Rheological, Structural, and Biological Trade-Offs in Bioink Design for 3D Bioprinting
Abstract
1. Introduction
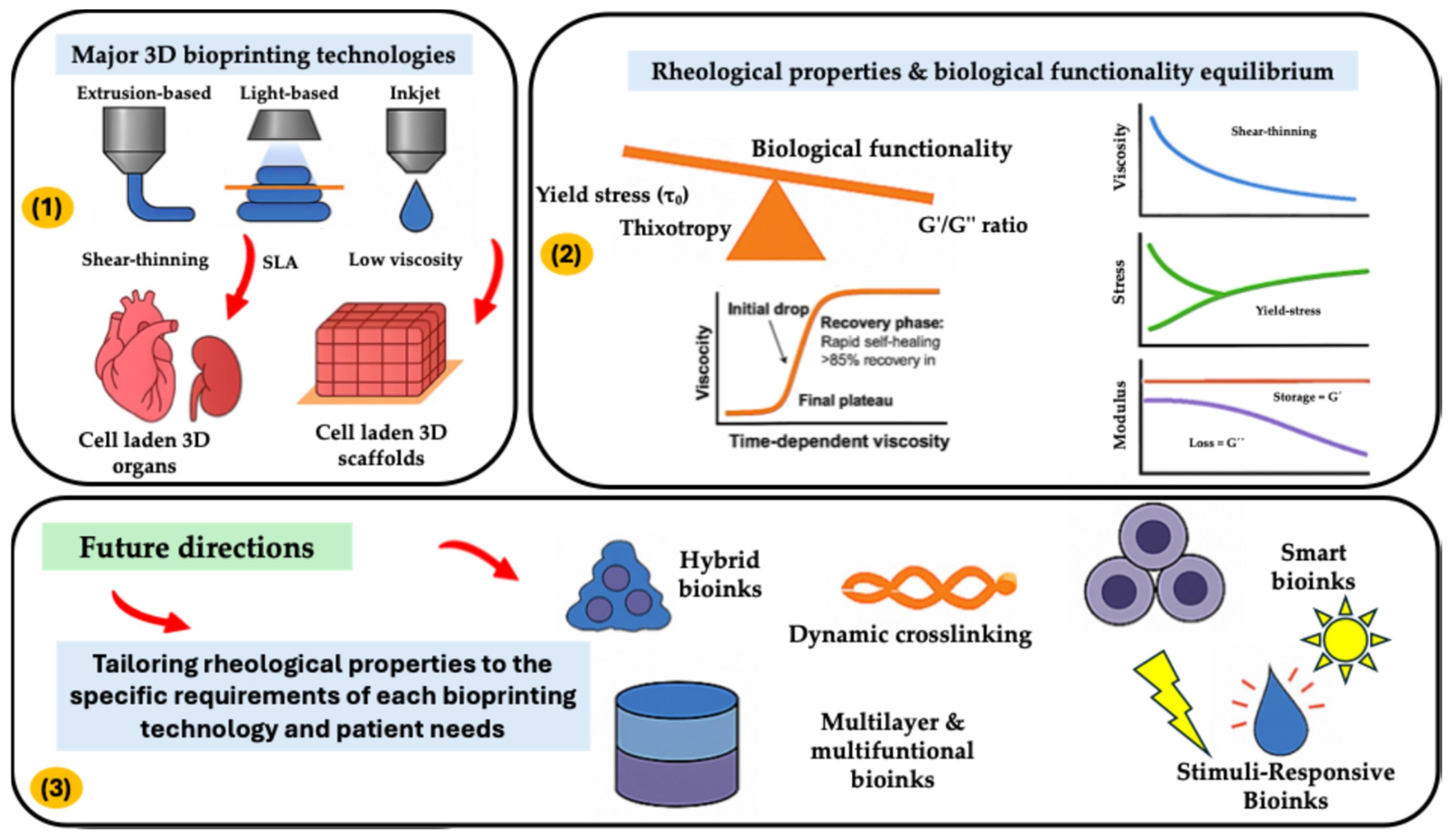
2. Fundamentals of Bioinks Rheology
2.1. Viscosity
2.2. Shear-Thinning Behavior or Pseudoplastic Behavior
2.3. Yield Stress
2.4. Viscoelasticity (G′/G″)
2.5. Thixotropy
2.6. Rheological Characterization Methods in Bioink Assessment
2.7. Rheological Influence on Bioprinting Performance Parameters
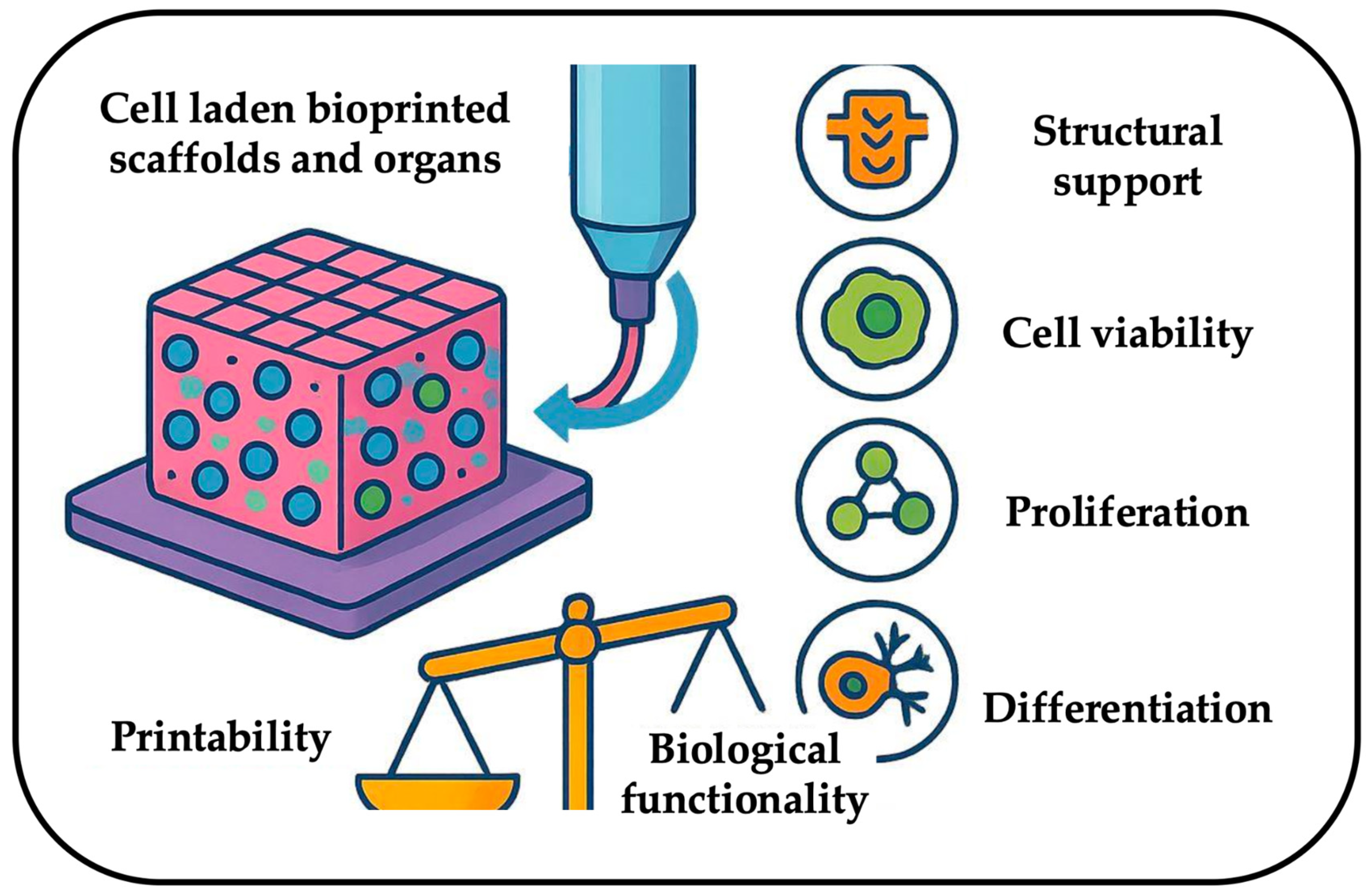
3. Biological Functionality of Bioinks
3.1. Requirements for Cell Viability
3.2. Requirements for Proliferation
3.3. Requirements for Differentiation
3.3.1. Cell Adhesion
3.3.2. Nutrient and Oxygen Transport
3.3.3. Biochemical Signaling
3.3.4. Appropriate Mechanical Properties
3.3.5. Presence of Differentiation Factors
3.3.6. Microenvironment
3.4. Requirements for Tissue Maturation
3.4.1. Natural vs. Synthetic Polymers
Natural Polymers in Bioinks
Synthetic Polymers in Bioinks
3.4.2. Cell-Adhesive Motifs
Key Cell-Adhesive Motifs and Their Functions
Impact on Tissue Maturation
3.4.3. Considerations for Bioink Design
3.4.4. Decellularized Extracellular Matrix (dECM)
4. The Rheology Trade-Off in Bioink Design: Balancing Printability and Functionality
5. Case Studies and Applications
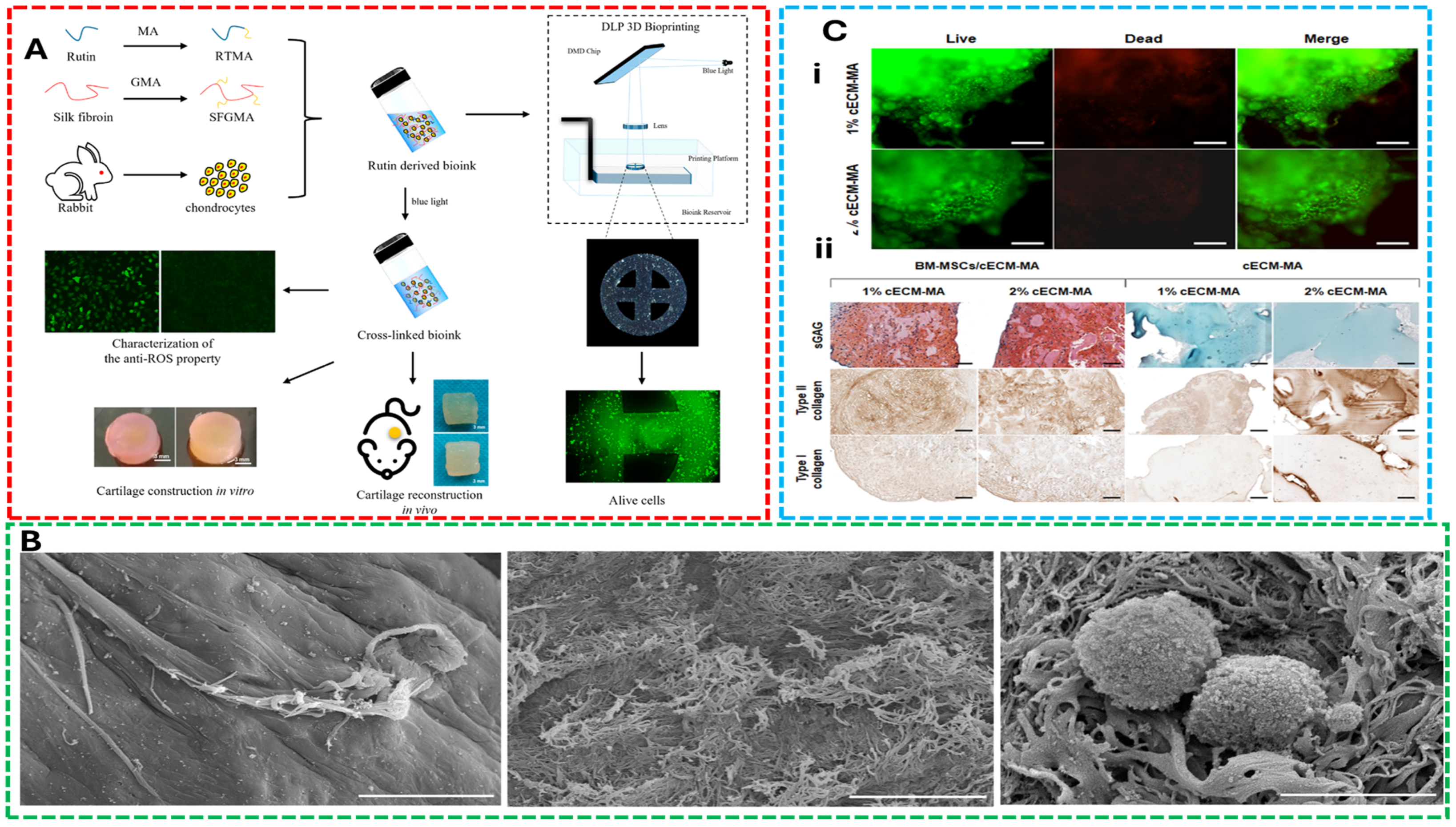
5.1. Concept 1: Bioinks for Cartilage Regeneration
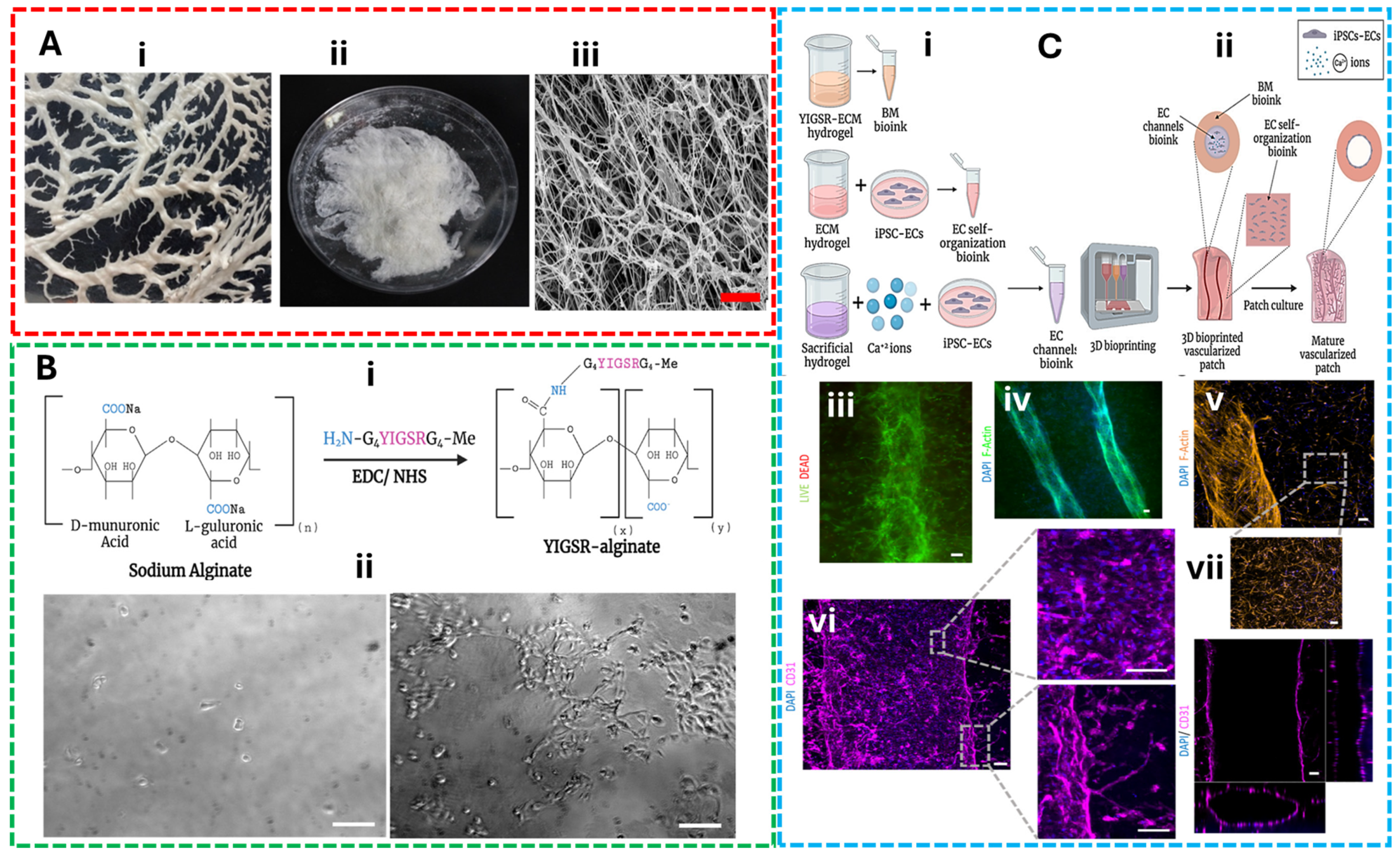
5.2. Concept 2: Bioinks for Vascularized Tissue
5.3. Concept 3: Bioinks for Bone Regeneration
5.4. Concept 4: Decellularized Extracellular Matrix (dECM)–Bioinks
6. Challenges and Future Perspectives
6.1. Standardization of Rheological and Biological Testing
6.2. Regulatory and Manufacturing Considerations
6.3. Future Perspectives
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dai, G.; Lee, V. Three-Dimensional Bioprinting and Tissue Fabrication: Prospects for Drug Discovery and Regenerative Medicine. Adv. Health Care Technol. 2015, 23, 23–35. [Google Scholar] [CrossRef]
- Gao, G.; Cui, X. Three-Dimensional Bioprinting in Tissue Engineering and Regenerative Medicine. Biotechnol. Lett. 2016, 38, 203–211. [Google Scholar] [CrossRef]
- Wei, Q.; An, Y.; Zhao, X.; Li, M.; Zhang, J. Three-Dimensional Bioprinting of Tissue-Engineered Skin: Biomaterials, Fabrication Techniques, Challenging Difficulties, and Future Directions: A Review. Int. J. Biol. Macromol. 2024, 266, 131281. [Google Scholar] [CrossRef] [PubMed]
- Jain, P.; Kathuria, H.; Dubey, N. Advances in 3D Bioprinting of Tissues/Organs for Regenerative Medicine and in-Vitro Models. Biomaterials 2022, 287, 121639. [Google Scholar] [CrossRef] [PubMed]
- Mirshafiei, M.; Rashedi, H.; Yazdian, F.; Rahdar, A.; Baino, F. Advancements in Tissue and Organ 3D Bioprinting: Current Techniques, Applications, and Future Perspectives. Mater. Des. 2024, 240, 112853. [Google Scholar] [CrossRef]
- Lee, K.; Silva, E.A.; Mooney, D.J. Growth Factor Delivery-Based Tissue Engineering: General Approaches and a Review of Recent Developments. J. R. Soc. Interface 2011, 8, 153–170. [Google Scholar] [CrossRef]
- Subbiah, R.; Guldberg, R.E. Materials Science and Design Principles of Growth Factor Delivery Systems in Tissue Engineering and Regenerative Medicine. Adv. Healthc. Mater. 2019, 8, 1801000. [Google Scholar] [CrossRef]
- Akhtar, Z.B. Advancements within Molecular Engineering for Regenerative Medicine and Biomedical Applications an Investigation Analysis towards A Computing Retrospective. J. Electron. Electromed. Eng. Med. Inform. 2024, 6, 54–72. [Google Scholar] [CrossRef]
- Fonseca, A.C.; Melchels, F.P.W.; Ferreira, M.J.S.; Moxon, S.R.; Potjewyd, G.; Dargaville, T.R.; Kimber, S.J.; Domingos, M. Emulating Human Tissues and Organs: A Bioprinting Perspective Toward Personalized Medicine. Chem. Rev. 2020, 120, 11093–11139. [Google Scholar] [CrossRef]
- Rasekh, M.; Arshad, M.S.; Ahmad, Z. Advances in Drug Delivery Integrated with Regenerative Medicine: Innovations, Challenges, and Future Frontiers. Pharmaceutics 2025, 17, 456. [Google Scholar] [CrossRef]
- Ho, D.; Quake, S.R.; McCabe, E.R.B.; Chng, W.J.; Chow, E.K.; Ding, X.; Gelb, B.D.; Ginsburg, G.S.; Hassenstab, J.; Ho, C.-M.; et al. Enabling Technologies for Personalized and Precision Medicine. Trends Biotechnol. 2020, 38, 497–518. [Google Scholar] [CrossRef] [PubMed]
- Alzoubi, L.; Aljabali, A.A.A.; Tambuwala, M.M. Empowering Precision Medicine: The Impact of 3D Printing on Personalized Therapeutic. AAPS PharmSciTech 2023, 24, 228. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Shi, C.; Wang, S.; Qi, R.; Gu, W.; Yu, F.; Zhang, G.; Qiu, F. Bridging the Gap: How Patient-Derived Lung Cancer Organoids Are Transforming Personalized Medicine. Front. Cell Dev. Biol. 2025, 13, 1554268. [Google Scholar] [CrossRef] [PubMed]
- Elango, J.; Lijnev, A.; Zamora-Ledezma, C.; Alexis, F.; Wu, W.; Marín, J.M.G.; Sanchez De Val, J.E.M. The Relationship of Rheological Properties and the Performance of Silk Fibroin Hydrogels in Tissue Engineering Application. Process Biochem. 2023, 125, 198–211. [Google Scholar] [CrossRef]
- Colosi, C.; Shin, S.R.; Manoharan, V.; Massa, S.; Costantini, M.; Barbetta, A.; Dokmeci, M.R.; Dentini, M.; Khademhosseini, A. Microfluidic Bioprinting of Heterogeneous 3D Tissue Constructs Using Low-Viscosity Bioink. Adv. Mater. 2016, 28, 677–684. [Google Scholar] [CrossRef]
- Lee, S.C.; Gillispie, G.; Prim, P.; Lee, S.J. Physical and Chemical Factors Influencing the Printability of Hydrogel-Based Extrusion Bioinks. Chem. Rev. 2020, 120, 10834–10886. [Google Scholar] [CrossRef]
- Fontes, A.B.; Marcomini, R.F. 3D Bioprinting: A Review of Materials, Processes and Bioink Properties. J. Eng. Exact Sci. 2020, 6, 0617–0639. [Google Scholar] [CrossRef]
- Malafaya, P.B.; Silva, G.A.; Reis, R.L. Natural–Origin Polymers as Carriers and Scaffolds for Biomolecules and Cell Delivery in Tissue Engineering Applications. Adv. Drug Deliv. Rev. 2007, 59, 207–233. [Google Scholar] [CrossRef]
- Gasperini, L.; Mano, J.F.; Reis, R.L. Natural Polymers for the Microencapsulation of Cells. J. R. Soc. Interface 2014, 11, 20140817. [Google Scholar] [CrossRef]
- Satchanska, G.; Davidova, S.; Petrov, P.D. Natural and Synthetic Polymers for Biomedical and Environmental Applications. Polymers 2024, 16, 1159. [Google Scholar] [CrossRef]
- Serafin, A.; Culebras, M.; Collins, M.N. Synthesis and Evaluation of Alginate, Gelatin, and Hyaluronic Acid Hybrid Hydrogels for Tissue Engineering Applications. Int. J. Biol. Macromol. 2023, 233, 123438. [Google Scholar] [CrossRef] [PubMed]
- Bjelić, D.; Finšgar, M. The Role of Growth Factors in Bioactive Coatings. Pharmaceutics 2021, 13, 1083. [Google Scholar] [CrossRef] [PubMed]
- Nuutila, K.; Samandari, M.; Endo, Y.; Zhang, Y.; Quint, J.; Schmidt, T.A.; Tamayol, A.; Sinha, I. In Vivo Printing of Growth Factor-Eluting Adhesive Scaffolds Improves Wound Healing. Bioact. Mater. 2022, 8, 296–308. [Google Scholar] [CrossRef] [PubMed]
- Nosenko, M.A.; Moysenovich, A.M.; Arkhipova, A.Y.; Atretkhany, K.-S.N.; Nedospasov, S.A.; Drutskaya, M.S.; Moisenovich, M.M. Fibroblasts Upregulate Expression of Adhesion Molecules and Promote Lymphocyte Retention in 3D Fibroin/Gelatin Scaffolds. Bioact. Mater. 2021, 6, 3449–3460. [Google Scholar] [CrossRef]
- Zheng, L.; Li, D.; Wang, L. Rheology and Printability of Biopolymeric Oil-in-water High Internal Phase Pickering Emulsions: A Review. Compr. Rev. Food Sci. Food Saf. 2025, 24, e70125. [Google Scholar] [CrossRef]
- Pal, R. Rheology of High Internal Phase Ratio Emulsions and Foams. Adv. Colloid Interface Sci. 2025, 339, 103426. [Google Scholar] [CrossRef]
- Kyle, S.; Jessop, Z.M.; Al-Sabah, A.; Whitaker, I.S. ‘Printability’ of Candidate Biomaterials for Extrusion Based 3D Printing: State-of-the-Art. Adv. Healthc. Mater. 2017, 6, 1700264. [Google Scholar] [CrossRef]
- Kitana, W.; Levario-Diaz, V.; Cavalcanti-Adam, E.A.; Ionov, L. Biofabrication of Composite Bioink-Nanofiber Constructs: Effect of Rheological Properties of Bioinks on 3D (Bio)Printing and Cells Interaction with Aligned Touch Spun Nanofibers. Adv. Healthc. Mater. 2024, 13, 2303343. [Google Scholar] [CrossRef]
- Bercea, M. Rheology as a Tool for Fine-Tuning the Properties of Printable Bioinspired Gels. Molecules 2023, 28, 2766. [Google Scholar] [CrossRef]
- Dobraszczyk, B.J.; Morgenstern, M.P. Rheology and the Breadmaking Process. J. Cereal Sci. 2003, 38, 229–245. [Google Scholar] [CrossRef]
- De Smedt, S.; Attaianese, B.; Cardinaels, R. Direct Ink Writing of Particle-Based Multiphase Materials: From Rheology to Functionality. Curr. Opin. Colloid Interface Sci. 2025, 75, 101889. [Google Scholar] [CrossRef]
- Chen, D.T.N.; Wen, Q.; Janmey, P.A.; Crocker, J.C.; Yodh, A.G. Rheology of Soft Materials. Annu. Rev. Condens. Matter Phys. 2010, 1, 301–322. [Google Scholar] [CrossRef]
- Zamora-Ledezma, C.; Narváez-Muñoz, C.; Almeida-Arellano, F.A.; Voiry, D.; Salameh, C.; Medina, E.; Carrión-Matamoros, L.M.; Navas-León, D.G.; Vizuete, K.; Debut, A.; et al. Vermiculite and Graphene Oxide 2D Layered Nanoparticle for Improving Rheology and Filtration in Water-Based Drilling Fluids Formulations. Results Eng. 2025, 26, 104796. [Google Scholar] [CrossRef]
- Ribeiro, A.; Blokzijl, M.M.; Levato, R.; Visser, C.W.; Castilho, M.; Hennink, W.E.; Vermonden, T.; Malda, J. Assessing Bioink Shape Fidelity to Aid Material Development in 3D Bioprinting. Biofabrication 2017, 10, 014102. [Google Scholar] [CrossRef]
- Oh, D.; Shirzad, M.; Chang Kim, M.; Chung, E.-J.; Nam, S.Y. Rheology-Informed Hierarchical Machine Learning Model for the Prediction of Printing Resolution in Extrusion-Based Bioprinting. Int. J. Bioprint. 2023, 9, 1280. [Google Scholar] [CrossRef]
- Abedi, K.; Keshvari, H.; Solati-Hashjin, M. Extrusion-Based Bioprinting: Considerations toward Gelatin-Alginate Bioink. Rapid Prototyp. J. 2024, 30, 1094–1104. [Google Scholar] [CrossRef]
- Cooke, M.E.; Rosenzweig, D.H. The Rheology of Direct and Suspended Extrusion Bioprinting. APL Bioeng. 2021, 5, 011502. [Google Scholar] [CrossRef]
- Rashad, A.; Gomez, A.; Gangrade, A.; Zehtabi, F.; Mandal, K.; Maity, S.; Ma, C.; Li, B.; Khademhosseini, A.; De Barros, N.R. Effect of Viscosity of Gelatin Methacryloyl-Based Bioinks on Bone Cells. Biofabrication 2024, 16, 045036. [Google Scholar] [CrossRef]
- Sánchez-Sánchez, R.; Rodríguez-Rego, J.M.; Macías-García, A.; Mendoza-Cerezo, L.; Díaz-Parralejo, A. Relationship between Shear-Thinning Rheological Properties of Bioinks and Bioprinting Parameters. Int. J. Bioprint. 2023, 9, 687. [Google Scholar] [CrossRef]
- Majumder, N.; Mishra, A.; Ghosh, S. Effect of Varying Cell Densities on the Rheological Properties of the Bioink. Bioprinting 2022, 28, e00241. [Google Scholar] [CrossRef]
- Barreiro Carpio, M.; Gonzalez Martinez, E.; Dabaghi, M.; Ungureanu, J.; Arizpe Tafoya, A.V.; Gonzalez Martinez, D.A.; Hirota, J.A.; Moran-Mirabal, J.M. High-Fidelity Extrusion Bioprinting of Low-Printability Polymers Using Carbopol as a Rheology Modifier. ACS Appl. Mater. Interfaces 2023, 15, 54234–54248. [Google Scholar] [CrossRef]
- Chirianni, F.; Vairo, G.; Marino, M. Influence of Extruder Geometry and Bio-Ink Type in Extrusion-Based Bioprinting via an in Silico Design Tool. Meccanica 2024, 59, 1285–1299. [Google Scholar] [CrossRef]
- Serna, J.A.; Florez, S.L.; Talero, V.A.; Briceño, J.C.; Muñoz-Camargo, C.; Cruz, J.C. Formulation and Characterization of a SIS-Based Photocrosslinkable Bioink. Polymers 2019, 11, 569. [Google Scholar] [CrossRef]
- Kamble, S.D.; Singh, S.; Kumar, A.; Dwivedi, G. Tissue Engineering and Regenerative Medicine: Advances and Applications, 1st ed.; CRC Press: Boca Raton, FL, USA, 2025; ISBN 978-1-003-53635-2. [Google Scholar]
- Cho, W.-W.; Park, W.; Cho, D.-W. Recent Trends in Embedded 3D Bioprinting of Vascularized Tissue Constructs. Biofabrication 2025, 17, 022002. [Google Scholar] [CrossRef]
- Gupta, D.; Derman, I.D.; Xu, C.; Huang, Y.; Ozbolat, I.T. Droplet-Based Bioprinting. Nat. Rev. Methods Primer 2025, 5, 25. [Google Scholar] [CrossRef]
- Mohanan, P.V. Compendium of 3D Bioprinting Technology, 1st ed.; CRC Press: Boca Raton, FL, USA, 2024; ISBN 978-1-003-50519-8. [Google Scholar]
- Kim, J. Characterization of Biocompatibility of Functional Bioinks for 3D Bioprinting. Bioengineering 2023, 10, 457. [Google Scholar] [CrossRef] [PubMed]
- Handral, H.K.; Natu, V.P.; Cao, T.; Fuh, J.Y.H.; Sriram, G.; Lu, W.F. Emerging Trends and Prospects of Electroconductive Bioinks for Cell-Laden and Functional 3D Bioprinting. Bio-Des. Manuf. 2022, 5, 396–411. [Google Scholar] [CrossRef]
- Shyam, R.; Palaniappan, A. Effect of Sterilization Techniques on Biomaterial Inks’ Properties and 3D Bioprinting Parameters. Bioprinting 2023, 33, e00294. [Google Scholar] [CrossRef]
- Chen, H.; Fei, F.; Li, X.; Nie, Z.; Zhou, D.; Liu, L.; Zhang, J.; Zhang, H.; Fei, Z.; Xu, T. A Facile, Versatile Hydrogel Bioink for 3D Bioprinting Benefits Long-Term Subaqueous Fidelity, Cell Viability and Proliferation. Regen. Biomater. 2021, 8, rbab026. [Google Scholar] [CrossRef]
- Chaubey, A.; Sharma, S.; Chauhan, L.; Tyagi, A. Advancements in Bio-Ink Formulations for Bio-Printing Tissues and Organs: Biomaterial Selection and Compatibility. Int. J. Pharm. Res. Dev. 2024, 6, 131–142. [Google Scholar] [CrossRef]
- Ouyang, L.; Yao, R.; Zhao, Y.; Sun, W. Effect of Bioink Properties on Printability and Cell Viability for 3D Bioplotting of Embryonic Stem Cells. Biofabrication 2016, 8, 035020. [Google Scholar] [CrossRef]
- Ng, W.L.; Huang, X.; Shkolnikov, V.; Suntornnond, R.; Yeong, W.Y. Polyvinylpyrrolidone-Based Bioink: Influence of Bioink Properties on Printing Performance and Cell Proliferation during Inkjet-Based Bioprinting. Bio-Des. Manuf. 2023, 6, 676–690. [Google Scholar] [CrossRef]
- Tarassoli, S.P.; Jessop, Z.M.; Jovic, T.; Hawkins, K.; Whitaker, I.S. Candidate Bioinks for Extrusion 3D Bioprinting—A Systematic Review of the Literature. Front. Bioeng. Biotechnol. 2021, 9, 616753. [Google Scholar] [CrossRef]
- Ying, G.; Jiang, N.; Maharjan, S.; Yin, Y.; Chai, R.; Cao, X.; Yang, J.; Miri, A.K.; Hassan, S.; Zhang, Y.S. Aqueous Two-Phase Emulsion Bioink-Enabled 3D Bioprinting of Porous Hydrogels. Adv. Mater. 2018, 30, 1805460. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.B.; Lee, H.; Kim, G.H. Strategy to Achieve Highly Porous/Biocompatible Macroscale Cell Blocks, Using a Collagen/Genipin-Bioink and an Optimal 3D Printing Process. ACS Appl. Mater. Interfaces 2016, 8, 32230–32240. [Google Scholar] [CrossRef] [PubMed]
- Hajiabbas, M.; Alemzadeh, I.; Vossoughi, M. A Porous Hydrogel-Electrospun Composite Scaffold Made of Oxidized Alginate/Gelatin/Silk Fibroin for Tissue Engineering Application. Carbohydr. Polym. 2020, 245, 116465. [Google Scholar] [CrossRef] [PubMed]
- Asadpour, S.; Kargozar, S.; Moradi, L.; Ai, A.; Nosrati, H.; Ai, J. Natural Biomacromolecule Based Composite Scaffolds from Silk Fibroin, Gelatin and Chitosan toward Tissue Engineering Applications. Int. J. Biol. Macromol. 2020, 154, 1285–1294. [Google Scholar] [CrossRef]
- Dzobo, K.; Motaung, K.S.C.M.; Adesida, A. Recent Trends in Decellularized Extracellular Matrix Bioinks for 3D Printing: An Updated Review. Int. J. Mol. Sci. 2019, 20, 4628. [Google Scholar] [CrossRef]
- Kim, B.S.; Kim, H.; Gao, G.; Jang, J.; Cho, D.-W. Decellularized Extracellular Matrix: A Step towards the next Generation Source for Bioink Manufacturing. Biofabrication 2017, 9, 034104. [Google Scholar] [CrossRef]
- Karimi, F.; O’Connor, A.J.; Qiao, G.G.; Heath, D.E. Integrin Clustering Matters: A Review of Biomaterials Functionalized with Multivalent Integrin-Binding Ligands to Improve Cell Adhesion, Migration, Differentiation, Angiogenesis, and Biomedical Device Integration. Adv. Healthc. Mater. 2018, 7, 1701324. [Google Scholar] [CrossRef]
- Kim, J.; Choi, Y.-J.; Gal, C.-W.; Sung, A.; Park, H.; Yun, H.-S. Development of an Alginate–Gelatin Bioink Enhancing Osteogenic Differentiation by Gelatin Release. Int. J. Bioprint. 2023, 9, 660. [Google Scholar] [CrossRef]
- Cohen, R.; Baruch, E.-S.; Cabilly, I.; Shapira, A.; Dvir, T. Modified ECM-Based Bioink for 3D Printing of Multi-Scale Vascular Networks. Gels 2023, 9, 792. [Google Scholar] [CrossRef] [PubMed]
- Ahlfeld, T.; Cubo-Mateo, N.; Cometta, S.; Guduric, V.; Vater, C.; Bernhardt, A.; Akkineni, A.R.; Lode, A.; Gelinsky, M. A Novel Plasma-Based Bioink Stimulates Cell Proliferation and Differentiation in Bioprinted, Mineralized Constructs. ACS Appl. Mater. Interfaces 2020, 12, 12557–12572. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.S.; Das, S.; Jang, J.; Cho, D.-W. Decellularized Extracellular Matrix-Based Bioinks for Engineering Tissue- and Organ-Specific Microenvironments. Chem. Rev. 2020, 120, 10608–10661. [Google Scholar] [CrossRef]
- Hao, X.; Miao, S.; Li, Z.; Wang, T.; Xue, B.; Chen, J.; Xian, C.; Bi, L. 3D Printed Structured Porous Hydrogel Promotes Osteogenic Differentiation of BMSCs. Mater. Des. 2023, 227, 111729. [Google Scholar] [CrossRef]
- Shao, L.; Gao, Q.; Xie, C.; Fu, J.; Xiang, M.; Liu, Z.; Xiang, L.; He, Y. Sacrificial Microgel-Laden Bioink-Enabled 3D Bioprinting of Mesoscale Pore Networks. Bio-Des. Manuf. 2020, 3, 30–39. [Google Scholar] [CrossRef]
- Gao, Q.; He, Y.; Fu, J.; Liu, A.; Ma, L. Coaxial Nozzle-Assisted 3D Bioprinting with Built-in Microchannels for Nutrients Delivery. Biomaterials 2015, 61, 203–215. [Google Scholar] [CrossRef]
- Mangiatordi, M. Design and Modeling of an Oxygen-Releasing Hydrogel for Myocardial Infarction Treatment. Ph.D. Thesis, Politecnico di Torino, Turin, Italy, 2020. [Google Scholar]
- Sharma, R.; Benwood, C.; Willerth, S.M. Drug-releasing Microspheres for Stem Cell Differentiation. Curr. Protoc. 2021, 1, e331. [Google Scholar] [CrossRef]
- Feng, Q.; Li, D.; Li, Q.; Li, H.; Wang, Z.; Zhu, S.; Lin, Z.; Cao, X.; Dong, H. Assembling Microgels via Dynamic Cross-Linking Reaction Improves Printability, Microporosity, Tissue-Adhesion, and Self-Healing of Microgel Bioink for Extrusion Bioprinting. ACS Appl. Mater. Interfaces 2022, 14, 15653–15666. [Google Scholar] [CrossRef]
- Cidonio, G.; Alcala-Orozco, C.R.; Lim, K.S.; Glinka, M.; Mutreja, I.; Kim, Y.-H.; Dawson, J.I.; Woodfield, T.B.F.; Oreffo, R.O.C. Osteogenic and Angiogenic Tissue Formation in High Fidelity Nanocomposite Laponite-Gelatin Bioinks. Biofabrication 2019, 11, 035027. [Google Scholar] [CrossRef]
- Hauptstein, J.; Forster, L.; Nadernezhad, A.; Groll, J.; Teßmar, J.; Blunk, T. Tethered TGF-Β1 in a Hyaluronic Acid-Based Bioink for Bioprinting Cartilaginous Tissues. Int. J. Mol. Sci. 2022, 23, 924. [Google Scholar] [CrossRef] [PubMed]
- Engler, A.J.; Sen, S.; Sweeney, H.L.; Discher, D.E. Matrix Elasticity Directs Stem Cell Lineage Specification. Cell 2006, 126, 677–689. [Google Scholar] [CrossRef] [PubMed]
- Chimene, D.; Kaunas, R.; Gaharwar, A.K. Hydrogel Bioink Reinforcement for Additive Manufacturing: A Focused Review of Emerging Strategies. Adv. Mater. 2020, 32, 1902026. [Google Scholar] [CrossRef]
- Morgan, F.L.C.; Moroni, L.; Baker, M.B. Dynamic Bioinks to Advance Bioprinting. Adv. Healthc. Mater. 2020, 9, 1901798. [Google Scholar] [CrossRef]
- Halloran, D.; Durbano, H.W.; Nohe, A. Bone Morphogenetic Protein-2 in Development and Bone Homeostasis. J. Dev. Biol. 2020, 8, 19. [Google Scholar] [CrossRef]
- Ko, K.R.; Lee, J.; Lee, D.; Nho, B.; Kim, S. Hepatocyte Growth Factor (HGF) Promotes Peripheral Nerve Regeneration by Activating Repair Schwann Cells. Sci. Rep. 2018, 8, 8316. [Google Scholar] [CrossRef]
- Vo, T.N.; Kasper, F.K.; Mikos, A.G. Strategies for Controlled Delivery of Growth Factors and Cells for Bone Regeneration. Adv. Drug Deliv. Rev. 2012, 64, 1292–1309. [Google Scholar] [CrossRef]
- Murphy, S.V.; De Coppi, P.; Atala, A. Opportunities and Challenges of Translational 3D Bioprinting. Nat. Biomed. Eng. 2019, 4, 370–380. [Google Scholar] [CrossRef]
- Sasmal, P.; Datta, P.; Wu, Y.; Ozbolat, I.T. 3D Bioprinting for Modelling Vasculature. Microphysiol. Syst. 2018, 2, 9. [Google Scholar] [CrossRef]
- Theus, A.S.; Ning, L.; Hwang, B.; Gil, C.; Chen, S.; Wombwell, A.; Mehta, R.; Serpooshan, V. Bioprintability: Physiomechanical and Biological Requirements of Materials for 3D Bioprinting Processes. Polymers 2020, 12, 2262. [Google Scholar] [CrossRef]
- Yang, J.; Li, Z.; Li, S.; Zhang, Q.; Zhou, X.; He, C. Tunable Metacrylated Silk Fibroin-Based Hybrid Bioinks for the Bioprinting of Tissue Engineering Scaffolds. Biomater. Sci. 2023, 11, 1895–1909. [Google Scholar] [CrossRef] [PubMed]
- Gorroñogoitia, I.; Urtaza, U.; Zubiarrain-Laserna, A.; Alonso-Varona, A.; Zaldua, A.M. A Study of the Printability of Alginate-Based Bioinks by 3D Bioprinting for Articular Cartilage Tissue Engineering. Polymers 2022, 14, 354. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Yang, M.; Zhou, Z.; Sun, J.; Meng, X.; Huang, Y.; Zhu, W.; Zhu, S.; He, N.; Zhu, X.; et al. An Anti-Oxidative Bioink for Cartilage Tissue Engineering Applications. J. Funct. Biomater. 2024, 15, 37. [Google Scholar] [CrossRef]
- Padhi, A.; Nain, A.S. ECM in Differentiation: A Review of Matrix Structure, Composition and Mechanical Properties. Ann. Biomed. Eng. 2020, 48, 1071–1089. [Google Scholar] [CrossRef]
- Yue, K.; Trujillo-de Santiago, G.; Alvarez, M.M.; Tamayol, A.; Annabi, N.; Khademhosseini, A. Synthesis, Properties, and Biomedical Applications of Gelatin Methacryloyl (GelMA) Hydrogels. Biomaterials 2015, 73, 254–271. [Google Scholar] [CrossRef]
- Webber, R.E.; Shull, K.R. Strain Dependence of the Viscoelastic Properties of Alginate Hydrogels. Macromolecules 2004, 37, 6153–6160. [Google Scholar] [CrossRef]
- Kafili, G.; Tamjid, E.; Niknejad, H.; Simchi, A. Concentration Modulated Microstructure and Rheological Properties of Nanofibrous Hydrogels Derived from Decellularized Human Amniotic Membrane for 3D Cell Culture. J. Mater. Sci. Mater. Eng. 2024, 19, 12. [Google Scholar] [CrossRef]
- Soliman, B.G.; Lindberg, G.C.J.; Jungst, T.; Hooper, G.J.; Groll, J.; Woodfield, T.B.F.; Lim, K.S. Stepwise Control of Crosslinking in a One-Pot System for Bioprinting of Low-Density Bioinks. Adv. Healthc. Mater. 2020, 9, 1901544. [Google Scholar] [CrossRef]
- Liao, C.; Li, Y.; Tjong, S.C. Graphene Nanomaterials: Synthesis, Biocompatibility, and Cytotoxicity. Int. J. Mol. Sci. 2018, 19, 3564. [Google Scholar] [CrossRef] [PubMed]
- Zandi, N.; Sani, E.S.; Mostafavi, E.; Ibrahim, D.M.; Saleh, B.; Shokrgozar, M.A.; Tamjid, E.; Weiss, P.S.; Simchi, A.; Annabi, N. Nanoengineered Shear-Thinning and Bioprintable Hydrogel as a Versatile Platform for Biomedical Applications. Biomaterials 2021, 267, 120476. [Google Scholar] [CrossRef]
- Das, S.; Kim, S.-W.; Choi, Y.-J.; Lee, S.; Lee, S.-H.; Kong, J.-S.; Park, H.-J.; Cho, D.-W.; Jang, J. Decellularized Extracellular Matrix Bioinks and the External Stimuli to Enhance Cardiac Tissue Development in Vitro. Acta Biomater. 2019, 95, 188–200. [Google Scholar] [CrossRef] [PubMed]
- Paxton, N.; Smolan, W.; Böck, T.; Melchels, F.; Groll, J.; Jungst, T. Proposal to Assess Printability of Bioinks for Extrusion-Based Bioprinting and Evaluation of Rheological Properties Governing Bioprintability. Biofabrication 2017, 9, 044107. [Google Scholar] [CrossRef] [PubMed]
- Rasouli, R.; Sweeney, C.; Frampton, J.P. Heterogeneous and Composite Bioinks for 3D-Bioprinting of Complex Tissue. Biomed. Mater. Devices 2025, 3, 108–126. [Google Scholar] [CrossRef] [PubMed]
- Tuftee, C.; Alsberg, E.; Ozbolat, I.T.; Rizwan, M. Emerging Granular Hydrogel Bioinks to Improve Biological Function in Bioprinted Constructs. Trends Biotechnol. 2024, 42, 339–352. [Google Scholar] [CrossRef]
- Brunel, L.G.; Hull, S.M.; Heilshorn, S.C. Engineered Assistive Materials for 3D Bioprinting: Support Baths and Sacrificial Inks. Biofabrication 2022, 14, 032001. [Google Scholar] [CrossRef]
- Yeh, M.-Y.; Zhao, J.-Y.; Hsieh, Y.-R.; Lin, J.-H.; Chen, F.-Y.; Chakravarthy, R.D.; Chung, P.-C.; Lin, H.-C.; Hung, S.-C. Reverse Thermo-Responsive Hydrogels Prepared from Pluronic F127 and Gelatin Composite Materials. RSC Adv. 2017, 7, 21252–21257. [Google Scholar] [CrossRef]
- Neumann, M.; Di Marco, G.; Iudin, D.; Viola, M.; Van Nostrum, C.F.; Van Ravensteijn, B.G.P.; Vermonden, T. Stimuli-Responsive Hydrogels: The Dynamic Smart Biomaterials of Tomorrow. Macromolecules 2023, 56, 8377–8392. [Google Scholar] [CrossRef]
- Khalid, M.Y.; Arif, Z.U.; Noroozi, R.; Zolfagharian, A.; Bodaghi, M. 4D Printing of Shape Memory Polymer Composites: A Review on Fabrication Techniques, Applications, and Future Perspectives. J. Manuf. Process. 2022, 81, 759–797. [Google Scholar] [CrossRef]
- Freeman, S.; Calabro, S.; Williams, R.; Jin, S.; Ye, K. Bioink Formulation and Machine Learning-Empowered Bioprinting Optimization. Front. Bioeng. Biotechnol. 2022, 10, 913579. [Google Scholar] [CrossRef]
- Qavi, I.; Halder, S.; Tan, G. Optimization of Printability of Bioinks with Multi-Response Optimization (MRO) and Artificial Neural Networks (ANN). Prog. Addit. Manuf. 2025, 10, 3573–3598. [Google Scholar] [CrossRef]
- Lee, J.; Oh, S.J.; An, S.H.; Kim, W.-D.; Kim, S.-H. Machine Learning-Based Design Strategy for 3D Printable Bioink: Elastic Modulus and Yield Stress Determine Printability. Biofabrication 2020, 12, 035018. [Google Scholar] [CrossRef] [PubMed]
- Stone, R.N.; Reeck, J.C.; Oxford, J.T. Advances in Cartilage Tissue Engineering Using Bioinks with Decellularized Cartilage and Three-Dimensional Printing. Int. J. Mol. Sci. 2023, 24, 5526. [Google Scholar] [CrossRef] [PubMed]
- Behan, K.; Dufour, A.; Garcia, O.; Kelly, D. Methacrylated Cartilage ECM-Based Hydrogels as Injectables and Bioinks for Cartilage Tissue Engineering. Biomolecules 2022, 12, 216. [Google Scholar] [CrossRef] [PubMed]
- Tong, X.; Ayushman, M.; Lee, H.-P.; Yang, F. Tuning Local Matrix Compliance Accelerates Mesenchymal Stem Cell Chondrogenesis in 3D Sliding Hydrogels. Biomaterials 2025, 317, 123112. [Google Scholar] [CrossRef]
- O’Donnell, S.; Levingstone, T. Development of Collagen-Based Bioinks for Cartilage Tissue Engineering Applications. AIP Conf. Proc. 2024, 3125, 030013. [Google Scholar] [CrossRef]
- Machour, M.; Meretzki, R.; Moshe-Haizler, Y.; Shuhmaher, M.; Safina, D.; Levy, M.M.; Levenberg, S. A Stiff Bioink for Hybrid Bioprinting of Vascularized Bone Tissue with Enhanced Mechanical Properties. Biomaterials 2024, 322, 123406. [Google Scholar] [CrossRef]
- Salg, G.A.; Blaeser, A.; Gerhardus, J.S.; Hackert, T.; Kenngott, H.G. Vascularization in Bioartificial Parenchymal Tissue: Bioink and Bioprinting Strategies. Int. J. Mol. Sci. 2022, 23, 8589. [Google Scholar] [CrossRef]
- Jeon, O.; Park, H.; Lee, M.S.; Alsberg, E. In Situ Cell-Only Bioprinting of Patterned Prevascular Tissue into Bioprinted High-Density Stem Cell-Laden Microgel Bioinks for Vascularized Bone Tissue Regeneration 2025. bioRxiv 2025. [Google Scholar] [CrossRef]
- Celikkin, N.; Mastrogiacomo, S.; Dou, W.; Heerschap, A.; Oosterwijk, E.; Walboomers, X.F.; Święszkowski, W. In Vitro and in Vivo Assessment of a 3D Printable Gelatin Methacrylate Hydrogel for Bone Regeneration Applications. J. Biomed. Mater. Res. B Appl. Biomater. 2022, 110, 2133–2145. [Google Scholar] [CrossRef]
- Badhe, R.V.; Chatterjee, A.; Bijukumar, D.; Mathew, M.T. Current Advancements in Bio-Ink Technology for Cartilage and Bone Tissue Engineering. Bone 2023, 171, 116746. [Google Scholar] [CrossRef]
- Kumar, P.; Sharma, J.; Kumar, R.; Najser, J.; Frantik, J.; Manuja, A.; Sunnam, N.; Praveenkumar, S. Advances in Bioink-Based 3D Printed Scaffolds: Optimizing Biocompatibility and Mechanical Properties for Bone Regeneration. Biomater. Sci. 2025, 13, 2556–2579. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Guo, R.; Zhang, Z.J. Bioink Formulations for Bone Tissue Regeneration. Front. Bioeng. Biotechnol. 2021, 9, 630488. [Google Scholar] [CrossRef]
- Distler, T.; Fournier, N.; Grünewald, A.; Polley, C.; Seitz, H.; Detsch, R.; Boccaccini, A.R. Polymer-Bioactive Glass Composite Filaments for 3D Scaffold Manufacturing by Fused Deposition Modeling: Fabrication and Characterization. Front. Bioeng. Biotechnol. 2020, 8, 552. [Google Scholar] [CrossRef]
- Wu, J.; Han, Y.; Fu, Q.; Hong, Y.; Li, L.; Cao, J.; Li, H.; Liu, Y.; Chen, Y.; Zhu, J.; et al. Application of Tissue-Derived Bioink for Articular Cartilage Lesion Repair. Chem. Eng. J. 2022, 450, 138292. [Google Scholar] [CrossRef]
- Xu, Y.; Jia, L.; Wang, Z.; Jiang, G.; Zhou, G.; Chen, W.; Chen, R. Injectable Photo-Crosslinking Cartilage Decellularized Extracellular Matrix for Cartilage Tissue Regeneration. Mater. Lett. 2020, 268, 127609. [Google Scholar] [CrossRef]
- ASTM F3659-24; Standard Guide for Bioinks Used in Bioprinting. ASTM International: West Conshohocken, PA, USA, 2024.
- GlobeNewswire FluidForm Bio Announces Advance in Bioprinted Tissue Therapies with Landmark Publication in Science Advances. 2025. Available online: https://www.biospace.com/press-releases/fluidform-bio-announces-advance-in-bioprinted-tissue-therapies-with-landmark-publication-in-science-advances (accessed on 25 April 2025).
- Molecular Devices 3D Bioprinting—BAB400 Bioprinter Solution. Available online: https://www.moleculardevices.com/products/3d-biology/3d-bioprinting-bab400-bioprinter-automation-solution (accessed on 13 August 2025).
- Sekar, M.P.; Budharaju, H.; Zennifer, A.; Sethuraman, S.; Vermeulen, N.; Sundaramurthi, D.; Kalaskar, D.M. Current Standards and Ethical Landscape of Engineered Tissues—3D Bioprinting Perspective. J. Tissue Eng. 2021, 12, 20417314211027677. [Google Scholar] [CrossRef] [PubMed]

| Parameter | Definition | Printability Impact | Cell Viability Impact |
|---|---|---|---|
| Shear-thinning | Viscosity decreases under shear | Enables extrusion | Reduces shear stress |
| Yield stress (τ0) | Minimum stress to initiate flow | Prevents nozzle clogging | Limits mixing forces |
| G′/G″ ratio | Elastic vs. viscous dominance | Structural integrity | Mitigates post-print deformation |
| Thixotropy | Time-dependent viscosity recovery | Prevents layer sagging | Protects cells post-extrusion |
| Shear-Thinning Behavior | Yield Stress and Structural Fidelity | Viscoelasticity (G′, G″) Dynamics | Thixotropic Recovery |
|---|---|---|---|
| High viscosity at rest (prevents cell settling). | Below τ0: Elastic solid behavior (maintains shape post-printing). | Ideal bioink: G′ > G″ at rest (elastic dominance for shape retention). | Rapid structural rebuild (<30 s) prevents layer fusion issues. |
| Sharp viscosity drop under shear (enables extrusion through nozzles) | Above τ0: Viscous flow (enables extrusion). | G″ > G′ during extrusion (viscous flow). | Enhanced cell viability due to reduced sustained shear stress |
| Critical shear rate for optimal extrusion marked (avoids cell damage). | τ0 range (2–10 Pa) for cell-friendly mixing and filament integrity | Loss tangent (tan δ = G″/G′) range: 0.25–0.45 for balance |
| Polymers | Natural | Synthetic |
|---|---|---|
| Advantages | Biocompatibility: Natural polymers are generally well-tolerated by the body, minimizing the risk of adverse immune responses. Bioactivity: Many natural polymers contain cell-binding motifs that promote cell adhesion, proliferation, and differentiation. Examples include RGD sequences in collagen and fibronectin. Biodegradability: Natural polymers are typically biodegradable, breaking down into non-toxic products that can be readily cleared by the body. Cell-Material Interactions: Natural polymers often mimic the natural ECM, providing a more physiologically relevant environment for cells. | Tunable Properties: Synthetic polymers can be precisely engineered to control their mechanical properties, degradation rate, and other characteristics. Reproducibility: Synthetic polymers are manufactured under controlled conditions, ensuring consistent batch-to-batch quality. Mechanical Strength: Synthetic polymers can be designed to provide high mechanical strength and durability. Processability: Synthetic polymers can be readily processed into a variety of shapes and structures using techniques such as electrospinning, 3D printing, and molding. |
| Limitations | Batch-to-Batch Variability: Natural polymers can exhibit significant variability in their composition and properties depending on the source and processing methods. Mechanical Properties: Natural polymers often lack the mechanical strength and durability required for load-bearing applications. Immunogenicity: Some natural polymers can elicit an immune response, particularly if they are derived from animal sources. Degradation Rate: Controlling the degradation rate of natural polymers can be challenging, potentially leading to premature scaffold collapse or delayed tissue regeneration. Processing Challenges: Some natural polymers can be difficult to process into desired shapes and structures. | Lack of Bioactivity: Synthetic polymers typically lack the inherent bioactivity of natural polymers, requiring modification to promote cell adhesion and differentiation. Biocompatibility Concerns: Some synthetic polymers can elicit adverse immune responses or release toxic degradation products. Hydrophobicity: Many synthetic polymers are hydrophobic, which can hinder cell adhesion and nutrient transport. Degradation Products: The degradation products of some synthetic polymers can be acidic, potentially causing inflammation and tissue damage. |
| Requirements for Tissue Maturation | Controlled Degradation: The degradation rate should match the rate of new tissue formation to provide structural support during the maturation process. Cell Adhesion Motifs: Incorporation of cell adhesion motifs (RGD) to promote cell attachment and spreading. Biocompatibility: Minimal inflammatory response and toxicity. Porosity: Sufficient porosity to allow for cell infiltration, nutrient transport, and waste removal. Mechanical Stability: Adequate mechanical strength to withstand physiological loads during the initial stages of tissue formation. | Biocompatibility: Non-toxic and non-immunogenic. Controlled Degradation: The degradation rate should be tailored to the rate of tissue formation. Mechanical Properties: Mechanical strength and elasticity should match the target tissue. Surface Modification: Surface modification with cell adhesion molecules (RGD peptides) to enhance cell attachment and spreading. Porosity: Interconnected pores to facilitate cell infiltration, nutrient transport, and waste removal. Sterilizability: Ability to be sterilized without compromising material properties. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elango, J.; Zamora-Ledezma, C. Rheological, Structural, and Biological Trade-Offs in Bioink Design for 3D Bioprinting. Gels 2025, 11, 659. https://doi.org/10.3390/gels11080659
Elango J, Zamora-Ledezma C. Rheological, Structural, and Biological Trade-Offs in Bioink Design for 3D Bioprinting. Gels. 2025; 11(8):659. https://doi.org/10.3390/gels11080659
Chicago/Turabian StyleElango, Jeevithan, and Camilo Zamora-Ledezma. 2025. "Rheological, Structural, and Biological Trade-Offs in Bioink Design for 3D Bioprinting" Gels 11, no. 8: 659. https://doi.org/10.3390/gels11080659
APA StyleElango, J., & Zamora-Ledezma, C. (2025). Rheological, Structural, and Biological Trade-Offs in Bioink Design for 3D Bioprinting. Gels, 11(8), 659. https://doi.org/10.3390/gels11080659










