Evaluation of Growth, Yield and Bioactive Compounds of Ethiopian Kale (Brassica carinata A. Braun) Microgreens under Different LED Light Spectra and Substrates
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Materials and Design
2.2. Growing Environment
2.3. Growth Measurements
2.4. Yield and Biomass Analysis
2.5. Phytochemical Analysis
2.5.1. Flavonoids
2.5.2. Carotenoids
2.5.3. Nitrates
2.5.4. Chlorophyll
2.6. Statistical Analysis
3. Results
3.1. Effect of LED Light and Substrate on Height, Leaf Area and Canopy Cover
3.2. Effect of LED Light and Substrate on Yield and Dry Weight
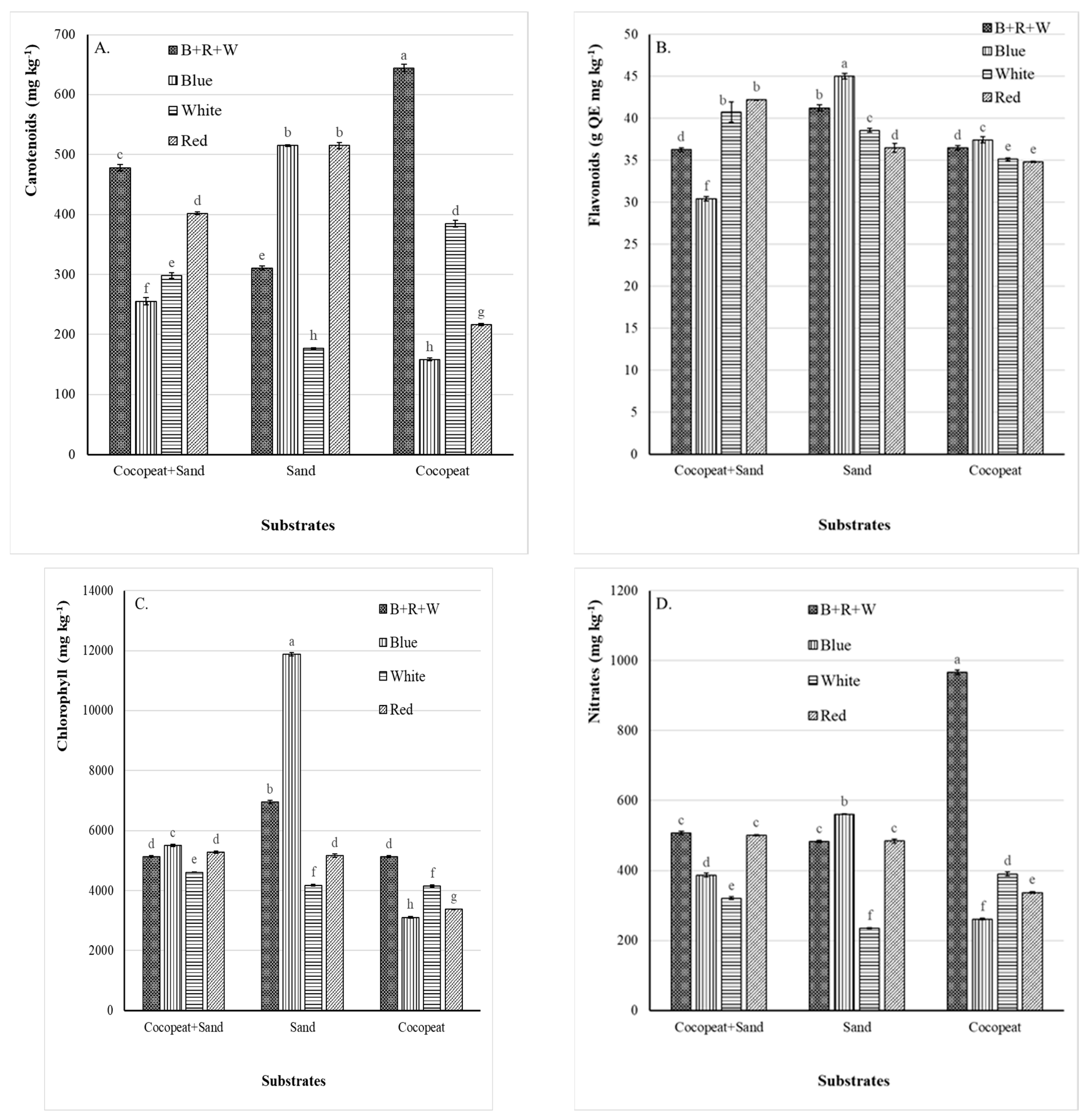
3.3. Effect of LED Light and Substrate on Phytochemical Content
4. Discussion
4.1. Effect of LED Light and Substrate on Height, Leaf Area and Canopy Cover
4.2. Effect of LED Light and Substrate on Yield and Biomass
4.3. Effect of LED Light and Substrate on Phytochemical Content
4.3.1. Carotenoids
4.3.2. Flavonoids
4.3.3. Chlorophyll
4.3.4. Nitrates
4.4. Interactive Effects of Light and Substrate on Phytochemicals
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Appolloni, E.; Pennisi, G.; Zauli, I.; Carotti, L.; Paucek, I.; Quaini, S. Beyond vegetables: Effects of indoor LED light on specialized metabolite biosynthesis in medicinal and aromatic plants, edible flowers, and microgreens. J. Sci. Food Agric. 2022, 102, 472–487. [Google Scholar] [CrossRef]
- Bulgari, R.; Negri, M.; Santoro, P.; Ferrante, A. Quality evaluation of indoor-grown microgreens cultivated on three different substrates. Horticulturae 2021, 7, 96. [Google Scholar] [CrossRef]
- Zhang, Y.; Xiao, Z.; Ager, E.; Kong, L.; Tan, L. Nutritional quality and health benefits of microgreens, a crop of modern agriculture. J. Future Foods 2021, 1, 58–66. [Google Scholar] [CrossRef]
- Verlinden, S. Microgreens: Definitions, product types, and production practices. Hortic. Res. 2020, 85–124. [Google Scholar] [CrossRef]
- Loedolff, B.; Brooks, J.; Stander, M.; Peters, S.; Kossmann, J. High light bio-fortification stimulates de novo synthesis of resveratrol in Diplotaxis tenuifolia (wild rocket) micro-greens. J. Funct. Food Health Dis. 2017, 7, 859–872. [Google Scholar] [CrossRef]
- Neugart, S.; Baldermann, S.; Ngwene, B.; Wesonga, J.; Schreiner, M. Indigenous leafy vegetables of Eastern Africa—A source of extraordinary secondary plant metabolites. Food Res. Int. 2017, 1, 411–422. [Google Scholar] [CrossRef]
- Odongo, G.A.; Schlotz, N.; Herz, C.; Hanschen, F.S.; Baldermann, S.; Neugart, S. The role of plant processing for the cancer preventive potential of Ethiopian kale (Brassica carinata). Food Nutr. Res. 2017, 1, 31–61. [Google Scholar] [CrossRef]
- Nakakaawa, L.; Gbala, I.D.; Cheseto, X.; Bargul, J.L.; Wesonga, J.M. Oral acute, sub-acute toxicity and phytochemical profile of Brassica carinata A. Braun microgreens ethanolic extract in Wistar rats. J. Ethnopharmacol. 2023, 6, 305. [Google Scholar] [CrossRef]
- Chen, H.; Tong, X.; Tan, L.; Kong, L. Consumers’ Acceptability and Perceptions toward the Consumption of Hydroponically and Soil Grown Broccoli Microgreens. J. Agric. Food Res. 2020, 2, 100051. [Google Scholar] [CrossRef]
- Poudel, P.; Duenas, A.E.K.; Di Gioia, F. Organic waste compost and spent mushroom compost as potential growing media components for the sustainable production of microgreens. Front. Plant Sci. 2023, 14, 1229157. [Google Scholar] [CrossRef]
- Di Gioia, F.; De Bellis, P.; Mininni, C.; Santamaria, P.; Serio, F. Physicochemical, agronomical and microbiological evaluation of alternative growing media for the production of rapini (Brassica rapa L.) microgreens. J. Sci. Food Agric. 2017, 97, 1212–1219. [Google Scholar] [CrossRef] [PubMed]
- Thepsilvisut, O.; Sukree, N.; Chutimanukul, P.; Athinuwat, D.; Chuaboon, W.; Poomipan, P. Efficacy of Agricultural and Food Wastes as the Growing Media for Sunflower and Water Spinach Microgreens Production. Horticulturae 2023, 9, 876. [Google Scholar] [CrossRef]
- Kyriacou, M.C.; El-Nakhel, C.; Pannico, A.; Graziani, G.; Soteriou, G.A.; Giordano, M. Phenolic constitution, phytochemical and macronutrient content in three species of microgreens as modulated by natural fiber and synthetic substrates. Antioxidants 2020, 9, 252. [Google Scholar] [CrossRef] [PubMed]
- Ying, Q. Exploration on Using Light-Emitting Diode Spectra to Improve the Quality and Yield of Microgreens in Controlled Environments. Ph.D. Thesis, The University of Guelph, Guelph, ON, Canada, 2020. [Google Scholar]
- Craver, J.K.; Gerovac, J.R.; Lopez, R.G.; Kopsell, D.A. Light intensity and light quality from sole-source light-emitting diodes impact phytochemical concentrations within brassica microgreens. J. Am. Soc. Hortic. 2017, 142, 3–12. [Google Scholar] [CrossRef]
- Brazaityte, A.; Viršile, A.; Jankauskiene, J.; Sakalauskiene, S.; Samuoliene, G.; Sirtautas, R. Effect of supplemental UV-A irradiation in solid-state lighting on the growth and phytochemical content of microgreens. Int. Agrophys. 2015, 29, 13–22. [Google Scholar] [CrossRef]
- Liu, K.; Gao, M.; Jiang, H.; Ou, S.; Li, X.; He, R. Light Intensity and Photoperiod Affect Growth and Nutritional Quality of Brassica Microgreens. Molecules 2022, 27, 883. [Google Scholar] [CrossRef] [PubMed]
- Semananda, N.P.K.; Ward, J.D.; Myers, B.R. A Semi-Systematic Review of Capillary Irrigation: The Benefits, Limitations, and Opportunities. Horticulturae 2018, 4, 23. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 7, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Patrignani, A.; Ochsner, T.E. Canopeo: A powerful new tool for measuring fractional green canopy cover. Agron. J. 2015, 107, 2312–2320. [Google Scholar] [CrossRef]
- Baba, S.A.; Malik, S.A. Determination of total phenolic and flavonoid content, antimicrobial and antioxidant activity of a root extract of Arisaema jacquemontii Blume. J. Taibah Univ. Sci. 2015, 9, 449–454. [Google Scholar] [CrossRef]
- Nyonje, W.A.; Makokha, A.O.; Abukutsa-Onyango, M.O. Anti-Nutrient, Phytochemical and Antiradical Evaluation of 10 Amaranth (Amaranthus spp.) Varieties Before and After Flowering. J. Agric. Sci. 2014, 6, 68. [Google Scholar] [CrossRef]
- Ritchie, R.J. Universal chlorophyll equations for estimating chlorophylls a, b, c, and d and total chlorophylls in natural assemblages of photosynthetic organisms using acetone, methanol, or ethanol solvents. Photosynthetica 2008, 46, 115–126. [Google Scholar] [CrossRef]
- Davis, P.A.; Burns, C. Photobiology in protected horticulture. Food Energy Secur. 2016, 5, 223–238. [Google Scholar] [CrossRef]
- Qian, H.; Liu, T.; Deng, M.; Miao, H.; Cai, C.; Shen, W. Effects of light quality on main health-promoting compounds and antioxidant capacity of Chinese kale sprouts. Food Chem. 2016, 196, 1232–1238. [Google Scholar] [CrossRef] [PubMed]
- Son, K.H.; Oh, M.M. Growth, photosynthetic and antioxidant parameters of two lettuce cultivars as affected by red, green, and blue light-emitting diodes. Hortic. Environ. Biotechnol. 2015, 56, 639–653. [Google Scholar] [CrossRef]
- Naznin, M.T.; Lefsrud, M.; Gravel, V.; Azad, M.O.K. Blue light added with red LEDs enhance growth characteristics, pigments content, and antioxidant capacity in lettuce, Spinach, Kale, Basil, and sweet pepper in a controlled environment. Plants 2019, 8, 93. [Google Scholar] [CrossRef] [PubMed]
- Ying, Q.; Kong, Y.; Zheng, Y. Applying blue light alone, or in combination with far-red light, during nighttime increases elongation without compromising yield and quality of indoor-grown microgreens. HortScience 2020, 55, 876–881. [Google Scholar] [CrossRef]
- Awang, Y.; Shazmi Shaharom, A.; Mohamad, R.B.; Selamat, A. Chemical and Physical Characteristics of Cocopeat-Based Media Mixtures and Their Effects on the Growth and Development of Celosia cristata. Am. J. Agric. Biol. Sci. 2009, 4, 63–71. [Google Scholar] [CrossRef]
- Son, K.H.; Oh, M.M. Leaf Shape, Growth, and Antioxidant Phenolic Compounds of Two Lettuce Cultivars Grown under Various Combinations of Blue and Red Light-emitting Diodes. HortScience 2013, 48, 988–995. [Google Scholar] [CrossRef]
- Gunjal, M.; Singh, J.; Kaur, J.; Kaur, S.; Nanda, V.; Mehta, C.M. Comparative analysis of morphological, nutritional, and bioactive properties of selected microgreens in alternative growing medium. S. Afr. J. Bot. 2024, 165, 188–201. [Google Scholar] [CrossRef]
- Lanoue, J.; St. Louis, S.; Little, C.; Hao, X. Continuous lighting can improve yield and reduce energy costs while increasing or maintaining nutritional contents of microgreens. Front. Plant Sci. 2022, 13, 983222. [Google Scholar] [CrossRef] [PubMed]
- Bulgari, R.; Baldi, A.; Ferrante, A.; Lenzi, A. Yield and quality of basil, Swiss chard, and rocket microgreens grown in a hydroponic system. N. Z. J. Crop Hortic. Sci. 2017, 45, 119–129. [Google Scholar] [CrossRef]
- Nolan, D.A. Effects of Seed Density and Other Factors on the Yield of Microgreens Grown Hydroponically on Burlap; Virginia Tech: Blacksburg, VA, USA, 2018; pp. 1–44. [Google Scholar]
- Thuong, V.T.; Minh, H.G. Effects of growing substrates and seed density on yield and quality of radish (Raphanus sativus) microgreens. Res. Crops 2020, 21, 579–586. [Google Scholar]
- Jones-Baumgardt, C.S. The Use of Light-Emitting Diodes for Microgreen Production in Controlled Environments; The University of Guelph: Guelph, ON, USA, 2019; pp. 1–111. [Google Scholar]
- Liu, J.; Pattey, E.; Miller, J.R.; McNairn, H.; Smith, A.; Hu, B. Estimating crop stresses, aboveground dry biomass and yield of corn using multi-temporal optical data combined with a radiation use efficiency model. Remote Sens. Environ. 2010, 114, 1167–1177. [Google Scholar] [CrossRef]
- Demir, K.; Sarıkamış, G.; Çakırer Seyrek, G. Effect of LED lights on the growth, nutritional quality and glucosinolate content of broccoli, cabbage and radish microgreens. Food Chem. 2023, 401, 134088. [Google Scholar] [CrossRef]
- Frede, K.; Winkelmann, S.; Busse, L.; Baldermann, S. The effect of LED light quality on the carotenoid metabolism and related gene expression in the genus Brassica. BMC Plant Biol. 2023, 23, 328. [Google Scholar] [CrossRef]
- Alrifai, O.; Hao, X.; Liu, R.; Lu, Z.; Marcone, M.F.; Tsao, R. LED-Induced Carotenoid Synthesis and Related Gene Expression in Brassica Microgreens. J. Agric. Food Chem. 2021, 69, 4674–4685. [Google Scholar] [CrossRef]
- Samuoliene, G.; Sirtautas, R.; Brazaityte, A.; Duchovskis, P. LED lighting and seasonality effects antioxidant properties of baby leaf lettuce. Food Chem. 2012, 134, 1494–1499. [Google Scholar] [CrossRef]
- Harbart, V.; Frede, K.; Fitzner, M.; Baldermann, S. Regulation of carotenoid and flavonoid biosynthetic pathways in Lactuca sativa var capitate L. in protected cultivation. Front. Plant Sci. 2023, 14, 1124750. [Google Scholar] [CrossRef]
- Landi, M.; Zivcak, M.; Sytar, O.; Brestic, M.; Allakhverdiev, S.I. Plasticity of photosynthetic processes and the accumulation of secondary metabolites in plants in response to monochromatic light environments. Biochim. Biophys. Acta Bioenerg. 2020, 1861, 148131. [Google Scholar] [CrossRef]
- Jiang, H.; Li, Y.; He, R.; Tan, J.; Liu, K.; Chen, Y. Effect of Supplemental UV-A Intensity on Growth and Quality of Kale under Red and Blue Light. Int. J. Mol. Sci. 2022, 23, 6819. [Google Scholar] [CrossRef]
- Wu, X.; Zhang, S.; Liu, X.; Shang, J.; Zhang, A.; Zhu, Z. Chalcone synthase (CHS) family members analysis from eggplant (Solanum melongena L.) in the flavonoid biosynthetic pathway and expression patterns in response to heat stress. PLoS ONE 2020, 15, e0226537. [Google Scholar] [CrossRef]
- Manivannan, A.; Soundararajan, P.; Park, Y.G.; Jeong, B.R. Physiological and Proteomic Insights Into Red and Blue Light-Mediated Enhancement of in vitro Growth in Scrophularia kakudensis—A Potential Medicinal Plant. Front. Plant Sci. 2021, 11, 607007. [Google Scholar] [CrossRef]
- Saleh, R. Growing Media Amendments and LED Light Interaction Effect on Microgreens Plant Growth and Biochemical Composition. Dalhouse University: Halifax, NS, USA, 2023. [Google Scholar]
- Barrett, D.M.; Beaulieu, J.C.; Shewfelt, R. Color, flavor, texture, and nutritional quality of fresh-cut fruits and vegetables: Desirable levels, instrumental and sensory measurement, and the effects of processing. Crit. Rev. Food Sci. Nutr. 2010, 50, 369–389. [Google Scholar] [CrossRef]
- Lobiuc, A.; Vasilache, V.; Pintilie, O.; Stoleru, T.; Burducea, M.; Oroian, M. Blue and red LED illumination improves growth and bioactive compounds contents in acyanic and cyanic Ocimum basilicum L. Microgreens. Molecules 2017, 22, 2111. [Google Scholar] [CrossRef] [PubMed]
- Pennisi, G.; Blasioli, S.; Cellini, A.; Maia, L.; Crepaldi, A.; Braschi, I. Unraveling the role of Red: Blue LED lights on resource use efficiency and nutritional properties of indoor grown sweet basil. Front. Plant Sci. 2019, 10, 00305. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.X.; Zang, J.; Xu, Z.G.; Guo, S.R.; Jiao, X.L.; Liu, X.Y. Effects of different light quality on growth, chlorophyll concentration and chlorophyll biosynthesis precursors of non-heading Chinese cabbage (Brassica campestris L.). Acta Physiol. Plant. 2013, 35, 2721–2726. [Google Scholar] [CrossRef]
- Ferrón-Carrillo, F.; Luis Guil-Guerrero, J.; María González-Fernández, J.; Lyashenko, S.; Battafarano, F. LED Enhances Plant Performance and Both Carotenoids and Nitrates Profiles in Lettuce. Plant Foods Hum. Nutr. 2021, 76, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Pinto, E.; Almeida, A.A.; Aguiar, A.A.; Ferreira, I.M.P.L.V.O. Comparison between the mineral profile and nitrate content of microgreens and mature lettuces. J. Food Compos. Anal. 2015, 37, 38–43. [Google Scholar] [CrossRef]
- Hanrahan, P.; Krueger, W. Reflection from Layered Surfaces due to Subsurface Scattering. Semin. Graph. Pap. 2023, 2, 279–288. [Google Scholar]
| Treatment | Height (cm) | Leaf Area (cm2) | Canopy Cover (%) |
|---|---|---|---|
| LED Lights | |||
| B | 9.9 (0.16) a | 57.62 (1.40) c | 50.68 (4.51) a |
| R | 9.2 (0.16) b | 57.36 (1.46) c | 44.45 (2.66) b |
| W | 9.7 (0.18) a | 63.43 (1.56) b | 56.39 (2.85) a |
| B + R + W | 9.8 (0.11) a | 68.11 (1.96) a | 55.15 (2.76) a |
| P | 0.011 | <0.001 | <0.001 |
| LSD0.05 | 0.39 | 4.32 | 5.87 |
| F Value | F (3,108) = 3.92 | F (3,108) = 11.18 | F (3,33) = 13.12 |
| Substrates | |||
| Sand | 9.8 (0.13) a | 60.0 (1.36) b | 56.0 (3.26) a |
| Cocopeat | 9.2 (0.12) b | 59.1 (1.44) c | 47.1 (2.07) b |
| Sand + Cocopeat | 9.9 (0.14) a | 65.6 (1.66) a | 51.9 (3.394) ab |
| P | <0.001 | 0.001 | 0.005 |
| LSD0.05 | 0.34 | 3.74 | 5.08 |
| F Value | F (3,108) = 11.86 | F (3,108) = 7.28 | F (3,33) = 12.02 |
| Treatment | Yield (g) | Dry Weight (g) |
|---|---|---|
| LED Lights | ||
| B | 17.9 (1.94) a | 1.2 (0.11) ab |
| R | 16.0 (0.86) a | 1.0 (0.06) c |
| W | 18.8 (2.36) a | 1.3 (0.17) a |
| B + R + W | 19.5 (2.22) a | 1.2 (0.10) ab |
| P | 0.339 | 0.053 |
| LSD0.05 | 3.73 | 0.23 |
| F (3,33) | 1.28 | 5.38 |
| Substrates | ||
| Cocopeat | 15.2 (1.75) b | 1.0 (0.08) a |
| Sand | 19.2 (1.54) a | 1.3 (0.10) b |
| Cocopeat + sand | 19.8 (1.76) a | 1.2 (0.13) ab |
| P | 0.013 | 0.016 |
| LSD0.05 | 3.23 | 0.20 |
| F (2,33) | 11.29 | 13.14 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maru, R.N.; Wesonga, J.; Okazawa, H.; Kavoo, A.; Neondo, J.O.; Mazibuko, D.M.; Maskey, S.; Orsini, F. Evaluation of Growth, Yield and Bioactive Compounds of Ethiopian Kale (Brassica carinata A. Braun) Microgreens under Different LED Light Spectra and Substrates. Horticulturae 2024, 10, 436. https://doi.org/10.3390/horticulturae10050436
Maru RN, Wesonga J, Okazawa H, Kavoo A, Neondo JO, Mazibuko DM, Maskey S, Orsini F. Evaluation of Growth, Yield and Bioactive Compounds of Ethiopian Kale (Brassica carinata A. Braun) Microgreens under Different LED Light Spectra and Substrates. Horticulturae. 2024; 10(5):436. https://doi.org/10.3390/horticulturae10050436
Chicago/Turabian StyleMaru, Ruth Nyambura, John Wesonga, Hiromu Okazawa, Agnes Kavoo, Johnstone O. Neondo, Dickson Mgangathweni Mazibuko, Sarvesh Maskey, and Francesco Orsini. 2024. "Evaluation of Growth, Yield and Bioactive Compounds of Ethiopian Kale (Brassica carinata A. Braun) Microgreens under Different LED Light Spectra and Substrates" Horticulturae 10, no. 5: 436. https://doi.org/10.3390/horticulturae10050436
APA StyleMaru, R. N., Wesonga, J., Okazawa, H., Kavoo, A., Neondo, J. O., Mazibuko, D. M., Maskey, S., & Orsini, F. (2024). Evaluation of Growth, Yield and Bioactive Compounds of Ethiopian Kale (Brassica carinata A. Braun) Microgreens under Different LED Light Spectra and Substrates. Horticulturae, 10(5), 436. https://doi.org/10.3390/horticulturae10050436







