Abstract
The use of fertigation, coupled with micro-irrigation, has continued to increase since it was first introduced in horticultural cropping systems. This combination provides a technical solution whereby nutrients and water can be supplied to the crop with high precision in terms of time and space, thereby allowing high nutrient use efficiency. However, the correct estimation of crop nutrient and water needs is fundamental to obtaining precise plant nutrition and high nutrient use efficiency in fertigated cropping systems. This paper illustrates the state-of-the-art and new perspectives for optimal nutrient management of vegetable crops cultivated under fertigation regimes. An overall description is reported for the most valuable technologies and techniques based on simulation models, soil testing, plant testing, and related decision support systems that can be adopted for efficient fertigation. However, it should be highlighted that only a few of the above technologies and techniques are practically available and/or easy to use by growers. Therefore, much more attention should be paid in the future to the transfer of research knowledge to farmers and technical advisors.
1. Introduction
Fertilization and irrigation are two of the most important factors in crop production, as they strongly affect the yield and quality of cultivated crops [1,2,3]. The total area cultivated by micro-irrigation worldwide increased from 1.1 million ha in 1986 [4] to roughly 11.1 million in 2015 [5]. This astonishing growth in the use of micro-irrigation is mainly due to (i) its high irrigation efficiency (up to 95% of applied water [6]), which partially contributes to solving the water shortage problems associated with both poor quality and low quantity in some cultivated areas and (ii) the progressive development of irrigation technologies that have substantially reduced the cost of equipment for micro-irrigation systems.
Horticultural crops are generally high-value, so irrigation is fundamental for obtaining high yield and quality for open-field-vegetable crops, and it is required for protected vegetable crops [7].
Fertigation is the agronomic operation in which fertilizer is dissolved in the irrigation water and delivered to the root zone by the irrigation system [8]. This combination provides the technical capacity for precise mineral nutrition, both spatially and temporally. The first scientific application of fertigation was in 1958 in the USA [9] using sprinklers while the combination with drip irrigation was first applied in Israel on tomato crops [10].
Fertigation generally allows for a significant increase of nutrient use efficiency in terms of plant nutrient recovery, with much higher results (up to 90%) than in other fertilizer application systems (40–45%) [11,12]. The main advantages of fertigation are the increased flexibility to split the fertilizer dose according to the uptake rate of the crop, an improved distribution of fertilizer in the root zone, and the possibility of maintaining a low (but constant) nutrient level in the soil solution. As a consequence, the use of fertigation has been found to reduce the run-off of mobile nutrients such as N by up to 70% compared with conventional fertilizer applications [12]. Other advantages and the main drawbacks of fertigation are summarized in Table 1.

Table 1.
The main advantages and drawbacks of fertigation.
Micro-irrigation systems enable higher water use efficiency only if combined with precise irrigation scheduling. In many areas of the world, the adoption of drip irrigation without precise irrigation management has not increased the water use efficiency of vegetable crops, and run-off of nutrients (mainly N) from the root zone has been observed. For example, in the greenhouse district of Almeria (Spain), Thompson et al. [13] reported that, in the last 20 years, the concentration of N (as nitrates, NO3−) in the aquifers increased from a few mg L−1 to more than 100 mg L−1. Similar results were observed in central California [14].
However, in the present review, irrigation scheduling is not analyzed in detail, as other reviews on this topic are reported in this special issue [15,16,17]. The state-of-the-art of the most used and promising methods for the optimal nutrient management of fertigated crops are here reported, which include soil testing, crop monitoring techniques and tools, and Decision Support Systems (DSSs).
2. Nutrients Applied by Fertigation
In theory, all water-soluble fertilizers could be applied through fertigation, but, for many years, the supply of P and microelements by fertigation was not advised in practice, as their precipitation can easily occur when the pH is higher than 7.0, resulting in clogging of the irrigation system [3,18].
Nitrogen can be applied in different chemical forms, the most common being nitrate (potassium nitrate, calcium nitrate, and magnesium nitrate salts), ammonium (ammonium nitrate, ammonium sulphate), and urea.
Nitrate is an anion not retained by the soil cation exchange capacity (CEC), thereby resulting in the most mobile nutrient beyond the crop root zone. Many authors have reported that NO3− moves in the soil transported by the irrigation water. Nitrate tends to accumulate at the periphery of the wetted soil volume and when water is applied in excess respect to the soil water capacity, nitrate is transported to the deeper soil layers (up to 100–150 cm), outside the crop root zone [3,12,19].
Ammonium (NH4+) is a cation retained by CEC; it is not very mobile in the soil and moves less than 10–15 cm from the emitter [20].
Urea is a very highly water-soluble compound and is not adsorbed by colloids; it moves in the soil easily, until its transformation into NH4+ [12,19]. The distribution of urea in the root zone profile depends on the length of irrigation and the characteristics of the soil. Haynes [21] reported that, in sandy soil, at the end of a fertigation with urea or NO3−, the distribution in the root zone for the two different fertilizers was similar. Both were more evenly distributed vertically and laterally in the soil profile up to 15 cm from the emitter.
Ammonium and urea are nitrified and contribute to the NO3− distribution and acidification in the root zone.
Phosphorus is present in the soil solution in anionic forms, but it is strongly adsorbed by the colloids of the soil. In general, it tends to accumulate within a few centimeters around the emitters. Therefore, the use of phosphate salts in fertigated systems has been previously limited [3,19]. Currently, phosphoric acid or urea-phosphate are instead commonly applied; in fact, their use contributes to prevention of possible clogging due to the precipitation of Ca and Mg and ensures a better distribution of P in the soil.
Potassium, like NH4+, is a nutrient subject to CEC retention, and it moves slowly in the root zone. In some conditions (low CEC and high K concentration in the fertigation solution), K can move beside the waterfront, with a better distribution along the wet root zone. In these conditions, K run-off can occur [3,19].
The application of metallic micronutrients (iron, manganese, zinc and copper) through irrigation water requires the use of chelated forms (mainly ethylenediaminetetraacetic acid, EDTA and ethylenediamine-N,N’-bis(2-hydroxyphenylacetic acid, EDDHA) to avoid precipitation [12].
The observed nutrient distribution in fertigation is mainly due to the specific characteristics of the single ions, and soil hydraulic and chemical properties (i.e., CEC) [22]. Nevertheless, other factors can contribute to reducing or increasing ion mobility in the soil; for example, ion dynamics in the soil also depend on fertigation and irrigation frequency.
Frequent supplies of low amounts of nutrients with irrigation water increased the availability of N, P and K in the root zone, and thus increased the yield and quality of tomatoes [23,24]. Badr and El-Yazied [25] tested the influence of different fertigation frequencies, (from one to 14 days) on tomatoes with two different amounts of N (200 and 300 kg N ha−1). The total crop N uptake was considerably higher in the treatment with the more frequent applications. The authors concluded that the supply of N in small and frequent doses (i.e., every day): (i) reduces the variation in nutrient concentration in the root zone and possible related salinity stress; (ii) increases nutrient availability for the crop; and (iii) reduces the risk of N leaching. Shedeed and colleagues [23] found that the daily application of N, P and K with irrigation water in tomato increased the availability of these nutrients in the root zone, improved yield and quality of the crop and reduced the N and K losses, particularly in sandy soils. However, other authors have reported that frequent fertigation regimes are not easy to manage and can increase water losses due to evaporation from the constantly wet soil surface [26]. In general, due to the low mobility in soil, many growers prefer to apply P pre-plant, together with 20–30% of total planned N and K fertilizers.
3. Optimal Fertigation Management
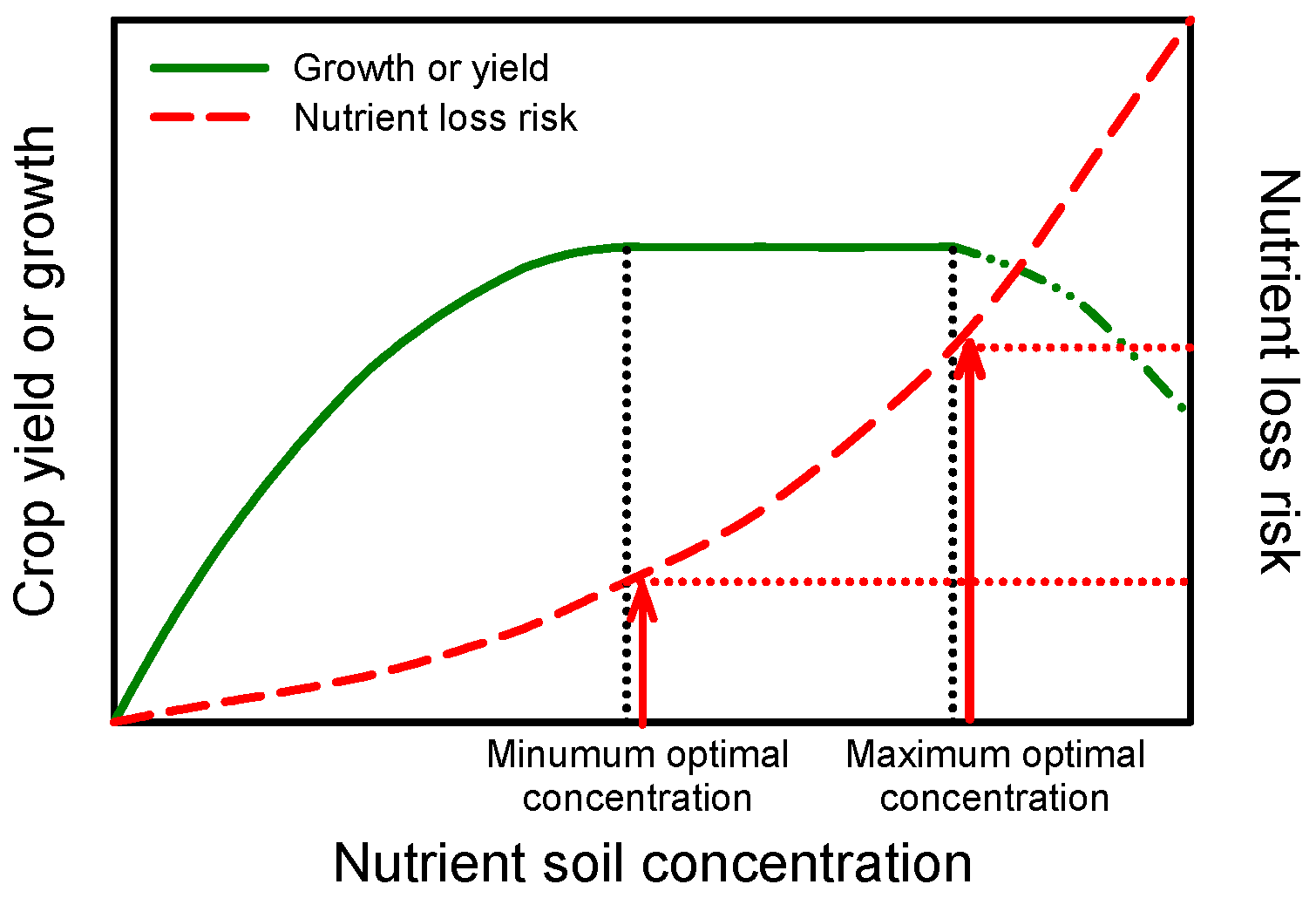
Figure 1 illustrates the relationship between nutrient soil concentration and the risk of fertilizer losses due to nutrient leaching (NO3 and K) or fixation (P and K). At a low soil nutrient concentration, plant growth and yield are limited. The increase of soil nutrient concentration produces an increase of crop growth/yield until a minimum optimal concentration of the considered nutrient in the root zone is achieved. Generally, exceeding the limit of the maximum optimal nutrient concentration will cause a reduction in produce quality, and then in growth and yield. Between the minimum and maximum, no significant response can be observed within a large soil nutrient concentration range.
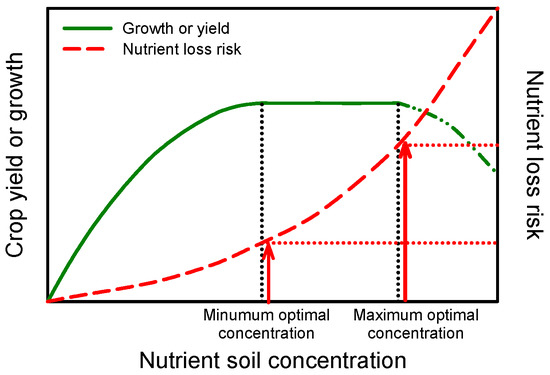
Figure 1.
Relationship between crop growth (or yield) and nutrient soil concentration in the root zone. The maintaining of the minimum optimal nutrient concentration allows for reducing nutrient loss risk (e.g., NO3− leaching) without affecting crop produce and quality.
For N management, many authors have reported that when a minimum optimal concentration of NO3− is present in the soil before sowing or transplanting, the maximum yield is guaranteed [27,28,29,30,31]. Generally, NO3− leaching is positively correlated with the concentration of NO3− in the soil [32]. The maintaining of a constant nutrient optimal concentration over the crop cycle reduces the risk of nutrient losses from the root zone, due to excessive rainfall or irrigation (Figure 1) [11,33].
The main goal in efficient fertigation management is to satisfy both the crop needs and to minimize nutrient losses. To attain this goal, it is necessary to accurately estimate three main variables: (i) crop nutrient requirement; (ii) soil nutrient availability; and (iii) nutrient delivery to match crop development.
Knowledge of the soil nutrient availability is critical for optimized fertigation management. Very often soils cultivated with vegetable crops contain an excessive amount of nutrients; in the above case, a pre-transplanting soil analysis is strategic to avoid the over-application of fertilizers.
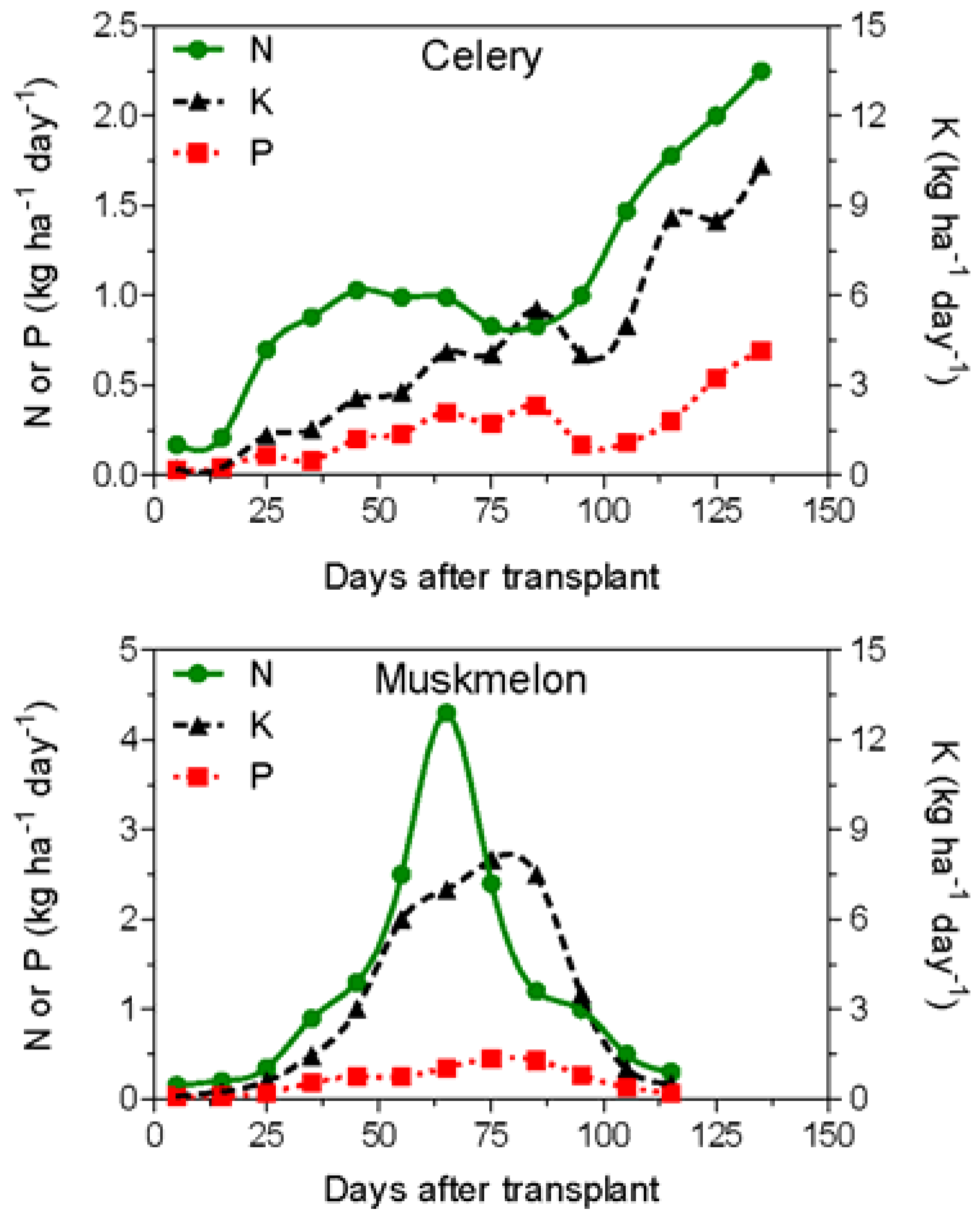
In the last 40 years, many authors have studied the nutrient requirement over the whole growth cycle for the most important vegetable crops. In general, N, P, and K crop uptake is correlated with crop biomass accumulation. Two different uptake patterns can be identified for vegetable crops (Figure 2): (i) leafy vegetable crops, such as lettuce, spinach, and celery, where the nutrient uptake curve can be well represented by exponential functions, with low nutrient requirement in the first part of the growing cycle and with a continuous increase of the nutrient uptake until the harvest; (ii) fruit crops (tomato, melon, pepper, etc.) characterized by a relatively low nutrient uptake until bloom and a maximum nutrient requirement during bloom and early-fruit development.
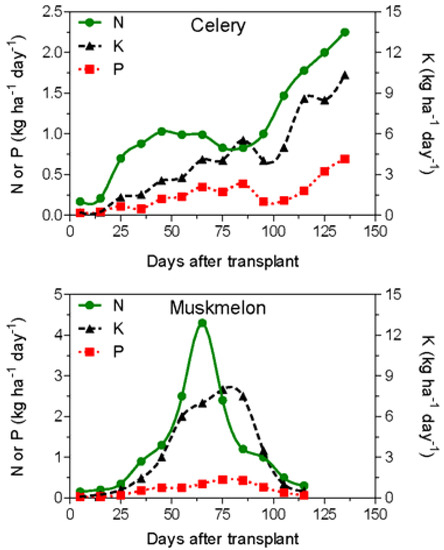
Figure 2.
Characteristic nutrient uptake rates of a fertigated leafy vegetable (celery; rearranged from Feigin et al. [39]) and a fruit crop (muskmelon; rearranged from Sagiv et al. [40]).
Generally, during the fruit development and harvesting phases (only for fruit vegetables), the nutrient crop uptake declines [27]. Table 2 reports data on nutrient crop uptake (expressed as kg ha−1 day−1) as a function of time after sowing or transplanting for some leafy and fruit vegetables: these data can be used for the calculation of the total amounts of nutrients to be supplied to the crop by fertigation.

Table 2.
Daily absorption rate of nitrogen (N), phosphorus (P) and potassium (K) (kg ha−1 day−1) of selected vegetables grown under drip irrigation after emergence or planting (adapted from Bar-Yosef [3]).
Although many new technologies have been developed and validated for precise nutrient and irrigation management, they have not been sufficiently exploited by growers. In practice, nutrient and water supply in cropping systems are mostly based on growers’ experiences, which is mainly of static (fixed) fertilization plans [13].
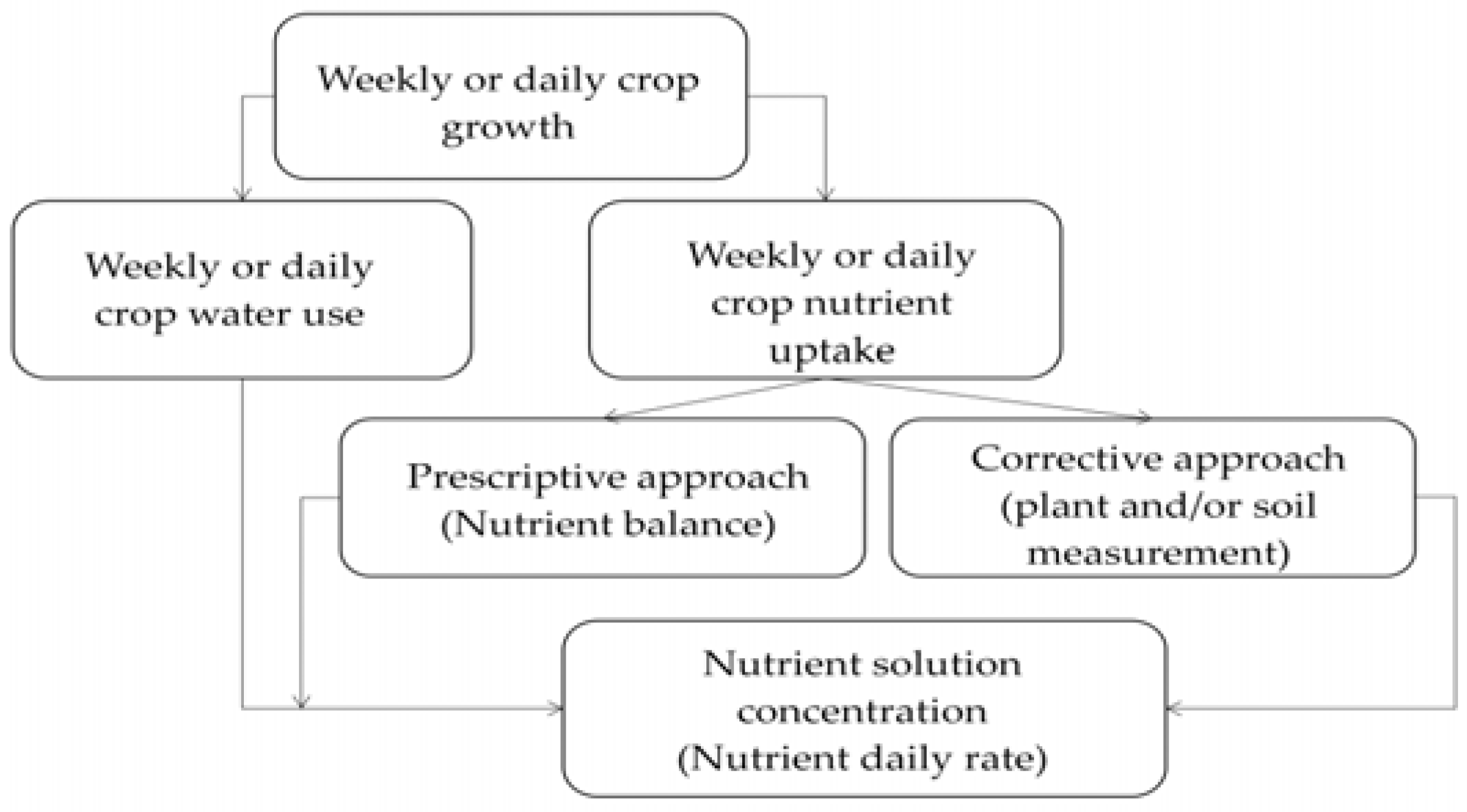
Two main approaches can be taken for efficient fertigation management (Figure 3). In the first case, the fertilizer dose can be estimated before cultivation, taking into account all variables that may change the concentration of nutrients in the root zone and data collected on the cultivation system, which are then used to draw in advance a “prescriptive” fertilization plan [41]. Following this approach, the desired (target) nutrient concentration can be calculated as the ratio between the amount of nutrient and water taken up by the crop in the same time period (Figure 3). In the second case, fertigation management is accomplished through a “corrective” approach: a nutrient solution is applied to the crop and its nutrient composition is periodically adjusted on the basis of plant and/or soil measurements, in order to avoid nutrient excesses or deficiencies [41]. Both approaches can be integrated for the same crop: the prescriptive fertilizer dose is then calculated before planting, and afterwards the fertilizer supply rate is fine-tuned according to the seasonal variations of crop nutritional status, detected by soil and/or plant measurements.
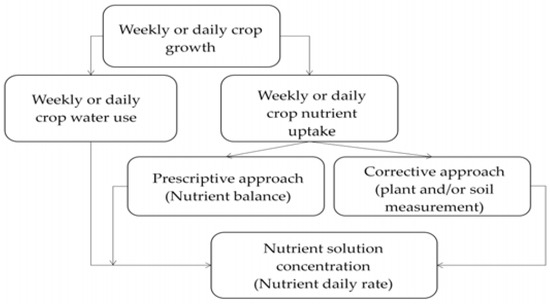
Figure 3.
Conceptual framework for the calculation of the fertilizer supply rate in fertigated crops, following different theoretical approaches.
4. Techniques for Prescriptive Fertigation Management
4.1. Simulation Models
Crop water and fertilizer requirements can both be estimated through simulation models. Most simulation models developed for agricultural purposes are algorithms based on mathematical formulas. Modeling cropping systems can predict (quantify) variables, which are useful for crop management as a function of other easier-to-measure variables. Mathematical models can then simulate crop nutrients and water requirements and can eventually be used to manage the nutrient supply in fertigated cropping systems. Mathematical models of plant nutrition have been applied at different micro and macro levels, from the cellular scale to the whole crop and for different purposes [42]. For example, mathematical models have been developed to simulate antagonisms among nutrient and non-nutrient ions at the root level [43], nutrient uptake at the whole plant level [44] or the effect of non-nutrient ions on plant growth and yield [45]. Modeling plant nutrition can be accomplished through static or dynamic models with regard to time [42]. Static models are based on fixed variables predetermined on the basis of averaged values such as expected historical yield and historical climate data. Dynamic models relate to real-time climate measurements or forecasted climatic data.
Many simplified models are based on the concept that the quantity of nutrient absorbed per water unit—namely nutrient uptake concentration—can be simulated per se as a function of plant growth stages, climate parameters, thermal time (i.e., growing degree days), and nutrient and non-nutrient (e.g., saline ions) concentration in the root zone, either with static or dynamic approaches. Based on these parameters, the nutrient requirements of tomatoes [46,47], melons [48], and other horticultural crops have been estimated [49,50]. In some cultivation systems, where the supply of fertilizers is practically equal to plant nutrient uptake (i.e., a soilless culture), uptake concentration has been used to simulate the nutrient requirement of horticultural crops to optimize nutrient management in closed and semi-closed cultivation systems [46,51]. The decision support system (DSS) Hydrotools is one example of a simulation model developed for the management of soilless cultivations based on the uptake concentration concept [52]. Using the same concept, Dutch researchers have developed the “Fertigation model” to calculate the crop nutrient demand of soil-grown greenhouse crops [49,50].
More complex simulation models estimate plant biomass accumulation and nutrient tissue concentration; the crop nutrient uptake can be estimated from the combination of these two parameters. This approach has been used at different levels of complexity to estimate the nutrient uptake of different vegetable species [53,54,55,56]. Plant biomass accumulation can be simulated following both empirical and mechanistic approaches. For the latter, non-rectangular hyperbola and other nonlinear equations have been proposed to fit the photoassimilation response to intercepted radiation [42,57,58,59], air carbon dioxide concentration and temperature [60,61], tissue age [61], nutrient tissue concentration [62,63], etc. Empirical models have been used to simulate biomass accumulation and plant phenology, as in the case of the Tomgro, a simulation model developed for tomato cultivation [64]. The variation of nutrient concentration in plant tissues can be simulated using different approaches [42] from empirical equations to more mechanistic models, which take into account the ion influx rate at root level [65,66].
However, crop nutrient uptake is not the only variable to be taken into account for the estimation of the crop fertilizer requirement. Under operative conditions, fertilizer delivery and recovery at the crop level are affected by inefficiencies, mostly due to both the adoption of poor techniques and the interference of environmental factors. However, the supply of fertilizer is not the only source of nutrients for the cropping system. To estimate the nutrient balance in the root zone, many models have been proposed that take into account the nutrient mineralization from organic materials and the nutrient losses due to immobilization, fixation, volatilization and leaching of different nutrient ions, [67,68,69,70,71]. Other tools have been developed to simulate the nutrient fluxes in the root zone, such as the Fussim [72] and Hydrus [73] models.
4.2. Decision Support Systems Based on Simulators
Depending on the cultivation system and simulation complexity, a number of models can be assembled together and integrated into more complex simulation tools, which can then simulate different processes involved in crop fertilization. These tools are known as decision support systems (DSSs), as they can give advice on the quantity of fertilizers to be delivered to the crop, taking into account the interactions between plant fertilization practice and many variables other than plant nutrient uptake. A compendium of the most potentially useful DSSs for fertigation management is provided in Table 3.

Table 3.
The main Decision Support Systems (DSSs) for fertigation management. For each DSS, the reference of the inventor(s), the main input and output, and the eventual validation work of the model are reported.
The DSS CropSyst [74] was originally developed for extensive cropping systems and has been successfully adapted to simulate the water and nutrient requirements of vegetable species [75]. Nitrogen and water balance can be simulated with the DSS VegSyst, which has been found to be effective for the fertilization of melon [56], sweet pepper [76] and tomato [77]. Another worthy example is EU-Rotate_N [78], a DSS that simulates the most significant N balance variables and can be applied for the fertigation control of horticultural crops and for the assessment of environmental impact indices, due to N leaching from the root zone. EU-Rotate_N has been successfully applied to simulate water/nitrogen uptake and leaching of horticultural crops cultivated in the Mediterranean area [79] and China [80]. Suarez-Rey and colleagues [81] calibrated and validated EU-Rotate_N on escarole and lettuce, and observed up to 57% reduction in the N supply, compared with standard growers’ practice. Based on EU-Rotate_N, Zhang and colleagues [82,83] developed and tested the SMCR_N model, which implements more mechanistic algorithms for the simulation of water and nutrient fluxes in the root zone. This allows for the simulation of crop yield response, N uptake and leaching for many different species of vegetable crops with a high level of accuracy.
The Fertirrigere DSS simulates water and nutrient (macronutrients) balance in the root zone and gives suggestions on the quantity of salts to be dissolved in the irrigation water for optimal fertigation management of processing tomatoes [54,55]. The application of Fertirrigere improved both the environmental and economic sustainability of crops in trials conducted under Mediterranean climate conditions [84]. GesCoN is a DSS that simulates water and N balance for the optimized fertilization management of vegetable crops grown in open areas [85]; the DSS has been validated in experimental trials carried out in the Mediterranean area and Florida [86].
In recent years, DSSs for agricultural purposes have rapidly become more widespread, due to the increased availability of computer Internet connections (e.g., Wi-Fi) and microcomputers including laptops, smart phones, and tablets. The DSSs can be stand-alone or web-based programs. The online DSS CropManage (https: cropmanage.ucanr.edu) has been developed to simulate both irrigation and N fertilizer requirements specifically for leafy vegetables grown in the central coast of California [87]. The software recommends fertilizer N based on keeping mineral N concentration in the root zone close to a minimum optimal soil threshold. In experimental trials, the use of this software reduced N fertilizer inputs by 30% with respect to the fertilizer plan of the conventional grower.
The nature of these simulation models and the derived DSSs means that they are affected by uncertainty. The number of parameters and their complexity can be a bottleneck in terms of their application: usually the more complex models are the most accurate and are more flexible in adapting to different scenarios, but they also require much more effort for calibration and/or use in different cultivation systems. However, modelling represents the only or at least one of the most valuable strategies to (i) estimate variables that are difficult to measure directly; (ii) simulate scenarios for making decisions in advance; (iii) simulate the behaviour of large cultivated area; and (iv) for training and educational programs.
Undoubtedly, a simulation-based DSS can be a useful tool for supporting fertigation management and making decisions in fertigated crops. However, it should be noted that only a few DSSs are practically available and/or easy to use by growers (e.g., CropManage, Fertirrigere).
5. Techniques for Corrective Fertigation Management
5.1. Plant Monitoring
Crop monitoring through different analytical approaches can be used to assess plant nutrient status and eventually to correct nutrient delivery in terms of both quantity and type of nutrients. The approach known as “speaking plant” or “plant testing” is based on the principle that the control of growing conditions should be based on the physiological status of the plant [99]. To accomplish the above goal, both direct and indirect measurements of the nutrient status in plant tissues can be carried out in the field.
Over the past 20 years, this approach has been mainly investigated in terms of N nutrition [100] because of its role in plant metabolism and for the environmental implications derived from N fertilizers when applied in excess. The main characteristics of a reliable “speaking plant” method are the ease and rapidity of the measurement, which allow for a sufficient number of measurements to obtain crop-representative data. The first “speaking plant” technique to be developed was petiole sap analysis, which is probably still the most common.
The analysis of the sap extracted from a leaf petiole has been used in many studies, for monitoring plant nutritional status and eventually optimizing plant fertilization. A significant correlation between NO3− and K concentration in petiole sap and N or K content in plant tissues has been observed for many vegetable crops [98,101,102]. However, experiments have highlighted the limits of this technique, and have shown that optimal values of nutrient concentration in the leaf petiole sap change as a function of (i) crop stage (values of sufficiency decline with crop age); (ii) cultivation system; (iii) species and cultivar and (iv) plant nutrient and water status [98].
Knowledge of the guide-values is thus critical to successfully apply the method, and this remains the main limitation for its practical application. However, reference values for N and also K are available for different types of vegetables grown in Florida [103] (Table 4), for melon and tomato in Almeria (Spain) [102], for potatoes in central and northern Europe [98], and for industrial tomato production in central Italy [101].

Table 4.
References values for different vegetable crop grown in Florida (Source: Hartz and Hochmuth [27]; Hochmuth [103]).
One of the most promising technologies for plant monitoring are optical sensors, which are developing rapidly. Tissue (leaf) chlorophyll fluorescence, reflectance, and transmittance can be measured with these sensors, thus obtaining crop indices that can be correlated with plant stress and nutrient status [104].
The optical measuring devices can be divided roughly into sensors able to measure portions of a plant or individual leaves and those able to acquire data from large parts of the canopy, and that could be installed on tractors, drones, robots, or even on airplanes or satellites. Among the portable instruments, the most widely used are the chlorophyll meter SPAD-502 (based on single-photon avalanche diode (SPAD) technology), originally developed by Konica-Minolta (Osaka, Japan), the Hydro N-tester (Yara International, Oslo, Norway) and Dualex Scientific+® (FORCE-A, Paris, France). The SPAD produces a dimensionless index positively correlated to the concentration of chlorophyll in the leaf blade, which is in turn related to the concentration of N in plant tissues. Once the values of N sufficiency are assessed, the corresponding SPAD index value can be used to support N fertilization [104,105,106,107]. Some authors have found variability in the SPAD index when the culture was carried out in the presence of abiotic stress [106]. SPAD measurement variability can be reduced after in-situ calibration of the instrument (e.g., non-fertilized parcel), as, for example, recommended by the manufacturers of some chlorophyll meters (i.e., N Tester® Yara, Yara International, Oslo, Norway). The variability of the SPAD index and the need to calibrate it for each species, cultivation conditions, etc., are the major reasons why, despite the fact that the first experiments occurred more than 20 years ago, commercial use for the management of fertilization has up until now been quite limited.
A new promising instrument is the Dualex Scientific+® (FORCE-A, Paris, France). The Dualex® can provide four different indices related to the concentration of chlorophyll, flavonols, anthocyanins and N derived by the nitrogen balance index (NBI®); it can be used to assess the nutritional status of the crop and the presence of stress [108]. The Dualex® has been used successfully by Tremblay and colleagues [109], for N fertilization of winter vegetable crops in the open field. The authors also observed a high correlation between the indices obtained with the Dualex®, the SPAD and nitrate concentrations in the leaf petioles.
Wu and colleagues [110] evaluated the effectiveness of various methods to manage the N nutrition of potatoes by comparing the SPAD index, the analysis of the leaf petiole sap and use of images acquired by QuickBird satellite (http://www.satimagingcorp.com/satellite-sensors/ quickbird/) with different types of spectral resolution for the calculation of the normalized difference vegetative index (NDVI). The best results were provided by the analysis of the petiole sap followed by SPAD, while information derived from satellite images were of little value for the practical management of the N fertilization.
Muñoz-Huerta et al. [104] published a review work summarizing the characteristics of optical sensors, including those based on the measurements of tissue reflectance. These types of sensors can be divided into two main categories: (i) those that measure the reflectance of the canopy through a multispectral sensor dependent on passive sunlight, such as CropScan® (Cropscan, Rochester, NE, USA) and FieldSpec® (ASD PANalytical, Boulder, CO, USA); (ii) those that measure reflectance by producing the source light themselves, thereby providing a more precise measurement independent of the ambient light, such as N Sensor™ (Yara, Grimsby, UK), Crop Circle® (Holland Scientific, Lincoln, The Netherlands), and GreenSeeker® (Trimble, Sunnyvale, CA, USA). Most research on reflectance meters has been conducted with cereal crops, although some studies are available for vegetable crops [107,111,112].
The use of multispectral images for estimating the nutrient content of the canopy and nutrient supply management have been assessed in many reports [113,114]. Canopy reflectance measurements are based on the relationships between visible and near infra-red (NIR) wavelengths and the crop canopy status [115,116]. The reflectance value of specific wavelengths can be used in the computation of different indices, of which the NDVI is the most common [104]. Indices related to healthy and stressed crops can be compared for the interpretation of collected data.
Finally, the “speaking plant” approach, which improves fertilization management, has been mainly applied in extensive cropping systems, where the growing cycles are relatively long and the growers have enough time to correct mineral fertilization if necessary. However, this approach cannot be successfully applied to vegetable crops that are characterized by short growing cycles (20–40 days) such as salad vegetables and radish crops.
5.2. Root Zone Monitoring
Soil testing to monitor nutrient availability in the root zone is a valuable alternative to plant monitoring. Fertigation can be modulated as a function of a target (i.e., optimal) concentration or quantity per soil volume of fertilizer, to be maintained in the root zone and to achieve optimal yield and quality for the crop. Following this approach, the nutrient availability in the root zone can be assessed through different monitoring techniques: (i) laboratory analysis in which nutrients are extracted by solutions containing barium chloride, ammonium acetate and calcium chloride, for example; (ii) analysis of soil or substrate aqueous extract (e.g., 1:2 V:V soil/water; [117]; and (iii) analysis of soil water solution extracted from the root zone by suction lysimeters [118].
Detailed laboratory analyses of soil samples are more useful for pre-transplant fertilization and for the evaluation of nutrient availability in the medium-long period, while for quick adjustments of fertigation during the cultivation cycle, the use of aqueous extracts appears to be more effective [100].
Over the last 20 years, Dutch researchers have developed corrective fertilization protocols for many greenhouse crops, based on the chemical analysis of aqueous extracts (1:2 V:V). Samples from the root zone are collected each 3–6 weeks, depending on the crop, and analyzed for target nutrients. The nutrient solution can then be adjusted after comparing analyzed values with pre-established thresholds, as shown in Table 5 [117,119]. The frequent nutrient addition with low water volume supplied by fertigation means that the concentration of nutrients in the aqueous extract is highly representative of the root zone nutrient status. The method can also be applied for monitoring saline (ballast) ions (e.g., Na, Cl) or simply EC.

Table 5.
Some examples of guideline nutrient concentrations (mmol L−1) and maximum electro-conductivity values (EC, dS m−1) of aqueous soil extracts (1 volume of wet soil: 2 volumes of water) for the base and dressing fertilization of some protected horticultural crops. The standard nutrient solutions advised for protected soil-grown vegetables cultivated in Dutch climate conditions are also reported.
Suction lysimeters allow an easy sampling of the soil solution in the root zone. This consists of a porous cup hermetically welded on a pipe into which a capillary is inserted. The capillary enables the collection of the circulating solution extracted from the soil after applying a vacuum (maintained for 24–36 h after the end of irrigation), using a pump or a simple syringe. The method has been widely investigated in Israel, Spain and Australia and has been found effective for the management of N in greenhouse crops. However, the uncertainty of the measurements, due to the large spatial variation of the soil nutrient solution concentration, is a weak point that limits the commercial use of this technique [118]. For example, Hartz and Hochmuth [27] proposed using them in vegetable crops with a threshold value of 5 mmol L−1. Hartz [120] later concluded that errors due to the high spatial variability of the soil solution may limit their application. Experimental work on tomatoes and melons has been carried out in protected cultivation but revealed a poor correlation between the nutritional status of the plant and the concentration of nutrients in the suction cup extract; the method therefore appears to be more suitable when excesses are avoided [102]. The aqueous extract obtained through different methods can be analyzed with a rapid determination kit and other analysis tools [121,122,123].
Several rapid chemical analysis systems are on the market that can be used to monitor the concentration of nutrients in water extracts, but the most effective are based on the use of photometry with visible light and ion-specific sensors such as ion selective optodes (ISO), ion selective electrodes (ISE), and ion-selective field effect transistors (ISFEs). There are many simple and portable photometers available (e.g., RQ Reflectoquant Flex™, Merck KGaA, Darmstadt, Germany) and several rapid kits based on color reactions.
Undoubtedly ion-specific sensors based on optical (ISO) or electrochemical (ISE or ISFET) reactions are particularly suitable for the fertigation of soilless crops where the nutrient solution can be easily collected [122]. Commercial examples of portable specific ion sensors are CardyNO3 Meter™, (Horiba Ltd., Kyoto, Japan) for the determination of NO3−, and the Nutrient Analyzer™ (Clean-grown, Wolverhampton, United Kingdom) for the determination of N-NO3, N-NH4, Ca, Mg, K, Cl, Na, pH and EC.
Some studies have recently proposed the use of NIR reflectance for the estimation of N and P content directly on the soil samples without extraction treatments [124,125,126]. Here, the measurement is carried out through spectroscopes or instruments in which the light source is provided by LED lamps emitting monochromatic light at wavelengths in the absorption range of the investigated element. In the future, these sensors could potentially estimate different chemical elements in soil.
5.3. Decision Support Systems Based on Crop Monitoring
Notwithstanding the extensive research activity recently conducted on plant monitoring for assessing crop nutritional status, very few applicative protocols have been developed based on this approach. The high variability of nutrient concentration in plant tissues and the need for knowledge of their respective critical thresholds, which may change as a function of cultivated species, plant/organ age, cultivation system and pedo-climatic conditions, may limit the application of this approach under a wide range of conditions, which is an important aspect when transferring technologies into practical operations.
For the above reasons, most DSSs based on corrective approaches relate to soil monitoring or mixed soil/plant monitoring methods. This approach was proposed by [97] with the CRA-W model; the DSS predicts the total N to be supplied as a function of expected yield and soil analysis, and 70% of the above quantity is given at transplant while the remaining 30% is supplied if necessary after evaluating plant status by rapid tissue analysis (e.g., using chlorophyll meters). The KNS (Kulturbegleitende Non-Sollwerte) method has been proposed for vegetable crops in Germany [93]; this DSS computes the N balance based on the assumption that a minimum nutrient buffer must be maintained in the root zone to ensure high yield and quality. KNS has been tested on 21 different vegetables with an average N reduction of 57% compared with standard growers’ practice [94]. An upgraded version of KNS is the N-Expert DSS [127]. However, the most popular approach based on soil testing is probably the “Fertigation Model” [49] initially developed by Dutch researchers [117,119] before being used across Europe. The method has been extensively adopted at the commercial level in the Netherlands [100], and by growers from Mediterranean countries such as Italy [52] and Greece [128]. In Italy, the method can be applied with the user-friendly software GreenFert [52] (http://www.cespevi.it/softunipi/greenfert.html).
6. Conclusions
Fertigation applied through micro-irrigation systems is undoubtedly one of the most effective strategies to improve nutrient use efficiency in agricultural systems. The possibility of supplying nutrients at a low rate and high frequency improves plant nutrient uptake and nutrient availability in the root zone, in addition to the reduced risk of nutrient loss. All of the above aspects positively influence the economic and environmental sustainability of agricultural activities.
When optimal management techniques are followed, the application of fertigation allows for high yield and quality of agricultural produce, and high income for growers. Scientifically-based criteria are adopted for determining the optimal type and application rate of fertilizers. The latter must be estimated on the basis of objective methodologies that take into account all of the agricultural system variables that can interact with the fertilization process. Accurate measurements are required to support the theory.
Many tools have been developed to accomplish the above goals, and other promising instruments and techniques will be soon available for the management of fertigated crops. Among all methods reviewed, decision support systems based on simulation models and soil testing approaches are the most widespread of these, but optical sensors appear to have much potential to integrate the former tools for precise control fertigation in intensive cropping systems.
Author Contributions
Luca Incrocci, Daniele Massa and Alberto Pardossi have contributed in equal part to the writing of the present article.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Goldberg, D.; Gornat, B.; Rimon, D. Drip Irrigation Principles, Design and Agricultural Practices; Drip Irrigation Scientific Publications: Kfar Shmariahu, Israel, 1976. [Google Scholar]
- Dasberg, S.; Bresler, E. Drip Irrigation Manual; Publ. No. 9; International Irrigation Information Center: Bet Dagan, Israel, 1985. [Google Scholar]
- Bar-Yosef, B. Advances in fertigation. In Advances in Agronomy; Sparks, D.L., Ed.; Academic Press: London, UK, 1999; Volume 65, pp. 1–77. [Google Scholar]
- Reinders, F.B. Micro-irrigation: World overview on technology and utilization. In Proceedings of the 7th International Micro-Irrigation Congress, Kuala Lumpur, Malaysia, 13–15 September 2007. [Google Scholar]
- International Commission on Irrigation and Drainage (ICID). Agricultural Water Management for Sustainable Rural Development: Annual Report 2015–2016; International Commission on Irrigation and Drainage: New Delhi, India, 2016; pp. 79–80. [Google Scholar]
- Howell, T.A. Irrigation Efficiency; Marcel Dekker: New York, NY, USA, 2003. [Google Scholar]
- Pardossi, A.; Incrocci, L. Traditional and new approaches to irrigation scheduling in vegetable crops. HortTechnology 2011, 21, 309–313. [Google Scholar]
- Hagin, J.; Lowengart, A. Fertigation for minimizing environmental pollution by fertilizers. Fert. Res. 1995, 43, 5–7. [Google Scholar] [CrossRef]
- Bryan, B.B.; Thomas, E.L. Distribution of Fertilizer Materials Applied through Sprinkler Irrigation Systems; Arkansas System Division of Agriculture Experiment Station: Fayetteville, AR, USA, 1958; p. 12. [Google Scholar]
- Sagiv, B.; Kafkafi, U. Fertilization and manuring of pepper plants in sandy soils. Hassadeh 1976, 56, 1726–1730. [Google Scholar]
- Agostini, F.; Tei, F.; Silgram, M.; Farneselli, M.; Benincasa, P.; Aller, M. Decreasing nitrate leaching in vegetable crops with better N management. In Genetic Engineering, Biofertilisation, Soil Quality and Organic Farming; Springer: Berlin, Germany, 2010; pp. 147–200. [Google Scholar]
- Solaimalai, A.; Baskar, M.; Sadasakthi, A.; Subburamu, K. Fertigation in high value crops—A review. Agric. Rev. 2005, 1, 1–13. [Google Scholar]
- Thompson, R.B.; Martínez-Gaitán, C.; Gallardo, M.; Giménez, C.; Fernández, M.D. Identification of irrigation and N management practices that contribute to nitrate leaching loss from an intensive vegetable production system by use of a comprehensive survey. Agric. Water Manag. 2007, 89, 261–274. [Google Scholar] [CrossRef]
- Harter, T.; Lund, J.R. Addressing Nitrate in California’s Drinking Water; Report for the State Water Resources Control Board; Center for Watershed Sciences, University of California: Davis, CA, USA, 2012. [Google Scholar]
- Alvino, A.; Marino, S. Remote and proximal sensing for precision agriculture and irrigation management. Horticulturae 2017, in press. [Google Scholar]
- Cahn, M. New approaches to irrigation scheduling of vegetables. Horticulturae 2017, 3, 28. [Google Scholar] [CrossRef]
- George, R. Sensor sense in irrigation management. Horticulturae 2017. submitted. [Google Scholar]
- Bar-Yosef, B. Advances in fertigation. Adv. Agron. 1999, 65, 1–77. [Google Scholar]
- Hanson, B.R.; Šimůnek, J.; Hopmans, J.W. Evaluation of urea-ammonium-nitrate fertigation with drip irrigation using numerical modeling. Agric. Water Manag. 2006, 86, 102–113. [Google Scholar] [CrossRef]
- Li, J.; Zhang, J.; Rao, M. Wetting patterns and nitrogen distributions as affected by fertigation strategies from a surface point source. Agric. Water Manag. 2004, 67, 89–104. [Google Scholar] [CrossRef]
- Haynes, R.J. Movement and transformations of fertigated nitrogen below trickle emitters and their effects on pH in the wetted soil volume. Fert. Res. 1990, 23, 105–112. [Google Scholar] [CrossRef]
- Abalos, D.; Sanchez-Martin, L.; Garcia-Torres, L.; van Groenigen, J.W.; Vallejo, A. Management of irrigation frequency and nitrogen fertilization to mitigate GHG and NO emissions from drip-fertigated crops. Sci. Total Environ. 2014, 490, 880–888. [Google Scholar] [CrossRef] [PubMed]
- Shedeed, S.I.; Zaghloul, S.M.; Yassen, A. Effect of method and rate of fertilizer application under drip irrigation on yield and nutrient uptake by tomato. Ozean J. Appl. Sci. 2009, 2, 139–147. [Google Scholar]
- Farneselli, M.; Benincasa, P.; Tosti, G.; Simonne, E.; Guiducci, M.; Tei, F. High fertigation frequency improves nitrogen uptake and crop performance in processing tomato grown with high nitrogen and water supply. Agric. Water Manag. 2015, 154, 52–58. [Google Scholar] [CrossRef]
- Badr, M.; El-Yazied, A. Effect of fertigation frequency from subsurface drip irrigation on tomato yield grown on sandy soil. Aust. J. Basic Appl. Sci. 2007, 1, 279–285. [Google Scholar]
- Simonne, E.; Studstill, D.; Hochmuth, R.C. Understanding water movement in mulched beds on sandy soils: An approach to ecologically sound fertigation in vegetable production. Acta Hortic. 2006, 700, 173–178. [Google Scholar] [CrossRef]
- Hartz, T.; Hochmuth, G.J. Fertility management of drip-irrigated vegetables. HortTechnology 1996, 6, 168–172. [Google Scholar]
- Everaarts, A.P.; de Moel, C.P. The effect of nitrogen and the method of application on yield and quality of white cabbage. Eur. J. Agric. 1998, 9, 203–211. [Google Scholar] [CrossRef]
- Hartz, T.K.; Bendixen, W.E.; Wierdsma, L. The value of presidedress soil nitrate testing as a nitrogen management tool in irrigated vegetable production. HortScience 2000, 35, 651–656. [Google Scholar]
- Heckman, J.R.; Morris, T.; Sims, J.T.; Sieczka, J.B.; Krogmann, U.; Nitzsche, P.; Ashley, R. Pre-sidedress soil nitrate test is effective for fall cabbage. HortScience 2002, 37, 113–117. [Google Scholar]
- Bottoms, T.G.; Smith, R.F.; Cahn, M.D.; Hartz, T.K. Nitrogen requirements and N status determination of lettuce. HortScience 2012, 47, 1768–1774. [Google Scholar]
- Tei, F.; Benincasa, P.; Guiducci, M. Nitrogen fertilisation on lettuce, processing tomato and sweet pepper: Yield, nitrogen uptake and the risk of nitrate leaching. Acta Hortic. 1999, 506, 61–67. [Google Scholar] [CrossRef]
- Lord, E.; Johnson, P.; Archer, J. Nitrate Sensitive Areas: A study of large scale control of nitrate loss in England. Soil Use Manag. 1999, 15, 201–207. [Google Scholar] [CrossRef]
- Bar-Yosef, B.; Kramer, S.; Ben Basat, S. Trickle Irrigation and Fertilization in the Northern Arava Valley in Israel: Fresh Tomatoes (cv. 675); Annual Report; Agricultural Research Organization: Bet-Dagan, Israel, 1982. [Google Scholar]
- Dafne, O. Nitrogen Effect on Dry Matter Production, Mineral Absorption, Yield and Quality of Processing Tomatoes; University of Jerusalem: Jerusalem, Israel, 1984. [Google Scholar]
- Bar-Yosef, B.; Kramer, S.; Ben Basat, S. Trickle Irrigation and Fertilization in the Northern Arava Valley in Israel: Egg plant; Annual Report; Agricultural Research Organization: Bet-Dagan, Israel, 1981. [Google Scholar]
- Feigin, A.; Sagiv, B. Broccoli Response to Manure and N Fertilization in a Loessial Soil in Southern Israel; Agricultural Research Organization: Bet Dagan, Israel, 1971. [Google Scholar]
- Bar-Yosef, B.; Sagiv, B. Trickle Irrigation and Fertilization of Iceberg Lettuce. In Proceedings of the 9th International Plant Nutrition Colloquium, Coventry, England, 22–27 August 1982; pp. 33–38. [Google Scholar]
- Feigin, A.; Sagiv, B.; Aviram, H.; Zipilevich, Y. Celery Response to Manure and N Fertilization in a Loessial Soil in Southern Israel; Report on Project 301–046; Agricultural Research Organization: Bet Dagan, Israel, 1976. [Google Scholar]
- Sagiv, B.; Bar-Yosef, B.; Eliah, E. Drip Irrigation and Fertilization of Spring Muskmelon at Besor; Pub. No. 17; Division of Scientific Publications, The Volcani Center: Bet Dagan, Israel, 1980.
- Giller, K.E.; Chalk, P.; Dobermann, A.; Hammond, L.; Heffer, P.; Ladha, J.K.; Nyamudeza, P.; Maene, L.; Ssali, H.; Freney, J. Emerging technologies to increase the efficiency of use of fertilizer nitrogen. In Agriculture and the Nitrogen Cycle; Mosier, A., Syers, J.K., Freney, J.R., Eds.; Island Press: New York, NY, USA, 2004; p. 344. [Google Scholar]
- Thornley, J.H.M.; Johnson, I.R. Plant and Crop Modelling: A Mathematical Approach to Plant and Crop Physiology; Clarendon Press: Oxford, UK, 1990; p. 670. [Google Scholar]
- Massa, D.; Mattson, N.S.; Lieth, H.J. Effects of saline root environment (NaCl) on nitrate and potassium uptake kinetics for rose plants: A Michaelis-Menten modelling approach. Plant Soil 2009, 318, 101–115. [Google Scholar] [CrossRef]
- Louison, L.; Omrane, A.; Ozier-Lafontaine, H.; Picart, D. Modeling plant nutrient uptake: Mathematical analysis and optimal control. Lect. Notes Pure Appl. 2015, 4, 193–203. [Google Scholar] [CrossRef]
- Karlberg, L.; Ben-Gal, A.; Jansson, P.E.; Shani, U. Modelling transpiration and growth in salinity-stressed tomato under different climatic conditions. Ecol. Model. 2006, 190, 15–40. [Google Scholar] [CrossRef]
- Massa, D.; Incrocci, L.; Maggini, R.; Bibbiani, C.; Carmassi, G.; Malorgio, F.; Pardossi, A. Simulation of crop water and mineral relations in greenhouse soilless culture. Environ. Model. Softw. 2011, 26, 711–722. [Google Scholar] [CrossRef]
- Thompson, R.B.; Gallardo, M.; Rodríguez, J.S.; Sánchez, J.A.; Magán, J.J. Effect of N uptake concentration on nitrate leaching from tomato grown in free-draining soilless culture under Mediterranean conditions. Sci. Hortic. 2013, 150, 387–398. [Google Scholar] [CrossRef]
- Pardossi, A.; Incrocci, L.; Malorgio, F.; Campiotti, C.A. The relationship between mineral nutrition and fruit yield components in melon grown in recirculating nutrient solution culture. Agric. Mediterr. 2004, 134, 8–14. [Google Scholar]
- Voogt, W.; Kipp, J.A.; de Graaf, R.; Spaans, L. A fertigation model for glasshouse crops grown in soil. Acta Hortic. 2000, 537, 495–502. [Google Scholar] [CrossRef]
- Voogt, W.; Steinbuch, F.; van Winkel, A. Evaluation of the ‘fertigation model’, a decision support system for water and nutrient supply for soil grown greenhouse crops. Acta Hortic. 2006, 718, 531–538. [Google Scholar] [CrossRef]
- Massa, D.; Incrocci, L.; Maggini, R.; Carmassi, G.; Campiotti, C.A.; Pardossi, A. Strategies to decrease water drainage and nitrate emission from soilless cultures of greenhouse tomato. Agric. Water Manag. 2010, 97, 971–980. [Google Scholar] [CrossRef]
- Incrocci, L. Gestire la nutrizione in serra con due software gratuiti. L’Inf. Agrar. 2012, 68, 50–51. [Google Scholar]
- Seginer, I. A dynamic model for nitrogen-stressed lettuce. Ann. Bot. 2003, 91, 623–635. [Google Scholar] [CrossRef] [PubMed]
- Battilani, A. Fertirrigere V2.11: A multi-target DSS to manage water and nutrient supply at macrozone level. Acta Hortic. 2006, 724, 111–118. [Google Scholar] [CrossRef]
- Battilani, A.; Bussières, P.; Dumas, Y. Fertirrigere: A simple tool-model for managing water and nutrient supply in drip-irrigated processing tomatoes. Acta Hortic. 2003, 613, 155–158. [Google Scholar] [CrossRef]
- Gallardo, M.; Giménez, C.; Martínez-Gaitán, C.; Stöckle, C.O.; Thompson, R.B.; Granados, M.R. Evaluation of the VegSyst model with muskmelon to simulate crop growth, nitrogen uptake and evapotranspiration. Agric. Water Manag. 2011, 101, 107–117. [Google Scholar] [CrossRef]
- Marcelis, L.F.M.; Heuvelink, E.; Goudriaan, J. Modelling biomass production and yield of horticultural crops: A review. Sci. Hortic. 1998, 74, 83–111. [Google Scholar] [CrossRef]
- Causton, D.R.; Dale, P.M. The monomolecular and rectangular hyperbola as empirical models of the response of photosynthetic rate to photon flux density, with applications to three Veronica species. Ann. Bot. 1990, 65, 389–394. [Google Scholar] [CrossRef]
- Wang, Y.P. A refinement to the two-leaf model for calculating canopy photosynthesis. Agr. For. Meteorol. 2000, 101, 143–150. [Google Scholar] [CrossRef]
- Cannell, M.G.R.; Thornley, J.H.M. Temperature and CO2 responses of leaf and canopy photosynthesis: A clarification using the non-rectangular hyperbola model of photosynthesis. Ann. Bot. 1998, 82, 883–892. [Google Scholar] [CrossRef]
- Lieth, J.H.; Pasian, C.C. A model for net photosynthesis of rose leaves as a function of photosynthetically active radiation, leaf temperature, and leaf age. J. Am. Soc. Hortic. Sci. 1990, 115, 486–491. [Google Scholar]
- Thornley, J.H.M. Dynamic model of leaf photosynthesis with acclimation to light and nitrogen. Ann. Bot. 1998, 81, 421–430. [Google Scholar] [CrossRef]
- Hikosaka, K. Optimality of nitrogen distribution among leaves in plant canopies. J. Plant Res. 2016, 129, 299–311. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.W.; Dayan, E.; Allen, L.H.; van Keulen, H.; Challa, H. A dynamic tomato growth and yield model (TOMGRO). Am. Soc. Agric. Eng. 1991, 34, 663–672. [Google Scholar] [CrossRef]
- Silberbush, M.; Lieth, J.H. Nitrate and potassium uptake by greenhouse roses (Rosa hybrida) along successive flower-cut cycles: A model and its calibration. Sci. Hortic. 2004, 101, 127–141. [Google Scholar] [CrossRef]
- Mattson, N.; Lieth, H. ‘Kardinal’ rose exhibits growth plasticity and enhanced nutrient absorption kinetics following nitrate, phosphate, and potassium deprivation. J. Am. Soc. Hort. Sci. 2008, 133, 341–350. [Google Scholar]
- Van Diepen, C.A.; Wolf, J.; van Keulen, H.; Rappoldt, C. WOFOST: A simulation model of crop production. Soil Use Manag. 1989, 5, 16–24. [Google Scholar] [CrossRef]
- Hutson, J.L.; Wagenet, R.J. Leaching Estimation and Chemistry Model: A Process-based Model of Water and Solute Movement, Transformation, Plant Uptake and Chemical Reaction in the Unsaturated Zone; The Department of Soil, Crop and Atmospheric Sciences, Cornell University: New York, NY, USA, 1991; p. 121. [Google Scholar]
- Mary, B.; Guérif, J. Intérêts et limites des modèles de prévision de l'évolution des matières organiques et de l‘azote dans le sol. Cah. Agric. 1994, 3, 247–257. [Google Scholar]
- Webb, T.H.; Lilburne, L.R.; Francis, G.S. Validation of the GLEAMS simulation model for estimating net nitrogen mineralisation and nitrate leaching under cropping in Canterbury, New Zealand. Aust. J. Soil Res. 2001, 39, 1015–1025. [Google Scholar] [CrossRef]
- Hansen, S.; Abrahamsen, P.; Petersen, C.T.; Styczen, M. Daisy: Model use, calibration, and validation. Trans. ASABE 2012, 55, 1315–1333. [Google Scholar] [CrossRef]
- Heinen, M. FUSSIM2: Brief description of the simulation model and application to fertigation scenarios. Agronomie 2001, 21, 285–296. [Google Scholar] [CrossRef]
- Šimůnek, J.; van Genuchten, M.T.; Šejna, M. Modeling Subsurface Water Flow and Solute Transport with HYDRUS and Related Numerical Software Packages; Garcia-Navarro, P., Ed.; Numerical Modelling of Hydrodynamics for Water Resources; Taylor & Francis Group: London, UK, 2007. [Google Scholar]
- Stöckle, C.O.; Donatelli, M.; Nelson, R. CropSyst, a cropping systems simulation model. Eur. J. Agric. 2003, 18, 289–307. [Google Scholar] [CrossRef]
- Martínez-Gaitán, C.; Gallardo, M.; Thompson, R.B.; Stöckle, C.O.; Granados, M.R.; Fernández, M.D.; Giménez, C. Use of CropSyst to simulate growth, ETc and N uptake for the development of irrigation and N fertiliser programs in intensive vegetable crop production. Acta Hortic. 2008, 802, 337–342. [Google Scholar] [CrossRef]
- Giménez, C.; Gallardo, M.; Martínez-Gaitán, C.; Stöckle, C.O.; Thompson, R.B.; Granados, M.R. VegSyst, a simulation model of daily crop growth, nitrogen uptake and evapotranspiration for pepper crops for use in an on-farm decision support system. Irrig. Sci. 2013, 31, 465–477. [Google Scholar] [CrossRef]
- Gallardo, M.; Thompson, R.B.; Giménez, C.; Padilla, F.M.; Stöckle, C.O. Prototype decision support system based on the VegSyst simulation model to calculate crop N and water requirements for tomato under plastic cover. Irrig. Sci. 2014, 32, 237–253. [Google Scholar] [CrossRef]
- Rahn, C.R.; Zhang, K.; Lillywhite, R.; Ramos, C.; Doltra, J.; de Paz, J.M.; Riley, H.; Fink, M.; Nendel, C.; Thorup-Kristensen, K.; et al. Eu-Rotate_N—A decision support system—To predict environmental and economic consequences of the management of nitrogen fertiliser in crop rotations. Eur. J. Hortic. Sci. 2010, 75, 20–32. [Google Scholar]
- Nendel, C.; Venezia, A.; Piro, F.; Ren, T.; Lillywhite, R.D.; Rahn, C.R. The performance of the EU-Rotate-N model in predicting the growth and nitrogen uptake of rotations of field vegetable crops in a Mediterranean environment. J. Agric. Sci. 2013, 151, 538–555. [Google Scholar] [CrossRef]
- Sun, Y.; Hu, K.; Fan, Z.; Wei, Y.; Lin, S.; Wang, J. Simulating the fate of nitrogen and optimizing water and nitrogen management of greenhouse tomato in North China using the EU-Rotate_N model. Agric. Water Manag. 2013, 128, 72–84. [Google Scholar] [CrossRef]
- Suárez-Rey, E.M.; Romero-Gámez, M.; Giménez, C.; Thompson, R.B.; Gallardo, M. Use of EU-Rotate_N and CropSyst models to predict yield, growth and water and N dynamics of fertigated leafy vegetables in a Mediterranean climate and to determine N fertilizer requirements. Agric. Syst. 2016, 149, 150–164. [Google Scholar] [CrossRef]
- Zhang, K.; Greenwood, D.J.; Spracklen, W.P.; Rahn, C.R.; Hammond, J.P.; White, P.J.; Burns, I.G. A universal agro-hydrological model for water and nitrogen cycles in the soil-crop system SMCR_N: Critical update and further validation. Agric. Water Manag. 2010, 97, 1411–1422. [Google Scholar] [CrossRef]
- Zhang, K.; Yang, D.; Greenwood, D.J.; Rahn, C.R.; Thorup-Kristensen, K. Development and critical evaluation of a generic 2-D agro-hydrological model (SMCR_N) for the responses of crop yield and nitrogen composition to nitrogen fertilizer. Agric. Ecosyst. Environ. 2009, 132, 160–172. [Google Scholar] [CrossRef]
- Massa, D.; Incrocci, L.; Pardossi, A.; Paoli, P.D.; Battilani, A. Application of a decision support system for increasing economic and environmental sustainability of processing tomato cultivated in Mediterranean climate. Acta Hortic. 2013, 971, 51–58. [Google Scholar] [CrossRef]
- Elia, A.; Conversa, G. A decision support system (GesCoN) for managing fertigation in open field vegetable crops. Part I-methodological approach and description of the software. Front. Plant Sci. 2015, 6, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Conversa, G.; Bonasia, A.; Di Gioia, F.; Elia, A. A decision support system (GesCoN) for managing fertigation in vegetable crops. Part II-model calibration and validation under different environmental growing conditions on field grown tomato. Front. Plant Sci. 2015, 6, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Cahn, M.; Smith, R.; Hartz, T. Improving Irrigation and Nitrogen Management in California Leafy Greens Production. In Proceedings of the NUTRIHORT: Nutrient Management, Innovative Techniques and Nutrient Legislation in Intensive Horticulture for an Improved Water Quality, Ghent, Belgium, 16–18 September 2013; pp. 65–68. [Google Scholar]
- Stockle, C.O.; Martin, S.A.; Campbell, G.S. CropSyst, a cropping systems simulation model: Water/nitrogen budgets and crop yield. Agric. Syst. 1994, 46, 335–359. [Google Scholar] [CrossRef]
- Soto, F.; Gallardo, M.; Giménez, C.; Peña-Fleitas, T.; Thompson, R.B. Simulation of tomato growth, water and N dynamics using the EU-Rotate_N model in Mediterranean greenhouses with drip irrigation and fertigation. Agric. Water Manag. 2014, 132, 46–59. [Google Scholar] [CrossRef]
- Sun, Y.; Hu, K.; Zhang, K.; Jiang, L.; Xu, Y. Simulation of nitrogen fate for greenhouse cucumber grown under different water and fertilizer management using the EU-Rotate_N model. Agric. Water Manag. 2012, 112, 21–32. [Google Scholar] [CrossRef]
- Gallardo, M.; Fernández, M.D.; Giménez, C.; Padilla, F.M.; Thompson, R.B. Revised VegSyst model to calculate dry matter production, critical N uptake and ETc of several vegetable species grown in Mediterranean greenhouses. Agric. Syst. 2016, 146, 30–43. [Google Scholar] [CrossRef]
- Cahn, M.; Hartz, T.; Smith, R.; Noel, B.; Johnson, L.; Melton, F. CropManage: An online decision support tool for irrigation and nutrient management. In Proceedings of the Western Nutrient Management Conference, Reno, NV, USA, 5–6 March 2015; pp. 9–13. [Google Scholar]
- Lorenz, H.P.; Schlaghecken, J.; Engl, G.; Maync, A.; Ziegler, J.; Kohl, M.; Strohmeyer, K. Ordnungsgemasse Stìckstoff-Versorgung im Freiland-Gemusebau nach dem “Kulturbegleìtenden Nmin-Sollwerte (KNS)-System”; Mìnìsterium fur Landwìrtschaft, Weinbau und Forsten: Rheìnland Pfalz, Maìnz, 1989; p. 85. [Google Scholar]
- Ziegler, J.; Strohmeier, K.; Brand, T. Nitrogen supply of vegetables based on the “KNS-system“. Acta Hortic. 1996, 428, 223–233. [Google Scholar] [CrossRef]
- Fink, M.; Sharpf, H.C. N-Expert—A decision support system for vegetable fertilization in the field. Acta Hortic. 1993, 339, 67–74. [Google Scholar] [CrossRef]
- Chen, Q.; Zhang, H.; Li, X.; Christie, P.; Horlacher, D.; Liebig, H.P. Use of a modified N-expert system for vegetable production in the Beijing region. J. Plant Nutr. 2005, 28, 475–487. [Google Scholar] [CrossRef]
- Goffart, J.P.; Olivier, M.; Frankinet, M. Crop nitrogen status assessment tools in a decision support system for nitrogen fertilization management of potato crops. Horttechnology 2011, 21, 282–286. [Google Scholar]
- Goffart, J.P.; Olivier, M.; Frankinet, M. Potato Crop Nitrogen Status Assessment to Improve N Fertilization Management and Efficiency: Past-Present-Future. Potato Res. 2008, 51, 355–383. [Google Scholar] [CrossRef]
- Nishina, H. Development of speaking plant approach technique for intelligent greenhouse. Agric. Agric. Sci. Proc. 2015, 3, 9–13. [Google Scholar] [CrossRef]
- Thompson, R.B.; Incrocci, L.; Voogt, W.; Pardossi, A.; Magán, J.J. Sustainable irrigation and nitrogen management of fertigated vegetable crops. Acta Hortic. 2017, 1150, 363–378. [Google Scholar] [CrossRef]
- Farneselli, M.; Tei, F.; Simonne, E. Reliability of petiole sap test for n nutritional status assessing in processing tomato. J. Plant Nutr. 2014, 37, 270–278. [Google Scholar] [CrossRef]
- Peña-Fleitas, M.T.; Gallardo, M.; Thompson, R.B.; Farneselli, M.; Padilla, F.M. Assessing crop N status of fertigated vegetable crops using plant and soil monitoring techniques. Ann. Appl. Biol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Hochmuth, G.J. Efficiency ranges for nitrate-nitrogen and potassium for vegetable petiole sap quick tests. HortTechnology 1994, 4, 218–222. [Google Scholar]
- Muñoz-Huerta, R.F.; Guevara-Gonzalez, R.G.; Contreras-Medina, L.M.; Torres-Pacheco, I.; Prado-Olivarez, J.; Ocampo-Velazquez, R.V. A review of methods for sensing the nitrogen status in plants: Advantages, disadvantages and recent advances. Sensors 2013, 13, 10823–10843. [Google Scholar] [CrossRef] [PubMed]
- Loh, F.C.W.; Grabosky, J.C.; Bassuk, N.L. Using the SPAD 502 meter to assess chlorophyll and nitrogen content of benjamin fig and cottonwood leaves. Horttechnology 2002, 12, 682–686. [Google Scholar]
- Swiader, J.M.; Moore, A. Spad-chlorophyll response to nitrogen fertilization and evaluation of nitrogen status in dryland and irrigated pumpkins. J. Plant Nutr. 2002, 25, 1089–1100. [Google Scholar] [CrossRef]
- Padilla, F.M.; Peña-Fleitas, M.T.; Gallardo, M.; Thompson, R.B. Threshold values of canopy reflectance indices and chlorophyll meter readings for optimal nitrogen nutrition of tomato. Ann. Appl. Biol. 2015, 166, 271–285. [Google Scholar] [CrossRef]
- Goulas, Y.; Cerovic, Z.G.; Cartelat, A.; Moya, I. Dualex: A new instrument for field measurements of epidermal ultraviolet absorbance by chlorophyll fluorescence. Appl. Opt. 2004, 43, 4488–4496. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, N.; Bélec, C.; Jenni, S.; Fortier, E.; Mellgren, R. The dualex—A new tool to determine nitrogen sufficiency in broccoli. Acta Hortic. 2009, 824, 121–132. [Google Scholar] [CrossRef]
- Wu, J.D.; Wang, D.; Rosen, C.J.; Bauer, M.E. Comparison of petiole nitrate concentrations, SPAD chlorophyll readings, and QuickBird satellite imagery in detecting nitrogen status of potato canopies. Field Crops Res. 2007, 101, 96–103. [Google Scholar] [CrossRef]
- Gianquinto, G.; Orsini, F.; Fecondini, M.; Mezzetti, M.; Sambo, P.; Bona, S. A methodological approach for defining spectral indices for assessing tomato nitrogen status and yield. Eur. J. Agric. 2011, 35, 135–143. [Google Scholar] [CrossRef]
- Padilla, F.M.; Teresa Peña-Fleitas, M.; Gallardo, M.; Thompson, R.B. Evaluation of optical sensor measurements of canopy reflectance and of leaf flavonols and chlorophyll contents to assess crop nitrogen status of muskmelon. Eur. J. Agric. 2014, 58, 39–52. [Google Scholar] [CrossRef]
- Baret, F.; Houlès, V.; Guérif, M. Quantification of plant stress using remote sensing observations and crop models: The case of nitrogen management. J. Exp. Bot. 2007, 58, 869–880. [Google Scholar] [CrossRef] [PubMed]
- Usha, K.; Singh, B. Potential applications of remote sensing in horticulture—A review. Sci. Hortic. 2013, 153, 71–83. [Google Scholar] [CrossRef]
- Fox, R.H.; Walthall, C.L. Crop monitoring technologies to assess nitrogen status. In Nitrogen in Agricultural Systems; American Society of Agronomy: Madison, WI, USA, 2008; pp. 647–674. [Google Scholar]
- Samborski, S.M.; Tremblay, N.; Fallon, E. Strategies to make use of plant sensor-based diagnostic information for nitrogen recommendations. Agron. J. 2009, 101, 800–816. [Google Scholar] [CrossRef]
- Sonneveld, C.; Voogt, W. Plant Nutrition of Greenhouse Crops; Springer: New York, NY, USA, 2009; p. 350. [Google Scholar]
- Granados, M.R.; Thompson, R.B.; Fernández, M.D.; Martínez-Gaitán, C.; Gallardo, M. Prescriptive-corrective nitrogen and irrigation management of fertigated and drip-irrigated vegetable crops using modeling and monitoring approaches. Agric. Water Manag. 2013, 119, 121–134. [Google Scholar] [CrossRef]
- Van den Bos, A.L.; de Kreij, C.; Voogt, W. Bemestingsadviesbasis Grond; Proefstation voor Bloemisterij en Glasgroente: Naaldwijk, 1999; p. 54. [Google Scholar]
- Hartz, T.K. The assessment of soil and crop nutrient status in the development of efficient fertilizer recommendations. Acta Hortic. 2003, 627, 231–240. [Google Scholar] [CrossRef]
- Thompson, R.B.; Gallardo, M.; Joya, M.; Segovia, C.; Martínez-Gaitán, C.; Granados, M.R. Evaluation of rapid analysis systems for on-farm nitrate analysis in vegetable cropping. Span. J. Agric. Res. 2009, 7, 200–211. [Google Scholar] [CrossRef]
- Bamsey, M.; Graham, T.; Thompson, C.; Berinstain, A.; Scott, A.; Dixon, M. Ion-specific nutrient management in closed systems: The necessity for ion-selective sensors in terrestrial and space-based agriculture and water management systems. Sensors 2012, 12, 13349–13392. [Google Scholar] [CrossRef] [PubMed]
- Parks, S.E.; Irving, D.E.; Milham, P.J. A critical evaluation of on-farm rapid tests for measuring nitrate in leafy vegetables. Sci. Hortic. 2012, 134, 1–6. [Google Scholar] [CrossRef]
- Bogrekci, I.; Lee, W.S. Spectral measurement of common soil phosphates. Trans. ASAE 2005, 48, 2371–2378. [Google Scholar] [CrossRef]
- Yokota, M.; Okada, T.; Yamaguchi, I. An optical sensor for analysis of soil nutrients by using LED light sources. Meas. Sci. Technol. 2007, 18, 2197–2201. [Google Scholar] [CrossRef]
- Bansod, S.J.; Thakare, S.S. Near Infrared spectroscopy based a portable soil nitrogen detector design. Int. J. Comput. Sci. Inf. Technol. 2014, 5, 3953–3956. [Google Scholar]
- Feller, C.; Fink, M. NMIN target values for field vegetables. Acta Hortic. 2002, 571, 195–201. [Google Scholar] [CrossRef]
- De Kreij, C.; Kavvadias, V.; Assimakopoulou, A.; Paraskevopoulos, A. Development of Fertigation for Trickle Irrigated Vegetables under Mediterranean Conditions. Int. J. Veg. Sci. 2007, 13, 81–99. [Google Scholar] [CrossRef]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).