Abstract
Composts of spent mushrooms substrates can be an alternative for the partial replacement of peat as growing media in horticulture. Three mature composts from Agaricus bisporus (Ag), Pleurotus ostreatus (Pl), and 70% Ag:30% Pl (AgPl) production were used as partial components of peat growing media, used at a 1:4 compost:peat ratio for growing red baby leaf lettuce. They showed higher yields, between 3 and 7 times more than that for peat itself, even under the pressure of the plant pathogen Pythium irregulare. AgPl showed the higher suppressiveness (50%) against Pythium irregulare than Ag- (38%) or Pl- (15%) supplemented media. The combination of these raw materials and a suitable composting process is important for obtaining mature compost for use as a partial component of peat-based growing media.
1. Introduction
At the present time, there is an increasing demand for proteins of plant origin, which cost less and are healthier than the proteins from animal sources [1]. Edible mushrooms belonging to the Basidiomycetes are an interesting alternative due to their high concentrations of proteins and vitamins. Agaricus bisporus (A. bisporus) and Pleurotus ostreatus (P. ostreatus) are the most commonly cultivated mushroom species.
Worldwide mushroom production is greater than 25 MT per year [2], producing an average 5 kg of spent mushroom substrate (SMS) per kilogram of mushroom. Accumulation of this waste over time has a negative impact on the environment [3,4], generating leachates that can contaminate the soil and surrounding water [5]. After mushroom harvest, SMS still holds high levels of organic matter and nutrients and could be of potential use in agriculture, horticulture, or disease management [4]. However, SMS requires stabilization for using in agriculture, due to the amount of labile organic matter, assuring at the same time the elimination of mushroom mycelia that invade the SMS [6]. The stabilization of SMS through a composting process could offer a sustainable alternative for agriculture [7,8]. The composting process involves the succession of microorganisms, which is directly affected by various factors such as the specific mix of raw materials, temperature, aeration, moisture, C/N ratio, and pH, among others [9,10].
Lettuce (Lactuca sativa L.) is the most common of the salad leaf crops and is mainly consumed fresh. Among lettuces types, baby leaf red lettuce has popularity, due to its easier and faster processing and high content of phytochemicals with health beneficial effects. The successful production of lettuce in soilless culture with a minimal level of pest control depends on uniform, high-quality seedling germination and growth in a substrate [11].
Peat is the main component of growing media for lettuce production, because of its ideal characteristics for cropping such as constant chemical and physical properties [12]. Nevertheless, peat is a non-renewable resource whose harvest produces a negative impact on global climate change, and which is susceptible to soilborne pathogens such as Pythium irregulare (P. irregulare) (causing damping-off diseases), characterized as virulent and fast spreading in baby leaf lettuce crops in Mediterranean areas [11].
Composts from SMS can be partial components of growing media [13,14]. Moreover, some have shown potential suppressive activity against plant pathogens [11,12,13,14,15]. There are different mechanisms involved in pathogen suppression, including nutrient and space competition, antibiosis, and mycoparasitism [16], and the induction of systemic resistance to biotic stresses such as disease and abiotic stresses [17].
Our hypothesis is that the use, as a component of plant growing media, of compost made from spent mushroom substrate (SMS) after culture of A. bisporus (Ag), or P. ostreatus (Pl), or a combination combination of 70% A. bisporus and 30% P. ostreatus (AgPl) mixed with peat (1:4; compost:peat) would increase germination and plant biomass production and reduce the effects of P. irregulare in red baby leaf lettuce grown under soilless conditions compared to peat alone as growing media. To test this hypothesis, several experiments were carried out with the following objectives: (1) to evaluate the composting process of SMS from Ag, Pl, and AgPl; (2) to evaluate whether the composts could be used as a component of soilless growing media (1:4; compost:peat) to produce red baby leaf lettuce; (3) to evaluate the suppressive capacity of the composts under biotic stress of P. irregulare; and, (4) to evaluate whether the suppressiveness of SMS compost from AgPl inoculated with the biocontrol agent Trichoderma harzianum (AgPl + T) as a component of soilless growing media could be increased.
2. Materials and Methods
2.1. Raw Materials
Spent mushroom substrates from A. bisporus culture and P. ostreatus culture were produced after 3–4 mushroom harvests. The substrate for A. bisporus production was principally made using cereal straw, poultry manure, calcium sulphate (CaSO4·2H2O) and water to reach 70% humidity. Limestone gravel (high-purity calcium carbonate) was added to buffer the pH to 7.5, and the compost reached temperatures around 70 °C and was turned 3–4 times. The substrate for P. ostreatus production was principally made using straw, 70% humidity, and was not composted. Both substrates for A. bisporus or P. ostreatus production were packed in plastic bags for mushroom production and they were distributed to the production sites, 30–40 farms in a radius of 20 km from the substrate production site. Once they were spent, they were moved to a recycling plant for their management, which involved removing the plastic, homogenizing the SMS, and placing in piles for composting. For this study, SMS was collected from the compost recycling plant Sustratos de la Rioja located in Pradejón (La Rioja, Spain). The main characteristics of the Ag SMS and Pl SMS can be found in Table 1.

Table 1.
Physicochemical and chemical properties during composting process.
2.2. The Composting Process
Three composting piles of 2500 tons were set up: 100% Ag SMS (Pile Ag), 100% Pl SMS (Pile Pl), and a mix of 70% Ag SMS and 30% Pl SMS (v/v) (Pile AgPl). The piles showed an initial water holding capacity of 70%, which was maintained to 50–60% by regular turning when the temperature was higher than 65 °C. The composting processes lasted around 130 days, including 50 days for the bio-oxidative phase and a maturation phase of 80 days. Sampling was performed throughout the composting process at 0, 20, 35, 90, and 130 days from the beginning, from three sites on each pile and mixed to obtain a representative sample.
2.3. Assessment of Composts as a Component of Growing Media for Red Baby Leaf Lettuce Cultivation and as a Suppressive Growing Media under P. irregulare Biotic Stress
A pot experiment was performed to assess the different composts obtained after the composting process as compost growing media for red baby leaf lettuce cultivation. Treatments were Ag, Pl, and AgPl composts mixed with commercial peat 315 (Blond/black 60/40 Turbas y Coco Mar Menor S.L.) at a 1:4 (w/w; compost:peat) ratio. This ratio was selected as optimal for avoiding seed germination inhibition. Peat alone was used as the control treatment. The main physicochemical and chemical characteristics of the peat were as follows: pH 5.6; electrical conductivity (EC) 1 mS cm−1; total C 466 g kg−1; total N 9.4 g kg−1; total P 0.3 g kg−1; and total K 0.9 g kg−1. Red baby leaf lettuce “Ligier RZ84-14” (Rijk Zwaan, De Lier, The Netherlands) was selected as the assayed crop and P. irregulare as the pathogen to evaluate compost suppressiveness.The pathogen (P. irregulare) was isolated in potato dextrose agar medium (PDA, Sharlau, Spain) culture from lettuce plants showing disease symptoms in a lettuce field, then selected based on phenotypic appearance, and re-cultured on PDA to ensure identity The P. irregulare inoculum was produced by mixing and blending 4-day-old mycelia onto PDA with 200 mL of sterile distilled water. Thirty replicate pots were prepared from each treatment: half (15) were not inoculated with the pathogen and were used to evaluate the effect of compost as a growing media; the other half were infected with the pathogen (6.75 mL) before planting, equivalent to 8.23 log copies of internally transcribed spacers (ITS) g−1 growing media.
For germination, the pots were placed in a growth chamber at 18 ± 1 °C at 80% relative humidity (RH) and in darkness for 48 h. After that, the pots were randomly distributed in a growth chamber at 24/18 °C day/night with a RH range of 60–70% for 25 days. The germination percentage was measured six days after sowing and was calculated as the ratio of germinated seeds divided by total seeds, multiplied by 100. The lettuce plants were collected 25 days after planting, and the fresh plant biomass was weighed.
2.4. Assessment of Composts Inoculated with T. harzianum as a Component of Growing Media and a Suppressive Growing Media under P. irregulare Biotic Stress for Red Baby Leaf Lettuce Cultivation
A pot experiment was performed with two treatments: AgPl and AgPl + T. The latter was inoculated with T. harzianum (CEBAS collection) to achieve a final concentration of 6.75 log copies ITS g−1 growing media. T. harzianum was produced and immobilized in bentonite (1:9) [18]. The experiment was set up as described in the prior section.
2.5. Chemical and Microbiological Properties
The pH and electrical conductivity (EC) were measured in a 1:10 (w/v) aqueous extract of the substrate media. The total organic carbon (TOC) and total nitrogen (N) were measured using a LECO TruSpec C/N Elemental Analyzer. P, K, Na, Ca, Mg, Fe, and heavy metals were determined by inductively coupled plasma-mass spectrophotometry (ICP-MS PQExCell, VG-Thermo Elemental, Winsford, Cheshire, UK), after HNO3/HClO4 high-pressure digestion. Total organic carbon (TOC) loss due to mineralization was calculated from the initial (X1) and final (X2) ash contents according to the following equation [19]:
TOC loss (%) = 100 – 100 [X1 (100 − X2)/X2 (100 − X1)]
The suppressiveness index was calculated according to the formulae of disease suppressiveness describe by Veeken et al. [20]. The abundance of P. irregulare and T. harzianum inoculated was measured in a real-time PCR system by quantitative 7500 Fast real-time PCR (qPCR), following the protocol described by Giménez et al. [11] with the specific primers for P. irregulare and T. harzianum previously described by López-Mondéjar et al. [21].
2.6. Statistical Analysis
Data were analysed using the IMB Statistics SPSS 26 software, and an ANOVA test was performed. When the F-statistic was significant, the differences between treatments were determined using Tukey’s test at α = 0.05, or Duncan’s multiple range test for non-homogeneous values at p = 0.05. Normality and homogeneity of the variances were checked using the Shapiro–Wilk and Levene tests, respectively.
3. Results
3.1. The Composting Process
The temperature in the piles increased until it reached values ranging from 47.76 to 54.32 °C; these temperatures were maintained for 55 days (thermophilic phase). The temperatures then decreased during the cooling and maturation phase (Table 1). The thermophilic phase duration for Pile Ag was 64 days, for Pile AgPl 52 days, and for the Pile Pl only 44 days. Both piles with Ag (Pile Ag and Pile AgPl) showed higher temperatures (>50 °C) than Pile Pl.
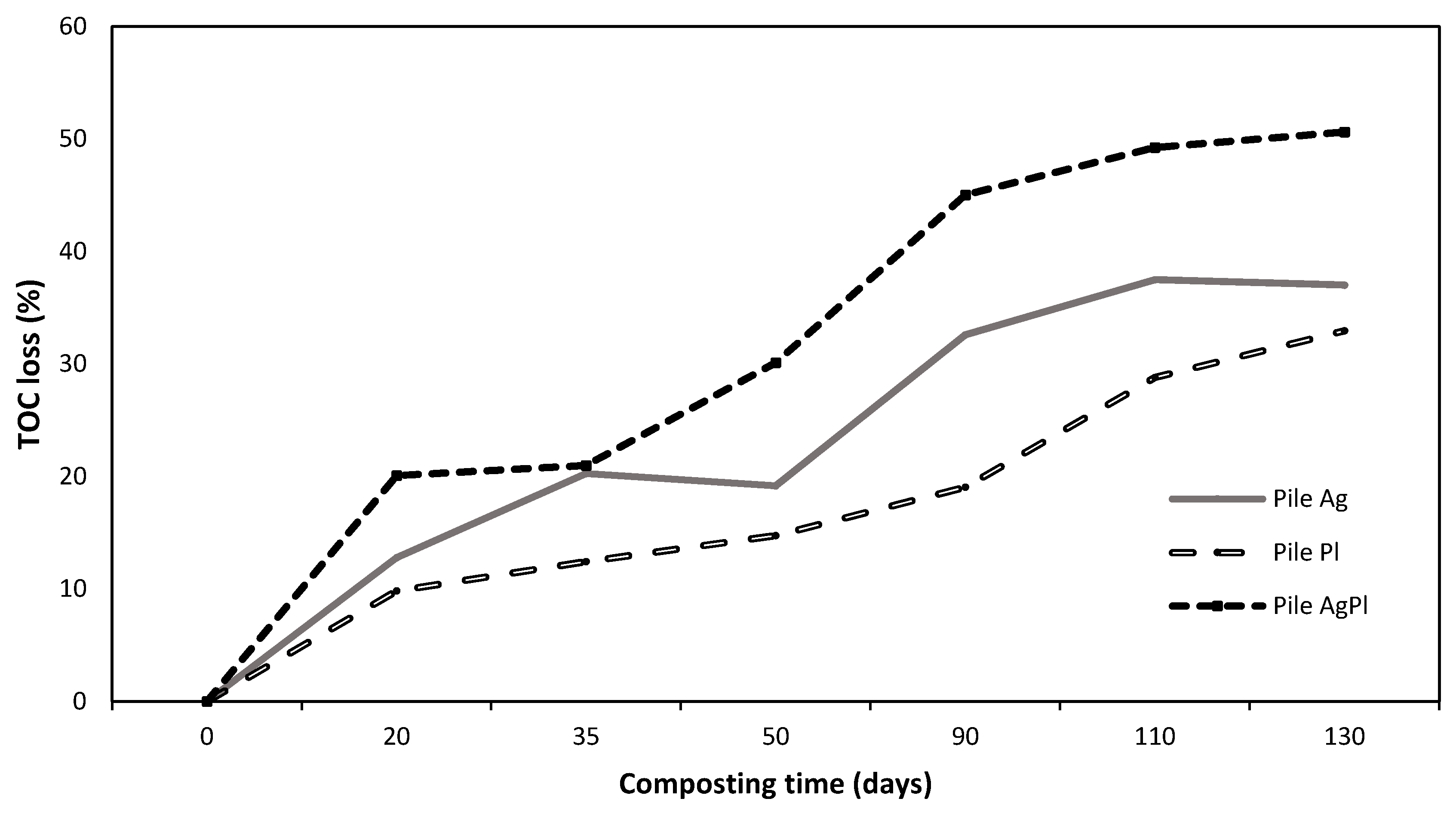
The variations in physicochemical and chemical parameters during the composting process are shown in Table 1. In general, the pH and EC increased during the composting process in the three piles. After 130 days, the pH reached values of 7.62 (Pile Ag), 7.57 (Pile AgPl), and 7.88 (Pile Pl), while the EC reached values of 7.67 (Pile Ag), 7.56 (Pile AgPl), and 7.11 (Pile Pl). The C/N ratios and total carbon (TC) of the three piles diminished during the composting process, although the C content was higher in Pile Pl during composting than in the other two piles. The highest percentage of TOC loss occurred in Pile AgPl (45%), followed by Pile Ag (30%) and Pile Pl (23%) (Figure 1). Inversely to C content, the total nitrogen (TN) content increased during the composting process, and Piles AgPl and Ag showed the highest TN content throughout the process (Table 1). In general, total P, K, Mg, and, especially, Ca also significantly increased during the composting process, with Pile Ag showing the highest values at the end of composting process. Total Cd, Cr, Mn, Zn, Cu, Cr, Pb, and Ni showed a similar trend to the other measured minerals, also increasing during composting (Table 2). Composts did not show evidence of Salmonella spp., Listeria spp., or Escherichia coli. Moreover, no animal pathogens were detected in the substrates before use in mushroom cultivation (data not shown).
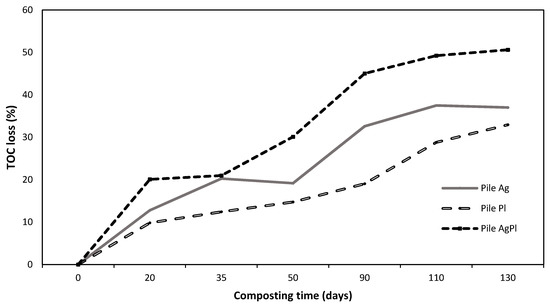
Figure 1.
Percentage of total organic carbon losses during the composting process of the different piles. 100% A. bisporus (Pile Ag), 100% P. ostreatus (Pile Pl), and 70% A. bisporus: 30% P. ostreatus (Pile AgPl).

Table 2.
Heavy metal concentration of 100% A. bisporus (Ag), 100% P. ostreatus (Pl), and 70% A. bisporus: 30% P. ostreatus (AgPl) composts.
3.2. Composts as a Growing Media Component
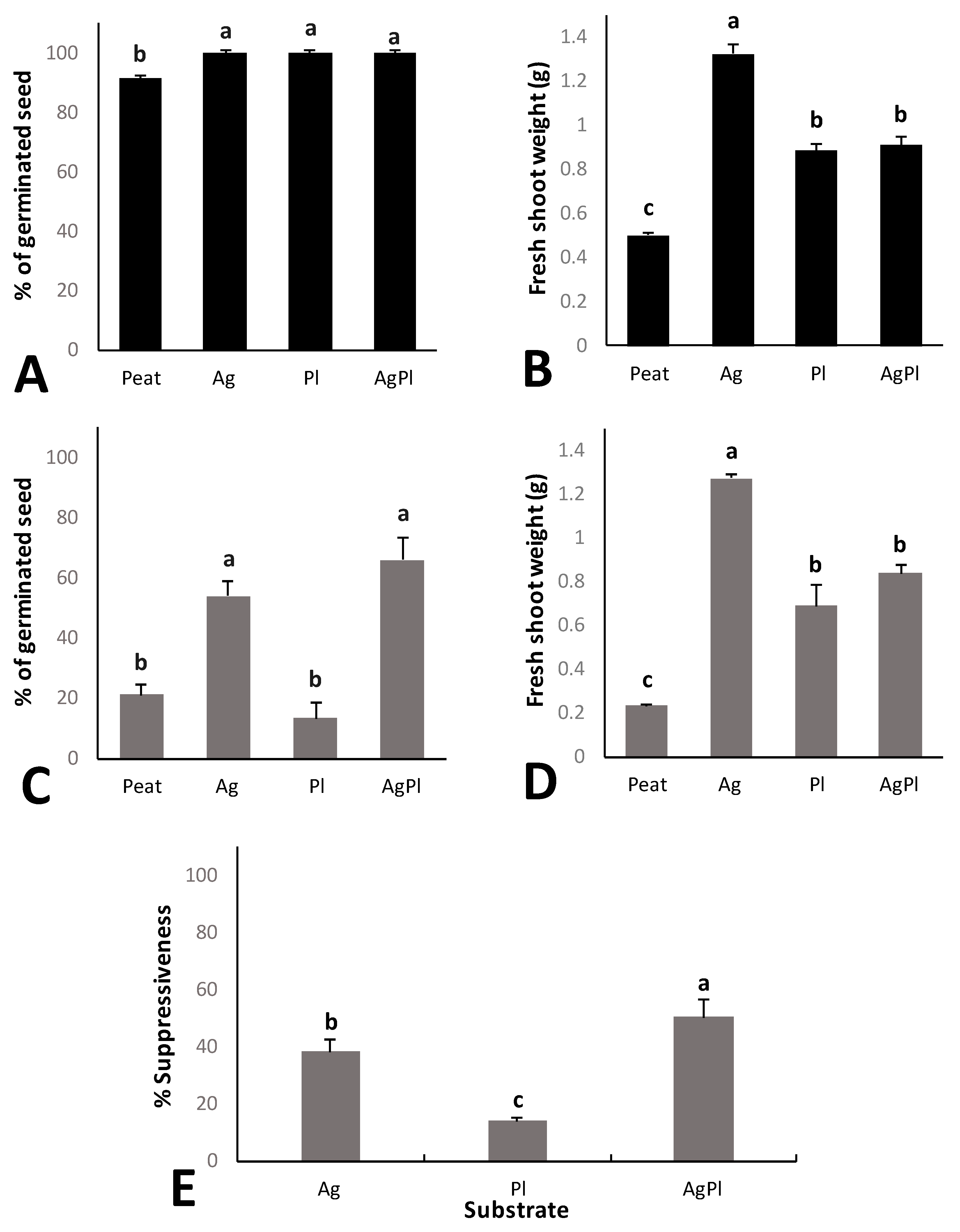
The percentage of red baby leaf lettuce seed germination in the different composts was significantly higher than the germination rate of plants grown in peat alone, and no significant differences between the composts were observed (Figure 2A). The fresh shoot weight of red baby leaf lettuce grown in the composts was also significantly higher than that grown in peat. Comparing the three composts, the highest fresh shoot weight was obtained for Ag (Figure 2B).
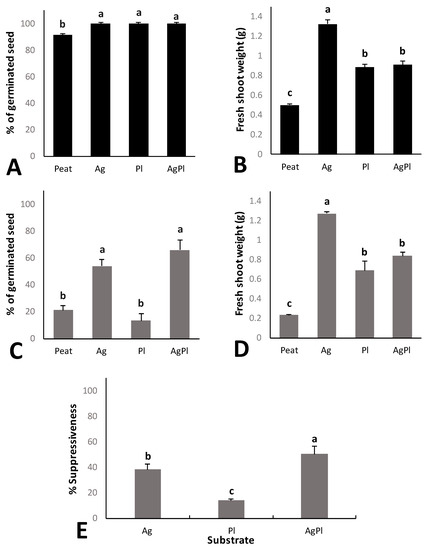
Figure 2.
Germinated seed percentage (A) and fresh shoot weight (B) of red baby leaf lettuce plants without pathogen. Germinated seed percentage with P. irregulare (C) and fresh shoot weight (D) of red baby leaf lettuce plants with P. irregulare. Suppressiveness index (%) against P. irregulare (E). Error bars represent the standard errors. Values with the same letter do not differ significantly according to Tukey’s test (α < 0.05). Compost growing media of 100% peat, and peat with 100% A. bisporus (Ag), 100% P. ostreatus (Pl), or 70% A. bisporus: 30% P. ostreatus (AgPl) added at a 1:4 compost:peat ratio.
3.3. Composts as a Component of Suppressive Growing Media against P. irregulare
Under P. irregulare pressure, red baby leaf lettuce seed germination was between 20% and 70%. The Ag and AgPl media showed significantly higher seed germination rates than Pl and peat (Figure 2C). Moreover, the fresh shoot weight was significantly higher in all compost growing media than in peat alone. Comparing the composts, the fresh shoot weight for Ag was significantly higher than the others, which did not differ (Figure 2D).
The suppressiveness index made it possible to separate suppression against the pathogen from the nutritional and biostimulant effects of the composts. AgPl showed the highest suppressiveness index against P. irregulare (Figure 2E). Both AgPl and Ag showed greater suppressiveness than Pl and peat. Differences were not observed in final P. irregulare concentration (Table 3).

Table 3.
Amount of P. irregulare in the different compost growing media.
3.4. Composts Amended with T. harzianum as a Component of Growing Media
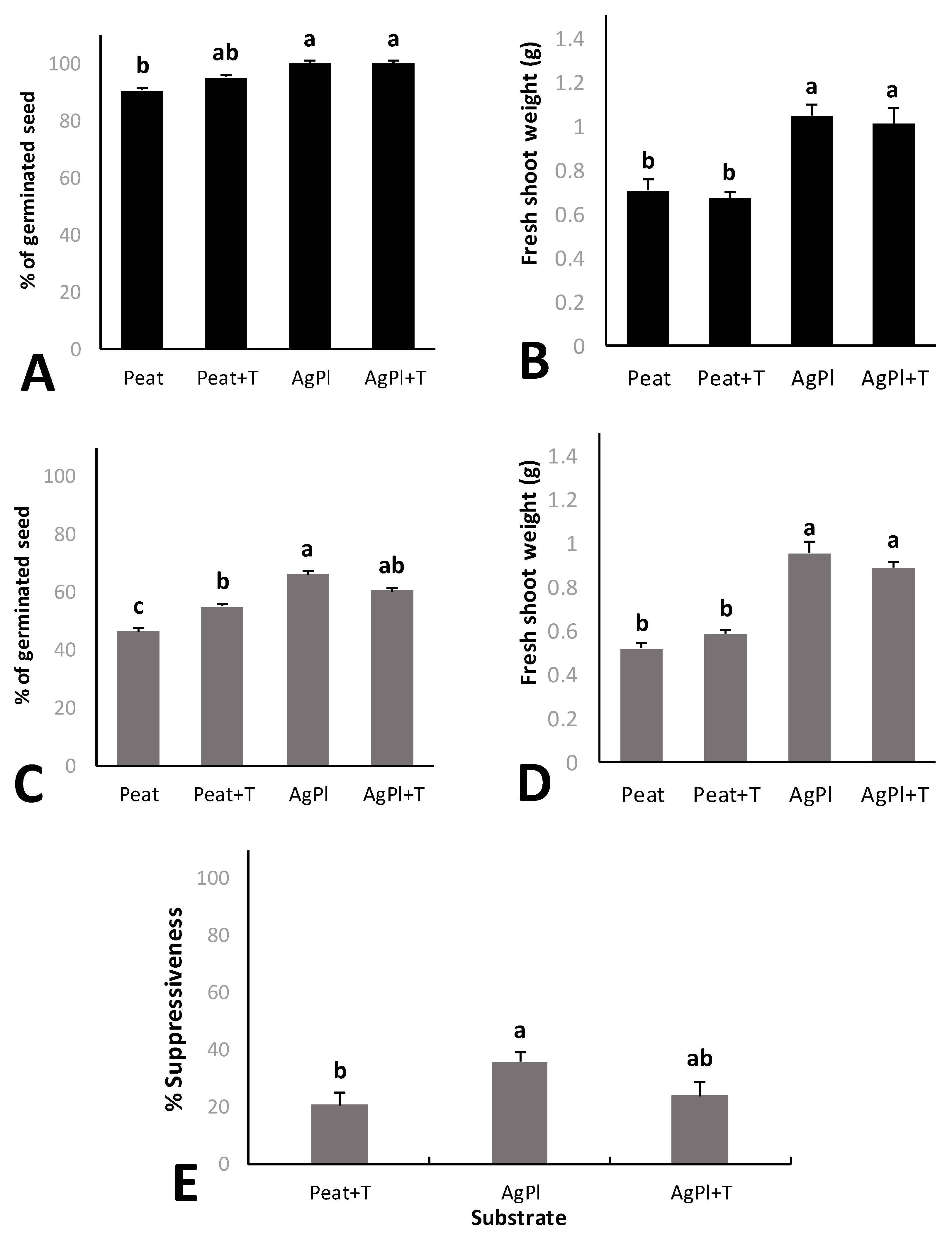
AgPl showed the best suppressiveness index and germination under P. irregulare biotic stress and a good value for fresh plant biomass weight. This compost was inoculated with T. harzianum (AgPl + T) in order to evaluate the possibility of increasing the effects against P. irregulare. Red baby leaf lettuce grown in AgPl and AgPl + T showed significantly higher germination rates and fresh shoot weights than lettuce grown in Peat and Peat + T (Figure 3A,B). No significant differences were observed between compost growing media either with or without T. harzianum (Figure 3A,B).
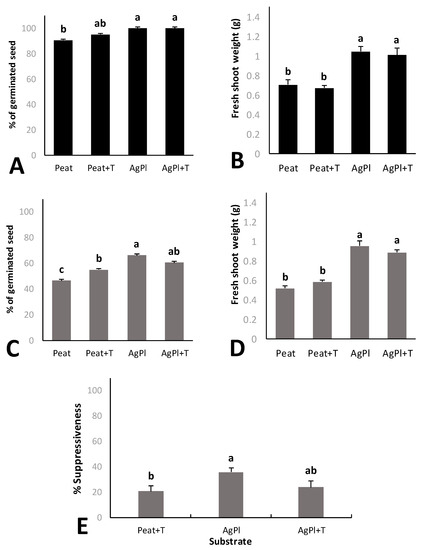
Figure 3.
Germinated seed percentage (A) and fresh shoot weight of baby red lettuce plants (B) without pathogen. Germinated seed percentage (C) and fresh shoot weight of baby red lettuce plants with P. irregulare (D). Suppressiveness index (%) against P. irregulare (E). Error bars represent the standard errors. Values with the same letter do not differ significantly according to Tukey’s post hoc test (α < 0.05). Peat; Peat + T. harzianum (Peat + T), (70% A. bisporus and 30% P. ostreatus) + T. harzianum (AgPl + T).
3.5. Composts Amended with T. harzianum as a Component of Suppressive Growing Media against P. irregulare: Effects on Red Baby Leaf Lettuce Seed Germination, Growth, and the Suppressiveness Index
Lettuce seed germination was significantly lower in Peat than in Peat + T and in both AgPl and AgPl + T (Figure 3C). No significant differences were observed between AgPl and AgPl + T. Both compost growing media also showed significantly higher fresh shoot weight than Peat and Peat + T (Figure 3D). T. harzianum did not increase the fresh shoot weight compared to its non-inoculated treatment. With respect to the suppressiveness index, T. harzianum was not found in either compost growing media or in peat under P. irregulare pressure (data not shown). Moreover, there were no differences between the amount of T. harzianum in Peat and AgPl showing, 4.44 and 4.51 log copies ITS g−1, respectively (Table 3).
4. Discussion
Composting has gained significant attention as an environmentally friendly way to dispose of utilized organic wastes, rather than sending them to a landfill [14]. However, it is necessary to develop adequate composting processes. The temperature profile, C/N ratio, and the evolution of the total organic C are three of the main parameters that indicate the progress of a composting process [22,23]. The temperatures profiles of the compost piles followed the stages frequently observed in the composting process. These stages included a thermophilic phase (>45 °C) resulting from the intense aerobic microbial metabolism that leads to the rapid breakdown of organic matter by microbes producing heat as an exothermic reaction, and a maturation stage with a temperature decrease (down to 35 °C) as the organic matter is stabilized and consequently microbial activity drops [24]. The length and temperatures of the thermophilic phase depend on the composition of the raw materials. Pile Ag and Pile AgPl showed higher temperatures (54 °C) than Pile Pl (48 °C), probably due to the fact that the A. bisporus SMS contained labile components, especially nitrogen, to reactivate the microbial biomass during the composting. This would increase the temperature to a greater extent and maintain it for longer than in Pile Pl [25].
During composting, the amount of organic matter tends to drop due to mineralization and carbon loss in the form of carbon dioxide. The highest TOC losses were found in both piles with A. bisporus (Pile Ag (30%) and Pile AgPl (45%)), probably due to the higher amounts of most labile components in the organic matter from A. bisporus SMS. In contrast to the C losses, the TN level increased during the composting process; this usually occurs in the composting process when organic matter loss is greater than ammonium loss [23] or nitrate leaching. The higher TN levels in Pile Ag and Pile AgPl could be due to the chicken manure, rich in organic nitrogen [3,4,5,6,7,8,9,10,11,12,13,14]. Similar results were also observed by González-Marcos et al. [3], who found a TOC reduction of 50% when composting a mix of A. bisporus SMS and by-products from a winery. During the composting process, the C/N ratio diminished significantly due to the C losses, and the piles reached values below 15–20, indicative of high-quality mature compost [14,26]. Both pH and EC are important factors that influence seed germination and plant growth rates. The pH values of the three final composts ranged between 7.57 and 7.88, adequate for use in agriculture. Nevertheless, a lower range for growing media (5.2–7.0) is recommended [9]. The ECs of the three SMSs assayed were also higher than those found in other agroindustrial wastes (>4 dS m−1) [9]. Furthermore, during composting, the mineralization of organic matter contributes to EC increases [27], reaching values ranging from 7.11 to 7.67. These EC values are not recommended in growing media [28], and some strategy must be applied to make the composts more suitable for use. One of those is the use of smaller ratios of compost as growing media. We used composts in at a 1:4 compost:peat ratio. As a result, both the EC and pH levels reached values within the range recommended. The composts displayed some characteristics ideal for agricultural application: [29] N > 1 g/100 g, P > 0.43 g/100 g, K > 0.41 g/100 g, Ca > 1.4 g/100 g, Mg > 0.2 g/100 g. The heavy metal content also increased in the three composts due to the composting process, although the levels were within the limits permitted in the current Spanish legal framework [30].
The use of these three composts as a growing media component for baby leaf lettuce cultivation increased the germination percentage and fresh plant weight over peat alone, mainly due to the nutrient content and a possible biostimulant effect [10]. Ag and AgPl resulted in the highest plant weights, even in presence of the P. irregulare pathogen. These characteristics make the three composts (Ag, AgPl, and Pl) attractive as at least a partial component of growing media, not only for their effect but also for the homogeneity of the raw materials. Moreover, spent mushroom composts from A. bisporus and P. ostreatus production would assure the same characteristics of the final composts, which is an important aspect of growing media materials, which should not result in differences in production from one batch to another [10]. Properties such as suppressiveness against certain plant diseases make it possible to reduce the use of chemical pesticides in agriculture. The disease-suppressive effects of composts of different origins and compositions have been widely studied, and different results have been obtained according to the compost type, pathogen to be controlled, environmental conditions, etc. [15]. The three compost growing media (Ag, Pl, AgPl) also showed a suppressive capacity against P. irregulare. AgPl followed by Ag showed the highest suppressiveness index. Disease suppression by composts is mainly attributed to the biotic factor [31], where beneficial microorganisms recolonize the compost [32]. The suppressive effects of composts are associated with the organic matter–microorganism–root consortia that occur in the plant rhizosphere. There are two main types of mechanisms via which composts help suppress plant pathogens: direct and indirect. In our assay, as no effect on the pathogen interaction was observed, the suppressive effect should be therefore mainly attributed to an indirect effect through the plant rather than through a direct interaction with the pathogen. Indirect mechanisms include the activation of plant disease-resistance genes or the improvement of plant nutrition and vigour, allowing the plant to grow in the presence of the pathogen and not be affected [10,11].
The difference observed between the suppressiveness of AgPl and Ag could be due to the presence in the combined AgPl compost of plant growth-promoting rhizobacteria (PGPR) and endophyte microorganisms, rending the host more resistant or tolerant to disease [33]. This would explain the fact that there was a suppressive effect when Ag and Pl were combined but not with Ag alone. The suppressiveness of composts has been studied in depth, and it can be generally concluded that the raw materials from which a given compost is prepared are crucial to the development of suppressive microbiota within it [33]. Kumbhar [34] observed, for instance, that compost from A. bisporus showed a beneficial effect in controlling some pests and diseases such as damping off, root rot of creeping grass, Verticillium disease, and Fusarium wilt in tomato.
The incorporation of T. harzianum into composts is a method used to induce or increase the natural suppressiveness of growing media [35]. The incorporation of T. harzianum into the AgPl compost did not appear to increase the compost’s natural suppressiveness, while the incorporation in peat was effective. It could be due to the raw materials in the composts or the addition of biocontrol microorganisms against T. harzianum, that did not permit T. harzianum growth. It is well documented that some species of Trichoderma are mushroom pathogens [36], and this forces mushroom growers to control them by using biocontrol microorganisms such as Bacillus spp. [37]. These could have been well established in the spent composts and therefore be part of their potential natural suppressiveness, yet they would not permit T. harzianum establishment.
5. Conclusions
We conclude that the composting process of spent mushroom substrates from A. bisporus, P. ostreatus, and a mix of 70%:30% mixture, respectively (Pile Ag, Pile Pl, and Pile AgPl) may produce quality, stabilized composts. The compost may be reintroduced into a production system and be a promising partial component (1:4, compost:peat) of organic growing media that could produce higher red baby leaf lettuce yields and provide some suppressive activity against P. irregulare. The compost obtained from the combination of both A. bisporus and P. ostreatus showed the highest suppressiveness against P. irregulare although the incorporation of T. harzianum did not increase the suppressiveness. A study of a compost microbial community before adding T. harzianum would be recommended to evaluate the establishment of the T. harzianum.
Author Contributions
Investigation, D.H., F.C., and J.A.P.; funding acquisition, J.A.P.; writing—original draft preparation, D.H.; writing—review and editing, D.H., M.R., F.C., J.A.S.-T., and J.A.P. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Spanish Ministry of Economy and Competitiveness (AGL2017-84085-C3-3-R); the Centre for the Development of Industrial Technology (CDTI) funding the project “Biotechnological management of organic waste for obtaining new bioproducts of new generation” code 99193 and the “Fundación Seneca de la Region de Murcia” within the Research Groups of Excellence programme of the Region of Murcia (19896/GERM/15).
Institutional Review Board Statement
Not applicable for studies not involving humans or animals.
Informed Consent Statement
Not applicable for studies not involving humans.
Data Availability Statement
The data presented in this study were obtained from red baby leaf lettuce (Lactuca sativa L., Ligier RZ84-14, Rijk Zwaan, De Lier, The Netherlands) culture.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| SMS | spent mushroom substrate |
| Ag | SMS of Agaricus bisporus |
| Pl | SMS of Pleurotus ostreatus |
| AgPl | mix of 70% SMS of Agaricus bisporus and 30% SMS of Pleurotus ostreatus |
References
- Jerzak, M.A.; Śmiglak-Krajewska, M. Globalization of the Market for Vegetable Protein Feed and Its Impact on Sustainable Agricultural Development and Food Security in EU Countries Illustrated by the Example of Poland. Sustainability 2020, 12, 888. [Google Scholar] [CrossRef]
- Phan, C.W.; Sabaratnam, V. Potential Uses of Spent Mushroom Substrate and Its Associated Lignocellulosic Enzymes. Appl. Microbiol. Biotechnol. 2012, 96, 863–873. [Google Scholar] [CrossRef] [PubMed]
- González-Marcos, A.; Alba-Elías, F.; Martínez-de-Pisón, F.J.; Alfonso-Cendón, J.; Castejón-Limas, M. Composting of Spent Mushroom Substrate and Winery Sludge. Compost Sci. Util. 2015, 23, 58–65. [Google Scholar] [CrossRef]
- Lau, K.L.; Tsang, Y.Y.; Chiu, S.W. Use of Spent Mushroom Compost to Bioremediate PAH-Contaminated Samples. Chemosphere 2003, 52, 1539–1546. [Google Scholar] [CrossRef]
- Stewart, D.P.C.; Cameron, K.C.; Cornforth, I.S. Inorganic-N Release from Spent Mushroom Compost under Laboratory and Field Conditions. Soil Biol. Biochem. 1998, 30, 1689–1699. [Google Scholar] [CrossRef]
- García-Delgado, C.; Yunta, F.; Eymar, E. Methodology for Polycyclic Aromatic Hydrocarbons Extraction from Either Fresh or Dry Spent Mushroom Compost and Quantification by High-Performance Liquid Chromatography–Photodiode Array Detection. Commun. Soil Sci. Plant Anal. 2013, 44, 817–825. [Google Scholar] [CrossRef]
- López-González, J.A.; Suárez-Estrella, F.; Vargas-García, M.C.; López, M.J.; Jurado, M.M.; Moreno, J. Dynamics of Bacterial Microbiota during Lignocellulosic Waste Composting: Studies upon Its Structure, Functionality and Biodiversity. Bioresour. Technol. 2015, 175, 406–416. [Google Scholar] [CrossRef] [PubMed]
- Medina, J.; Monreal, C.; Barea, J.M.; Arriagada, C.; Borie, F.; Cornejo, P. Crop Residue Stabilization and Application to Agricultural and Degraded Soils: A Review. J. Waste Manag. 2015, 42, 41–54. [Google Scholar] [CrossRef] [PubMed]
- Khater, E.S.G. Some physical and chemical properties of compost. Int. J. Waste Resour. 2015, 5, 1–5. [Google Scholar] [CrossRef]
- Pascual, J.A.; Ceglie, F.; Tuzel, Y.; Koller, M.; Koren, A.; Hitchings, R.; Tittarelli, F. Organic Substrate for Transplant Production in Organic Nurseries. A Review. Agron. Sustain. Dev. 2018, 38, 35. [Google Scholar] [CrossRef]
- Giménez, A.; Fernández, J.A.; Pascual, J.A.; Ros, M.; López-Serrano, M.; Egea-Gilabert, C. An Agroindustrial Compost as Alternative to Peat for Production of Baby Leaf Red Lettuce in a Floating System. Sci. Hortic. (Amst.) 2019, 246, 907–915. [Google Scholar] [CrossRef]
- Pane, C.; Spaccini, R.; Piccolo, A.; Scala, F.; Bonanomi, G. Compost Amendments Enhance Peat Suppressiveness to Pythium Ultimum, Rhizoctonia Solani and Sclerotinia Minor. Biol. Control 2011, 56, 115–124. [Google Scholar] [CrossRef]
- Medina, E.; Paredes, C.; Pérez-Murcia, M.D.; Bustamante, M.A.; Moral, R. Spent Mushroom Substrates as Component of Growing Media for Germination and Growth of Horticultural Plants. Bioresour. Technol. 2009, 100, 4227–4232. [Google Scholar] [CrossRef] [PubMed]
- Paula, F.S.; Tatti, E.; Abram, F.; Wilson, J.; O’Flaherty, V. Stabilisation of Spent Mushroom Substrate for Application as a Plant Growth-Promoting Organic Amendment. J. Environ. Manag. 2017, 196, 476–486. [Google Scholar] [CrossRef]
- Blaya, J.; Lloret, E.; Ros, M.; Pascual, J.A. Identification of Predictor Parameters to Determine Agro-Industrial Compost Suppressiveness against Fusarium Oxysporum and Phytophthora Capsici Diseases in Muskmelon and Pepper Seedlings. J. Sci. Food Agric. 2015, 95, 1482–1490. [Google Scholar] [CrossRef]
- Benhamou, N.; Chet, I. Cellular and Molecular Mechanisms Involved in the Interaction between Trichoderma Harzianum and Pythium Ultimum. Appl. Environ. Microbiol. 1997, 63, 2095–2099. [Google Scholar] [CrossRef]
- Shoresh, M.; Harman, G.E.; Mastouri, F. Induced Systemic Resistance and Plant Responses to Fungal Biocontrol Agents. Annu. Rev. Phytopathol. 2010, 48, 21–43. [Google Scholar] [CrossRef]
- Zohar-Perez, C.; Chernin, L.; Chet, I.; Nussinovitch, A. Structure of dried cellular alginate matrix containing fillers provides extra protection for microorganisms against UVC radiation. Radiat. Res. 2003, 1, 198–204. [Google Scholar] [CrossRef]
- Paredes, C.; Roig, A.; Bernal, M.P.; Sánchez-Monedero, M.A.; Cegarra, J. Evolution of Organic Matter and Nitrogen during Co-Composting of Olive Mill Wastewater with Solid Organic Wastes. Biol. Fertil. Soils 2000, 32, 222–227. [Google Scholar] [CrossRef]
- Veeken, A.H.M.; Blok, W.J.; Curci, F.; Coenen, G.C.M.; Termorshuizen, A.J.; Hamelers, H.V.M. Improving Quality of Composted Biowaste to Enhance Disease Suppressiveness of Compost-Amended, Peat-Based Potting Mixes. Soil Biol. Biochem. 2005, 37, 2131–2140. [Google Scholar] [CrossRef]
- López-Mondéjar, R.; Antón, A.; Raidl, S.; Ros, M.; Pascual, J.A. Quantification of the Biocontrol Agent Trichoderma Harzianum with Real-Time TaqMan PCR and Its Potential Extrapolation to the Hyphal Biomass. Bioresour. Technol. 2010, 101, 2888–2891. [Google Scholar] [CrossRef] [PubMed]
- Bustamante, M.A.; Paredes, C.; Marhuenda-Egea, F.C.; Pérez-Espinosa, A.; Bernal, M.P.; Moral, R. Co-Composting of Distillery Wastes with Animal Manures: Carbon and Nitrogen Transformations in the Evaluation of Compost Stability. Chemosphere 2008, 72, 551–557. [Google Scholar] [CrossRef] [PubMed]
- Bernal, M.P.; Alburquerque, J.A.; Moral, R. Composting of Animal Manures and Chemical Criteria for Compost Maturity Assessment. A Review. Bioresour. Technol. 2009, 100, 5444–5453. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.; Liang, F.; Yu, A.; Li, B.; Yang, L. Evaluation of Stability and Maturity during Forced-Aeration Composting of Chicken Manure and Sawdust at Different C/N Ratios. Chemosphere 2010, 78, 614–619. [Google Scholar] [CrossRef]
- Paredes, C.; Medina, E.; Moral, R.; Pérez-Murcia, M.D.; Moreno-Caselles, J.; Bustamante, M.A.; Cecilia, J.A. Characterization of the Different Organic Matter Fractions of Spent Mushroom Substrate. Commun. Soil Sci. Plant Anal. 2009, 40, 150–161. [Google Scholar] [CrossRef]
- Bernal, M.P.; Sánchez-Monedero, M.A.; Paredes, C.; Roig, A. Carbon Mineralization from Organic Wastes at Different Composting Stages during Their Incubation with Soil. Agric. Ecosyst. Environ. 1998, 69, 175–189. [Google Scholar] [CrossRef]
- Farrell, M.; Jones, D.L. Critical Evaluation of Municipal Solid Waste Composting and Potential Compost Markets. Bioresour. Technol. 2009, 100, 4301–4310. [Google Scholar] [CrossRef] [PubMed]
- Noguera, P.; Abad, M.; Puchades, R.; Maquieira, A.; Noguera, V. Influence of Particle Size on Physical and Chemical Properties of Coconut Coir Dust as Container Medium. Commun. Soil Sci. Plant. Anal. 2003, 34, 593–605. [Google Scholar] [CrossRef]
- Debertoldi, M.; Vallini, G.; Pera, A. The Biology of Composting: A Review. Waste Manag. Res. 1983, 1, 157–176. [Google Scholar] [CrossRef]
- Real Decreto 506/2013, de 28 de Junio, Sobre Productos Fertilizantes; Ministerio de la Presidencia: Madrid, Spain, 2013.
- Noble, R.; Roberts, S.J. Eradication of Plant Pathogens and Nematodes during Composting: A Review. Plant Pathol. 2004, 53, 548–568. [Google Scholar] [CrossRef]
- Alabouvette, C.; Olivain, C.; Steinberg, C. Biological Control of Plant Diseases: The European Situation. Eur. J. Plant. Pathol. 2006, 114, 329–341. [Google Scholar] [CrossRef]
- Castaño, R.; Borrero, C.; Avilés, M. Organic Matter Fractions by SP-MAS 13C NMR and Microbial Communities Involved in the Suppression of Fusarium Wilt in Organic Growth Media. Biol. Control. 2011, 58, 286–293. [Google Scholar] [CrossRef]
- Kumbhar, A.S. Assessment of Growth Promotion and Disease Suppressing Ability of Spent Mushroom Substrate. Master Thesis, Dr. Panjabrao Deshmukh Krishi Vidyapeeth University, Akola, India, June 2012. [Google Scholar]
- Blaya, J.; López-Mondéjar, R.; Lloret, E.; Pascual, J.A.; Ros, M. Changes Induced by Trichoderma Harzianum in Suppressive Compost Controlling Fusarium Wilt. Pestic. Biochem. Physiol. 2013, 107, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Velázquez-Cedeño, M.A.; Farnet, A.M.; Ferré, E.; Savoie, J.M. Variations of Lignocellulosic Activities in Dual Cultures of Pleurotus Ostreatus and Trichoderma Longibrachiatum on Unsterilized Wheat Straw. Mycologia 2004, 96, 712–719. [Google Scholar] [CrossRef]
- Colavolpe, M.B.; Mejía, S.J.; Albertó, E. Efficiency of treatments for controlling Trichoderma spp. during spawning in cultivation of lignicolous mushrooms. Braz. J. Microbiol. 2014, 45, 1263–1270. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).