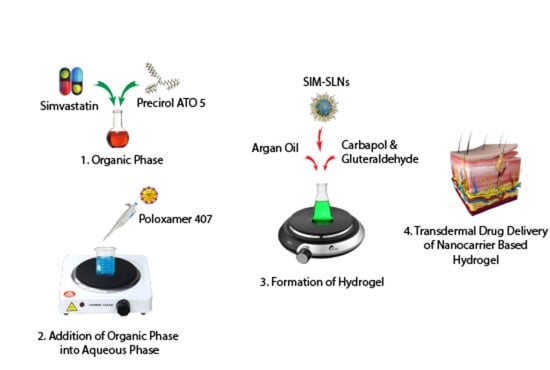
Hydrogel Containing Solid Lipid Nanoparticles Loaded with Argan Oil and Simvastatin: Preparation, In Vitro and Ex Vivo Assessment
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
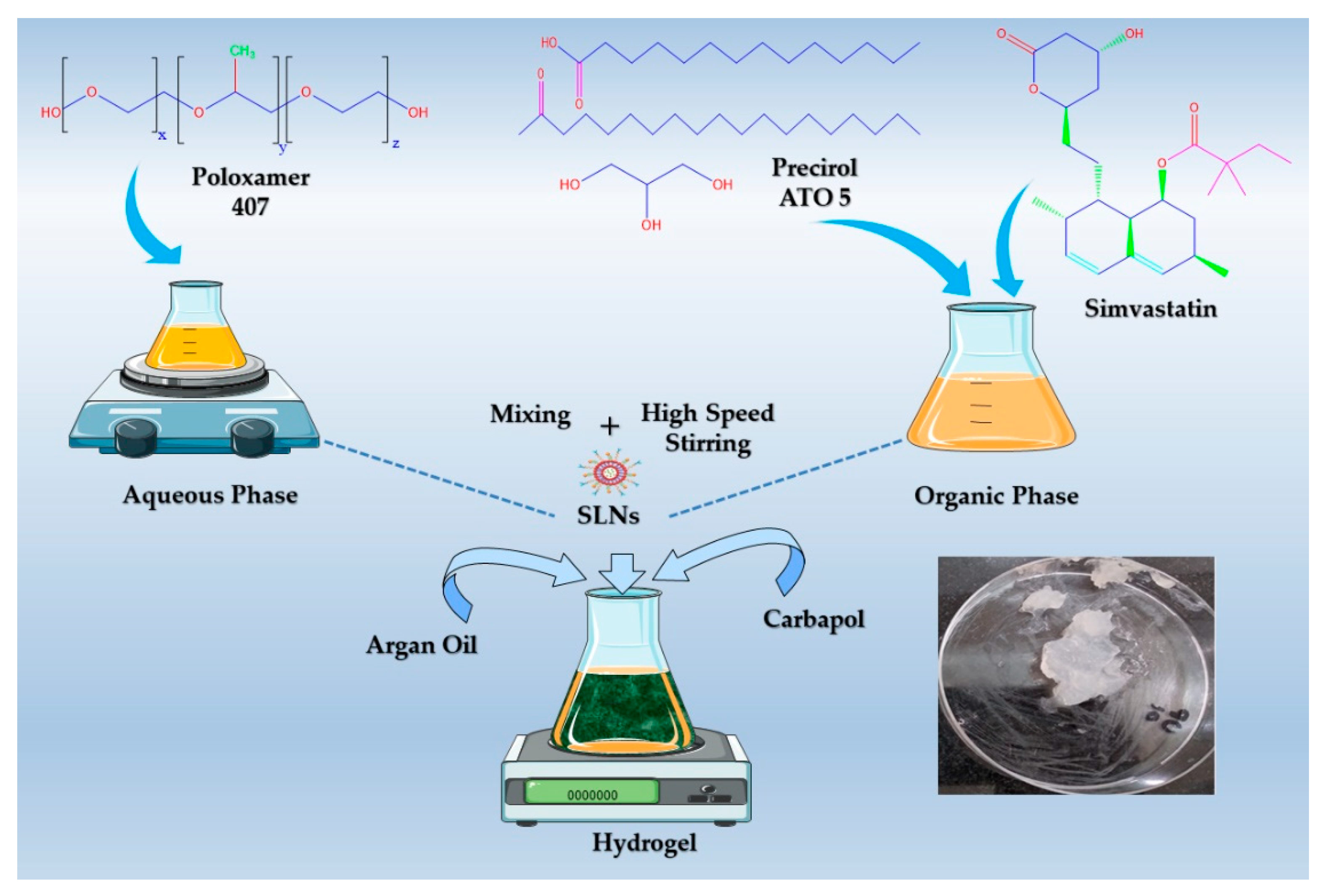
Preparation of SIM-SLNs
2.3. Physicochemical Characterization of Nanoparticles
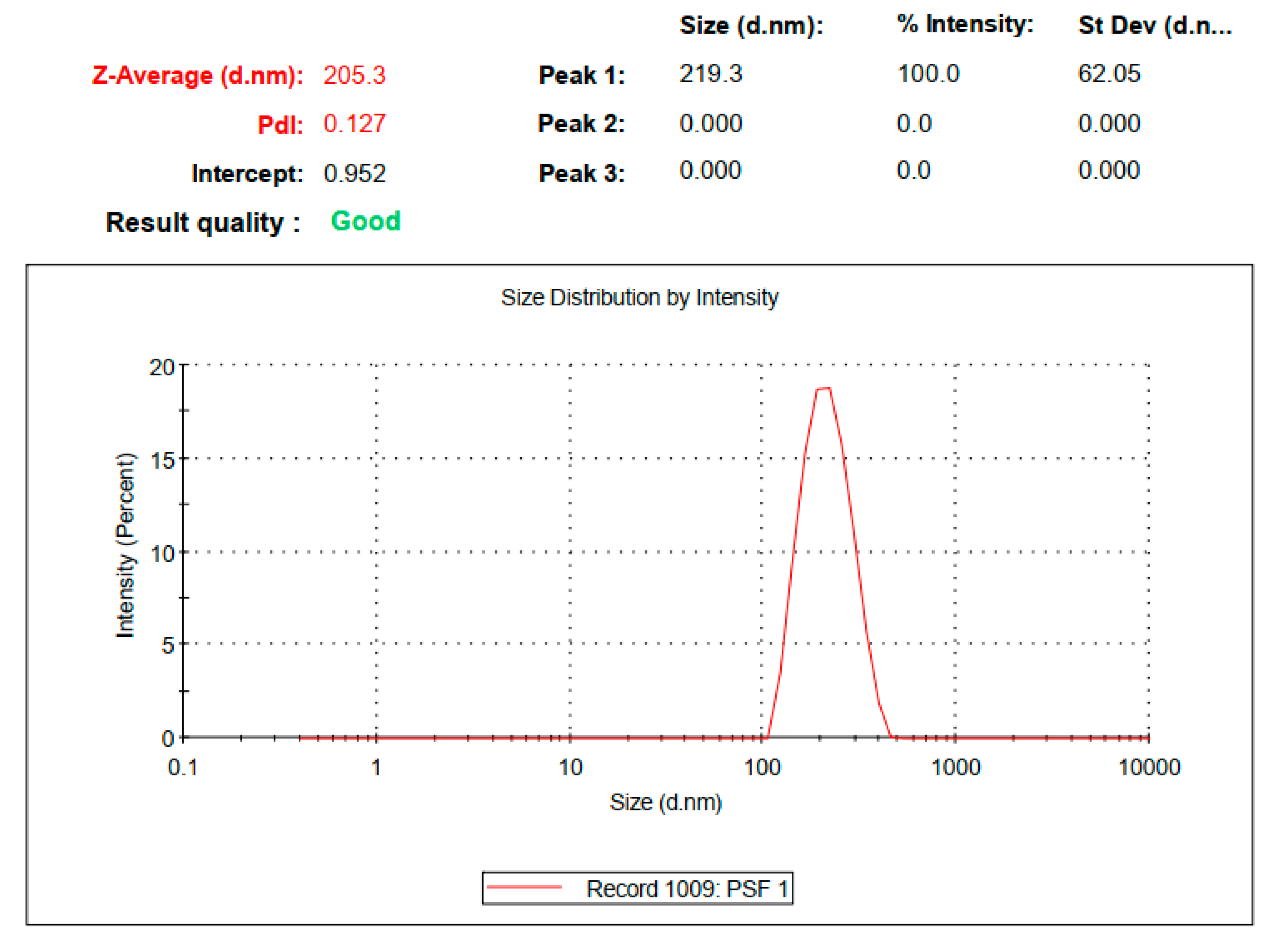
2.3.1. Particle Size, Polydispersity Index, Zeta Potential and Entrapment Efficiency
2.3.2. Scanning Electron Microscopy (SEM)
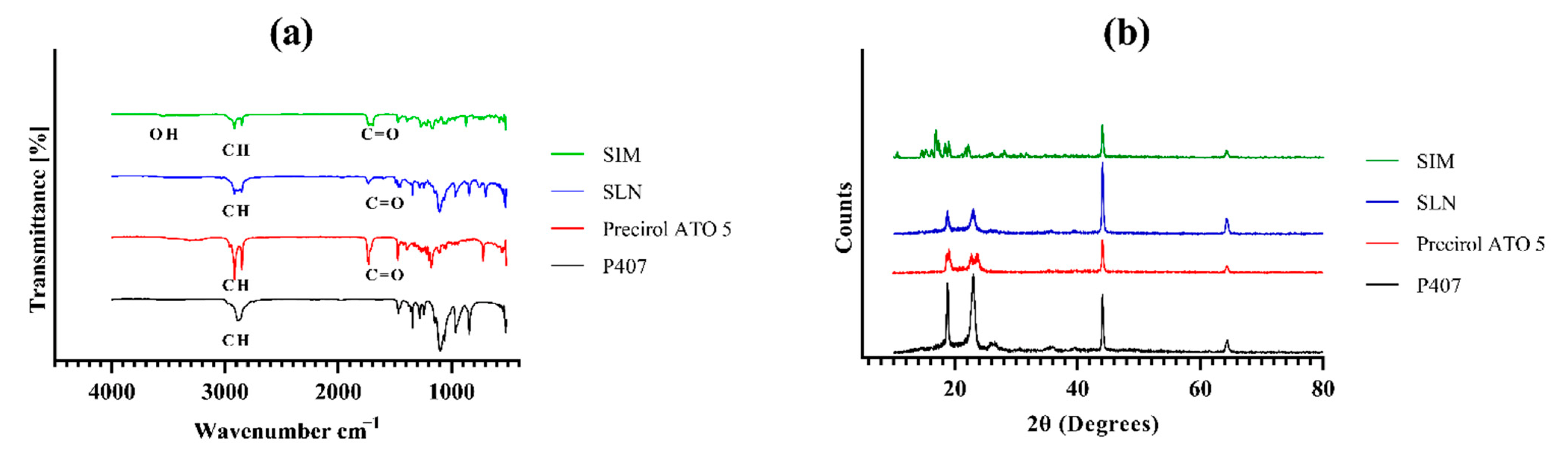
2.3.3. Fourier Transform Infrared Spectroscopy (FTIR)
2.3.4. Powder X-ray Diffractometry (PXRD)
2.4. Formulation of SIM-SLNs Loaded Transdermal Hydrogel
2.5. Characterization of Transdermal Hydrogel
2.5.1. Physical Appearance and pH Measurement
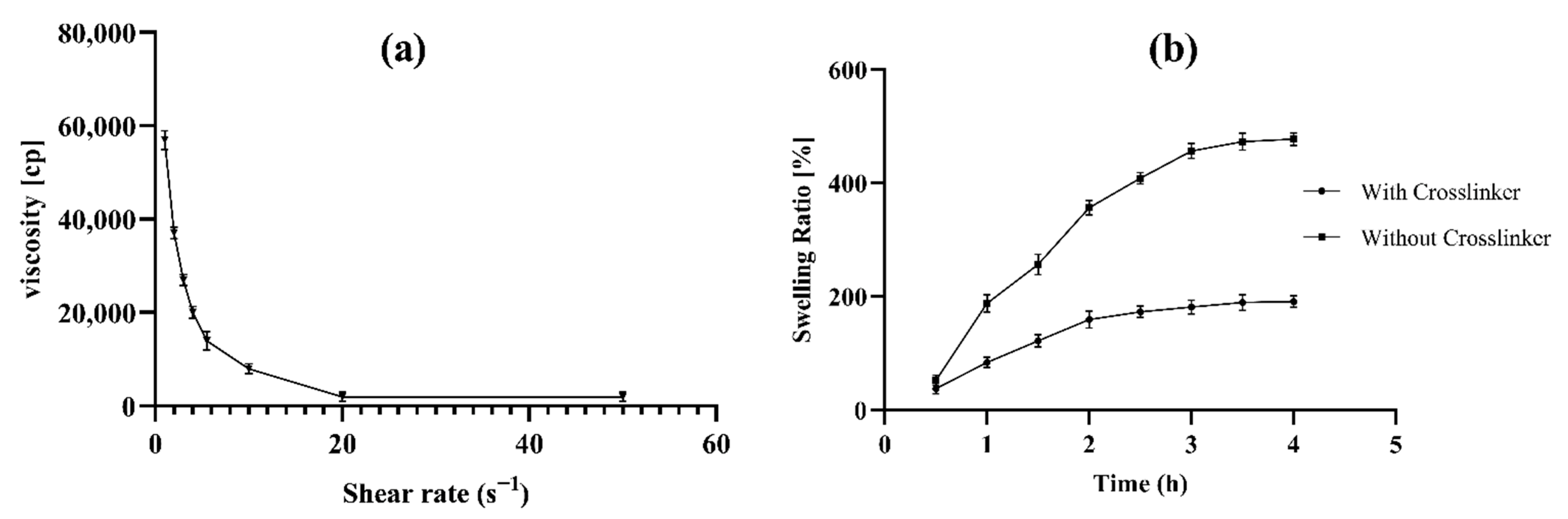
2.5.2. Rheological Study and Spreadability of Transdermal Hydrogel
2.5.3. Swelling index, Bio-adhesion and Extrudability
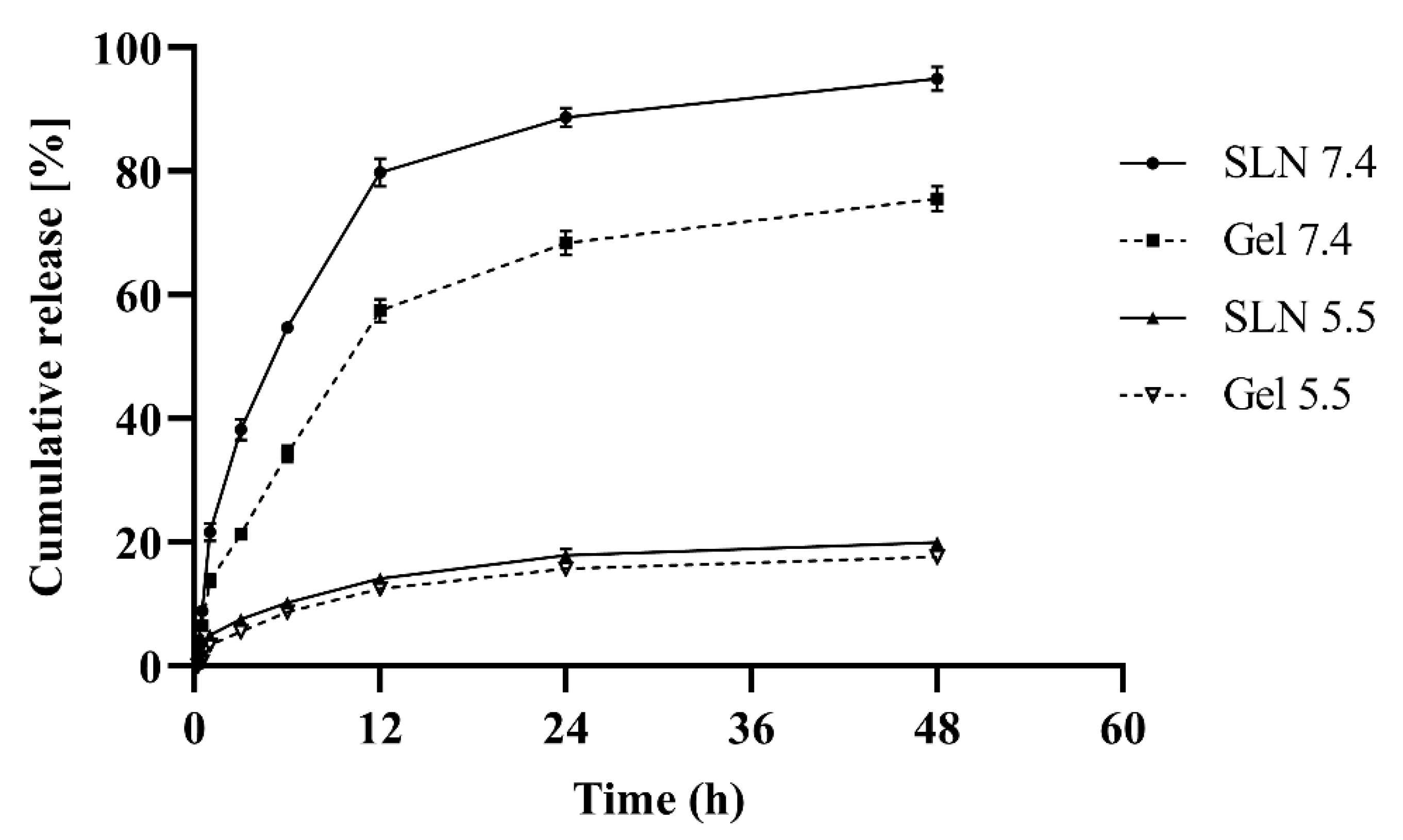
2.6. In Vitro Drug Release Studies
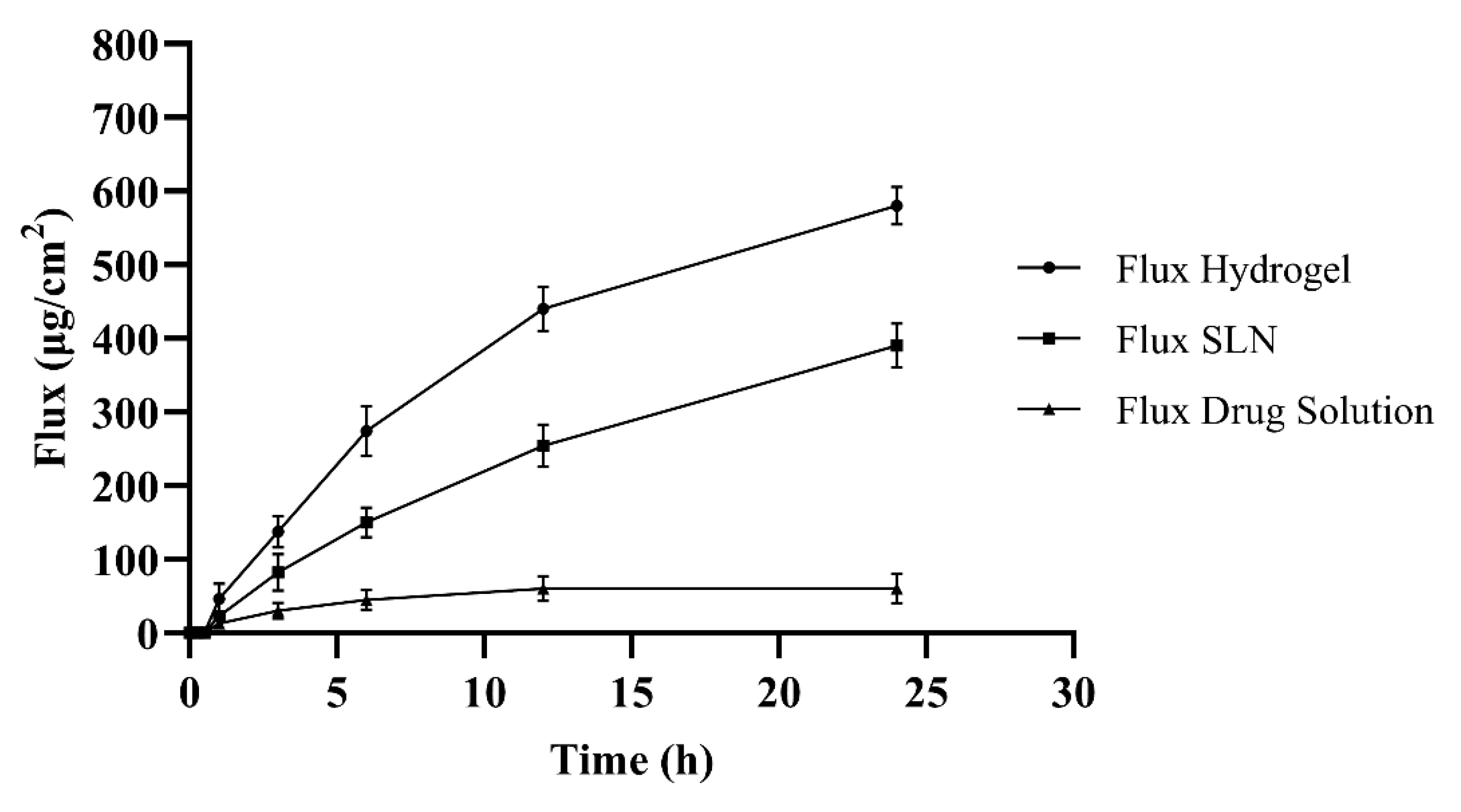
2.7. Permeation Studies
2.8. Ex Vivo Penetration Analysis through Fluorescent Microscope
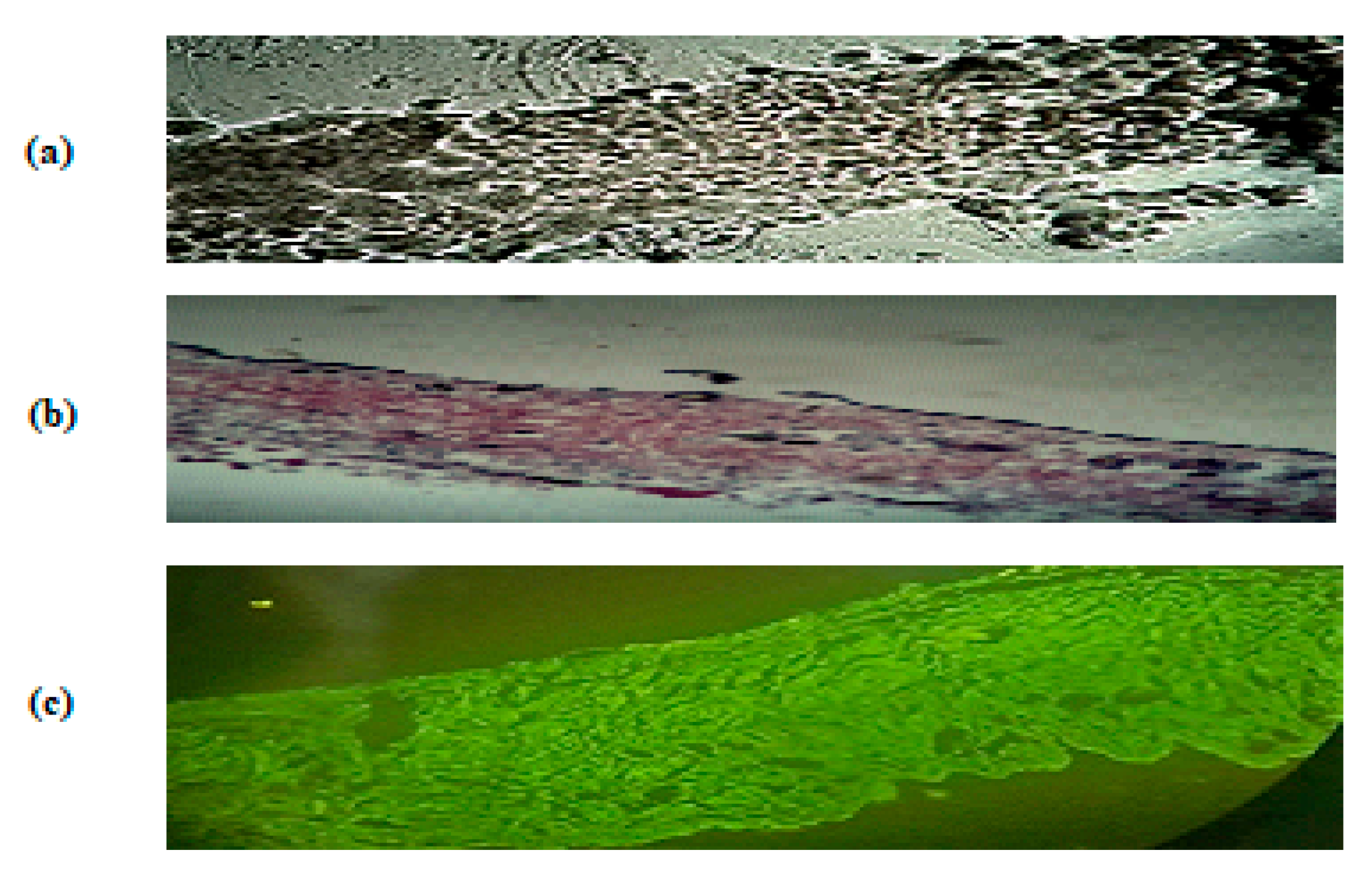
2.9. Evaluation of Skin Structure after Treatment with Hydrogel
2.10. Stability Studies
3. Results and Discussion
3.1. Preparation and Physicochemical Properties of SIM-SLNs
3.2. Particle Morphology
3.3. FTIR Analysis
3.4. Degree of Crystallinity of SIM-SLNs
3.5. Physicochemical Evaluation of Transdermal Hydrogel
3.5.1. Physical Appearance and pH of Formulation
3.5.2. Spreadability and Rheological Behaviour of Transdermal Hydrogel
3.5.3. Swelling index, Bioadhesion and Extrudability
3.6. In-Vitro Release
3.7. Skin Permeability
3.8. Ex Vivo Penetration Analysis through Fluorescent Microscope
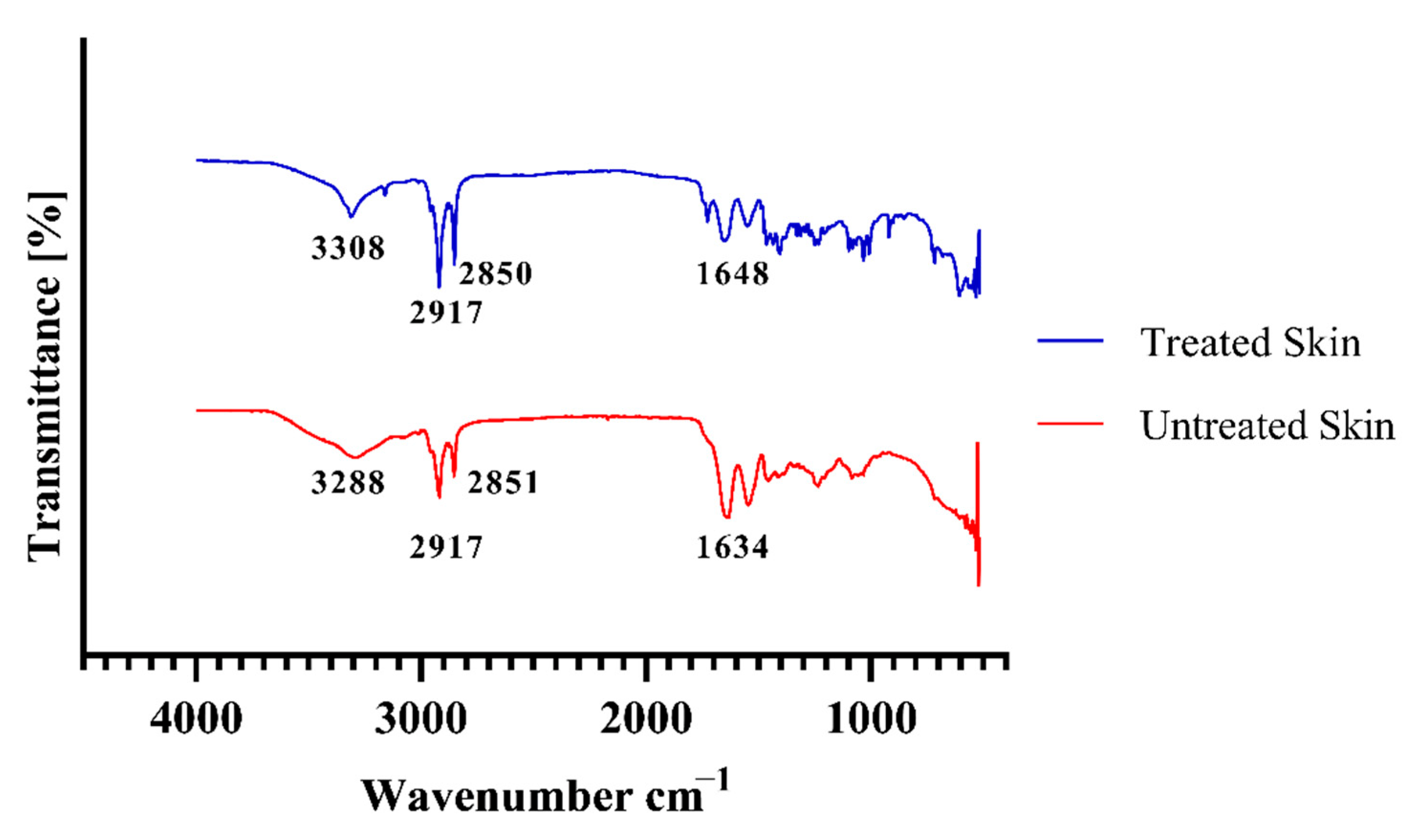
3.9. Evaluation of Skin Structure after Treatment with Hydrogel
3.10. Stability Studies of Hydrogel
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Ethical Approval
References
- Adi, D.; Abuzhalihan, J.; Wang, Y.-H.; Baituola, G.; Wu, Y.; Xie, X.; Fu, Z.Y.; Yang, Y.N.; Ma, X.; Li, X.M.; et al. IDOL gene variant is associated with hyperlipidemia in Han population in Xinjiang, China. Sci. Rep. 2020, 10, 14280. [Google Scholar] [CrossRef] [PubMed]
- Asbahani, A.; El Miladi, K.; Badri, W.; Sala, M.; Addi, E.H.A.; Casabianca, H.; Mousadik, A.; El Hartmann, D.; Jilale, A.; Renaud, F.N.R.; et al. Essential oils: From extraction to encapsulation. Int. J. Pharm. 2015, 483, 220–243. [Google Scholar] [CrossRef] [PubMed]
- Badreddine, A.; Zarrouk, A.; Meddeb, W.; Nury, T.; Rezig, L.; Debbabi, M.; Bessam, F.Z.; Brahmi, F.; Vejux, A.; Mejri, M.; et al. Chapter 10—Antioxidant and neuroprotective properties of Mediterranean oils: Argan oil, olive oil, and milk thistle seed oil. In Oxidative Stress and Dietary Antioxidants in Neurological Diseases; Martin, C.R., Preedy, V.R., Eds.; Academic Press: London, UK, 2020; pp. 143–154. Available online: https://www.sciencedirect.com/science/article/pii/B9780128177808099941 (accessed on 22 February 2022). [CrossRef]
- Tan, W.Y.T.; Young, B.E.; Lye, D.C.; Chew, D.E.K.; Dalan, R. Statin use is associated with lower disease severity in COVID-19 infection. Sci. Rep. 2020, 10, 17458. [Google Scholar] [CrossRef]
- Dolatabadi, J.E.N.; Hamishehkar, H.; Eskandani, M.; Valizadeh, H. Formulation, characterization and cytotoxicity studies of alendronate sodium-loaded solid lipid nanoparticles. Colloids Surf. B Biointerfaces 2014, 117, 21–28. [Google Scholar] [CrossRef]
- Nutan, B.; Chandel, A.K.S.; Biswas, A.; Kumar, A.; Yadav, A.; Maiti, P.; Jewrajka, S.K. Gold Nanoparticle Promoted Formation and Biological Properties of Injectable Hydrogels. Biomacromolecules 2020, 21, 3782–3794. [Google Scholar] [CrossRef]
- Yang, Z.; Peng, H.; Wang, W.; Liu, T. Crystallization behavior of poly(ε-caprolactone)/layered double hydroxide nanocomposites. J. Appl. Polym. Sci. 2010, 116, 2658–2667. [Google Scholar] [CrossRef]
- Ahmed, T.A.; Badr-Eldin, S.M.; Ahmed, O.A.A.; Aldawsari, H. Intranasal optimized solid lipid nanoparticles loaded in situ gel for enhancing trans-mucosal delivery of simvastatin. J. Drug Deliv. Sci. Technol. 2018, 48, 499–508. [Google Scholar] [CrossRef]
- Thoniyot, P.; Tan, M.J.; Karim, A.A.; Young, D.J.; Loh, X.J. Nanoparticle–Hydrogel Composites: Concept, Design, and Applications of These Promising, Multi-Functional Materials. Adv. Sci. 2015, 2, 1400010. [Google Scholar] [CrossRef]
- Qindeel, M.; Ullah, M.H.; Fakhar-ud-Din; Ahmed, N.; Rehman, A. Recent trends, challenges and future outlook of transdermal drug delivery systems for rheumatoid arthritis therapy. J. Control. Release 2020, 327, 595–615. [Google Scholar] [CrossRef]
- Xia, M.; Cheng, Y.; Meng, Z.; Jiang, X.; Chen, Z.; Theato, P.; Zhu, M. A novel nanocomposite hydrogel with precisely tunable UCST and LCST. Macromol. Rapid Commun. 2015, 36, 477–482. [Google Scholar] [CrossRef] [PubMed]
- Duan, Y.; Dhar, A.; Patel, C.; Khimani, M.; Neogi, S.; Sharma, P.; Siva Kumar, N.; Vekariya, R.L. A brief review on solid lipid nanoparticles: Part and parcel of contemporary drug delivery systems. RSC Adv. 2020, 10, 26777–26791. [Google Scholar] [CrossRef]
- Ma, G.; Lin, W.; Wang, Z.; Zhang, J.; Qian, H.; Xu, L.; Yuan, Z.; Chen, S. Development of polypeptide-based zwitterionic amphiphilic micelles for nanodrug delivery. J. Mater. Chem. B 2016, 4, 5256–5264. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, S.; Pavithran, M.; Viswanath, A.; Narayanan, D.; Mohan, C.C.; Manzoor, K.; Menon, D. Sequentially releasing dual-drug-loaded PLGA-casein core/shell nanomedicine: Design, synthesis, biocompatibility and pharmacokinetics. Acta Biomater. 2014, 10, 2112–2124. [Google Scholar] [CrossRef] [PubMed]
- Martínez Rivas, C.J.; Tarhini, M.; Badri, W.; Miladi, K.; Greige-Gerges, H.; Nazari, Q.A.; Galindo Rodríguez, S.A.; Román, R.Á.; Fessi, H.; Elaissari, A. Nanoprecipitation process: From encapsulation to drug delivery. Int. J. Pharm. 2017, 532, 66–81. [Google Scholar] [CrossRef]
- Tella, A.C.; Okoro, H.K.; Sokoya, S.O.; Adimula, V.O.; Olatunji, S.O.; Zvinowanda, C.; Ngila, J.C.; Shaibu, R.O.; Adeyemi, O.G. Synthesis, Characterization and Antifungal Activity of Fe(III) Metal–Organic Framework and its Nano-Composite. Chem. Afr. 2020, 3, 119–126. [Google Scholar] [CrossRef] [Green Version]
- Mohammadi-Samani, S.; Salehi, H.; Entezar-Almahdi, E.; Masjedi, M. Preparation and characterization of sumatriptan loaded solid lipid nanoparticles for transdermal delivery. J. Drug Deliv. Sci. Technol. 2020, 57, 101719. [Google Scholar] [CrossRef]
- Alvarez-Figueroa, M.J.; Narváez-Araya, D.; Armijo-Escalona, N.; Carrasco-Flores, E.A.; González-Aramundiz, J.V. Design of Chitosan Nanocapsules with Compritol 888 ATO® for Imiquimod Transdermal Administration. Evaluation of Their Skin Absorption by Raman Microscopy. Pharm. Res. 2020, 37, 195. [Google Scholar] [CrossRef]
- Eltellawy, Y.A.; El-Kayal, M.; Abdel-Rahman, R.F.; Salah, S.; Shaker, D.S. Optimization of transdermal atorvastatin calcium–Loaded proniosomes: Restoring lipid profile and alleviating hepatotoxicity in poloxamer 407-induced hyperlipidemia. Int. J. Pharm. 2021, 593, 120163. [Google Scholar] [CrossRef]
- Luo, Y.; Teng, Z.; Li, Y.; Wang, Q. Solid lipid nanoparticles for oral drug delivery: Chitosan coating improves stability, controlled delivery, mucoadhesion and cellular uptake. Carbohydr. Polym. 2015, 122, 221–229. [Google Scholar] [CrossRef]
- Saqib, M.; Shabbir Ali Bhatti, A.; Ahmad, N.M.; Ahmed, N.; Shahnaz, G.; Lebaz, N.; Elaissari, A. Amphotericin b loaded polymeric nanoparticles for treatment of leishmania infections. Nanomaterials 2020, 10, 1152. [Google Scholar] [CrossRef] [PubMed]
- Rana, I.; Khan, N.; Ansari, M.M.; Shah, F.A.; Din, F.U.; Sarwar, S.; Imran, M.; Qureshi, O.S.; Choi, H.I.; Lee, C.H.; et al. Solid lipid nanoparticles-mediated enhanced antidepressant activity of duloxetine in lipopolysaccharide-induced depressive model. Colloids Surf. B Biointerfaces 2020, 194, 111209. [Google Scholar] [CrossRef] [PubMed]
- Harisa, G.I.; Alomrani, A.H.; Badran, M.M. Simvastatin-loaded nanostructured lipid carriers attenuate the atherogenic risk of erythrocytes in hyperlipidemic rats. Eur. J. Pharm. Sci. 2017, 96, 62–71. [Google Scholar] [CrossRef] [PubMed]
- Qindeel, M.; Ahmed, N.; Sabir, F.; Khan, S.; Ur-Rehman, A. Development of novel pH-sensitive nanoparticles loaded hydrogel for transdermal drug delivery. Drug Dev. Ind. Pharm. 2019, 45, 629–641. [Google Scholar] [CrossRef] [PubMed]
- Jana, S.; Manna, S.; Nayak, A.K.; Sen, K.K.; Basu, S.K. Carbopol gel containing chitosan-egg albumin nanoparticles for transdermal aceclofenac delivery. Colloids Surf. B Biointerfaces 2014, 114, 36–44. [Google Scholar] [CrossRef]
- Zeb, A.; Qureshi, O.S.; Yu, C.H.; Akram, M.; Kim, H.S.; Kim, M.S.; Kang, J.H.; Majid, A.; Chang, S.Y.; Bae, O.N.; et al. Enhanced anti-rheumatic activity of methotrexate-entrapped ultradeformable liposomal gel in adjuvant-induced arthritis rat model. Int. J. Pharm. 2017, 525, 92–100. [Google Scholar] [CrossRef]
- Siddiqui, B.; ur.Rehman, A.; Haq, I.-U.; Ahmad, N.M.; Ahmed, N. Development, optimisation, and evaluation of nanoencapsulated diacerein emulgel for potential use in osteoarthritis. J. Microencapsul. 2020, 37, 595–608. [Google Scholar] [CrossRef]
- Seong, J.S.; Yun, M.E.; Park, S.N. Surfactant-stable and pH-sensitive liposomes coated with N-succinyl-chitosan and chitooligosaccharide for delivery of quercetin. Carbohydr. Polym. 2018, 181, 659–667. [Google Scholar] [CrossRef]
- Naz, K.; Shahnaz, G.; Ahmed, N.; Qureshi, N.A.; Sarwar, H.S.; Imran, M.; Khan, G.M. Formulation and In Vitro Characterization of Thiolated Buccoadhesive Film of Fluconazole. AAPS PharmSciTech 2017, 18, 1043–1055. [Google Scholar] [CrossRef]
- Jamadar, M.J.; Shaikh, R.H. Preparation and Evaluation of Herbal Gel Formulation. J. Pharaceutical Res. Educ. 2017, 1, 201–224. [Google Scholar]
- Qureshi, O.S.; Kim, H.S.; Zeb, A.; Choi, J.S.; Kim, H.S.; Kwon, J.E.; Kim, M.S.; Kang, J.H.; Ryou, C.; Park, J.S.; et al. Sustained release docetaxel-incorporated lipid nanoparticles with improved pharmacokinetics for oral and parenteral administration. J. Microencapsul. 2017, 34, 250–261. [Google Scholar] [CrossRef] [PubMed]
- Azeez, L.; Lateef, A.; Adejumo, A.L.; Adeleke, J.T.; Adetoro, R.O.; Mustapha, Z. Adsorption Behaviour of Rhodamine B on Hen Feather and Corn Starch Functionalized with Green Synthesized Silver Nanoparticles (AgNPs) Mediated with Cocoa Pods Extracts. Chem. Afr. 2020, 3, 237–250. [Google Scholar] [CrossRef] [Green Version]
- Dar, M.J.; Din, F.U.; Khan, G.M. Sodium stibogluconate loaded nano-deformable liposomes for topical treatment of leishmaniasis: Macrophage as a target cell. Drug Deliv. 2018, 25, 1595–1606. [Google Scholar] [CrossRef]
- Panchagnula, R.; Bokalial, R.; Sharma, P.; Khandavilli, S. Transdermal delivery of naloxone: Skin permeation, pharmacokinetic, irritancy and stability studies. Int. J. Pharm. 2005, 293, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Giuliano, E.; Paolino, D.; Fresta, M.; Cosco, D. Drug-Loaded Biocompatible Nanocarriers Embedded in Poloxamer 407 Hydrogels as Therapeutic Formulations. Medicines 2018, 6, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahmed, S.; Gull, A.; Aqil, M.; Danish Ansari, M.; Sultana, Y. Poloxamer-407 thickened lipid colloidal system of agomelatine for brain targeting: Characterization, brain pharmacokinetic study and behavioral study on Wistar rats. Colloids Surf. B Biointerfaces 2019, 181, 426–436. [Google Scholar] [CrossRef]
- Schröder, A.; Sprakel, J.; Schroën, K.; Spaen, J.N.; Berton-Carabin, C.C. Coalescence stability of Pickering emulsions produced with lipid particles: A microfluidic study. J. Food Eng. 2018, 234, 63–72. [Google Scholar] [CrossRef]
- Patil, S.G.; Jadhav, V.M.; Kadam, V.J. Formulation and Evaluation of Herbal Gel. Indian J. Nat. Prod. Resour. 2012, 3, 501–505. [Google Scholar]
- De Mendoza, A.E.H.; Campanero, M.A.; Mollinedo, F.; Blanco-Prieto, M.J. Lipid nanomedicines for anticancer drug therapy. J. Biomed. Nanotechnol. 2009, 5, 323–343. [Google Scholar] [CrossRef] [Green Version]
- Qureshi, O.S.; Zeb, A.; Akram, M.; Kim, M.S.; Kang, J.H.; Kim, H.S.; Majid, A.; Han, I.; Chang, S.Y.; Bae, O.N.; et al. Enhanced acute anti-inflammatory effects of CORM-2-loaded nanoparticles via sustained carbon monoxide delivery. Eur. J. Pharm. Biopharm. 2016, 108, 187–195. [Google Scholar] [CrossRef]
- Dudhipala, N.; Veerabrahma, K. Improved anti-hyperlipidemic activity of Rosuvastatin Calcium via lipid nanoparticles: Pharmacokinetic and pharmacodynamic evaluation. Eur. J. Pharm. Biopharm. 2017, 110, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Jaafar-Maalej, C.; Diab, R.; Andrieu, V.; Elaissari, A.; Fessi, H. Ethanol injection method for hydrophilic and lipophilic drug-loaded liposome preparation. J. Liposome Res. 2010, 20, 228–243. [Google Scholar] [CrossRef] [PubMed]
- Tarhini, M.; Benlyamani, I.; Hamdani, S.; Agusti, G.; Fessi, H.; Greige-Gerges, H.; Bentaher, A.; Elaissari, A. Protein-based nanoparticle preparation via nanoprecipitation method. Materials 2018, 11, 394. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Md, S.; Kuldeep Singh, J.K.A.; Waqas, M.; Pandey, M.; Choudhury, H.; Habib, H.; Hussain, F.; Hussain, Z. Nanoencapsulation of betamethasone valerate using high pressure homogenization–solvent evaporation technique: Optimization of formulation and process parameters for efficient dermal targeting. Drug Dev. Ind. Pharm. 2019, 45, 323–332. [Google Scholar] [CrossRef]
- Jiang, T.; Han, N.; Zhao, B.; Xie, Y.; Wang, S. Enhanced dissolution rate and oral bioavailability of simvastatin nanocrystal prepared by sonoprecipitation. Drug Dev. Ind. Pharm. 2012, 38, 1230–1239. [Google Scholar] [CrossRef]
- Rauf, A.; Razzaq, S.; Tabish, T.A.; Tahseen, S.; Sandhu, M.A.; Shahnaz, G. Investigating the intracellular bactericidal effects of rifampicin loaded S-protected thiomeric chitosan nanocargoes against Mycobacterium tuberculosis. J. Drug Deliv. Sci. Technol. 2021, 61, 102184. [Google Scholar] [CrossRef]
- Hasan, A.S.; Socha, M.; Lamprecht, A.; Ghazouani, F.; El Sapin, A.; Hoffman, M.; Maincent, P.; Ubrich, N. Effect of the microencapsulation of nanoparticles on the reduction of burst release. Int. J. Pharm. 2007, 344, 53–61. [Google Scholar] [CrossRef]
- Chen, H.; Chang, X.; Du, D.; Li, J.; Xu, H.; Yang, X. Microemulsion-based hydrogel formulation of ibuprofen for topical delivery. Int. J. Pharm. 2006, 315, 52–58. [Google Scholar] [CrossRef]
- Gangurde, A.B.; Amin, P.D. Microencapsulation by Spray Drying of Vitamin A Palmitate from Oil to Powder and Its Application in Topical Delivery System. J. Encapsul. Adsorpt. Sci. 2017, 07, 10–39. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Yang, Q.; Zhao, Y.; Long, S.; Zheng, J. Dual physically crosslinked double network hydrogels with high toughness and self-healing properties. Soft Matter 2017, 13, 911–920. [Google Scholar] [CrossRef]
- Mirzaei, B.E.; Ramazani, A.; Shafiee, M.; Danaei, M. Studies on glutaraldehyde crosslinked chitosan hydrogel properties for drug delivery systems. Int. J. Polym. Mater. Polym. Biomater. 2013, 62, 605–611. [Google Scholar] [CrossRef]
- Puri, A.; Frempong, D.; Mishra, D.; Dogra, P. Microneedle-mediated transdermal delivery of naloxone hydrochloride for treatment of opioid overdose. Int. J. Pharm. 2021, 604, 120739. [Google Scholar] [CrossRef] [PubMed]
- Din, F.U.; Saleem, S.; Aleem, F.; Ahmed, R.; Huda, N.U.; Ahmed, S.; Khaleeq, N.; Shah, K.U.; Ullah, I.; Zeb, A.; et al. Advanced colloidal technologies for the enhanced bioavailability of drugs. Cogent Med. 2018, 5, 1480572. [Google Scholar] [CrossRef]
- Kawar, D.; Abdelkader, H. Hyaluronic acid gel-core liposomes (hyaluosomes) enhance skin permeation of ketoprofen. Pharm. Dev. Technol. 2019, 24, 947–953. [Google Scholar] [CrossRef]
- Khalid, A.; Ahmed, N.; Qindeel, M.; Asad, M.I.; Khan, G.M.; Rehman, A.U. Development of novel biopolymer-based nanoparticles loaded cream for potential treatment of topical fungal infections. Drug Dev. Ind. Pharm. 2021, 47, 1090–1099. [Google Scholar] [CrossRef]

| Formulation Code | Lipid Phase | Aqueous Phase (20 mL Water) | |
|---|---|---|---|
| Lipid (mg) | Solvent (mL) | P407 (%) | |
| A | 50 | 2 | 0.25 |
| B | 50 | 2 | 0.5 |
| C | 50 | 2 | 0.75 |
| D | 50 | 2 | 1 |
| E | 50 | 2 | 1.5 |
| F | 30 | 2 | 0.5 |
| G | 40 | 2 | 0.5 |
| H | 50 | 2 | 0.5 |
| I | 60 | 2 | 0.5 |
| J | 70 | 2 | 0.5 |
| Formulation Code | Entrapment Efficiency | Particle Size (nm) | PDI | Observation |
|---|---|---|---|---|
| A | 65 | 230 | 0.400 | Non uniform a and unstable b |
| B | 79 | 225 | 0.452 | Uniform and stable c |
| C | 75 | 180 | 0.327 | Uniform d and stable |
| D | 68 | 175 | 0.252 | Uniform and stable |
| E | 63 | 166 | 0.174 | Uniform and stable |
| F | 74 | 177 | 0.268 | Uniform and stable |
| G | 77 | 182 | 0.372 | Uniform and stable |
| H | 82 | 205 | 0.127 | Uniform and stable |
| I | 79 | 241 | 0.407 | Non uniform and stable |
| J | 71 | 551 | 0.793 | Non uniform and unstable |
| Time (Months) | Physical Appearance | pH | ||
|---|---|---|---|---|
| Phase Separation | Color | Grittiness | ||
| 0 | No | No change | None | 5.3 |
| 3 | No | No change | None | 5.5 |
| 6 | No | No change | None | 5.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, M.F.A.; Ur.Rehman, A.; Howari, H.; Alhodaib, A.; Ullah, F.; Mustafa, Z.u.; Elaissari, A.; Ahmed, N. Hydrogel Containing Solid Lipid Nanoparticles Loaded with Argan Oil and Simvastatin: Preparation, In Vitro and Ex Vivo Assessment. Gels 2022, 8, 277. https://doi.org/10.3390/gels8050277
Khan MFA, Ur.Rehman A, Howari H, Alhodaib A, Ullah F, Mustafa Zu, Elaissari A, Ahmed N. Hydrogel Containing Solid Lipid Nanoparticles Loaded with Argan Oil and Simvastatin: Preparation, In Vitro and Ex Vivo Assessment. Gels. 2022; 8(5):277. https://doi.org/10.3390/gels8050277
Chicago/Turabian StyleKhan, Muhammad Farhan Ali, Asim Ur.Rehman, Haidar Howari, Aiyeshah Alhodaib, Faiz Ullah, Zia ul Mustafa, Abdelhamid Elaissari, and Naveed Ahmed. 2022. "Hydrogel Containing Solid Lipid Nanoparticles Loaded with Argan Oil and Simvastatin: Preparation, In Vitro and Ex Vivo Assessment" Gels 8, no. 5: 277. https://doi.org/10.3390/gels8050277
APA StyleKhan, M. F. A., Ur.Rehman, A., Howari, H., Alhodaib, A., Ullah, F., Mustafa, Z. u., Elaissari, A., & Ahmed, N. (2022). Hydrogel Containing Solid Lipid Nanoparticles Loaded with Argan Oil and Simvastatin: Preparation, In Vitro and Ex Vivo Assessment. Gels, 8(5), 277. https://doi.org/10.3390/gels8050277