be refined. As the PS fraction is reduced, the efficiency of the cross-polarization decreases. Finally, an observation of the relative intensities between cross-polarization spectra gives an immediate diagnostic of the relative rigidity of a given copolymer series. For example, based on the relative spectral intensities shown in Figure 1, sample 3 appears to have a stronger rigid phase component compared to samples 1 and 2. We believe that some fluid parts of sample 3 have to be considered as rigid because the lithium ions strongly coordinated the PEGMA chains for forming “ionomer” as demonstrated in previous publication [12]; those parts are “frozen” and the ratio reported in Table 1 is based on GPC analysis.
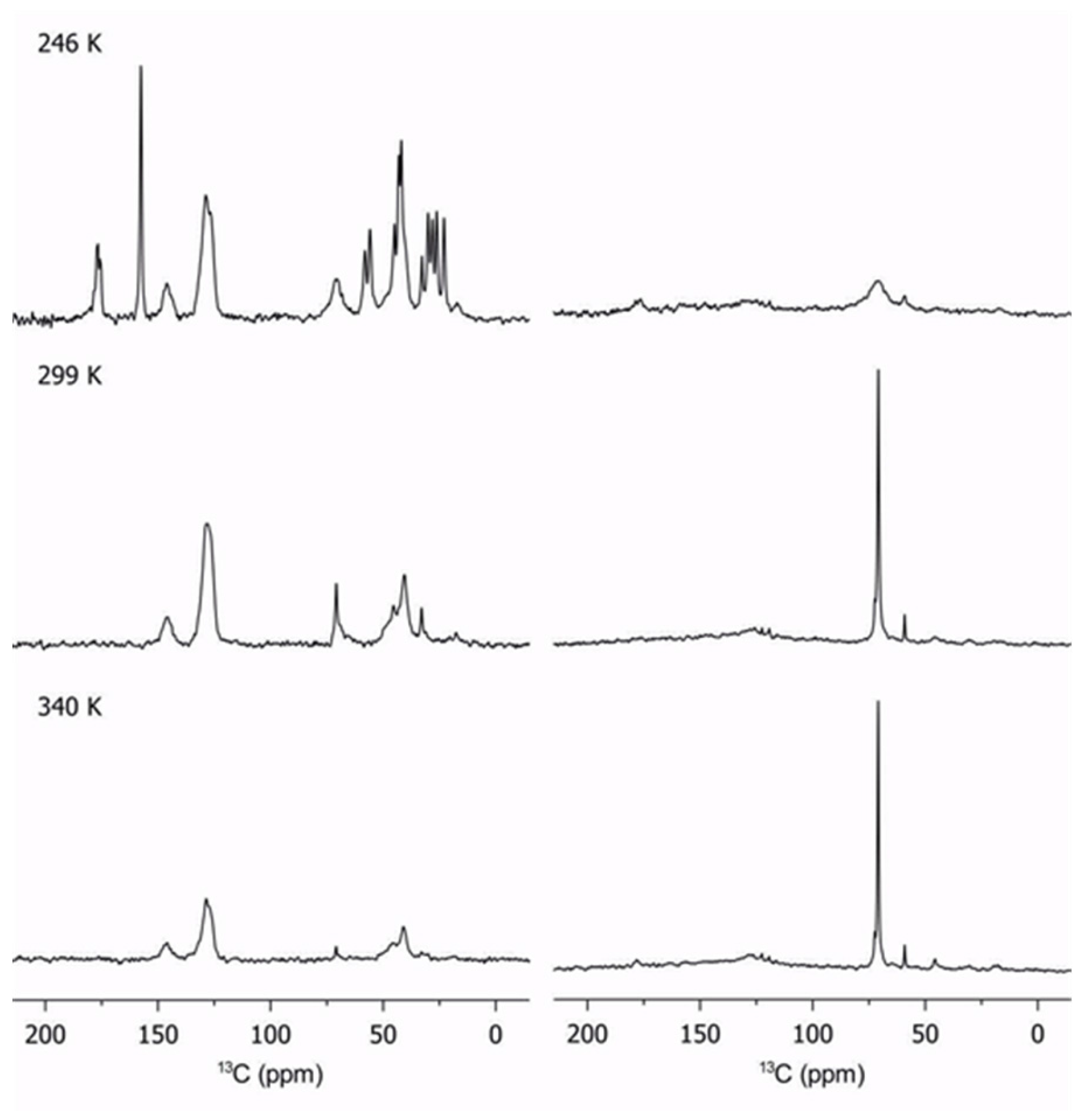
The same methodology is used to determine changes in internal dynamics with temperature. Figure 2 shows the evolution of 13C spectra for sample 3 with temperature.
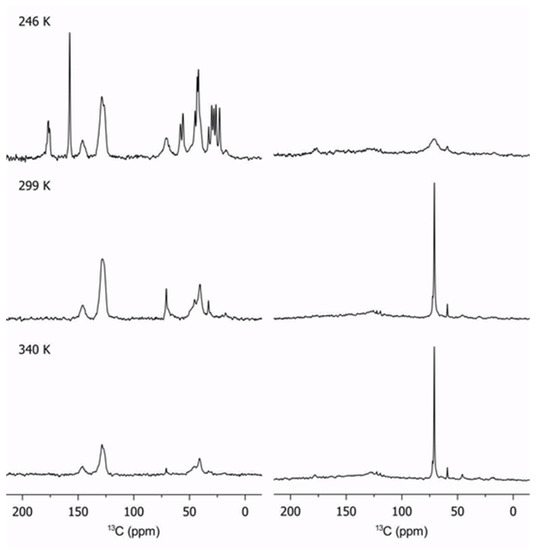
Figure 2.
Evolution of cross-polarization (left column) and direct-pulse excitation (right column) of sample 3 with temperature. Note the decrease in intensity of the rigid segments as temperature is increased (left column).
The effect of temperature of the PEGMA phase is unambiguous: as temperature decreased, the mobility of the PEGMA chains is reduced as expected. The very low signal on the pendant chains on the DP spectrum, and the appearance of the carbonyl peak at 175 ppm on the CP spectrum, reflects the strong stiffening of the chains at 246 K. This effect is directly correlated to the poor ionic conductivity at low temperatures, especially near the glass transition point (225 K). The glass transition of poly(styrene) is ca. 373 K, and the signal at 30 ppm is characteristic of the poly(styrene) backbone, indicating that PS is already rigid at the highest temperatures studied in this work. The poly(styrene) phase was not observed in the DP spectra because the temperatures were too far from the glass transition. No major changes in the PEGMA segments are observed between 299 K and 340 K since they are not affected by the melting of chains.
2.3. Lithium Diffusion in the Membranes by 7Li Solid-State NMR
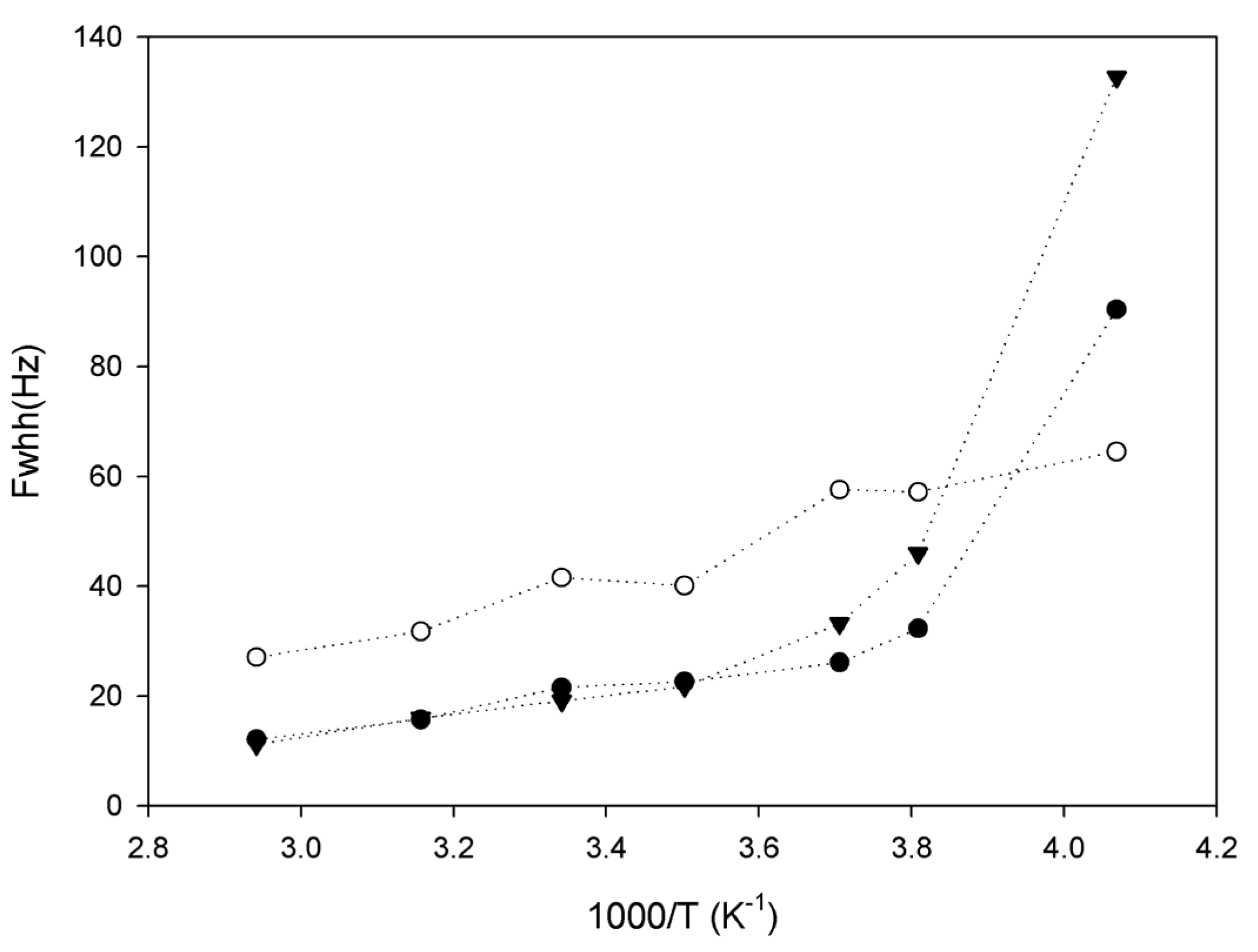
The 13C spectra provide information on the polymer rigidity, and 7Li NMR is useful to monitor the mobility of Li+ ions, which are qualitatively correlated to the conductivity of the material. Due to the very high mobility of lithium in our samples, 7Li spectra consist of a single sharp peak with full-widths at half height as low as 12 Hz, which reach a maximum at low temperatures of 120 Hz (Figure 3). This is almost two orders of magnitude lower than the linewidths reported for polyurethane-poly(dimethylsiloxane) copolymers [17]. The highest ionic conductivity reported in Table 1 is 2.54 × 10−4 Scm−1 at 60 °C, which is higher (about 3 times) than those reported earlier [17], so we can evaluate qualitatively the ionic conductivity of a polymer by this method. Interestingly, 1H decoupling appears to have no effect on the 7Li linewidth in our samples: thus, the mobility of lithium ions is sufficient to completely eliminate 1H–7Li dipolar couplings.
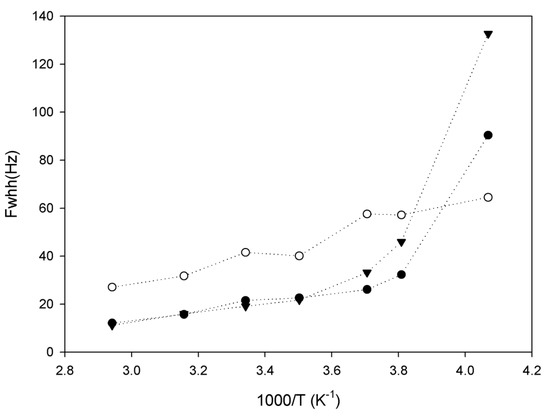
Figure 3.
Evolution of 7Li linewidth as a function of inverse temperature for sample 1 (black triangles), sample 2 (open circles) and sample 3 (black circles).
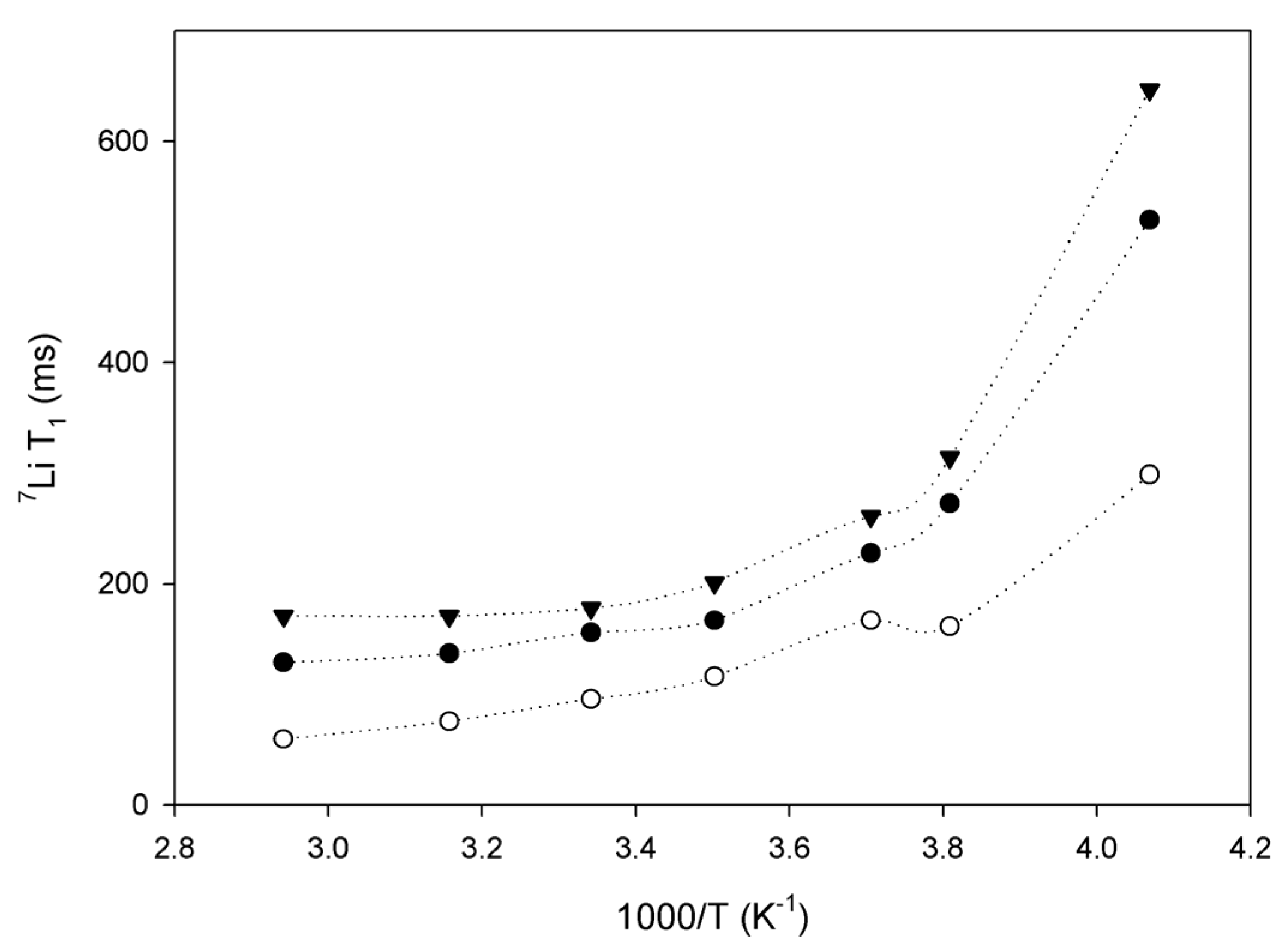
While 7Li linewidths are influenced by motions with correlation times shorter than μs, faster motions with correlation times in the nanoseconds will contribute to longitudinal relaxation (T1) of the NMR signal. Thus, T1 characterizes fast motion that facilitates lithium diffusion.
Sample 2 in Figure 4 has the fastest lithium motion (shorter T1 relaxation times), which is not in agreement with the ionic conductivity reported in Table 1. Also, it appears that temperature had less influence on the lithium mobility in this polymer. Mobility in samples 1 and 3 dropped around 263 K, which is related to the crystallization of poly(ethylene glycol) pendant chains in graft copolymers. Figure 2 shows that low temperatures have a great effect on solidification of the polymer chains (PEGMA). The close correlation between the mobility of lithium ions and the motions of PEGMA pendant chains suggests a strong association between the lithium ions and PEGMA groups.
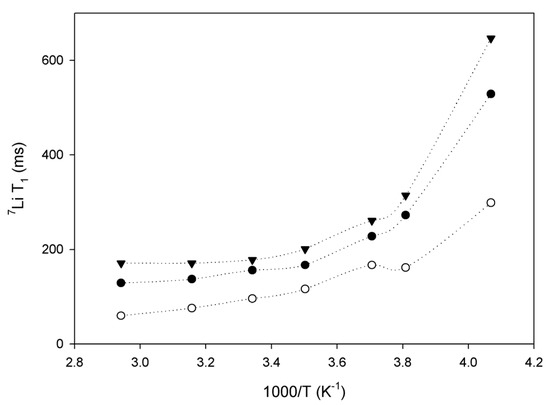
Figure 4.
7Li longitudinal relaxation (T1) as a function of inverse temperature for sample 1 (black triangles), sample 2 (open circles) and sample 3 (black circles).
Sample 3 shows the lowest lithium mobility (see Figure 4), despite the highest ratio of PEGMA. We explain this behavior as resulting from the strangling of the polymeric chains due to the high molecular weight (1,000,000 g mol−1), which hinders the motion of lithium [12].
Lin et al. [17] calculated the activation energy (Ea) of lithium diffusion from the slope of the curve at low temperatures by the Arrhenius relationship. This information is relevant for sample 3 because it is impossible to obtain using AC impedance measurements because this polymer cannot be prepared as a thin film. Table 2 reports the results using the two methods. A qualitative correlation is observed with an approximate difference of a factor of 2 between the two methods, the same factor was also reported in reference [17]. The relaxation time depends on the size of the mobile segments of the polymer electrolyte; one should note that there would be a range of sizes of the mobile polymeric segments and the relaxation time would be some sort of average value. Thus it would be a rough and approximate measure. The activation energy and conductivity as determined by the impedance measurements would be more significant in comparing the conductivity mechanisms of different polymers. Moreover, AC impedance allowed the measurements of long-scale motion while the solid-state NMR 7Li measurements are related with local motion of Li+, so that can contribute also for the difference of values measured for conductivity and by consequence Ea. Nevertheless, the NMR measurements indicate that the activation energy of polymers 2 and 3 is similar. It also important to note that the result obtained for sample 1 by solid-state NMR is within the same magnitude of the normal thermal fluctuation (4 kT = 9.6 kJ), thus the value is probably none applicable in this case.

Table 2.
Energies of activation recorded by AC Impedance and 7Li longitudinal relaxation (T1).
3. Conclusions
The development of well-define micro-structured polymers offers a new trend in solid polymer electrolytes. Solid-state NMR is a perfect tool for identifying the mobile and rigid parts in polymers. A combined 13C and 7Li NMR approach to correlate polymers and lithium motilities was investigated and successfully applied to comb-like copolymers. Our results strongly suggest that there is an interaction between the PEGMA pendant groups and lithium ions. Solid-state NMR is very useful when materials cannot be prepared in a form amenable to classical techniques such as AC impedance because the activation energy can be calculated in the solid state. This NMR technique is useful to evaluate the viability of polymers as SPEs without preparing films, and thus can be used for preliminary testing.
Acknowledgments
This work was supported by Hydro-Québec.
Author Contributions
Jean-Christophe Daigle and Alexandre A. Arnold conceived and designed the experiments; Alexandre A. Arnold performed the experiments; all the authors analysed the data and wrote the paper.
Conflicts of Interest
The authors declare no conflict of interest.
Appendix A
The spectra were recorded on a Bruker Avance III HD operating at frequencies of 400.03, 100.60 and 155.47 MHz for 1H, 13C and 7Li, respectively, using a triple resonance 1.9 mm MAS probe in double resonance mode. The samples had a mass of 15 mg and the s spinning frequency was 20 kHz. 13C spectra were obtained using 1.5 ms cross-polarization ramped from 70 to 100% of the maximum amplitude or a 90° pulse. In both cases, the 13C radio-frequency field was ca. 80 kHz. The cross-polarization spectra of rigid segments were obtained under high-power 1H TPPM decoupling with a 100 kHz radio-frequency field. The direct-pulse excitation spectra of the mobile segments were obtained under low-power GARP decoupling at a 1H radio-frequency field of 3.5 kHz. In both cases, the acquisition times were 25 ms and the recycle delays were 5 s. 7Li spectra were produced using 2.5 μs long 90° pulses with a 200 ms acquisition time and a 5 s recycle delay. The 7Li longitudinal relaxation times were measured using the inversion recovery pulse sequence incrementing the relaxation delays in 12 steps between 1 ms and 5 s. All spectra were processed and fitted with Topspin3.5.2 and the dynamics center module. Note that sample temperature was calibrated taking into account additional heating due to rotor friction or radio-frequency fields.
References
- Murata, K.; Izuchi, S.; Yoshihisa, Y. An overview of the research and development of solid polymer electrolyte batteries. Electrochim. Acta 2000, 45, 1501–1508. [Google Scholar] [CrossRef]
- Tarascon, J.M.; Armand, M. Issues and challenges facing rechargeable lithium batteries. Nature 2001, 414, 359. [Google Scholar] [CrossRef] [PubMed]
- Hammami, A.; Raymond, N.; Armand, M. Runaway risk of forming toxic compounds. Nature 2003, 424, 635. [Google Scholar] [CrossRef] [PubMed]
- Manthiram, A.; Yu, X.; Wang, S. Lithium battery chemistries enabled by solid-state electrolytes. Nat. Rev. Mater. 2017, 2, 16103. [Google Scholar] [CrossRef]
- Wright, P.V. Electrical conductivity in ionic complexes of poly(ethylene oxide). Br. Polym. J. 1975, 7, 319–327. [Google Scholar] [CrossRef]
- Armand, M.B.; Chabagno, J.M.; Duclot, M.J. Polyethers as Solid Electrolytes; Elsevier: Amsterdam, The Netherlands, 1979; pp. 131–136. [Google Scholar]
- Marzantowicz, M.; Dygas, J.R.; Krok, F.; Florjańczyk, Z.; Zygadło-Monikowska, E. Influence of crystalline complexes on electrical properties of peo: Litfsi electrolyte. Electrochim. Acta 2007, 53, 1518–1526. [Google Scholar] [CrossRef]
- Singh, M.; Odusanya, O.; Wilmes, G.M.; Eitouni, H.B.; Gomez, E.D.; Patel, A.J.; Chen, V.L.; Park, M.J.; Fragouli, P.; Iatrou, H.; et al. Effect of molecular weight on the mechanical and electrical properties of block copolymer electrolytes. Macromolecules 2007, 40, 4578–4585. [Google Scholar] [CrossRef]
- Monroe, C.; Newman, J. Dendrite growth in lithium/polymer systems: A propagation model for liquid electrolytes under galvanostatic conditions. J. Electrochem. Soc. 2003, 150, A1377–A1384. [Google Scholar] [CrossRef]
- Monroe, C.; Newman, J. The impact of elastic deformation on deposition kinetics at lithium/polymer interfaces. J. Electrochem. Soc. 2005, 152, A396–A404. [Google Scholar] [CrossRef]
- Bouchet, R.; Maria, S.; Meziane, R.; Aboulaich, A.; Lienafa, L.; Bonnet, J.-P.; Phan, T.N.T.; Bertin, D.; Gigmes, D.; Devaux, D.; et al. Single-ion bab triblock copolymers as highly efficient electrolytes for lithium-metal batteries. Nat. Mater. 2013, 12, 452–457. [Google Scholar] [CrossRef] [PubMed]
- Daigle, J.-C.; Vijh, A.; Hovington, P.; Gagnon, C.; Hamel-Pâquet, J.; Verreault, S.; Turcotte, N.; Clément, D.; Guerfi, A.; Zaghib, K. Lithium battery with solid polymer electrolyte based on comb-like copolymers. J. Power Sources 2015, 279, 372–383. [Google Scholar] [CrossRef]
- Villaluenga, I.; Inceoglu, S.; Jiang, X.; Chen, X.C.; Chintapalli, M.; Wang, D.R.; Devaux, D.; Balsara, N.P. Nanostructured single-ion-conducting hybrid electrolytes based on salty nanoparticles and block copolymers. Macromolecules 2017, 50, 1998–2005. [Google Scholar] [CrossRef]
- Sun, J.; Stone, G.M.; Balsara, N.P.; Zuckermann, R.N. Structure–conductivity relationship for peptoid-based peo–mimetic polymer electrolytes. Macromolecules 2012, 45, 5151–5156. [Google Scholar] [CrossRef]
- Devaux, D.; Glé, D.; Phan, T.N.T.; Gigmes, D.; Giroud, E.; Deschamps, M.; Denoyel, R.; Bouchet, R. Optimization of block copolymer electrolytes for lithium metal batteries. Chem. Mater. 2015, 27, 4682–4692. [Google Scholar] [CrossRef]
- Gorecki, W.; Jeannin, M.; Belorizky, E.; Roux, C.; Armand, M. Physical properties of solid polymer electrolyte PEO(LiTFSI) complexes. J. Phys. Condens. Matter 1995, 7, 6823. [Google Scholar] [CrossRef]
- Lin, C.-L.; Kao, H.-M.; Wu, R.-R.; Kuo, P.-L. Multinuclear solid-state nmr, dsc, and conductivity studies of solid polymer electrolytes based on polyurethane/poly(dimethylsiloxane) segmented copolymers. Macromolecules 2002, 35, 3083–3096. [Google Scholar] [CrossRef]
- Hayamizu, K.; Akiba, E.; Bando, T.; Aihara, Y.; Price, W.S. Nmr studies on poly(ethylene oxide)-based polymer electrolytes with different cross-linking doped with LIN(SO2CF3)2. Restricted diffusion of the polymer and lithium ion and time-dependent diffusion of the anion. Macromolecules 2003, 36, 2785–2792. [Google Scholar] [CrossRef]
- Yang, L.-Y.; Wei, D.-X.; Xu, M.; Yao, Y.-F.; Chen, Q. Transferring lithium ions in nanochannels: A PEO/Li+ solid polymer electrolyte design. Angew. Chem. Int. Ed. 2014, 53, 3631–3635. [Google Scholar] [CrossRef] [PubMed]
- Harry, K.J.; Hallinan, D.T.; Parkinson, D.Y.; MacDowell, A.A.; Balsara, N.P. Detection of subsurface structures underneath dendrites formed on cycled lithium metal electrodes. Nat. Mater. 2014, 13, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Daigle, J.-C.; Asakawa, Y.; Vijh, A.; Hovington, P.; Armand, M.; Zaghib, K. Exceptionally stable polymer electrolyte for a lithium battery based on cross-linking by a residue-free process. J. Power Sources 2016, 332, 213–221. [Google Scholar] [CrossRef]
- Spiess, H.W. Molecular motion, phase separation and internal surfaces in rubber-elastic polymers. Angew. Makromol. Chem. 1992, 202–203, 331–342. [Google Scholar] [CrossRef]
- Spiess, H.W.; Schmidt-Rohr, K. Multidimensional solid-state NMR studies of chain motions in polymers. Polym. Prepr. (Am. Chem. Soc. Div. Polym. Chem.) 1992, 33, 68–69. [Google Scholar]
- Bluemich, B.; Bluemich, P.; Guenther, E.; Jansen, J.; Schauss, G.; Spiess, H.W. NMR Imaging of Polymers: Methods and Applications; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 1992. [Google Scholar]
- Bluemich, B.; Bluemler, P.; Guenther, E.; Spiess, H.W. Methods and applications of NMR imaging in polymer research. Polym. Prepr. (Am. Chem. Soc. Div. Polym. Chem.) 1992, 33, 759–760. [Google Scholar]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).