Dissolved Organic Matter in the Global Ocean: A Primer
Abstract
:1. Introduction
2. DOC Concentrations and Reactivity
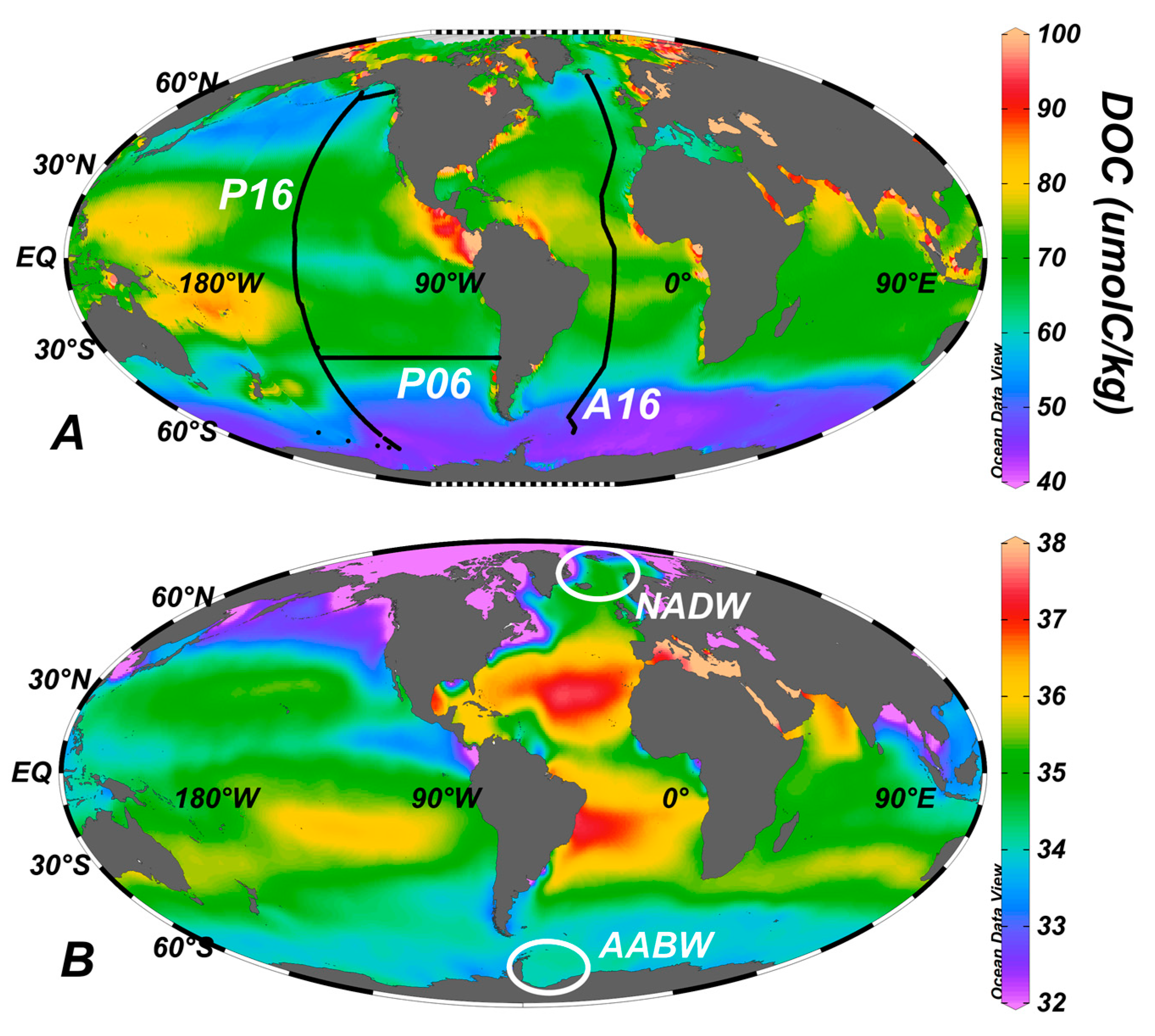
3. Global Distribution
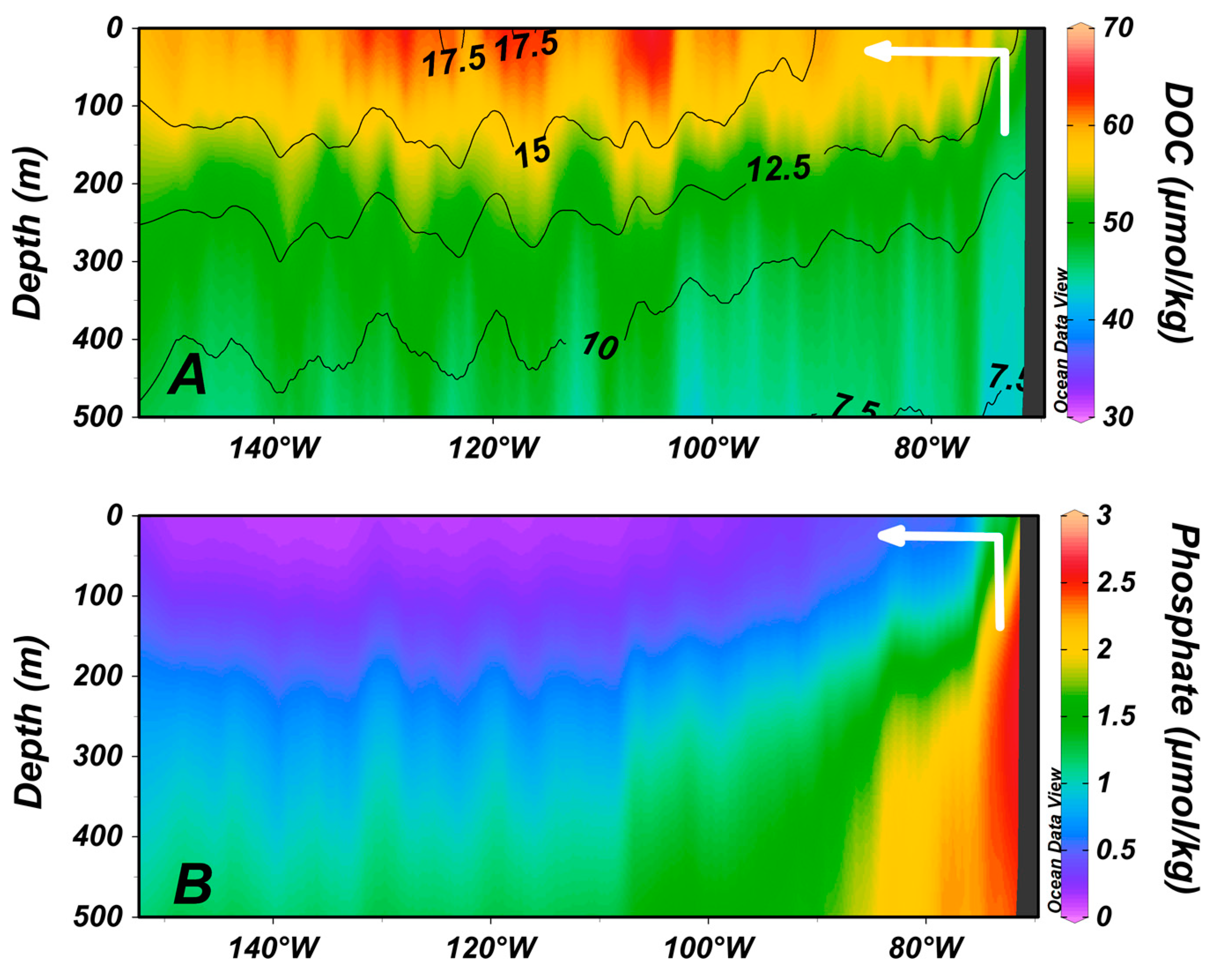
4. Zones of Net DOC Production
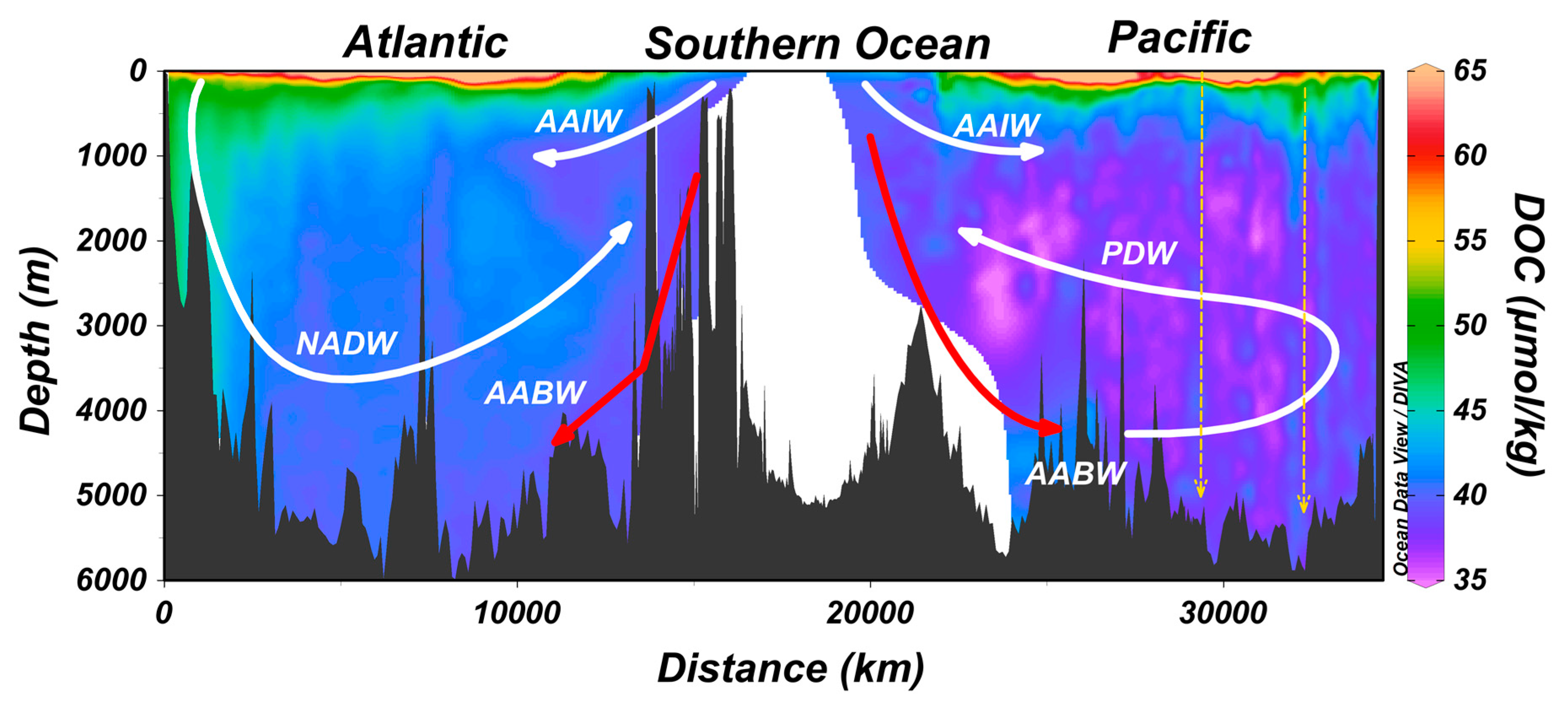
5. Export of Surface Accumulated DOC to Depth with the Ocean’s Overturning Circulation
6. Zones of Deep Ocean DOC Enrichment Due to Sinking Biogenic Particles
7. Composition of Ocean Dissolved Organic Matter
8. Closing with Consideration of Marine Gels
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Hansell, D.A.; Carlson, C.A.; Repeta, D.J.; Schlitzer, R. Dissolved organic matter in the ocean: A controversy stimulates new insights. Oceanography 2009, 22, 202–211. [Google Scholar] [CrossRef]
- Carlson, C.A.; Hansell, D.A. DOC sources, sinks and fluxes. In Biogeochemistry of Marine Dissolved Organic Matter, 2nd ed.; Elsevier: London, UK, 2014; pp. 65–126. [Google Scholar]
- Chin, W.C.; Orellana, M.V.; Verdugo, P. Spontaneous assembly of marine dissolved organic matter into polymer gels. Nature 1998, 391, 568–572. [Google Scholar] [CrossRef]
- Orellana, M.V.; Matrai, P.A.; Leck, C.; Rauschenberg, C.D.; Lee, A.M.; Coz, E. Marine microgels as a source of cloud condensation nuclei in the high Arctic. Proc. Natl. Acad. Sci. USA 2011, 108, 13612–13617. [Google Scholar] [CrossRef] [Green Version]
- Verdugo, P. Marine Microgels. In Annual Review of Marine Science; Annual Reviews: San Mateo, CA, USA, 2012; Volume 4, pp. 375–400. [Google Scholar]
- Orellana, M.V.; Leck, C. Marine Microgels. In Biogeochemistry of Marine Dissolved Organic Matter, 2nd ed.; Academic Press: Boston, MA, USA, 2015; pp. 451–480. [Google Scholar] [CrossRef]
- Hansell, D.A.; Carlson, C.A. Biogeochemistry of Marine Dissolved Organic Matter, 2nd ed.; Elsevier: Waltham, MA, USA, 2014. [Google Scholar]
- Hansell, D.A. Recalcitrant Dissolved Organic Carbon Fractions. In Annual Review of Marine Science; Annual Reviews: San Mateo, CA, USA, 2013; Volume 5, pp. 421–445. [Google Scholar]
- Lonborg, C.; Carreira, C.; Jickells, T.; Alvarez-Salgado, X.A. Impacts of global change on ocean dissolved organic carbon (DOC) cycling. Front. Mar. Sci. 2020, 7, 466. [Google Scholar] [CrossRef]
- Bauer, J.E.; Bianchi, T.S. Dissolved Organic Carbon Cycling and Transformation. In Treatise on Estuarine and Coastal Science; Wolanski, E., McLusky, D.S., Eds.; Academic Press: Waltham, MA, USA, 2011; Volume 5, pp. 7–67. [Google Scholar]
- Ogawa, H.; Tanoue, E. Dissolved organic matter in oceanic waters. J. Oceanogr. 2003, 59, 129–147. [Google Scholar] [CrossRef]
- Moran, M.A.; Kujawinski, E.B.; Stubbins, A.; Fatland, R.; Aluwihare, L.I.; Buchan, A.; Crump, B.C.; Dorrestein, P.C.; Dyhrman, S.T.; Hess, N.J.; et al. Deciphering ocean carbon in a changing world. Proc. Natl. Acad. Sci. USA 2016, 113, 3143–3151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wagner, S.; Schubotz, F.; Kaiser, K.; Hallmann, C.; Waska, H.; Rossel, P.E.; Hansmann, R.; Elvert, M.; Middelburg, J.J.; Engel, A.; et al. Soothsaying DOM: A current perspective on the future of oceanic dissolved organic carbon. Front. Mar. Sci. 2020, 7, 341. [Google Scholar] [CrossRef]
- Bauer, J.E.; Williams, P.M.; Druffel, E.R.M. C-14 Activity of dissolved organic carbon fractions in the North-central Pacific and Sargasso Sea. Nature 1992, 357, 667–670. [Google Scholar] [CrossRef]
- Roshan, S.; DeVries, T. Efficient dissolved organic carbon production and export in the oligotrophic ocean. Nat. Commun. 2017, 8, 8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schlitzer, R. Ocean Data View; Alfred Wegener Institute: Bremerhaven, Germany, 2021. [Google Scholar]
- Hansell, D.A.; Carlson, C.A.; Amon, R.M.W.; Álvarez-Salgado, X.A.; Yamashita, Y.; Romera-Castillo, C.; Bif, M.B. Compilation of Dissolved Organic Matter (DOM) Data Obtained from Global Ocean Observations from 1994 to 2020; NOAA National Centers for Environmental Information: Washington, DC, USA, 2021. [CrossRef]
- Hansell, D.A.; Carlson, C.A. Net community production of dissolved organic carbon. Glob. Biogeochem. Cycles 1998, 12, 443–453. [Google Scholar] [CrossRef]
- Bif, M.B.; Hansell, D.A. Seasonality of dissolved organic carbon in the upper northeast Pacific Ocean. Glob. Biogeochem. Cycles 2019, 33, 526–539. [Google Scholar] [CrossRef]
- Romera-Castillo, C.; Letscher, R.T.; Hansell, D.A. New nutrients exert fundamental control on dissolved organic carbon accumulation in the surface Atlantic Ocean. Proc. Natl. Acad. Sci. USA 2016, 113, 10497–10502. [Google Scholar] [CrossRef] [Green Version]
- Carlson, C.A.; Hansell, D.A.; Nelson, N.B.; Siegel, D.A.; Smethie, W.M.; Khatiwala, S.; Meyers, M.M.; Halewood, E. Dissolved organic carbon export and subsequent remineralization in the mesopelagic and bathypelagic realms of the North Atlantic basin. Deep-Sea Res. Part Ii-Top. Stud. Oceanogr. 2010, 57, 1433–1445. [Google Scholar] [CrossRef]
- Hansell, D.A.; Carlson, C.A.; Schlitzer, R. Net removal of major marine dissolved organic carbon fractions in the subsurface ocean. Glob. Biogeochem. Cycles 2012, 26, GB1016. [Google Scholar] [CrossRef]
- Baetge, N.; Graff, J.R.; Behrenfeld, M.J.; Carlson, C.A. Net community production, dissolved organic carbon accumulation, and vertical export in the western North Atlantic. Front. Mar. Sci. 2020, 7, 227. [Google Scholar] [CrossRef]
- Bercovici, S.K.; Huber, B.A.; DeJong, H.B.; Dunbar, R.B.; Hansell, D.A. Dissolved organic carbon in the Ross Sea: Deep enrichment and export. Limnol. Oceanogr. 2017, 62, 2593–2603. [Google Scholar] [CrossRef]
- Fontela, M.; Garcia-Ibanez, M.I.; Hansell, D.A.; Mercier, H.; Perez, F.F. Dissolved Organic Carbon in the North Atlantic Meridional Overturning Circulation. Sci. Rep. 2016, 6, 26931. [Google Scholar] [CrossRef] [Green Version]
- Hanawa, K.; Talley, L.D. Chapter 5.4 Mode waters. In International Geophysics; Siedler, G., Church, J., Gould, J., Eds.; Academic Press: Cambridge, MA, USA, 2001; Volume 77, pp. 373–386. [Google Scholar]
- Broek, T.A.B.; Walker, B.D.; Guilderson, T.P.; Vaughn, J.S.; Mason, H.E.; McCarthy, M.D. Low molecular weight dissolved organic carbon: Aging, compositional changes, and selective utilization during global ocean circulation. Glob. Biogeochem. Cycles 2020, 34, e2020GB006547. [Google Scholar] [CrossRef]
- Flerus, R.; Lechtenfeld, O.J.; Koch, B.P.; McCallister, S.L.; Schmitt-Kopplin, P.; Benner, R.; Kaiser, K.; Kattner, G. A molecular perspective on the ageing of marine dissolved organic matter. Biogeosciences 2012, 9, 1935–1955. [Google Scholar] [CrossRef] [Green Version]
- Bercovici, S.K.; Koch, B.P.; Lechtenfeld, O.J.; McCallister, S.L.; Schmitt-Kopplin, P.; Hansell, D.A. Aging and molecular changes of dissolved organic matter between two deep oceanic end-members. Glob. Biogeochem. Cycles 2018, 32, 1449–1456. [Google Scholar] [CrossRef]
- Turner, J.T. Zooplankton fecal pellets, marine snow, phytodetritus and the ocean’s biological pump. Prog. Oceanogr. 2015, 130, 205–248. [Google Scholar] [CrossRef]
- Noji, T.T.; Borsheim, K.Y.; Rey, F.; Nortvedt, R. Dissolved organic carbon associated with sinking particles can be crucial for estimates of vertical carbon flux. Sarsia 1999, 84, 129–135. [Google Scholar] [CrossRef]
- Orellana, M.V.; Hansell, D.A. Ribulose-1,5-bisphosphate carboxylase/oxygenase (RuBisCO): A long-lived protein in the deep ocean. Limnol. Oceanogr. 2012, 57, 826–834. [Google Scholar] [CrossRef] [Green Version]
- Collins, J.R.; Edwards, B.R.; Thamatrakoln, K.; Ossolinski, J.E.; DiTullio, G.R.; Bidle, K.D.; Doney, S.C.; Van Mooy, B.A.S. The multiple fates of sinking particles in the North Atlantic Ocean. Glob. Biogeochem. Cycles 2015, 29, 1471–1494. [Google Scholar] [CrossRef] [Green Version]
- Lopez, C.N.; Robert, M.; Galbraith, M.; Bercovici, S.K.; Orellana, M.V.; Hansell, D.A. High temporal variability of total organic carbon in the deep northeastern Pacific. Front. Earth Sci. 2020, 8, 80. [Google Scholar] [CrossRef]
- Repeta, D.J.; Aluwihare, L.I. Radiocarbon analysis of neutral sugars in high-molecular-weight dissolved organic carbon: Implications for organic carbon cycling. Limnol. Oceanogr. 2006, 51, 1045–1053. [Google Scholar] [CrossRef] [Green Version]
- Hansman, R.L.; Griffin, S.; Watson, J.T.; Druffel, E.R.M.; Ingalls, A.E.; Pearson, A.; Aluwihare, L.I. The radiocarbon signature of microorganisms in the mesopelagic ocean. Proc. Natl. Acad. Sci. USA 2009, 106, 6513–6518. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Repeta, D.J. Chemical Characterization and Cycling of Dissolved Organic Matter. In Biogeochemistry of Marine Dissolved Organic Matter, 2nd ed.; Elsevier: London, UK, 2014; pp. 21–63. [Google Scholar]
- Riedel, T.; Dittmar, T. A method detection limit for the analysis of natural organic matter via Fourier Transform Ion Cyclotron Resonance Mass Spectrometry. Anal. Chem. 2014, 86, 8376–8382. [Google Scholar] [CrossRef]
- Hertkorn, N.; Ruecker, C.; Meringer, M.; Gugisch, R.; Frommberger, M.; Perdue, E.M.; Witt, M.; Schmitt-Kopplin, P. High-precision frequency measurements: Indispensable tools at the core of the molecular-level analysis of complex systems. Anal. Bioanal. Chem. 2007, 389, 1311–1327. [Google Scholar] [CrossRef] [Green Version]
- Nebbioso, A.; Piccolo, A. Molecular characterization of dissolved organic matter (DOM): A critical review. Anal. Bioanal. Chem. 2013, 405, 109–124. [Google Scholar] [CrossRef] [PubMed]
- Baltar, F.; Alvarez-Salgado, X.A.; Aristegui, J.; Benner, R.; Hansell, D.A.; Herndl, G.J.; Lonborg, C. What Is refractory organic matter in the ocean? Front. Mar. Sci. 2021, 8, 327. [Google Scholar] [CrossRef]
- Arakawa, N.; Aluwihare, L.I.; Simpson, A.J.; Soong, R.; Stephens, B.M.; Lane-Coplen, D. Carotenoids are the likely precursor of a significant fraction of marine dissolved organic matter. Sci. Adv. 2017, 3, 11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tanaka, T.; Fillmore, D.; Sun, S.-T.; Nishio, I.; Swislow, G.; Shah, A. Phase transitions in ionic gels. Phys. Rev. Lett. 1980, 45, 1636–1639. [Google Scholar] [CrossRef]
- Shibayama, M.; Tanaka, T.; Han, C.C. Small angle neutron scattering study on poly(N-isopropyl acrylamide) gels near their volume-phase transition temperature. J. Chem. Phys. 1992, 97, 13. [Google Scholar] [CrossRef]
- Chen, C.-S.; Anaya, J.M.; Chen, E.Y.T.; Farr, E.; Chin, W.-C. Ocean Warming–Acidification Synergism Undermines Dissolved Organic Matter Assembly. PLoS ONE 2015, 10, e0118300. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T. Phase transitions of gels. In Polyelectrolyte Gels; American Chemical Society: Washington, DC, USA, 1992; Volume 480, pp. 1–21. [Google Scholar]
- Boyer, T.P.; Antonov, J.I.; Baranova, O.K.; Coleman, C.; Garcia, H.E.; Grodsky, A.; Johnson, D.R.; Locarnini, R.A.; Mishonov, A.V.; O’Brien, T.D.; et al. World Ocean Database 2013; National Oceanic and Atmospheric Administration: Washington, DC, USA, 2013. [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hansell, D.A.; Orellana, M.V. Dissolved Organic Matter in the Global Ocean: A Primer. Gels 2021, 7, 128. https://doi.org/10.3390/gels7030128
Hansell DA, Orellana MV. Dissolved Organic Matter in the Global Ocean: A Primer. Gels. 2021; 7(3):128. https://doi.org/10.3390/gels7030128
Chicago/Turabian StyleHansell, Dennis A., and Mónica V. Orellana. 2021. "Dissolved Organic Matter in the Global Ocean: A Primer" Gels 7, no. 3: 128. https://doi.org/10.3390/gels7030128
APA StyleHansell, D. A., & Orellana, M. V. (2021). Dissolved Organic Matter in the Global Ocean: A Primer. Gels, 7(3), 128. https://doi.org/10.3390/gels7030128




