Formulation and Evaluation of Topical Nano-Lipid-Based Delivery of Butenafine: In Vitro Characterization and Antifungal Activity
Abstract
:1. Introduction
2. Result and Discussion
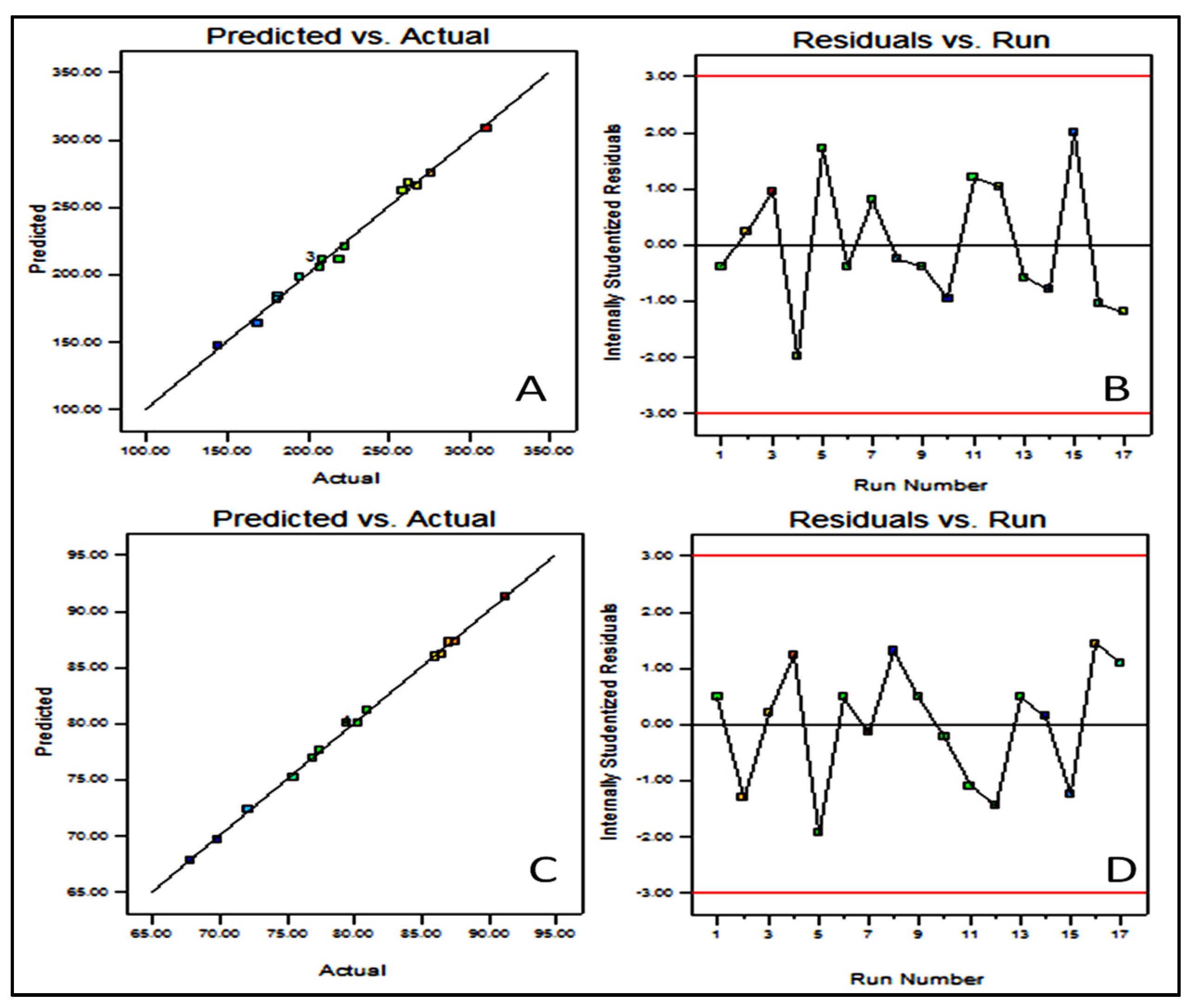
2.1. Optimization
2.2. Effect of Independent Variables on Vesicle Size (Y1)
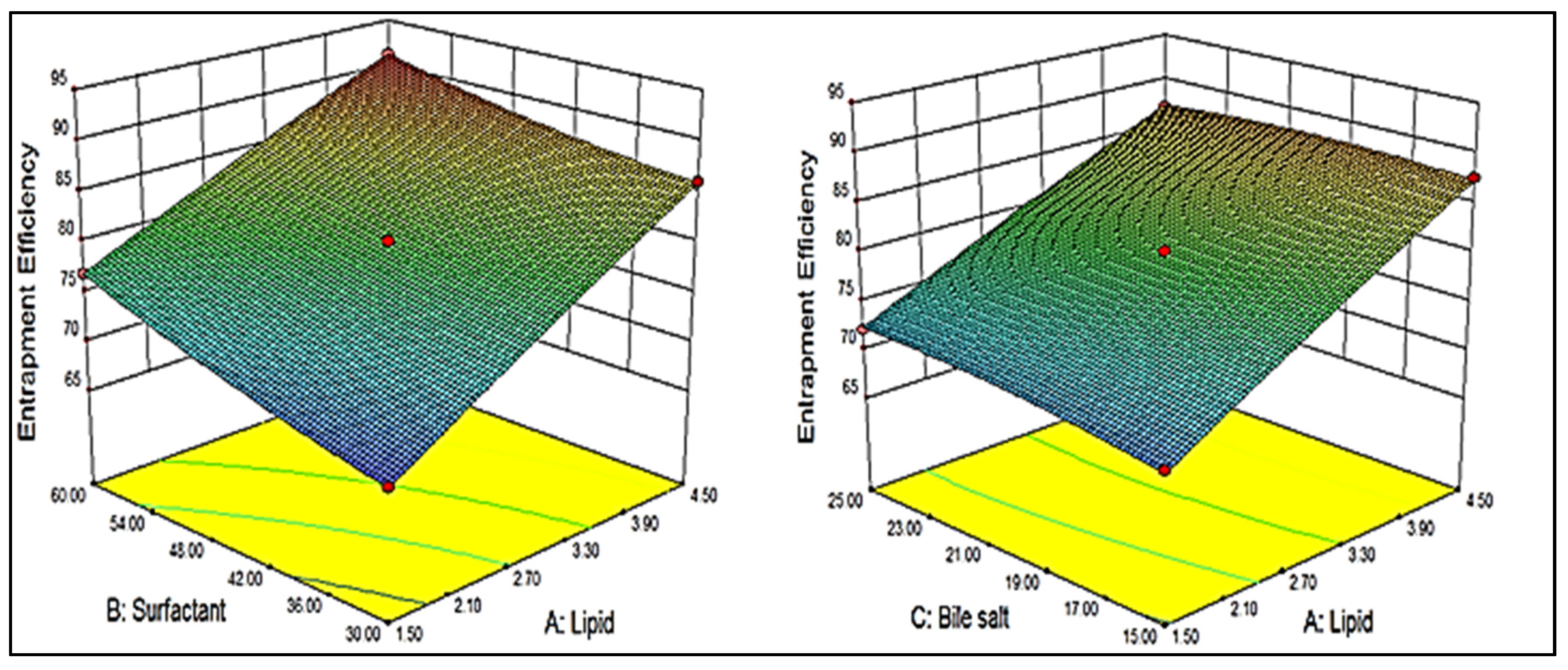
2.3. Effect of Independent Variables on Entrapment Efficiency (Y2)
2.4. Point Prediction
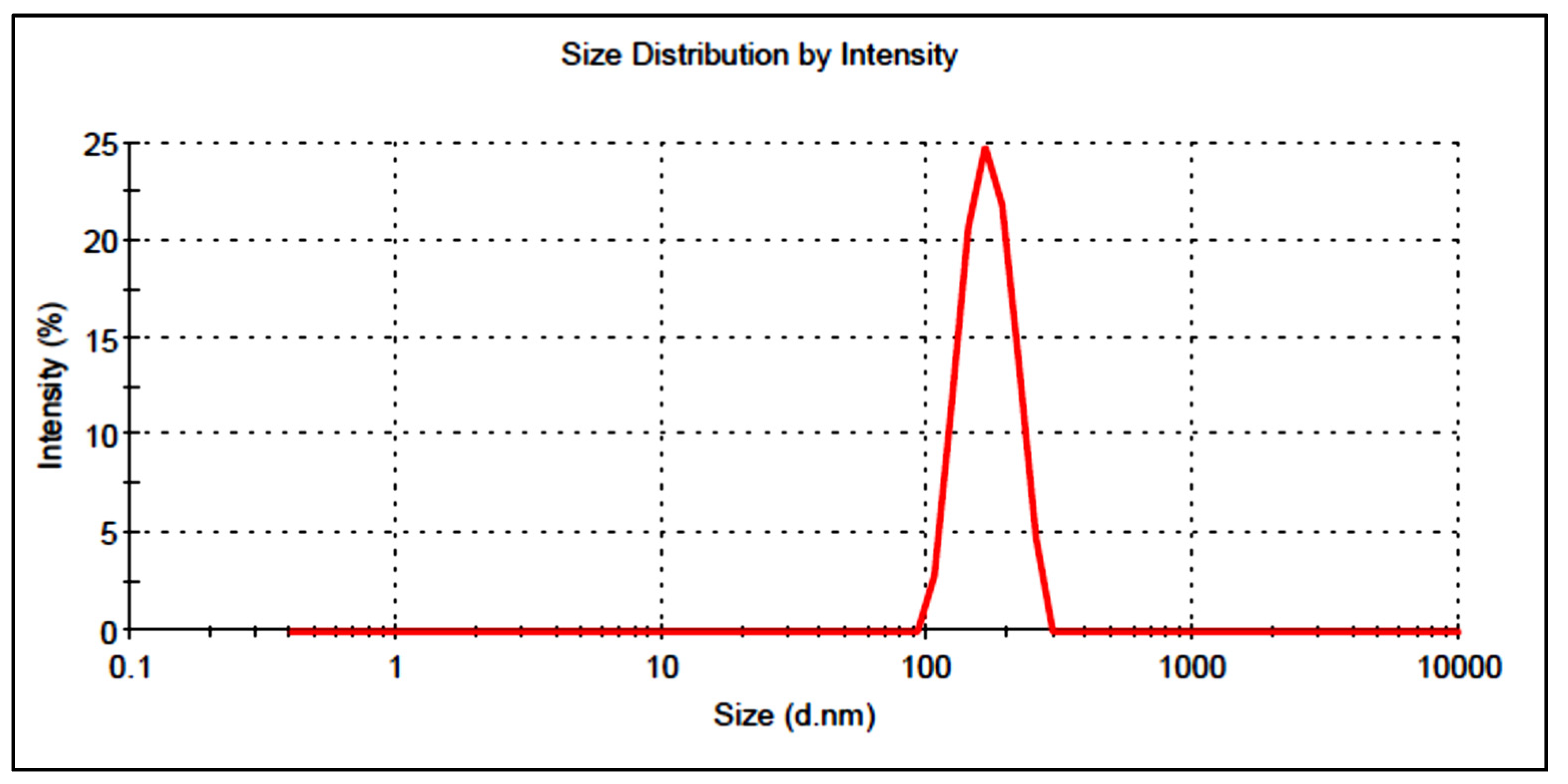
2.5. Vesicle Size, PDI, and Zeta Potential
2.6. Entrapment Efficiency
2.7. Thermal Analysis
2.8. Development of BN-BSo-Gel
2.9. Viscosity, pH, and Drug Content
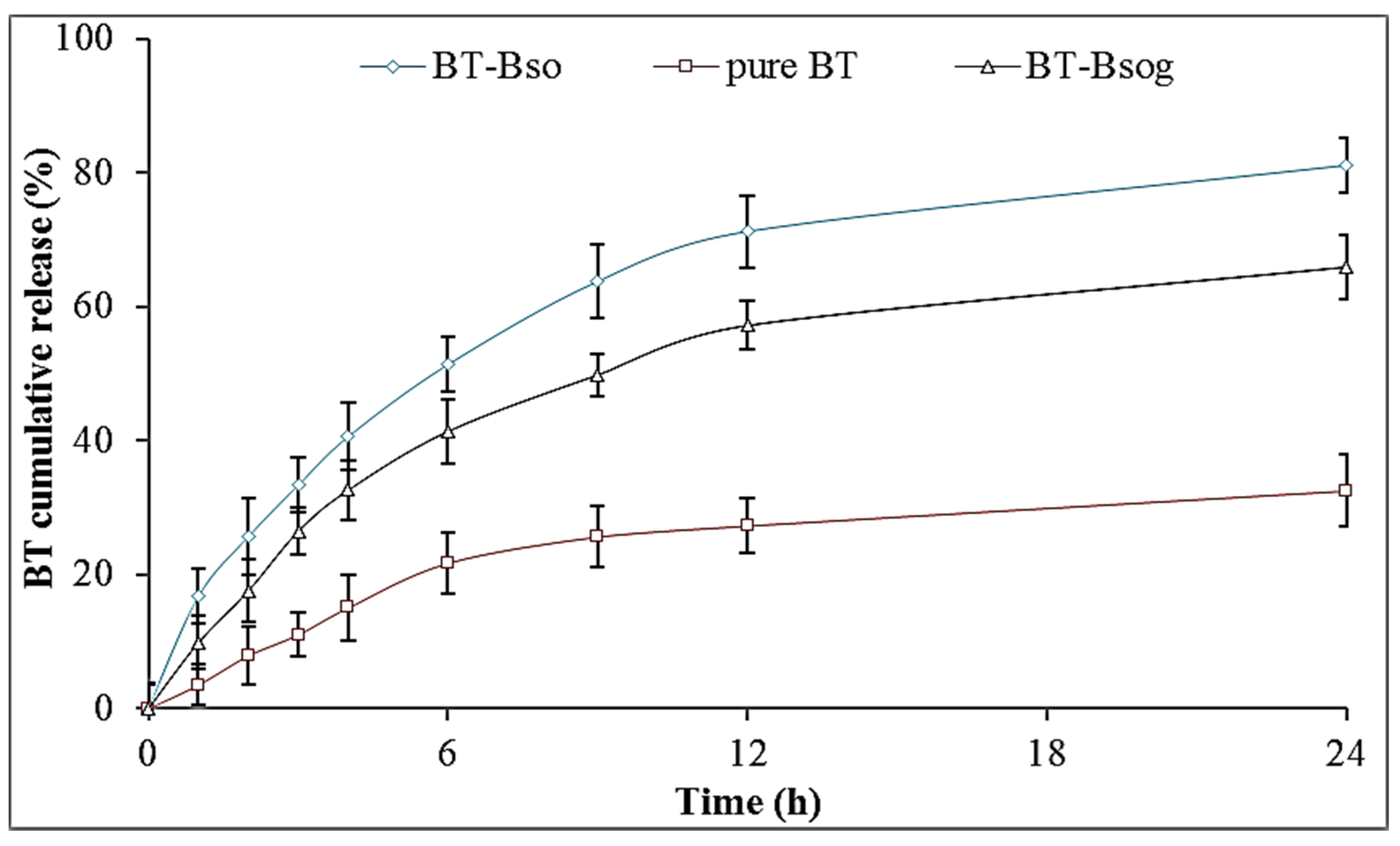
2.10. In Vitro Release Study
2.11. In Vitro Permeation Study
2.12. Irritation Study
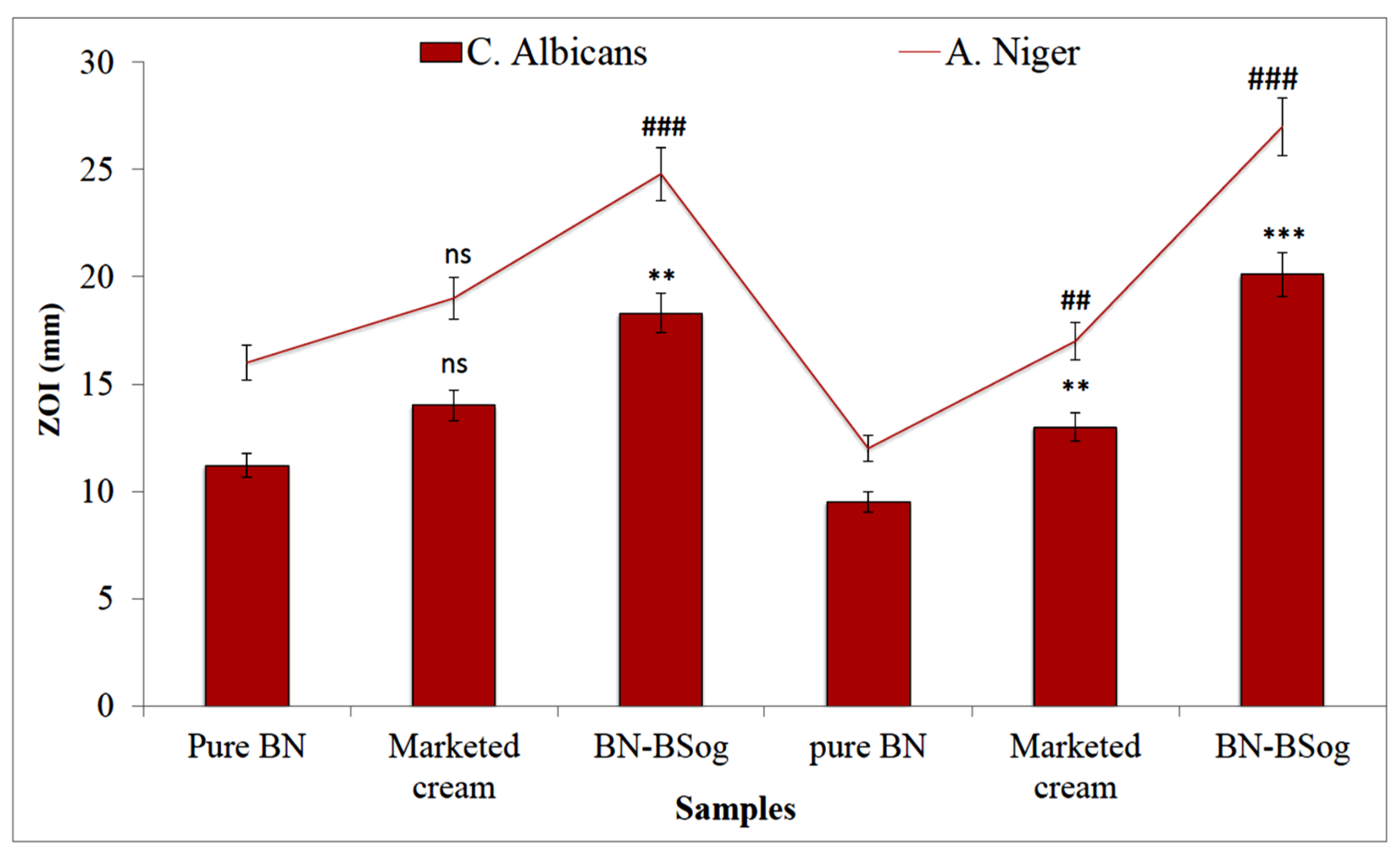
2.13. Antifungal Study
3. Conclusions
4. Materials and Experimental
4.1. Materials
4.2. Experimental
4.2.1. Preliminary Study
4.2.2. Optimization
4.2.3. Formulation of Bilosomes
4.3. Characterization of BN-BS
4.3.1. Particle Characterization
4.3.2. Entrapment Efficiency
4.3.3. Thermal Analysis
4.3.4. Development of BN-BSs Loaded Gel
4.3.5. Gel Characterization
4.3.6. In Vitro Release Study
4.3.7. Permeation Study
4.3.8. Irritation Study
4.3.9. Antifungal Study
4.3.10. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mendes, I.T.; Ruela, A.L.M.; Carvalho, F.C.; Freitas, J.T.J.; Bonfilio, R.; Pereira, G.R. Development and characterization of nanostructured lipid carrier-based gels for the transdermal delivery of donepezil. Colloids Surf. B Biointerfaces 2019, 177, 274–281. [Google Scholar] [CrossRef] [PubMed]
- Al-Mahallawi, A.M.; Abdelbary, A.A.; Aburahma, M.H. Investigating the potential of employing bilosomes as a novel vesicular carrier for transdermal delivery of tenoxicam. Int. J. Pharm. 2015, 485, 329–340. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.; Pradhan, M.; Nag, M.; Singh, M.R. Vesicular system: Versatile carrier for transdermal delivery of bioactives. Artif. Cells Nanomed. Biotechnol. 2015, 43, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Kassem, M.A.; Sayed, S. Bilosomes as Promising Nanovesicular Carriers for Improved Transdermal Delivery: Construction, in vitro Optimization, ex vivo Permeation and in vivo Evaluation. Int. J. Nanomed. 2020, 15, 9783–9798. [Google Scholar] [CrossRef] [PubMed]
- Niu, X.Q.; Zhang, D.P.; Bian, Q.; Feng, X.F.; Li, H.; Rao, Y.F.; Shen, Y.M.; Geng, F.N.; Yuan, A.R.; Ying, X.Y.; et al. Mechanism investigation of ethosomes transdermal permeation. Int. J. Pharm. X 2019, 1, 100027. [Google Scholar] [CrossRef]
- Zhang, Y.; Jing, Q.; Hu, H.; He, Z.; Wu, T.; Guo, T.; Feng, N. Sodium dodecyl sulfate improved stability and transdermal delivery of salidroside-encapsulated niosomes via effects on zeta potential. Int. J. Pharm. 2020, 580, 119183. [Google Scholar] [CrossRef]
- Pandit, A.P.; Omase, S.B.; Mute, V.M. A chitosan film containing quercetin-loaded transfersomes for treatment of secondary osteoporosis. Drug Deliv. Transl. Res. 2020, 10, 1495–1506. [Google Scholar] [CrossRef]
- Maione-Silva, L.; de Castro, E.G.; Nascimento, T.L.; Cintra, E.R.; Moreira, L.C.; Cintra, B.A.S.; Valadares, M.C.; Lima, E.M. Ascorbic acid encapsulated into negatively charged liposomes exhibits increased skin permeation, retention and enhances collagen synthesis by fibroblasts. Sci. Rep. 2019, 9, 522. [Google Scholar] [CrossRef] [Green Version]
- Zafar, A.; Alruwaili, N.K.; Imam, S.S.; Hadal Alotaibi, N.; Alharbi, K.S.; Afzal, M.; Ali, R.; Alshehri, S.; Alzarea, S.I.; Elmowafy, M.; et al. Bioactive Apigenin loaded oral nano bilosomes: Formulation optimization to preclinical assessment. Saudi Pharm. J. 2021, 29, 269–279. [Google Scholar] [CrossRef]
- El Menshawe, S.F.; Aboud, H.M.; Elkomy, M.H.; Kharshoum, R.M.; Abdeltwab, A.M. A novel nanogel loaded with chitosan decorated bilosomes for transdermal delivery of terbutaline sulfate: Artificial neural network optimization, in vitro characterization and in vivo evaluation. Drug Deliv. Transl. Res. 2020, 10, 471–485. [Google Scholar] [CrossRef]
- Khalil, R.M.; Abdelbary, A.; Kocova El-Arini, S.; Basha, M.; El-Hashemy, H.A. Evaluation of bilosomes as nanocarriers for transdermal delivery of tizanidine hydrochloride: In vitro and ex vivo optimization. J. Liposome Res. 2019, 29, 171–182. [Google Scholar] [CrossRef] [PubMed]
- Albash, R.; El-Nabarawi, M.A.; Refai, H.; Abdelbary, A.A. Tailoring of PEGylated bilosomes for promoting the transdermal delivery of olmesartan medoxomil: In-vitro characterization, ex-vivo permeation and in-vivo assessment. Int. J. Nanomed. 2019, 14, 6555–6574. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fetih, G. Fluconazole-loaded niosomal gels as a topical ocular drug delivery system for corneal fungal infections. J. Drug Deliv. Sci. Technol. 2016, 35, 8–15. [Google Scholar] [CrossRef]
- Soliman, O.A.E.; Mohamed, E.A.; Khatera, N.A.A. Enhanced ocular bioavailability of fluconazole from niosomal gels and microemulsions: Formulation, optimization, and in vitro-in vivo evaluation. Pharm. Dev. Technol. 2019, 24, 48–62. [Google Scholar] [CrossRef]
- Garg, A.K.; Maddiboyina, B.; Alqarni, M.H.S.; Alam, A.; Aldawsari, H.M.; Rawat, P.; Singh, S.; Kesharwani, P. Solubility enhancement, formulation development and antifungal activity of luliconazole niosomal gel-based system. J. Biomater. Sci. Polym. Ed. 2021, 32, 1009–1023. [Google Scholar] [CrossRef]
- Kumar, N.; Goindi, S. Development, characterization and preclinical evaluation of nanosized liposomes of itraconazole for topical application: 32 full factorial design to estimate the relationship between formulation components. J. Drug Deliv. Sci. Technol. 2021, 66, 102785. [Google Scholar] [CrossRef]
- Shetty, S.; Jose, J.; Kumar, L.; Charyulu, R.N. Novel ethosomal gel of clove oil for the treatment of cutaneous candidiasis. J. Cosmet. Dermatol. 2019, 18, 862–869. [Google Scholar] [CrossRef]
- Rao, S.; Barot, T.; Rajesh, K.S.; Jha, L.L. Formulation, optimization and evaluation of microemulsion based gel of Butenafine Hydrochloride for topical delivery by using simplex lattice mixture design. J. Pharm. Investig. 2016, 46, 1–12. [Google Scholar] [CrossRef]
- Ahmed, M.M.; Fatima, F.; Anwer, M.K.; Ibnouf, E.O.; Kalam, M.A.; Alshamsan, A.; Aldawsari, M.F.; Alalaiwe, A.; Ansari, M.J. Formulation and in vitro evaluation of topical nanosponge-based gel containing butenafine for the treatment of fungal skin infection. Saudi Pharm. J. 2021, 29, 467–477. [Google Scholar] [CrossRef]
- Alshehri, S.; Imam, S.S. Formulation and evaluation of butenafine loaded PLGA-nanoparticulate laden chitosan nano gel. Drug Deliv. 2021, 28, 2348–2360. [Google Scholar] [CrossRef]
- Mahdi, W.A.; Bukhari, S.I.; Imam, S.S.; Alshehri, S.; Zafar, A.; Yasir, M. Formulation and Optimization of Butenafine-Loaded Topical Nano Lipid Carrier-Based Gel: Characterization, Irritation Study, and Anti-Fungal Activity. Pharmaceutics 2021, 13, 1087. [Google Scholar] [CrossRef] [PubMed]
- Fachel, F.N.S.; Medeiros-Neves, B.; Dal Prá, M.; Schuh, R.S.; Veras, K.S.; Bassani, V.L.; Koester, L.S.; Henriques, A.T.; Braganhol, E.; Teixeira, H.F. Box-Behnken design optimization of mucoadhesive chitosan-coated nanoemulsions for rosmarinic acid nasal delivery-In vitro studies. Carbohydr. Polym. 2018, 199, 572–582. [Google Scholar] [CrossRef]
- Virupakshappa, P.K.; Krishnaswamy, M.B.; Mishra, G.; Mehkri, M.A. Optimization of Crude Oil and PAHs Degradation by Stenotrophomonas rhizophila KX082814 Strain through Response Surface Methodology Using Box-Behnken Design. Biotechnol. Res. Int. 2016, 2016, 4769542. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jazuli, I.; Annu Nabi, B.; Moolakkadath, T.; Alam, T.; Baboota, S.; Ali, J. Optimization of Nanostructured Lipid Carriers of Lurasidone Hydrochloride Using Box-Behnken Design for Brain Targeting: In Vitro and In Vivo Studies. J. Pharm. Sci. 2019, 108, 3082–3090. [Google Scholar] [CrossRef] [PubMed]
- Yusuf, M.; Sharma, V.; Pathak, K. Nanovesicles for transdermal delivery of felodipine: Development, characterization, and pharmacokinetics. Int. J. Pharm. Investig. 2014, 4, 119–130. [Google Scholar] [PubMed] [Green Version]
- Yang, G.; Wu, F.; Chen, M.; Jin, J.; Wang, R.; Yuan, Y. Formulation design, characterization, and in vitro and in vivo evaluation of nanostructured lipid carriers containing a bile salt for oral delivery of gypenosides. Int. J. Nanomed. 2019, 14, 2267–2280. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gonzalez-Rodriguez, M.L.; Arroyo, C.M.; Cozar-Bernal, M.J.; Gonzalez, R.P.; Leon, J.M.; Calle, M.; Canca, D.; Rabasco, A.M. Deformability properties of timolol-loaded transfersomes based on the extrusion mechanism. Statistical optimization of the process. Drug Dev. Ind. Pharm. 2016, 42, 1683–1694. [Google Scholar] [CrossRef]
- Abdelbary, G.; El-Gendy, N. Niosome-encapsulated gentamicin for ophthalmic controlled delivery. AAPS PharmSciTech 2008, 9, 740–747. [Google Scholar] [CrossRef]
- Aboud, H.M.; Ali, A.A.; El-Menshawe, S.F.; Elbary, A.A. Nanotransfersomes of carvedilol for intranasal delivery: Formulation, characterization and in vivo evaluation. Drug Deliv. 2016, 23, 2471–2481. [Google Scholar] [CrossRef] [Green Version]
- Talele, P.; Sahu, S.; Mishra, A.K. Physicochemical characterization of solid lipid nanoparticles comprised of glycerol monostearate and bile salts. Colloids Surf. B Biointerfaces 2018, 172, 517–525. [Google Scholar] [CrossRef]
- Mosallam, S.; Sheta, N.M.; Elshafeey, A.H.; Abdelbary, A.A. Fabrication of Highly Deformable Bilosomes for Enhancing the Topical Delivery of Terconazole: In Vitro Characterization, Microbiological Evaluation, and In Vivo Skin Deposition Study. AAPS PharmSciTech 2021, 22, 74. [Google Scholar] [CrossRef] [PubMed]
- Uprit, S.; Kumar Sahu, R.; Roy, A.; Pare, A. Preparation and characterization of minoxidil loaded nanostructured lipid carrier gel for effective treatment of alopecia. Saudi Pharm. J. 2019, 21, 379–385. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fasolo, D.; Pippi, B.; Meirelles, G.; Zorzi, G.; Fuentefria, A.M.; Poser, G.V.; Teixeira, H.F. Topical delivery of antifungal Brazilian red propolis benzophenones-rich extract by means of cationic lipid nanoemulsions optimized by means of Box-Behnken Design. J. Drug Deliv. Sci. Technol. 2020, 56, 101573. [Google Scholar] [CrossRef]
- Zhang, W.; Zhao, X.; Yu, G.; Suo, M. Optimization of propofol loaded niosomal gel for transdermal delivery. J. Biomater. Sci. Polym. Ed. 2021, 32, 858–873. [Google Scholar] [CrossRef] [PubMed]
- Higuchi, T. Mechanism of sustained- action medication. Theoretical analysis of rate of release of solid drugs dispersed in solid matrices. J. Pharm. Sci. 1963, 52, 1145–1149. [Google Scholar] [CrossRef] [PubMed]
- Ansari, M.; Kazemipour, M.; Aklamli, M. The study of drug permeation through natural membranes. Int. J. Pharm. 2006, 327, 6–11. [Google Scholar] [CrossRef] [PubMed]
- Haigh, J.M.; Smith, E.W. The selection and use of natural and synthetic membranes for in vitro diffusion experiments. Eur. J. Pharm. Sci. 1994, 2, 311–330. [Google Scholar] [CrossRef]
- Vinardell, M.; Mitjans, M. Alternative methods for eye and skin irritation tests: An overview. J. Pharm. Sci. 2008, 97, 46–59. [Google Scholar] [CrossRef]



| Formulation Code | Independent Constraints | Dependent Constraints | |||
|---|---|---|---|---|---|
| Lipid (%) | Surfactant (mg) | Bile Salt (mg) | Vesicle Size (nm) | Entrapment Efficiency (%) | |
| A | B | C | Y1 | Y2 | |
| F1 | 1.5 | 30 | 20 | 181.5 ± 4.5 | 67.79 ± 1.5 |
| F2 | 4.5 | 30 | 20 | 310.1 ± 7.4 | 85.99 ± 1.8 |
| F3 | 1.5 | 60 | 20 | 144.4 ± 4.8 | 76.88 ± 1.4 |
| F4 | 4.5 | 60 | 20 | 222.8 ± 5.2 | 91.29 ± 2.1 |
| F5 | 1.5 | 45 | 15 | 180.8 ± 3.5 | 69.87 ± 1.3 |
| F6 | 4.5 | 45 | 15 | 262.7 ± 6.2 | 87.56 ± 1.8 |
| F7 | 1.5 | 45 | 25 | 168.9 ± 4.1 | 72.12 ± 1.4 |
| F8 | 4.5 | 45 | 25 | 276.3 ± 6.3 | 86.99 ± 1.7 |
| F9 | 3 | 30 | 15 | 268.2 ± 5.9 | 77.38 ± 1.4 |
| F10 | 3 | 60 | 15 | 208.2 ± 4.2 | 80.99 ± 1.6 |
| F11 | 3 | 30 | 25 | 258.8 ± 5.7 | 75.46 ± 1.4 |
| F12 | 3 | 60 | 25 | 195.3 ± 4.5 | 86.50 ± 1.7 |
| F13 * | 3 | 45 | 20 | 209.6 ± 4.7 | 80.23 ± 2.7 |
| F14 * | 3 | 45 | 20 | 208.1 ± 4.4 | 80.98 ± 2.1 |
| F15 * | 3 | 45 | 20 | 209.3 ± 3.5 | 80.12 ± 1.4 |
| F16 * | 3 | 45 | 20 | 209.9 ± 4.8 | 80.21 ± 1.9 |
| F17 * | 3 | 45 | 20 | 207.9 ± 5.1 | 80.56 ± 1.3 |
| Model | Y₁ (Vesicle Size, nm) | ||||
|---|---|---|---|---|---|
| SD | R² | Adjusted R² | Predicted R² | p-Value | |
| Linear | 12.85 | 0.9275 | 0.9108 | 0.8625 | <0.0001 |
| 2FI | 11.54 | 0.9551 | 0.9281 | 0.8283 | 0.1717 |
| Quadratic | 5.36 | 0.9932 | 0.9845 | 0.9328 | 0.0029 |
| Y₂ (Entrapment efficiency, %) | |||||
| Linear | 1.37 | 0.9634 | 0.9549 | 0.9242 | <0.0001 |
| 2FI | 0.70 | 0.9926 | 0.9881 | 0.9654 | 0.0009 |
| Quadratic | 0.38 | 0.9985 | 0.9966 | 0.9876 | 0.0077 |
| Source | Particle Size (nm) | Entrapment Efficiency (%) | ||||||
|---|---|---|---|---|---|---|---|---|
| Sum of the Square | DF | F-Value | p-Value | Sum of the Square | DF | F-Value | p-Value | |
| Model | 29,427.84 | 9 | 114.00 | <0.0001 * | 663.11 | 9 | 521.14 | <0.0001 * |
| A | 19,700.13 | 1 | 686.83 | <0.0001 * | 530.89 | 1 | 3755.08 | <0.0001 * |
| B | 7727.73 | 1 | 269.42 | <0.0001 * | 105.42 | 1 | 745.62 | <0.0001 * |
| C | 52.69 | 1 | 1.84 | 0.2174 ** | 3.47 | 1 | 24.56 | 0.0016 * |
| AB | 651.02 | 1 | 22.70 | 0.0020 * | 3.59 | 1 | 25.40 | 0.0015 * |
| AC | 162.31 | 1 | 5.66 | 0.0490 * | 1.99 | 1 | 14.06 | 0.0072 * |
| BC | 3.12 | 1 | 0.11 | 0.7514 ** | 13.80 | 1 | 97.62 | <0.0001 * |
| A2 | 49.05 | 1 | 1.71 | 0.2323 ** | 0.29 | 1 | 2.08 | 0.1929 ** |
| B2 | 206.58 | 1 | 7.20 | 0.0314 * | 1.97 | 1 | 13.91 | 0.0074 * |
| C2 | 855.84 | 1 | 29.84 | 0.0009 * | 1.88 | 1 | 13.33 | 0.0082 * |
| Residual | 200.78 | 7 | <0.0001 * | 0.99 | 7 | |||
| Lack of Fit | 116.15 | 3 | 1.83 | <0.0001 * | 0.46 | 3 | 1.18 | 0.4221 ** |
| Pure Error | 84.63 | 4 | 0.52 | 4 | ||||
| Total | 29,628.62 | 16 | 664.10 | 16 | ||||
| Samples | Egg | Score | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Nonirritant | Mild Irritant | Moderate Irritant | Severe Irritant | Overall Score | ||||||||||||
| Time (min) | Time (min) | Time (min) | Time (min) | |||||||||||||
| 0 | 0.5 | 2 | 5 | 0.5 | 2 | 5 | 0.5 | 2 | 5 | 0 | 0.5 | 2 | 5 | |||
| BN-BSog | Egg 1 | 0 | 0 | 0 | 0.0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Egg 2 | 0 | 0 | 0 | 0.0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||
| Egg 3 | 0 | 0 | 0 | 0.2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||
| Mean score | 0 | 0 | 0 | 0.2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||
| Positive control (SLS, 2%) | Egg 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 14 | 17 | 19 | 17.44 |
| Egg 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 16 | 18 | 19 | ||
| Egg 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 16 | 18 | 20 | ||
| Mean score | 0 | 0 | 0 | 0. | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 15.33 | 17.66 | 19.33 | ||
| Negative control (NS, 0.9%) | Egg 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Egg 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||
| Egg 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||
| Mean score | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||
| Independent Variables | Dependent Variables | |
|---|---|---|
| Lower (−1) | Upper (+1) | |
| Lipid (%) | 1.5 | 4.5 |
| Surfactant (Span 60, mg) | 30 | 60 |
| Bile salt (mg) | 15 | 25 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zafar, A.; Imam, S.S.; Alruwaili, N.K.; Yasir, M.; Alsaidan, O.A.; Alshehri, S.; Ghoneim, M.M.; Khalid, M.; Alquraini, A.; Alharthi, S.S. Formulation and Evaluation of Topical Nano-Lipid-Based Delivery of Butenafine: In Vitro Characterization and Antifungal Activity. Gels 2022, 8, 133. https://doi.org/10.3390/gels8020133
Zafar A, Imam SS, Alruwaili NK, Yasir M, Alsaidan OA, Alshehri S, Ghoneim MM, Khalid M, Alquraini A, Alharthi SS. Formulation and Evaluation of Topical Nano-Lipid-Based Delivery of Butenafine: In Vitro Characterization and Antifungal Activity. Gels. 2022; 8(2):133. https://doi.org/10.3390/gels8020133
Chicago/Turabian StyleZafar, Ameeduzzafar, Syed Sarim Imam, Nabil K. Alruwaili, Mohd Yasir, Omar Awad Alsaidan, Sultan Alshehri, Mohammed M. Ghoneim, Mohammad Khalid, Ali Alquraini, and Salman S. Alharthi. 2022. "Formulation and Evaluation of Topical Nano-Lipid-Based Delivery of Butenafine: In Vitro Characterization and Antifungal Activity" Gels 8, no. 2: 133. https://doi.org/10.3390/gels8020133
APA StyleZafar, A., Imam, S. S., Alruwaili, N. K., Yasir, M., Alsaidan, O. A., Alshehri, S., Ghoneim, M. M., Khalid, M., Alquraini, A., & Alharthi, S. S. (2022). Formulation and Evaluation of Topical Nano-Lipid-Based Delivery of Butenafine: In Vitro Characterization and Antifungal Activity. Gels, 8(2), 133. https://doi.org/10.3390/gels8020133









