Can Moringa Leaf Spray Treatment Increase the Nutraceutical Properties of Radish Baby Leaf?
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plants Materials
2.2. Moringa oleifera Leaf Extract
2.3. Measurement of Growth Parameters
2.4. Chlorophyll a Fluorescence
2.5. Quantification of Chlorophyll and Carotenoids
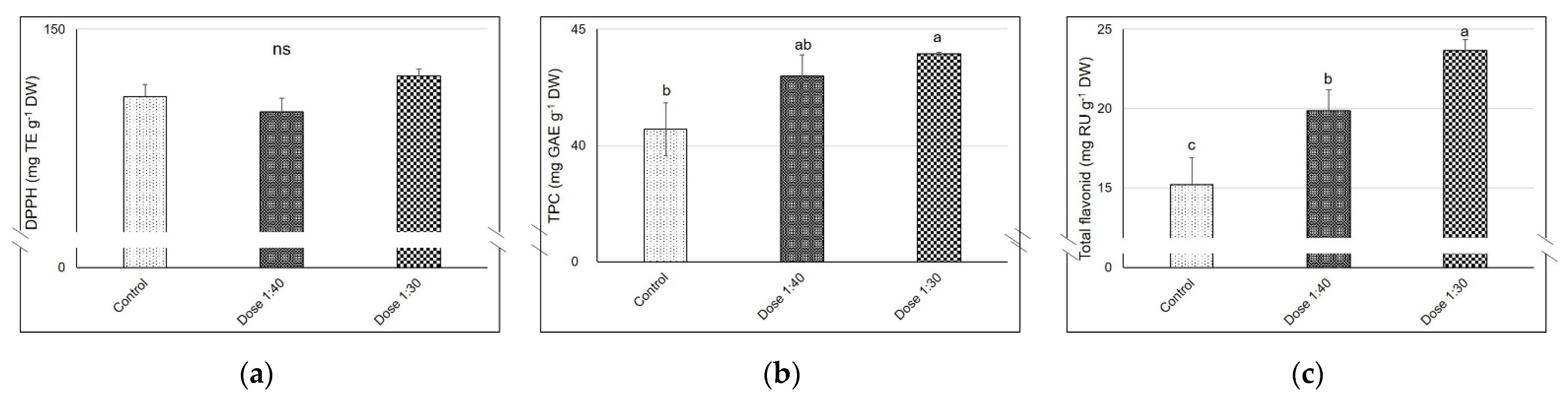
2.6. 2.2-Diphenyl-1-Picrylhydrazyl (DPPH) Radical-Scavenging Activity and Total Phenolic and Flavonoid Contents
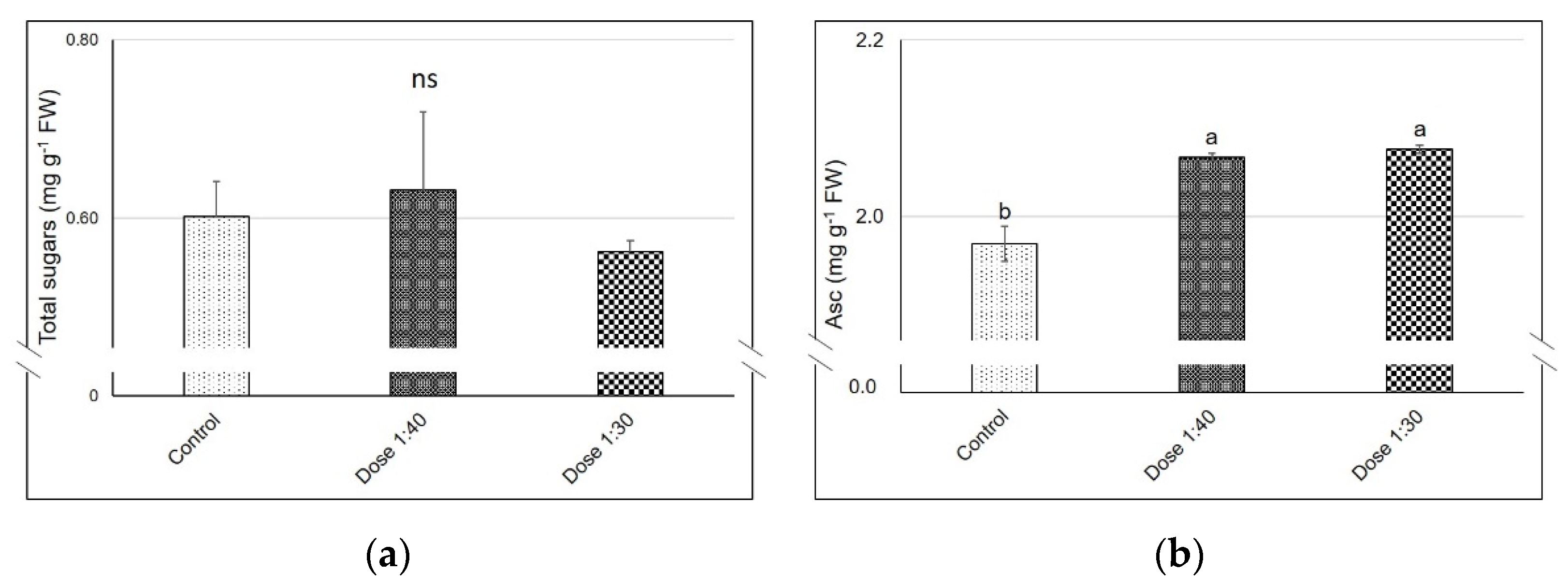
2.7. Total Sugars
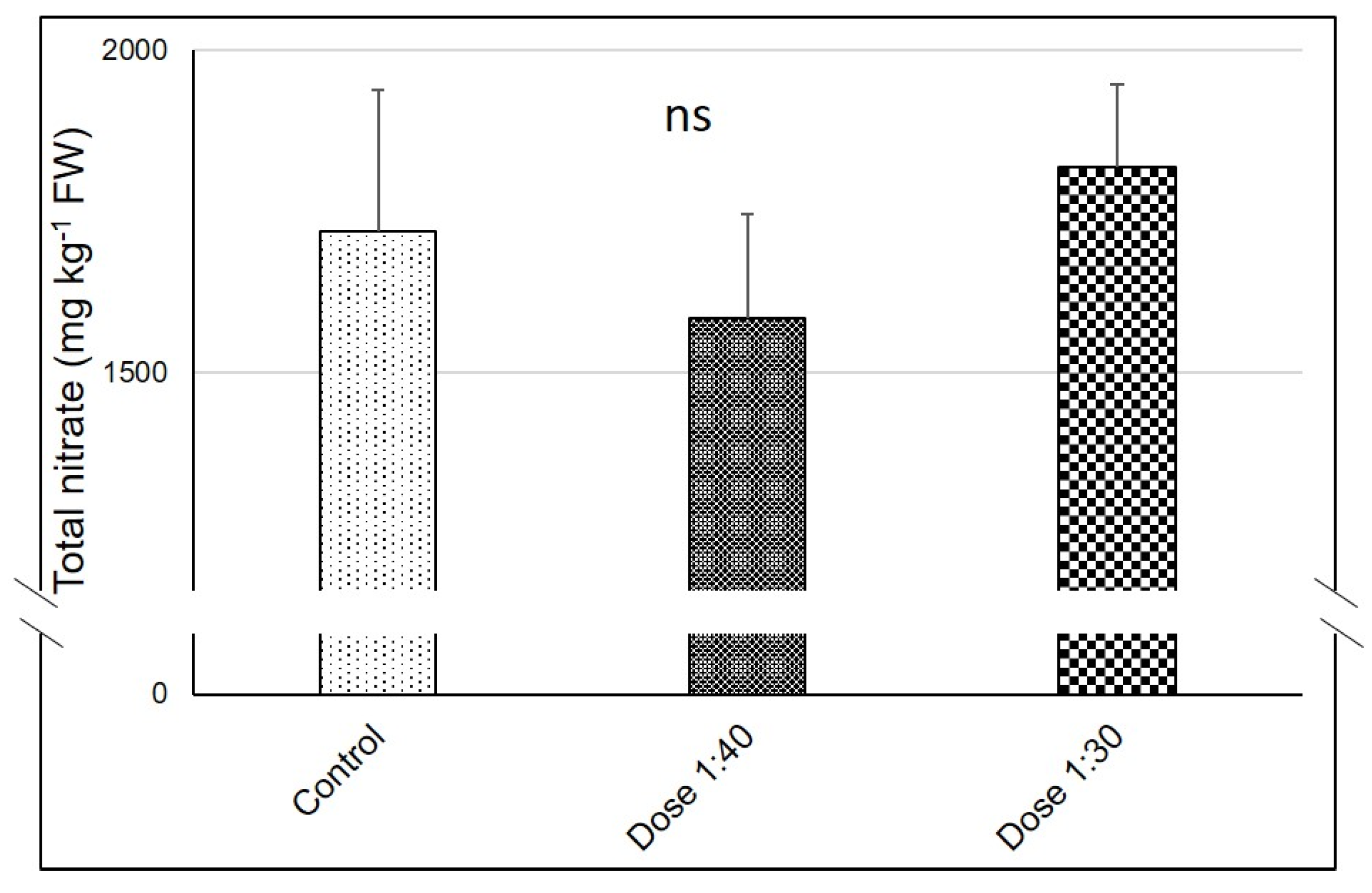
2.8. Ascorbic Acid (Asc) Analysis and Nitrate Concentration
2.9. Statistical Analysis
3. Results
3.1. Plantlets Characteristics and Photosytem Efficiency
3.2. Chlorophyll a, b, and Total, and Carotenoids
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Miller-Cebert, R.L.; Sistani, N.A.; Cebert, E. Comparative mineral composition among canola cultivars and other cruciferous leafy greens. J. Food Compos. Anal. 2009, 22, 112–116. [Google Scholar] [CrossRef]
- Clarkson, G.J.; O’Byrne, E.E.; Rothwell, S.D.; Taylor, G. Identifying traits to improve postharvest processability in baby leaf salad. Postharvest Biol. Technol. 2003, 30, 287–298. [Google Scholar] [CrossRef]
- Di Bella, M.C.; Niklas, A.; Toscano, S.; Picchi, V.; Romano, D.; Lo Scalzo, R.; Branca, F. Morphometric characteristics, polyphenols and ascorbic acid variation in Brassica oleracea L. novel foods: Sprouts, microgreens and baby leaves. Agronomy 2020, 10, 782. [Google Scholar] [CrossRef]
- Santos, J.; Oliveira, M.B.P.P.; Ibáñez, E.; Herrero, M. Phenolic profile evolution of different ready-to-eat baby-leaf vegetables during storage. J. Chromatogr. A 2014, 1327, 118–131. [Google Scholar] [CrossRef] [Green Version]
- Ryder, E.J. The New salad crop revolution. In Proceedings of the Fifth National Symposium Trends in New Crops and New Uses, Atlanta, GA, USA, 10–13 November 2001; pp. 408–412. [Google Scholar]
- Tsouvaltzis, P.; Brecht, J.K. Changes in Quality and Antioxidant Enzyme Activities of Bunched and Topped Radish (Raphanus sativus L.) Plants during Storage at 5 or 10C. J. Food Qual. 2014, 37, 157–167. [Google Scholar] [CrossRef]
- Curtis, I.S. The noble radish: Past, present and future. Trends Plant Sci. 2003, 8, 305–307. [Google Scholar] [CrossRef]
- Beevi, S.S.; Narasu, M.L.; Gowda, B.B. Polyphenolics profile, antioxidant and radical scavenging activity of leaves and stem of Raphanus sativus L. Plant Foods Hum. Nutr. 2010, 65, 8–17. [Google Scholar] [CrossRef]
- Takaya, Y.; Kondo, Y.; Furukawa, T.; Niwa, M. Antioxidant constituents of radish sprout (Kaiware-daikon), Raphanus sativus L. J. Agric. Food Chem. 2003, 51, 8061–8066. [Google Scholar] [CrossRef]
- Savci, S. Investigation of effect of chemical fertilizers on environment. APCBEE Procedia 2012, 1, 287–292. [Google Scholar] [CrossRef] [Green Version]
- Phiri, C.; Mbewe, D.N. Influence of Moringa oleifera leaf extracts on germination and seedling survival of three common legumes. Int. J. Agric. Biol. 2010, 12, 315–317. Available online: http://www.fspublishers.org/published_papers/35759_..pdf (accessed on 14 June 2022).
- Phiri, C. Influence of Moringa oleifera leaf extracts on germination and early seedling development of major cereals. Agric. Biol. J. N. Am. 2010, 1, 774–777. [Google Scholar] [CrossRef]
- Nagar, P.K.; Iyer, R.I.; Sircar, P.K. Cytokinins in developing fruits of Moringa pterigosperma Gaertn. Physiol. Plant. 1982, 55, 45–50. [Google Scholar] [CrossRef]
- Siddhuraju, P.; Becker, K. Antioxidant properties of various solvent extracts of total phenolic constituents from three different agroclimatic origins of drumstick tree (Moringa oleifera Lam.) leaves. J. Agric. Food Chem. 2003, 51, 2144–2155. [Google Scholar] [CrossRef]
- Anwar, F.; Rashid, U. Physico-chemical characteristics of Moringa oleifera seeds and seed oil from a wild provenance of Pakistan. Pak. J. Bot. 2007, 39, 1443–1453. [Google Scholar]
- Oluduro, A.O. Evaluation of antimicrobial properties and nutritional potentials of Moringa oleifera Lam. leaf in South-Western Nigeria. Malays. J. Microbiol. 2012, 8, 59–67. [Google Scholar]
- Jacob, S.J.P.; Shenbagaraman, S. Evaluation of antioxidant and antimicrobial activities of the selected green leafy vegetables. Int. J. PharmTech Res. 2011, 3, 148–152. [Google Scholar]
- Dehshahri, S.H.; Wink, M.; Afsharypuor, S.; Asghari, G.; Mohagheghzadeh, A. Antioxidant activity of methanolic leaf extract of Moringa peregrina (Forssk.). Fiori. Res. Pharm. Sci. 2012, 7, 111. [Google Scholar]
- Pakade, V.; Cukrowska, E.; Chimuka, L. Comparison of antioxidant activity of Moringa oleifera and selected vegetables in South Africa. S. Afr. J. Sci. 2013, 109, 1–5. Available online: https://hdl.handle.net/10520/EJC133141 (accessed on 20 May 2022). [CrossRef]
- Toscano, S.; Ferrante, A.; Branca, F.; Romano, D. Enhancing the quality of two species of baby leaves sprayed with Moringa leaf extract as biostimulant. Agronomy 2021, 11, 1399. [Google Scholar] [CrossRef]
- Nofal, E.M.S.; Khalafallah, M.M.; Shahin, S.M.; Montasser, H.M.M.S. Usage of magnetic iron to raise tolerance of some ornamental trees and shrubs to soil salinity in case of horseradish tree (Moringa oleifera Lam.). Appl. Ecol. Environ. Res. 2021, 19, 1177–1190. [Google Scholar] [CrossRef]
- Ali, O.; Ramsubhag, A.; Jayaraman, J. Biostimulant properties of seaweed extracts in plants: Implications towards sustainable crop production. Plants 2021, 10, 531. [Google Scholar] [CrossRef]
- Zulfiqar, F.; Casadesús, A.; Brockman, H.; Munné-Bosch, S. An overview of plant-based natural biostimulants for sustainable horticulture with a particular focus on moringa leaf extracts. Plant Sci. 2020, 295, 110194. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K. Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes. Method Enzymol. 1987, 148, 350–382. [Google Scholar]
- Wang, Y.; Gao, Y.; Ding, H.; Liu, S.; Han, X.; Gui, J.; Liu, D. Subcritical ethanol extraction of flavonoids from Moringa oleifera leaf and evaluation of antioxidant activity. Food Chem. 2017, 218, 152–158. [Google Scholar] [CrossRef]
- Yemm, E.W.; Willis, A.J. The estimation of carbohydrates in plant extracts by anthrone. Biochem. J. 1954, 57, 508–514. [Google Scholar] [CrossRef] [Green Version]
- Cocetta, G.; Rossoni, M.; Gardana, C.; Mignani, I.; Ferrante, A.; Spinardi, A. Methyl jasmonate affects phenolic metabolism and gene expression in blueberry (Vaccinium corymbosum). Physiol. Plant. 2015, 153, 269–283. [Google Scholar] [CrossRef]
- Janghel, E.K.; Gupta, V.K.; Rai, M.K.; Rai, J.K. Micro determination of ascorbic acid using methyl viologen. Talanta 2007, 72, 1013–1016. [Google Scholar] [CrossRef]
- Cataldo, D.A.; Haroon, M.; Sehrader, L.E.; Youngs, V.L. Rapid colorimetric determination of nitrate in plant tissue by titration of salicylic acid. Commun. Soil Sci. Plant Anal. 1975, 6, 71–80. [Google Scholar] [CrossRef]
- Chojnacka, K. Innovative bio-products for agriculture. Open Chem. 2015, 13, 932–937. [Google Scholar] [CrossRef]
- Jahan, I. Foliar application of moringa leaf extract as a bio-stimulant on growth, yield and nutritional quality of brinjal. J. Agric. Food Environ. 2020, 1, 94–99. [Google Scholar]
- Xu, L.; Geelen, D. Developing Biostimulants from Agro-Food and Industrial By-Products. Front. Plant Sci. 2018, 9, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khan, A.U.; Ullah, F.; Khan, N.; Mehmood, S.; Fahad, S.; Datta, R.; Irshad, I.; Danish, S.; Saud, S.; Alaraidh, I.A. Production of organic fertilizers from rocket seed (Eruca sativa L.), chicken peat and Moringa oleifera leaves for growing linseed under water deficit stress. Sustainability 2021, 13, 59. [Google Scholar] [CrossRef]
- Sandhu, R.K.; Nandwani, D. Effect of Stimplex® on yield and growth performance of organic leafy greens in Tennessee. In Proceedings of the 43rd Annual Meeting of the Plant Growth Regulation Society of America, Raleigh, NC, USA, 17–21 July 2016; pp. 63–69. [Google Scholar]
- Bulgari, R.; Franzoni, G.; Ferrante, A. Biostimulants application in horticultural crops under abiotic stress conditions. Agronomy 2019, 9, 306. [Google Scholar] [CrossRef] [Green Version]
- Abd El-Mageed, T.A.; Semida, W.M.; Rady, M.M. Moringa leaf extract as biostimulant improves water use efficiency, physio-biochemical attributes of squash plants under deficit irrigation. Agric. Water Manag. 2017, 193, 46–54. [Google Scholar] [CrossRef]
- Arif, Y.; Bajguz, A.; Hayat, S. Moringa oleifera extract as a natural plant biostimulant. J. Plant Growth Regul. 2022, 1–16. [Google Scholar] [CrossRef]
- Hoque, T.S.; Rana, M.S.; Zahan, S.A.; Jahan, I.; Abedin, M.A. Moringa Leaf Extract as a Bio-Stimulant on Growth, Yield and Nutritional Improvement in Cabbage. Asian J. Med. Biol. Res. 2020, 6, 196–203. [Google Scholar] [CrossRef]
- Abdalla, M.M. The potential of Moringa oleifera extract as a biostimulant in enhancing the growth, biochemical and hormonal contents in rocket (Eruca vesicaria subsp. sativa) plants. Int. J. Plant Physiol. Biochem. 2013, 5, 42–49. [Google Scholar] [CrossRef]
- Mehmood, A.; Naveed, K.; Ayub, Q.; Alamri, S.; Siddiqui, M.H.; Wu, C.; Depeng, W.; Shah Saud, J.B.; Subhan, D.; Rahul, D.; et al. Exploring the potential of moringa leaf extract as bio stimulant for improving yield and quality of black cumin oil. Sci. Rep. 2021, 11, 24217. [Google Scholar] [CrossRef]
- Scaglia, B.; Pognani, M.; Adani, F. The anaerobic digestion process capability to produce biostimulant: The case study of the dissolved organic matter (DOM) vs. auxin-like property. Sci. Total Environ. 2017, 589, 36–45. [Google Scholar] [CrossRef]
- Makkar, H.A.; Becker, K. Nutrional value and antinutritional components of whole and ethanol extracted Moringa oleifera leaves. Anim. Feed Sci. Technol. 1996, 63, 211–228. [Google Scholar] [CrossRef]
- Moyo, B.; Masika, P.J.; Hugo, A.; Muchenje, V. Nutritional characterization of Moringa (Moringa oleifera Lam.) leaves. Afr. J. Biotechnol. 2011, 10, 12925–12933. [Google Scholar] [CrossRef] [Green Version]
- Foidl, N.; Makkar, H.P.S.; Becker, K. The Potential of Moringa oleifera for Agricultural and Industrial Uses. What Dev. Potential Moringa Prod. 2001, 20. Available online: https://betuco-wp.be/Agroforestry/Tree/Moringa%20Oleifera%20-%20Potential%20for%20agricultural%20and%20industrial%20uses.pdf (accessed on 20 May 2022).
- Hassanein, R.A.; Abdelkader, A.F.; Fara-mawy, H.M. Moringa leaf extracts as biostimulants-inducing salinity tolerance in the sweet basil plant. Egypt. J. Bot. 2019, 59, 303–318. [Google Scholar] [CrossRef]
- Hala, H.; El-Noor, A.; Ewais, N.A. Effect of Moringa oleifera leaf extract (MLE) on pepper seed germination, seedlings improvement, growth, fruit yield and its quality. Middle East J. Agric. Res. 2017, 6, 448–463. [Google Scholar]
- Merwad, A.R.M. Effect of humic and fulvic substances and Moringa leaf extract on Sudan grass plants grown under saline conditions. Can. J. Soil Sci. 2017, 97, 703–716. [Google Scholar] [CrossRef] [Green Version]
- Matthew, A. Moringa leaf extract on the growth and yield of pepper (Capsicum annuum L.). ARPN J. Agric. Biol. Sci. 2016, 11, 107–109. [Google Scholar]
- Emongor, V.E. Effects of Moringa (Moringa oleifera) leaf extract on growth, yield and yield components of snap beans (Phaseolus vulgaris). Br. J. Appl. Sci. Technol. 2015, 6, 114–122. [Google Scholar] [CrossRef]
- Rady, M.M.; Mohamed, G.F.; Abdalla, A.M.; Ahmed, Y.H. Integrated application of salicylic acid and Moringa oleifera leaf extract alleviates the salt-induced adverse effects in common bean plants. Int. J. Agric. Technol. 2015, 11, 1595–1614. [Google Scholar]
- Ozobia, A.P. Comparative assessment of effect of Moringa extracts, NPK fertilizer and poultry manure on soil properties and growth performance of Solanium menlongina in Abuja, North Central Region of Nigeria. Int. J. Agric. Crop. Sci. 2014, 2, 88–93. [Google Scholar]
- Khan, F.; Pandey, P.; Ahmad, V.; Upadhyay, T.K. Moringa oleifera methanolic leaves extract induces apoptosis and G0/G1 cell cycle arrest via downregulation of Hedgehog Signaling Pathway in human prostate PC-3 cancer cells. J. Food Biochem. 2020, 44, e13338. [Google Scholar] [CrossRef]
- Toscano, S.; Romano, D.; Massa, D.; Bulgari, R.; Franzoni, G.; Ferrante, A. Biostimulant applications in low input horticultural cultivation systems. Italus Hortus 2019, 25, 27–36. [Google Scholar] [CrossRef]
- Sheng, M.; Tang, M.; Chen, H.; Yang, B.; Zhang, F.; Huang, Y. Influence of arbuscular mycorrhizae on photosynthesis and water status of maize plants under salt stress. Mycorrhiza 2008, 18, 287–296. [Google Scholar] [CrossRef]
- Bulgari, R.; Cocetta, G.; Trivellini, A.; Ferrante, A. Borage extracts affect wild rocket quality and influence nitrate and carbon metabolism. Physiol. Mol. Biol. Plants 2020, 26, 649–660. [Google Scholar] [CrossRef] [PubMed]
- Yaseen, A.A.; Takacs-Hajos, M. Evaluation of moringa (Moringa oleifera Lam.) leaf extract on bioactive compounds of lettuce (Lactuca sativa L.) grown under glasshouse environment. J. King Saud Univ. Sci. 2022, 34, 101916. [Google Scholar] [CrossRef]
- Aslam, M.; Sultana, B.; Anwar, F.; Munir, H. Foliar spray of selected plant growth regulators affected the biochemical and antioxidant attributes of spinach in a field experiment. Turk. J. Agric. For. 2016, 40, 136–145. [Google Scholar] [CrossRef]
- Yakhin, O.I.; Lubyanov, A.A.; Yakhin, I.A.; Brown, P.H. Biostimulants in plant science: A global perspective. Front. Plant Sci. 2017, 7, 2049. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nasir, M.; Khan, A.S.; Basra, S.A.; Malik, A.U. Foliar application of moringa leaf extract, potassium and zinc influence yield and fruit quality of ‘Kinnow’ mandarin. Sci. Hortic. 2016, 210, 227–235. [Google Scholar] [CrossRef]
- Bulgari, R.; Podetta, N.; Cocetta, G.; Piaggesi, A.; Ferrante, A. The effect of a complete fertilizer for leafy vegetables production in family and urban gardens. Bulg. J. Agric. Sci. 2014, 20, 1361–1367. Available online: http://www.agrojournal.org/20/06-12.pdf (accessed on 15 June 2022).
- Yasmeen, A.; Basra, S.M.A.; Farooq, M.; Hussain, N. Exogenous application of moringa leaf extract modulates the antioxidant enzyme system to improve wheat performance under saline conditions. Plant Growth Regul. 2013, 69, 225–233. [Google Scholar] [CrossRef]
| Treatments | Fresh Biomass (FW, g plant−1) | Dry Biomass (DW, %) | Total Leaf Area (cm2 plant−1) | Unit Leaf Area (cm2) | Wx (cm) | Wy (cm) |
|---|---|---|---|---|---|---|
| Control | 0.514 ± 0.016 c | 15.640 ± 0.339 | 12.601 ± 0.516 c | 4.475 ± 0.118 c | 1.861 ± 0.039 c | 3.731 ± 0.052 c |
| 1:40 | 0.602 ± 0.020 b | 16.208 ± 0.321 | 14.935 ± 0.569 b | 5.301 ± 0.125 b | 2.058 ± 0.030 b | 3.970 ± 0.061 b |
| 1:30 | 0.800 ± 0.022 a | 16.796 ± 0.321 | 20.460 ± 0.652 a | 6.781 ± 0.219 a | 2.358 ± 0.049 a | 4.556 ± 0.073 a |
| Significance | *** | ns | *** | *** | *** | *** |
| Treatments | L* | a* | b* | F0 | Fm | Fv/Fm |
|---|---|---|---|---|---|---|
| Control | 54.390 ± 0.449 | −21.008 ± 0.248 a | 34.031 ± 0.660 b | 444.000 ± 18.369 | 1779.889 ± 57.664 | 0.748 ± 0.012 b |
| 1:40 | 55.082 ± 0.450 | −21.362 ± 0.158 a | 35.107 ± 0.567 ab | 428.556 ± 33.311 | 1935.000 ± 147.016 | 0.778 ± 0.002 a |
| 1:30 | 55.470 ± 0.482 | −22.001 ± 0.197 b | 36.403 ± 0.733 a | 433.778 ± 11.201 | 2037.222 ± 59.715 | 0.786 ± 0.004 a |
| Significance | ns | ** | * | ns | ns | ** |
| Treatments | Chl a(µg mg−1 FW) | Chl b(µg mg−1 FW) | Total Chl (µg mg−1 FW) | Carotenoids (µg mg−1 FW) | Chl a/Chl b | Chl/Car |
|---|---|---|---|---|---|---|
| Control | 0.410 ± 0.021 b | 0.155 ± 0.010 c | 0.572 ± 0.041 b | 0.097 ± 0.003 c | 2.654 ± 0.002 b | 3.284 ± 0.187 b |
| 1:40 | 0.699 ± 0.023 a | 0.229 ± 0.016 a | 0.923 ± 0.036 a | 0.173 ± 0.004 a | 3.051 ± 0.005 a | 4.923 ± 0.191 a |
| 1:30 | 0.682 ± 0.014 a | 0.208 ± 0.00 b | 0.826 ± 0.016 a | 0.152 ± 0.008 b | 2.983 ± 0.004 a | 4.453 ± 0.112 a |
| Significance | *** | *** | *** | *** | *** | ** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Romano, D.; La Fornara, G.; Tribulato, A.; Toscano, S. Can Moringa Leaf Spray Treatment Increase the Nutraceutical Properties of Radish Baby Leaf? Horticulturae 2022, 8, 671. https://doi.org/10.3390/horticulturae8080671
Romano D, La Fornara G, Tribulato A, Toscano S. Can Moringa Leaf Spray Treatment Increase the Nutraceutical Properties of Radish Baby Leaf? Horticulturae. 2022; 8(8):671. https://doi.org/10.3390/horticulturae8080671
Chicago/Turabian StyleRomano, Daniela, Giovanni La Fornara, Alessandro Tribulato, and Stefania Toscano. 2022. "Can Moringa Leaf Spray Treatment Increase the Nutraceutical Properties of Radish Baby Leaf?" Horticulturae 8, no. 8: 671. https://doi.org/10.3390/horticulturae8080671
APA StyleRomano, D., La Fornara, G., Tribulato, A., & Toscano, S. (2022). Can Moringa Leaf Spray Treatment Increase the Nutraceutical Properties of Radish Baby Leaf? Horticulturae, 8(8), 671. https://doi.org/10.3390/horticulturae8080671









