Use of Chicken Feather Peptone and Sugar Beet Molasses as Low Cost Substrates for Xanthan Production by Xanthomonas campestris MO-03
Abstract
:1. Introduction
2. Results and Discussion
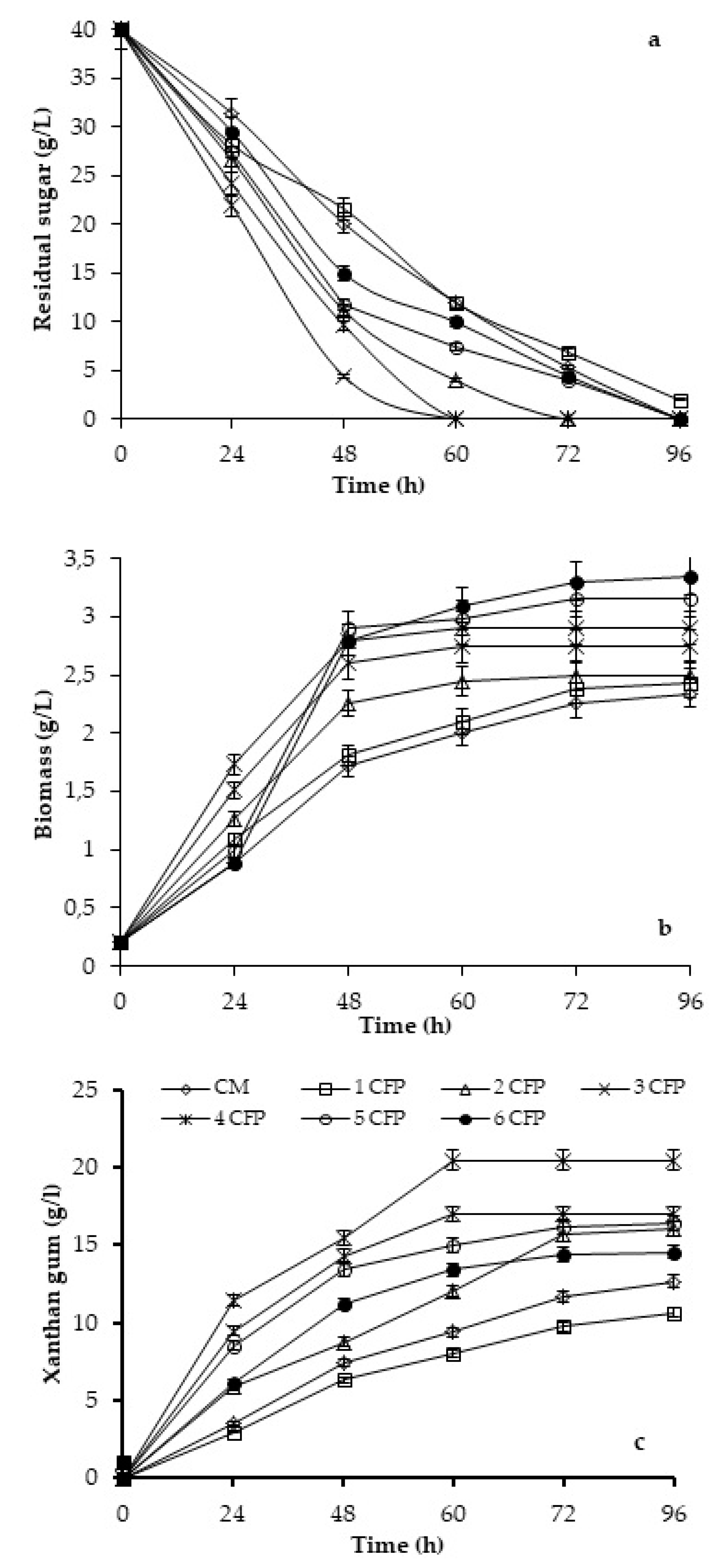
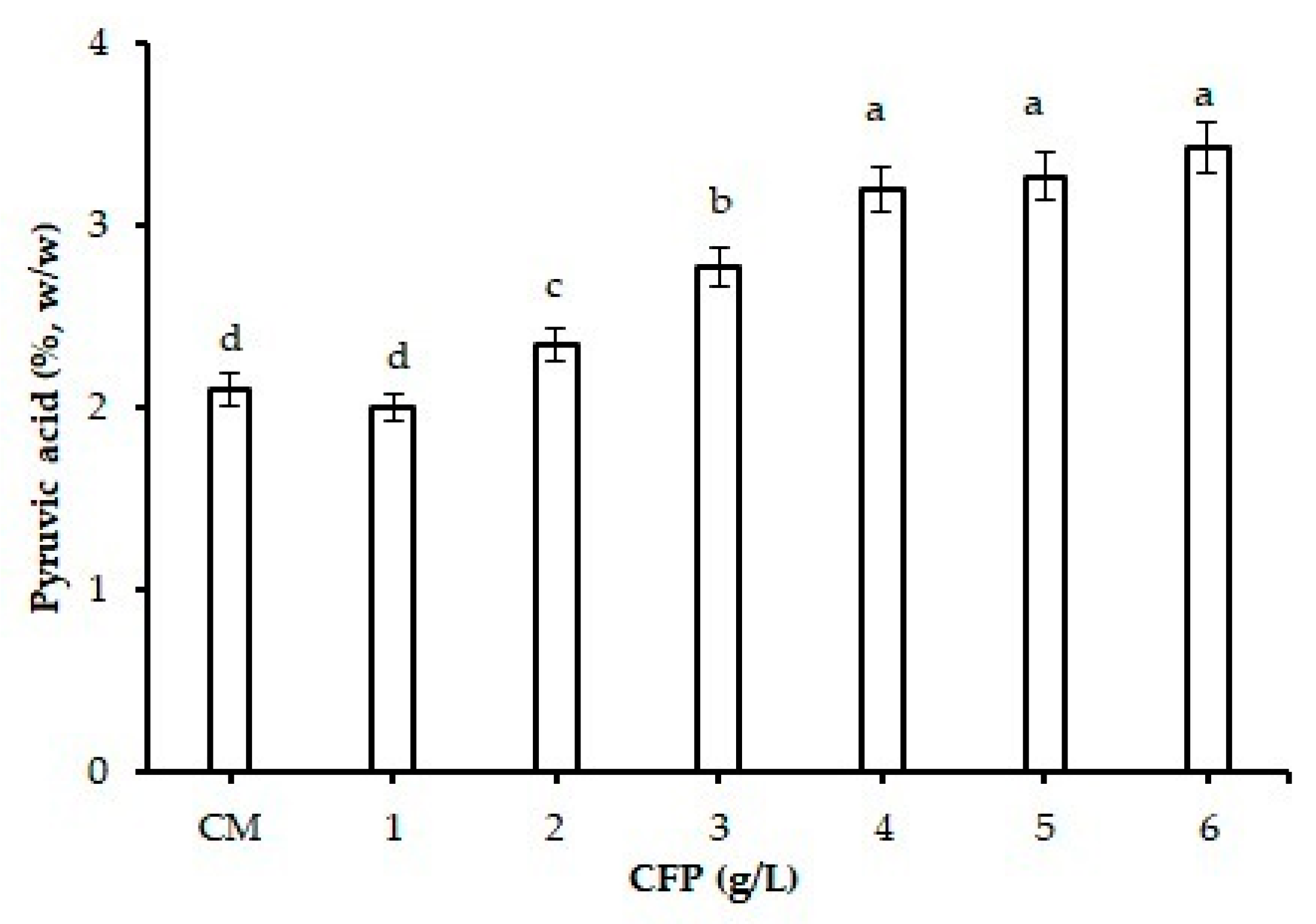
2.1. Effect of CFP on Xanthan Gum Production
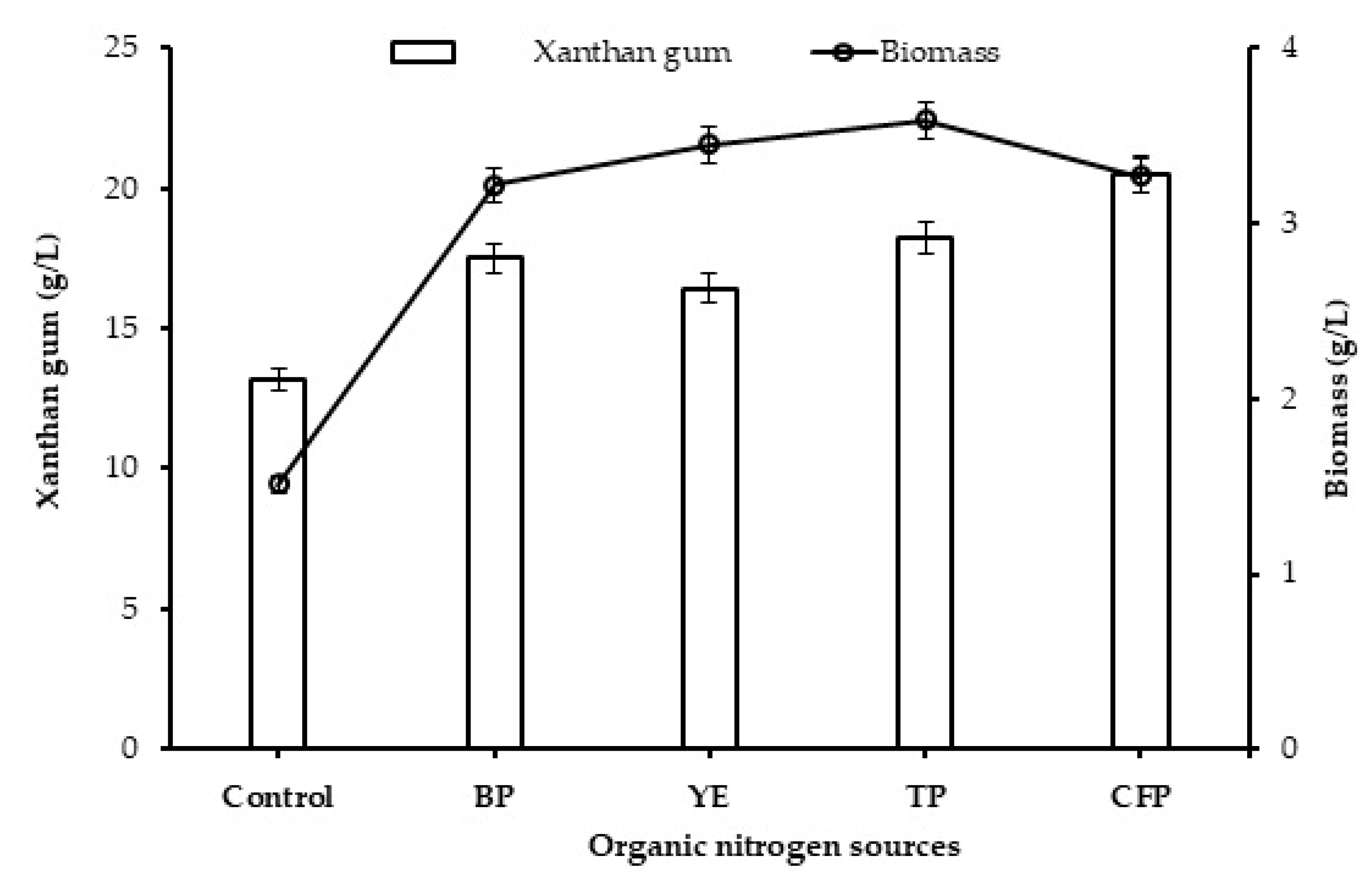
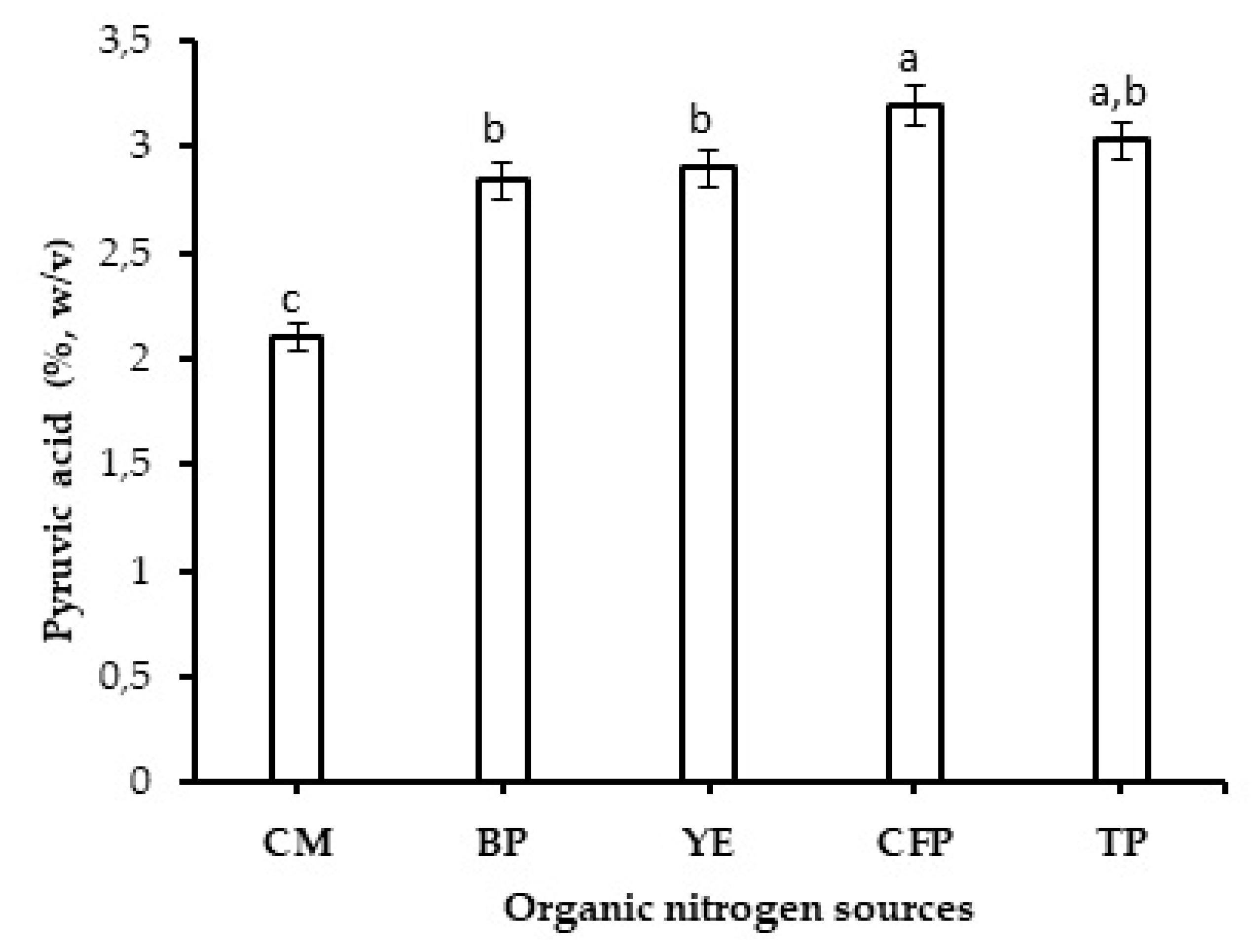
2.2. Effect of Organic Nitrogen Sources on Production of Xanthan
3. Materials and Methods
3.1. Microorganism
3.2. Hydrolysis of Chicken Feathers
3.3. Pretreatment of Molasses
3.4. Media and Culture Conditions
3.5. Analytical Methods
3.6. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ozdal, M. Determination of carbon and nitrogen sources for the production of mycelial biomass and exopolysaccharide by Lepista nuda in liquid culture. Turkish J. Agric. Food Sci. Technol. 2018, 6, 581–585. [Google Scholar]
- Garcia-Ochoa, F.; Santos, V.E.; Casas, J.A.; Gómez, E. Xanthan gum: Production, recovery, and properties. Biotechnol. Adv. 2000, 18, 549–579. [Google Scholar] [CrossRef]
- Faria, S.; Vieira, P.A.; Resende, M.M.; França, F.P.; Cardoso, V.L. A comparison between shaker and bioreactor performance based on the kinetic parameters of xanthan gum production. Appl. Biochem. Biotechnol. 2009, 156, 45–58. [Google Scholar] [CrossRef] [PubMed]
- Petri, D.F. Xanthan gum: A versatile biopolymer for biomedical and technological applications. J. Appl. Polym. Sci. 2015, 132, 42035. [Google Scholar] [CrossRef]
- Wang, Z.; Wu, J.; Gao, M.J.; Zhu, L.; Zhan, X.B. High production of xanthan gum by a glycerol-tolerant strain Xanthomonas campestris WXLB-006. Prep. Biochem. Biotechnol. 2017, 47, 468–472. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Rao, K.M.; Han, S.S. Application of xanthan gum as polysaccharide in tissue engineering: A review. Carbohydr. Polym. 2017, 180, 128–144. [Google Scholar] [CrossRef] [PubMed]
- Kurt, A.; Toker, O.S.; Tornuk, F. Effect of xanthan and locust bean gum synergistic interaction on characteristics of biodegradable edible film. Int. J. Biol. Macromol. 2017, 102, 1035–1044. [Google Scholar] [CrossRef]
- Kalogiannis, S.; Iakovidou, G.; Liakopoulou-Kyriakides, M.; Kyriakidis, D.A.; Skaracis, G.N. Optimization of xanthan gum production by Xanthomonas campestris grown in molasses. Process Biochem. 2003, 39, 249–256. [Google Scholar] [CrossRef]
- Cheng, R.; Lin, L.; Zhang, Y. Hydrogen peroxide (H2O2) supply significantly improves xanthan gum production mediated by Xanthomonas campestris in vitro. J. Ind. Microbiol. Biotechnol. 2009, 39, 799–803. [Google Scholar] [CrossRef]
- Letisse, F.; Chevallereau, P.; Simon, J.L.; Lindley, N.D. Kinetic analysis of growth and xanthan gum production with Xanthomonas campestris on sucrose, using sequentially consumed nitrogen sources. Appl. Microbiol. Biotechnol. 2001, 55, 417–422. [Google Scholar] [CrossRef]
- Li, P.; Li, T.; Zeng, Y.; Li, X.; Jiang, X.; Wang, Y.; Xie, T.; Zhang, Y. Biosynthesis of xanthan gum by Xanthomonas campestris LRELP-1 using kitchen waste as the sole substrate. Carbohydr. Polym. 2016, 151, 684–691. [Google Scholar] [CrossRef] [PubMed]
- Niknezhad, S.V.; Asadollahi, M.A.; Zamani, A.; Biria, D.; Doostmohammadi, M. Optimization of xanthan gum production using cheese whey and response surface methodology. Food Sci. Biotechnol. 2015, 24, 453–460. [Google Scholar] [CrossRef]
- Bhatia, S.K.; Kumar, N.; Bhatia, R.K. Stepwise bioprocess for exopolysaccharide production using potato starch as carbon source. 3 Biotech 2015, 5, 735–739. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, J.A.; Cardoso, L.G.; de Jesus Assis, D.; Gomes, G.V.P.; Oliveira, M.B.P.P.; de Souza, C.O.; Druzian, J.I. Xanthan Gum Production by Xanthomonas campestris pv. campestris IBSBF 1866 and 1867 from Lignocellulosic Agroindustrial Wastes. Appl. Biochem. Biotechnol. 2018, 3, 750–763. [Google Scholar] [CrossRef]
- Gilani, S.L.; Najafpour, G.D.; Heydarzadeh, H.D.; Zare, H. Kinetic models for xanthan gum production using Xanthomonas campestris from molasses. Chem. Ind. Chem. Eng. Q. 2011, 17, 179–187. [Google Scholar] [CrossRef]
- Nakata, H.; Tamura, M.; Shintani, T.; Gomi, K. Evaluation of baker’s yeast strains exhibiting significant growth on Japanese beet molasses and compound analysis of the molasses types. J. Biosci. Bioeng. 2014, 117, 715–719. [Google Scholar] [CrossRef]
- USDA. Turkey Sugar Annual Report, Gain Report Number: TR8013. Available online: https://gain.fas.usda.gov/Recent%20GAIN%20Publications/Sugar%20Annual_Ankara_Turkey_4-13-2018.pdfm (accessed on 12 October 2018).
- Patinvoh, R.J.; Feuk-Lagerstedt, E.; Lundin, M.; Horváth, I.S.; Taherzadeh, M.J. Biological Pretreatment of Chicken Feather and Biogas Production from Total Broth. Appl. Biochem. Biotechnol. 2016, 180, 1401–1415. [Google Scholar] [CrossRef]
- Ozdal, M.; Gurkok, S.; Ozdal, O.G. Optimization of rhamnolipid production by Pseudomonas aeruginosa OG1 using waste frying oil and chicken feather peptone. 3 Biotech 2017, 7, 117. [Google Scholar] [CrossRef]
- Paul, T.; Das, A.; Mandal, A.; Halder, S.K.; DasMohapatra, P.K.; Pati, B.R.; Mondal, K.C. Production and purification of keratinase using chicken feather bioconversion by a newly isolated Aspergillus fumigatus TKF1: Detection of valuable metabolites. Biomass Conv. Biorefin. 2014, 4, 137–148. [Google Scholar] [CrossRef]
- Ozdal, M.; Kurbanoglu, E.B. Citric acid production by Aspergillus niger from agro-industrial by-products: Molasses and chicken feather peptone. Waste Biomass Valorization 2018. [Google Scholar] [CrossRef]
- Benesova, P.; Kucera, D.; Marova, I.; Obruca, S. Chicken feather hydrolysate as inexpensive complex nitrogen source for PHA production by Cupriavidus necator on waste frying oils. Lett. Appl. Microbiol. 2017, 65, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Ozdal, M.; Kurbanoglu, E.B. Valorisation of chicken feathers for xanthan gum production using Xanthomonas campestris MO-03. J. Genet. Eng. Biotechnol. 2018, 16, 259–263. [Google Scholar] [CrossRef]
- Mesomo, M.; Silva, M.F.; Boni, G.; Padilha, F.F.; Mazutti, M.A.; Mossi, A.; Oliveira, D.; Cansian, R.L.; Di Luccio, M.; Treichel, H. Xanthan gum produced by Xanthomonas campestris from cheese whey: Production optimization and rheological characterization. J. Sci. Food Agric. 2009, 89, 2440–2445. [Google Scholar] [CrossRef]
- Carignatto, C.R.R.; Oliveira, K.S.M.; de Lima, V.M.G.; de Oliva Neto, P. New culture medium to xanthan production by Xanthomonas campestris pv. campestris. Indian J. Microbiol. 2011, 51, 283–288. [Google Scholar] [CrossRef] [PubMed]
- Kurbanoglu, E.B.; Ozdal, M.; Ozdal, O.G.; Algur, O.F. Enhanced production of prodigiosin by Serratia marcescens MO-1 using ram horn peptone. Braz. J. Microbiol. 2015, 46, 631–637. [Google Scholar] [CrossRef] [PubMed]
- Souw, P.; Demain, A.L. Nutritional studies on xanthan production by Xanthomonas campestris NRRL B1459. Appl. Environ. Microbiol. 1979, 37, 1186–1192. [Google Scholar] [PubMed]
- Murad, H.A.; Mohamed, S.H.; Abu-El-Khair, A.G. Impact of amino acids, nitrogen source and buffering system on xanthan yield produced on hydrolyzed whey lactose. Biotechnology 2017, 16, 69–76. [Google Scholar] [CrossRef]
- Lo, Y.M.; Yang, S.T.; Min, D.B. Effects of yeast extract and glucose on xanthan production and cell growth in batch culture of Xanthomonas campestris. Appl. Microbiol. Biotechnol. 1997, 47, 689–694. [Google Scholar] [CrossRef]
- Kurbanoglu, E.B.; Kurbanoglu, N.I. Ram horn hydrolysate as enhancer of xanthan production in batch culture of Xanthomonas campestris EBK-4 isolate. Process Biochem. 2007, 42, 1146–1149. [Google Scholar] [CrossRef]
- Rosalam, S.; England, R. Review of xanthan gum production from unmodified starches by Xanthomonas comprestris sp. Enzyme Microb. Technol. 2006, 39, 197–207. [Google Scholar] [CrossRef]
- Lopes, B.D.; Lessa, V.L.; Silva, B.M.; Carvalho, M.A.D.; Schnitzler, E.; Lacerda, L.G. Xanthan gum: Properties, production conditions, quality and economic perspective. J. Food Nutr. Res. 2015, 54, 185–194. [Google Scholar]
- De Vuyst, L.; Vermeire, A. Use of industrial medium components for xanthan production by Xanthomonas campestris NRRL-B-1459. Appl. Microbiol. Biotechnol. 1994, 42, 187–191. [Google Scholar] [CrossRef]
- Cadmus, M.C.; Knutson, C.A., Jr. Production of high-pyruvate xanthan gum on synthetic medium. Patent No. 4,394,447, 1983. [Google Scholar]
- Candia, J.L.F.; Deckwer, W.D. Effect of the nitrogen source on pyruvate content and rheological properties of xanthan. Biotechnol. Prog. 1999, 15, 446–452. [Google Scholar] [CrossRef] [PubMed]
- Sloneker, J.H.; Orentas, D.G. Pyruvic acid, a unique component of an exocellular bacterial polysaccharide. Nature 1962, 104, 478–479. [Google Scholar] [CrossRef]
- Bawa, R.; Hall, R.E.; Kabra, B.P.; Teague, J.E.; Cagle, G.D.; Markwardt, K.L.; Shah, M.V. Gelling Ophthalmic Compositions Containing Xanthan Gum. U.S. Patent No 6,261,547, 2001. [Google Scholar]
- Kelco Biospecialties Ltd. Production of Xanthan Having a Low Pyruvate Content. Patent No. EP0066961A1, 1982. [Google Scholar]
- Mabrouk, M.E.; ElAhwany, A.M.; Beliah, M.M.; Sabry, S.A. Xanthan production by a novel mutant strain of Xanthomonas campestris: Application of statistical design for optimization of process parameters. Life Sci. J. 2013, 10, 1660–1667. [Google Scholar]
- Moosavi, A.; Karbassi, A. Bioconversıon of sugar-beet molasses into xanthan gum. J. Food Process Preserv. 2010, 34, 316–322. [Google Scholar] [CrossRef]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.T.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
| Media | Xanthan Gum (g/L) | Pyruvic Acid Content (%) | References |
|---|---|---|---|
| Molasses (17.5%), 4 g/L K2HPO4, 10 g/L yeast extract | 53 | 1.2–2.3% | [8] |
| Molasses (6%), 10 g/L yeast extract, 3 mL mineral solution | 17.1 | ND | [15] |
| Molasses (10%), corn steep liquor (2%), phosphate (0.1%), citrate (0.1%) | 22.8 | ~2 | [33] |
| Molasses (10%), 10 g/L KH2PO4, 0.3 g/L MgCl2, 0.5 g/L citric acid | 27.9 | ND | [39] |
| Molasses (10%), 3.0 g/L K2HPO4, 0.25 g/L MgSO4, 1.4 g/L (NH4)2SO4, 1.0 g/L CaCO3 | 19.8 | ND | [40] |
| Molasses (8%), 3.0 g/L KH2PO4, 0.25 g/L MgSO4, 0.25 g/L CaCO3, 4 g/L CFP | 20.5 | 3.2 | This study |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ozdal, M.; Başaran Kurbanoglu, E. Use of Chicken Feather Peptone and Sugar Beet Molasses as Low Cost Substrates for Xanthan Production by Xanthomonas campestris MO-03. Fermentation 2019, 5, 9. https://doi.org/10.3390/fermentation5010009
Ozdal M, Başaran Kurbanoglu E. Use of Chicken Feather Peptone and Sugar Beet Molasses as Low Cost Substrates for Xanthan Production by Xanthomonas campestris MO-03. Fermentation. 2019; 5(1):9. https://doi.org/10.3390/fermentation5010009
Chicago/Turabian StyleOzdal, Murat, and Esabi Başaran Kurbanoglu. 2019. "Use of Chicken Feather Peptone and Sugar Beet Molasses as Low Cost Substrates for Xanthan Production by Xanthomonas campestris MO-03" Fermentation 5, no. 1: 9. https://doi.org/10.3390/fermentation5010009
APA StyleOzdal, M., & Başaran Kurbanoglu, E. (2019). Use of Chicken Feather Peptone and Sugar Beet Molasses as Low Cost Substrates for Xanthan Production by Xanthomonas campestris MO-03. Fermentation, 5(1), 9. https://doi.org/10.3390/fermentation5010009





