Advances and Perspectives in the Use of Biocontrol Agents against Fungal Plant Diseases
Abstract
:1. Introduction
2. Biocontrol Agents and Their Activity against Fungal Plant Pathogens
2.1. BCAs against Soil-Borne Pathogens
2.1.1. Microbial Consortia
2.2. BCAs against Air-Borne Pathogens
2.3. BCAs against Postharvest Pathogens
3. BCAs under Evaluation or Already Approved as Biofungicides in the EU
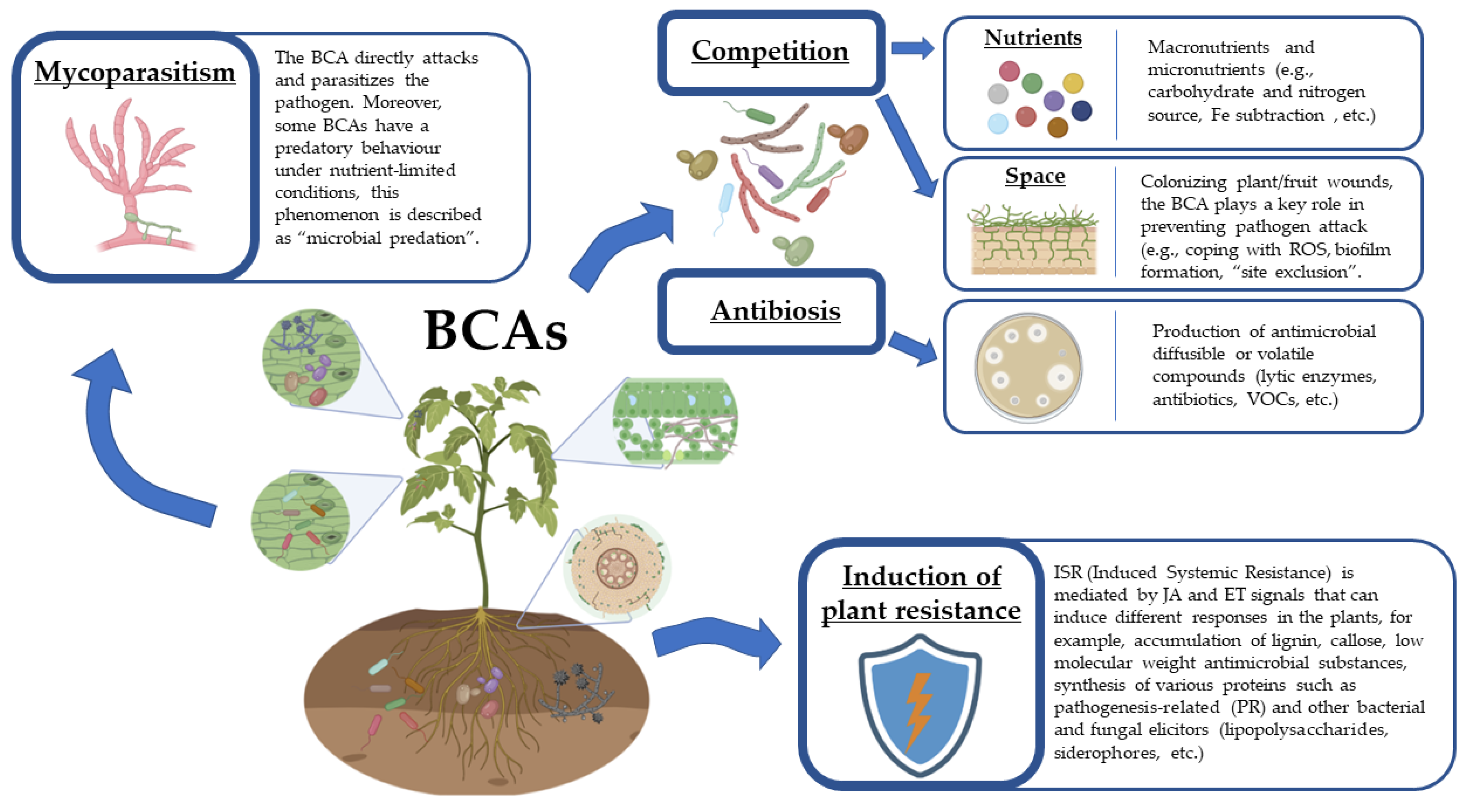
4. Mechanisms of Action of Biocontrol Agents
4.1. Mycoparasitism
4.2. Antibiosis
4.3. Competition
4.4. Induced Resistance
5. Molecular Approaches to Potentiate the Effectiveness of BCAs against Fungal Pathogens
5.1. NGS Techniques to Elucidate the Mechanisms of Action of Fungal, Yeasts, and Bacterial BCAs
5.2. Functional Genomics to Identify Fungal, Yeast, and Bacterial Genes Important for Biocontrol
6. Issues and Constraints in the Registration and Commercial Development of Biocontrol Agents against Phytopathogenic Fungi
6.1. Development of a Stable Formulate
6.2. Patent Application
6.3. Registration of Active Ingredients and Formulation Process
- -
- Protection of human and environmental safety;
- -
- Ensuring and maintaining quality standards;
- -
- Protection of technological invention and rights [287].
- -
- In the registration dossier, the data required are differentiated for the active ingredient and the formulate [288].
- -
- Data requirements for an active substance (Technical Grade Active Ingredient, TGAI) usually include:
- -
- Identity and purity;
- -
- Physical and chemical or biological properties;
- -
- Further information on use, production processes, and related areas;
- -
- Analytical methods used to identify the active ingredient;
- -
- Effects on Human health;
- -
- Residues (often confused with persistence);
- -
- Fate and behavior in the environment;
- -
- Effects on non-target organisms;
- -
- Summary of all.
- -
- Data requirements for the formulated product (FP):
- -
- Identity and composition of the formulation;
- -
- Physical and chemical properties;
- -
- Application, labelling, and packaging;
- -
- Further information;
- -
- Analytical methods;
- -
- Efficacy data;
- -
- Toxicology and exposure;
- -
- Residues;
- -
- Fate and behavior in the environment;
- -
- Effects on non-target organisms;
- -
- Summary.
7. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rajaram, S.; Dubin, H.J. Plant Diseases, Global Food Security and the Role of R. Glenn Anderson. In Plant Diseases and Food Security in the 21st Century; Scott, P., Strange, R., Korsten, L., Gullino, M.L., Eds.; Springer International Publishing: Cham, Switzerland, 2021; pp. 35–45. ISBN 978-3-030-57899-2. [Google Scholar]
- Castoria, R.; Logrieco, A. Mycotoxins in fruits and major fruit-derived products-an overview. Microb. Biotechnol. Hortic. 2007, 2, 305–344. [Google Scholar]
- Sanzani, S.M.; Reverberi, M.; Geisen, R. Mycotoxins in harvested fruits and vegetables: Insights in producing fungi, biological role, conducive conditions, and tools to manage postharvest contamination. Postharvest Biol. Technol. 2016, 122, 95–105. [Google Scholar] [CrossRef]
- Pirttilä, A.M.; Mohammad Parast Tabas, H.; Baruah, N.; Koskimäki, J.J. Biofertilizers and Biocontrol Agents for Agriculture: How to Identify and Develop New Potent Microbial Strains and Traits. Microorganisms 2021, 9, 817. [Google Scholar] [CrossRef] [PubMed]
- Tariq, M.; Khan, A.; Asif, M.; Khan, F.; Ansari, T.; Shariq, M.; Siddiqui, M.A. Biological control: A sustainable and practical approach for plant disease management. Acta Agric. Scand. Sect. B—Soil Plant Sci. 2020, 70, 507–524. [Google Scholar] [CrossRef]
- Kumar, J.; Ramlal, A.; Mallick, D.; Mishra, V. An Overview of Some Biopesticides and Their Importance in Plant Protection for Commercial Acceptance. Plants 2021, 10, 1185. [Google Scholar] [CrossRef]
- Köhl, J.; Kolnaar, R.; Ravensberg, W.J. Mode of Action of Microbial Biological Control Agents against Plant Diseases: Relevance Beyond Efficacy. Front. Plant Sci. 2019, 10, 845. [Google Scholar] [CrossRef] [Green Version]
- Ons, L.; Bylemans, D.; Thevissen, K.; Cammue, B.P.A. Combining Biocontrol Agents with Chemical Fungicides for Integrated Plant Fungal Disease Control. Microorganisms 2020, 8, 1930. [Google Scholar] [CrossRef]
- RBH, W.; Golding, J. Advances in Postharvest Fruit and Vegetable Technology; CRC Press: Boca Raton, FL, USA, 2015; ISBN 978-1-4822-1697-7. [Google Scholar]
- Frederiks, C.; Wesseler, J.H.H. A comparison of the EU and US regulatory frameworks for the active substance registration of microbial biological control agents. Pest Manag. Sci. 2019, 75, 87–103. [Google Scholar] [CrossRef] [Green Version]
- Shoda, M. Bacterial control of plant diseases. J. Biosci. Bioeng. 2000, 89, 515–521. [Google Scholar] [CrossRef]
- Vinale, F.; Sivasithamparam, K.; Ghisalberti, E.L.; Marra, R.; Woo, S.L.; Lorito, M. Trichoderma–plant–pathogen interactions. Soil Biol. Biochem. 2008, 40, 1–10. [Google Scholar] [CrossRef]
- Ampt, E.A.; van Ruijven, J.; Raaijmakers, J.M.; Termorshuizen, A.J.; Mommer, L. Linking ecology and plant pathology to unravel the importance of soil-borne fungal pathogens in species-rich grasslands. Eur. J. Plant Pathol. 2019, 154, 141–156. [Google Scholar] [CrossRef] [Green Version]
- Cacciola, S.O.; Gullino, M.L. Emerging and re-emerging fungus and oomycete soil-borne plant diseases in Italy. Phytopathol. Mediterr. 2019, 58, 451–472. [Google Scholar] [CrossRef]
- Schlatter, D.; Kinkel, L.; Thomashow, L.; Weller, D.; Paulitz, T. Disease Suppressive Soils: New Insights from the Soil Microbiome. Phytopathology 2017, 107, 1284–1297. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bastida, F.; Eldridge, D.J.; García, C.; Kenny Png, G.; Bardgett, R.D.; Delgado-Baquerizo, M. Soil microbial diversity–biomass relationships are driven by soil carbon content across global biomes. ISME J. 2021, 15, 2081–2091. [Google Scholar] [CrossRef] [PubMed]
- Jayaraman, S.; Naorem, A.K.; Lal, R.; Dalal, R.C.; Sinha, N.K.; Patra, A.K.; Chaudhari, S.K. Disease-Suppressive Soils—Beyond Food Production: A Critical Review. J. Soil Sci. Plant Nutr. 2021, 21, 1437–1465. [Google Scholar] [CrossRef]
- Montesinos, E.; Bonaterra, A. Pesticides, Microbial. In Encyclopedia of Microbiology, 3rd ed.; Schaechter, M., Ed.; Academic Press: Oxford, UK, 2009; pp. 110–120. ISBN 978-0-12-373944-5. [Google Scholar]
- Rodríguez, M.A.; Rothen, C.; Lo, T.E.; Cabrera, G.M.; Godeas, A.M. Suppressive soil against Sclerotinia sclerotiorum as a source of potential biocontrol agents: Selection and evaluation of Clonostachys rosea BAFC1646. Biocontrol Sci. Technol. 2015, 25, 1388–1409. [Google Scholar] [CrossRef] [Green Version]
- Brescia, F.; Pertot, I.; Puopolo, G. Lysobacter. In Beneficial Microbes in Agro-Ecology: Bacteria and Fungi; Amaresan, N., Senthil Kumar, M., Annapurna, K., Kumar, K., Sankaranarayanan, A., Eds.; Academic Press: Cambridge, MA, USA, 2020; Chapter 16; pp. 313–338. ISBN 978-0-12-823414-3. [Google Scholar]
- Alfiky, A.; Weisskopf, L. Deciphering Trichoderma–Plant–Pathogen Interactions for Better Development of Biocontrol Applications. J. Fungi 2021, 7, 61. [Google Scholar] [CrossRef]
- Zin, N.A.; Badaluddin, N.A. Biological functions of Trichoderma spp. for agriculture applications. Ann. Agric. Sci. 2020, 65, 168–178. [Google Scholar] [CrossRef]
- del Carmen, H.; Rodríguez, M.; Evans, H.C.; de Abreu, L.M.; de Macedo, D.M.; Ndacnou, M.K.; Bekele, K.B.; Barreto, R.W. New species and records of Trichoderma isolated as mycoparasites and endophytes from cultivated and wild coffee in Africa. Sci. Rep. 2021, 11, 5671. [Google Scholar] [CrossRef]
- Kandula, D.R.W.; Jones, E.E.; Stewart, A.; McLean, K.L.; Hampton, J.G. Trichoderma species for biocontrol of soil-borne plant pathogens of pasture species. Biocontrol Sci. Technol. 2015, 25, 1052–1069. [Google Scholar] [CrossRef]
- González-Pérez, E.; Ortega-Amaro, M.A.; Salazar-Badillo, F.B.; Bautista, E.; Douterlungne, D.; Jiménez-Bremont, J.F. The Arabidopsis-Trichoderma interaction reveals that the fungal growth medium is an important factor in plant growth induction. Sci. Rep. 2018, 8, 16427. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perrin, R. Interactions between mycorrhizae and diseases caused by soil-borne fungi. Soil Use Manag. 1990, 6, 189–194. [Google Scholar] [CrossRef]
- Singh, R.; Adholeya, A.; Mukerji, K.G. Mycorrhiza in Control of Soil Borne Pathogens. In Mycorrhizal Biology; Mukerji, K.G., Chamola, B.P., Singh, J., Eds.; Springer: Boston, MA, USA, 2000; pp. 173–196. ISBN 978-1-4615-4265-0. [Google Scholar]
- Harrier, L.A.; Watson, C.A. The potential role of arbuscular mycorrhizal (AM) fungi in the bioprotection of plants against soil-borne pathogens in organic and/or other sustainable farming systems. Pest Manag. Sci. 2004, 60, 149–157. [Google Scholar] [CrossRef]
- Bücking, H. The Role of the Mycorrhizal Symbiosis in Nutrient Uptake of Plants and the Regulatory Mechanisms Underlying These Transport Processes. In Plant Science; Liepold, E., Ed.; IntechOpen: Rijeka, Croatia, 2012; Chapter 4. [Google Scholar]
- Soudzilovskaia, N.A.; Vaessen, S.; Barcelo, M.; He, J.; Rahimlou, S.; Abarenkov, K.; Brundrett, M.C.; Gomes, S.; Merckx, V.; Tedersoo, L. FungalRoot: Global online database of plant mycorrhizal associations. New Phytol. 2020, 227, 955–966. [Google Scholar] [CrossRef] [PubMed]
- Goicoechea, N. Mycorrhizal Fungi as Bioprotectors of Crops Against Verticillium Wilt—A Hypothetical Scenario Under Changing Environmental Conditions. Plants 2020, 9, 1468. [Google Scholar] [CrossRef] [PubMed]
- Azcón-Aguilar, C.; Jaizme-Vega, M.C.; Calvet, C. The contribution of arbuscular mycorrhizal fungi to the control of soil-borne plant pathogens. In Mycorrhizal Technology in Agriculture; Gianinazzi, S., Schüepp, H., Barea, J.M., Haselwandter, K., Eds.; Birkhäuser Basel: Basel, Switzerland, 2002; pp. 187–197. ISBN 978-3-0348-8117-3. [Google Scholar]
- Shaikh, S.S.; Sayyed, R.Z. Role of Plant Growth-Promoting Rhizobacteria and Their Formulation in Biocontrol of Plant Diseases. In Plant Microbes Symbiosis: Applied Facets; Arora, N.K., Ed.; Springer: New Delhi, India, 2015; pp. 337–351. ISBN 978-81-322-2068-8. [Google Scholar]
- Labuschagne, N.; Pretorius, T.; Idris, A.H. Plant Growth Promoting Rhizobacteria as Biocontrol Agents against Soil-Borne Plant Diseases. In Plant Growth and Health Promoting Bacteria; Maheshwari, D.K., Ed.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 211–230. ISBN 978-3-642-13612-2. [Google Scholar]
- Weller, D.M. Pseudomonas Biocontrol Agents of Soilborne Pathogens: Looking Back Over 30 Years. Phytopathology 2007, 97, 250–256. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rahman, M. Bacillus spp.: A Promising Biocontrol Agent of Root, Foliar, and Postharvest Diseases of Plants. In Bacilli and Agrobiotechnology; Islam, M.T., Rahman, M., Pandey, P., Jha, C.K., Aeron, A., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 113–141. ISBN 978-3-319-44409-3. [Google Scholar]
- Bukhat, S.; Imran, A.; Javaid, S.; Shahid, M.; Majeed, A.; Naqqash, T. Communication of plants with microbial world: Exploring the regulatory networks for PGPR mediated defense signaling. Microbiol. Res. 2020, 238, 126486. [Google Scholar] [CrossRef]
- LeBlanc, N. Bacteria in the genus Streptomyces are effective biological control agents for management of fungal plant pathogens: A meta-analysis. BioControl 2021, 67, 111–121. [Google Scholar] [CrossRef]
- Barea, J.-M.; Pozo, M.J.; Azcón, R.; Azcón-Aguilar, C. Microbial co-operation in the rhizosphere. J. Exp. Bot. 2005, 56, 1761–1778. [Google Scholar] [CrossRef] [Green Version]
- Handelsman, J.; Stabb, E.V. Biocontrol of Soilborne Plant Pathogens. Plant Cell 1996, 8, 1855–1869. [Google Scholar] [CrossRef]
- De Curtis, F.; Lima, G.; Vitullo, D.; De Cicco, V. Biocontrol of Rhizoctonia solani and Sclerotium rolfsii on tomato by delivering antagonistic bacteria through a drip irrigation system. Crop Prot. 2010, 29, 663–670. [Google Scholar] [CrossRef]
- Mousa, W.K.; Shearer, C.; Limay-Rios, V.; Ettinger, C.L.; Eisen, J.A.; Raizada, M.N. Root-hair endophyte stacking in finger millet creates a physicochemical barrier to trap the fungal pathogen Fusarium graminearum. Nat. Microbiol. 2016, 1, 16167. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Palmieri, D.; Vitale, S.; Lima, G.; Di Pietro, A.; Turrà, D. A bacterial endophyte exploits chemotropism of a fungal pathogen for plant colonization. Nat. Commun. 2020, 11, 5264. [Google Scholar] [CrossRef] [PubMed]
- El-Tarabily, K.A.; Sivasithamparam, K. Potential of yeasts as biocontrol agents of soil-borne fungal plant pathogens and as plant growth promoters. Mycoscience 2006, 47, 25–35. [Google Scholar] [CrossRef]
- Botha, A. Yeasts in Soil. In Biodiversity and Ecophysiology of Yeasts; Péter, G., Rosa, C., Eds.; Springer: Berlin/Heidelberg, Germany, 2006; pp. 221–240. ISBN 978-3-540-30985-7. [Google Scholar]
- Yurkov, A.M. Yeasts of the soil–obscure but precious. Yeast 2018, 35, 369–378. [Google Scholar] [CrossRef] [Green Version]
- Robbins, C.; Thiergart, T.; Hacquard, S.; Garrido-Oter, R.; Gans, W.; Peiter, E.; Schulze-Lefert, P.; Spaepen, S. Root-Associated Bacterial and Fungal Community Profiles of Arabidopsis thaliana Are Robust Across Contrasting Soil P Levels. Phytobiomes J. 2017, 2, 24–34. [Google Scholar] [CrossRef] [Green Version]
- Cecilia Mestre, M.; Rosa, C.A.; Safar, S.V.B.; Libkind, D.; Fontenla, S.B. Yeast communities associated with the bulk-soil, rhizosphere and ectomycorrhizosphere of a Nothofagus pumilio forest in northwestern Patagonia, Argentina. FEMS Microbiol. Ecol. 2011, 78, 531–541. [Google Scholar] [CrossRef]
- Joubert, P.M.; Doty, S.L. Endophytic Yeasts: Biology, Ecology and Applications. In Endophytes of Forest Trees: Biology and Applications; Pirttilä, A.M., Frank, A.C., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 3–14. ISBN 978-3-319-89833-9. [Google Scholar]
- Ferraz, P.; Cássio, F.; Lucas, C. Potential of Yeasts as Biocontrol Agents of the Phytopathogen Causing Cacao Witches’ Broom Disease: Is Microbial Warfare a Solution? Front. Microbiol. 2019, 10, 1766. [Google Scholar] [CrossRef] [Green Version]
- El-Tarabily, K.A. Suppression of Rhizoctonia solani diseases of sugar beet by antagonistic and plant growth-promoting yeasts. J. Appl. Microbiol. 2004, 96, 69–75. [Google Scholar] [CrossRef]
- Niu, B.; Wang, W.; Yuan, Z.; Sederoff, R.R.; Sederoff, H.; Chiang, V.L.; Borriss, R. Microbial Interactions within Multiple-Strain Biological Control Agents Impact Soil-Borne Plant Disease. Front. Microbiol. 2020, 11, 2452. [Google Scholar] [CrossRef]
- Xu, X.-M.; Jeffries, P.; Pautasso, M.; Jeger, M.J. Combined Use of Biocontrol Agents to Manage Plant Diseases in Theory and Practice. Phytopathology 2011, 101, 1024–1031. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Palmieri, D.; Vitullo, D.; De Curtis, F.; Lima, G. A microbial consortium in the rhizosphere as a new biocontrol approach against fusarium decline of chickpea. Plant Soil 2017, 412, 425–439. [Google Scholar] [CrossRef]
- Liu, K.; McInroy, J.A.; Hu, C.-H.; Kloepper, J.W. Mixtures of Plant-Growth-Promoting Rhizobacteria Enhance Biological Control of Multiple Plant Diseases and Plant-Growth Promotion in the Presence of Pathogens. Plant Dis. 2017, 102, 67–72. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vargas-Inciarte, L.; Fuenmayor-Arrieta, Y.; Luzardo-Méndez, M.; Costa-Jardin, M.D.; Vera, A.; Carmona, D.; Homen-Pereira, M.; Costa-Jardin, P.D.; San-Blas, E. Use of different Trichoderma species in cherry type tomatoes (Solanum lycopersicum L.) against Fusarium oxysporum wilt in tropical greenhouses. Agron. Costarric. 2019, 43, 85–100. [Google Scholar]
- Minchev, Z.; Kostenko, O.; Soler, R.; Pozo, M.J. Microbial Consortia for Effective Biocontrol of Root and Foliar Diseases in Tomato. Front. Plant Sci. 2021, 12, 2428. [Google Scholar] [CrossRef] [PubMed]
- Moretti, M.; Grunau, A.; Minerdi, D.; Gehrig, P.; Roschitzki, B.; Eberl, L.; Garibaldi, A.; Gullino, M.L.; Riedel, K. A proteomics approach to study synergistic and antagonistic interactions of the fungal–bacterial consortium Fusarium oxysporum wild-type MSA 35. Proteomics 2010, 10, 3292–3320. [Google Scholar] [CrossRef]
- Win, T.T.; Bo, B.; Malec, P.; Fu, P. The effect of a consortium of Penicillium sp. and Bacillus spp. in suppressing banana fungal diseases caused by Fusarium sp. and Alternaria sp. J. Appl. Microbiol. 2021, 131, 1890–1908. [Google Scholar] [CrossRef]
- Gonda, M.; Garmendia, G.; Rufo, C.; León Peláez, Á.; Wisniewski, M.; Droby, S.; Vero, S. Biocontrol of Aspergillus flavus in Ensiled Sorghum by Water Kefir Microorganisms. Microorganisms 2019, 7, 253. [Google Scholar] [CrossRef] [Green Version]
- Zhimo, V.Y.; Biasi, A.; Kumar, A.; Feygenberg, O.; Salim, S.; Vero, S.; Wisniewski, M.; Droby, S. Yeasts and Bacterial Consortia from Kefir Grains Are Effective Biocontrol Agents of Postharvest Diseases of Fruits. Microorganisms 2020, 8, 428. [Google Scholar] [CrossRef] [Green Version]
- Jawed, K.; Yazdani, S.S.; Koffas, M.A.G. Advances in the development and application of microbial consortia for metabolic engineering. Metab. Eng. Commun. 2019, 9, e00095. [Google Scholar] [CrossRef]
- García-Jiménez, B.; Torres-Bacete, J.; Nogales, J. Metabolic modelling approaches for describing and engineering microbial communities. Comput. Struct. Biotechnol. J. 2021, 19, 226–246. [Google Scholar] [CrossRef] [PubMed]
- Ponomarova, O.; Patil, K.R. Metabolic interactions in microbial communities: Untangling the Gordian knot. Curr. Opin. Microbiol. 2015, 27, 37–44. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mazzola, M.; Freilich, S. Prospects for Biological Soilborne Disease Control: Application of Indigenous versus Synthetic Microbiomes. Phytopathology 2016, 107, 256–263. [Google Scholar] [CrossRef] [Green Version]
- Sarma, B.K.; Yadav, S.K.; Singh, S.; Singh, H.B. Microbial consortium-mediated plant defense against phytopathogens: Readdressing for enhancing efficacy. Soil Biol. Biochem. 2015, 87, 25–33. [Google Scholar] [CrossRef]
- Izquierdo-García, L.F.; González-Almario, A.; Cotes, A.M.; Moreno-Velandia, C.A. Trichoderma virens Gl006 and Bacillus velezensis Bs006: A compatible interaction controlling Fusarium wilt of cape gooseberry. Sci. Rep. 2020, 10, 6857. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pliego, C.; Ramos, C.; de Vicente, A.; Cazorla, F.M. Screening for candidate bacterial biocontrol agents against soilborne fungal plant pathogens. Plant Soil 2011, 340, 505–520. [Google Scholar] [CrossRef] [Green Version]
- Sayeed Akhtar, M.; Siddiqui, Z.A. Biocontrol of a root-rot disease complex of chickpea by Glomus intraradices, Rhizobium sp. and Pseudomonas straita. Crop Prot. 2008, 27, 410–417. [Google Scholar] [CrossRef]
- Szczech, M.; Shoda, M. Biocontrol of Rhizoctonia Damping-off of Tomato by Bacillus subtilis Combined with Burkholderia cepacia. J. Phytopathol. 2004, 152, 549–556. [Google Scholar] [CrossRef]
- Frąc, M.; Hannula, S.E.; Bełka, M.; Jędryczka, M. Fungal Biodiversity and Their Role in Soil Health. Front. Microbiol. 2018, 9, 707. [Google Scholar] [CrossRef] [Green Version]
- Sleutel, S.; Bouckaert, L.; Buchan, D.; Van Loo, D.; Cornelis, W.M.; Sanga, H.G. Manipulation of the soil pore and microbial community structure in soil mesocosm incubation studies. Soil Biol. Biochem. 2012, 45, 40–48. [Google Scholar] [CrossRef]
- Rachwał, K.; Gustaw, K.; Kazimierczak, W.; Waśko, A. Is soil management system really important? comparison of microbial community diversity and structure in soils managed under organic and conventional regimes with some view on soil properties. PLoS ONE 2021, 16, e0256969. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Milner, H.; Díaz-Pérez, J.C.; Ji, P. Effects of soil management practices on soil microbial communities and development of southern blight in vegetable production. Appl. Soil Ecol. 2015, 91, 58–67. [Google Scholar] [CrossRef]
- Massart, S.; Martinez-Medina, M.; Jijakli, M.H. Biological control in the microbiome era: Challenges and opportunities. Biol. Control 2015, 89, 98–108. [Google Scholar] [CrossRef]
- Janakiev, T.; Dimkić, I.; Unković, N.; Ljaljević Grbić, M.; Opsenica, D.; Gašić, U.; Stanković, S.; Berić, T. Phyllosphere Fungal Communities of Plum and Antifungal Activity of Indigenous Phenazine-Producing Pseudomonas synxantha against Monilinia laxa. Front. Microbiol. 2019, 10, 2287. [Google Scholar] [CrossRef] [Green Version]
- Legein, M.; Smets, W.; Vandenheuvel, D.; Eilers, T.; Muyshondt, B.; Prinsen, E.; Samson, R.; Lebeer, S. Modes of Action of Microbial Biocontrol in the Phyllosphere. Front. Microbiol. 2020, 11, 1619. [Google Scholar] [CrossRef]
- Parasuraman, P.; Pattnaik, S.; Busi, S. Phyllosphere Microbiome: Functional Importance in Sustainable Agriculture. In New and Future Developments in Microbial Biotechnology and Bioengineering; Singh, J.S., Singh, D.P., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; Chapter 10; pp. 135–148. ISBN 978-0-444-64191-5. [Google Scholar]
- Lindow, S. Phyllosphere microbiology: A perspective. In Microbial Ecology of Aerial Plant Surfaces; Athenaeum Press: Cambridge, MA, USA, 2006; pp. 1–20. [Google Scholar] [CrossRef]
- Lindow, S.E.; Leveau, J.H.J. Phyllosphere microbiology. Curr. Opin. Biotechnol. 2002, 13, 238–243. [Google Scholar] [CrossRef]
- Rishbeth, J.; Lumsden, R.D.; Gibbs, J.N.; Hamilton, W.D.; Cook, R.J. Biological Control of Air-Borne Pathogens [and Discussion]. Philos. Trans. R. Soc. Lond. B Biol. Sci. 1988, 318, 265–281. [Google Scholar]
- Bacterial Weapons of Fungal Destruction: Phyllosphere-Targeted Biological Control of Plant Diseases, with Emphasis on Sclerotinia Stem Rot and Blackleg Diseases in Canola (Brassica napus L.). In Environmental and Microbial Relationships; Kubicek, C.P.; Druzhinina, I.S. (Eds.) Springer: Berlin/Heidelberg, Germany, 2007; pp. 189–199. ISBN 978-3-540-71840-6. [Google Scholar]
- Griffin, E.A.; Carson, W.P. The Ecology and Natural History of Foliar Bacteria with a Focus on Tropical Forests and Agroecosystems. Bot. Rev. 2015, 81, 105–149. [Google Scholar] [CrossRef]
- Agrios, G.N. Control of Plant Diseases. In Plant Pathology, 5th ed.; Agrios, G.N., Ed.; Academic Press: San Diego, CA, USA, 2005; Chapter 9; pp. 293–353. ISBN 978-0-12-044565-3. [Google Scholar]
- Wilson, M. Biocontrol of aerial plant diseases in agriculture and horticulture: Current approaches and future prospects. J. Ind. Microbiol. Biotechnol. 1997, 19, 188–191. [Google Scholar] [CrossRef]
- Heiniger, U.; Rigling, D. Biological Control of Chestnut blight in Europe. Annu. Rev. Phytopathol. 1994, 32, 581–599. [Google Scholar] [CrossRef]
- Choi, G.H.; Nuss, D.L. Hypovirulence of Chestnut Blight Fungus Conferred by an Infectious Viral cDNA. Science 1992, 257, 800–803. [Google Scholar] [CrossRef] [PubMed]
- Kunova, A.; Pizzatti, C.; Cerea, M.; Gazzaniga, A.; Cortesi, P. New formulation and delivery method of Cryphonectria parasitica for biological control of chestnut blight. J. Appl. Microbiol. 2017, 122, 180–187. [Google Scholar] [CrossRef] [PubMed]
- Carro-Huerga, G.; Compant, S.; Gorfer, M.; Cardoza, R.E.; Schmoll, M.; Gutiérrez, S.; Casquero, P.A. Colonization of Vitis vinifera L. by the Endophyte Trichoderma sp. Strain T154: Biocontrol Activity against Phaeoacremonium minimum. Front. Plant Sci. 2020, 11, 1170. [Google Scholar] [CrossRef]
- Vorholt, J.A. Microbial life in the phyllosphere. Nat. Rev. Microbiol. 2012, 10, 828–840. [Google Scholar] [CrossRef]
- Ceballos, I.; Mosquera, S.; Angulo, M.; Mira, J.J.; Argel, L.E.; Uribe-Velez, D.; Romero-Tabarez, M.; Orduz-Peralta, S.; Villegas, V. Cultivable Bacteria Populations Associated with Leaves of Banana and Plantain Plants and Their Antagonistic Activity against Mycosphaerella fijiensis. Microb. Ecol. 2012, 64, 641–653. [Google Scholar] [CrossRef] [PubMed]
- Khare, S.; Lawhon, S.D.; Drake, K.L.; Nunes, J.E.S.; Figueiredo, J.F.; Rossetti, C.A.; Gull, T.; Everts, R.E.; Lewin, H.A.; Galindo, C.L.; et al. Systems Biology Analysis of Gene Expression during In Vivo Mycobacterium avium paratuberculosis Enteric Colonization Reveals Role for Immune Tolerance. PLoS ONE 2012, 7, e42127. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Cariño, H.F.; Guadarrama-Mendoza, P.C.; Sánchez-López, V.; Cuervo-Parra, J.A.; Ramírez-Reyes, T.; Dunlap, C.A.; Valadez-Blanco, R. Biocontrol of Alternaria alternata and Fusarium oxysporum by Trichoderma asperelloides and Bacillus paralicheniformis in tomato plants. Antonie Van Leeuwenhoek 2020, 113, 1247–1261. [Google Scholar] [CrossRef] [PubMed]
- Kazerooni, E.A.; Maharachchikumbura, S.S.N.; Al-Sadi, A.M.; Kang, S.-M.; Yun, B.-W.; Lee, I.-J. Biocontrol Potential of Bacillus amyloliquefaciens against Botrytis pelargonii and Alternaria alternata on Capsicum annuum. J. Fungi 2021, 7, 472. [Google Scholar] [CrossRef] [PubMed]
- Andreolli, M.; Zapparoli, G.; Angelini, E.; Lucchetta, G.; Lampis, S.; Vallini, G. Pseudomonas protegens MP12: A plant growth-promoting endophytic bacterium with broad-spectrum antifungal activity against grapevine phytopathogens. Microbiol. Res. 2019, 219, 123–131. [Google Scholar] [CrossRef]
- Demissie, Z.A.; Witte, T.; Robinson, K.A.; Sproule, A.; Foote, S.J.; Johnston, A.; Harris, L.J.; Overy, D.P.; Loewen, M.C. Transcriptomic and Exometabolomic Profiling Reveals Antagonistic and Defensive Modes of Clonostachys rosea Action against Fusarium graminearum. Mol. Plant-Microbe Interact. 2020, 33, 842–858. [Google Scholar] [CrossRef]
- Nowak-Thompson, B.; Gould, S.J.; Kraus, J.; Loper, J.E. Production of 2,4-diacetylphloroglucinol by the biocontrol agent Pseudomonas fluorescens Pf-5. Can. J. Microbiol. 1994, 40, 1064–1066. [Google Scholar] [CrossRef]
- Kidarsa, T.A.; Goebel, N.C.; Zabriskie, T.M.; Loper, J.E. Phloroglucinol mediates cross-talk between the pyoluteorin and 2,4-diacetylphloroglucinol biosynthetic pathways in Pseudomonas fluorescens Pf-5. Mol. Microbiol. 2011, 81, 395–414. [Google Scholar] [CrossRef]
- Gross, H.; Stockwell, V.O.; Henkels, M.D.; Nowak-Thompson, B.; Loper, J.E.; Gerwick, W.H. The Genomisotopic Approach: A Systematic Method to Isolate Products of Orphan Biosynthetic Gene Clusters. Chem. Biol. 2007, 14, 53–63. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miotto-Vilanova, L.; Jacquard, C.; Courteaux, B.; Wortham, L.; Michel, J.; Clément, C.; Barka, E.A.; Sanchez, L. Burkholderia phytofirmans PsJN Confers Grapevine Resistance against Botrytis cinerea via a Direct Antimicrobial Effect Combined with a Better Resource Mobilization. Front. Plant Sci. 2016, 7, 1236. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ait Barka, E.; Gognies, S.; Nowak, J.; Audran, J.-C.; Belarbi, A. Inhibitory effect of endophyte bacteria on Botrytis cinerea and its influence to promote the grapevine growth. Biol. Control 2002, 24, 135–142. [Google Scholar] [CrossRef]
- Sawana, A.; Adeolu, M.; Gupta, R.S. Molecular signatures and phylogenomic analysis of the genus Burkholderia: Proposal for division of this genus into the emended genus Burkholderia containing pathogenic organisms and a new genus Paraburkholderia gen. nov. harboring environmental species. Front. Genet. 2014, 5, 429. [Google Scholar] [CrossRef] [Green Version]
- De Vrieze, M.; Pandey, P.; Bucheli, T.D.; Varadarajan, A.R.; Ahrens, C.H.; Weisskopf, L.; Bailly, A. Volatile Organic Compounds from Native Potato-associated Pseudomonas as Potential Anti-oomycete Agents. Front. Microbiol. 2015, 6, 1295. [Google Scholar] [CrossRef] [Green Version]
- Spadaro, D.; Droby, S. Development of biocontrol products for postharvest diseases of fruit: The importance of elucidating the mechanisms of action of yeast antagonists. Trends Food Sci. Technol. 2016, 47, 39–49. [Google Scholar] [CrossRef]
- Lima, G.; De Curtis, F.; Piedimonte, D.; Spina, A.M.; De Cicco, V. Activity of antagonists and natural compounds against powdery mildew of cucurbits: Laboratory and field trials. J. Plant Pathol. 2002, 84, 185. [Google Scholar]
- De Curtis, F.; De Cicco, V.; Lima, G. Efficacy of biocontrol yeasts combined with calcium silicate or sulphur for controlling durum wheat powdery mildew and increasing grain yield components. Field Crops Res. 2012, 134, 36–46. [Google Scholar] [CrossRef]
- De Curtis, F.; Ianiri, G.; Raiola, A.; Ritieni, A.; Succi, M.; Tremonte, P.; Castoria, R. Integration of biological and chemical control of brown rot of stone fruits to reduce disease incidence on fruits and minimize fungicide residues in juice. Crop Prot. 2019, 119, 158–165. [Google Scholar] [CrossRef]
- Stone, B.W.G.; Weingarten, E.A.; Jackson, C.R. The Role of the Phyllosphere Microbiome in Plant Health and Function. Annu. Plant Rev. Online 2018, 1, 533–556. [Google Scholar]
- Ritpitakphong, U.; Falquet, L.; Vimoltust, A.; Berger, A.; Métraux, J.-P.; L’Haridon, F. The microbiome of the leaf surface of Arabidopsis protects against a fungal pathogen. New Phytol. 2016, 210, 1033–1043. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmidt, C.S.; Mrnka, L.; Lovecká, P.; Frantík, T.; Fenclová, M.; Demnerová, K.; Vosátka, M. Bacterial and fungal endophyte communities in healthy and diseased oilseed rape and their potential for biocontrol of Sclerotinia and Phoma disease. Sci. Rep. 2021, 11, 3810. [Google Scholar] [CrossRef]
- Wisniewski, M.; Wilson, C.L. Biological control of postharvest diseases of fruits and vegetables: Recent advances. Hortscience 1992, 27, 94–98. [Google Scholar] [CrossRef] [Green Version]
- Roberts, R.G. Integrating biological control into postharvest disease management strategies. Hortscience 1994, 29, 758–762. [Google Scholar] [CrossRef]
- Janisiewicz, W.J.; Jeffers, S.N. Efficacy of commercial formulation of two biofungicides for control of blue mold and gray mold of apples in cold storage. Crop Prot. 1997, 16, 629–633. [Google Scholar] [CrossRef]
- Obagwu, J.; Korsten, L. Integrated control of citrus green and blue molds using Bacillus subtilis in combination with sodium bicarbonate or hot water. Postharvest Biol. Technol. 2003, 28, 187–194. [Google Scholar] [CrossRef]
- Stéphane, C.; Brion, D.; Jerzy, N.; Christophe, C.; Ait, B.E. Use of Plant Growth-Promoting Bacteria for Biocontrol of Plant Diseases: Principles, Mechanisms of Action, and Future Prospects. Appl. Environ. Microbiol. 2005, 71, 4951–4959. [Google Scholar] [CrossRef] [Green Version]
- Dukare, A.S.; Paul, S.; Nambi, V.E.; Gupta, R.K.; Singh, R.; Sharma, K.; Vishwakarma, R.K. Exploitation of microbial antagonists for the control of postharvest diseases of fruits: A review. Crit. Rev. Food Sci. Nutr. 2019, 59, 1498–1513. [Google Scholar] [CrossRef]
- Pimenta, R.S.; Morais, P.B.; Rosa, C.A.; Corrêa, A. Utilization of Yeasts in Biological Control Programs. In Yeast Biotechnology: Diversity and Applications; Satyanarayana, T., Kunze, G., Eds.; Springer: Dordrecht, The Netherlands, 2009; pp. 199–214. ISBN 978-1-4020-8292-4. [Google Scholar]
- Raaijmakers, J.M.; Vlami, M.; de Souza, J.T. Antibiotic production by bacterial biocontrol agents. Antonie Van Leeuwenhoek 2002, 81, 537–547. [Google Scholar] [CrossRef] [PubMed]
- Lachance, M.-A. Yeast Biodiversity: How Many and How Much? In Biodiversity and Ecophysiology of Yeasts; Péter, G., Rosa, C., Eds.; Springer: Berlin/Heidelberg, Germany, 2006; pp. 1–9. ISBN 978-3-540-30985-7. [Google Scholar]
- Shanmuganathan, N. Yeasts as a Biocontrol for Microbial Diseases of Fruit. U.S. Patent No. 5,525,132, 11 June 1996. [Google Scholar]
- Wilson, C.; Ghaouth, A.E. Composition containing 2-deoxy-D-glucose and Candida saitoana and a method of use for the biological control of postharvest diseases. U.S. Patent No. 5,591,429, 7 January 1997. [Google Scholar]
- Lima, G.; Ippolito, A.; Nigro, F.; Romanazzi, G.; Schena, L.; Gatto, M.; Salerno, M. Lotta biologica contro marciumi postraccolta di uva da tavola, fragola e actinidia con Aureobasidium pullulans e Candida oleophila. Inf. Agrar. 1996, 45, 79–84. [Google Scholar]
- Lima, G.; Ippolito, A.; Nigro, F.; Salerno, M. Effectiveness of Aureobasidium pullulans and Candida oleophila against postharvest strawberry rots. Postharvest Biol. Technol. 1997, 10, 169–178. [Google Scholar] [CrossRef]
- Weiss, A.; Mögel, G.; Kunz, S. Development of “Boni-Pro-tect”-a Yeast Preparation For Use In the Control of Post-harvest Diseases of Apples. In Proceedings of the 12th International Conference on Cultivation Technique and Phytopathological Problems in Organic Fruit-Growing, Weinsberg, Germany, 31 January–2 February 2006. [Google Scholar]
- Spadaro, D.; Gullino, M.L. State of the art and future prospects of the biological control of postharvest fruit diseases. Int. J. Food Microbiol. 2004, 91, 185–194. [Google Scholar] [CrossRef]
- Lima, G.; Spina, A.M.; Castoria, R.; De Curtis, F.; De Cicco, V. Integration of Biocontrol Agents and Food-Grade Additives for Enhancing Protection of Stored Apples from Penicillium expansum. J. Food Prot. 2005, 68, 2100–2106. [Google Scholar] [CrossRef]
- Lima, G.; De Curtis, F.; De Cicco, V. Interaction of microbial biocontrol agents and fungicides in the control of postharvest disease. Stewart Postharvest Rev. 2008, 4, 1–7. [Google Scholar] [CrossRef]
- Cañamás, T.P.; Viñas, I.; Torres, R.; Usall, J.; Solsona, C.; Teixidó, N. Field applications of improved formulations of Candida sake CPA-1 for control of Botrytis cinerea in grapes. Biol. Control 2011, 56, 150–158. [Google Scholar] [CrossRef]
- Calvo-Garrido, C.; Viñas, I.; Elmer, P.; Usall, J.; Teixidó, N. Candida sake CPA-1 and other biologically based products as potential control strategies to reduce sour rot of grapes. Lett. Appl. Microbiol. 2013, 57, 356–361. [Google Scholar] [CrossRef]
- Zhang, H.; Mahunu, G.K.; Castoria, R.; Yang, Q.; Apaliya, M.T. Recent developments in the enhancement of some postharvest biocontrol agents with unconventional chemicals compounds. Trends Food Sci. Technol. 2018, 78, 180–187. [Google Scholar] [CrossRef]
- Castoria, R.; Wright, S.A.I.; Droby, S. Biological Control of Mycotoxigenic Fungi in Fruits. In Mycotoxins in Fruits and Vegetables; Barkai-Golan, R., Paster, N., Eds.; Academic Press: San Diego, CA, USA, 2008; Chapter 16; pp. 311–333. ISBN 978-0-12-374126-4. [Google Scholar]
- Zheng, X.; Yang, Q.; Zhang, X.; Apaliya, M.T.; Ianiri, G.; Zhang, H.; Castoria, R. Biocontrol Agents Increase the Specific Rate of Patulin Production by Penicillium expansum but Decrease the Disease and Total Patulin Contamination of Apples. Front. Microbiol. 2017, 8, 1240. [Google Scholar] [CrossRef]
- Lima, G.; Castoria, R.; De Curtis, F.; Raiola, A.; Ritieni, A.; De Cicco, V. Integrated control of blue mould using new fungicides and biocontrol yeasts lowers levels of fungicide residues and patulin contamination in apples. Postharvest Biol. Technol. 2011, 60, 164–172. [Google Scholar] [CrossRef]
- Castoria, R.; Morena, V.; Caputo, L.; Panfili, G.; De Curtis, F.; De Cicco, V. Effect of the Biocontrol Yeast Rhodotorula glutinis Strain LS11 on Patulin Accumulation in Stored Apples. Phytopathology 2005, 95, 1271–1278. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Castoria, R.; Mannina, L.; Durán-Patrón, R.; Maffei, F.; Sobolev, A.P.; De Felice, D.V.; Pinedo-Rivilla, C.; Ritieni, A.; Ferracane, R.; Wright, S.A.I. Conversion of the Mycotoxin Patulin to the Less Toxic Desoxypatulinic Acid by the Biocontrol Yeast Rhodosporidium kratochvilovae Strain LS11. J. Agric. Food Chem. 2011, 59, 11571–11578. [Google Scholar] [CrossRef] [Green Version]
- Ianiri, G.; Idnurm, A.; Castoria, R. Transcriptomic responses of the basidiomycete yeast Sporobolomyces sp. to the mycotoxin patulin. BMC Genom. 2016, 17, 210. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.; Bao, Y.; Shen, D.; Feng, W.; Yu, T.; Zhang, J.; Zheng, X.D. Biocontrol of Alternaria alternata on cherry tomato fruit by use of marine yeast Rhodosporidium paludigenum Fell & Tallman. Int. J. Food Microbiol. 2008, 123, 234–239. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, P.; Xia, J.; Yu, T.; Lou, B.; Wang, J.; Zheng, X.D. Effect of water activity on stress tolerance and biocontrol activity in antagonistic yeast Rhodosporidium paludigenum. Int. J. Food Microbiol. 2010, 143, 103–108. [Google Scholar] [CrossRef]
- Zhu, R.; Yu, T.; Guo, S.; Hu, H.A.O.; Zheng, X.; Karlovsky, P. Effect of the Yeast Rhodosporidium paludigenum on Postharvest Decay and Patulin Accumulation in Apples and Pears. J. Food Prot. 2015, 78, 157–163. [Google Scholar] [CrossRef]
- Liu, M.; Zhang, W.; Yao, J.; Niu, J. Production, purification, characterization, and biological properties of Rhodosporidium paludigenum polysaccharide. PLoS ONE 2021, 16, e0246148. [Google Scholar] [CrossRef]
- Wei, M.; Dhanasekaran, S.; Godana, E.A.; Yang, Q.; Sui, Y.; Zhang, X.; Ngolong Ngea, G.L.; Zhang, H. Whole-genome sequencing of Cryptococcus podzolicus Y3 and data-independent acquisition-based proteomic analysis during OTA degradation. Food Control 2022, 136, 108862. [Google Scholar] [CrossRef]
- Ngolong Ngea, G.L.; Yang, Q.; Castoria, R.; Zhang, X.; Routledge, M.N.; Zhang, H. Recent trends in detecting, controlling, and detoxifying of patulin mycotoxin using biotechnology methods. Compr. Rev. Food Sci. Food Saf. 2020, 19, 2447–2472. [Google Scholar] [CrossRef]
- Vero, S.; Garmendia, G.; González, M.B.; Bentancur, O.; Wisniewski, M. Evaluation of yeasts obtained from Antarctic soil samples as biocontrol agents for the management of postharvest diseases of apple (Malus × domestica). FEMS Yeast Res. 2013, 13, 189–199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, H.; Wisniewski, M.E.; Abdelfattah, A.; Zheng, X. Biocontrol activity of a cold-adapted yeast from Tibet against gray mold in cherry tomato and its action mechanism. Extremophiles 2017, 21, 789–803. [Google Scholar] [CrossRef] [PubMed]
- De García, V.; Brizzio, S.; Libkind, D.; Buzzini, P.; Van Broock, M. Biodiversity of cold-adapted yeasts from glacial meltwater rivers in Patagonia, Argentina. FEMS Microbiol. Ecol. 2007, 59, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Janisiewicz, W.J.; Tworkoski, T.J.; Kurtzman, C.P. Biocontrol Potential of Metchnikowia pulcherrima Strains against Blue Mold of Apple. Phytopathology 2001, 91, 1098–1108. [Google Scholar] [CrossRef] [Green Version]
- Kurtzman, C.P.; Droby, S. Metschnikowia fructicola, a New Ascosporic Yeast with Potential for Biocontrol of Postharvest Fruit Rots. Syst. Appl. Microbiol. 2001, 24, 395–399. [Google Scholar] [CrossRef] [Green Version]
- Piombo, E.; Sela, N.; Wisniewski, M.; Hoffmann, M.; Gullino, M.L.; Allard, M.W.; Levin, E.; Spadaro, D.; Droby, S. Genome Sequence, Assembly and Characterization of Two Metschnikowia fructicola Strains Used as Biocontrol Agents of Postharvest Diseases. Front. Microbiol. 2018, 9, 593. [Google Scholar] [CrossRef] [Green Version]
- Pereyra, M.M.; Díaz, M.A.; Soliz-Santander, F.F.; Poehlein, A.; Meinhardt, F.; Daniel, R.; Dib, J.R. Screening Methods for Isolation of Biocontrol Epiphytic Yeasts against Penicillium digitatum in Lemons. J. Fungi 2021, 7, 166. [Google Scholar] [CrossRef]
- Lima, G.; De Curtis, F.; Castoria, R.; De Cicco, V. Activity of the Yeasts Cryptococcus laurentii and Rhodotorula glutinis against Post-harvest Rots on Different Fruits. Biocontrol Sci. Technol. 1998, 8, 257–267. [Google Scholar] [CrossRef]
- Palmieri, D.; Barone, G.; Cigliano, R.A.; De Curtis, F.; Lima, G.; Castoria, R.; Ianiri, G. Complete genome sequence of the biocontrol yeast Papiliotrema terrestris strain LS28. G3 Genes|Genomes|Genetics 2021, 11, jkab332. [Google Scholar] [CrossRef]
- Lahlali, R.; Ezrari, S.; Radouane, N.; Kenfaoui, J.; Esmaeel, Q.; El Hamss, H.; Belabess, Z.; Barka, E.A. Biological Control of Plant Pathogens: A Global Perspective. Microorganisms 2022, 10, 596. [Google Scholar] [CrossRef]
- Corke, A.T.K. Biological Control of Plant Pathogens. By Kenneth F. Baker and R. James Cook. San Francisco: Freeman (1974), pp. 433, £5.90. Exp. Agric. 1975, 11, 159. [Google Scholar] [CrossRef]
- Raymaekers, K.; Ponet, L.; Holtappels, D.; Berckmans, B.; Cammue, B.P.A. Screening for novel biocontrol agents applicable in plant disease management—A review. Biol. Control 2020, 144, 104240. [Google Scholar] [CrossRef]
- Benl tez, T.A.; Rincón, A.M.; Limón, M.C.; Codón, A.C. Biocontrol mechanisms of Trichoderma strains. Int. Microbiol. 2004, 7, 249–260. [Google Scholar]
- Huang, H.C.; Erickson, R.S. Overwintering of Coniothyrium minkans, a mycoparasite of Sclerotinia sclerotiorum, on the Canadian Prairies. Australas. Plant Pathol. 2011, 31, 291–293. [Google Scholar] [CrossRef]
- Tu, J.C. Hyperparasitism of Streptomyces albus on a Destructive Mycoparasite Nectria inventa. J. Phytopathol. 1986, 117, 71–76. [Google Scholar] [CrossRef]
- Nofal, A.M.; El-Rahman, M.A.; Abdelghany, T.M.; Abd El-Mongy, M. Mycoparasitic nature of Egyptian Trichoderma isolates and their impact on suppression Fusarium wilt of tomato. Egypt. J. Biol. Pest Control 2021, 31, 103. [Google Scholar] [CrossRef]
- Moreno-Ruiz, D.; Lichius, A.; Turrà, D.; Di Pietro, A.; Zeilinger, S. Chemotropism Assays for Plant Symbiosis and Mycoparasitism Related Compound Screening in Trichoderma atroviride. Front. Microbiol. 2020, 11, 3006. [Google Scholar] [CrossRef] [PubMed]
- Horner, N.R.; Grenville-Briggs, L.J.; van West, P. The oomycete Pythium oligandrum expresses putative effectors during mycoparasitism of Phytophthora infestans and is amenable to transformation. Fungal Biol. 2012, 116, 24–41. [Google Scholar] [CrossRef] [Green Version]
- Deacon, J.W.; Henry, C.M. Methods used to study Pythium oligandrum, an aggressive parasite of other fungi. Ann. Appl. Biol. 1978, 89, 141–142. [Google Scholar] [CrossRef]
- Heydari, A.; Pessarakli, M. A review on biological control of fungal plant pathogens using microbial antagonists. J. Biol. Sci. 2010, 10, 273–290. [Google Scholar] [CrossRef] [Green Version]
- Tapio, E.; Pohto-Lahdenperä, A. Scanning electron microscopy of hyphal interaction between Streptomyces griseoviridis and some plant pathogenic fungi. Agric. Food Sci. 1991, 63, 435–441. [Google Scholar] [CrossRef]
- Ziedan, E.-S.H.; Farrag, E.S.; El-Mohamedy, R.S.; Abd Alla, M.A. Streptomyces alni as a biocontrol agent to root-rot of grapevine and increasing their efficiency by biofertilisers inocula. Arch. Phytopathol. Plant Prot. 2010, 43, 634–646. [Google Scholar] [CrossRef]
- Trunk, K.; Peltier, J.; Liu, Y.-C.; Dill, B.D.; Walker, L.; Gow, N.A.R.; Stark, M.J.R.; Quinn, J.; Strahl, H.; Trost, M.; et al. The type VI secretion system deploys antifungal effectors against microbial competitors. Nat. Microbiol. 2018, 3, 920–931. [Google Scholar] [CrossRef]
- Pal, K.K.; McSpadden Gardener, B. Biological Control of Plant Pathogens. Plant Health Instr. 2006, 1–25. [Google Scholar] [CrossRef] [Green Version]
- Benhamou, N.; Chet, I. Cellular and Molecular Mechanisms Involved in the Interaction between Trichoderma harzianum and Pythium ultimum. Appl. Environ. Microbiol. 1997, 63, 2095–2099. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hutchings, M.I.; Truman, A.W.; Wilkinson, B. Antibiotics: Past, present and future. Curr. Opin. Microbiol. 2019, 51, 72–80. [Google Scholar] [CrossRef]
- Di Francesco, A.; Martini, C.; Mari, M. Biological control of postharvest diseases by microbial antagonists: How many mechanisms of action? Eur. J. Plant Pathol. 2016, 145, 711–717. [Google Scholar] [CrossRef]
- Nunes, C.A. Biological control of postharvest diseases of fruit. Eur. J. Plant Pathol. 2012, 133, 181–196. [Google Scholar] [CrossRef]
- Raaijmakers, J.M.; Paulitz, T.C.; Steinberg, C.; Alabouvette, C.; Moënne-Loccoz, Y. The rhizosphere: A playground and battlefield for soilborne pathogens and beneficial microorganisms. Plant Soil 2009, 321, 341–361. [Google Scholar] [CrossRef] [Green Version]
- Hossain, M.T.; Khan, A.; Chung, E.J.; Rashid, M.H.-O.; Chung, Y.R. Biological Control of Rice Bakanae by an Endophytic Bacillus oryzicola YC7007. Plant Pathol. J. 2016, 32, 228–241. [Google Scholar] [CrossRef] [Green Version]
- Meziane, H.; Gavriel, S.; Ismailov, Z.; Chet, I.; Chernin, L.; Höfte, M. Control of green and blue mould on orange fruit by Serratia plymuthica strains IC14 and IC1270 and putative modes of action. Postharvest Biol. Technol. 2006, 39, 125–133. [Google Scholar] [CrossRef]
- Pretorius, D.; van Rooyen, J.; Clarke, K.G. Enhanced production of antifungal lipopeptides by Bacillus amyloliquefaciens for biocontrol of postharvest disease. New Biotechnol. 2015, 32, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Haas, D.; Keel, C. Regulation of antibiotic production in root-colonizing Pseudomonas spp. and relevance for biological control of plant disease. Annu. Rev. Phytopathol. 2003, 41, 117–153. [Google Scholar] [CrossRef] [PubMed]
- Brescia, F.; Vlassi, A.; Bejarano, A.; Seidl, B.; Marchetti-Deschmann, M.; Schuhmacher, R.; Puopolo, G. Characterisation of the Antibiotic Profile of Lysobacter capsici AZ78, an Effective Biological Control Agent of Plant Pathogenic Microorganisms. Microorganisms 2021, 9, 1320. [Google Scholar] [CrossRef] [PubMed]
- Poppe, L.; Vanhoutte, S.; Höfte, M. Modes of Action of Pantoea agglomerans CPA-2, an Antagonist of Postharvest Pathogens on Fruits. Eur. J. Plant Pathol. 2003, 109, 963–973. [Google Scholar] [CrossRef]
- Pacios-Michelena, S.; Aguilar González, C.N.; Alvarez-Perez, O.B.; Rodriguez-Herrera, R.; Chávez-González, M.; Arredondo Valdés, R.; Ascacio Valdés, J.A.; Govea Salas, M.; Ilyina, A. Application of Streptomyces Antimicrobial Compounds for the Control of Phytopathogens. Front. Sustain. Food Syst. 2021, 5, 1–13. [Google Scholar] [CrossRef]
- Verma, M.; Brar, S.K.; Tyagi, R.D.; Surampalli, R.Y.; Valéro, J.R. Antagonistic fungi, Trichoderma spp.: Panoply of biological control. Biochem. Eng. J. 2007, 37, 1–20. [Google Scholar] [CrossRef]
- Harman, G.E.; Howell, C.R.; Viterbo, A.; Chet, I.; Lorito, M. Trichoderma species—Opportunistic, avirulent plant symbionts. Nat. Rev. Microbiol. 2004, 2, 43–56. [Google Scholar] [CrossRef]
- Choińska, R.; Piasecka-Jóźwiak, K.; Chabłowska, B.; Dumka, J.; Łukaszewicz, A. Biocontrol ability and volatile organic compounds production as a putative mode of action of yeast strains isolated from organic grapes and rye grains. Antonie Van Leeuwenhoek 2020, 113, 1135–1146. [Google Scholar] [CrossRef]
- Song, G.C.; Ryu, C.-M. Two Volatile Organic Compounds Trigger Plant Self-Defense against a Bacterial Pathogen and a Sucking Insect in Cucumber under Open Field Conditions. Int. J. Mol. Sci. 2013, 14, 9803–9819. [Google Scholar] [CrossRef] [Green Version]
- Bevan, E.A.; Makower, M. The physiological basis of the killer character in yeast. In Proceedings of the International Congress of Genetics, The Hague, The Netherland, 2–10 September 1963; Pergamon Press: Oxford, UK, 1963; Volume 1, pp. 202–203. [Google Scholar]
- Farkas, Z.; Márki-Zay, J.; Kucsera, J.; Vágvölgyi, C.; Golubev, W.; Pfeiffer, I. Characterization of two different toxins of Wickerhamomyces anomalus (pichia anomala) VKM Y-159. Acta Biol. Hung. 2012, 63, 277–287. [Google Scholar] [CrossRef] [PubMed]
- de Lima, J.R.; Gonçalves, L.R.B.; Brandão, L.R.; Rosa, C.A.; Viana, F.M.P. Isolation, identification, and activity in vitro of killer yeasts against Colletotrichum gloeosporioides isolated from tropical fruits. J. Basic Microbiol. 2013, 53, 590–599. [Google Scholar] [CrossRef] [PubMed]
- Giovati, L.; Ciociola, T.; De Simone, T.; Conti, S.; Magliani, W. Wickerhamomyces Yeast Killer Toxins’ Medical Applications. Toxins 2021, 13, 655. [Google Scholar] [CrossRef]
- Schmitt, M.J.; Breinig, F. The viral killer system in yeast: From molecular biology to application. FEMS Microbiol. Rev. 2002, 26, 257–276. [Google Scholar] [CrossRef]
- Grzegorczyk, M.; Żarowska, B.; Restuccia, C.; Cirvilleri, G. Postharvest biocontrol ability of killer yeasts against Monilinia fructigena and Monilinia fructicola on stone fruit. Food Microbiol. 2017, 61, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Fredericks, L.R.; Lee, M.D.; Crabtree, A.M.; Boyer, J.M.; Kizer, E.A.; Taggart, N.T.; Roslund, C.R.; Hunter, S.S.; Kennedy, C.B.; Willmore, C.G.; et al. The Species-Specific Acquisition and Diversification of a K1-like Family of Killer Toxins in Budding Yeasts of the Saccharomycotina. PLOS Genet. 2021, 17, e1009341. [Google Scholar] [CrossRef]
- Brown, D.W. The KP4 killer protein gene family. Curr. Genet. 2011, 57, 51–62. [Google Scholar] [CrossRef]
- Schmitt, M.J.; Breinig, F. Yeast viral killer toxins: Lethality and self-protection. Nat. Rev. Microbiol. 2006, 4, 212–221. [Google Scholar] [CrossRef]
- Walker, G.M. Pichia anomala: Cell physiology and biotechnology relative to other yeasts. Antonie Van Leeuwenhoek 2011, 99, 25–34. [Google Scholar] [CrossRef] [Green Version]
- Moura, V.S.; Pollettini, F.L.; Ferraz, L.P.; Mazzi, M.V.; Kupper, K.C. Purification of a killer toxin from Aureobasidium pullulans for the biocontrol of phytopathogens. J. Basic Microbiol. 2021, 61, 77–87. [Google Scholar] [CrossRef]
- Ghoul, M.; Mitri, S. The Ecology and Evolution of Microbial Competition. Trends Microbiol. 2016, 24, 833–845. [Google Scholar] [CrossRef] [PubMed]
- Turrà, D.; Di Pietro, A. Chemotropic sensing in fungus–plant interactions. Curr. Opin. Plant Biol. 2015, 26, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Bais, H.P.; Weir, T.L.; Perry, L.G.; Gilroy, S.; Vivanco, J.M. The Role of Root Exudates in Rhizosphere Interactions with Plants and Other Organisms. Annu. Rev. Plant Biol. 2006, 57, 233–266. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, J.; Sui, Y.; Wisniewski, M.; Droby, S.; Liu, Y. Review: Utilization of antagonistic yeasts to manage postharvest fungal diseases of fruit. Int. J. Food Microbiol. 2013, 167, 153–160. [Google Scholar] [CrossRef]
- Castoria, R.; Caputo, L.; De Curtis, F.; De Cicco, V. Resistance of Postharvest Biocontrol Yeasts to Oxidative Stress: A Possible New Mechanism of Action. Phytopathology 2003, 93, 564–572. [Google Scholar] [CrossRef] [Green Version]
- Castoria, R.; Miccoli, C.; Barone, G.; Palmieri, D.; De Curtis, F.; Lima, G.; Heitman, J.; Ianiri, G. Molecular Tools for the Yeast Papiliotrema terrestris LS28 and Identification of Yap1 as a Transcription Factor Involved in Biocontrol Activity. Appl. Environ. Microbiol. 2021, 87, e02910-20. [Google Scholar] [CrossRef]
- Liu, J.; Wisniewski, M.; Droby, S.; Norelli, J.; Hershkovitz, V.; Tian, S.; Farrell, R. Increase in antioxidant gene transcripts, stress tolerance and biocontrol efficacy of Candida oleophila following sublethal oxidative stress exposure. FEMS Microbiol. Ecol. 2012, 80, 578–590. [Google Scholar] [CrossRef]
- Filonow, A.B. Role of Competition for Sugars by Yeasts in the Biocontrol of Gray Mold of Apple. Biocontrol Sci. Technol. 1998, 8, 243–256. [Google Scholar] [CrossRef]
- van Loon, L.C. Helping Plants To Defend Themselves: Biocontrol By Disease-Suppressing Rhizobacteria. In Highlights in European Plant Biotechnology Research and Technology Transfer; de Vries, G.E., Metzlaff, K., Eds.; Elsevier: Amsterdam, The Netherlands, 2000; Volume 6, pp. 203–213. ISBN 0168-7972. [Google Scholar]
- Segarra, G.; Casanova, E.; Avilés, M.; Trillas, I. Trichoderma asperellum Strain T34 Controls Fusarium Wilt Disease in Tomato Plants in Soilless Culture through Competition for Iron. Microb. Ecol. 2010, 59, 141–149. [Google Scholar] [CrossRef]
- Saravanakumar, D.; Ciavorella, A.; Spadaro, D.; Garibaldi, A.; Gullino, M.L. Metschnikowia pulcherrima strain MACH1 outcompetes Botrytis cinerea, Alternaria alternata and Penicillium expansum in apples through iron depletion. Postharvest Biol. Technol. 2008, 49, 121–128. [Google Scholar] [CrossRef]
- Calvente, V.; Benuzzi, D.; de Tosetti, M.I.S. Antagonistic action of siderophores from Rhodotorula glutinis upon the postharvest pathogen Penicillium expansum. Int. Biodeterior. Biodegrad. 1999, 43, 167–172. [Google Scholar] [CrossRef]
- Di Francesco, A.; Baraldi, E. How siderophore production can influence the biocontrol activity of Aureobasidium pullulans against Monilinia laxa on peaches. Biol. Control 2021, 152, 104456. [Google Scholar] [CrossRef]
- Oberegger, H.; Schoeser, M.; Zadra, I.; Abt, B.; Haas, H. SREA is involved in regulation of siderophore biosynthesis, utilization and uptake in Aspergillus nidulans. Mol. Microbiol. 2001, 41, 1077–1089. [Google Scholar] [CrossRef]
- Klein, M.N.; Kupper, K.C. Biofilm production by Aureobasidium pullulans improves biocontrol against sour rot in citrus. Food Microbiol. 2018, 69, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Castoria, R.; De Curtis, F.; Lima, G.; De Cicco, V. β-1,3-glucanase activity of two saprophytic yeasts and possible mode of action as biocontrol agents against postharvest diseases. Postharvest Biol. Technol. 1997, 12, 293–300. [Google Scholar] [CrossRef]
- Di Francesco, A.; Ugolini, L.; D’Aquino, S.; Pagnotta, E.; Mari, M. Biocontrol of Monilinia laxa by Aureobasidium pullulans strains: Insights on competition for nutrients and space. Int. J. Food Microbiol. 2017, 248, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Sangiorgio, D.; Cellini, A.; Donati, I.; Pastore, C.; Onofrietti, C.; Spinelli, F. Facing Climate Change: Application of Microbial Biostimulants to Mitigate Stress in Horticultural Crops. Agronomy 2020, 10, 794. [Google Scholar] [CrossRef]
- Kerry, B.R. Rhizosphere Interactions and the Exploitation of Microbial Agents for the Biological Control of Plant-Parasitic Nematodes. Annu. Rev. Phytopathol. 2000, 38, 423–441. [Google Scholar] [CrossRef] [Green Version]
- Ramamoorthy, V.; Viswanathan, R.; Raguchander, T.; Prakasam, V.; Samiyappan, R. Induction of systemic resistance by plant growth promoting rhizobacteria in crop plants against pests and diseases. Crop Prot. 2001, 20, 1–11. [Google Scholar] [CrossRef]
- Khanna, K.; Kohli, S.K.; Ohri, P.; Bhardwaj, R. Plants-nematodes-microbes crosstalk within soil: A trade-off among friends or foes. Microbiol. Res. 2021, 248, 126755. [Google Scholar] [CrossRef]
- Bhavanam, S.; Stout, M.J. Assessment of Silicon-and Mycorrhizae-Mediated Constitutive and Induced Systemic Resistance in Rice, Oryza sativa L., against the Fall Armyworm, Spodoptera frugiperda Smith. Plants 2021, 10, 2126. [Google Scholar] [CrossRef] [PubMed]
- Beneduzi, A.; Ambrosini, A.; Passaglia, L.M.P. Plant growth-promoting rhizobacteria (PGPR): Their potential as antagonists and biocontrol agents. Genet. Mol. Biol. 2012, 35, 1044–1051. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Conrath, U.; Beckers, G.J.M.; Langenbach, C.J.G.; Jaskiewicz, M.R. Priming for Enhanced Defense. Annu. Rev. Phytopathol. 2015, 53, 97–119. [Google Scholar] [CrossRef] [PubMed]
- van Loon, L.C.; Bakker, P.A.H.M.; Pieterse, C.M.J. Systemic resistance induced by rhizosphere bacteria. Annu. Rev. Phytopathol. 1998, 36, 453–483. [Google Scholar] [CrossRef] [Green Version]
- Yariv, B.; Arik, M.; Yechiel, S.; Ilan, C.; Ada, V. Synthetic Ultrashort Cationic Lipopeptides Induce Systemic Plant Defense Responses against Bacterial and Fungal Pathogens. Appl. Environ. Microbiol. 2009, 75, 5373–5379. [Google Scholar] [CrossRef] [Green Version]
- Kuć, J. Concepts and Direction of Induced Systemic Resistance in Plants and its Application. Eur. J. Plant Pathol. 2001, 107, 7–12. [Google Scholar] [CrossRef]
- van Peer, R.; Niemann, G.J.; Schippers, B. Induced resistance and phytoalexin accumulation in biological control of Fusarium wilt of carnation by Pseudomonas sp. strain WCS417r. Phytopathology 1991, 81, 728–734. [Google Scholar] [CrossRef]
- Wei, G. Induction of systemic resistance of cucumber to Colletotrichum orbiculare by select strains of plant growth-promoting rhizobacteria. Phytopathology 1991, 81, 1508–1512. [Google Scholar] [CrossRef]
- Barka, E.A.; Belarbi, A.; Hachet, C.; Nowak, J.; Audran, J.-C. Enhancement of in vitro growth and resistance to gray mould of Vitis vinifera co-cultured with plant growth-promoting rhizobacteria. FEMS Microbiol. Lett. 2000, 186, 91–95. [Google Scholar] [CrossRef] [Green Version]
- Lahoz, E.; Contillo, R.; Porrone, F. Induction of Systemic Resistance to Erysiphe orontii Cast in Tobacco by Application on Roots of an Isolate of Gliocladium roseum Bainier. J. Phytopathol. 2004, 152, 465–470. [Google Scholar] [CrossRef]
- Brooks, D.S.; Gonzalez, C.F.; Appel, D.N.; Filer, T.H. Evaluation of Endophytic Bacteria as Potential Biological-Control Agents for Oak Wilt. Biol. Control 1994, 4, 373–381. [Google Scholar] [CrossRef]
- Khan, N.; Martínez-Hidalgo, P.; Ice, T.A.; Maymon, M.; Humm, E.A.; Nejat, N.; Sanders, E.R.; Kaplan, D.; Hirsch, A.M. Antifungal Activity of Bacillus Species against Fusarium and Analysis of the Potential Mechanisms Used in Biocontrol. Front. Microbiol. 2018, 9, 2363. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agostini, R.B.; Rius, S.P.; Vargas, W.A.; Campos-Bermudez, V.A. Proteome impact on maize silks under the priming state induced by Trichoderma root colonization. Planta 2021, 253, 115. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.; Aggarwal, R.; Bashyal, B.M.; Darshan, K.; Parmar, P.; Saharan, M.S.; Hussain, Z.; Solanke, A.U. Transcriptome Reprogramming of Tomato Orchestrate the Hormone Signaling Network of Systemic Resistance Induced by Chaetomium globosum. Front. Plant Sci. 2021, 12, 721193. [Google Scholar] [CrossRef]
- Roylawar, P.; Khandagale, K.; Randive, P.; Shinde, B.; Murumkar, C.; Ade, A.; Singh, M.; Gawande, S.; Morelli, M. Piriformospora indica Primes Onion Response against Stemphylium Leaf Blight Disease. Pathogen 2021, 10, 1085. [Google Scholar] [CrossRef]
- Shaw, S.; Le Cocq, K.; Paszkiewicz, K.; Moore, K.; Winsbury, R.; de Torres Zabala, M.; Studholme, D.J.; Salmon, D.; Thornton, C.R.; Grant, M.R. Transcriptional reprogramming underpins enhanced plant growth promotion by the biocontrol fungus Trichoderma hamatum GD12 during antagonistic interactions with Sclerotinia sclerotiorum in soil. Mol. Plant Pathol. 2016, 17, 1425–1441. [Google Scholar] [CrossRef] [Green Version]
- Sharma, V.; Salwan, R.; Sharma, P.N.; Gulati, A. Integrated Translatome and Proteome: Approach for Accurate Portraying of Widespread Multifunctional Aspects of Trichoderma. Front. Microbiol. 2017, 8, 1602. [Google Scholar] [CrossRef]
- Zhao, H.; Zhou, T.; Xie, J.; Cheng, J.; Chen, T.; Jiang, D.; Fu, Y. Mycoparasitism illuminated by genome and transcriptome sequencing of Coniothyrium minitans, an important biocontrol fungus of the plant pathogen Sclerotinia sclerotiorum. Microb. Genom. 2020, 6, e000345. [Google Scholar] [CrossRef]
- Darshan, K.; Aggarwal, R.; Bashyal, B.M.; Singh, J.; Shanmugam, V.; Gurjar, M.S.; Solanke, A.U. Transcriptome Profiling Provides Insights Into Potential Antagonistic Mechanisms Involved in Chaetomium globosum against Bipolaris sorokiniana. Front. Microbiol. 2020, 11, 2971. [Google Scholar] [CrossRef]
- Broberg, M.; Dubey, M.; Iqbal, M.; Gudmundssson, M.; Ihrmark, K.; Schroers, H.-J.; Funck Jensen, D.; Brandström Durling, M.; Karlsson, M. Comparative genomics highlights the importance of drug efflux transporters during evolution of mycoparasitism in Clonostachys subgenus Bionectria (Fungi, Ascomycota, Hypocreales). Evol. Appl. 2021, 14, 476–497. [Google Scholar] [CrossRef]
- Westermann, A.J.; Barquist, L.; Vogel, J. Resolving host–pathogen interactions by dual RNA-seq. PLOS Pathog. 2017, 13, e1006033. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Espindula, E.; Sperb, E.R.; Bach, E.; Passaglia, L.M.P. The combined analysis as the best strategy for Dual RNA-Seq mapping. Genet. Mol. Biol. 2020, 42, e20190215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Westermann, A.J.; Gorski, S.A.; Vogel, J. Dual RNA-seq of pathogen and host. Nat. Rev. Microbiol. 2012, 10, 618–630. [Google Scholar] [CrossRef]
- Lysøe, E.; Dees, M.W.; Brurberg, M.B. A Three-Way Transcriptomic Interaction Study of a Biocontrol Agent (Clonostachys rosea), a Fungal Pathogen (Helminthosporium solani), and a Potato Host (Solanum tuberosum). Mol. Plant-Microbe Interact. 2017, 30, 646–655. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hershkovitz, V.; Sela, N.; Taha-Salaime, L.; Liu, J.; Rafael, G.; Kessler, C.; Aly, R.; Levy, M.; Wisniewski, M.; Droby, S. De-novo assembly and characterization of the transcriptome of Metschnikowia fructicola reveals differences in gene expression following interaction with Penicillium digitatumand grapefruit peel. BMC Genom. 2013, 14, 168. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Chen, L.; Sun, Y.; Zhao, L.; Zheng, X.; Yang, Q.; Zhang, X. Investigating Proteome and Transcriptome Defense Response of Apples Induced by Yarrowia lipolytica. Mol. Plant-Microbe Interact. 2017, 30, 301–311. [Google Scholar] [CrossRef] [Green Version]
- Rueda-Mejia, M.P.; Nägeli, L.; Lutz, S.; Hayes, R.D.; Varadarajan, A.R.; Grigoriev, I.V.; Ahrens, C.H.; Freimoser, F.M. Genome, transcriptome and secretome analyses of the antagonistic, yeast-like fungus Aureobasidium pullulans to identify potential biocontrol genes. Microb. Cell 2021, 8, 184–202. [Google Scholar] [CrossRef]
- Laur, J.; Ramakrishnan, G.B.; Labbé, C.; Lefebvre, F.; Spanu, P.D.; Bélanger, R.R. Effectors involved in fungal–fungal interaction lead to a rare phenomenon of hyperbiotrophy in the tritrophic system biocontrol agent–powdery mildew–plant. New Phytol. 2018, 217, 713–725. [Google Scholar] [CrossRef] [Green Version]
- Li, J.-Y.; Gao, T.-T.; Wang, Q. Comparative and Functional Analyses of Two Sequenced Paenibacillus polymyxa Genomes Provides Insights into Their Potential Genes Related to Plant Growth-Promoting Features and Biocontrol Mechanisms. Front. Genet. 2020, 11, 1374. [Google Scholar] [CrossRef]
- Nelkner, J.; Torres Tejerizo, G.; Hassa, J.; Lin, T.W.; Witte, J.; Verwaaijen, B.; Winkler, A.; Bunk, B.; Spröer, C.; Overmann, J.; et al. Genetic Potential of the Biocontrol Agent Pseudomonas brassicacearum (Formerly P. trivialis) 3Re2-7 Unraveled by Genome Sequencing and Mining, Comparative Genomics and Transcriptomics. Genes 2019, 10, 601. [Google Scholar] [CrossRef] [Green Version]
- Hennessy, R.C.; Glaring, M.A.; Olsson, S.; Stougaard, P. Transcriptomic profiling of microbe–microbe interactions reveals the specific response of the biocontrol strain P. fluorescens In5 to the phytopathogen Rhizoctonia solani. BMC Res. Notes 2017, 10, 376. [Google Scholar] [CrossRef] [Green Version]
- Penttilä, M.; Nevalainen, H.; Rättö, M.; Salminen, E.; Knowles, J. A versatile transformation system for the cellulolytic filamentous fungus Trichoderma reesei. Gene 1987, 61, 155–164. [Google Scholar] [CrossRef]
- Woo, S.L.; Scala, F.; Ruocco, M.; Lorito, M. The Molecular Biology of the Interactions between Trichoderma spp., Phytopathogenic Fungi, and Plants. Phytopathology 2006, 96, 181–185. [Google Scholar] [CrossRef] [Green Version]
- Adnan, M.; Islam, W.; Shabbir, A.; Khan, K.A.; Ghramh, H.A.; Huang, Z.; Chen, H.Y.H.; Lu, G. Plant defense against fungal pathogens by antagonistic fungi with Trichoderma in focus. Microb. Pathog. 2019, 129, 7–18. [Google Scholar] [CrossRef]
- Silva, R.N.; Monteiro, V.N.; Steindorff, A.S.; Gomes, E.V.; Noronha, E.F.; Ulhoa, C.J. Trichoderma/pathogen/plant interaction in pre-harvest food security. Fungal Biol. 2019, 123, 565–583. [Google Scholar] [CrossRef]
- Monfil, V.O.; Casas-Flores, S. Molecular Mechanisms of Biocontrol in Trichoderma spp. and Their Applications in Agriculture. In Biotechnology and Biology of Trichoderma; Gupta, V.K., Schmoll, M., Herrera-Estrella, A., Upadhyay, R.S., Druzhinina, I., Tuohy, M.G., Eds.; Elsevier: Amsterdam, The Netherlands, 2014; Chapter 32; pp. 429–453. ISBN 978-0-444-59576-8. [Google Scholar]
- Mukherjee, P.K.; Horwitz, B.A.; Herrera-Estrella, A.; Schmoll, M.; Kenerley, C.M. Trichoderma Research in the Genome Era. Annu. Rev. Phytopathol. 2013, 51, 105–129. [Google Scholar] [CrossRef]
- Zeilinger, S.; Reithner, B.; Scala, V.; Peissl, I.; Lorito, M.; Mach, R.L. Signal transduction by Tga3, a novel G protein alpha subunit of Trichoderma atroviride. Appl. Environ. Microbiol. 2005, 71, 1591–1597. [Google Scholar] [CrossRef] [Green Version]
- Rocha-Ramírez, V.; Omero, C.; Chet, I.; Horwitz, B.A.; Herrera-Estrella, A. Trichoderma atroviride G-Protein α-Subunit Gene tga1 Is Involved in Mycoparasitic Coiling and Conidiation. Eukaryot. Cell 2002, 1, 594–605. [Google Scholar] [CrossRef] [Green Version]
- Omann, M.R.; Lehner, S.; Escobar Rodríguez, C.; Brunner, K.; Zeilinger, S. The seven-transmembrane receptor Gpr1 governs processes relevant for the antagonistic interaction of Trichoderma atroviride with its host. Microbiology 2012, 158, 107–118. [Google Scholar] [CrossRef]
- Ruocco, M.; Lanzuise, S.; Vinale, F.; Marra, R.; Turrà, D.; Woo, S.L.; Lorito, M. Identification of a New Biocontrol Gene in Trichoderma atroviride: The Role of an ABC Transporter Membrane Pump in the Interaction with Different Plant-Pathogenic Fungi. Mol. Plant-Microbe Interact. 2009, 22, 291–301. [Google Scholar] [CrossRef] [Green Version]
- Mukherjee, P.K.; Kenerley, C.M. Regulation of Morphogenesis and Biocontrol Properties in Trichoderma virens by a VELVET Protein, Vel1. Appl. Environ. Microbiol. 2010, 76, 2345–2352. [Google Scholar] [CrossRef] [Green Version]
- Dautt-Castro, M.; Estrada-Rivera, M.; Olguin-Martínez, I.; del Carmen Rocha-Medina, M.; Islas-Osuna, M.A.; Casas-Flores, S. TBRG-1 a Ras-like protein in Trichoderma virens involved in conidiation, development, secondary metabolism, mycoparasitism, and biocontrol unveils a new family of Ras-GTPases. Fungal Genet. Biol. 2020, 136, 103292. [Google Scholar] [CrossRef]
- Nygren, K.; Dubey, M.; Zapparata, A.; Iqbal, M.; Tzelepis, G.D.; Durling, M.B.; Jensen, D.F.; Karlsson, M. The mycoparasitic fungus Clonostachys rosea responds with both common and specific gene expression during interspecific interactions with fungal prey. Evol. Appl. 2018, 11, 931–949. [Google Scholar] [CrossRef] [Green Version]
- Lv, B.; Jiang, N.; Hasan, R.; Chen, Y.; Sun, M.; Li, S. Cell Wall Biogenesis Protein Phosphatase CrSsd1 Is Required for Conidiation, Cell Wall Integrity, and Mycoparasitism in Clonostachys rosea. Front. Microbiol. 2020, 11, 1640. [Google Scholar] [CrossRef]
- Iqbal, M.; Dubey, M.; Broberg, A.; Viketoft, M.; Jensen, D.F.; Karlsson, M. Deletion of the Nonribosomal Peptide Synthetase Gene nps1 in the Fungus Clonostachys rosea Attenuates Antagonism and Biocontrol of Plant Pathogenic Fusarium and Nematodes. Phytopathology 2019, 109, 1698–1709. [Google Scholar] [CrossRef]
- Fatema, U.; Broberg, A.; Jensen, D.F.; Karlsson, M.; Dubey, M. Functional analysis of polyketide synthase genes in the biocontrol fungus Clonostachys rosea. Sci. Rep. 2018, 8, 15009. [Google Scholar] [CrossRef]
- Sun, Z.-B.; Wang, Q.; Sun, M.-H.; Li, S.-D. The Mitogen-Activated Protein Kinase Gene Crmapk Is Involved in Clonostachys chloroleuca Mycoparasitism. Mol. Plant-Microbe Interact. 2020, 33, 902–910. [Google Scholar] [CrossRef]
- Sun, Z.-B.; Sun, M.-H.; Zhou, M.; Li, S.-D. Transformation of the endochitinase gene Chi67-1 in Clonostachys rosea 67-1 increases its biocontrol activity against Sclerotinia sclerotiorum. AMB Express 2017, 7, 1. [Google Scholar] [CrossRef] [Green Version]
- Zeng, F.; Gong, X.; Hamid, M.I.; Fu, Y.; Jiatao, X.; Cheng, J.; Li, G.; Jiang, D. A fungal cell wall integrity-associated MAP kinase cascade in Coniothyrium minitans is required for conidiation and mycoparasitism. Fungal Genet. Biol. 2012, 49, 347–357. [Google Scholar] [CrossRef]
- Daguerre, Y.; Edel-Hermann, V.; Steinberg, C. Fungal Genes and Metabolites Associated with the Biocontrol of Soil-borne Plant Pathogenic Fungi. In Fungal Metabolites; Mérillon, J.-M., Ramawat, K.G., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 33–104. ISBN 978-3-319-25001-4. [Google Scholar]
- Yehuda, H.; Droby, S.; Bar-Shimon, M.; Wisniewski, M.; Goldway, M. The effect of under- and overexpressed CoEXG1-encoded exoglucanase secreted by Candida oleophila on the biocontrol of Penicillium digitatum. Yeast 2003, 20, 771–780. [Google Scholar] [CrossRef]
- Bar-Shimon, M.; Yehuda, H.; Cohen, L.; Weiss, B.; Kobeshnikov, A.; Daus, A.; Goldway, M.; Wisniewski, M.; Droby, S. Characterization of extracellular lytic enzymes produced by the yeast biocontrol agent Candida oleophila. Curr. Genet. 2004, 45, 140–148. [Google Scholar] [CrossRef]
- Grevesse, C.; Lepoivre, P.; Jijakli, M.H. Characterization of the Exoglucanase-Encoding Gene PaEXG2 and Study of Its Role in the Biocontrol Activity of Pichia anomala Strain K. Phytopathology 2003, 93, 1145–1152. [Google Scholar] [CrossRef] [Green Version]
- Friel, D.; Pessoa, N.M.G.; Vandenbol, M.; Jijakli, M.H. Separate and Combined Disruptions of Two Exo-β-1,3-Glucanase Genes Decrease the Efficiency of Pichia anomala (Strain K) Biocontrol against Botrytis cinerea on Apple. Mol. Plant-Microbe Interact. 2007, 20, 371–379. [Google Scholar] [CrossRef] [Green Version]
- Fiori, S.; Fadda, A.; Giobbe, S.; Berardi, E.; Migheli, Q. Pichia angusta is an effective biocontrol yeast against postharvest decay of apple fruit caused by Botrytis cinerea and Monilia fructicola. FEMS Yeast Res. 2008, 8, 961–963. [Google Scholar] [CrossRef] [Green Version]
- Gore-Lloyd, D.; Sumann, I.; Brachmann, A.O.; Schneeberger, K.; Ortiz-Merino, R.A.; Moreno-Beltrán, M.; Schläfli, M.; Kirner, P.; Santos Kron, A.; Rueda-Mejia, M.P.; et al. Snf2 controls pulcherriminic acid biosynthesis and antifungal activity of the biocontrol yeast Metschnikowia pulcherrima. Mol. Microbiol. 2019, 112, 317–332. [Google Scholar] [CrossRef] [Green Version]
- Sui, Y.; Sun, Z.; Zou, Y.; Li, W.; Jiang, M.; Luo, Y.; Liao, W.; Wang, Y.; Gao, X.; Liu, J.; et al. The Rlm1 transcription factor in Candida oleophila contributes to abiotic stress resistance and biocontrol efficacy against postharvest gray mold of kiwifruit. Postharvest Biol. Technol. 2020, 166, 111222. [Google Scholar] [CrossRef]
- Zhang, Z.; Chen, J.; Li, B.; He, C.; Chen, Y.; Tian, S. Influence of Oxidative Stress on Biocontrol Activity of Cryptococcus laurentii against Blue Mold on Peach Fruit. Front. Microbiol. 2017, 8, 151. [Google Scholar] [CrossRef] [Green Version]
- de Werra, P.; Péchy-Tarr, M.; Keel, C.; Maurhofer, M. Role of gluconic acid production in the regulation of biocontrol traits of Pseudomonas fluorescens CHA0. Appl. Environ. Microbiol. 2009, 75, 4162–4174. [Google Scholar] [CrossRef] [Green Version]
- Dutta, S.; Yu, S.-M.; Jeong, S.C.; Lee, Y.H. High-throughput analysis of genes involved in biocontrol performance of Pseudomonas fluorescens NBC275 against Gray mold. J. Appl. Microbiol. 2020, 128, 265–279. [Google Scholar] [CrossRef] [Green Version]
- Murata, K.; Suenaga, M.; Kai, K. Genome Mining Discovery of Protegenins A–D, Bacterial Polyynes Involved in the Antioomycete and Biocontrol Activities of Pseudomonas protegens. ACS Chem. Biol. 2021. [Google Scholar] [CrossRef]
- Chaudhary, T.; Dixit, M.; Gera, R.; Shukla, A.K.; Prakash, A.; Gupta, G.; Shukla, P. Techniques for improving formulations of bioinoculants. 3 Biotech 2020, 10, 199. [Google Scholar] [CrossRef]
- Berninger, T.; González López, Ó.; Bejarano, A.; Preininger, C.; Sessitsch, A. Maintenance and assessment of cell viability in formulation of non-sporulating bacterial inoculants. Microb. Biotechnol. 2018, 11, 277–301. [Google Scholar] [CrossRef] [Green Version]
- Behle, R.; Birthisel, T. Formulations of Entomopathogens as Bioinsecticides. In Mass Production of Beneficial Organisms; Morales-Ramos, J.A., Rojas, M.G., Shapiro-Ilan, D.I., Eds.; Academic Press: San Diego, CA, USA, 2014; Chapter 14; pp. 483–517. ISBN 978-0-12-391453-8. [Google Scholar]
- Butu, M.; Rodino, S.; Butu, A. Biopesticide formulations-current challenges and future perspectives. In Advances in Bio-Inoculant Science; Rakshit, A., Meena, V.S., Abhilash, P.C., Sarma, B.K., Singh, H.B., Fraceto, L., Parihar, M., Singh, A.K., Eds.; Woodhead Publishing: Sawston, UK, 2022; Chapter 3; pp. 19–29. ISBN 978-0-12-823355-9. [Google Scholar]
- Jangir, M.; Sharma, S.; Sharma, S. Development of next-generation formulation against Fusarium oxysporum and unraveling bioactive antifungal metabolites of biocontrol agents. Sci. Rep. 2021, 11, 22895. [Google Scholar] [CrossRef]
- Melin, P.; Schnürer, J.; Håkansson, S. Formulation and stabilisation of the biocontrol yeast Pichia anomala. Antonie Van Leeuwenhoek 2011, 99, 107–112. [Google Scholar] [CrossRef]
- Cumagun, C.J.R. Advances in Formulation of Trichoderma for Biocontrol. In Biotechnology and Biology of Trichoderma; Gupta, V.K., Schmoll, M., Herrera-Estrella, A., Upadhyay, R.S., Druzhinina, I., Tuohy, M.G., Eds.; Elsevier: Amsterdam, The Netherlands, 2014; Chapter 39; pp. 527–531. ISBN 978-0-444-59576-8. [Google Scholar]
- Bashan, Y.; de-Bashan, L.E.; Prabhu, S.R.; Hernandez, J.-P. Advances in plant growth-promoting bacterial inoculant technology: Formulations and practical perspectives (1998–2013). Plant Soil 2014, 378, 1–33. [Google Scholar] [CrossRef] [Green Version]
- Sriram, S.; Roopa, K.P.; Savitha, M.J. Extended shelf-life of liquid fermentation derived talc formulations of Trichoderma harzianum with the addition of glycerol in the production medium. Crop Prot. 2011, 30, 1334–1339. [Google Scholar] [CrossRef]
- Montesinos, E. Development, registration and commercialization of microbial pesticides for plant protection. Int. Microbiol. 2003, 6, 245–252. [Google Scholar] [CrossRef]
- Popp, J.; Pető, K.; Nagy, J. Pesticide productivity and food security. A review. Agron. Sustain. Dev. 2013, 33, 243–255. [Google Scholar] [CrossRef]
- (EFSA), E.F.S.A.; Alvarez, F.; Arena, M.; Auteri, D.; Borroto, J.; Brancato, A.; Carrasco Cabrera, L.; Castoldi, A.F.; Chiusolo, A.; Colagiorgi, A.; et al. Peer review of the pesticide risk assessment of the active substance sheep fat. EFSA J. 2022, 20, e07073. [Google Scholar] [CrossRef]

| Substance | Category | Status | Date of Approval/Expiration | Authorised | Commercial Name | Target Pathogens | Mechanism of Action |
|---|---|---|---|---|---|---|---|
| Bacillus amyloliquefaciens strain QST 713 | BA, FU | Approved | 1 February 2007 30 April 2022 | BE, BG, CY, CZ, DE, DK, EE, EL, ES, FI, FR, HR, IE, IT, LT, LU, LV, NL, PL, PT, RO, SE, SI, SK, UK | SERENADE ASO® | Large spectrum activity (Botrytis spp., Monilia spp., Sclerotinia spp., Rhizoctonia spp., Alternaria spp., Aspergillus spp., Phomopsis spp., Phytophthora spp., Pythium spp., Verticillium spp., etc) | antagonism, competition, antibiosis, activates plant defences |
| Bacillus amyloliquefaciens strain AT-332 | FU | Pending | \ \ | \ | \ | \ | \ |
| Bacillus amyloliquefaciens strain FZB42 | FU | Pending | \ \ | \ | \ | \ | \ |
| Bacillus amyloliquefaciens strain MBI 600 | FU | Approved | 16 September 2016 16 September 2026 | BE, CY, CZ, DE, DK, EL, FI, FR, HU, IT, LT, NL, PL, PT, RO, SE, UK | SERIFEL® | Botrytis cinerea, Sclerotinia spp. | antagonism, competition, antibiosis, activates plant defences |
| Bacillus amyloliquefaciens strain FZB24 | FU | Approved | 1 June 2017 1 June 2032 | BE, BG, CY, CZ, DE, EL, FR, IT, NL, PL, RO, SI, UK | TAEAGRO® | Powdery mildews diseases, Botrytis sp. | antagonism, competition, antibiosis, activates plant defences |
| Bacillus amyloliquefaciens subsp. plantarum strain D747 | FU | Approved | 1 April 2015 31 March 2025 | BE, CY, DK, EL, ES, FR, IE, IT, NL, PL, SE, SI, UK | AMYLO-X® | Botrytis cinerea, Monilinia spp., Sclerotinia spp. | antagonism, competition, antibiosis, activates plant defences |
| Bacillus nakamurai strain F727 | FU | Pending | \ \ | \ | \ | \ | \ |
| Bacillus pumilus strain QST 2808 | FU | Approved | 1 September 2014 31 August 2025 | CY, CZ, EL, FR, HR, IE, IT, NL, RO, SI, UK | SONATA® | Powdery mildews diseases | antagonism, competition, antibiosis, activates plant defences |
| Pseudomonas chlororaphis strain MA342 | FU | Approved | 1 October 2004 30 April 2022 | BE, DE, DK, ES, FI, FR, IT, LT, LU, NL, PT, SE, UK | PRORADIX® | Rhizoctonia spp., Helmintosporium solani, Fusarium spp. | antagonism, competition, antibiosis. |
| Streptomyces strain K61 | FU | Approved | 01 July 2021 30 June 2036 | BE, CY, DK, EE, FI, FR, HU, IT, LT, LV, NL, SE, UK | LALSTOP K61® WP | Damping-off and wilt and root diseases (Alternaria spp., Rhizoctonia spp., Fusarium spp., Phytophthora spp., Pythium spp.), | antagonism, mycoparasitism, competition and antibiosis. |
| Streptomyces lydicus strain WYEC 108 | BA, FU | Approved | 1 January 2015 31 December 2025 | ACTINOVATE® AG | Powdery and downey mildew, Botrytis spp., Alternaria spp. and other aerial borne pathogens, Pythium spp., Phytophthora spp., Fusarium spp., Rhizoctonia spp., Verticillium spp., Phymatotrichum omnivorum (cotton root rot) and other root decay fungi | antagonism, competition, activates plant defences, antibiosis | |
| Zucchini yellow mosaic virus (ZYMV mild strain) | FU | Not approved | \ \ | \ | \ | \ | \ |
| Zucchini yellow mosaic virus - weak strain | FU | Approved | 01 June 2013 31 May 2023 | FR | \ | \ | \ |
| Substance | Category | Status | Date of Approval/Expiration | Authorised | Commercial Name | Target Pathogens | Mechanism of Action |
|---|---|---|---|---|---|---|---|
| Ampelomyces quisqualis strain AQ10 | FU | Approved | 01 August 2018 31 July 2033 | BE, CY, DE, EL, ES, FR, IE, IT, LU, NL, SI, SK, UK | AQ10® WG | Powdery mildews diseases | mycoparasitism, competition |
| Aspergillus flavus strain MUCL 54911 | FU | Pending | \ \ | \ | \ | \ | \ |
| Aureobasidium pullulans strains DSM 14940 and DSM 14941 | BA, FU | Approved | 01 February 2014 31 January 2025 | BE, DE, EL, ES, FR, HU, IT, LU, NL, PL, PT, RO, SI, SK, UK | BLOSSOM PROTECT NEW® | Gleosporium spp., Penicillium spp., Stemphylium spp. e Botrytis spp. | antagonism, competition, antibiosis. |
| Candida oleophila strain O | FU | Approved | 01 October 2013 31 December 2024 | BE, EL, ES, FR, IT, NL, PL, UK | NEXY® | Post-harvest diseases (Botrytis cinerea, Penicillium spp., Colletotrichum musae) | antagonism, competition |
| Clonostachys rosea strain J1446 | FU | Approved | 01 April 2019 31 March 2034 | BE, CY, DE, DK, EE, ES, FI, FR, IE, NL, PL, SE, SI, UK | LALSTOP G46® WG | Damping-off and wilt and root diseases (Rhizoctonia spp., Fusarium spp., Phytophthora spp., Pythium spp., Verticillium spp.), Macrophomina phaseolina, Botrytis cinerea, Didymella bryoniae | mycoparasitism, competition, antagonism |
| Coniothyrium minitans strain CON/M/91-08 (DSM 9660) | FU | Approved | 1 Auugust 2017 31 July 2032 | BE, BG, CZ, DE, DK, EL, ES, FI, FR, HU, IE, IT, LU, NL, PL, PT, SE, SK, UK | CONTANS®WG | Sclerotinia sclerotiorum e Sclerotinia minor | mycoparasitism |
| Pseudozyma flocculosa | FU | Not approved | \ \ | \ | \ | \ | \ |
| Pythium oligandrum strain M1 | FU | Approved | 01 May 2009 30 April 2022 | AT, BG, CY, CZ, DE, EL, ES, FR, HU, IT, PL, PT, RO, SE, SI, SK, UK | POLYVERSUM® | Botrytis cinerea, Sclerotinia spp. | mycoparasitism, activates plant defences |
| Saccharomyces cerevisiae strain LAS02 | FU | Approved | 6 July2016 6 July2031 | EL, FR, PL | JULIETTA® | Botrytis spp., Monilia spp. | antagonism, competition |
| Trichoderma asperellum strain ICC012 | FU | Approved | 1 May 2009 30 April 2022 | DE, FR, IT, PT | TUSAL® | Phytophthora spp., Fusarium spp., Rhizoctonia solani, Pythium spp., Sclerotinia sclerotiorum | competition, antagonism, mycoparasitism and antibiosis. |
| Trichoderma asperellum strain T34 | FU | Approved | 1 June 2013 31 May 2023 | BE, CZ, DE, ES, FR, HU, IE, IT, NL, PL, PT, RO, SI, UK | T34 BIOCONTROL® | Wilt and root diseases (Fusarium spp., Pythium spp.) | competition, antagonism, activates plant defences, mycoparasitism and antibiosis |
| Trichoderma asperellum strain T25 | FU | Approved | 1 May 2009 30 April 2022 | DE, FR, IT, PT | TUSAL® | Phytophthora spp., Fusarium spp., Rhizoctonia solani, Pythium spp., Sclerotinia sclerotiorum | competition, antagonism, activates plant defences, mycoparasitism and antibiosis |
| Trichoderma asperellum strain TV1 | FU | Approved | 1 May 2009 30 April 2022 | DE, FR, IT, PT | PATRIOT GOLD® | Wilt and root diseases (Pythium spp., Phytophthora capsici, Rhizoctonia solani, Verticillium spp.) | competition, antagonism, activates plant defences, mycoparasitism and antibiosis |
| Trichoderma atroviride strain IMI 206040 | FU | Approved | 1 May 2009 30 April 2022 | IT | REMEDIER® | Wilt and root diseases (Rhizoctonia solani, Pythium spp., Sclerotinia sclerotiorum, Verticillium dahliae, Thielaviopsis basicola, Sclerotium rolfsii, Phytophthora spp., Armillaria mellea), Esca (Stereum hirsutum, Phellinus igniarius, Phaeomoniella chlamydospora, Fomitiporia mediterranea, Phaeocremonium aleophilumm), brown spot of pear (Stemphylium vesicarium) | competition, antagonism, activates plant defences, mycoparasitism and antibiosis |
| Trichoderma atroviride strain T11 | FU | Approved | 1 May 2009 30 April 2022 | IT | TUSAL® | Phytophthora spp., Fusarium spp., Rhizoctonia solani, Pythium spp., Sclerotinia sclerotiorum | competition, antagonism, activates plant defences, mycoparasitism and antibiosis |
| Trichoderma atroviride strain I-1237 | FU | Approved | 1 June 2013 31 May 2023 | CY, ES, FR, IT, PT | ESQUIVE® WP | Esca (Stereum hirsutum, Phellinus igniarius, Phaeomoniella chlamydospora, Fomitiporia mediterranea, Phaeocremonium aleophilumm), eutypiosis (Eutypa lata), Black Dead Arm = BDA (Botryospaeria obtuse) | antagonism, mycoparasitism, competition and antibiosis |
| Trichoderma atroviride strain SC1 | FU | Approved | 6 July 2016 6 July 2031 | BE, CY, CZ, DE, EL, ES, FR, HR, HU, IT, LU, NL, PL, PT, RO, SI | VINTEC® | Esca (Stereum hirsutum, Phellinus igniarius, Phaeomoniella chlamydospora, Fomitiporia mediterranea, Phaeocremonium aleophilumm), eutypiosis (Eutypa lata), Black Dead Arm = BDA (Botryospaeria obtuse), Botrytis spp. | antagonism, mycoparasitism, competition and antibiosis |
| Trichoderma gamsii strain ICC080 | FU | Approved | 1 May 2009 30 April 2022 | CY, DE, EL, ES, FR, IT, NL, PT | REMEDIER® | Wilt and root diseases (Rhizoctonia solani, Pythium spp., Sclerotinia sclerotiorum, Verticillium dahliae, Thielaviopsis basicola, Sclerotium rolfsii, Phytophthora spp., Armillaria mellea), Esca (Stereum hirsutum, Phellinus igniarius, Phaeomoniella chlamydospora, Fomitiporia mediterranea, Phaeocremonium aleophilumm), brown spot of pear (Stemphylium vesicarium) | competition, antagonism, activates plant defences, mycoparasitism and antibiosis |
| Trichoderma harzianum strain T-22 | FU | Approved | 39934 44681 | BE, DK, EE, ES, IE, IT, PL, PT, SK | TRIANUM® | Pythium spp., Rhizoctonia spp., Fusarium spp., Sclerotinia spp. | competition, antagonism, mycoparasitism, activates plant defences |
| Trichoderma harzianum strain ITEM 908 | FU | Approved | 39934 44681 | BE, DK, EE, ES, IE, IT, PL, PT, SK | TRIANUM® | Pythium spp., Rhizoctonia spp., Fusarium spp., Sclerotinia spp. | competition, antagonism, mycoparasitism, activates plant defences |
| Trichoderma harzianum strain B97 | FU | Pending | \ \ | \ | \ | \ | \ |
| Trichoderma polysporum strain IMI 206039 | FU | Not approved | 1 May 2009 30 April 2019 | SE | \ | \ | \ |
| Verticillium albo-atrum strain WCS850 | FU | Approved | 1 November 2019 31 October2034 | DE, DK, NL, SE, UK | DUTCH TRIG® | Ophyostoma novo-ulmi | activates plant defences against |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palmieri, D.; Ianiri, G.; Del Grosso, C.; Barone, G.; De Curtis, F.; Castoria, R.; Lima, G. Advances and Perspectives in the Use of Biocontrol Agents against Fungal Plant Diseases. Horticulturae 2022, 8, 577. https://doi.org/10.3390/horticulturae8070577
Palmieri D, Ianiri G, Del Grosso C, Barone G, De Curtis F, Castoria R, Lima G. Advances and Perspectives in the Use of Biocontrol Agents against Fungal Plant Diseases. Horticulturae. 2022; 8(7):577. https://doi.org/10.3390/horticulturae8070577
Chicago/Turabian StylePalmieri, Davide, Giuseppe Ianiri, Carmine Del Grosso, Giuseppe Barone, Filippo De Curtis, Raffaello Castoria, and Giuseppe Lima. 2022. "Advances and Perspectives in the Use of Biocontrol Agents against Fungal Plant Diseases" Horticulturae 8, no. 7: 577. https://doi.org/10.3390/horticulturae8070577
APA StylePalmieri, D., Ianiri, G., Del Grosso, C., Barone, G., De Curtis, F., Castoria, R., & Lima, G. (2022). Advances and Perspectives in the Use of Biocontrol Agents against Fungal Plant Diseases. Horticulturae, 8(7), 577. https://doi.org/10.3390/horticulturae8070577








