Ethanol Production from a Mixture of Waste Tissue Paper and Food Waste through Saccharification and Mixed-Culture Fermentation
Abstract
1. Introduction
2. Materials and Methods
2.1. Material
2.1.1. Tissue Paper
2.1.2. Food Waste
2.1.3. Yeast and Medium
2.2. Separate Hydrolysis Fermentation with Mixed Yeast Cultures
2.2.1. Fermentation Performance of Mixed Yeast Cultures
2.2.2. The Impact of Mixed Substrates on Fermentation Performance
2.2.3. Impact of pH Regulation on Fermentation Performance
2.2.4. Impact of Mixed Culture Ratios on Fermentation Performance
2.2.5. Impact of Dynamic Temperature Control on Fermentation Performance
2.3. Analytical Methods
2.3.1. Determination of Yeast Concentration and Sugar
2.3.2. Determination of Alcohols and Acids
3. Results
3.1. Characteristics of Food Waste and Waste Tissue Paper
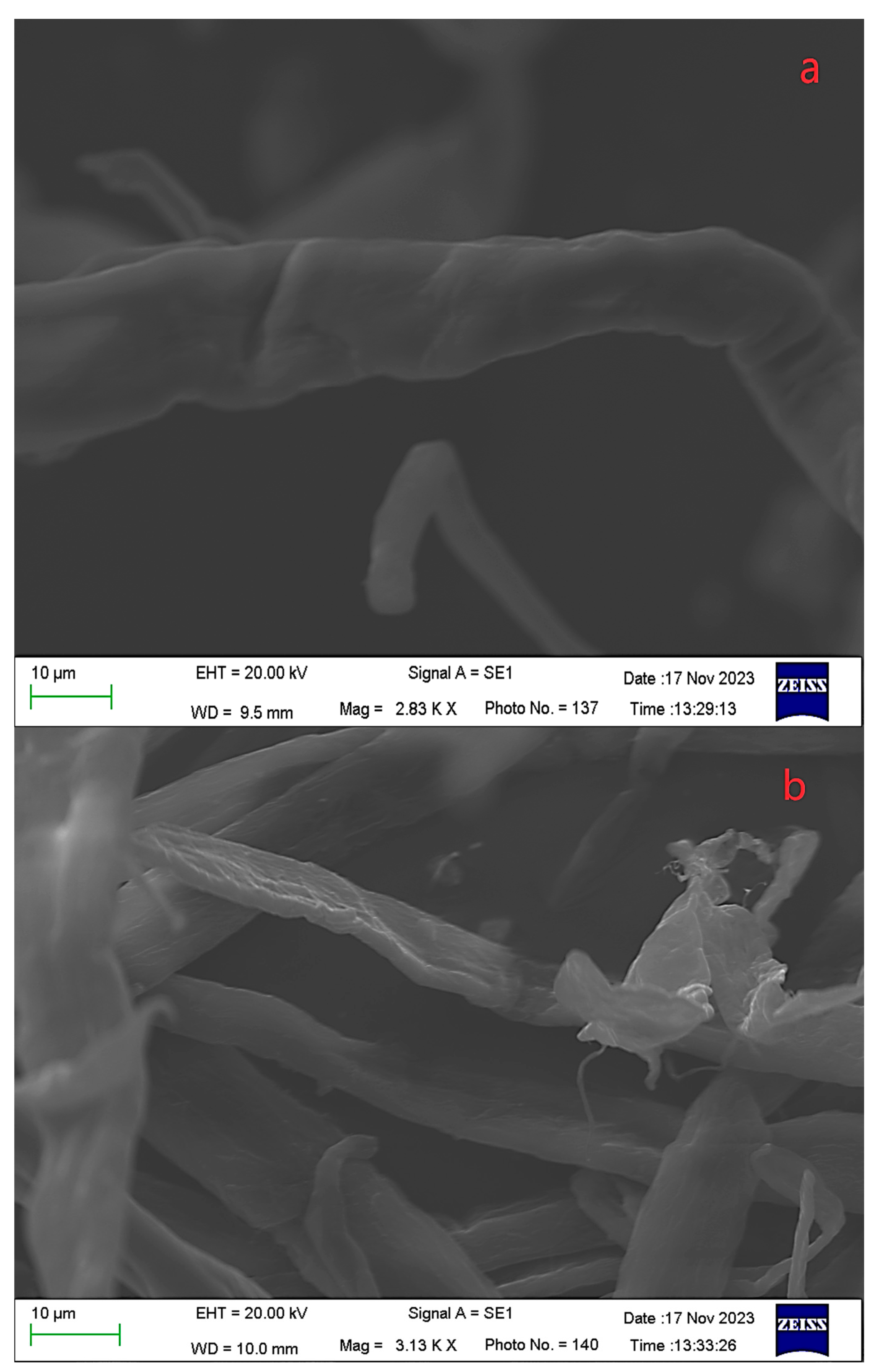
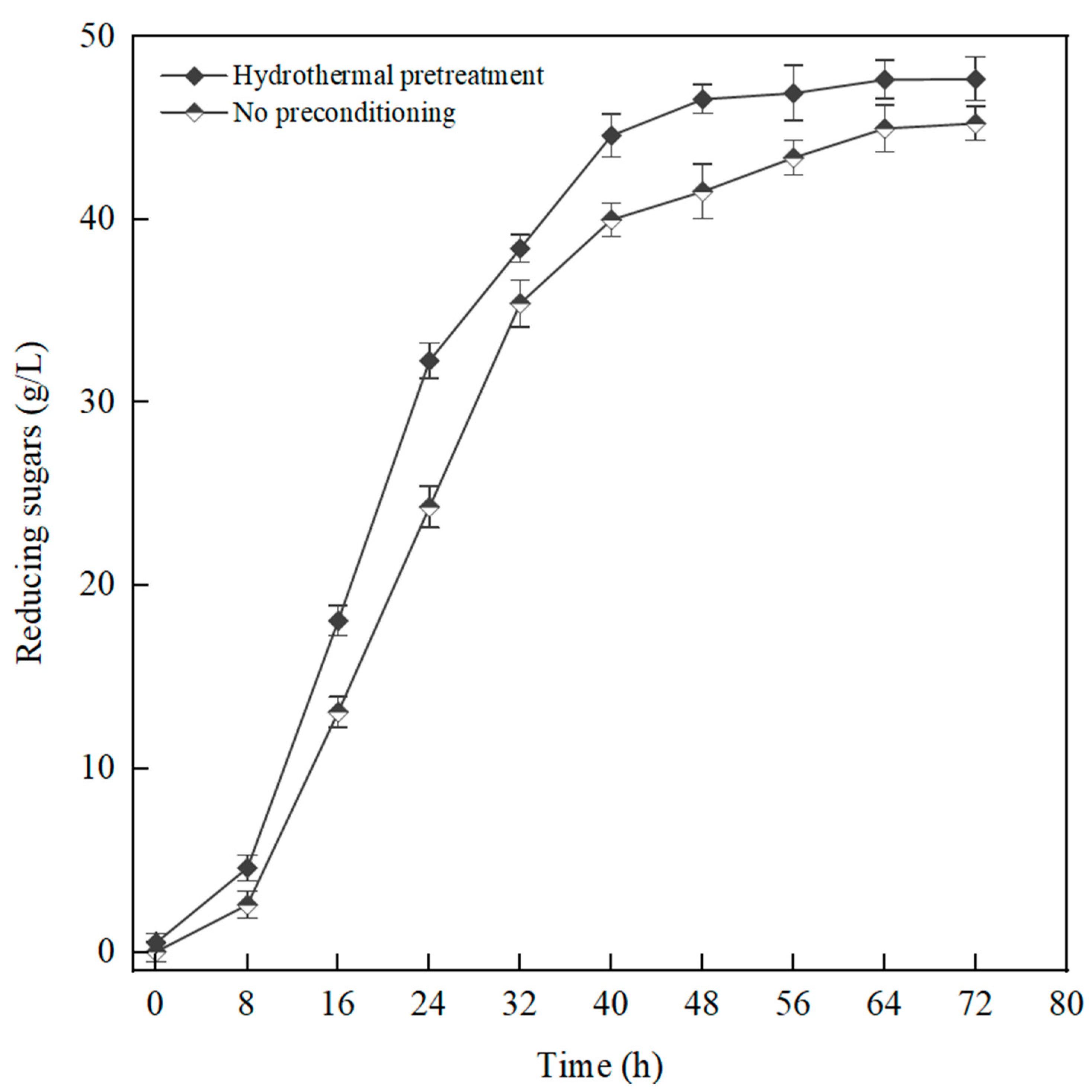
3.2. Impact of Hydrothermal Pretreatment on the Saccharification of Waste Tissue Paper
3.3. Separate Hydrolysis Fermentation with Mixed Yeast Cultures
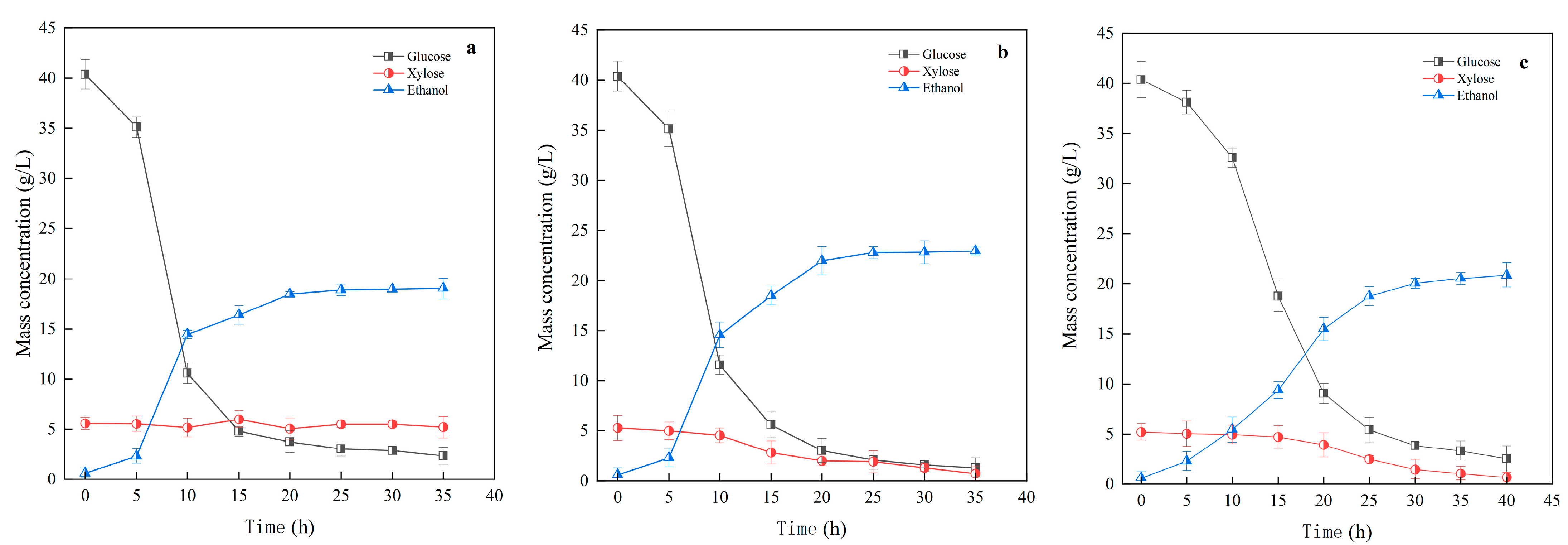
3.3.1. Impact of Mixed and Single Yeast Cultures on Ethanol Fermentation
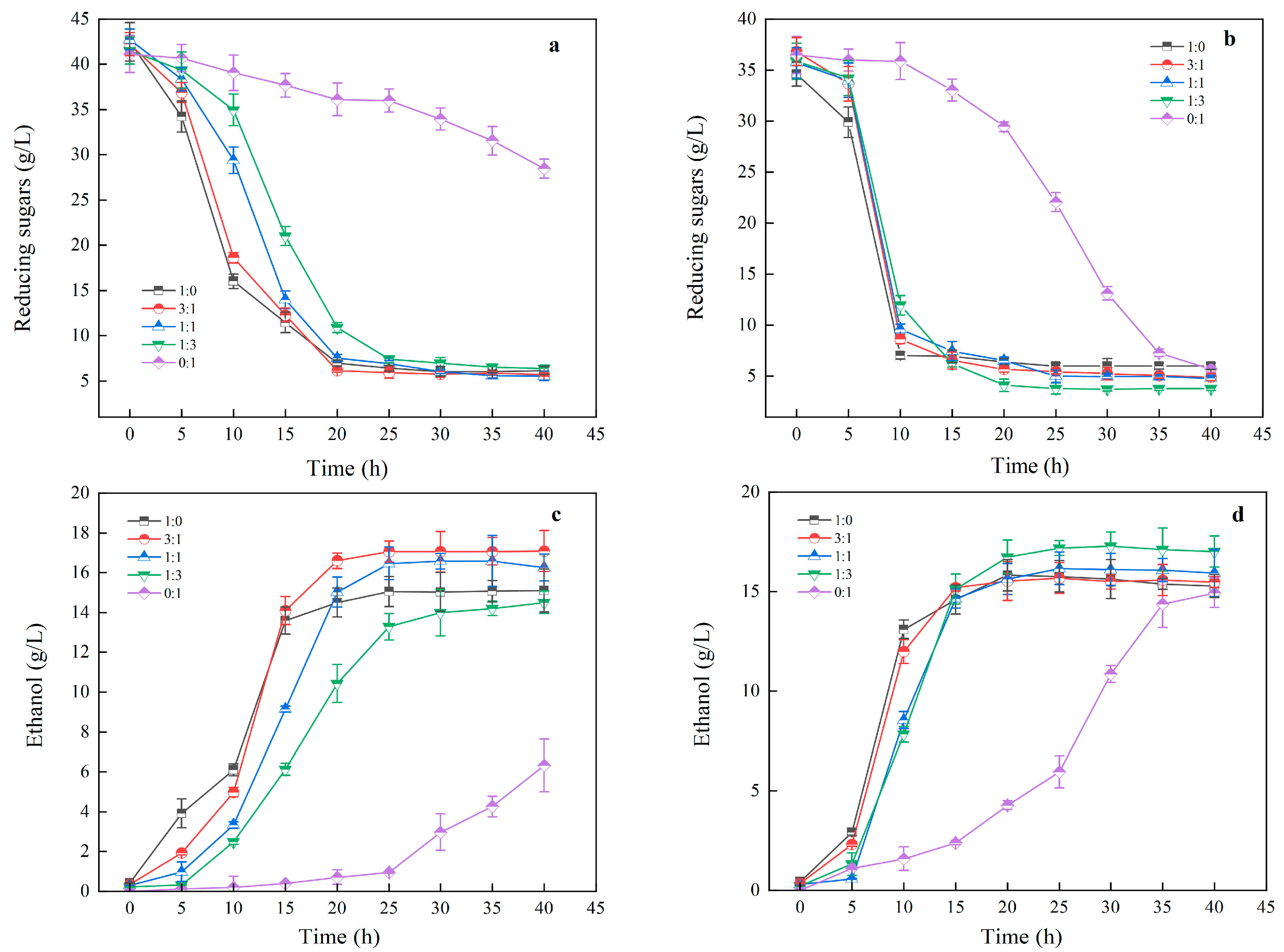
3.3.2. Impact of Mixed Substrates on Ethanol Fermentation
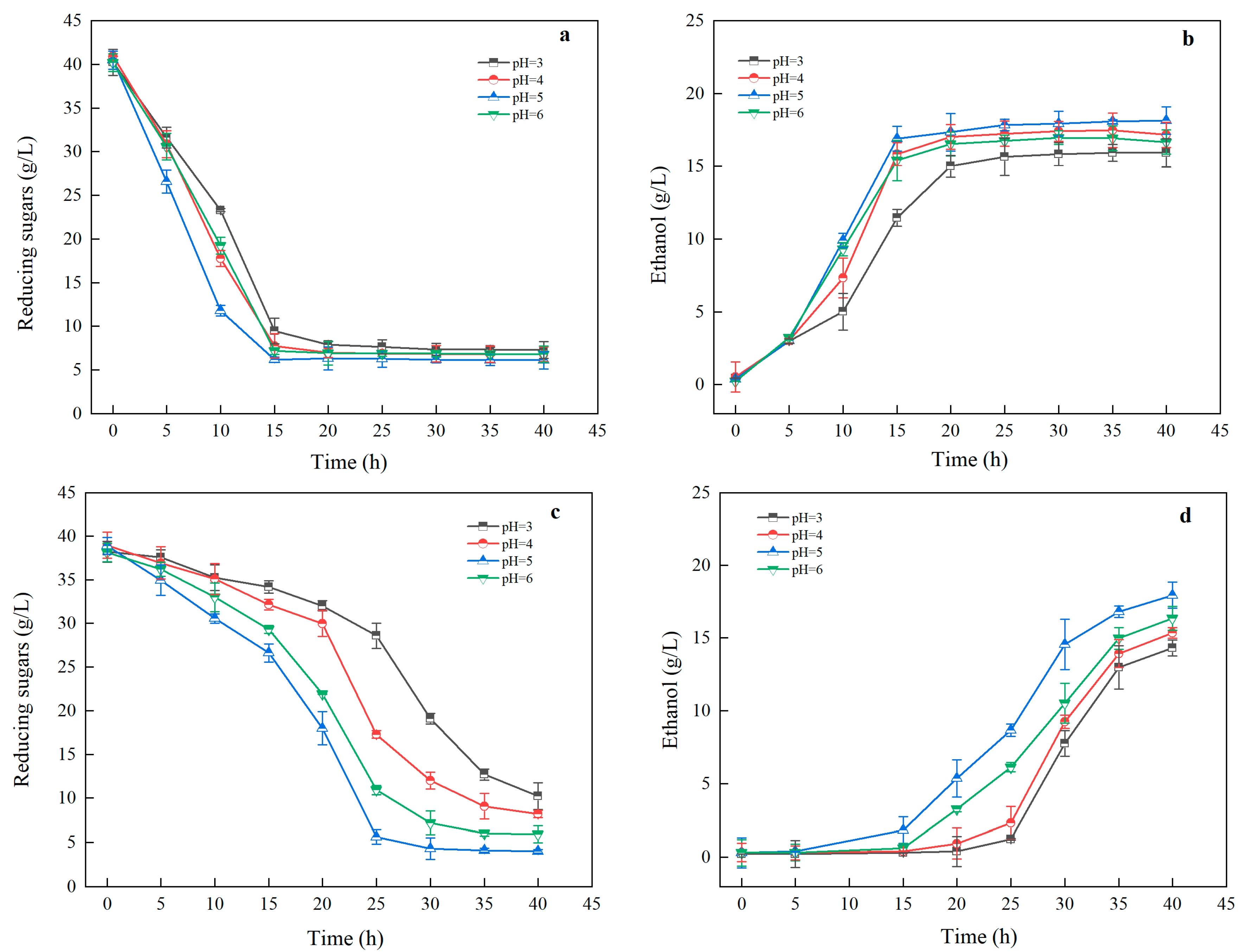
3.3.3. Impact of pH on Ethanol Fermentation
3.3.4. Impact of Mixed Culture Ratios on Ethanol Fermentation
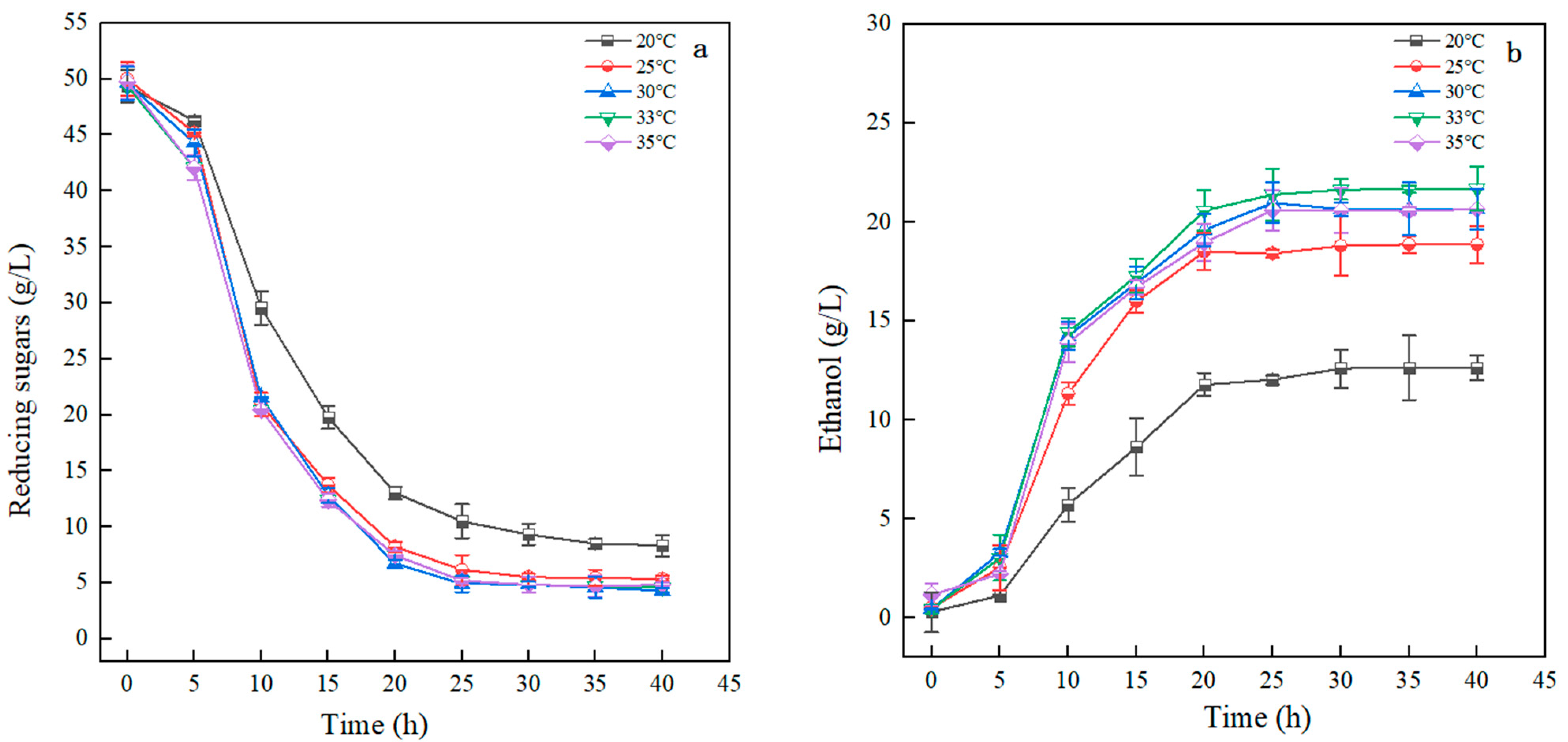
3.3.5. Impact of Temperature on Ethanol Fermentation
3.3.6. Non-Isothermal Dynamic Fermentation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Vassilev, I.; Hernandez, P.A.; Batlle-Vilanova, P.; Freguia, S.; Krömer, J.O.; Keller, J.; Ledezma, P.; Virdis, B. Microbial electrosynthesis of isobutyric, butyric, caproic acids, and corresponding alcohols from carbon dioxide. ACS Sustain. Chem. Eng. 2018, 6, 8485–8493. [Google Scholar] [CrossRef]
- Lin, Y.; Tanaka, S. Ethanol fermentation from biomass resources: Current state and prospects. Appl. Microbiol. Biotechnol. 2006, 69, 627–642. [Google Scholar] [CrossRef]
- Balat, M.; Balat, H. Recent trends in global production and utilization of bio-ethanol fuel. Appl. Energy 2009, 86, 2273–2282. [Google Scholar] [CrossRef]
- Dubey, A.K.; Gupta, P.K.; Garg, N.; Naithani, S. Bioethanol production from waste paper acid pretreated hydrolyzate with xylose fermenting pichia stipitis. Carbohydr. Polym. 2012, 88, 825–829. [Google Scholar] [CrossRef]
- Zhang, Z.; Macquarrie, D.J.; De Bruyn, M.; Budarin, V.L.; Hunt, A.J.; Gronnow, M.J.; Fan, J.; Shuttleworth, P.S.; Clark, J.H.; Matharu, A.S. Low-temperature microwave-assisted pyrolysis of waste office paper and the application of bio-oil as an al adhesive. Green Chem. Int. J. Green Chem. Resour. GC 2015, 17, 260–270. [Google Scholar] [CrossRef]
- Ioelovich, M. Waste paper-promising feedstock for bioethanol production. J. Sci. Res. Rep. 2014, 3, 905–916. [Google Scholar]
- Elliston, A.; Collins, S.R.A.; Wilson, D.R.; Roberts, I.N.; Waldron, K.W. High concentrations of cellulosic ethanol achieved by fed batch semi simultaneous saccharification and fermentation of waste-paper. Bioresour. Technol. 2013, 134, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Brummer, V.; Jurena, T.; Hlavacek, V.; Omelkova, J.; Bebar, L.; Gabriel, P.; Stehlik, P. Enzymatic hydrolysis of pretreated waste paper–source of raw material for production of liquid biofuels. Bioresour. Technol. 2014, 152, 543–547. [Google Scholar] [CrossRef] [PubMed]
- Pivnenko, K.; Eriksson, E.; Astrup, T.F. Waste paper for recycling: Overview and identification of potentially critical substances. Waste Manag. 2015, 45, 134–142. [Google Scholar] [CrossRef]
- Nishimura, H.; Tan, L.; Sun, Z.; Tang, Y.; Kida, K.; Morimura, S. Efficient production of ethanol from waste paper and the biochemical methane potential of stillage eluted from ethanol fermentation. Waste Manag. 2016, 48, 644–651. [Google Scholar] [CrossRef]
- Dinsdale, R.M.; Premier, G.C.; Hawkes, F.R.; Hawkes, D.L. Two-stage anaerobic co-digestion of waste activated sludge and fruit/vegetable waste using inclined tubular digesters. Bioresour. Technol. 2000, 72, 159–168. [Google Scholar] [CrossRef]
- Wang, Y.F.; Tan, L.; Wang, T.; Sun, Z.Y.; Tang, Y.Q.; Kida, K. Production of ethanol from kitchen waste by using flocculating saccharomyces cerevisiae kf-7. Environ. Technol. 2017, 38, 316–325. [Google Scholar] [CrossRef]
- Claassen, P.A.M.; van Lier, J.B.; Contreras, A.M.L.; van Niel, E.W.J.; Sijtsma, L. Utilisation of biomass for the supply of energy carriers. Appl. Microbiol. Biotechnol. 1999, 52, 741–755. [Google Scholar] [CrossRef]
- Lebeau, T.; Jouenne, T.; Junter, G.A. Continuous alcoholic fermentation of glucose/xylose mixtures by co-immobilized saccharomyces cerevisiae and candida shehatae. Appl. Microbiol. Biotechnol. 1998, 50, 309–313. [Google Scholar] [CrossRef]
- Limei, Y.; Ping, X.; Lijuan, Y.; Jing, L. Determination and analysis of reducing sugar in four kinds of fruits. Guangdong Chem. Ind. 2021, 48, 243–244, 248. [Google Scholar]
- Raud, M.; Tutt, M.; Olt, J.; Kikas, T. Dependence of the hydrolysis efficiency on the lignin content in lignocellulosic material. Int. J. Hydrogen Energy 2016, 41, 16338–16343. [Google Scholar] [CrossRef]
- Sharma, V.; Nargotra, P.; Sharma, S.; Bajaj, B.K. Efficacy and functional mechanisms of a novel combinatorial pretreatment approach based on deep eutectic solvent and ultrasonic waves for bioconversion of sugarcane bagasse. Renew. Energy 2021, 163, 1910–1922. [Google Scholar] [CrossRef]
- Donohoe, B.S.; Decker, S.R.; Melvin, P.T.; Himmel, M.E.; Vinzant, T.B. Visualizing lignin coalescence and migration through maize cell walls following thermochemical pretreatment. Biotechnol. Bioeng. 2008, 5, 913–925. [Google Scholar] [CrossRef] [PubMed]
- Hickert, L.R.; Da Cunha-Pereira, F.; de Souza-Cruz, P.B.; Rosa, C.A.; Ayub, M.A.Z. Ethanogenic fermentation of co-cultures of candida shehatae hm 52.2 and saccharomyces cerevisiae icv d254 in synthetic medium and rice hull hydrolysate. Bioresour. Technol. 2013, 131, 508–514. [Google Scholar] [CrossRef]
- Gírio, F.M.; Fonseca, C.; Carvalheiro, F.; Duarte, L.C.; Marques, S.; Bogel-Bukasik, R. Hemicelluloses for fuel ethanol: A review. Bioresour. Technol. 2010, 101, 4775–4800. [Google Scholar] [CrossRef]
- Wei, C.; Lan, S.; Xu, Z. Ultracytochemical localization and functional analysis of atpase during the endosperm development. Agric. Sci. China 2003, 2, 144–148. [Google Scholar]
- Liu, A.; Wang, D.; Wang, Z.; Wu, X.; Qi, H.; Deng, M.; Wang, K.; Chen, L. A saccharomyces cerevisiae strain with high tolerance:breeding by gradual acclimation and fermentation performance. Microbiol. China 2024, 51, 1018–1032. [Google Scholar]
- Xu, W.; Ln, C.; Fu, X.-Y.; Ma, T.-T.; Fu, D.-W.; Wang, W. Isolation of low ph resistant yeast and its adaptability to acid stress environment. Sci. Technol. Food Ind. 2019, 40, 112–117. [Google Scholar]
- Qin, Z.; Lai, J.; Liu, B.; Liu, J. Raman spectroscopic analysis of ethanol fermentation atvarious initial pH levels. Chin. J. Lasers 2013, 40, 0215001. [Google Scholar]
- Palmqvist, E.; Hahn-Hägerdal, B. Fermentation of Lignocellulosic Hydrolysates. II: Inhibitors and Mechanisms of Inhibition. Bioresour. Technol. 2000, 74, 25–33. [Google Scholar] [CrossRef]
- Mussatto, S.I.; Roberto, I.C. Chemical characterization and liberation of pentose sugars from brewer’s spent grain. J. Chem. Technol. Biotechnol. 2006, 81, 268–274. [Google Scholar] [CrossRef]
- Palmqvist, E.; Hahn-Hägerdal, B. Fermentation of lignocellulosic hydrolysates. I: Inhibition and detoxi®cation. Bioresour. Technol. 2000, 74, 17–24. [Google Scholar] [CrossRef]
- Tengerdy, R.P.; Szakacs, G. Bioconversion of lignocellulose in solid substrate fermentation. Biochem. Eng. J. 2003, 13, 169–179. [Google Scholar] [CrossRef]
- Yalcin, S.K.; Ozbas, Z.Y. Effects of ph and temperature on growth and glycerol production kinetics of two indigenous wine strains of saccharomyces cerevisiae from turkey. Braz. J. Microbiol. 2008, 39, 325–332. [Google Scholar] [CrossRef]
- Gamero, A.; Tronchoni, J.; Querol, A.; Belloch, C. Production of aroma compounds by cryotolerant saccharomyces species and hybrids at low and moderate fermentation temperatures. J. Appl. Microbiol. 2013, 114, 1405–1414. [Google Scholar] [CrossRef]
- Marquardt, J.; Yao, L.; Okada, H.; Svitkina, T.; Bi, E. The lkb1-like kinase elm1 controls septin hourglass assembly and stability by regulating filament pairing. Curr. Biol. 2020, 30, 2386–2394. [Google Scholar] [CrossRef] [PubMed]
- Phisalaphong, M.; Srirattana, N.; Tanthapanichakoon, W. Mathematical modeling to investigate temperature effect on kinetic parameters of ethanol fermentation. Biochem. Eng. J. 2006, 28, 36–43. [Google Scholar] [CrossRef]
- Peñuela-Martínez, A.E.; Moreno-Riascos, S.; Medina-Rivera, R. Influence of temperature-controlled fermentation on the quality of mild coffee (coffea arabica l.) Cultivated at different elevations. Agriculture 2023, 13, 1132. [Google Scholar] [CrossRef]
- Yiguan, L.I.; Qi, W.; Xuan, H.E.; Yintao, G.; Congju, C.; Qingzhen, T.; Jianxin, C. Production of sesame-flavor baijiu in pilot-scale of temperature-controlled fermentation. Food Ferment. Ind. 2020, 46, 125–130. [Google Scholar]
- Pan, Z. Effects of ambient temperature on the fen liquor making. Liquor. Mak. 2015, 42, 57–59. [Google Scholar]


| Components | Content (%) | |
|---|---|---|
| Tissue Paper | Food Waste | |
| Moisture | 2.9 ± 0.6 | 75.1 ± 0.1 |
| Dry biomass | ||
| Ash | 0.37 ± 0.019 | 9.32 ± 0.27 |
| Cellulose | 61.13 ± 3.07 | 15.2 ± 1.18 |
| Hemicellulose | 28.27 ± 1.85 | ND a |
| Lignin | 7.69 ± 1.53 | ND |
| Starch | ND | 40.3 ± 0.1 |
| Protein | 1.1 ± 0.20 | 17.4 ± 0.30 |
| Fat | ND | 2.35 ± 0.18 |
| C | 41.71 ± 2.01 | 38.97 ± 2.27 |
| N | 0.106 ± 0.0 | 3.09 ± 0.1 |
| H | 6.28 ± 0.30 | 6.033 ± 0.25 |
| Na | ND | 1.59 ± 0.37 |
| K | ND | 0.007 ± 0.0 |
| C/N | 393.49 | 12.61 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, H.; Wang, Y.; Lv, P.; Zhou, J.; Gao, M.; Qian, D.; Song, B.; Wang, Q. Ethanol Production from a Mixture of Waste Tissue Paper and Food Waste through Saccharification and Mixed-Culture Fermentation. Fermentation 2024, 10, 194. https://doi.org/10.3390/fermentation10040194
Ma H, Wang Y, Lv P, Zhou J, Gao M, Qian D, Song B, Wang Q. Ethanol Production from a Mixture of Waste Tissue Paper and Food Waste through Saccharification and Mixed-Culture Fermentation. Fermentation. 2024; 10(4):194. https://doi.org/10.3390/fermentation10040194
Chicago/Turabian StyleMa, Hongzhi, Yueyao Wang, Pin Lv, Jun Zhou, Ming Gao, Dayi Qian, Bo Song, and Qunhui Wang. 2024. "Ethanol Production from a Mixture of Waste Tissue Paper and Food Waste through Saccharification and Mixed-Culture Fermentation" Fermentation 10, no. 4: 194. https://doi.org/10.3390/fermentation10040194
APA StyleMa, H., Wang, Y., Lv, P., Zhou, J., Gao, M., Qian, D., Song, B., & Wang, Q. (2024). Ethanol Production from a Mixture of Waste Tissue Paper and Food Waste through Saccharification and Mixed-Culture Fermentation. Fermentation, 10(4), 194. https://doi.org/10.3390/fermentation10040194







