Handling and Risk Mitigation of Nanoscale Graphene and Related Materials: Some Considerations and Recommendations
Abstract
1. Introduction
2. Materials and Methods
Nomenclature
- Graphene as a generic term for nanoscale graphene
- Graphene nanoplatelets (GNP)
- Graphene nanosheets (GNS)
- Graphene oxide (GO)
- Reduced graphene oxide (rGO)
- Functionalised graphene (fG)
- Carbon nanotube (CNT)
3. Respiratory Hazards Associated with Graphene Nanoplatelets (GNP)
- Graphene nanoparticles with a high specific surface area at the nanoscale can have higher reactivity;
- Graphene-based aerosols can penetrate at a higher rate when their mass median aerodynamic diameter is <3 μm;
- Graphene of size <1 μm was found to be going through lungs less than that of smaller sized materials;
- GO sheets of larger size were assessed to be having a stronger antibacterial activity to Escherichia coli (E-coli) than those of smaller sheets;
- Cell membranes can easily be affected by sharp edges of the sheets;
- The lung macrophages were less susceptible to when the aspect ratio of nanomaterials are >3 and a length of at least 10 μm;
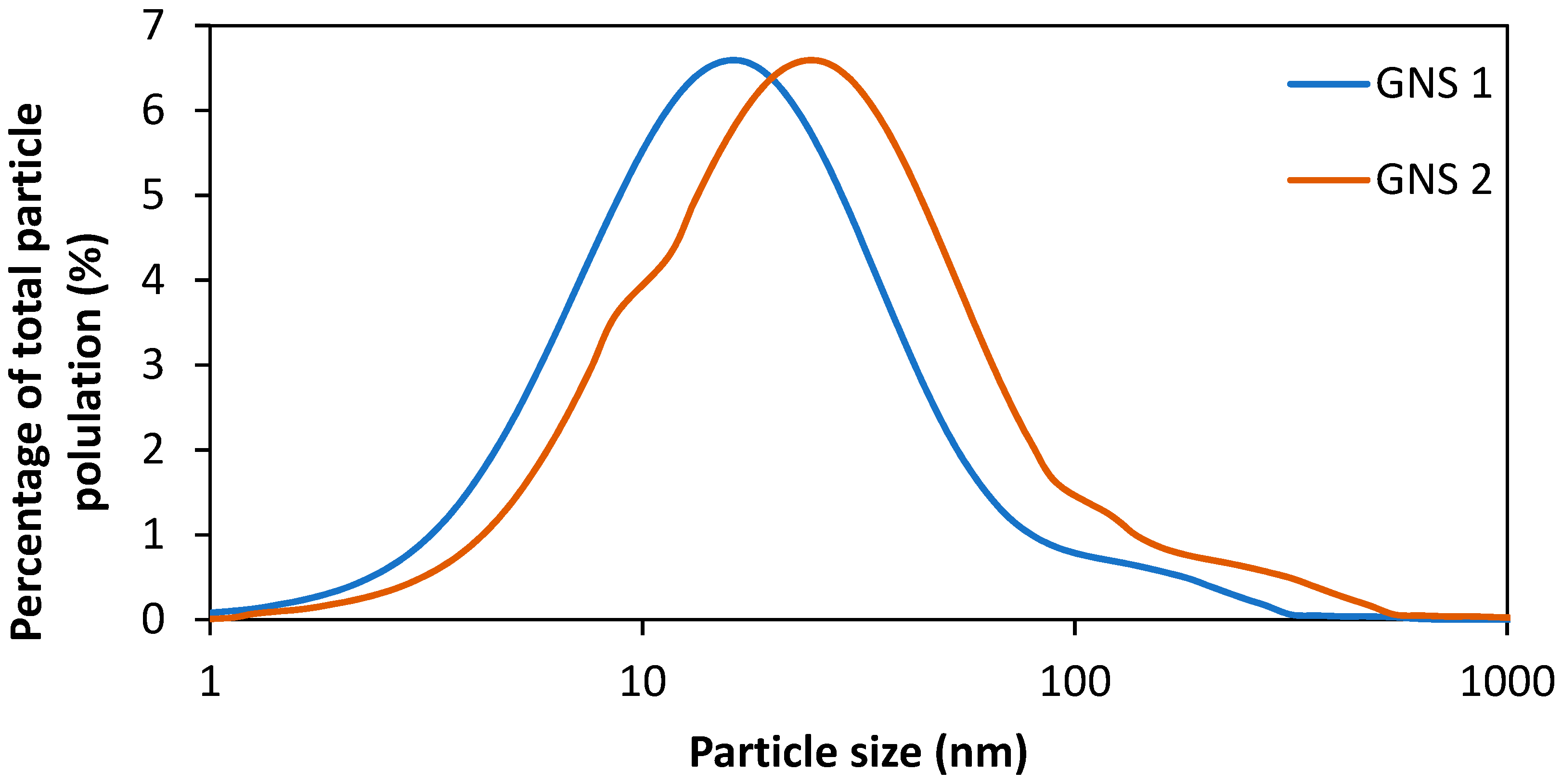
- Increased agglomeration/aggregation of GNP can reduce the hemolytic activity;
- Aggregations/agglomerate can be formed by high concentrations of nanoparticles, by which translocation may be reduced in biological blockades;
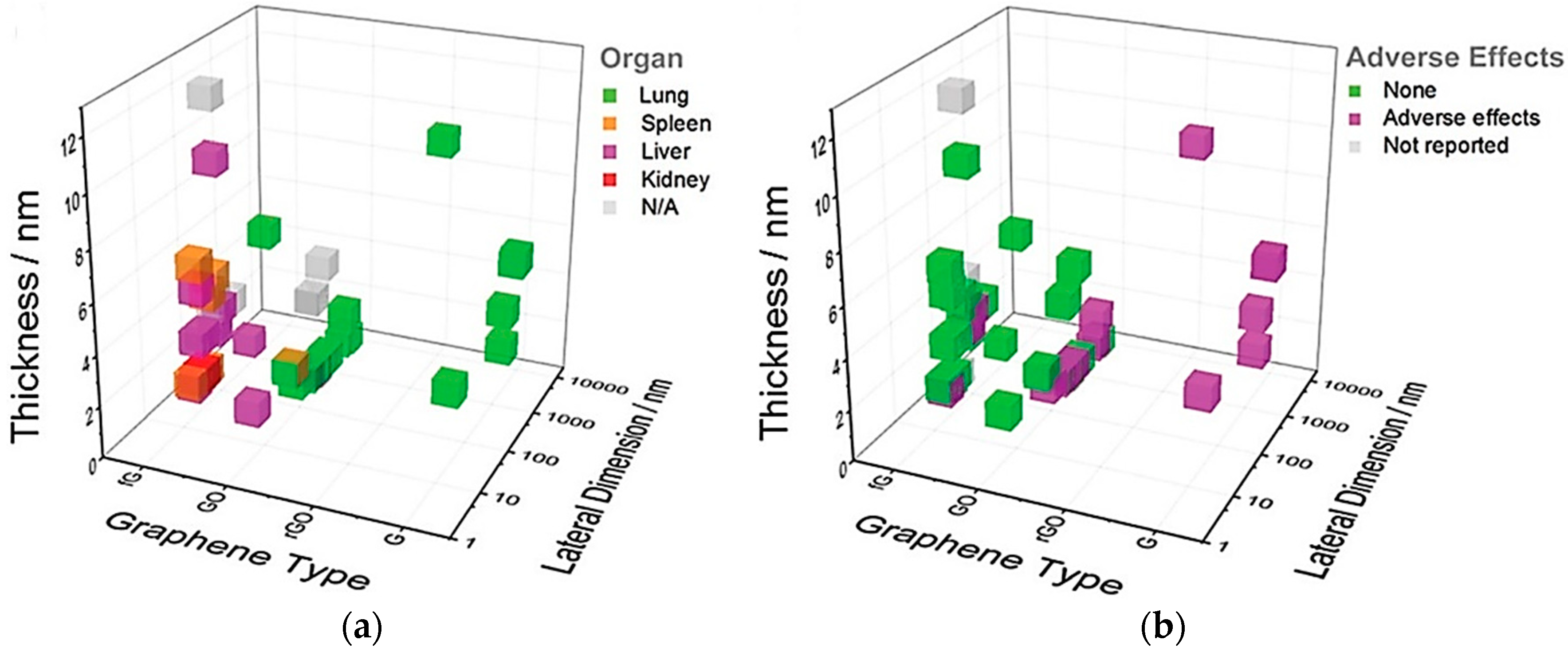
- Human neutrophil based myeloxidase can degrade highly dispersed graphene nanoparticles, while the agglomerated state of nanoparticles does not get degraded by the same (Figure 3 shows the effect of different graphene materials on human organs upon exposure); however, the degradation of GNP may not be directly related to the organs where the GNP precipitate;
- Hydrophilic graphene is found to be more biocompatible than hydrophobic graphene;
- Biological membranes, such as Gram-positive versus Gram-negative bacteria can be influenced by altered surface charges;
- Surface contaminants of graphene may cause potential harm to the human system.
4. Considerations and Recommendations
- Given that the extent of any potential airborne particle contamination is unknown, it is recommended that a particle survey should be undertaken to quantify any airborne particle contamination with graphene. If airborne contamination is detected, such a survey would help with risk mitigation measures such as the provision of local, flexible enclosures, or rebalancing local ventilation systems in a laboratory.
- Identification of processes and procedures in which graphene is used, and where it could become airborne. It would help in the understanding of how local ventilation systems are used in each of these processes, and inform how risk mitigation measures could be improved. Possible exposure monitoring devices would be helpful to identify ENM contamination. Strategic goals should be developed in order to protect researchers using ENM and to translate the research findings of the effects of ENM into workplace practice efficiently [19].
- Gaining confirmation from manufactures of P3 masks would provide adequate protection for graphene with a Dae of 3 microns and, if necessary, ensure the masks are individually fitted.
- To reduce the risk of handling graphene:
- Limiting the amount of nanomaterials or batch size of samples would reduce the risk of airborne contamination and exposure to the skin [20];
- Containing graphene in a closed cabinet surrounded by appropriate materials is necessary to avoid spills [21];
- When preparing dispersions, contain graphene as much as possible, particularly while it is being subjected to ultrasonication;
- It is also wise to inform co-workers/researchers who are not working with ENM to alert them how to mitigate risks associated with such materials;
- Safe-by-design tactics changing the biological activity via reducing the toxicity-related effects will have high effectiveness in occupational risk control hierarchy, which would highly suitable for ENM.
- Reviewing of clean up and waste disposal procedures is necessary to ensure these are adequate and up to date. Vacuum cleaners fitted with high efficiency particulate arrestance (HEPA)filters should be used. Waste containing or contaminated with graphene should be separately bagged and sent out for deep burial by authorised personnel.
- A fume hood with HEPA filters and a weighing balance should be dedicated for graphene to avoid any sort of contamination. All of the weighing processes are carried out under the fume hood. Likely consequences of exposure to graphene particles over 2 μm are respiratory or skin irritation/inflammation.
- Use a vacuum cleaner with a HEPA filter for loose powder:
- Small powder spills (typically involving less than 5 mg of material) of nano or micron-sized graphene must be removed using cloth/gauze/absorbent using appropriate removal material (for example, soapy water). The contaminated surfaces must be cleaned several times until no trace of nanomaterial is visible. One can use a clear cloth to make the surface is cleaned enough. The contaminated clean-up materials should be disposed of in an appropriate way [11,20,22];
- Large spills need to be taken care of by professionally trained cleaners;
- Figure 4 shows nano-enclosure and protective measures while handling ENM.
- Facilities handling nanomaterials should have a clear and concise safe work instruction (SWI) manual, log sheet and drill press document to allow users (particularly new users) to be aware of safe handling and housekeeping.
- A proper and detailed risk assessment should be carried out for any class of nanomaterials that is under usage or experimentation. This will inform the technician and users on how to mitigate the problem when it arises. The document should be regularly updated and revised upon necessity [23].
- Disposal of nanoparticles [24]:
- Graphene materials should not be disposed of as general waste compounds in sinks or drains;
- If any ENM is dispersed in any acid medium or reductants, it should be neutralised before disposal;
- Decanting liquids containing graphene should be disposed of with the help of chemical waste disposal companies;
- The appropriate disposal protocols should be sufficiently communicated to all the users and non-users, and integration between different scientific disciplines should be properly carried out.
5. Summary
Funding
Conflicts of Interest
References and Notes
- Code of Federal Regulations: Title 40—Protection of Environment: Chapter 1—Environmental Protection Agency (Continued). Subchapter R-Toxic Substances Control Act. Part 721—Significant New Uses of Chemical Substances; Environmental Protection Agency: Washington, DC, USA, 2005.
- Kurian, A.S.; Mohan, V.B.; Bhattacharyya, D. Embedded large strain sensors with graphene-carbon black-silicone rubber composites. Sens. Actuators A: Phys. 2018, 282, 206–214. [Google Scholar] [CrossRef]
- Mohan, V.B.; Brown, R.; Jayaraman, K.; Bhattacharyya, D. Optimisation of hybridisation effect in graphene reinforced polymer nanocomposites. Adv. Compos. Mater. 2017, 1–17. [Google Scholar] [CrossRef]
- Mohan, V.B.; Jayaraman, K.; Bhattacharyya, D. Relevance of adhesion in particulate/fibre- polymer composites and particle coated fibre yarns: A critical review. Rev. Adhes. Adhes. 2016, 4, 119–151. [Google Scholar] [CrossRef]
- Mohan, V.B.; Jayaraman, K.; Bhattacharyya, D. Fabrication of highly conductive graphene particle-coated fiber yarns using polymeric binders through efficient coating techniques. Adv. Polym. Technol. 2018. [Google Scholar] [CrossRef]
- Mohan, V.B.; Krebs, B.J.; Bhattacharyya, D. Development of novel highly conductive 3D printable hybrid polymer-graphene composites. Mater. Today Commun. 2018, 17, 554–561. [Google Scholar] [CrossRef]
- Mohan, V.B.; Lau, K.-T.; Hui, D.; Bhattacharyya, D. Graphene-based materials and their composites: A review on production, applications and product limitations. Compos. Part B: Eng. 2018, 142, 200–220. [Google Scholar] [CrossRef]
- SEM of graphene nanoplatelets—Supplied by Emfutur Technologies Spain.
- Li, D.; Müller, M.B.; Gilje, S.; Kaner, R.B.; Wallace, G.G. Processable aqueous dispersions of graphene nanosheets. Nat. Nanotechnol. 2008, 3, 101. [Google Scholar] [CrossRef] [PubMed]
- Schinwald, A.; Murphy, F.A.; Jones, A.; MacNee, W.; Donaldson, K. Graphene-based nanoplatelets: A new risk to the respiratory system as a consequence of their unusual aerodynamic properties. ACS Nano 2012, 6, 736–746. [Google Scholar] [CrossRef] [PubMed]
- Bianco, A. Graphene: Safe or toxic? The two faces of the medal. Angew. Chem. Int. Ed. 2013, 52, 4986–4997. [Google Scholar] [CrossRef] [PubMed]
- Park, M.V.; Bleeker, E.A.; Brand, W.; Cassee, F.R.; van Elk, M.; Gosens, I.; de Jong, W.H.; Meesters, J.A.; Peijnenburg, W.J.; Quik, J.T. Considerations for safe innovation: The case of Graphene. ACS Nano 2017, 11, 9574–9593. [Google Scholar] [CrossRef] [PubMed]
- Graphene Toxicity—The Interview by The Graphene Counsil dated July 2014.
- Nowack, B.; Ranville, J.F.; Diamond, S.; Gallego-Urrea, J.A.; Metcalfe, C.; Rose, J.; Horne, N.; Koelmans, A.A.; Klaine, S.J. Potential scenarios for nanomaterial release and subsequent alteration in the environment. Environ. Toxicol. Chem. 2012, 31, 50–59. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Kiratipaiboon, C.; Porter, D.W.; Rojanasakul, L.W.; Dinu, C.Z.; Wang, K.; Yang, Y.; Rojanasakul, Y. Predicting Nanotube Fibrogenicity through Stem Cell-Mediated Fibroblast Focus and Spheroid Formation. Nano Lett. 2018, 18, 6500–6508. [Google Scholar] [CrossRef] [PubMed]
- Bradley, D. Is graphene safe? Mater. Today 2012, 15, 230. [Google Scholar] [CrossRef]
- Lo, L.M.; Hammond, D.; Bartholomew, I.; Almaguer, D.; Heitbrink, W.; Topmiller, J. Engineering Controls for Nano-Scale Graphene Platelets During Manufacturing and Handling Processes; Dep. Health Hum. Serv. Cent. Dis. Control. Prev. Natl Inst Occup. Safety Health: Washington, DC, USA, 2011.
- Bussy, C.; Jasim, D.; Lozano, N.; Terry, D.; Kostarelos, K. The current graphene safety landscape—A literature mining exercise. Nanoscale 2015, 7, 6432–6435. [Google Scholar] [CrossRef] [PubMed]
- Kuempel, E.D.; Geraci, C.L.; Schulte, P.A. Risk assessment and risk management of nanomaterials in the workplace: Translating research to practice. Ann. Occup. Hyg. 2012, 56, 491–505. [Google Scholar] [PubMed]
- Bussy, C.; Ali-Boucetta, H.; Kostarelos, K. Safety considerations for graphene: Lessons learnt from carbon nanotubes. Acc. Chem. Res. 2012, 46, 692–701. [Google Scholar] [CrossRef] [PubMed]
- Laux, P.; Tentschert, J.; Riebeling, C.; Braeuning, A.; Creutzenberg, O.; Epp, A.; Fessard, V.; Haas, K.H.; Haase, A.; Hund-Rinke, K. Nanomaterials: Certain aspects of application, risk assessment and risk communication. Arch. Toxicol. 2018, 92, 121–141. [Google Scholar] [CrossRef] [PubMed]
- Freeland, J.; Hulme, J.; Kinnison, D.; Mitchell, A.; Veitch, P.; Aitken, R.; Hankin, S.; Poland, C.; Bard, D.; Gibson, R.; et al. Working Safely with Nanomaterials in Research & Development by The UK NanoSafety Partnership Group (UKNSPG). Available online: https://www.safenano.org/media/64896/Working%20Safely%20with%20Nanomaterials%20-%20Release%201%200%20-%20Aug2012.pdf (accessed on 15 May 2019).
- Seabra, A.B.; Paula, A.J.; de Lima, R.; Alves, O.L.; Durán, N. Nanotoxicity of graphene and graphene oxide. Chem. Res. Toxicol. 2014, 27, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Kong, B.; Seog, J.H.; Graham, L.M.; Lee, S.B. Experimental considerations on the cytotoxicity of nanoparticles. Nanomedicine 2011, 6, 929–941. [Google Scholar] [CrossRef] [PubMed]


© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mohan, V.B. Handling and Risk Mitigation of Nanoscale Graphene and Related Materials: Some Considerations and Recommendations. C 2019, 5, 36. https://doi.org/10.3390/c5030036
Mohan VB. Handling and Risk Mitigation of Nanoscale Graphene and Related Materials: Some Considerations and Recommendations. C. 2019; 5(3):36. https://doi.org/10.3390/c5030036
Chicago/Turabian StyleMohan, Velram Balaji. 2019. "Handling and Risk Mitigation of Nanoscale Graphene and Related Materials: Some Considerations and Recommendations" C 5, no. 3: 36. https://doi.org/10.3390/c5030036
APA StyleMohan, V. B. (2019). Handling and Risk Mitigation of Nanoscale Graphene and Related Materials: Some Considerations and Recommendations. C, 5(3), 36. https://doi.org/10.3390/c5030036