From Nano-Gels to Marine Snow: A Synthesis of Gel Formation Processes and Modeling Efforts Involved with Particle Flux in the Ocean
Abstract
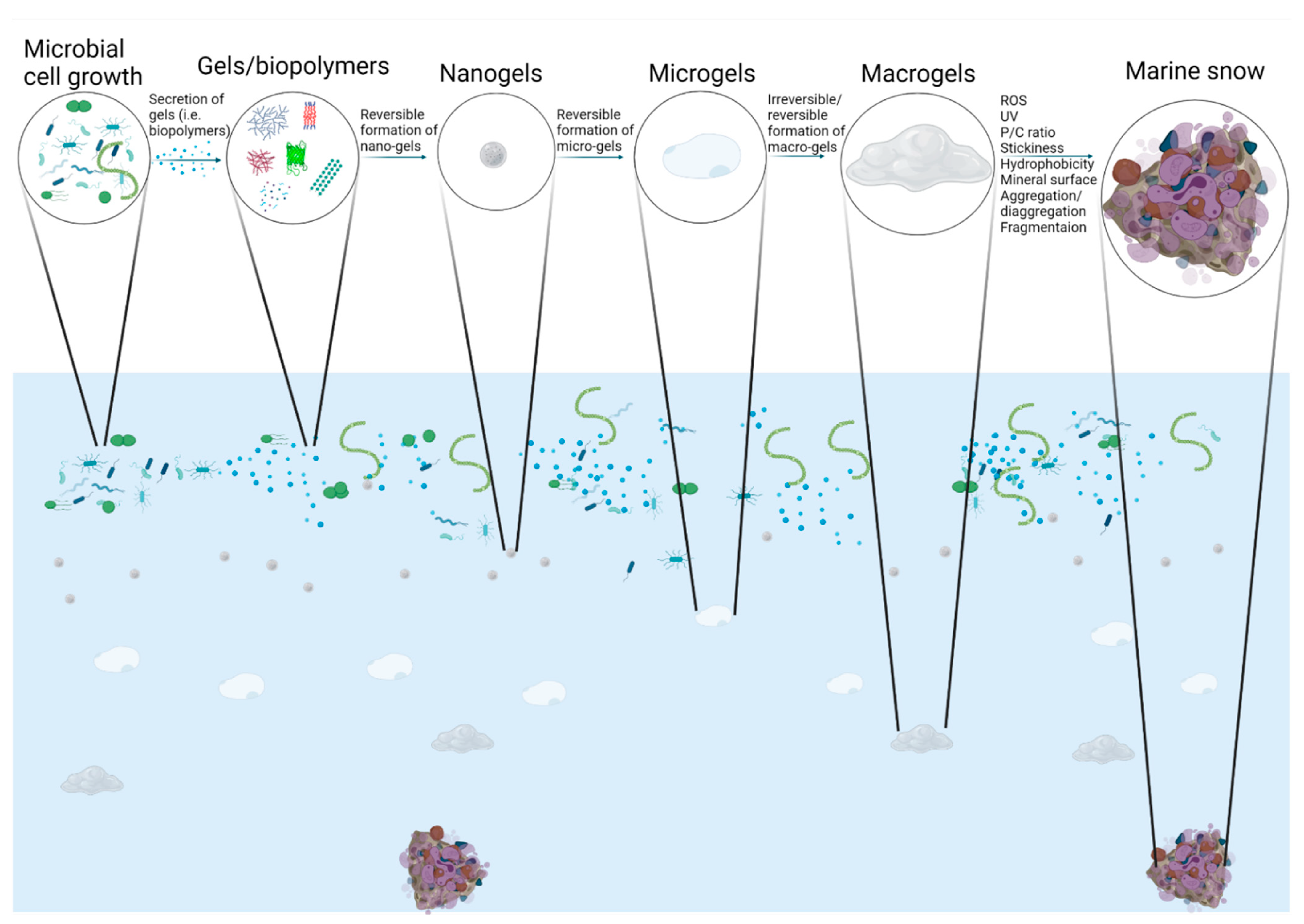
1. Introduction
2. Colloidal Nanogels (or Macromolecules) and Microgels
3. TEP and CSP
4. Marine Snow
5. Modeling Efforts
6. Marine Gels and the Ocean’s Carbon Cycle
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Azam, F.; Fenchel, T.; Field, J.G.; Gray, J.S.; Meyer-Reil, L.A.; Thingstad, F. The ecological role of water-column microbes in the sea. Mar. Ecol. Prog. Ser. 1983, 10, 257–263. [Google Scholar] [CrossRef]
- Falkowski, P.G.; Barber, R.T.; Smetacek, V. Biogeochemical controls and feedbacks on ocean primary production. Science 1998, 281, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Ducklow, H.W.; Steinberg, D.K.; Buesseler, K.O. Upper ocean carbon export and the biological pump. Oceanography 2001, 14, 50–58. [Google Scholar] [CrossRef]
- Verdugo, P.; Alldredge, A.L.; Azam, F.; Kirchman, D.L.; Passow, U.; Santschi, P.H. The oceanic gel phase: A bridge in the DOM–POM continuum. Mar. Chem. 2004, 92, 67–85. [Google Scholar] [CrossRef]
- Mari, X.; Passow, U.; Migon, C.; Burd, A.B.; Legendre, L. Transparent exopolymer particles: Effects on carbon cycling in the ocean. Prog. Oceanogr. 2017, 151, 13–37. [Google Scholar] [CrossRef]
- Polimene, L.; Sailley, S.; Clark, D.; Mitra, A.; Allen, J.I. Biological or microbial carbon pump? The role of phytoplankton stoichiometry in ocean carbon sequestration. J. Plankton Res. 2017, 39, 180–186. [Google Scholar] [CrossRef]
- Orellana, M.V.; Leck, C. Chapter 9–Marine microgels. In Biogeochemistry of Marine Dissolved Organic Matter, 2nd ed.; Hansell, D.A., Carlson, C.A., Eds.; Academic Press: Boston, MA, USA, 2015; pp. 451–480. [Google Scholar] [CrossRef]
- Azam, F.; Smith, D.C.; Steward, G.F.; Hagström, Å. Bacteria-organic matter coupling and its significance for oceanic carbon cycling. Micro. Ecol. 1994, 28, 167–179. [Google Scholar] [CrossRef] [PubMed]
- Decho, A.W. Microbial exopolymer secretions in ocean environments: Their role (s) in food webs and marine processes. Oceanogr. Mar. Biol. Annu. Rev. 1990, 28, 73–153. [Google Scholar]
- Buesseler, K.O.; Lamborg, C.H.; Boyd, P.W.; Lam, P.J.; Trull, T.W.; Bidigare, R.R.; Bishop, J.K.; Casciotti, K.L.; Dehairs, F.; Elskens, M.; et al. Revisiting carbon flux through the ocean’s twilight zone. Science 2007, 316, 567–570. [Google Scholar] [CrossRef]
- Armstrong, R.A.; Lee, C.; Hedges, J.I.; Honjo, S.; Wakeham, S.G. A new, mechanistic model for organic carbon fluxes in the ocean based on the quantitative association of POC with ballast minerals. Deep Sea Res. II Top. Stud. Oceanogr. 2001, 49, 219–236. [Google Scholar] [CrossRef]
- Martin, J.H.; Knauer, G.A.; Karl, D.M.; Broenkow, W.W. VERTEX: Carbon cycling in the northeast Pacific. Deep Sea Res. Part A Oceanogr. Res. Pap. 1987, 34, 267–285. [Google Scholar] [CrossRef]
- Verdugo, P.; Santschi, P.H. Polymer dynamics of DOC networks and gel formation in seawater. Deep Sea Res. Part II Top. Stud. Oceanogr. 2010, 57, 1486–1493. [Google Scholar] [CrossRef]
- Verdugo, P. Marine microgels. Ann. Rev. Mar. Sci. 2012, 15, 4375–4400. [Google Scholar] [CrossRef]
- Arrieta, J.M.; Mayol, E.; Hansman, R.L.; Herndl, G.J.; Dittmar, T.; Duarte, C.M. Dilution limits dissolved organic carbon utilization in the deep ocean. Science 2015, 348, 331–333. [Google Scholar] [CrossRef] [PubMed]
- Santschi, P.H.; Balnois, E.; Wilkinson, K.J.; Zhang, J.; Buffle, J.; Guo, L. Fibrillar polysaccharides in marine macromolecular organic matter as imaged by atomic force microscopy and transmission electron microscopy. Limnol. Oceanogr. 1998, 43, 896–908. [Google Scholar] [CrossRef]
- Santschi, P.H.; Guo, L.; Walsh, I.D.; Quigley, M.S.; Baskaran, M. Boundary exchange and scavenging of radionuclides in continental margin waters of the Middle Atlantic Bight: Implications for organic carbon fluxes. Cont. Shelf Res. 1999, 19, 609–636. [Google Scholar] [CrossRef]
- Mari, X.; Burd, A. Seasonal size spectra of transparent exopolymeric particles (TEP) in a coastal sea and comparison with those predicted using coagulation theory. Mar. Ecol. Prog. Ser. 1998, 163, 63–76. [Google Scholar] [CrossRef]
- Zhou, J.; Mopper, K.; Passow, U. The role of surface-active carbohydrates in the formation of transparent exopolymer particles by bubble adsorption of seawater. Limnol. Oceanogr. 1998, 43, 1860–1871. [Google Scholar] [CrossRef]
- Passow, U. Formation of transparent exopolymer particles, TEP, from dissolved precursor material. Mar. Ecol. Prog, Ser. 2000, 192, 1–11. [Google Scholar] [CrossRef]
- Alldredge, A.L.; Silver, M.W. Characteristics, dynamics and significance of marine snow. Prog. Oceanogr. 1988, 20, 41–82. [Google Scholar] [CrossRef]
- Alldredge, A.L.; Jackson, G.A. Aggregation in marine systems. Deep-Sea Res. Part II Top. Stud. Oceanogr. 1995, 42, 1–7. [Google Scholar] [CrossRef]
- Jackson, G.A.; Burd, A.B. Aggregation in the marine environment. Environ. Sci. Technol. 1998, 32, 2805–2814. [Google Scholar] [CrossRef]
- Leppard, G.G.; West, M.M.; Flannigan, D.T.; Carson, J.; Lott, J.N. A classification scheme for marine organic colloids in the Adriatic Sea: Colloid speciation by transmission electron microscopy. Can. J. Fish. Aq. Sci. 1997, 54, 2334–2349. [Google Scholar] [CrossRef]
- Navarro, E.; Baun, A.; Behra, R.; Hartmann, N.B.; Filser, J.; Miao, A.J.; Quigg, A.; Santschi, P.H.; Sigg, L. Environmental behavior and ecotoxicity of engineered nanoparticles to algae, plants, and fungi. Ecotoxicology 2008, 17, 372–386. [Google Scholar] [CrossRef] [PubMed]
- Quigg, A.; Chin, W.C.; Chen, C.S.; Zhang, S.; Jiang, Y.; Miao, A.J.; Schwehr, K.A.; Xu, C.; Santschi, P.H. Direct and indirect toxic effects of engineered nanoparticles on algae: Role of natural organic matter. ACS Sustain. Chem. Eng. 2013, 1, 686–702. [Google Scholar] [CrossRef]
- Chanton, J.; Zhao, T.; Rosenheim, B.E.; Joye, S.; Bosman, S.; Brunner, C.; Yeager, K.M.; Diercks, A.R.; Hollander, D. Using natural abundance radiocarbon to trace the flux of petrocarbon to the seafloor following the Deepwater Horizon oil spill. Environ. Sci. Technol. 2015, 49, 847–854. [Google Scholar] [CrossRef]
- Daly, K.L.; Passow, U.; Chanton, J.; Hollander, D. Assessing the impacts of oil-associated marine snow formation and sedimentation during and after the Deepwater Horizon oil spill. Anthropocene 2016, 13, 18–33. [Google Scholar] [CrossRef]
- Passow, U.; Ziervogel, K. Marine snow sedimented oil released during the Deepwater Horizon spill. Oceanography 2016, 29, 118–125. [Google Scholar] [CrossRef]
- Quigg, A.; Passow, U.; Chin, W.C.; Xu, C.; Doyle, S.; Bretherton, L.; Kamalanathan, M.; Williams, A.K.; Sylvan, J.B.; Finkel, Z.V.; et al. The role of microbial exopolymers in determining the fate of oil and chemical dispersants in the ocean. Limnol. Oceanogr. Lett. 2016, 1, 3–26. [Google Scholar] [CrossRef]
- Michels, J.; Stippkugel, A.; Lenz, M.; Wirtz, K.; Engel, A. Rapid aggregation of biofilm-covered microplastics with marine biogenic particles. Proc. R. Soc. B 2018, 285, 20181203. [Google Scholar] [CrossRef]
- Summers, S.; Henry, T.; Gutierrez, T. Agglomeration of nano-and microplastic particles in seawater by autochthonous and de novo-produced sources of exopolymeric substances. Mar. Poll. Bull. 2018, 130, 258–267. [Google Scholar] [CrossRef] [PubMed]
- Porter, A.; Lyons, B.P.; Galloway, T.S.; Lewis, C. Role of marine snows in microplastic fate and bioavailability. Environ. Sci. Technol. 2018, 52, 7111–7119. [Google Scholar] [CrossRef] [PubMed]
- Kvale, K.F.; Friederike Prowe, A.E.; Oschlies, A. A critical examination of the role of marine snow and zooplankton fecal pellets in removing ocean surface microplastic. Front. Mar. Sci. 2020, 6, 808. [Google Scholar] [CrossRef]
- Passow, U.; Ziervogel, K.; Asper, V.; Diercks, A. Marine snow formation in the aftermath of the Deepwater Horizon oil spill in the Gulf of Mexico. Environ. Res. Lett. 2012, 7, 035301. [Google Scholar] [CrossRef]
- Shiu, R.F.; Vazquez, C.I.; Chiang, C.Y.; Chiu, M.H.; Chen, C.S.; Ni, C.W.; Gong, G.C.; Quigg, A.; Santschi, P.H.; Chin, W.C. Nano-and microplastics trigger secretion of protein-rich extracellular polymeric substances from phytoplankton. Sci. Total Environ. 2020, 748, 141469. [Google Scholar] [CrossRef] [PubMed]
- Santschi, P.H.; Chin, W.-C.; Quigg, A.; Xu, C.; Kamalanathan, M.; Lin, P.; Shiu, R.-F. Marine gel interactions with hydrophilic and hydrophobic pollutants. Gels 2021, 7, 83. [Google Scholar] [CrossRef]
- Žutić, V.; Svetličić, V. Interfacial Processes in Marine Chemistry; Springer: Berlin/Heidelberg, Germany, 2000; pp. 149–165. [Google Scholar]
- Thornton, D.C. Diatom aggregation in the sea: Mechanisms and ecological implications. Eur. J. Phycol. 2002, 37, 149–161. [Google Scholar] [CrossRef]
- Passow, U. Transparent exopolymer particles (TEP) in aquatic environments. Prog. Oceanogr. 2002, 55, 287–333. [Google Scholar] [CrossRef]
- Verdugo, P. Dynamics of marine biopolymer networks. Polym. Bull. 2007, 58, 139–143. [Google Scholar] [CrossRef]
- Burd, A.B.; Jackson, G.A. Particle aggregation. Ann. Rev. Mar. Sci. 2009, 1, 65–90. [Google Scholar] [CrossRef]
- Decho, A.W.; Gutierrez, T. Microbial extracellular polymeric substances (EPSs) in ocean systems. Front. Microbiol. 2017, 8, 922. [Google Scholar] [CrossRef] [PubMed]
- Riley, G.A. Organic aggregates in seawater and the dynamics of their formation and utilization. Limnol. Oceanogr. 1963, 8, 372–381. [Google Scholar] [CrossRef]
- Hansell, D.A. Recalcitrant dissolved organic carbon fractions. Ann. Rev. Mar. Sci. 2013, 5, 421–445. [Google Scholar] [CrossRef] [PubMed]
- Chin, W.C.; Orellana, M.V.; Verdugo, P. Spontaneous assembly of marine dissolved organic matter into polymer gels. Nature 1998, 391, 568–572. [Google Scholar] [CrossRef]
- Ding, Y.X.; Chin, W.C.; Verdugo, P. Development of a fluorescence quenching assay to measure the fraction of organic carbon present in self-assembled gels in seawater. Mar. Chem. 2007, 106, 456–462. [Google Scholar] [CrossRef]
- Moon, A.; Oviedo, A.; Ng, C.; Tuthill, J.; Dmitrijeva, J. Bacterial colonization of marine gel. In Proceedings of the American Society for Limnology and Oceanography Aquatic Sciences Meeting, Santa Fe, NM, USA, 4–9 February 2007. [Google Scholar]
- Verdugo, P.; Orellana, M.V.; Chin, W.C.; Petersen, T.W.; van den Eng, G.; Benner, R.; Hedges, J.I. Marine biopolymer self-assembly: Implications for carbon cycling in the ocean. Faraday Discuss. 2008, 139, 393–398. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Wen, L.S.; Tang, D.; Santschi, P.H. Re-examination of cross-flow ultrafiltration for sampling aquatic colloids: Evidence from molecular probes. Mar. Chem. 2000, 69, 75–90. [Google Scholar] [CrossRef]
- Edwards, S.F.; Grant, J.W. The effect of entanglements of diffusion in a polymer melt. J. Phys. A Math. Nucl. Gen. 1973, 6, 1169. [Google Scholar] [CrossRef]
- Edwards, S.F. The dynamics of polymer networks. J. Phys. A Math. Nucl. Gen. 1974, 7, 318. [Google Scholar] [CrossRef]
- Edwards, S.F. The theory of macromolecular networks. Biorheology 1986, 23, 589–603. [Google Scholar] [CrossRef]
- Grosberg, A.Y.; Khokhlov, A.R. Giant Molecules; Academic Press: Cambridge, MA, USA, 1997. [Google Scholar]
- Doi, M.; Edwards, S.F. The Theory of Polymer Dynamics; Oxford University Press: Oxford, UK, 1988. [Google Scholar]
- De Gennes, P.G.; Leger, L. Dynamics of entangled polymer chains. Ann. Rev. Phys. Chem. 1982, 33, 49–61. [Google Scholar] [CrossRef]
- Orellana, M.V.; Petersen, T.W.; Diercks, A.H.; Donohoe, S.; Verdugo, P.; van den Engh, G. Marine microgels: Optical and proteomic fingerprints. Mar. Chem. 2007, 105, 229–239. [Google Scholar] [CrossRef]
- Ding, Y.X.; Chin, W.C.; Rodriguez, A.; Hung, C.C.; Santschi, P.H.; Verdugo, P. Amphiphilic exopolymers from Sagittula stellata induce DOM self-assembly and formation of marine microgels. Mar. Chem. 2008, 112, 11–19. [Google Scholar] [CrossRef]
- Orellana, M.V.; Verdugo, P. Ultraviolet radiation blocks the organic carbon exchange between the dissolved phase and the gel phase in the ocean. Limnol. Oceanogr. 2003, 48, 1618–1623. [Google Scholar] [CrossRef]
- Orellana, M.V.; Matrai, P.A.; Leck, C.; Rauschenberg, C.D.; Lee, A.M.; Coz, E. Marine microgels as a source of cloud condensation nuclei in the high Arctic. Proc. Natl. Acad. Sci. USA 2011, 108, 13612–13617. [Google Scholar] [CrossRef]
- Tanaka, T.; Fillmore, D.; Sun, S.T.; Nishio, I.; Swislow, G.; Shah, A. Phase transitions in ionic gels. Phys. Rev. Lett. 1980, 45, 1636. [Google Scholar] [CrossRef]
- Orellana, M.N.; Hansell, D.A. Ribulose-1, 5-bisphosphate carboxylase/oxygenase (RuBisCO): A long-lived protein in the deep ocean. Limnol. Oceanogr. 2012, 57, 826–834. [Google Scholar] [CrossRef]
- Sun, L.; Xu, C.; Zhang, S.; Lin, P.; Schwehr, K.A.; Quigg, A.; Chiu, M.H.; Chin, W.C.; Santschi, P.H. Light-induced aggregation of microbial exopolymeric substances. Chemosphere 2017, 181, 675–681. [Google Scholar] [CrossRef]
- Sun, L.; Chiu, M.H.; Xu, C.; Lin, P.; Schwehr, K.A.; Bacosa, H.; Kamalanathan, M.; Quigg, A.; Chin, W.C.; Santschi, P.H. The effects of sunlight on the composition of exopolymeric substances and subsequent aggregate formation during oil spills. Mar. Chem. 2018, 203, 49–54. [Google Scholar] [CrossRef]
- Sun, L.; Chin, W.C.; Chiu, M.H.; Xu, C.; Lin, P.; Schwehr, K.A.; Quigg, A.; Santschi, P.H. Sunlight induced aggregation of dissolved organic matter: Role of proteins in linking organic carbon and nitrogen cycling in seawater. Sci. Total Environ. 2019, 654, 872–877. [Google Scholar] [CrossRef]
- Sun, L.; Xu, C.; Lin, P.; Quigg, A.; Chin, W.C.; Santschi, P.H. Photo-oxidation of proteins facilitates the preservation of high molecular weight dissolved organic nitrogen in the ocean. Mar. Chem. 2021, 229, 103907. [Google Scholar] [CrossRef]
- Xu, C.; Chin, W.C.; Lin, P.; Chen, H.; Chiu, M.H.; Waggoner, D.C.; Xing, W.; Sun, L.; Schwehr, K.A.; Hatcher, P.G.; et al. Comparison of microgels, extracellular polymeric substances (EPS) and transparent exopolymeric particles (TEP) determined in seawater with and without oil. Mar. Chem. 2019, 215, 103667. [Google Scholar] [CrossRef]
- Santschi, P.H.; Xu, C.; Schwehr, K.A.; Lin, P.; Sun, L.; Chin, W.C.; Kamalanathan, M.; Bacosa, H.P.; Quigg, A. Can the protein/carbohydrate (P/C) ratio of exopolymeric substances (EPS) be used as a proxy for their ‘stickiness’ and aggregation propensity? Mar. Chem. 2020, 218, 103734. [Google Scholar] [CrossRef]
- Ortega-Retuerta, E.; Passow, U.; Duarte, C.M.; Reche, I. Effects of ultraviolet B radiation on (not so) transparent exopolymer particles. Biogeosciences 2009, 6, 3071–3080. [Google Scholar] [CrossRef]
- Song, W.; Zhao, C.; Mu, S.; Pan, X.; Zhang, D.; Al-Misned, F.A.; Mortuza, M.G. Effects of irradiation and pH on fluorescence properties and flocculation of extracellular polymeric substances from the cyanobacterium Chroococcus minutus. Colloids Surf. B Biointerfaces 2015, 128, 115–118. [Google Scholar] [CrossRef]
- McCarthy, M.; Hedges, J.; Benner, R. Major biochemical composition of dissolved high molecular weight organic matter in seawater. Mar. Chem. 1996, 55, 281–297. [Google Scholar] [CrossRef]
- Hung, C.C.; Guo, L.; Santschi, P.H.; Alvarado-Quiroz, N.; Haye, J.M. Distributions of carbohydrate species in the Gulf of Mexico. Mar. Chem. 2003, 81, 119–135. [Google Scholar] [CrossRef]
- Hung, C.C.; Guo, L.; Roberts, K.A.; Santschi, P.H. Upper ocean carbon flux determined by the 234Th approach and sediment traps using size-fractionated POC and 234Th data from the Gulf of Mexico. Geochem. J. 2004, 38, 601–611. [Google Scholar] [CrossRef]
- Bhaskar, P.V.; Bhosle, N.B. Microbial extracellular polymeric substances in marine biogeochemical processes. Curr. Sci. 2005, 88, 45–53. [Google Scholar]
- Ding, Y.; Hung, C.C.; Santschi, P.H.; Verdugo, P.; Chin, W.C. Spontaneous assembly of exopolymers from phytoplankton. Terr. Atmos. Ocean. Sci. 2009, 20, 741–747. [Google Scholar] [CrossRef]
- Gutierrez, T.; Teske, A.; Ziervogel, K.; Passow, U.; Quigg, A. Microbial exopolymers: Sources, chemico-physiological properties, and ecosystem effects in the marine environment. Front. Microbiol. 2018, 9, 1822. [Google Scholar] [CrossRef]
- Doyle, S.M.; Whitaker, E.A.; De Pascuale, V.; Wade, T.L.; Knap, A.H.; Santschi, P.H.; Quigg, A.; Sylvan, J.B. Rapid formation of microbe-oil aggregates and changes in community composition in coastal surface water following exposure to oil and the dispersant Corexit. Front. Microbiol. 2018, 9, 689. [Google Scholar] [CrossRef]
- Doyle, S.M.; Lin, G.; Morales-McDevitt, M.; Wade, T.L.; Quigg, A.; Sylvan, J.B. Niche Partitioning between Coastal and Offshore Shelf Waters Results in Differential Expression of Alkane and Polycyclic Aromatic Hydrocarbon Catabolic Pathways. mSystems 2020, 5, e00668-20. [Google Scholar] [CrossRef]
- Kamalanathan, M.; Doyle, S.M.; Xu, C.; Achberger, A.M.; Wade, T.L.; Schwehr, K.; Santschi, P.H.; Sylvan, J.B.; Quigg, A. Exoenzymes as a signature of microbial response to marine environmental conditions. mSystems 2020, 5, e00290-20. [Google Scholar] [CrossRef]
- Stoderegger, K.E.; Herndl, G.J. Dynamics in bacterial cell surface properties assessed by fluorescent stains and confocal laser scanning microscopy. Aquat. Microb. Ecol. 2004, 36, 29–40. [Google Scholar] [CrossRef]
- Xu, C.; Zhang, S.; Chuang, C.Y.; Miller, E.J.; Schwehr, K.A.; Santschi, P.H. Chemical composition and relative hydrophobicity of microbial exopolymeric substances (EPS) isolated by anion exchange chromatography and their actinide-binding affinities. Mar. Chem. 2011, 126, 27–36. [Google Scholar] [CrossRef]
- Schwehr, K.A.; Xu, C.; Chiu, M.H.; Zhang, S.; Sun, L.; Lin, P.; Beaver, M.; Jackson, C.; Agueda, O.; Bergen, C.; et al. Protein: Polysaccharide ratio in exopolymeric substances controlling the surface tension of seawater in the presence or absence of surrogate Macondo oil with and without Corexit. Mar. Chem. 2018, 206, 84–92. [Google Scholar] [CrossRef]
- Diamant, H.; Andelman, D. Onset of self-assembly in polymer-surfactant systems. EPL (Europhys. Lett.) 1999, 48, 170. [Google Scholar] [CrossRef][Green Version]
- Arnosti, C. Microbial extracellular enzymes and the marine carbon cycle. Ann. Rev. Mar. Sci. 2011, 3, 401–425. [Google Scholar] [CrossRef]
- Kamalanathan, M.; Xu, C.; Schwehr, K.; Bretherton, L.; Beaver, M.; Doyle, S.M.; Genzer, J.; Hillhouse, J.; Sylvan, J.B.; Santschi, P.; et al. Extracellular enzyme activity profile in a chemically enhanced water accommodated fraction of surrogate oil: Toward understanding microbial activities after the Deepwater Horizon oil spill. Front. Microbiol. 2018, 9, 798. [Google Scholar] [CrossRef]
- Chin, W.C.; Orellana, M.V.; Quesada, I.; Verdugo, P. Secretion in unicellular marine phytoplankton: Demonstration of regulated exocytosis in Phaeocystis globosa. Plant Cell Physiol. 2004, 45, 535–542. [Google Scholar] [CrossRef]
- Chiu, M.H.; Khan, Z.A.; Garcia, S.G.; Le, A.D.; Kagiri, A.; Ramos, J.; Tsai, S.M.; Drobenaire, H.W.; Santschi, P.H.; Quigg, A.; et al. Effect of engineered nanoparticles on exopolymeric substances release from marine phytoplankton. Nanoscale Res. Lett. 2017, 12, 1–7. [Google Scholar] [CrossRef]
- Shiu, R.F.; Chiu, M.H.; Vazquez, C.I.; Tsai, Y.Y.; Le, A.; Kagiri, A.; Xu, C.; Kamalanathan, M.; Bacosa, H.P.; Doyle, S.M.; et al. Protein to carbohydrate (P/C) ratio changes in microbial extracellular polymeric substances induced by oil and Corexit. Mar. Chem. 2020, 223, 103789. [Google Scholar] [CrossRef]
- Alldredge, A.L.; Passow, U.; Logan, B.E. The abundance and significance of a class of large, transparent organic particles in the ocean. Deep Sea Res. Part I Oceanogr. Res. Pap. 1993, 40, 1131–1140. [Google Scholar] [CrossRef]
- Brunner, E.; Richthammer, P.; Ehrlich, H.; Paasch, S.; Simon, P.; Ueberlein, S.; van Pée, K.H. Chitin-based organic networks: An integral part of cell wall biosilica in the diatom Thalassiosira pseudonana. Angew. Chem. Int. Ed. 2009, 48, 9724–9727. [Google Scholar] [CrossRef] [PubMed]
- Spinde, K.; Kammer, M.; Freyer, K.; Ehrlich, H.; Vournakis, J.N.; Brunner, E. Biomimetic silicification of fibrous chitin from diatoms. Chem. Mat. 2011, 23, 2973–2978. [Google Scholar] [CrossRef]
- Nagata, T.; Yamada, Y.; Fukuda, H. Transparent Exopolymer Particles in Deep Oceans: Synthesis and Future Challenges. Gels 2021, 7, 75. [Google Scholar] [CrossRef]
- Busch, K.; Endres, S.; Iversen, M.H.; Michels, J.; Nöthig, E.M.; Engel, A. Bacterial colonization and vertical distribution of marine gel particles (TEP and CSP) in the Arctic Fram Strait. Front. Mar. Sci. 2017, 4, 166. [Google Scholar] [CrossRef]
- Engel, A.; Endres, S.; Galgani, L.; Schartau, M. Marvelous marine microgels: On the distribution and impact of gel-like particles in the oceanic water-column. Front. Mar. Sci. 2020, 7, 405. [Google Scholar] [CrossRef]
- Alldredge, A.L.; Gotschalk, C. In situ settling behavior of marine snow 1. Limnol. Oceanogr. 1988, 33, 339–351. [Google Scholar] [CrossRef]
- Alldredge, A.L.; Passow, U.; Haddock, H.D. The characteristics and transparent exopolymer particle (TEP) content of marine snow formed from thecate dinoflagellates. J. Plankton Res. 1998, 20, 393–406. [Google Scholar] [CrossRef]
- Chen, C.S.; Shiu, R.F.; Hsieh, Y.Y.; Xu, C.; Vazquez, C.I.; Cui, Y.; Hsu, I.C.; Quigg, A.; Santschi, P.H.; Chin, W.C. Stickiness of extracellular polymeric substances on different surfaces via magnetic tweezers. Sci. Total Environ. 2021, 757, 143766. [Google Scholar] [CrossRef]
- Buffle, J.; Wilkinson, K.J.; Stoll, S.; Filella, M.; Zhang, J. A generalized description of aquatic colloidal interactions: The three-colloidal component approach. Environ. Sci. Technol. 1998, 32, 2887–2899. [Google Scholar] [CrossRef]
- Flander-Putrle, V.; Malej, A. The evolution and phytoplankton composition of mucilaginous aggregates in the northern Adriatic Sea. Harmful Algae 2008, 7, 752–761. [Google Scholar] [CrossRef]
- Danovaro, R.; Fonda, U.S.; Pusceddu, A. Climate change and the potential spreading of marine mucilage and microbial pathogens in the Mediterranean Sea. PLoS ONE 2009, 4, e7006. [Google Scholar] [CrossRef]
- Sereda, T.J.; Mant, C.T.; Sönnichsen, F.D.; Hodges, R.S. Reversed-phase chromatography of synthetic amphipathic α-helical peptides as a model for ligand/receptor interactions Effect of changing hydrophobic environment on the relative hydrophilicity/hydrophobicity of amino acid side-chains. J. Chromatogr. 1994, 676, 139–153. [Google Scholar] [CrossRef]
- Monera, O.D.; Sereda, T.J.; Zhou, N.E.; Kay, C.M.; Hodges, R.S. Relationship of sidechain hydrophobicity and alpha-helical propensity on the stability of the single-stranded amphipathic alpha-helix. J. Pept. Sci. 1995, 1, 319–329. [Google Scholar] [CrossRef]
- Jackson, G.A. A model of the formation of marine algal flocs by physical coagulation processes. Deep Sea Res. Part A Oceanogr. Res. Pap. 1990, 37, 1197–1211. [Google Scholar] [CrossRef]
- Jackson, G.A.; Kiørboe, T. Maximum phytoplankton concentrations in the sea. Limnol. Oceanogr. 2008, 53, 395–399. [Google Scholar] [CrossRef]
- Jackson, G.A. TEP and coagulation during a mesocosm experiment. Deep Sea Res. Part II Top. Stud. Oceanogr. 1995, 42, 215–222. [Google Scholar] [CrossRef]
- Oguz, T. Modeling aggregate dynamics of transparent exopolymer particles (TEP) and their interactions with a pelagic food web. Mar. Ecol. Prog. Ser. 2017, 582, 15–31. [Google Scholar] [CrossRef]
- Jokulsdottir, T.; Archer, D. A stochastic, Lagrangian model of sinking biogenic aggregates in the ocean (SLAMS 1.0): Model formulation, validation and sensitivity. Geosci. Model Dev. 2016, 9, 1455–1476. [Google Scholar] [CrossRef]
- Dissanayake, A.L.; Burd, A.B.; Daly, K.L.; Francis, S.; Passow, U. Numerical modeling of the interactions of oil, marine snow, and riverine sediments in the ocean. J. Geophys. Res. Ocean. 2018, 123, 5388–5405. [Google Scholar] [CrossRef]
- Aumont, O.; Éthé, C.; Tagliabue, A.; Bopp, L.; Gehlen, M. PISCES-v2: An ocean biogeochemical model for carbon and ecosystem studies. Geosci. Model Dev. 2015, 8, 2465–2513. [Google Scholar] [CrossRef]
- Maerz, J.; Six, K.D.; Stemmler, I.; Ahmerkamp, S.; Ilyina, T. Microstructure and composition of marine aggregates as co-determinants for vertical particulate organic carbon transfer in the global ocean. Biogeoscience 2020, 17, 1765–1803. [Google Scholar] [CrossRef]
- Passow, U.; Alldredge, A.L. Aggregation of a diatom bloom in a mesocosm: The role of transparent exopolymer particles (TEP). Deep Sea Res. Part II Top. Stud. Oceanogr. 1995, 42, 99–109. [Google Scholar] [CrossRef]
- Trudnowska, E.; Lacour, L.; Ardyna, M.; Rogge, A.; Irisson, J.O.; Waite, A.M.; Babin, M.; Stemmann, L. Marine snow morphology illuminates the evolution of phytoplankton blooms and determines their subsequent vertical export. Nat. Commun. 2021, 12, 2816. [Google Scholar] [CrossRef] [PubMed]
- Hedges, J.I. Global biogeochemical cycles: Progress and problems. Mar. Chem. 1992, 39, 67–93. [Google Scholar] [CrossRef]
- Lampitt, R.S.; Salter, I.; de Cuevas, B.A.; Hartman, S.; Larkin, K.E.; Pebody, C.A. Long-term variability of downward particle flux in the deep northeast Atlantic: Causes and trends. Deep Sea Res. Part II Top. Stud. Oceanogr. 2010, 57, 1346–1361. [Google Scholar] [CrossRef]
- Smith, K.L.; Ruhl, H.A.; Kahru, M.; Huffard, C.L.; Sherman, A.D. Deep ocean communities impacted by changing climate over 24 y in the abyssal northeast Pacific Ocean. Proc. Natl. Acad. Sci. USA 2013, 110, 19838–19841. [Google Scholar] [CrossRef]
- Passow, U.; De La Rocha, C.L. Accumulation of mineral ballast on organic aggregates. Glob. Biogeochem. Cycles 2006, 20. [Google Scholar] [CrossRef]
- Briggs, N.; Dall’Olmo, G.; Claustre, H. Major role of particle fragmentation in regulating biological sequestration of CO2 by the oceans. Science 2020, 367, 791–793. [Google Scholar] [CrossRef]
- Collins, J.R.; Edwards, B.R.; Thamatrakoln, K.; Ossolinski, J.E.; DiTullio, G.R.; Bidle, K.D.; Doney, S.C.; Van Mooy, B.A. The multiple fates of sinking particles in the North Atlantic Ocean. Glob. Biogeochem. Cycles 2015, 9, 1471–1494. [Google Scholar] [CrossRef]
- Borchard, C.; Engel, A. Organic matter exudation by Emiliania huxleyi under simulated future ocean conditions. Biogeoscience 2012, 9, 3405–3423. [Google Scholar] [CrossRef]
- Endres, S.; Galgani, L.; Riebesell, U.; Schulz, K.G.; Engel, A. Stimulated bacterial growth under elevated p CO2: Results from an off-shore mesocosm study. PLoS ONE 2014, 9, e99228. [Google Scholar] [CrossRef]
- Engel, A. Direct relationship between CO2 uptake and transparent exopolymer particles production in natural phytoplankton. J. Plankton Res. 2002, 24, 49–53. [Google Scholar] [CrossRef]
- Liu, J.; Weinbauer, M.G.; Maier, C.; Dai, M.; Gattuso, J.P. Effect of ocean acidification on microbial diversity and on microbe-driven biogeochemistry and ecosystem functioning. Aquat. Microb. Ecol. 2010, 61, 291–305. [Google Scholar] [CrossRef]
- Piontek, J.; Händel, N.; Langer, G.; Wohlers, J.; Riebesell, U.; Engel, A. Effects of rising temperature on the formation and microbial degradation of marine diatom aggregates. Aquat. Microb. Ecol. 2009, 54, 305–318. [Google Scholar] [CrossRef][Green Version]
- Arrigo, K.R. Marine manipulations. Nature 2007, 450, 491–492. [Google Scholar] [CrossRef]
- Otero, A.; Vincenzini, M. Nostoc (Cyanophyceae) goes nude: Extracellular polysaccharides serve as a sink for reducing power under unbalanced C:N metabolism. J. Phycol. 2004, 40, 74–81. [Google Scholar] [CrossRef]
- Passow, U.; Laws, E.A. Ocean acidification as one of multiple stressors: Growth response of Thalassiosira weissflogii (diatom) under temperature and light stress. Mar. Ecol. Prog. Ser. 2015, 541, 75–90. [Google Scholar] [CrossRef]
- Piccolo, A. The supramolecular structure of humic substances. Soil Sci. 2001, 166, 810–832. [Google Scholar] [CrossRef]
- Wells, M.J.; Stretz, H.A. Supramolecular architectures of natural organic matter. Sci. Total Environ. 2019, 671, 1125–1133. [Google Scholar] [CrossRef]
| Acronym | Carbon Pool | Operational Definition |
|---|---|---|
| DOC | dissolved organic carbon | Fraction of organic carbon that passes through a filter (either 0.7 µm GF/F or 0.4 µm polycarbonate filter); It contains polymers (e.g., carbohydrates, proteins) that spontaneously self-assemble; these free biopolymers form nanogels. |
| DOM | dissolved organic matter | Material that passes through a filter (either 0.7 µm GF/F or 0.4 µm polycarbonate filter); includes colloidal particles and macromolecules. |
| POC | particulate organic carbon | Fraction of carbon retained by the filter. |
| POM | particulate organic matter | Fraction of organic matter retained by the filter. |
| EPS | extracellular polymeric substances or exopolymeric substances | Protein and polysaccharide rich materials with smaller amounts of nucleic acids and lipids. |
| TEP | transparent exopolymeric particles | Alcian-blue stainable transparent particles that are formed from acid polysaccharides. |
| CSP | Commassie stained particles | Protein-rich Commassie stainable particles. |
| marine snow | Composite particles (algae, bacteria, feces) in a matrix of EPS which is visible to the naked eye. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Quigg, A.; Santschi, P.H.; Burd, A.; Chin, W.-C.; Kamalanathan, M.; Xu, C.; Ziervogel, K. From Nano-Gels to Marine Snow: A Synthesis of Gel Formation Processes and Modeling Efforts Involved with Particle Flux in the Ocean. Gels 2021, 7, 114. https://doi.org/10.3390/gels7030114
Quigg A, Santschi PH, Burd A, Chin W-C, Kamalanathan M, Xu C, Ziervogel K. From Nano-Gels to Marine Snow: A Synthesis of Gel Formation Processes and Modeling Efforts Involved with Particle Flux in the Ocean. Gels. 2021; 7(3):114. https://doi.org/10.3390/gels7030114
Chicago/Turabian StyleQuigg, Antonietta, Peter H. Santschi, Adrian Burd, Wei-Chun Chin, Manoj Kamalanathan, Chen Xu, and Kai Ziervogel. 2021. "From Nano-Gels to Marine Snow: A Synthesis of Gel Formation Processes and Modeling Efforts Involved with Particle Flux in the Ocean" Gels 7, no. 3: 114. https://doi.org/10.3390/gels7030114
APA StyleQuigg, A., Santschi, P. H., Burd, A., Chin, W.-C., Kamalanathan, M., Xu, C., & Ziervogel, K. (2021). From Nano-Gels to Marine Snow: A Synthesis of Gel Formation Processes and Modeling Efforts Involved with Particle Flux in the Ocean. Gels, 7(3), 114. https://doi.org/10.3390/gels7030114









