Chitosan Microgels and Nanoparticles via Electrofluidodynamic Techniques for Biomedical Applications
Abstract
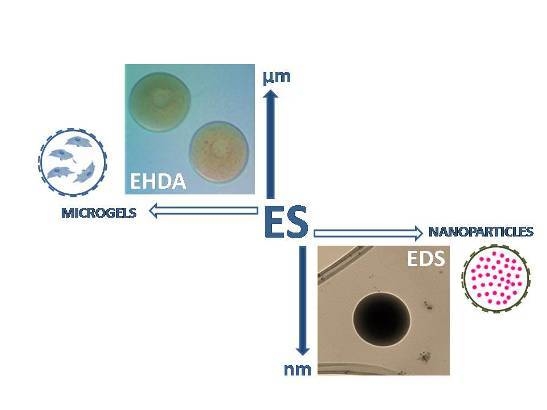
:1. Introduction

- (a)
- EDS involves the deposition of charged droplets on a grounded plate, by the breaking of polymer jet into nano-droplets under the solution overcharging conditions to form individual nanoparticles or agglomerates as a function of the local surface charge.
- (b)
- EHDA is based on the deposition of charged droplets in a crosslinking agent solution—i.e., calcium chloride (CaCl2) for alginate particles—prior to the solution overcharging, by the perturbation and cutting of polymer jet until the formation of microsized particles.
2. Results
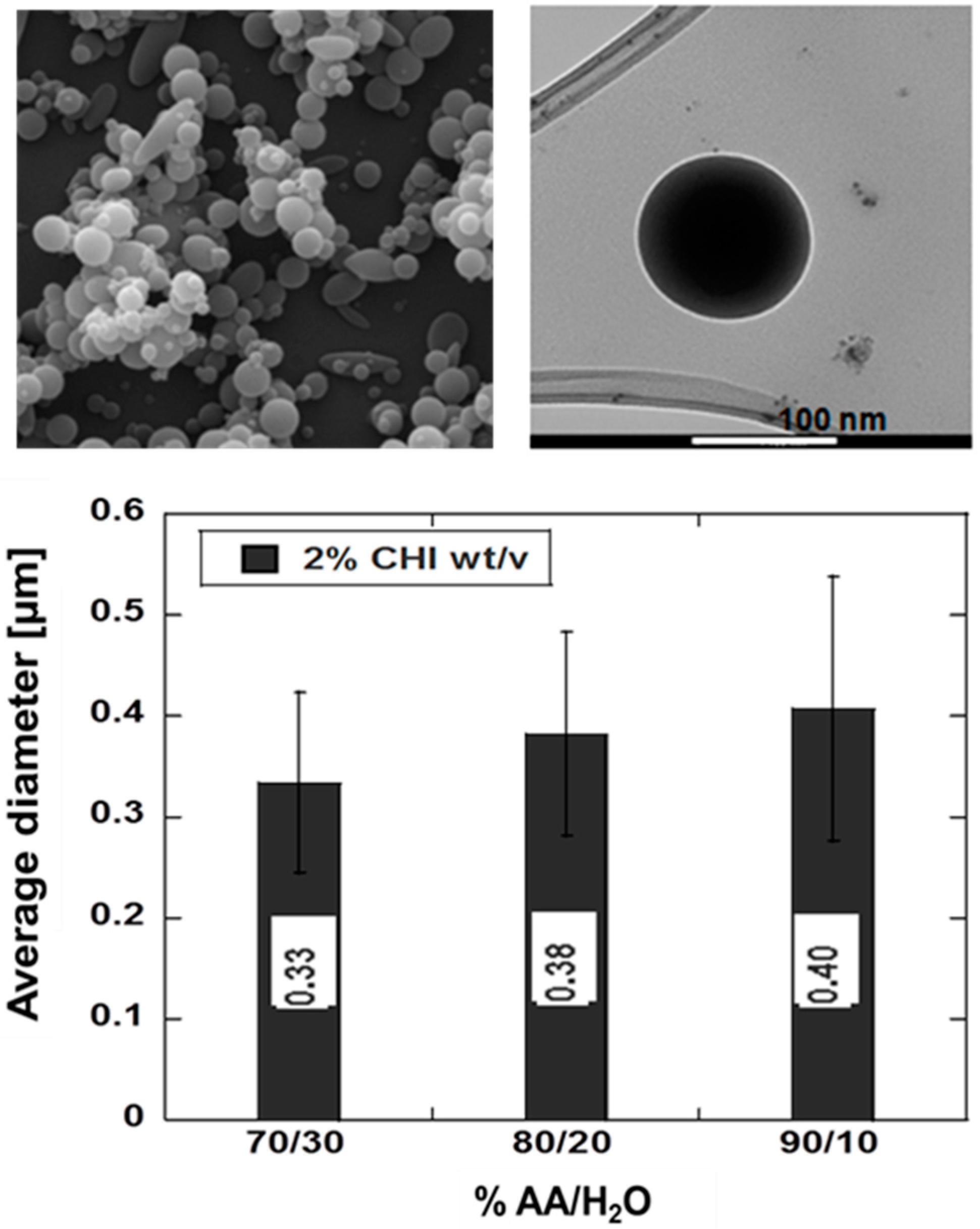
2.1. Microgels

2.2. Nanoparticles

3. Discussion
4. Conclusions and Future Trends
5. Materials and Methods
5.1. Microgels
5.2. Nanoparticles
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hamidi, M.; Azadi, A.; Rafiei, P. Hydrogel nanoparticles in drug delivery. Adv. Drug Deliv. Rev. 2008, 60, 1638–1649. [Google Scholar] [CrossRef] [PubMed]
- Tabata, Y.; Horiguchi, I.; Lutolf, M.P.; Sakai, Y. Development of Bioactive hydrogels capsules for the 3D expansion of pluripotent stem cells in bioreactors. Biomater. Sci. 2014, 2, 176–183. [Google Scholar] [CrossRef]
- Hoffman, A.S. Hydrogels for biomedical applications. Adv. Drug Deliv. Rev. 2002, 54, 3–12. [Google Scholar] [CrossRef]
- De Koker, S.; Richard, H.; de Geest, B.G. Polymeric Multilayer Capsules for Drug Delivery. Chem. Soc. Rev. 2012, 41, 2867–2884. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.Y.; Mooney, D.J. Hydrogels for tissue engineering. Chem. Rev. 2001, 101, 1869–1879. [Google Scholar] [CrossRef] [PubMed]
- Guarino, V.; Gloria, A.; Raucci, M.G.; Ambrosio, L. Hydrogel-Based Platforms for the Regeneration of Osteochondral Tissue and Intervertebral Disc. Polymers 2012, 4, 1590–1612. [Google Scholar] [CrossRef]
- Chen, S.; Lewallen, M.; Xie, T. Adhesion in the stem cell niche: Biological roles and regulation. Development 2013, 14, 255–265. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, C.; Pereira, P.; Gama, M. Self-Assembled Hydrogel Nanoparticles for Drug Delivery Applications. Materials 2010, 3, 1420–1460. [Google Scholar] [CrossRef] [Green Version]
- Nair, L.S.; Laurencin, C.T. Biodegradable polymers as biomaterials. Prog. Polym. Sci. 2007, 32, 762–798. [Google Scholar] [CrossRef]
- Tessmar, J.K.; Gopferich, A.M. Customized PEG-derived copolymers for tissue-engineering applications. Macromol. Biosci. 2007, 7, 23–39. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Marchant, R.E. Design properties of hydrogel tissue-engineering scaffolds. Expert. Rev. Med. Devices 2011, 8, 607–626. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Wong, E.H.H.; Yan, Y.; Cui, J.; Dai, Q.; Guo, J.; Qiao, G.G.; Caruso, F. The role of capsule stiffness on cellular processing. Chem. Sci. 2015, 6, 3505–3514. [Google Scholar] [CrossRef]
- Patel, V.R.; Amiji, M.M. Preparation and characterization of freeze-dried chitosan-poly (ethylene oxide) hydrogels for site-specific antibiotic delivery in the stomach. Pharm. Res. 1996, 13, 588–593. [Google Scholar] [CrossRef] [PubMed]
- Prabaharan, M.; Rodriguez-Perez, M.A.; de Saja, J.A.; Mano, J.F. Preparation and characterization of poly (L-lactic acid)–chitosan hybrid scaffolds with drug release capability. J. Biomed. Mater. Res. Part B Appl. Biomater. 2007, 81, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Jaworek, A. Electrospray droplet sources for thin film deposition. J. Mater. Sci. 2007, 42, 266–297. [Google Scholar] [CrossRef]
- Jaworek, A. Micro- and nanoparticle production by electrospraying. Powder Technol. 2007, 176, 18–35. [Google Scholar] [CrossRef]
- Guarino, V.; Altobelli, R.; Cirillo, V.; Cummaro, A.; Ambrosio, L. Additive electrospraying: A route to process electrospun scaffolds for controlled molecular release. Adv. Pol. Technol. 2015, 26, 1359–1369. [Google Scholar] [CrossRef]
- Hao, J.; Wang, F.; Wang, X.; Zhang, D.; Bi, Y.; Gao, Y.; Zhao, X.; Zhang, Q. Development and optimization of baicalin-loaded solid lipid nanoparticles prepared by coacervation method using central composite design. Eur. J. Pharma. Sci. 2012, 47, 497–505. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Ng, W.K.; Shen, S.; Kim, S.; Tan, R.B. Scalable ionic gelation synthesis of chitosan nanoparticles for drug delivery in static mixers. Carbohydr. Polymm. 2013, 94, 940–945. [Google Scholar] [CrossRef] [PubMed]
- Sridhar, R.; Ramakrishna, S. Electrosprayed nanoparticles for drug delivery and pharmaceutical applications. Biomaterials 2013, 3. [Google Scholar] [CrossRef] [PubMed]
- Muller, R.H.; Jacobs, C.; Kayser, O. Nanosuspensions as particulate drug formulations in therapy. Rationale for development and what we can expect for the future. Adv. Drug Deliv. Rev. 2001, 47, 3–19. [Google Scholar] [CrossRef]
- Dispenza, C.; Rigogliuso, S.; Grimaldi, N.; Sabatino, M.A.; Bulone, D.; Bondi, M.L.; Ghersi, G. Structure and biological evaluation of amino-functionalized PVP nanogels for fast cellular internalization. React. Funct. Polym. 2013, 73, 1103–1113. [Google Scholar] [CrossRef]
- Arya, N.; Chakraborty, S.; Dube, N.; Katti, D.S. Electrospraying: A facile technique for synthesis of chitosan-based micro/nanospheres for drug delivery applications. J. Biomed. Mater. Res. Part B Appl. Biomater. 2009, 88B, 17–31. [Google Scholar] [CrossRef] [PubMed]
- Hartman, R.P.A.; Brunner, D.J.; Camelot, D.M.A.; Marijnissen, J.C.M.; Scarlett, B. Jet break-up in electrohydrodynaminc atomization in the cone-jet mode. J. Aerosol. Sci. 2000, 31, 65–95. [Google Scholar] [CrossRef]
- Croisier, F.; Jérôme, C. Chitosan-based biomaterials for tissue engineering. Eur. Pol. J. 2013, 49, 4780–4792. [Google Scholar] [CrossRef]
- Pancholi, K.; Ahras, N.; Stride, E.; Edirisinghe, M. Novel electrohydrodynamic preparation of porous chitosan particles for drug delivery. J. Mater. Sci. Mater. Med. 2009, 20, 917–923. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.P.H. The electrohydrodynamic atomization of liquids. IEEE Trans. Ind. Appl. 1986, 22, 527–535. [Google Scholar] [CrossRef]
- Bock, N.; Dargaville, T.R.; Woodruff, M.A. Electrospraying of polymers with therapeutic molecules: State of the art. Prog. Polym. Sci. 2012, 37, 1510–1551. [Google Scholar] [CrossRef]
- Cha, J.; Lee, W.B.; Park, C.R.; Cho, Y.W.; Ahn, C.H.; Kwon, I.C. Preparation and characterization of cisplatin-incorporated chitosan hydrogels, microparticles, and nanoparticles. Macromol. Res. 2006, 14, 573–578. [Google Scholar] [CrossRef]
- Huang, Y.-C.; Lam, U.-I. Chitosan/Fucoidan pH Sensitive Nanoparticles for Oral Delivery System. J. Chin. Chem. Soc. 2011, 58, 779–785. [Google Scholar] [CrossRef]
- Wan Abdul Khodir, W.; Guarino, V.; Alvarez-Perez, M.A.; Cafiero, C.; Ambrosio, L. Trapping of Tetracycline Loaded Nanoparticles into PCL fibre networks in periodontal regeneration therapy. J. Bioact Comp. Pol. 2013, 28, 258–273. [Google Scholar] [CrossRef]
- Qian, X.-L.; Liu, H.; Wang, S.-L. pH-sensitive strontium carbonate nanoparticles as new anticancer vehicles for controlled etoposide release. Int. J. Nanomed. 2012, 7, 5781–5792. [Google Scholar]
- Tang, K.; Gomez, J. Generation by electrospray of monodisperse water droplets for targeted drug delivery by inhalation. J. Aerosol. Sci. 1994, 25, 1237–1249. [Google Scholar] [CrossRef]
- Ciach, T. Application of electro-hydro-dynamic atomization in drug delivery. J. Drug Del Sci. Technol. 2007, 17, 367–375. [Google Scholar] [CrossRef]
- Cao, L.; Luo, J.; Tu, K.; Wang, L.Q.; Jiang, H. Generation of nano-sized core–shell particles using a coaxial tri-capillary electrospray-template removal method. Colloids Surf. B Biointerfaces 2014, 115, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Labbaf, S.; Deb, S.; Cama, G.; Stride, E.; Edirisinghe, M. Preparation of multicompartment sub-micron particles using a triple-needle electrohydrodynamic device. J. Colloid Interface Sci. 2013, 409, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Lahann, J. Recent progress in nano-biotechnology: Compartmentalized micro- and nanoparticles via electrohydrodynamic co-jetting. Small 2011, 7, 1149–1156. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guarino, V.; Altobelli, R.; Ambrosio, L. Chitosan Microgels and Nanoparticles via Electrofluidodynamic Techniques for Biomedical Applications. Gels 2016, 2, 2. https://doi.org/10.3390/gels2010002
Guarino V, Altobelli R, Ambrosio L. Chitosan Microgels and Nanoparticles via Electrofluidodynamic Techniques for Biomedical Applications. Gels. 2016; 2(1):2. https://doi.org/10.3390/gels2010002
Chicago/Turabian StyleGuarino, Vincenzo, Rosaria Altobelli, and Luigi Ambrosio. 2016. "Chitosan Microgels and Nanoparticles via Electrofluidodynamic Techniques for Biomedical Applications" Gels 2, no. 1: 2. https://doi.org/10.3390/gels2010002
APA StyleGuarino, V., Altobelli, R., & Ambrosio, L. (2016). Chitosan Microgels and Nanoparticles via Electrofluidodynamic Techniques for Biomedical Applications. Gels, 2(1), 2. https://doi.org/10.3390/gels2010002