Synthesis of Cationic Quaternized Nanolevan Derivative for Small Molecule and Nucleic Acid Delivery
Abstract
1. Introduction
2. Results and Discussion
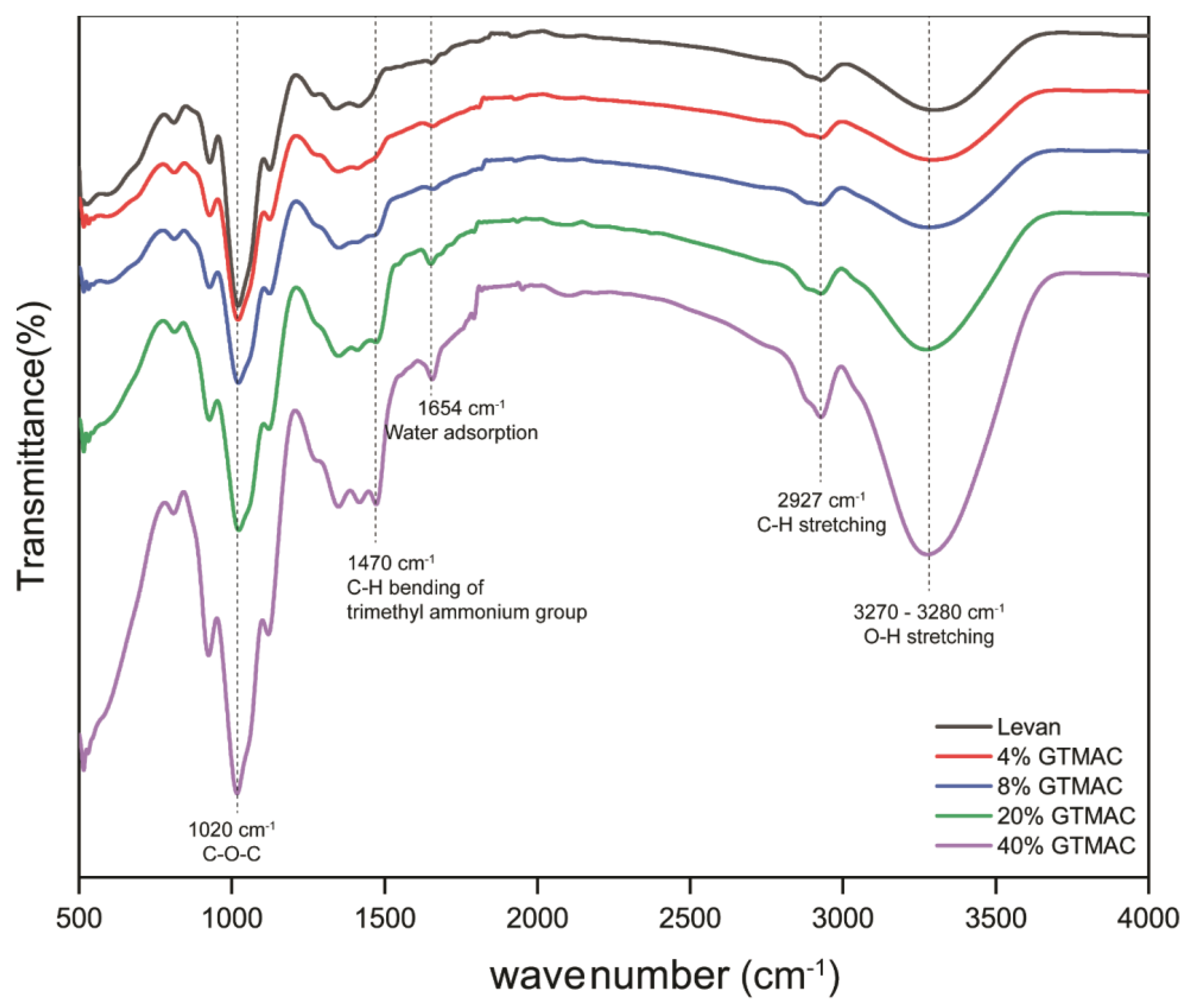
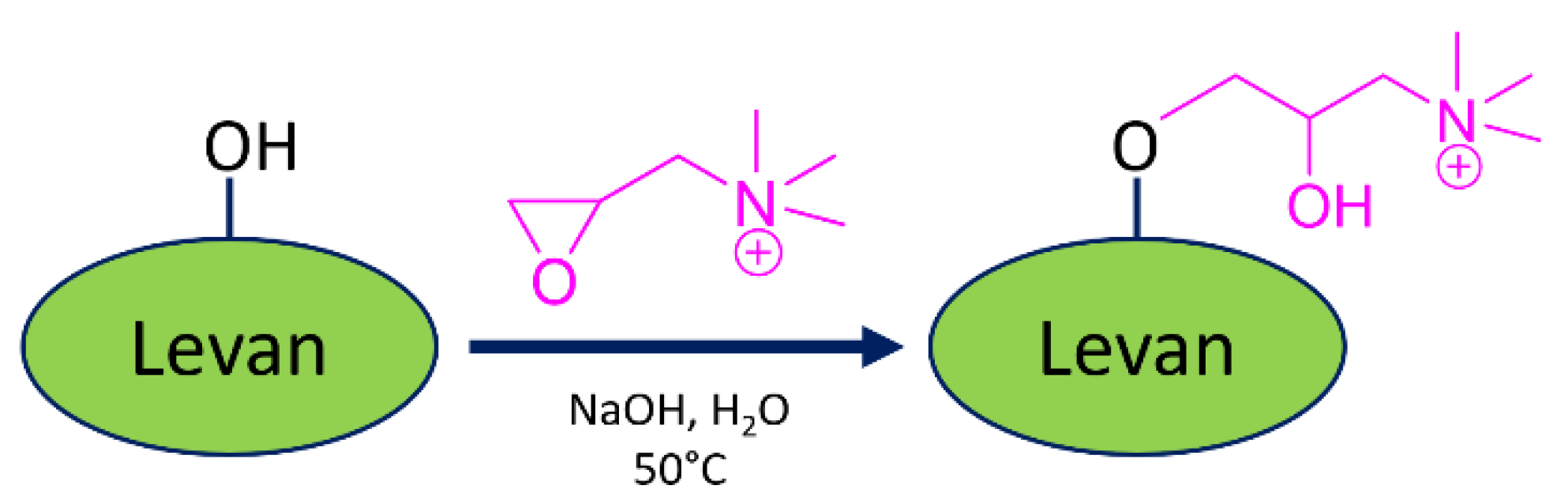
2.1. Chemical Modification of Levan and Characterization
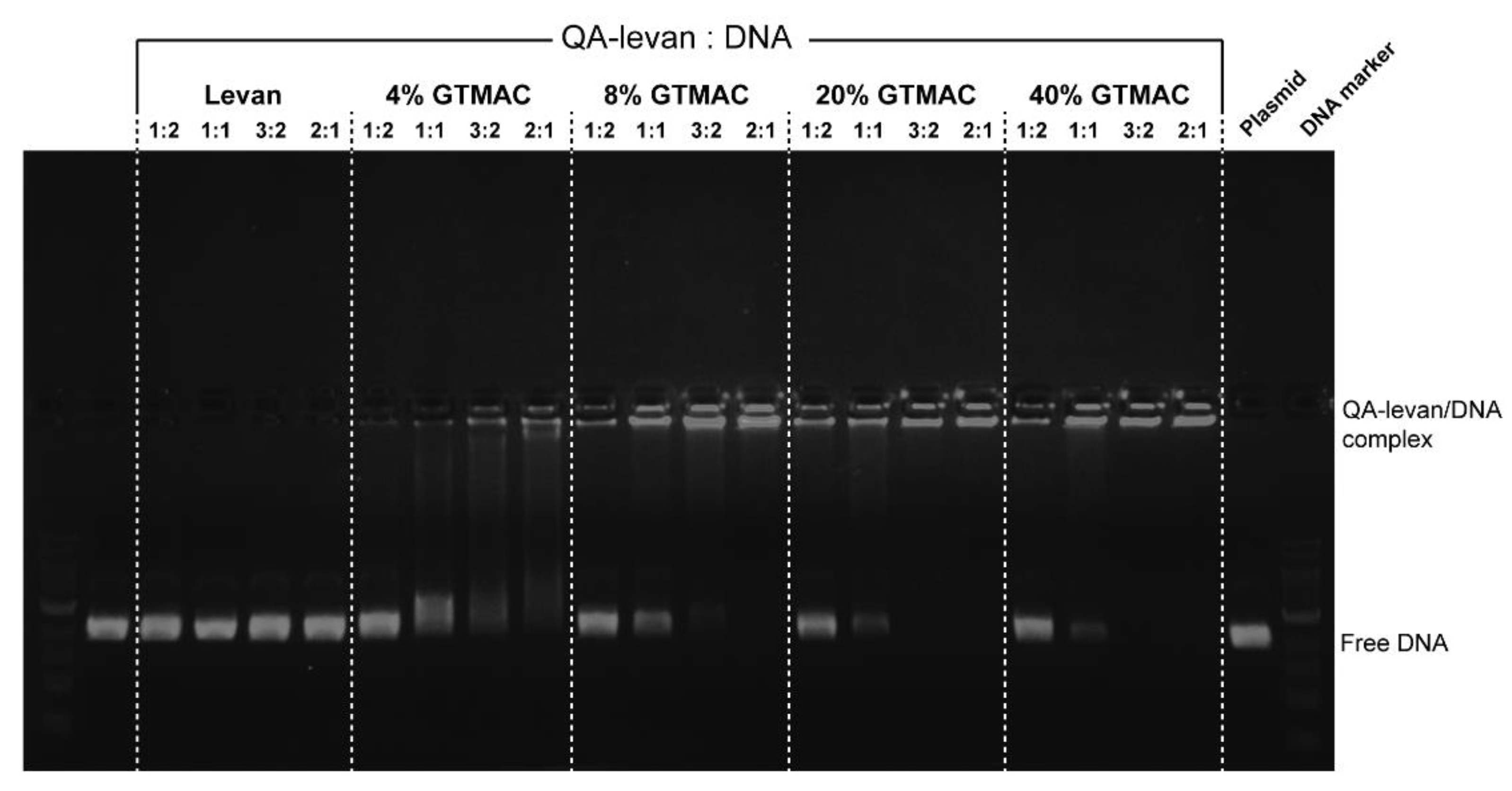
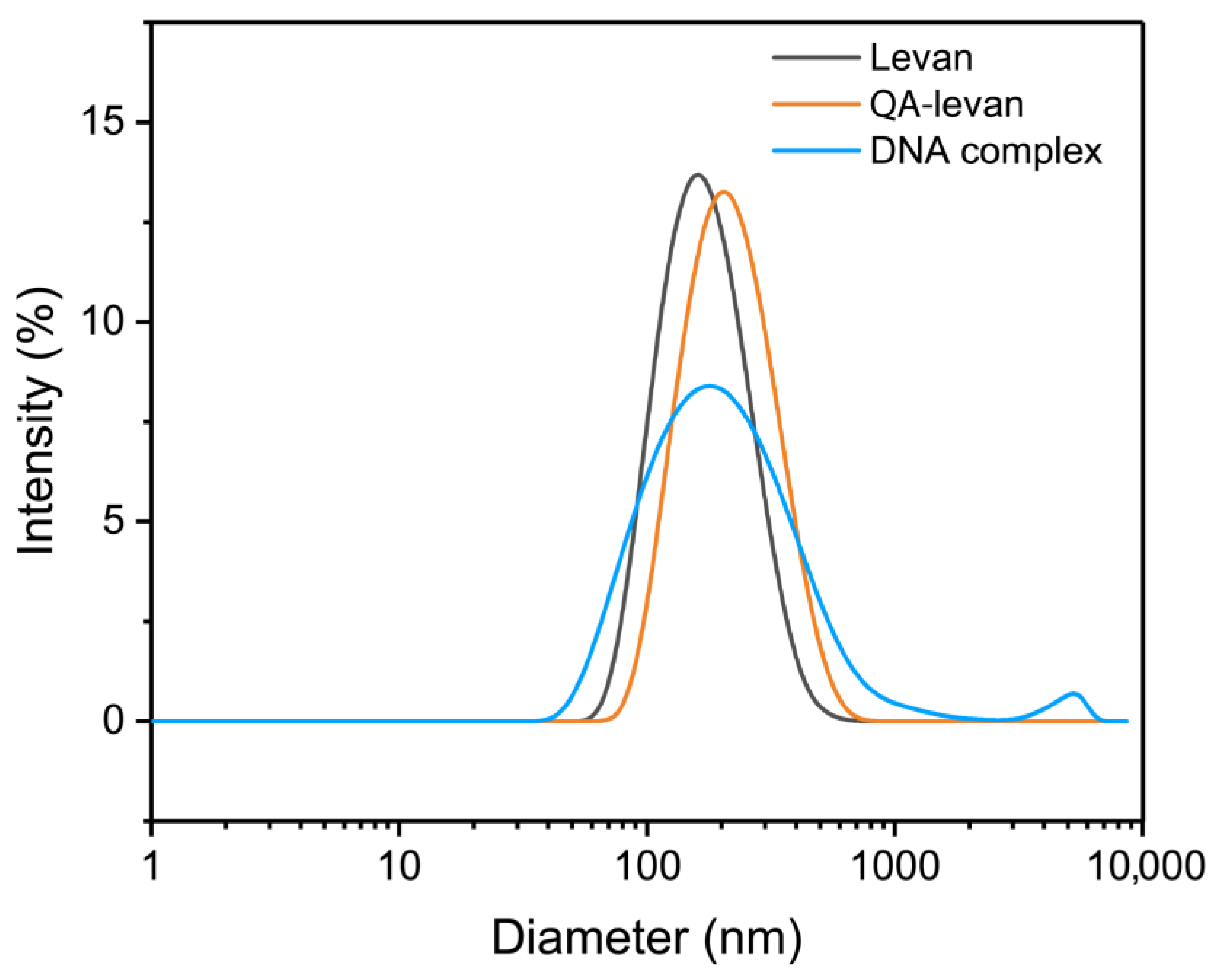
2.2. Formation of QA−Levan/DNA Polyplexes
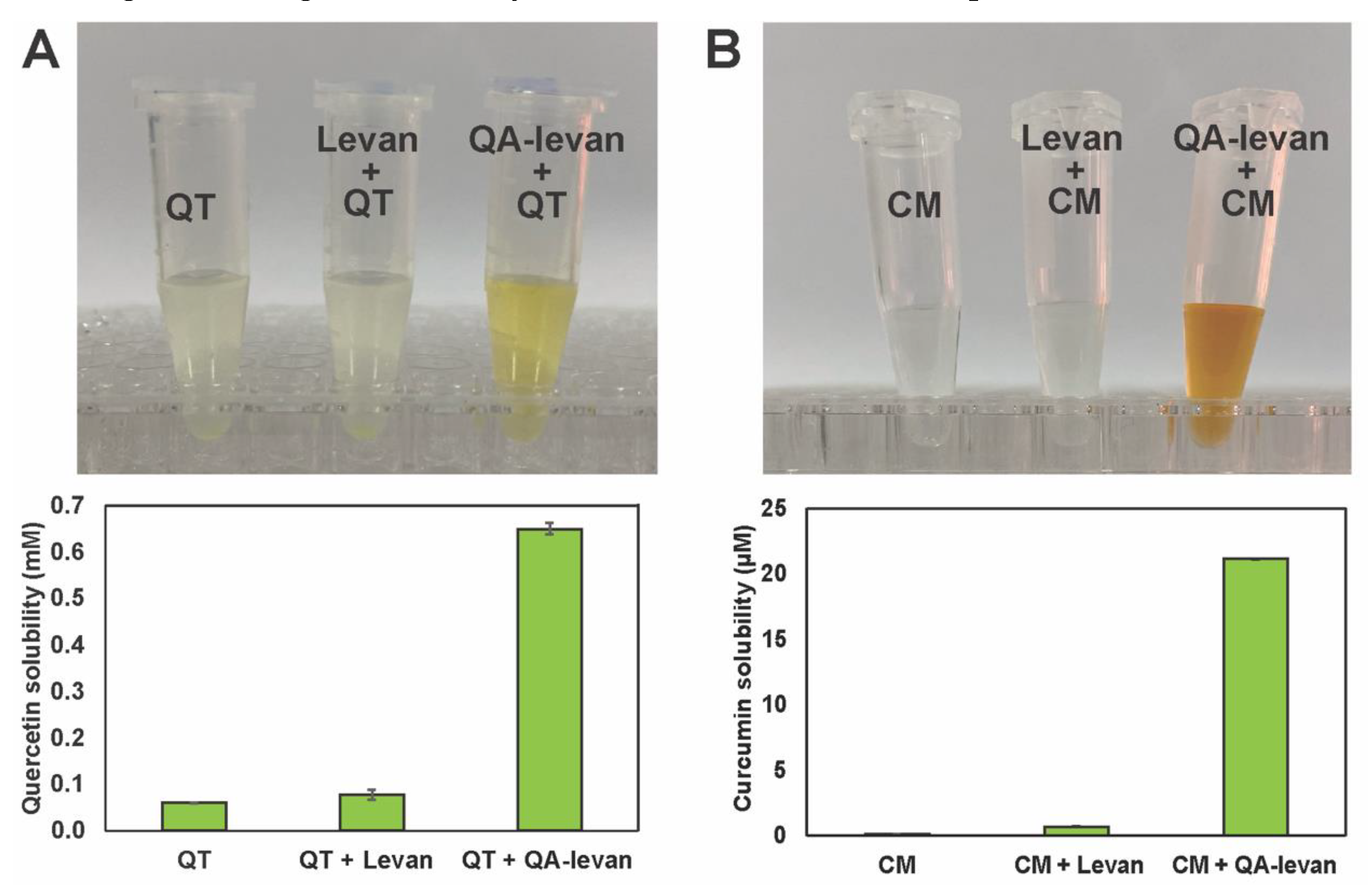
2.3. Drug Delivery Application of QA−Levan
2.4. Cytotoxicity to Human Cancer Cell Line
3. Conclusions
4. Materials and Methods
4.1. Chemicals
4.2. Preparation of Levan
4.3. Preparation of Quaternary Ammonium Levan (QA-Levan)
4.4. Characterization
4.4.1. Fourier Transform Infrared (FT-IR) Analysis
4.4.2. CHN Analysis
4.4.3. 1H-NMR
4.4.4. Zeta Sizer
4.5. Formation of QA-Levan/DNA Polyplexes
4.6. Encapsulation of Small Molecules
4.7. Cytotoxicity
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Plucinski, A.; Lyu, Z.; Schmidt, B.V.K.J. Polysaccharide nanoparticles: From fabrication to applications. J. Mater. Chem. B 2021, 9, 7030–7062. [Google Scholar] [CrossRef] [PubMed]
- Heo, R.; You, D.G.; Um, W.; Choi, K.Y.; Jeon, S.; Park, J.-S.; Choi, Y.; Kwon, S.; Kim, K.; Kwon, I.C.; et al. Dextran sulfate nanoparticles as a theranostic nanomedicine for rheumatoid arthritis. Biomaterials 2017, 131, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Mizrahy, S.; Peer, D. Polysaccharides as building blocks for nanotherapeutics. Chem. Soc. Rev. 2012, 41, 2623–2640. [Google Scholar] [CrossRef]
- Nicolaus, B.; Kambourova, M.; Oner, E.T. Exopolysaccharides from extremophiles: From fundamentals to biotechnology. Environ. Technol. 2010, 31, 1145–1158. [Google Scholar] [CrossRef] [PubMed]
- Miramontes-Corona, C.; Escalante, A.; Delgado, E.; Corona-González, R.I.; Vázquez-Torres, H.; Toriz, G. Hydrophobic agave fructans for sustained drug delivery to the human colon. React. Funct. Polym. 2020, 146, 104396. [Google Scholar] [CrossRef]
- Phengnoi, P.; Charoenwongpaiboon, T.; Wangpaiboon, K.; Klaewkla, M.; Nakapong, S.; Visessanguan, W.; Ito, K.; Pichyangkura, R.; Kuttiyawong, K. Levansucrase from Bacillus amyloliquefaciens KK9 and Its Y237S Variant Producing the High Bioactive Levan-Type Fructooligosaccharides. Biomolecules 2020, 10, 692. [Google Scholar] [CrossRef]
- Kang, H.K.; Seo, M.Y.; Seo, E.S.; Kim, D.; Chung, S.Y.; Kimura, A.; Day, D.F.; Robyt, J.F. Cloning and expression of levansucrase from Leuconostoc mesenteroides B-512 FMC in Escherichia coli. Biochim. Biophys. Acta (BBA)—Gene Struct. Expr. 2005, 1727, 5–15. [Google Scholar] [CrossRef]
- Silbir, S.; Dagbagli, S.; Yegin, S.; Baysal, T.; Goksungur, Y. Levan production by Zymomonas mobilis in batch and continuous fermentation systems. Carbohydr. Polym. 2014, 99, 454–461. [Google Scholar] [CrossRef]
- Charoenwongpaiboon, T.; Wangpaiboon, K.; Septham, P.; Jiamvoraphong, N.; Issaragrisil, S.; Pichyangkura, R.; Lorthongpanich, C. Production and bioactivities of nanoparticulated and ultrasonic-degraded levan generated by Erwinia tasmaniensis levansucrase in human osteosarcoma cells. Int. J. Biol. Macromol. 2022, 221, 1121–1129. [Google Scholar] [CrossRef]
- Charoenwongpaiboon, T.; Wangpaiboon, K.; Panpetch, P.; Field, R.A.; Barclay, J.E.; Pichyangkura, R.; Kuttiyawong, K. Temperature-dependent inulin nanoparticles synthesized by Lactobacillus reuteri 121 inulosucrase and complex formation with flavonoids. Carbohydr. Polym. 2019, 223, 115044. [Google Scholar] [CrossRef]
- Nakapong, S.; Pichyangkura, R.; Ito, K.; Iizuka, M.; Pongsawasdi, P. High expression level of levansucrase from Bacillus licheniformis RN-01 and synthesis of levan nanoparticles. Int. J. Biol. Macromol. 2013, 54, 30–36. [Google Scholar] [CrossRef]
- De Siqueira, E.C.; Rebouças, J.d.S.; Pinheiro, I.O.; Formiga, F.R. Levan-based nanostructured systems: An overview. Int. J. Pharm. 2020, 580, 119242. [Google Scholar] [CrossRef]
- Cinan, E.; Cesur, S.; Erginer Haskoylu, M.; Gunduz, O.; Toksoy Oner, E. Resveratrol-Loaded Levan Nanoparticles Produced by Electrohydrodynamic Atomization Technique. Nanomaterials 2021, 11, 2582. [Google Scholar] [CrossRef]
- Kim, S.-J.; Bae, P.K.; Chung, B.H. Self-assembled levan nanoparticles for targeted breast cancer imaging. Chem. Commun. 2015, 51, 107–110. [Google Scholar] [CrossRef]
- Koşarsoy Ağçeli, G.; Cihangir, N. Nano-sized biopolymer levan: Its antimicrobial, anti-biofilm and anti-cancer effects. Carbohydr. Res. 2020, 494, 108068. [Google Scholar] [CrossRef]
- Maciel, J.C.; Andrad, P.L.; Neri, D.F.M.; Carvalho, L.B.; Cardoso, C.A.; Calazans, G.M.T.; Albino Aguiar, J.; Silva, M.P.C. Preparation and characterization of magnetic levan particles as matrix for trypsin immobilization. J. Magn. Magn. Mater. 2012, 324, 1312–1316. [Google Scholar] [CrossRef]
- Costa, R.R.; Neto, A.I.; Calgeris, I.; Correia, C.R.; Pinho, A.C.M.; Fonseca, J.; Öner, E.T.; Mano, J.F. Adhesive nanostructured multilayer films using a bacterial exopolysaccharide for biomedical applications. J. Mater. Chem. B 2013, 1, 2367–2374. [Google Scholar] [CrossRef]
- Erginer, M.; Akcay, A.; Coskunkan, B.; Morova, T.; Rende, D.; Bucak, S.; Baysal, N.; Ozisik, R.; Eroglu, M.S.; Agirbasli, M.; et al. Sulfated levan from Halomonas smyrnensis as a bioactive, heparin-mimetic glycan for cardiac tissue engineering applications. Carbohydr. Polym. 2016, 149, 289–296. [Google Scholar] [CrossRef]
- Sung, Y.K.; Kim, S.W. Recent advances in polymeric drug delivery systems. Biomater. Res. 2020, 24, 12. [Google Scholar] [CrossRef]
- Polsinelli, I.; Caliandro, R.; Salomone-Stagni, M.; Demitri, N.; Rejzek, M.; Field, R.A.; Benini, S. Comparison of the Levansucrase from the epiphyte Erwinia tasmaniensis vs. its homologue from the phytopathogen Erwinia amylovora. Int. J. Biol. Macromol. 2019, 127, 496–501. [Google Scholar] [CrossRef]
- Charoenwongpaiboon, T.; Supraditaporn, K.; Klaimon, P.; Wangpaiboon, K.; Pichyangkura, R.; Issaragrisil, S.; Lorthongpanich, C. Effect of alternan versus chitosan on the biological properties of human mesenchymal stem cells. RSC Adv. 2019, 9, 4370–4379. [Google Scholar] [CrossRef] [PubMed]
- Selvi, S.S.; Hasköylü, M.E.; Genç, S.; Toksoy Öner, E. Synthesis and characterization of levan hydrogels and their use for resveratrol release. J. Bioact. Compat. Polym. 2021, 36, 464–480. [Google Scholar] [CrossRef]
- Demirci, T.; Hasköylü, M.E.; Eroğlu, M.S.; Hemberger, J.; Toksoy Öner, E. Levan-based hydrogels for controlled release of Amphotericin B for dermal local antifungal therapy of Candidiasis. Eur. J. Pharm. Sci. 2020, 145, 105255. [Google Scholar] [CrossRef] [PubMed]
- Moraes, F.C.; Antunes, J.C.; Forero Ramirez, L.M.; Aprile, P.; Franck, G.; Chauvierre, C.; Chaubet, F.; Letourneur, D. Synthesis of cationic quaternized pullulan derivatives for miRNA delivery. Int. J. Pharm. 2020, 577, 119041. [Google Scholar] [CrossRef]
- Sarker, N.C.; Ray, P.; Pfau, C.; Kalavacharla, V.; Hossain, K.; Quadir, M. Development of Functional Nanomaterials from Wheat Bran Derived Arabinoxylan for Nucleic Acid Delivery. J. Agric. Food Chem. 2020, 68, 4367–4373. [Google Scholar] [CrossRef]
- Chen, K.-Y.; Zeng, S.-Y. Preparation and Characterization of Quaternized Chitosan Coated Alginate Microspheres for Blue Dextran Delivery. Polymers 2017, 9, 210. [Google Scholar] [CrossRef]
- Amar-Lewis, E.; Azagury, A.; Chintakunta, R.; Goldbart, R.; Traitel, T.; Prestwood, J.; Landesman-Milo, D.; Peer, D.; Kost, J. Quaternized starch-based carrier for siRNA delivery: From cellular uptake to gene silencing. J. Control Release 2014, 185, 109–120. [Google Scholar] [CrossRef]
- Xiao, B.; Wan, Y.; Wang, X.; Zha, Q.; Liu, H.; Qiu, Z.; Zhang, S. Synthesis and characterization of N-(2-hydroxy)propyl-3-trimethyl ammonium chitosan chloride for potential application in gene delivery. Colloids Surf. B Biointerfaces 2012, 91, 168–174. [Google Scholar] [CrossRef]
- Thomas, J.J.; Rekha, M.R.; Sharma, C.P. Dextran–glycidyltrimethylammonium chloride conjugate/DNA nanoplex: A potential non-viral and haemocompatible gene delivery system. Int. J. Pharm. 2010, 389, 195–206. [Google Scholar] [CrossRef]
- Suksiri, P.; Sansanaphongpricha, K.; Muangsin, N.; Krusong, K. Development of positively-charged cycloamylose, CAQ as efficient nanodelivery system for siRNA. Biochem. Eng. J. 2023, 191, 108767. [Google Scholar] [CrossRef]
- Sardo, C.; Farra, R.; Licciardi, M.; Dapas, B.; Scialabba, C.; Giammona, G.; Grassi, M.; Grassi, G.; Cavallaro, G. Development of a simple, biocompatible and cost-effective Inulin-Diethylenetriamine based siRNA delivery system. Eur. J. Pharm. Sci. 2015, 75, 60–71. [Google Scholar] [CrossRef]
- Lesjak, M.; Beara, I.; Simin, N.; Pintać, D.; Majkić, T.; Bekvalac, K.; Orčić, D.; Mimica-Dukić, N. Antioxidant and anti-inflammatory activities of quercetin and its derivatives. J. Funct. Foods 2018, 40, 68–75. [Google Scholar] [CrossRef]
- Charoenwongpaiboona, T.; Laowtammathronb, C.; Lorthongpanichb, C. Therapeutic opportunities for cancers presented by natural and synthetic compounds targeting the Hippo signaling pathway. ScienceAsia 2021, 47, 665–672. [Google Scholar] [CrossRef]
- Gupta, S.C.; Patchva, S.; Koh, W.; Aggarwal, B.B. Discovery of curcumin, a component of golden spice, and its miraculous biological activities. Clin. Exp. Pharmacol. Physiol. 2012, 39, 283–299. [Google Scholar] [CrossRef]
- Gunathilake, T.M.S.U.; Ching, Y.C.; Uyama, H.; Hai, N.D.; Chuah, C.H. Enhanced curcumin loaded nanocellulose: A possible inhalable nanotherapeutic to treat COVID-19. Cellulose 2022, 29, 1821–1840. [Google Scholar] [CrossRef]
- Luckanagul, J.A.; Pitakchatwong, C.; Ratnatilaka Na Bhuket, P.; Muangnoi, C.; Rojsitthisak, P.; Chirachanchai, S.; Wang, Q.; Rojsitthisak, P. Chitosan-based polymer hybrids for thermo-responsive nanogel delivery of curcumin. Carbohydr. Polym. 2018, 181, 1119–1127. [Google Scholar] [CrossRef]
- Charoenwongpaiboon, T.; Wangpaiboon, K.; Pichyangkura, R.; Nepogodiev, S.A.; Wonganan, P.; Mahalapbutr, P.; Field, R.A. Characterization of a nanoparticulate exopolysaccharide from Leuconostoc holzapfelii KM01 and its potential application in drug encapsulation. Int. J. Biol. Macromol. 2021, 187, 690–698. [Google Scholar] [CrossRef]
- He, C.; Hu, Y.; Yin, L.; Tang, C.; Yin, C. Effects of particle size and surface charge on cellular uptake and biodistribution of polymeric nanoparticles. Biomaterials 2010, 31, 3657–3666. [Google Scholar] [CrossRef]
- Samal, S.K.; Dash, M.; Van Vlierberghe, S.; Kaplan, D.L.; Chiellini, E.; van Blitterswijk, C.; Moroni, L.; Dubruel, P. Cationic polymers and their therapeutic potential. Chem. Soc. Rev. 2012, 41, 7147–7194. [Google Scholar] [CrossRef]
- Bakrania, A.K.; Variya, B.C.; Patel, S.S. Role of β-Interferon Inducer (DEAE-Dextran) in Tumorigenesis by VEGF and NOTCH1 Inhibition along with Apoptosis Induction. Front. Pharmacol. 2017, 8, 930. [Google Scholar] [CrossRef]
- Zhan, T.; Bai, Q.; Zhao, Z. Antiproliferative effects of levan polysaccharide against colorectal cancer cells mediated through oxidative stress-stimulated HOTAIR/Akt signaling pathway: In vitro. Arab. J. Chem. 2021, 14, 103389. [Google Scholar] [CrossRef]


| Sample | %N | %C | %H | N/C |
|---|---|---|---|---|
| Levan | ND | 42.29 | 6.92 | ND |
| 4%GTMAC modified levan | 0.70 | 39.59 | 6.46 | 0.02 |
| 8%GTMAC modified levan | 1.24 | 40.32 | 6.65 | 0.03 |
| 20%GTMAC modified levan | 2.86 | 41.03 | 7.34 | 0.07 |
| 40%GTMAC modified levan | 2.45 | 36.72 | 6.92 | 0.07 |
| Sample | Diameter (nm) | ξ-Potential Mean ± SEM (mV) |
|---|---|---|
| Levan | 157.9 ± 0.2 | −7.05 ± 0.19 |
| QA-levan | 193.8 ± 1.5 | 53.7 ± 0.53 |
| QA-levan/DNA complex | 170.7 ± 1.7 | 47.9 ± 0.70 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Charoenwongphaibun, C.; Lorthongpanich, C.; Septham, P.; Wangpaiboon, K.; Panpetch, P.; Pichyangkura, R.; Charoenwongpaiboon, T.; Kuttiyawong, K. Synthesis of Cationic Quaternized Nanolevan Derivative for Small Molecule and Nucleic Acid Delivery. Gels 2023, 9, 188. https://doi.org/10.3390/gels9030188
Charoenwongphaibun C, Lorthongpanich C, Septham P, Wangpaiboon K, Panpetch P, Pichyangkura R, Charoenwongpaiboon T, Kuttiyawong K. Synthesis of Cationic Quaternized Nanolevan Derivative for Small Molecule and Nucleic Acid Delivery. Gels. 2023; 9(3):188. https://doi.org/10.3390/gels9030188
Chicago/Turabian StyleCharoenwongphaibun, Chonnipha, Chanchao Lorthongpanich, Prapasri Septham, Karan Wangpaiboon, Pawinee Panpetch, Rath Pichyangkura, Thanapon Charoenwongpaiboon, and Kamontip Kuttiyawong. 2023. "Synthesis of Cationic Quaternized Nanolevan Derivative for Small Molecule and Nucleic Acid Delivery" Gels 9, no. 3: 188. https://doi.org/10.3390/gels9030188
APA StyleCharoenwongphaibun, C., Lorthongpanich, C., Septham, P., Wangpaiboon, K., Panpetch, P., Pichyangkura, R., Charoenwongpaiboon, T., & Kuttiyawong, K. (2023). Synthesis of Cationic Quaternized Nanolevan Derivative for Small Molecule and Nucleic Acid Delivery. Gels, 9(3), 188. https://doi.org/10.3390/gels9030188








