Review on Sol-Gel Synthesis of Perovskite and Oxide Nanomaterials
Abstract
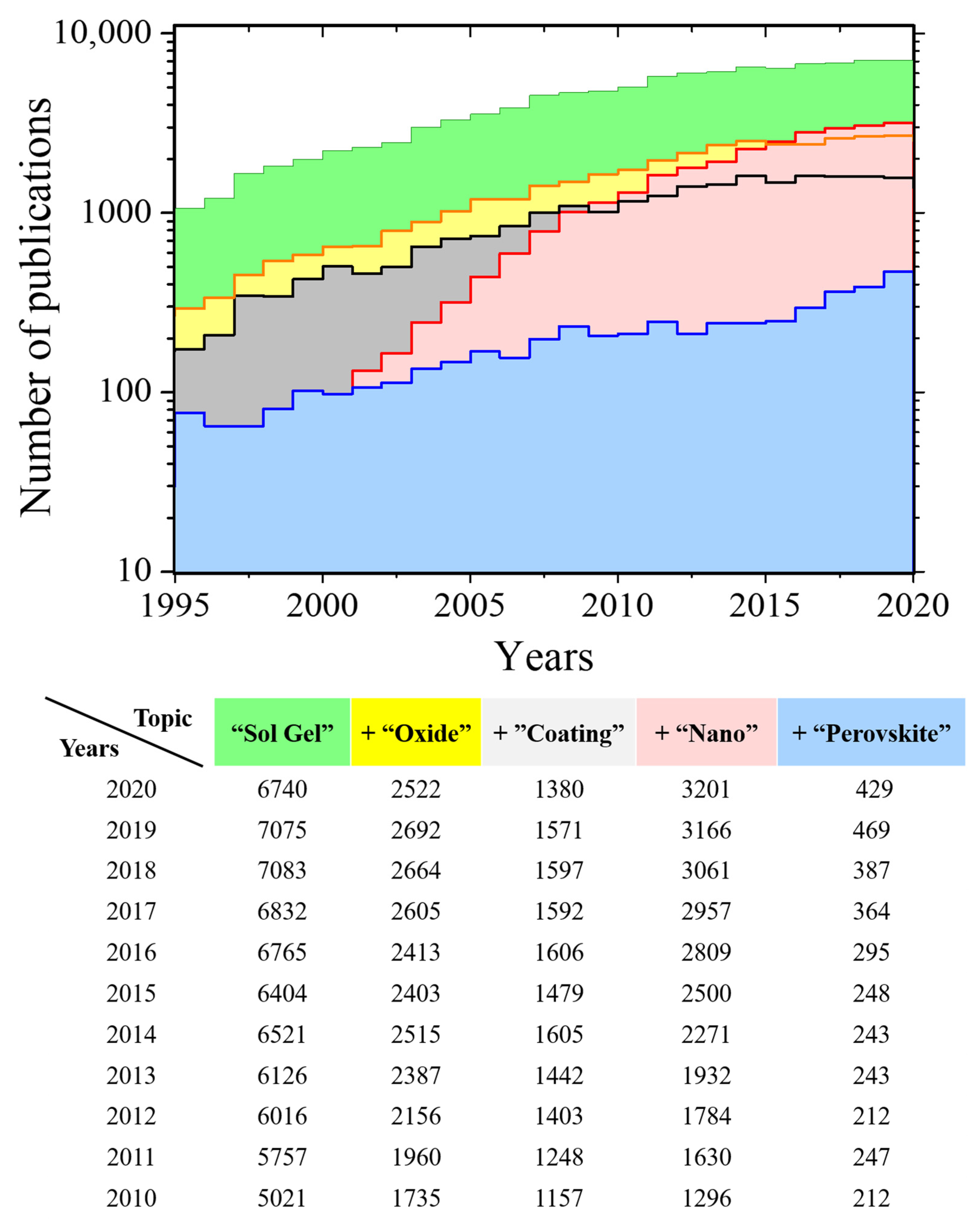
1. Introduction
- Possibility of obtaining special products such as powders, films or coatings, microspheres, fibers;
- Obtaining new solids with improved properties;
- High purity and homogeneity of the materials obtained;
- Saving energy during the process;
- Full control over the particle size and morphology;
- The solution and reaction step allows to incorporate easily, uniformly and quantitatively some trace elements, achieving a uniform doping at the molecular level.
- Compared with the solid-phase reaction, the chemical reaction is simpler and only requires a lower synthesis temperature (<220 °C).
- Very sensible to moisture;
- Difficult to scale up;
- Can include several steps and is a time-consuming process;
- Dimension and volume changes during different steps.
2. Synthesis of Perovskites
2.1. Sol-Gel Synthesis of Strontium and Barium Titanates and Its Derivate
2.2. Synthesis of LaMnO3 Nanoparticles
3. Synthesis of Vanadium Oxides
4. Synthesis of Zinc Oxides
5. Bioorganic Material in Sol-Gel Preparation
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Danks, A.E.; Hall, S.R.; Schnepp, Z. The evolution of ‘sol–gel’ chemistry as a technique for materials synthesis. Mater. Horizons 2016, 3, 91–112. [Google Scholar] [CrossRef]
- Ye, C.-Q. Sol-Gel Processes of Functional Powders and Films. In Chemical Reactions in Inorganic Chemistry; IntechOpen: London, UK, 2018. [Google Scholar]
- Purcar, V.; Şomoghi, R.; Niţu, S.G.; Nicolae, C.-A.; Alexandrescu, E.; Gîfu, I.C.; Gabor, A.R.; Stroescu, H.; Ianchiş, R.; Căprărescu, S.; et al. The Effect of Different Coupling Agents on Nano-ZnO Materials Obtained via the Sol–Gel Process. Nanomaterials 2017, 7, 439. [Google Scholar] [CrossRef] [PubMed]
- Fu, M.; Li, Y.; Wu, S.; Lu, P.; Liu, J.; Dong, F. Sol–gel preparation and enhanced photocatalytic performance of Cu-doped ZnO nanoparticles. Appl. Surf. Sci. 2011, 258, 1587–1591. [Google Scholar] [CrossRef]
- Purcar, V.; Rădiţoiu, V.; Dumitru, A.; Nicolae, C.-A.; Frone, A.N.; Anastasescu, M.; Rădiţoiu, A.; Raduly, M.F.; Gabor, R.A.; Căprărescu, S. Antireflective coating based on TiO2 nanoparticles modified with coupling agents via acid-catalyzed sol-gel method. Appl. Surf. Sci. 2019, 487, 819–824. [Google Scholar] [CrossRef]
- Spataru, C.I.; Purcar, V.; Ghiurea, M.; Radovici, C.; Stanga, G.; Donescu, D. Effects of the nanoassociation of hexadecyltrimethoxysilane precursors on the sol–gel process. J. Sol-Gel Sci. Technol. 2013, 65, 344–352. [Google Scholar] [CrossRef]
- Znaidi, L. Sol–gel-deposited ZnO thin films: A review. Mater. Sci. Eng. B 2010, 174, 18–30. [Google Scholar] [CrossRef]
- Ebelmen, M. Sur les combinations des acides borique et silicique avec les élhers. Ann. Chim. Phys. 1846, 16, 129. [Google Scholar]
- Graham, T. XXXV.—On the properties of silicic acid and other analogous colloidal substances. J. Chem. Soc. 1864, 17, 318–327. [Google Scholar] [CrossRef]
- Ostwald, W. Studien über die Bildung und Umwandlung fester Körper. Z. Phys. Chem. 1897, 22U, 289–330. [Google Scholar] [CrossRef]
- Rayleigh, L. LXXVIII. Periodic precipitates. Lond. Edinb. Dublin Philos. Mag. J. Sci. 1919, 38, 738–740. [Google Scholar] [CrossRef]
- Liesegang, R.E. Ueber einige Eigenschaften von Gallerten. Z. Chem. Ind. Kolloide 1896, 11, 212, Reprint: Liesegang, R.E. Ueber einige Eigenschaften von Gallerten. Z. Chem. Ind. Kolloide 1907, 1, 212. [Google Scholar] [CrossRef]
- Roy, R.; Osborn, E.F. The system Al2O3-SiO2-H2O. Am. Mineral. 1954, 39, 853–885. [Google Scholar]
- Roy, R. Ceramics by the Solution-Sol-Gel Route. Science 1987, 238, 1664–1669. [Google Scholar] [CrossRef] [PubMed]
- Dislich, H. New Routes to Multicomponent Oxide Glasses. Angew. Chem. Int. Ed. 1971, 10, 363–370. [Google Scholar] [CrossRef]
- Sakka, S. History of the Sol–Gel Chemistry and Technology. In Handbook of Sol-Gel Science and Technology; Springer: Singapore, 2016; pp. 1–27. [Google Scholar]
- Web of Science. Available online: https://www.webofscience.com (accessed on 1 December 2021).
- Parashar, M.; Shukla, V.K.; Singh, R. Metal oxides nanoparticles via sol–gel method: A review on synthesis, characterization and applications. J. Mater. Sci. Mater. Electron. 2020, 31, 3729–3749. [Google Scholar] [CrossRef]
- Issa, A.A.; El-Azazy, M.; Luyt, A.S. Kinetics of alkoxysilanes hydrolysis: An empirical approach. Sci. Rep. 2019, 9, 17624. [Google Scholar] [CrossRef]
- Athayde, D.; Souza, D.F.; Silva, A.; Vasconcelos, D.; Nunes, E.H.; da Costa, J.C.D.; Vasconcelos, W. Review of perovskite ceramic synthesis and membrane preparation methods. Ceram. Int. 2016, 42, 6555–6571. [Google Scholar] [CrossRef]
- Pechini, M.P. Patent: Method of Preparing Lead and Alkaline Earth Titanates and Niobates and Coating Method Using the Same to Form a Capacitor. Available online: https://patents.google.com/patent/US3330697A/en (accessed on 3 November 2021).
- Kurajica, S. A Brief Review on the Use of Chelation Agents in Sol-gel Synthesis with Emphasis on β-Diketones and β-Ketoesters. Chem. Biochem. Eng. Q. 2019, 33, 295–301. [Google Scholar] [CrossRef]
- Shlapa, Y.; Solopan, S.; Belous, A. Nanoparticles of La1-Sr MnO3 (0.23 ≤ x ≤ 0.25) manganite: Features of synthesis and crystallochemical properties. J. Magn. Magn. Mater. 2020, 510, 166902. [Google Scholar] [CrossRef]
- Wang, G.; Jia, G.; Wang, J.; Kong, H.; Lu, Y.; Zhang, C. Novel rare earth activator ions-doped perovskite-type La4Ti3O12 phosphors: Facile synthesis, structure, multicolor emissions, and potential applications. J. Alloys Compd. 2021, 877, 160217. [Google Scholar] [CrossRef]
- Garcia, A.; Bispo, A., Jr.; Lima, S.; Pires, A. Effects of the Pechini’s modified synthetic route on structural and photophysical properties of Eu3+ or Tb3+-doped LaAlO3. Mater. Res. Bull. 2021, 143, 111462. [Google Scholar] [CrossRef]
- Osman, N.; Samat, A.A.; Mat, A.N.C.; Malek, N.I.A.; Andas, J. Chelating agent size effect on thermal decomposition, phase formation and morphology characteristics of Y3+ doped Ba(Ce,Zr)O3 ceramics powder prepared by a sol-gel process. Ceram. Int. 2021, 48, 2289–2297. [Google Scholar] [CrossRef]
- Tewatia, K.; Sharma, A.; Sharma, M.; Kumar, A. Factors affecting morphological and electrical properties of Barium Titanate: A brief review. Mater. Today Proc. 2021, 44, 4548–4556. [Google Scholar] [CrossRef]
- Santander-Syro, A.F.; Copie, O.; Kondo, T.; Fortuna, F.; Pailhès, S.; Weht, R.; Qiu, X.G.; Bertran, F.; Nicolaou, A.; Taleb-Ibrahimi, A.; et al. Two-dimensional electron gas with universal subbands at the surface of SrTiO3. Nat. Cell Biol. 2011, 469, 189–193. [Google Scholar] [CrossRef]
- Pai, Y.-Y.; Tylan-Tyler, A.; Irvin, P.; Levy, J. Physics of SrTiO3-based heterostructures and nanostructures: A review. Rep. Prog. Phys. 2017, 81, 036503. [Google Scholar] [CrossRef]
- Szafraniak, B.; Fuśnik, Ł.; Xu, J.; Gao, F.; Brudnik, A.; Rydosz, A. Semiconducting Metal Oxides: SrTiO3, BaTiO3 and BaSrTiO3 in Gas-Sensing Applications: A Review. Coatings 2021, 11, 185. [Google Scholar] [CrossRef]
- Shi, X.; Wu, H.; Liu, Q.; Zhou, W.; Lu, S.; Shao, Z.; Dargusch, M.; Chen, Z.-G. SrTiO3-based thermoelectrics: Progress and challenges. Nano Energy 2020, 78, 105195. [Google Scholar] [CrossRef]
- Ji, X.; Zhu, Y.; Lian, X.; Fan, B.; Liu, X.; Xiao, P.; Zhang, Y. Hydroxylation mechanism of phase regulation of nanocrystal BaTiO3 synthesized by a hydrothermal method. Ceram. Int. 2021, 48, 2281–2288. [Google Scholar] [CrossRef]
- Kholodkova, A.; Danchevskaya, M.; Ivakin, Y.; Smirnov, A.; Ponomarev, S.; Fionov, A.; Kolesov, V. Solid state synthesis of barium titanate in air and in supercritical water: Properties of powder and ceramics. Ceram. Int. 2019, 45, 23050–23060. [Google Scholar] [CrossRef]
- Prado, L.R.; de Resende, N.; Silva, R.; Egues, S.M.; Banda, G. Influence of the synthesis method on the preparation of barium titanate nanoparticles. Chem. Eng. Process. Process. Intensif. 2016, 103, 12–20. [Google Scholar] [CrossRef]
- Kržmanc, M.M.; Klement, D.; Jančar, B.; Suvorov, D. Hydrothermal conditions for the formation of tetragonal BaTiO3 particles from potassium titanate and barium salt. Ceram. Int. 2015, 41, 15128–15137. [Google Scholar] [CrossRef]
- Habib, A.; Haubner, R.; Stelzer, N. Effect of temperature, time and particle size of Ti precursor on hydrothermal synthesis of barium titanate. Mater. Sci. Eng. B 2008, 152, 60–65. [Google Scholar] [CrossRef]
- Surmenev, R.; Chernozem, R.; Skirtach, A.; Bekareva, A.; Leonova, L.; Mathur, S.; Ivanov, Y.F.; Surmeneva, M. Hydrothermal synthesis of barium titanate nano/microrods and particle agglomerates using a sodium titanate precursor. Ceram. Int. 2021, 47, 8904–8914. [Google Scholar] [CrossRef]
- Özen, M.; Mertens, M.; Snijkers, F.; Cool, P. Template-free aqueous tape casting of hydrothermally synthesized barium titanate powder and the fabrication of highly {001}-{100} textured tapes. Ceram. Int. 2018, 44, 9720–9727. [Google Scholar] [CrossRef]
- Dunne, P.W.; Starkey, C.L.; Munn, A.S.; Tang, S.V.; Luebben, O.; Shvets, I.; Ryder, A.G.; Casamayou-Boucau, Y.; Morrison, L.; Lester, E.H. Bench- and pilot-scale continuous-flow hydrothermal production of barium strontium titanate nanopowders. Chem. Eng. J. 2016, 289, 433–441. [Google Scholar] [CrossRef]
- Magnone, E.; Kim, J.R.; Park, J.H. The effect of the hydrothermal synthesis variables on barium titanate powders. Ceram. Int. 2016, 42, 10030–10036. [Google Scholar] [CrossRef]
- Lee, H.-W.; Moon, S.; Choi, C.-H.; Kim, D.K. Synthesis and Size Control of Tetragonal Barium Titanate Nanopowders by Facile Solvothermal Method. J. Am. Ceram. Soc. 2012, 95, 2429–2434. [Google Scholar] [CrossRef]
- Moreno, J.; Dominguez, J.M.; Vicente, L. Synthesis and characterization of MTiO3(M = Mg, Ca, Sr, Ba) Sol-Gel. J. Mater. Chem. 1995, 5, 509–512. [Google Scholar] [CrossRef]
- Fuentes, S.; Zárate, R.; Chávez, E.; Muñoz, P.; Ayala, M.; Espinoza-González, R.; Leyton, P. Synthesis and characterization of BaTiO3 nanoparticles in oxygen atmosphere. J. Alloys Compd. 2010, 505, 568–572. [Google Scholar] [CrossRef]
- Demirörs, A.; Imhof, A. BaTiO3, SrTiO3, CaTiO3, and BaxSr1−xTiO3 Particles: A General Approach for Monodisperse Colloidal Perovskites. Chem. Mater. 2009, 21, 3002–3007. [Google Scholar] [CrossRef]
- Zhang, X.; Cui, B.; Wang, J.; Jin, Q. The effect of a barium titanate xerogel precursor on the grain size and densification of fine-grained BaTiO3 ceramics. Ceram. Int. 2019, 45, 10626–10632. [Google Scholar] [CrossRef]
- Fuentes, S.; Muñoz, P.; Barraza, N.; Chávez-Ángel, E.; Torres, C.M.S. Structural characterisation of slightly Fe-doped SrTiO3 grown via a sol–gel hydrothermal synthesis. J. Sol-Gel Sci. Technol. 2015, 75, 593–601. [Google Scholar] [CrossRef]
- Fuentes, S.; Chávez-Ángel, E.; Padilla-Campos, L.; Diaz-Droguett, D. Influence of reactant type on the Sr incorporation grade and structural characteristics of Ba1−xSrxTiO3 (x = 0–1) grown by sol–gel-hydrothermal synthesis. Ceram. Int. 2013, 39, 8823–8831. [Google Scholar] [CrossRef]
- Fuentes, S.; Espinoza-González, R.; Rosales, M.; León, J. Effects of Eu3+ on the morphological, structural and optical properties of BaTiO3@ZnO:Eu nanoparticles. J. Alloys Compd. 2020, 846, 156452. [Google Scholar] [CrossRef]
- Yin, X.; Wang, S.; Wang, B.; Shen, L. Perovskite-type LaMn1−xBxO3+δ (B = Fe, CO and Ni) as oxygen carriers for chemical looping steam methane reforming. Chem. Eng. J. 2021, 422, 128751. [Google Scholar] [CrossRef]
- Yin, X.; Shen, L.; Wang, S.; Wang, B.; Shen, C. Double adjustment of Co and Sr in LaMnO3+δ perovskite oxygen carriers for chemical looping steam methane reforming. Appl. Catal. B Environ. 2021, 301, 120816. [Google Scholar] [CrossRef]
- Mohamed, A.E.-M.A.; Álvarez-Alonso, P.; Hernando, B. The intrinsic exchange bias effect in the LaMnO3 and LaFeO3 compounds. J. Alloys Compd. 2021, 850, 156713. [Google Scholar] [CrossRef]
- Li, Y.; Xue, L.; Fan, L.; Yan, Y. The effect of citric acid to metal nitrates molar ratio on sol–gel combustion synthesis of nanocrystalline LaMnO3 powders. J. Alloys Compd. 2009, 478, 493–497. [Google Scholar] [CrossRef]
- Chen, Y.; Ma, J.; Li, D.; Zhu, C.; Zhang, W.; Miao, C. LaMnO3 nanocomposite double network hydrogel electrodes with enhanced electrochemical and mechanical performance for flexible supercapacitors. J. Alloys Compd. 2021, 888, 161555. [Google Scholar] [CrossRef]
- Li, C.; Yu, Z.; Liu, H.; Chen, K. High surface area LaMnO3 nanoparticles enhancing electrochemical catalytic activity for rechargeable lithium-air batteries. J. Phys. Chem. Solids 2018, 113, 151–156. [Google Scholar] [CrossRef]
- Shaterian, M.; Enhessari, M.; Rabbani, D.; Asghari, M.; Salavati-Niasari, M. Synthesis, characterization and photocatalytic activity of LaMnO3 nanoparticles. Appl. Surf. Sci. 2014, 318, 213–217. [Google Scholar] [CrossRef]
- Calvo, J.A.O.; Ayo, B.P.; De La Torre, U.; González-Velasco, J.R. Key factors in Sr-doped LaBO3 (B = Co or Mn) perovskites for NO oxidation in efficient diesel exhaust purification. Appl. Catal. B Environ. 2017, 213, 198–210. [Google Scholar] [CrossRef]
- Du, D.; Zheng, R.; He, M.; Zhao, C.; Zhou, B.; Li, R.; Xu, H.; Wen, X.; Zeng, T.; Shu, C. A-site cationic defects induced electronic structure regulation of LaMnO3 perovskite boosts oxygen electrode reactions in aprotic lithium–oxygen batteries. Energy Storage Mater. 2021, 43, 293–304. [Google Scholar] [CrossRef]
- Yoo, H.D.; Jokisaari, J.R.; Yu, Y.-S.; Kwon, B.J.; Hu, L.; Kim, S.; Han, S.-D.; Lopez, M.; Lapidus, S.H.; Nolis, G.M.; et al. Intercalation of Magnesium into a Layered Vanadium Oxide with High Capacity. ACS Energy Lett. 2019, 4, 1528–1534. [Google Scholar] [CrossRef]
- Cestarolli, D.T.; Guerra, E.M. Vanadium Pentoxide (V2O5): Their Obtaining Methods and Wide Applications. In Transition Metal Compounds-Synthesis, Properties, and Application; IntechOpen: London, UK, 2021. [Google Scholar]
- Chandrappa, G.; Steunou, N.; Cassaignon, S.; Bauvais, C.; Biswas, P.; Livage, J. Vanadium Oxide: From Gels to Nanotubes. J. Sol-Gel Sci. Technol. 2003, 26, 593–596. [Google Scholar] [CrossRef]
- O’Dwyer, C.; Lavayen, V.; Fuenzalida, D.; Newcomb, S.B.; Ana, M.A.S.; Benavente, E.; González, G.; Torres, C.M.S. Six-fold rotationally symmetric vanadium oxide nanostructures by a morphotropic phase transition. Phys. Status Solidi (b) 2007, 244, 4157–4160. [Google Scholar] [CrossRef]
- Shao, J.; Li, X.; Qu, Q.; Zheng, H. One-step hydrothermal synthesis of hexangular starfruit-like vanadium oxide for high power aqueous supercapacitors. J. Power Sources 2012, 219, 253–257. [Google Scholar] [CrossRef]
- Chernova, N.A.; Roppolo, M.; Dillon, A.C.; Whittingham, M.S. Layered vanadium and molybdenum oxides: Batteries and electrochromics. J. Mater. Chem. 2009, 19, 2526–2552. [Google Scholar] [CrossRef]
- Perera, S.D.; Patel, B.; Bonso, J.; Grunewald, M.; Ferraris, J.P.; Balkus, K.J. Vanadium Oxide Nanotube Spherical Clusters Prepared on Carbon Fabrics for Energy Storage Applications. ACS Appl. Mater. Interfaces 2011, 3, 4512–4517. [Google Scholar] [CrossRef]
- O’Dwyer, C.; Navas, D.; Lavayen, V.; Benavente, E.; Santa Ana, M.A.; Gonzalez, G.; Newcomb, S.B.; Sotomayor Torres, C.M. Nano-Urchin: The Formation and Structure of High-Density Spherical Clusters of Vanadium Oxide Nanotubes. Chem. Mater. 2006, 18, 3016–3022. [Google Scholar] [CrossRef]
- Ma, Y.; Wu, M.; Jin, X.; Shu, R.; Hu, C.; Xu, T.; Li, J.; Meng, X.; Cao, X. (NH4)2V7O16 Microbricks as a Novel Anode for Aqueous Lithium-Ion Battery with Good Cyclability. Chem.-A Eur. J. 2021, 27, 12341–12351. [Google Scholar] [CrossRef]
- Heo, J.W.; Bu, H.; Hyoung, J.; Hong, S.-T. Ammonium Vanadium Bronze, (NH4)2V7O16, as a New Lithium Intercalation Host Material. Inorg. Chem. 2020, 59, 4320–4327. [Google Scholar] [CrossRef] [PubMed]
- Navas, D.; Donoso, J.P.; Magon, C.; Sotomayor-Torres, C.M.; Moreno, M.; Lozano, H.; Benavente, E.; González, G. Ammonium hexadeca-oxo-heptavanadate microsquares. A new member in the family of the V7O16 mixed-valence nanostructures. New J. Chem. 2019, 43, 17548–17556. [Google Scholar] [CrossRef]
- Sabir, S.; Arshad, M.; Chaudhari, S.K. Zinc Oxide Nanoparticles for Revolutionizing Agriculture: Synthesis and Applications. Sci. World J. 2014, 2014, 1–8. [Google Scholar] [CrossRef]
- Narayana, A.; Bhat, S.A.; Fathima, A.; Lokesh, S.V.; Surya, S.G.; Yelamaggad, C.V. Green and low-cost synthesis of zinc oxide nanoparticles and their application in transistor-based carbon monoxide sensing. RSC Adv. 2020, 10, 13532–13542. [Google Scholar] [CrossRef]
- Shaba, E.Y.; Jacob, J.O.; Tijani, J.O.; Suleiman, M.A.T. A critical review of synthesis parameters affecting the properties of zinc oxide nanoparticle and its application in wastewater treatment. Appl. Water Sci. 2021, 11, 1–41. [Google Scholar] [CrossRef]
- Di Marcantonio, M.; Gellner, S.; Namanga, J.E.; Frohleiks, J.; Gerlitzki, N.; Vollkommer, F.; Bacher, G.; Nannen, E. Performance Enhancement by ZnO Nanoparticle Layer in Hybrid Ionic Transition Metal Complex-Light-Emitting Electrochemical Cells (iTMC-LECs). Adv. Mater. Technol. 2017, 2, 1600215. [Google Scholar] [CrossRef]
- Wibowo, A.; Marsudi, M.A.; Amal, M.I.; Ananda, M.B.; Stephanie, R.; Ardy, H.; Diguna, L.J. ZnO nanostructured materials for emerging solar cell applications. RSC Adv. 2020, 10, 42838–42859. [Google Scholar] [CrossRef]
- Dodoo-Arhin, D.; Asiedu, T.; Agyei-Tuffour, B.; Nyankson, E.; Obada, D.; Mwabora, J. Photocatalytic degradation of Rhodamine dyes using zinc oxide nanoparticles. Mater. Today Proc. 2020, 38, 809–815. [Google Scholar] [CrossRef]
- Kołodziejczak-Radzimska, A.; Jesionowski, T. Zinc Oxide—From Synthesis to Application: A Review. Materials 2014, 7, 2833–2881. [Google Scholar] [CrossRef] [PubMed]
- Aal, N.A.; Al-Hazmi, F.; Al-Ghamdi, A.A.; A Alghamdi, A.; El-Tantawy, F.; Yakuphanoglu, F. Novel rapid synthesis of zinc oxide nanotubes via hydrothermal technique and antibacterial properties. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 135, 871–877. [Google Scholar] [CrossRef]
- Manikandan, B.; Endo, T.; Kaneko, S.; Murali, K.R.; John, R. Properties of sol gel synthesized ZnO nanoparticles. J. Mater. Sci. Mater. Electron. 2018, 29, 9474–9485. [Google Scholar] [CrossRef]
- Galdámez-Martinez, A.; Santana, G.; Güell, F.; Martínez-Alanis, P.R.; Dutt, A. Photoluminescence of ZnO Nanowires: A Review. Nanomater. 2020, 10, 857. [Google Scholar] [CrossRef] [PubMed]
- Meenakshi, G.; Sivasamy, A. Synthesis and characterization of zinc oxide nanorods and its photocatalytic activities towards degradation of 2,4-D. Ecotoxicol. Environ. Saf. 2017, 135, 243–251. [Google Scholar] [CrossRef]
- Navas, D.; Ibañez, A.; González, I.; Palma, J.L.; Dreyse, P. Controlled dispersion of ZnO nanoparticles produced by basic precipitation in solvothermal processes. Heliyon 2020, 6, e05821. [Google Scholar] [CrossRef]
- Nagar, A.; Kumar, A.; Parveen, S.; Kumar, A.; Dhasmana, H.; Husain, S.; Verma, A.; Jain, V. Zinc oxide nanoflowers synthesized by sol-gel technique for field emission displays (FEDs). Mater. Today Proc. 2020, 32, 402–406. [Google Scholar] [CrossRef]
- Kiriarachchi, H.D.; Abouzeid, K.M.; Bo, L.; El-Shall, M.S. Growth Mechanism of Sea Urchin ZnO Nanostructures in Aqueous Solutions and Their Photocatalytic Activity for the Degradation of Organic Dyes. ACS Omega 2019, 4, 14013–14020. [Google Scholar] [CrossRef]
- Cheng, J.H.; Xu, S.; Ding, C.H. Uniform Nano/Micron-Sized ZnO Spheres with Controllable Diameter. Adv. Mater. Res. 2012, 496, 268–271. [Google Scholar] [CrossRef]
- Pourrahimi, A.M.; Liu, D.; Pallon, L.K.H.; Andersson, R.L.; Abad, A.M.; Lagarón, J.-M.; Hedenqvist, M.S.; Ström, V.; Gedde, U.W.; Olsson, R.T. Water-based synthesis and cleaning methods for high purity ZnO nanoparticles—comparing acetate, chloride, sulphate and nitrate zinc salt precursors. RSC Adv. 2014, 4, 35568–35577. [Google Scholar] [CrossRef]
- Samei, M.; Sarrafzadeh, M.-H.; Faramarzi, M.A. The impact of morphology and size of zinc oxide nanoparticles on its toxicity to the freshwater microalga, Raphidocelis subcapitata. Environ. Sci. Pollut. Res. 2018, 26, 2409–2420. [Google Scholar] [CrossRef]
- Anand, K.; Varghese, S.; Krishnamoorthy, A. Role of surfactants on the stability of nano-zinc oxide dispersions. Part. Sci. Technol. 2015, 35, 67–70. [Google Scholar] [CrossRef]
- Wu, P.-Y.; Pike, J.; Zhang, F.; Chan, S.-W. Low-Temperature Synthesis of Zinc Oxide Nanoparticles. Int. J. Appl. Ceram. Technol. 2006, 3, 272–278. [Google Scholar] [CrossRef]
- Chieng, B.W.; Loo, Y.Y. Synthesis of ZnO nanoparticles by modified polyol method. Mater. Lett. 2012, 73, 78–82. [Google Scholar] [CrossRef]
- Karak, N.; Barik, P.; Kundu, T.K. Visible luminescence from triethanolamine-modified zinc oxide nanoparticles. In AIP Conference Proceedings; American Institute of Physics: College Park, MD, USA, 2013; pp. 181–182. [Google Scholar]
- Tseng, Y.-K.; Chuang, M.-H.; Chen, Y.-C.; Wu, C.-H. Synthesis of 1D, 2D, and 3D ZnO Polycrystalline Nanostructures Using the Sol-Gel Method. J. Nanotechnol. 2012, 2012, 712850. [Google Scholar] [CrossRef]
- Haque, J.; Bellah, M.; Hassan, R.; Rahman, S. Synthesis of ZnO nanoparticles by two different methods & comparison of their structural, antibacterial, photocatalytic and optical properties. Nano Express 2020, 1, 010007. [Google Scholar] [CrossRef]
- Shokuhfar, A.; Samei, J.; Kandjani, A.E.; Vaezi, M.R. Synthesis of ZnO Nanoparticles via Sol-Gel Process Using Triethanolamine as a Novel Surfactant. Defect Diffus. Forum 2008, 273-276, 626–631. [Google Scholar] [CrossRef]
- Haque, A.; Mahalakshmi, S. Effect of Triethanolamine on Zinc Oxide Nanoparticles. Mater. Focus 2013, 2, 469–474. [Google Scholar] [CrossRef]
- Liu, X.; Wang, C.; Cai, B.; Xiao, X.; Guo, S.; Fan, Z.; Li, J.; Duan, X.; Liao, L. Rational Design of Amorphous Indium Zinc Oxide/Carbon Nanotube Hybrid Film for Unique Performance Transistors. Nano Lett. 2012, 12, 3596–3601. [Google Scholar] [CrossRef]
- Shao, D.; Wei, Q.; Zhang, L.; Cai, Y.; Jiang, S. Surface functionalization of carbon nanofibers by sol–gel coating of zinc oxide. Appl. Surf. Sci. 2008, 254, 6543–6546. [Google Scholar] [CrossRef]
- Han, T.Y.-J.; Worsley, M.A.; Baumann, T.F.; Satcher, J.J.H. Synthesis of ZnO coated activated carbon aerogel by simple sol–gel route. J. Mater. Chem. 2011, 21, 330–333. [Google Scholar] [CrossRef]
- Zhou, X.; Wu, G.; Wu, J.; Yang, H.; Wang, J.; Gao, G. Carbon black anchored vanadium oxide nanobelts and their post-sintering counterpart (V2O5 nanobelts) as high performance cathode materials for lithium ion batteries. Phys. Chem. Chem. Phys. 2014, 16, 3973–3982. [Google Scholar] [CrossRef]
- Zhou, X.; Wu, G.; Gao, G.; Cui, C.; Yang, H.; Shen, J.; Zhou, B.; Zhang, Z. The synthesis, characterization and electrochemical properties of Multi-Wall Carbon Nanotube-induced vanadium oxide nanosheet composite as a novel cathode material for lithium ion batteries. Electrochim. Acta 2012, 74, 32–38. [Google Scholar] [CrossRef]
- Kim, I.-H.; Kim, J.-H.; Cho, B.W.; Lee, Y.-H.; Kim, K.-B. Synthesis and Electrochemical Characterization of Vanadium Oxide on Carbon Nanotube Film Substrate for Pseudocapacitor Applications. J. Electrochem. Soc. 2006, 153, A989–A996. [Google Scholar] [CrossRef]
- Kumar, A. Sol gel synthesis of zinc oxide nanoparticles and their application as nano-composite electrode material for supercapacitor. J. Mol. Struct. 2020, 1220, 128654. [Google Scholar] [CrossRef]
- Bar-Cohen, Y. Biomimetics—using nature to inspire human innovation. Bioinspir. Biomim. 2006, 1, P1–P12. [Google Scholar] [CrossRef]
- Gao, F.; Qu, J.; Zhao, Z.; Wang, Z.; Qiu, J. Nitrogen-doped activated carbon derived from prawn shells for high-performance supercapacitors. Electrochim. Acta 2016, 190, 1134–1141. [Google Scholar] [CrossRef]
- Redondo, E.; Carretero-González, J.; Goikolea, E.; Ségalini, J.; Mysyk, R. Effect of pore texture on performance of activated carbon supercapacitor electrodes derived from olive pits. Electrochim. Acta 2015, 160, 178–184. [Google Scholar] [CrossRef]
- Li, B.; Dai, F.; Xiao, Q.; Yang, L.; Shen, J.; Zhang, C.; Cai, M. Nitrogen-doped activated carbon for a high energy hybrid supercapacitor. Energy Environ. Sci. 2016, 9, 102–106. [Google Scholar] [CrossRef]
- Wang, C.; Wang, X.; Lu, H.; Li, H.; Zhao, X. Cellulose-derived hierarchical porous carbon for high-performance flexible supercapacitors. Carbon 2018, 140, 139–147. [Google Scholar] [CrossRef]
- Maiti, U.N.; Lim, J.; Lee, K.E.; Lee, W.J.; Kim, S.O. Three-Dimensional Shape Engineered, Interfacial Gelation of Reduced Graphene Oxide for High Rate, Large Capacity Supercapacitors. Adv. Mater. 2013, 26, 615–619. [Google Scholar] [CrossRef]
- Cao, X.; Yin, Z.; Zhang, H. Three-dimensional graphene materials: Preparation, structures and application in supercapacitors. Energy Environ. Sci. 2014, 7, 1850–1865. [Google Scholar] [CrossRef]
- Yu, Z.; Tetard, L.; Zhai, L.; Thomas, J. Supercapacitor electrode materials: Nanostructures from 0 to 3 dimensions. Energy Environ. Sci. 2015, 8, 702–730. [Google Scholar] [CrossRef]
- Wen, P.; Gong, P.; Sun, J.; Wang, J.; Yang, S. Design and synthesis of Ni-MOF/CNT composites and rGO/carbon nitride composites for an asymmetric supercapacitor with high energy and power density. J. Mater. Chem. A 2015, 3, 13874–13883. [Google Scholar] [CrossRef]
- Ding, B.; Guo, D.; Wang, Y.; Wu, X.; Fan, Z. Functionalized graphene nanosheets decorated on carbon nanotubes networks for high performance supercapacitors. J. Power Sources 2018, 398, 113–119. [Google Scholar] [CrossRef]
- Biswal, M.; Banerjee, A.; Deo, M.; Ogale, S. From dead leaves to high energy density supercapacitors. Energy Environ. Sci. 2013, 6, 1249–1259. [Google Scholar] [CrossRef]
- Abioye, A.M.; Ani, F.N. Advancement in the production of activated carbon from biomass using microwave heating. J. Teknol. 2017, 79, 79–88. [Google Scholar] [CrossRef][Green Version]
- Lu, B.; Xiao, Z.; Zhu, H.; Xiao, W.; Wu, W.; Wang, D. Enhanced capacitive properties of commercial activated carbon by re-activation in molten carbonates. J. Power Sources 2015, 298, 74–82. [Google Scholar] [CrossRef]
- Hu, Y.; Tong, X.; Zhuo, H.; Zhong, L.; Peng, X. Biomass-Based Porous N-Self-Doped Carbon Framework/Polyaniline Composite with Outstanding Supercapacitance. ACS Sustain. Chem. Eng. 2017, 5, 8663–8674. [Google Scholar] [CrossRef]
- Yan, J.; Wang, Q.; Wei, T.; Fan, Z. Recent Advances in Design and Fabrication of Electrochemical Supercapacitors with High Energy Densities. Adv. Energy Mater. 2014, 4, 1300816. [Google Scholar] [CrossRef]
- Chen, X.; Paul, R.; Dai, L. Carbon-based supercapacitors for efficient energy storage. Natl. Sci. Rev. 2017, 4, 453–489. [Google Scholar] [CrossRef]
- Hao, P.; Zhao, Z.; Tian, J.; Li, H.; Sang, Y.; Yu, G.; Cai, H.; Liu, H.; Wong, C.P.; Umar, A. Hierarchical porous carbon aerogel derived from bagasse for high performance supercapacitor electrode. Nanoscale 2014, 6, 12120–12129. [Google Scholar] [CrossRef]
- Xu, M.; Yu, Q.; Liu, Z.; Lv, J.; Lian, S.; Hu, B.; Mai, L.; Zhou, L. Tailoring porous carbon spheres for supercapacitors. Nanoscale 2018, 10, 21604–21616. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Cai, Y.; Zhao, X.; Liang, Y.; Zheng, M.; Hu, H.; Dong, H.; Jiang, S.; Liu, Y.; Xiao, Y. Sulfur-doped nanoporous carbon spheres with ultrahigh specific surface area and high electrochemical activity for supercapacitor. J. Power Sources 2017, 360, 373–382. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, Y.; Ke, Q.; Ho, K.H.; Hu, Y.; Wang, J. Tuning the porous texture and specific surface area of nanoporous carbons for supercapacitor electrodes by adjusting the hydrothermal synthesis temperature. J. Mater. Chem. A 2013, 1, 12962–12970. [Google Scholar] [CrossRef]
- Wang, J.; Shen, L.; Ding, B.; Nie, P.; Deng, H.; Dou, H.; Zhang, X. Fabrication of porous carbon spheres for high-performance electrochemical capacitors. RSC Adv. 2014, 4, 7538–7544. [Google Scholar] [CrossRef]
- Hu, B.; Wang, K.; Wu, L.; Yu, S.-H.; Antonietti, M.; Titirici, M.-M. Engineering Carbon Materials from the Hydrothermal Carbonization Process of Biomass. Adv. Mater. 2010, 22, 813–828. [Google Scholar] [CrossRef] [PubMed]
- Pan, L.; Wang, Y.; Hu, H.; Li, X.; Liu, J.; Guan, L.; Tian, W.; Wang, X.; Li, Y.; Wu, M. 3D self-assembly synthesis of hierarchical porous carbon from petroleum asphalt for supercapacitors. Carbon 2018, 134, 345–353. [Google Scholar] [CrossRef]
- Liu, M.-C.; Lu, C.; Xu, Y.; Hu, Y.-X.; Li, J.; Zhang, H.; Zhang, Y.-S.; Zhang, B.-M.; Kong, L.-B.; Liu, W.-W.; et al. Three-Dimensional Interconnected Reticular Porous Carbon From Corn Starch By a Sample Sol–Gel Method Toward High-Performance Supercapacitors With Aqueous and Ionic Liquid Electrolytes. ACS Sustain. Chem. Eng. 2019, 7, 18690–18699. [Google Scholar] [CrossRef]
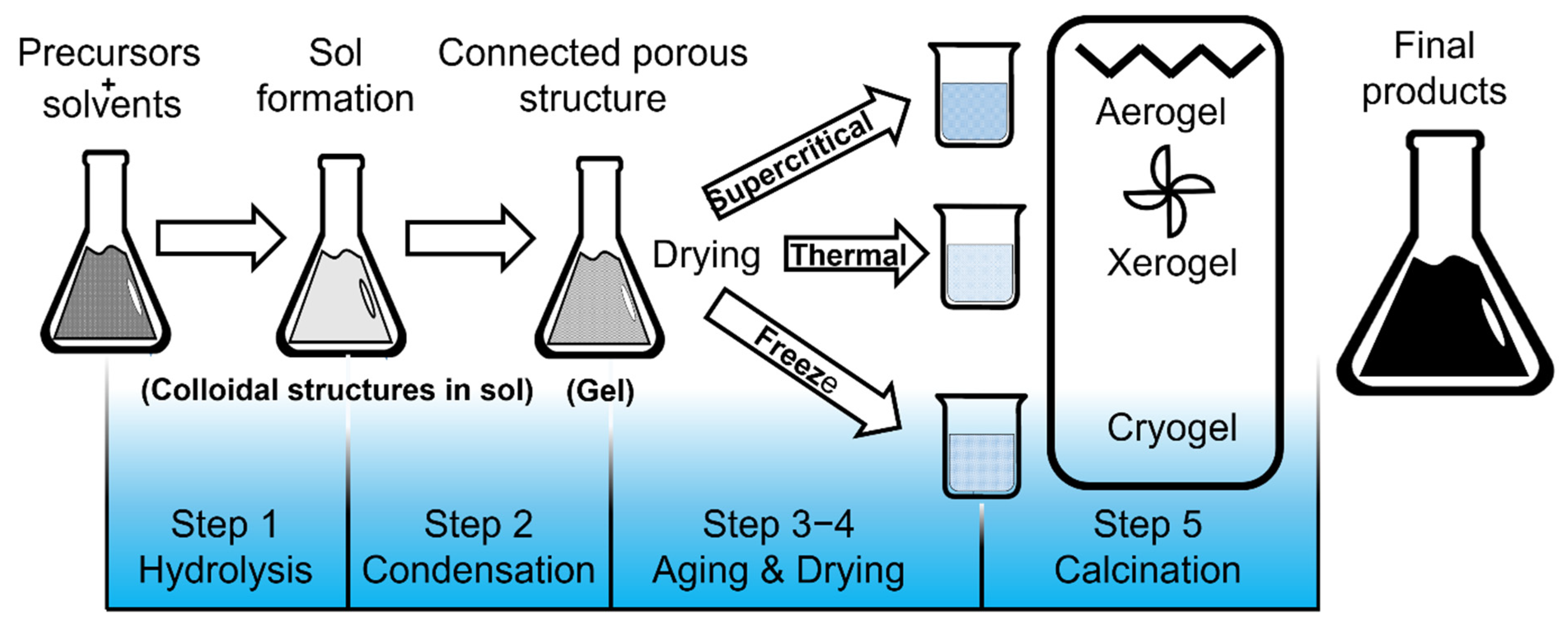
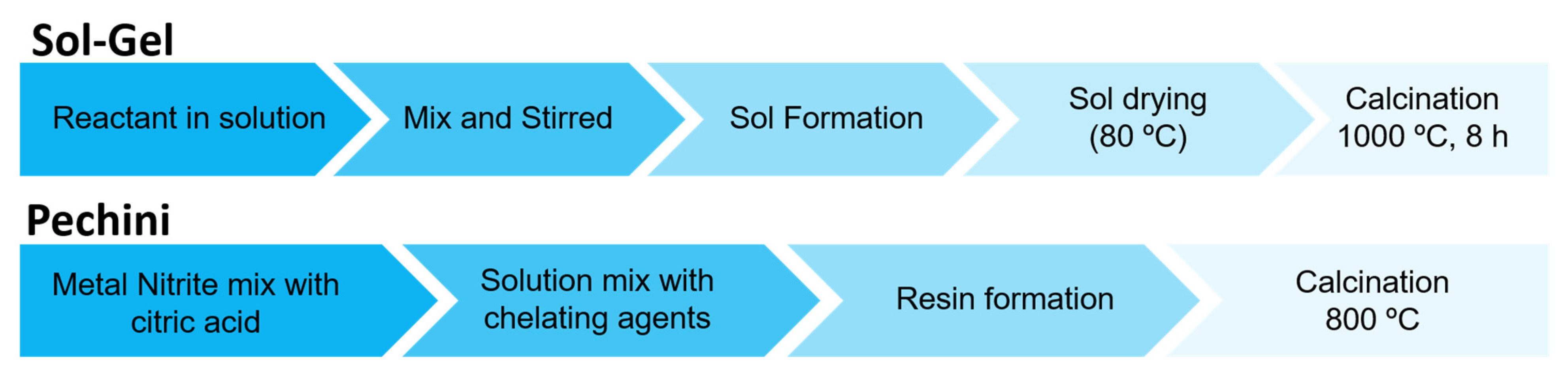





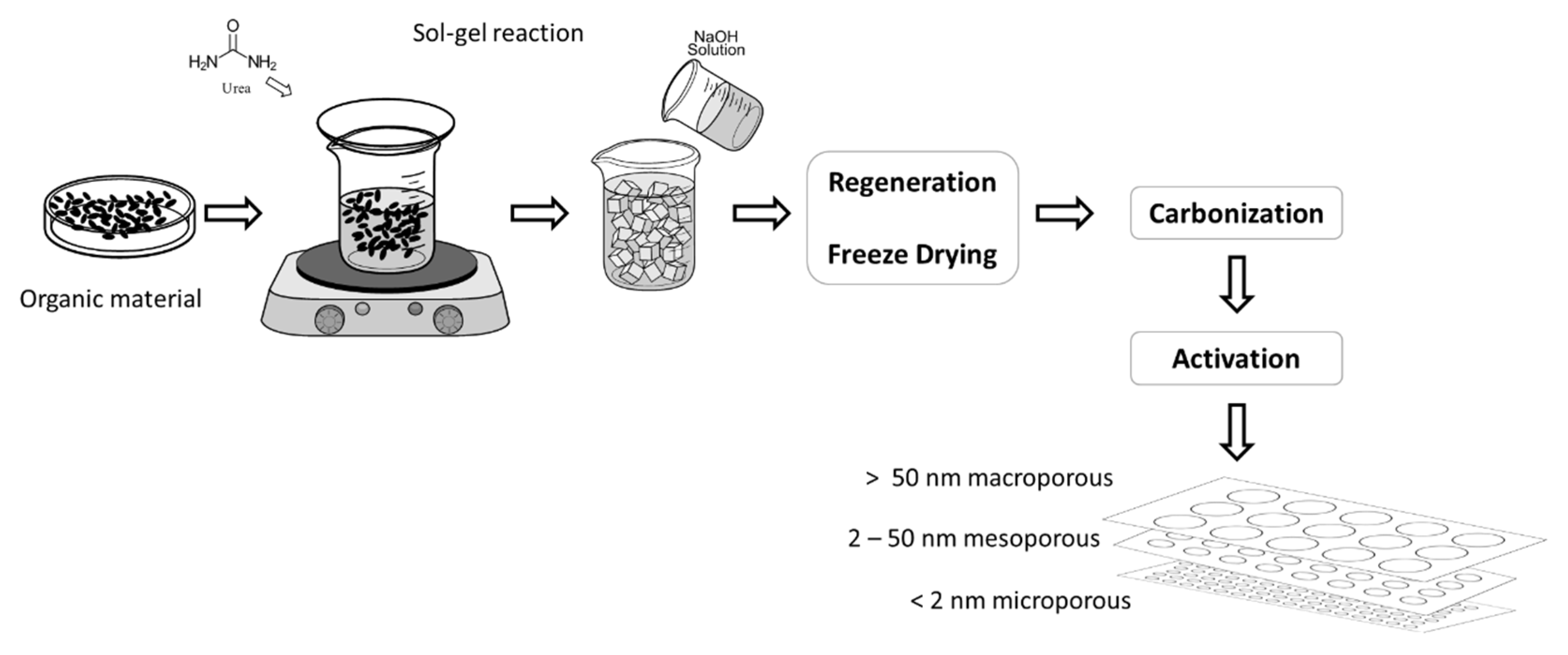

| Synthesis Methods | Particle Size | Agglomeration | Purity | Precursors | Calcination Temperature | Observations |
|---|---|---|---|---|---|---|
| Sol-Gel | >10 nm | Moderate | Excellent | Alkoxide or Acetylacetonates | 800 °C | Obtaining of uniform and small sized powders |
| Sol-Gel Pechini | >10 nm | Moderate | Excellent | Nitrates | 800–1000 °C | Accurate control of the final material composition |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Navas, D.; Fuentes, S.; Castro-Alvarez, A.; Chavez-Angel, E. Review on Sol-Gel Synthesis of Perovskite and Oxide Nanomaterials. Gels 2021, 7, 275. https://doi.org/10.3390/gels7040275
Navas D, Fuentes S, Castro-Alvarez A, Chavez-Angel E. Review on Sol-Gel Synthesis of Perovskite and Oxide Nanomaterials. Gels. 2021; 7(4):275. https://doi.org/10.3390/gels7040275
Chicago/Turabian StyleNavas, Daniel, Sandra Fuentes, Alejandro Castro-Alvarez, and Emigdio Chavez-Angel. 2021. "Review on Sol-Gel Synthesis of Perovskite and Oxide Nanomaterials" Gels 7, no. 4: 275. https://doi.org/10.3390/gels7040275
APA StyleNavas, D., Fuentes, S., Castro-Alvarez, A., & Chavez-Angel, E. (2021). Review on Sol-Gel Synthesis of Perovskite and Oxide Nanomaterials. Gels, 7(4), 275. https://doi.org/10.3390/gels7040275








