Cross-Linked Gel Polymer Electrolyte Based on Multiple Epoxy Groups Enabling Conductivity and High Performance of Li-Ion Batteries
Abstract
1. Introduction
2. Results and Discussion
2.1. Ionic Conductivity and Electrochemical Properties
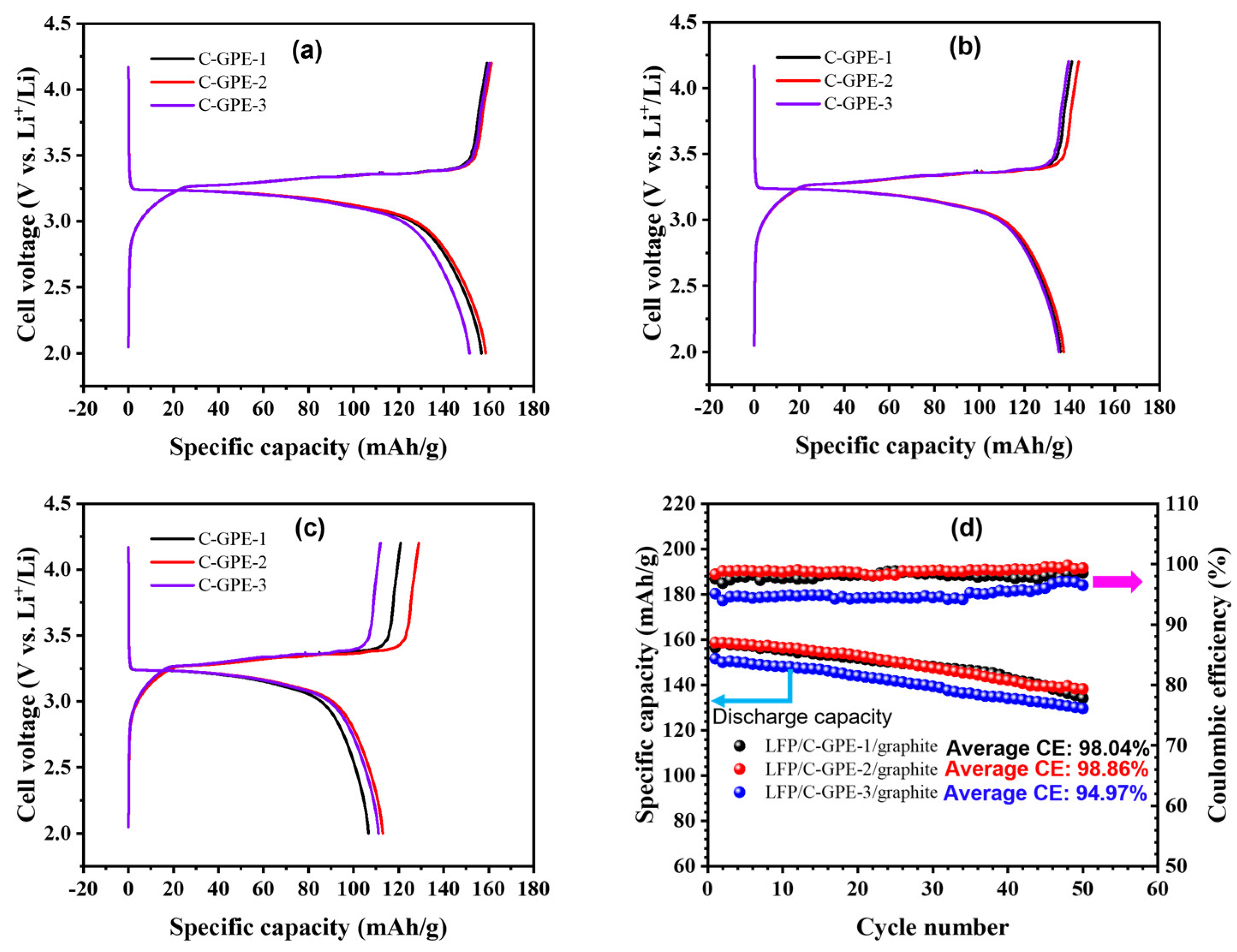
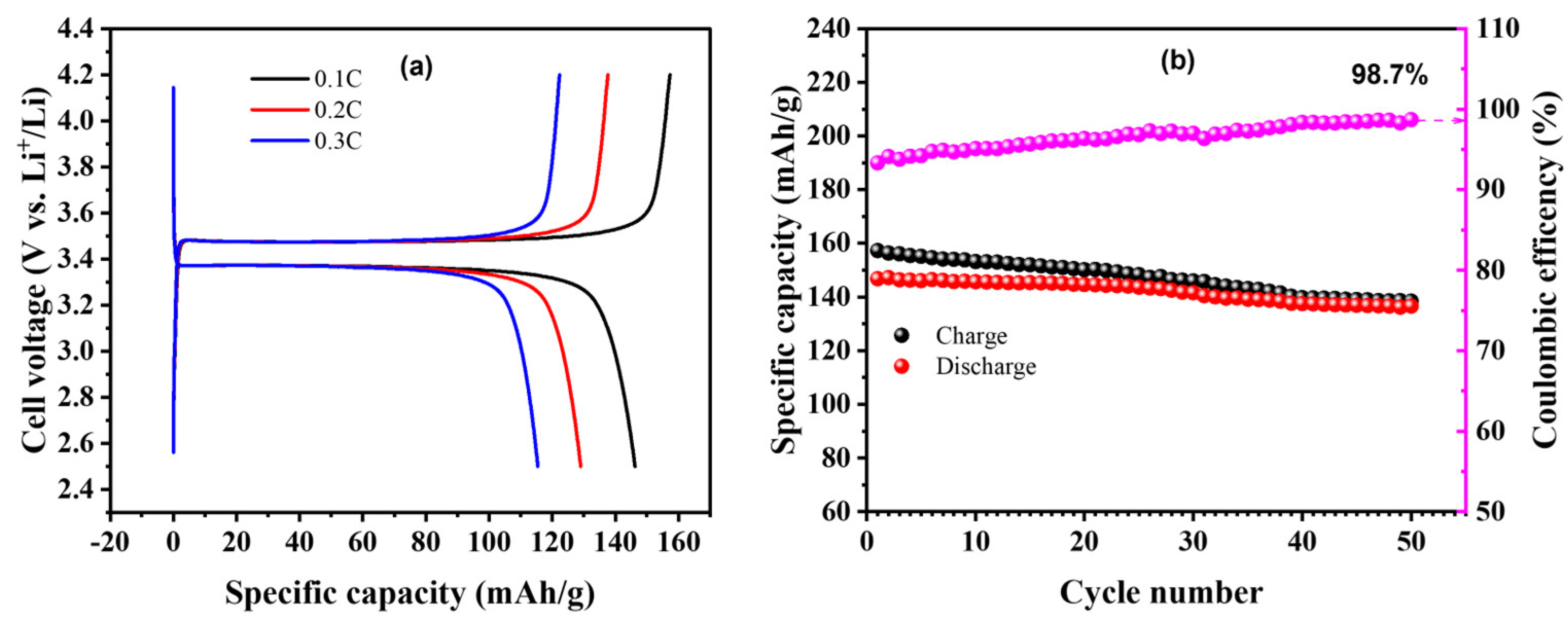
2.2. Battery Performances
2.3. Morphological Characterization
2.4. Characterization of As-Synthesized C-GPEs
2.5. In Situ Cross-Linking Gel Polymer Electrolytes
3. Conclusions
4. Materials and Methods
4.1. Sources
4.2. Characterization and Measurements
4.3. Electrochemical Measurements
4.4. In Situ-Polymerized Cross-Linked Gel Polymer Electrolytes (C-GPEs)
4.5. Preparation of LFP Cathode
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liu, K.; Liu, Y.; Lin, D.; Pei, A.; Cui, Y. Materials for lithium-ion battery safety. Sci. Adv. 2018, 4, eaas9820. [Google Scholar] [CrossRef] [PubMed]
- Xia, S.; Wu, X.; Zhang, Z.; Cui, Y.; Liu, W. Practical Challenges and Future Perspectives of All-Solid-State Lithium-Metal Batteries. Chemistry 2019, 5, 753–785. [Google Scholar] [CrossRef]
- Liu, Q.; Cai, B.; Li, S.; Yu, Q.; Lv, F.; Kang, F.; Wang, Q.; Li, B. Long-cycling and safe lithium metal batteries enabled by the synergetic strategy of ex situ anodic pretreatment and an in-built gel polymer electrolyte. J. Mater. Chem. A 2020, 8, 7197–7204. [Google Scholar] [CrossRef]
- Zhu, J.; Zhang, J.; Zhao, R.; Zhao, Y.; Liu, J.; Xu, N.; Wan, X.; Li, C.; Ma, Y.; Zhang, H.; et al. In situ 3D crosslinked gel polymer electrolyte for ultra-long cycling, high-voltage, and high-safety lithium metal batteries. Energy Storage Mater. 2023, 57, 92–101. [Google Scholar] [CrossRef]
- Wan, J.; Xie, J.; Kong, X.; Liu, Z.; Liu, K.; Shi, F.; Pei, A.; Chen, H.; Chen, W.; Chen, J.; et al. Ultrathin, flexible, solid polymer composite electrolyte enabled with aligned nanoporous host for lithium batteries. Nat. Nanotechnol. 2019, 14, 705–711. [Google Scholar] [CrossRef]
- Zhang, W.; Jin, L.; Lee, S.; Bae, W.; Park, S.; Jeon, M.; Kim, W.; Jang, H. In situ induced crosslinking highly conductive solid polymer electrolyte with intimated electrodes interfacial compatibility for safe Li-ion batteries. J. Power Sources 2023, 557, 232568. [Google Scholar] [CrossRef]
- Long, L.; Wang, S.; Xiao, M.; Meng, Y. Polymer electrolytes for lithium polymer batteries. J. Mater. Chem. A 2016, 4, 10038–10069. [Google Scholar] [CrossRef]
- Park, S.; Sohn, J.-Y.; Hwang, I.-T.; Shin, J.; Yun, J.-M.; Eom, K.; Shin, K.; Lee, Y.-M.; Jung, C.-H. In-situ preparation of gel polymer electrolytes in a fully-assembled lithium ion battery through deeply-penetrating high-energy electron beam irradiation. Chem. Eng. J. 2023, 452, 139339. [Google Scholar] [CrossRef]
- Wu, K.; Cui, J.; Yi, J.; Liu, X.; Ning, F.; Liu, Y.; Zhang, J. Biodegradable Gel Electrolyte Suppressing Water-Induced Issues for Long-Life Zinc Metal Anodes. ACS Appl. Mater. Interfaces 2022, 14, 34612–34619. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, J.; Han, W.; Zhang, J.; Ding, G.; Dong, S.; Cui, G. Review—In Situ Polymerization for Integration and Interfacial Protection Towards Solid State Lithium Batteries. J. Electrochem. Soc. 2020, 167, 0700527. [Google Scholar] [CrossRef]
- Liang, S.; Yan, W.; Wu, X.; Zhang, Y.; Zhu, Y.; Wang, H.; Wu, Y. Gel polymer electrolytes for lithium ion batteries: Fabrication, characterization and performance. Solid. State Ion. 2018, 318, 2–18. [Google Scholar] [CrossRef]
- Ma, C.; Cui, W.; Liu, X.; Ding, Y.; Wang, Y. In situ preparation of gel polymer electrolyte for lithium batteries: Progress and perspectives. InfoMat 2021, 4, e12232. [Google Scholar] [CrossRef]
- Chen, D.; Zhu, M.; Kang, P.; Zhu, T.; Yuan, H.; Lan, J.; Yang, X.; Sui, G. Self-Enhancing Gel Polymer Electrolyte by In Situ Construction for Enabling Safe Lithium Metal Battery. Adv. Sci. 2022, 9, e2103663. [Google Scholar] [CrossRef]
- Xiao, Q.; Deng, C.; Wang, Q.; Zhang, Q.; Yue, Y.; Ren, S. In Situ Cross-Linked Gel Polymer Electrolyte Membranes with Excellent Thermal Stability for Lithium Ion Batteries. ACS Omega 2019, 4, 95–103. [Google Scholar] [CrossRef]
- Ren, W.; Zhang, Y.; Lv, R.; Guo, S.; Wu, W.; Liu, Y.; Wang, J. In-situ formation of quasi-solid polymer electrolyte for improved lithium metal battery performances at low temperatures. J. Power Sources 2022, 542, 231773. [Google Scholar] [CrossRef]
- Fu, F.; Zheng, Y.; Jiang, N.; Liu, Y.; Sun, C.; Zhang, A.; Teng, H.; Sun, L.; Xie, H. A Dual-Salt PEO-based polymer electrolyte with Cross-Linked polymer network for High-Voltage lithium metal batteries. Chem. Eng. J. 2022, 450, 137776. [Google Scholar] [CrossRef]
- Zhang, W.; Yoon, S.; Jin, L.; Lim, H.; Jeon, M.; Jang, H.; Ahmed, F.; Kim, W. Lithium Salt Catalyzed Ring-Opening Polymerized Solid-State Electrolyte with Comparable Ionic Conductivity and Better Interface Compatibility for Li-Ion Batteries. Membranes 2022, 12, 330. [Google Scholar] [CrossRef]
- Zhang, W.; Ryu, T.; Yoon, S.; Jin, L.; Jang, G.; Bae, W.; Kim, W.; Ahmed, F.; Jang, H. Synthesis and Characterization of Gel Polymer Electrolyte Based on Epoxy Group via Cationic Ring-Open Polymerization for Lithium-Ion Battery. Membranes 2022, 12, 439. [Google Scholar] [CrossRef]
- Wang, Q.; Xu, X.; Hong, B.; Bai, M.; Li, J.; Zhang, Z.; Lai, Y. Molecular engineering of a gel polymer electrolyte via in-situ polymerization for high performance lithium metal batteries. Chem. Eng. J. 2022, 428, 131331. [Google Scholar] [CrossRef]
- Cheng, H.; Zhu, J.; Jin, H.; Gao, C.; Liu, H.; Cai, N.; Liu, Y.; Zhang, P.; Wang, M. In situ initiator-free gelation of highly concentrated lithium bis(fluorosulfonyl)imide-1,3-dioxolane solid polymer electrolyte for high performance lithium-metal batteries. Mater. Today Energy 2021, 20, 100623. [Google Scholar] [CrossRef]
- Castillo, J.; Santiago, A.; Judez, X.; Garbayo, I.; Coca Clemente, J.A.; Morant-Miñana, M.C.; Villaverde, A.; González-Marcos, J.A.; Zhang, H.; Armand, M.; et al. Safe, Flexible, and High-Performing Gel-Polymer Electrolyte for Rechargeable Lithium Metal Batteries. Chem. Mater. 2021, 33, 8812–8821. [Google Scholar] [CrossRef]
- Niu, Y.-B.; Yin, Y.-X.; Wang, W.-P.; Wang, P.-F.; Ling, W.; Xiao, Y.; Guo, Y.-G. In Situ Copolymerizated Gel Polymer Electrolyte with Cross-Linked Network for Sodium-Ion Batteries. CCS Chem. 2020, 2, 589–597. [Google Scholar] [CrossRef]
- Zhou, L.; Liu, S.; Li, W.; Song, H.; Du, L.; Cui, Z. Highly conductive Poly(ε-caprolactone) and chitosan based polymer electrolyte for lithium metal battery. J. Power Sources 2023, 553, 2322271. [Google Scholar] [CrossRef]
- Hu, J.J.; Long, G.K.; Liu, S.; Li, G.R.; Gao, X.P. A LiFSI-LiTFSI binary-salt electrolyte to achieve high capacity and cycle stability for a Li-S battery. Chem. Commun. 2014, 50, 14647–14650. [Google Scholar] [CrossRef]
- Cai, Y.; Zhang, H.; Cao, Y.; Wang, Q.; Cao, B.; Zhou, Z.; Lv, F.; Song, W.; Duo, D.; Yu, L. Synthesis, application and industrialization of LiFSI: A review and perspective. J. Power Sources 2022, 535, 231481. [Google Scholar] [CrossRef]
- Bai, P.; Ji, X.; Zhang, J.; Zhang, W.; Hou, S.; Su, H.; Li, M.; Deng, T.; Cao, L.; Liu, S.; et al. Formation of LiF-rich Cathode-Electrolyte Interphase by Electrolyte Reduction. Angew. Chem. Int. Ed. Engl. 2022, 61, e202202731. [Google Scholar] [CrossRef]
- Li, Z.; Liu, Q.; Deng, Y.; Zhou, M.; Tang, W.; Dong, H.; Zhao, W.; Liu, R. In situ cross-linked plastic crystal electrolytes toward superior lithium metal batteries. Mater. Today Energy 2023, 31, 101198. [Google Scholar] [CrossRef]
- Wei, J.; Yue, H.; Shi, Z.; Li, Z.; Li, X.; Yin, Y.; Yang, S. In Situ Gel Polymer Electrolyte with Inhibited Lithium Dendrite Growth and Enhanced Interfacial Stability for Lithium-Metal Batteries. ACS Appl. Mater. Interfaces 2021, 13, 32486–32494. [Google Scholar] [CrossRef]
- Qi, M.; Xu, Y.J.; Rao, W.H.; Luo, X.; Chen, L.; Wang, Y.Z. Epoxidized soybean oil cured with tannic acid for fully bio-based epoxy resin. RSC Adv. 2018, 8, 26948–26958. [Google Scholar] [CrossRef]
- Liu, Z.; Erhan, S.Z. Ring-Opening Polymerization of Epoxidized Soybean Oil. J. Am. Oil Chem. Soc. 2009, 87, 437–444. [Google Scholar] [CrossRef]
- Öztürk, C.; Küsefoğlu, S.H. Polymerization of epoxidized soybean oil with maleinized soybean oil and maleic anhydride grafted polypropylene mixtures. J. Appl. Polym. Sci. 2010, 118, 3311–3317. [Google Scholar] [CrossRef]
- Zhang, H.; Arcelus, O.; Carrasco, J. Role of asymmetry in the physiochemical and electrochemical behaviors of perfluorinated sulfonimide anions for lithium batteries: A DFT study. Electrochim. Acta 2018, 280, 290–299. [Google Scholar] [CrossRef]
- Wang, Y.; Cao, Z.; Ma, Z.; Liu, G.; Cheng, H.; Zou, Y.; Cavallo, L.; Li, Q.; Ming, J. Weak Solvent—Solvent Interaction Enables High Stability of Battery Electrolyte. ACS Energy Lett. 2023, 8, 1477–1484. [Google Scholar] [CrossRef]
- Mackanic, D.G.; Michaels, W.; Lee, M.; Feng, D.; Lopez, J.; Qin, J.; Cui, Y.; Bao, Z. Crosslinked Poly(tetrahydrofuran) as a Loosely Coordinating Polymer Electrolyte. Adv. Energy Mater. 2018, 8, 1800703. [Google Scholar] [CrossRef]
- Wang, Q.; Dong, T.; Zhou, Q.; Cui, Z.; Shangguan, X.; Lu, C.; Lv, Z.; Chen, K.; Huang, L.; Zhang, H.; et al. An in-situ generated composite solid-state electrolyte towards high-voltage lithium metal batteries. Sci. China Chem. 2022, 65, 934–942. [Google Scholar] [CrossRef]
- Shin, H.; Choi, S.J.; Choi, S.; Jang, B.Y.; Jeong, J.; Cho, Y.-G.; Lee, S.-Y.; Song, H.-K.; Yu, J.H.; Kim, T.-H. In situ gel electrolyte network guaranteeing ionic communication between solid electrolyte and cathode. J. Power Sources 2022, 546, 231926. [Google Scholar] [CrossRef]
- Chiu, L.-L.; Chung, S.-H. Composite gel-polymer electrolyte for high-loading polysulfide cathodes. J. Mater. Chem. A 2022, 10, 13719. [Google Scholar] [CrossRef]
- Kang, P.; Chen, D.; Wu, L.; Yang, X.; Sui, G. Insight into poly(1,3-dioxolane)-based polymer electrolytes and their interfaces with lithium Metal: Effect of electrolyte compositions. Chem. Eng. J. 2023, 455, 140931. [Google Scholar] [CrossRef]
- Lin, Y.; Wang, T.; Zhang, L.; Peng, X.; Huang, B.; Wu, M.; Zhao, T. In-situ forming lithiophilic-lithiophobic gradient interphases for dendrite-free all-solid-state Li metal batteries. Nano Energy 2022, 99, 107395. [Google Scholar] [CrossRef]
- Ahmed, F.; Choi, I.; Ryu, T.; Yoon, S.; Rahman, M.M.; Zhang, W.; Jang, H.; Kim, W. Highly conductive divalent fluorosulfonyl imide based electrolytes improving Li-ion battery performance: Additive potentiating electrolytes action. J. Power Sources 2020, 455, 227980. [Google Scholar] [CrossRef]
- Kasetaite, S.; De la Flor, S.; Serra, A.; Ostrauskaite, J. Effect of Selected Thiols on Cross-Linking of Acrylated Epoxidized Soybean Oil and Properties of Resulting Polymers. Polymers 2018, 10, 439. [Google Scholar] [CrossRef] [PubMed]
- Kerner, M.; Plylahan, N.; Scheers, J.; Johansson, P. Thermal stability and decomposition of lithium bis(fluorosulfonyl)imide (LiFSI) salts. RSC Adv. 2016, 6, 23327–23334. [Google Scholar] [CrossRef]
- Huang, S.; Cui, Z.; Qiao, L.; Xu, G.; Zhang, J.; Tang, K.; Liu, X.; Wang, Q.; Zhou, X.; Zhang, B.; et al. An in-situ polymerized solid polymer electrolyte enables excellent interfacial compatibility in lithium batteries. Electrochim. Acta 2019, 299, 820–827. [Google Scholar] [CrossRef]
- Nair, J.R.; Shaji, I.; Ehteshami, N.; Thum, A.; Diddens, D.; Heuer, A.; Winter, M. Solid Polymer Electrolytes for Lithium Metal Battery via Thermally Induced Cationic Ring-Opening Polymerization (CROP) with an Insight into the Reaction Mechanism. Chem. Mater. 2019, 31, 3118–3133. [Google Scholar] [CrossRef]
- Zhao, Q.; Liu, X.; Stalin, S.; Khan, K.; Archer, L.A. Solid-state polymer electrolytes with in-built fast interfacial transport for secondary lithium batteries. Nat. Energy 2019, 4, 365–373. [Google Scholar] [CrossRef]
- Chen, B.; Xu, Q.; Huang, Z.; Zhao, Y.; Chen, S.; Xu, X. One-pot preparation of new copolymer electrolytes with tunable network structure for all-solid-state lithium battery. J. Power Sources 2016, 331, 322–331. [Google Scholar] [CrossRef]
- Yao, W.; Zhang, Q.; Qi, F.; Zhang, J.; Liu, K.; Li, J.; Chen, W.; Du, Y.; Jin, Y.; Liang, Y.; et al. Epoxy containing solid polymer electrolyte for lithium ion battery. Electrochim. Acta 2019, 318, 302–313. [Google Scholar] [CrossRef]
- Ma, Y.; Sun, Q.; Wang, S.; Zhou, Y.; Song, D.; Zhang, H.; Shi, X.; Zhang, L. Li salt initiated in-situ polymerized solid polymer electrolyte: New insights via in-situ electrochemical impedance spectroscopy. Chem. Eng. J. 2022, 429, 132483. [Google Scholar] [CrossRef]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, W.; Bae, W.; Jin, L.; Park, S.; Jeon, M.; Kim, W.; Jang, H. Cross-Linked Gel Polymer Electrolyte Based on Multiple Epoxy Groups Enabling Conductivity and High Performance of Li-Ion Batteries. Gels 2023, 9, 384. https://doi.org/10.3390/gels9050384
Zhang W, Bae W, Jin L, Park S, Jeon M, Kim W, Jang H. Cross-Linked Gel Polymer Electrolyte Based on Multiple Epoxy Groups Enabling Conductivity and High Performance of Li-Ion Batteries. Gels. 2023; 9(5):384. https://doi.org/10.3390/gels9050384
Chicago/Turabian StyleZhang, Wei, Wansu Bae, Lei Jin, Sungjun Park, Minhyuk Jeon, Whangi Kim, and Hohyoun Jang. 2023. "Cross-Linked Gel Polymer Electrolyte Based on Multiple Epoxy Groups Enabling Conductivity and High Performance of Li-Ion Batteries" Gels 9, no. 5: 384. https://doi.org/10.3390/gels9050384
APA StyleZhang, W., Bae, W., Jin, L., Park, S., Jeon, M., Kim, W., & Jang, H. (2023). Cross-Linked Gel Polymer Electrolyte Based on Multiple Epoxy Groups Enabling Conductivity and High Performance of Li-Ion Batteries. Gels, 9(5), 384. https://doi.org/10.3390/gels9050384








