Improved Tolerance for Onion Thrips and Iris Yellow Spot in Onion Plant Introductions after Two Selection Cycles
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Planting and Field Design
2.3. Data Collection
2.4. Enzyme-Linked Immunosorbent Assay (ELISA)
2.5. Data Analysis
3. Results and Discussion
3.1. Virus Detection and IYS Spread
3.2. Leaf Color and Waxiness
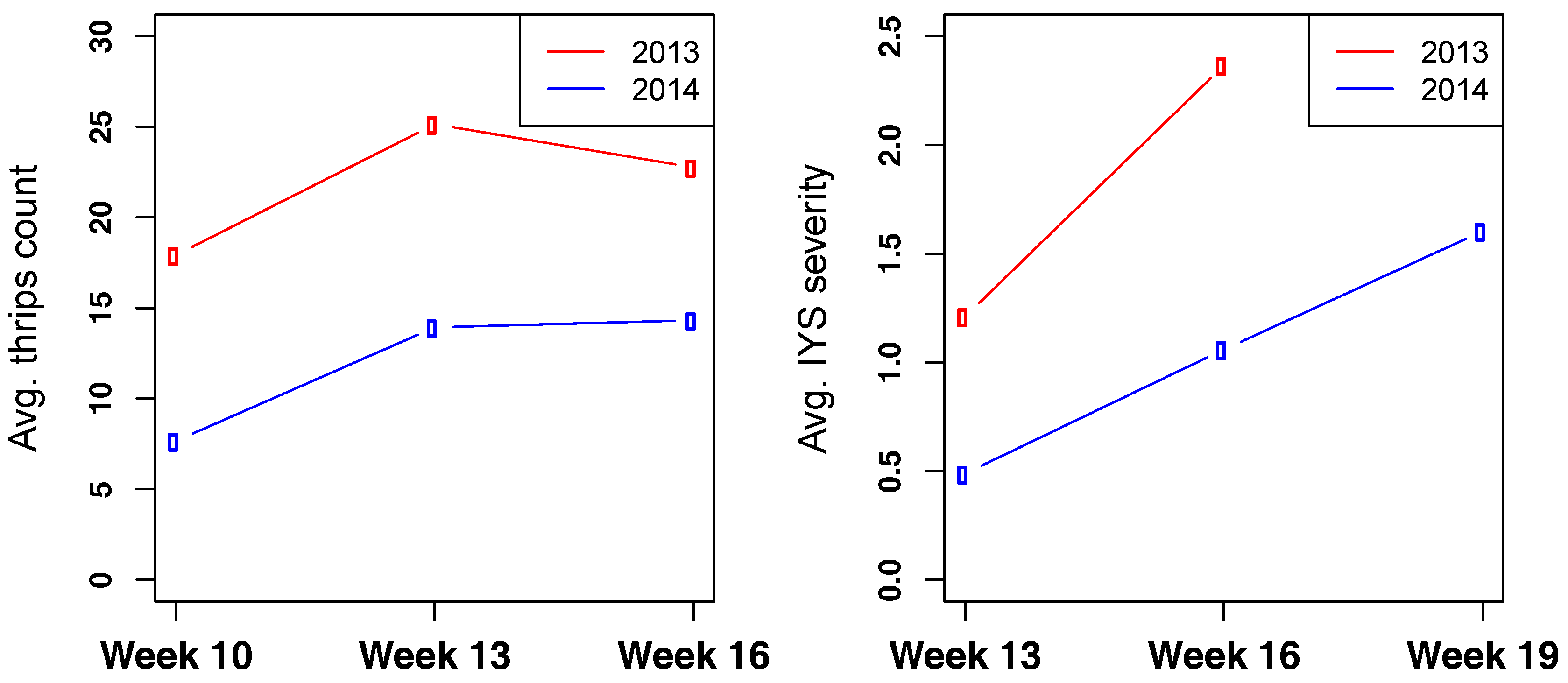
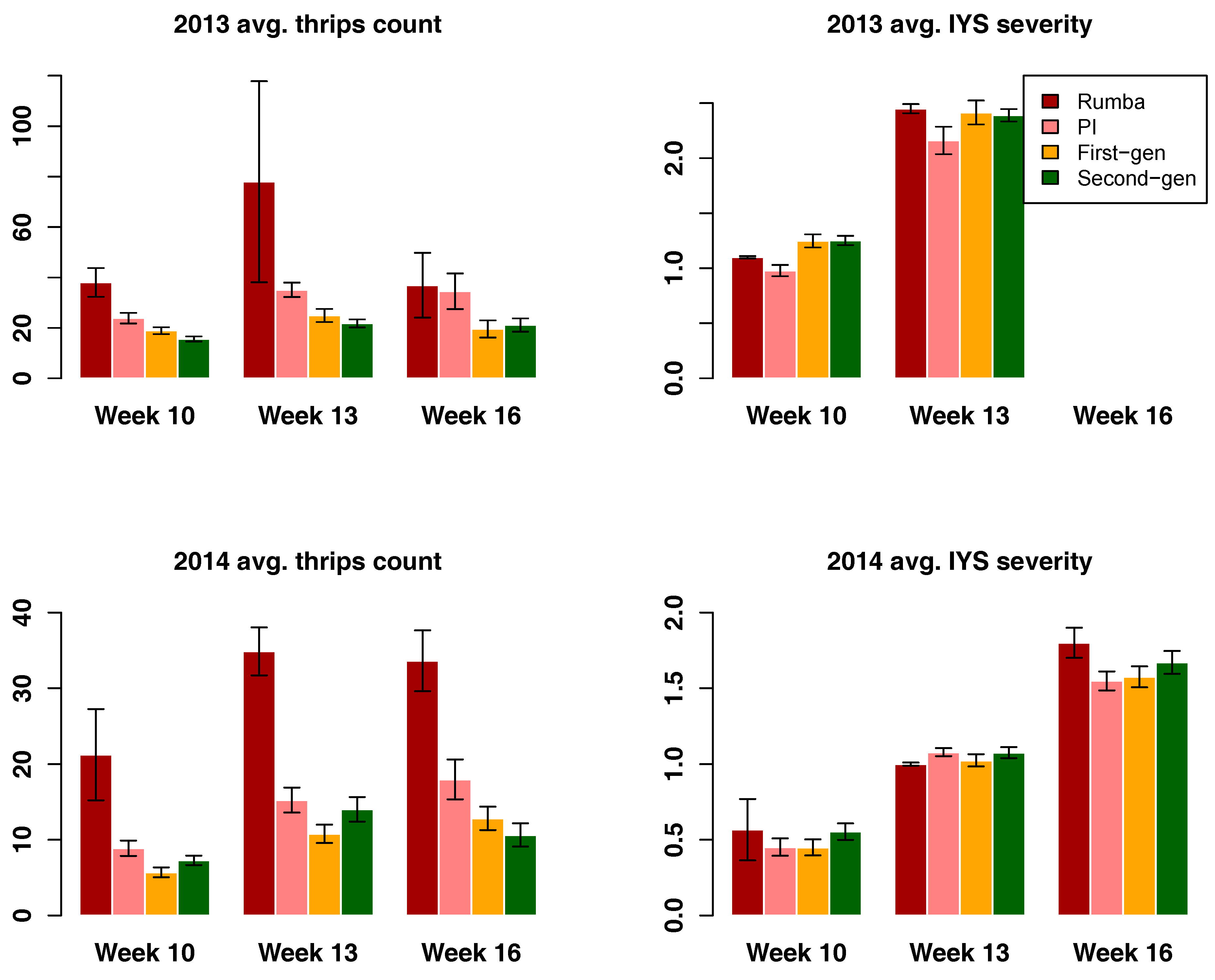
3.3. Thrips and IYS Severity
3.4. Selection Progress for Thrips and IYS
3.5. Future Direction towards Developing Better Germplasm
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gent, D.H.; du Toit, L.J.; Fichtner, S.F.; Mohan, S.K.; Pappu, H.R.; Schwartz, H.F. Iris yellow spot virus: An emerging threat to onion bulb and seed production. Plant Dis. 2006, 90, 1468–1480. [Google Scholar] [CrossRef]
- Cortes, I.; Livieratos, I.C.; Derks, A.; Peters, D.; Kormelink, R. Molecular and serological characterization of iris yellow spot virus, a new and distinct tospovirus species. Phytopathology 1998, 88, 1276–1282. [Google Scholar] [CrossRef] [PubMed]
- Coutts, B.A.; McMichael, L.A.; Tesoriero, L.; Rodoni, B.C.; Wilson, C.R.; Wilson, A.J.; Persley, D.M.; Jones, R.A.C. Iris yellow spot virus found infecting onions in three Australian states. Australas. Plant Pathol. 2003, 32, 555–557. [Google Scholar] [CrossRef]
- Pozzer, L.; Bezerra, I.C.; Kormelink, R.; Prins, M.; Peters, D.; Resende, R.D.; de Avila, A.C. Characterization of a tospovirus isolate of iris yellow spot virus associated with a disease in onion fields in Brazil. Plant Dis. 1999, 83, 345–350. [Google Scholar] [CrossRef]
- Rosales, M.; Pappu, H.; Lopez, L.; Mora, R.; Aljaro, A. 1457801. Iris yellow spot virus in onion in Chile. Plant Dis. 2005, 89, 1245. [Google Scholar] [CrossRef]
- Ravi, K.; Kitkaru, A.; Winter, S. Iris yellow spot virus in onions: A new tospovirus record from India. Plant Pathol. 2006, 55, 288. [Google Scholar] [CrossRef]
- Ghotbi, T.; Shahraeen, N.; Winter, S. Occurrence of tospoviruses in ornamental and weed species in Markazi and Tehran provinces in Iran. Plant Dis. 2005, 89, 425–429. [Google Scholar] [CrossRef]
- Gera, A.; Cohen, J.; Salomon, R.; Raccah, B. Iris yellow spot tospovirus detected in onion (Allium cepa) in Israel. Plant Dis. 1998, 82, 127. [Google Scholar] [CrossRef]
- Tomassoli, L.; Tiberini, A.; Masenga, V.; Vicchi, V.; Turina, M. Characterization of Iris yellow spot virus isolates from onion crops in northern Italy. J. Plant Pathol. 2009, 91, 733–739. [Google Scholar]
- Murai, T. Current status of the onion thrips, Thrips tabaci, as a pest thrips in Japan. Agrochem. Jpn. 2004, 84, 7–10. [Google Scholar]
- Ward, L.I.; Perez-Egusquiza, Z.; Fletcher, J.D.; Corona, F.M.O.; Tang, J.Z.; Liefting, L.W.; Martin, E.J.; Quinn, B.D.; Pappu, H.R.; Clover, G.R.G. First report of Iris yellow spot virus on Allium cepa in New Zealand. Plant Pathol. 2009, 58, 406. [Google Scholar] [CrossRef]
- Nischwitz, C.; Pappu, H.R.; Mullis, S.W.; Sparks, A.N.; Langston, D.R.; Csinos, A.S.; Gitaitis, R.D. Phylogenetic analysis of Iris yellow spot virus isolates from onion (Allium cepa) in Georgia (USA) and Peru. J. Phytopathol. 2007, 155, 531–535. [Google Scholar] [CrossRef]
- Robène-Soustrade, I.; Hostachy, B.; Roux-Cuvelier, M.; Minatchy, J.; Hédont, M.; Pallas, R.; Couteau, A.; Cassam, N.; Wuster, G. First report of Iris yellow spot virus in onion bulb- and seed-production fields in Réunion Island. Plant Pathol. 2006, 55, 288. [Google Scholar] [CrossRef]
- Córdoba-Sellés, C.; Martínez-Priego, L.; Muńoz-Gómez, R.; Jordá-Gutiérrez, C. Iris yellow spot virus: A new onion disease in Spain. Plant Dis. 2005, 89, 1243. [Google Scholar] [CrossRef]
- Hall, J.; Mohan, K.; Knott, E.; Moyer, J. Tospoviruses associated with scape blight of onion (Allium cepa) seed crops in Idaho. Plant Dis. 1993, 77, 952. [Google Scholar] [CrossRef]
- Pappu, H.; Matheron, M. Characterization of Iris yellow spot virus from onion in Arizona. Plant Health Prog. 2008, 9, 44. [Google Scholar] [CrossRef]
- Poole, G.J.; Pappu, H.R.; Davis, R.M.; Turini, T.A. Increasing outbreaks and impact of Iris yellow spot virus in bulb and seed onion crops in the Imperial and Antelope Valleys of California. Plant Health Prog. 2007, 8, 50. [Google Scholar] [CrossRef]
- Schwartz, H.; Brown Jr, W.; Blunt, T.; Gent, D. Iris yellow spot virus on onion in Colorado. Plant Dis. 2002, 86, 560. [Google Scholar] [CrossRef]
- Bag, S.; Singh, J.; Davis, R.; Chounet, W.; Pappu, H. Iris yellow spot virus in onion in Nevada and northern California. Plant Dis. 2009, 93, 674. [Google Scholar] [CrossRef]
- Creamer, R.; Sanogo, S.; Moya, A.; Romero, J.; Molina-Bravo, R.; Cramer, C. Iris yellow spot virus on onion in New Mexico. Plant Dis. 2004, 88, 1049. [Google Scholar] [CrossRef]
- Hoepting, C.; Schwartz, H.; Pappu, H. First report of Iris yellow spot virus on onion in New York. Plant Dis. 2007, 91, 327. [Google Scholar] [CrossRef]
- Crowe, F.; Pappu, H. Outbreak of Iris yellow spot virus in onion seed crops in central Oregon. Plant Dis. 2005, 89, 105. [Google Scholar] [CrossRef]
- Miller, M.; Saldana, R.; Black, M.; Pappu, H. First report of Iris yellow spot virus on onion (Allium cepa) in Texas. Plant Dis. 2006, 90, 1359. [Google Scholar] [CrossRef]
- du Toit, L.; Pappu, H.; Druffel, K.; Pelter, G. Iris yellow spot virus in onion bulb and seed crops in Washington. Plant Dis. 2004, 88, 222. [Google Scholar] [CrossRef]
- Diaz-Montano, J.; Fuchs, M.; Nault, B.A.; Fail, J.; Shelton, A.M. Onion thrips (Thysanoptera: Thripidae): A global pest of increasing concern in onion. J. Econ. Entomol. 2011, 104, 1–13. [Google Scholar] [CrossRef]
- Shock, C.C.; Feibert, E.; Jensen, L.; Mohan, S.K.; Saunders, L.D. Onion variety response to Iris yellow spot virus. HortTechnology 2008, 18, 539–544. [Google Scholar] [CrossRef]
- Kritzman, A.; Beckelman, H.; Alexandrov, S.; Cohen, J.; Lampel, M.; Zeidan, M.; Raccah, B.; Gera, A. Lisianthus leaf necrosis: A new disease of lisianthus caused by Iris yellow spot virus. Plant Dis. 2000, 84, 1185–1189. [Google Scholar] [CrossRef]
- Srinivasan, R.; Sundaraj, S.; Pappu, H.R.; Diffie, S.; Riley, D.G.; Gitaitis, R.D. Transmission of Iris yellow spot virus by Frankliniella fusca and Thrips tabaci (Thysanoptera: Thripidae). J. Econ. Entomol. 2012, 105, 40–47. [Google Scholar] [CrossRef]
- Diaz-Montano, J.; Fuchs, M.; Nault, B.A.; Shelton, A.M. Evaluation of onion cultivars for resistance to onion thrips (Thysanoptera: Thripidae) and Iris yellow spot virus. J. Econ. Entomol. 2010, 103, 925–937. [Google Scholar] [CrossRef]
- Kritzman, A.; Lampel, M.; Raccah, B.; Gera, A. Distribution and transmission of Iris yellow spot virus. Plant Dis. 2001, 85, 838–842. [Google Scholar] [CrossRef]
- Nagata, T.; Almeida, A.C.L.; Resende, R.O.; de Ávila, A.C. The identification of the vector species of Iris yellow spot tospovirus occurring on onion in Brazil. Plant Dis. 1999, 83, 399. [Google Scholar] [CrossRef]
- Riley, D.G.; Joseph, S.V.; Srinivasan, R.; Diffie, S. Thrips vectors of tospoviruses. J. Integr. Pest Manag. 2011, 2, I1–I10. [Google Scholar] [CrossRef]
- Gent, D.H.; Schwartz, H.F.; Khosla, R. Distribution and incidence of Iris yellow spot virus in Colorado and its relation to onion plant population and yield. Plant Dis. 2004, 88, 446–452. [Google Scholar] [CrossRef]
- Schwartz, H.; Gent, D. High Plains IPM Guide, a Cooperative Effort of the University of Wyoming; University of Nebraska, Colorado State University and Montana State University Montana State University, 2004. Available online: https://wiki.bugwood.org/index.php?title=HPIPM:Onion_Iris_Yellow_Spot&oldid=56075 (accessed on 15 January 2019).
- Mau, R.F.L.; Kessing, J.L.M. Thrips tabaci (Linderman). Crop Knowl. Master 2019. Available online: http://www.extento.hawaii.edu/kbase/crop/Type/t_tabaci.htm (accessed on 5 February 2019).
- Cranshaw, W.S.; Schweissing, F. Control of organophosphate resistant onion thrips, 1986. Insectic. Acaric. Tests 1989, 14, 128. [Google Scholar]
- Mahaffey, L.A. Diversity, Seasonal Biology, and IPM of Onion-Infesting Thrips in Colorado; Colorado State University: Fort Collins, CO, USA, 2006. [Google Scholar]
- Aldosari, S.A. Development of an IPM System for Onion Thrips (Thrips Tabaci Lindeman) as a Pest of Bulb Onions. Ph.D. Thesis, Colorado State University, Fort Collins, CO, USA, 1997. [Google Scholar]
- Jones, H.; Bailey, S.; Emsweller, S. Thrips resistance in the onion. Hilgardia 1934, 8, 213–232. [Google Scholar] [CrossRef]
- Coudriet, D.L.; Kishaba, A.N.; McCreight, J.D.; Bohn, G.W. Varietial resistance in onions to thrips. J. Econ. Entomol. 1979, 72, 614–615. [Google Scholar] [CrossRef]
- Molenaar, N.D. Genetics, thrips (Thrips tabaci L.) resistance and epicuticular wax characteristics of nonglossy and glossy onions (Allium cepa L.). Diss. Abstr. Int. B Sci. Eng. 1984, 45, 224. [Google Scholar]
- Damon, S.J.; Groves, R.L.; Havey, M.J. Variation for Epicuticular Waxes on Onion Foliage and Impacts on Numbers of Onion Thrips. J. Am. Soc. Hortic. Sci. 2014, 139, 495–501. [Google Scholar] [CrossRef]
- KIRK, W.D. Ecologically seIective coIoured traps. Ecol. Entomol. 1984, 9, 35–41. [Google Scholar] [CrossRef]
- Diaz-Montano, J.; Fail, J.; Deutschlander, M.; Nault, B.A.; Shelton, A.M. Characterization of resistance, evaluation of the attractiveness of plant odors, and effect of leaf color on different onion cultivars to onion thrips (Thysanoptera: Thripidae). J. Econ. Entomol. 2012, 105, 632–641. [Google Scholar] [CrossRef] [PubMed]
- Mohseni-Moghadam, M.; Cramer, C.S.; Steiner, R.L.; Creamer, R. Evaluating winter-sown onion entries for Iris yellow spot virus susceptibility. HortScience 2011, 46, 1224–1229. [Google Scholar] [CrossRef]
- Boateng, C.O.; Schwartz, H.F.; Havey, M.J.; Otto, K. Evaluation of Onion Germplasm for Resistance to Iris Yellow Spot (Iris Yellow Spot Virus) and Onion Thrips, Thrips tabaci. Southwest. Entomol. 2014, 39, 237–260. [Google Scholar] [CrossRef]
- Multani, P.S.; Cramer, C.S.; Steiner, R.L.; Creamer, R. Screening winter-sown onion entries for Iris yellow spot virus tolerance. HortScience 2009, 44, 627–632. [Google Scholar] [CrossRef]
- Cramer, C.S. Onion germplasm lines selected for reduced Iris yellow spot symptom expression. N.M. Agr. Expt. Stn. Germ. Rel. Not. 2013, 5. [Google Scholar]
- Cramer, C.S.; Singh, N.; Kamal, N.; Pappu, H.R. Screening Onion Plant Introduction Accessions for Tolerance to Onion Thrips and Iris Yellow Spot. Hortscience 2014, 49, 1253–1261. [Google Scholar] [CrossRef]
- Kamal, N.; Cramer, C.S. Selection Progress for Resistance to Iris Yellow Spot in Onions. HortScience 2018, 53, 1088–1094. [Google Scholar] [CrossRef]
- U.S. Department of Agriculture; National Genetic Resources Program. Germplasm Resources Information Network–(GRIN); National Germplasm Resources Laboratory: Beltsville, MD, USA, 2015. [Google Scholar]
- NM Climate Center. Leyendecker II PSRC. Available online: https://weather.nmsu.edu/ziamet/station/nmcc-da-5/ (accessed on 5 February 2019).
- Walker, S.J. Bulb Onion Culture and Management for Southern New Mexico; New Mexico State University: Las Cruces, New Mexico, 2009; pp. 1–16. [Google Scholar]
- Cramer, C.S.; Kamal, N.; Singh, N. Evaluating Iris Yellow Spot Disease Incidence and Severity in Onion Germplasm of Varying Leaf Characteristics. HortScience 2017, 52, 527–532. [Google Scholar] [CrossRef]
- Copeland, R. Assaying levels of plant virus by ELISA. In Plant Virology Protocols; Springer: Berlin/Heidelberg, Germany, 1998; pp. 455–460. [Google Scholar]
- Louie, R. Vascular puncture of maize kernels for the mechanical transmission of maize white line mosaic virus and other viruses of maize. Phytopathology 1995, 85, 139–143. [Google Scholar] [CrossRef]
- Zhang, L.; Zitter, T.; Lulkin, E. Artificial inoculation of maize white line mosaic virus into corn and wheat. Phytopathology 1991, 81, 397–400. [Google Scholar] [CrossRef]
- Fraser, L.; Matthews, R. Efficient mechanical inoculation of turnip yellow mosaic virus using small volumes of inoculum. J. Gen. Virol. 1979, 44, 565–568. [Google Scholar] [CrossRef]
- Salazar, L.; Jayasinghe, U. Fundamentals of Purification of Plant Viruses; International Potato Centre: Lima, Peru, 1999; pp. 1–10. [Google Scholar]
- Kalmus, H. The use of abrasives in the transmission of plant viruses. Ann. Appl. Biol. 1945, 32, 230–234. [Google Scholar] [CrossRef] [PubMed]
- Beraha, L.; Varzandeh, M.; Thornberry, H. Mechanism of the action of abrasives on infection by tobacco mosaic virus. Virology 1955, 1, 141–151. [Google Scholar] [CrossRef]

| Onion Thrips | IYS | IYS | |||||
|---|---|---|---|---|---|---|---|
| (Number/Plant) x | Severity Rating w | Incidence v | |||||
| Entry z | 10 y | 13 | 16 | 13 | 16 | 19 | 13 |
| 2013 | |||||||
| PI 172703 | 24.9 | 46.4 | 14.5 | 1.3 | 2.6 | 100.0 | |
| NMSU 10-580-1 | 14.7 | 10.8 *** | 4.3 | 1.9 * | 3.3 + | 100.0 | |
| NMSU 12-257 | 6.9 ** | 9.5 *** | 4.7 | 1.5 | 2.7 | 100.0 | |
| NMSU 12-258 | 10.9 * | 16.4 ** | 7.4 | 1.2 | 2.9 | 100.0 | |
| NMSU 12-260 | 10.3 * | 8.7 *** | 4.2 | 1.8 * | 3.2 + | 100.0 | |
| NMSU 12-261 | 10.3 * | 7.3 *** | 8.9 | 2.3 *** | 3.4 * | 100.0 | |
| NMSU 12-774 | 12.0 * | 8.6 *** | 2.8 | 1.8 * | 3.1 | 96.7 | |
| NMSU 10-583-1 | 24.6 | 17.3 ** | 9.4 | 1.2 | 2.7 | 100.0 | |
| NMSU 12-279 | 11.1 * | 7.7 *** | 1.9 | 1.7 + | 2.7 | 100.0 | |
| NMSU 10-579-1 | 17.9 | 25.9 | 7.9 | 1.4 | 2.3 | 100.0 | |
| NMSU 12-255 | 7.0 ** | 18.7 ** | 11.5 | 1.2 | 2.2 | 100.0 | |
| PI 546140 | 34.3 | 35.3 | 11.4 | 1.1 | 2.4 | 100.0 | |
| NMSU 10-700 | 24.3 | 35.4 | 23.6 | 0.9 | 1.7 * | 86.7 | |
| NMSU 12-298 | 12.1 *** | 18.7 | 14.1 | 1 | 1.7 + | 100.0 | |
| NMSU 12-299 | 17.9 * | 33.8 | 32.6 | 1 | 2 | 96.7 | |
| PI 289689 | 9.7 | 13.9 | 0.9 | 1.1 | 2.9 | 96.7 | |
| NMSU 10-618-1 | 15 | 22.2 | 3.3 | 1.2 | 2.5 | 100.0 | |
| NMSU 12-285 | 13.3 | 14 | 6.4 | 1.1 | 2.1 * | 100.0 | |
| PI 258956 | 13.8 | 40.3 | 71.2 | 0.9 | 1.9 | 86.7 | |
| NMSU 10-634-1 | 29.6 * | 45.7 | 57.3 | 1 | 1.8 | 96.7 | |
| NMSU 12-289 | 12.4 | 48.1 | 53 | 0.8 | 1.7 | 80.0 | |
| PI 239633 | 14.7 | 19.1 | 34.5 | 0.7 | 0.9 | 80 | |
| NMSU 10-589-1 | 14.5 | 26.1 | 19.4 | 1 | 1.6+ | 100.0 *** | |
| NMSU 12-283 | 5.7 | 20.3 | 44.4 | 1.0 + | 2.0 ** | 90.0 *** | |
| NMSU 10-596-1 | 8.9 | 14.3 | 26.5 | 2.2 *** | 3.0 *** | 100.0 *** | |
| NMSU 12-325 | 7.8 | 23.4 | 29.2 | 1 | 1.8 * | 86.7 *** | |
| Rumba | 38 | 78 | 37 | 0.9 | 2.1 | 100 | |
| 2014 | |||||||
| PI 172703 | 16.3 | 27.1 | 9.1 | 0.7 | 1.1 | 2.1 | 50.0 |
| NMSU 10-580-1 | 7.7 * | 17.1 | 8.1 | 0.5 | 1 | 1.6 | 46.7 |
| NMSU 12-257 | 6.6 ** | 8.1 ** | 6.5 | 0.6 | 1.3 + | 1.3 * | 41.3 |
| NMSU 12-258 | 6.2 ** | 6.3 ** | 9 | 0.6 | 1.1 | 1.6 + | 43.3 |
| NMSU 12-260 | 5.0 ** | 15.0+ | 5.7 | 0.8 | 1 | 2.2 | 46.7 |
| NMSU 12-261 | 7.5 * | 11.4 * | 6.5 | 0.7 | 0.9 | 1.3 * | 47.7 |
| NMSU 12-774 | 6.5 ** | 18.7 | 7.1 | 0.7 | 1.1 | 1.4 * | 66.7 |
| NMSU 10-583-1 | 5.1 ** | 13.2 * | 6.7 | 0.5 | 1.1 | 1.7 | 40.0 |
| NMSU 12-279 | 5.2 ** | 12.1 * | 7 | 0.5 | 1.2 | 1.9 | 46.7 |
| NMSU 10-579-1 | 0.7 * | 11.4 * | 8.6 | 0.3 | 0.7 * | 1.7 | 26.7 |
| NMSU 12-255 | 13.4 | 15.3+ | 10.5 | 0.8 | 1 | 1.7 | 61.7 |
| PI 546140 | 12.3 | 27.2 | 20.3 | 0.6 | 1.2 | 2 | 56.7 |
| NMSU 10-700 | 7.7 | 20.1 | 12.9 | 0.4 | 1.2 | 1.9 | 43.3 |
| NMSU 12-298 | 5.3 * | 18.6 | 8.8+ | 0.3 | 0.9 | 1.7 | 30.0 |
| NMSU 12-299 | 9.2 | 28.3 | 17.8 | 0.7 | 1 | 1.4 * | 60.0 |
| PI 289689 | 6.3 | 5.5 | 0.8 | 0.5 | 1.2 | 46.7 | |
| NMSU 10-618-1 | 4.2 | 8.2 | 1.1 | 0.4 | 1.1 | 2 | 40.0 |
| NMSU 12-285 | 7.2 | 4.8 | 0 | 0.2 | 1.1 | 1.9 | 13.3+ |
| PI 258956 | 6.7 | 16.1 | 47.4 | 0.4 | 0.9 | 1.1 | 36.7 |
| NMSU 10-634-1 | 10.4 | 11.6 | 13.7 *** | 0.3 | 0.9 | 1.3 | 30.0 |
| NMSU 12-289 | 9.2 | 18.8 | 22.1 *** | 0.4 | 1 | 1.2 | 40.0 |
| PI 239633 | 0.8 | 6.5 | 9.1 | 0.5 | 0.9 | 1.3 | 50.0 |
| NMSU 10-589-1 | 5.1 | 9.3 | 12.7 | 0.9 | 1 | 1.4 | 83.3 |
| NMSU 12-283 | 5.3 | 14 | 26.1 * | 0.7 | 1 | 1.4 | 70.0 |
| NMSU 10-596-1 | 2.2 | 4.2 | 16.6 | 0.4 | 1.2 | 1.4 | 33.3 |
| NMSU 12-325 | 11.2 * | 20.1+ | 20.6 | 0.4 | 1.3 | 2.5 ** | 43.3 |
| Rumba | 21.2 | 34.9 | 33.6 | 0.6 | 1 | 1.8 | 56.7 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Singh, N.; Cramer, C.S. Improved Tolerance for Onion Thrips and Iris Yellow Spot in Onion Plant Introductions after Two Selection Cycles. Horticulturae 2019, 5, 18. https://doi.org/10.3390/horticulturae5010018
Singh N, Cramer CS. Improved Tolerance for Onion Thrips and Iris Yellow Spot in Onion Plant Introductions after Two Selection Cycles. Horticulturae. 2019; 5(1):18. https://doi.org/10.3390/horticulturae5010018
Chicago/Turabian StyleSingh, Narinder, and Christopher S. Cramer. 2019. "Improved Tolerance for Onion Thrips and Iris Yellow Spot in Onion Plant Introductions after Two Selection Cycles" Horticulturae 5, no. 1: 18. https://doi.org/10.3390/horticulturae5010018
APA StyleSingh, N., & Cramer, C. S. (2019). Improved Tolerance for Onion Thrips and Iris Yellow Spot in Onion Plant Introductions after Two Selection Cycles. Horticulturae, 5(1), 18. https://doi.org/10.3390/horticulturae5010018





