Divergent Physiological Functions of Four Atg22-like Proteins in Conidial Germination, Development, and Virulence of the Entomopathogenic Fungus Beauveria bassiana
Abstract
1. Introduction
2. Materials and Methods
2.1. Strains, Media and Growth Conditions
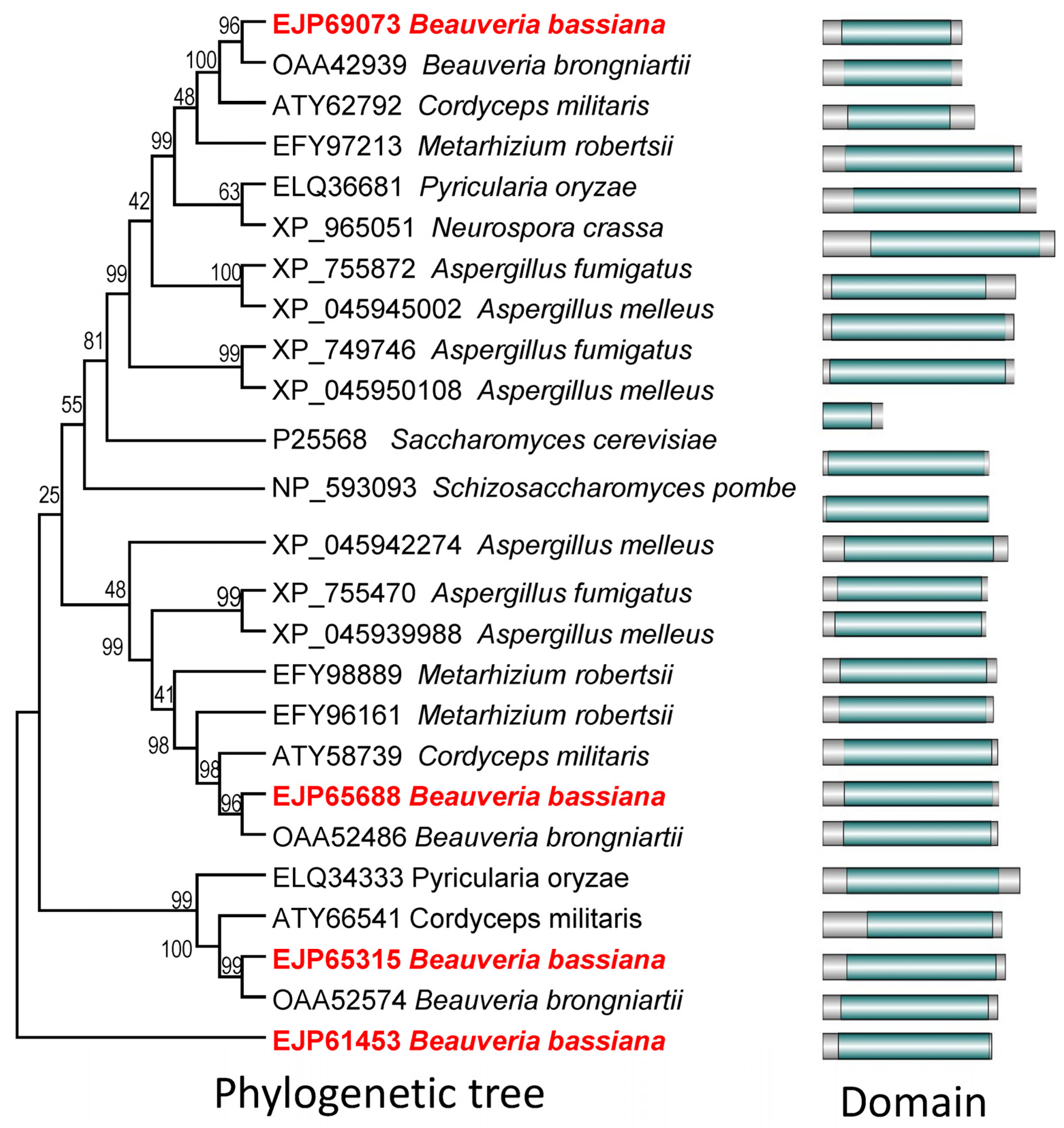
2.2. Bioinformatic Analyses and Sub-Cellular Localizations of Atg22-like Proteins in B. bassiana
2.3. Targeted Gene Disruption
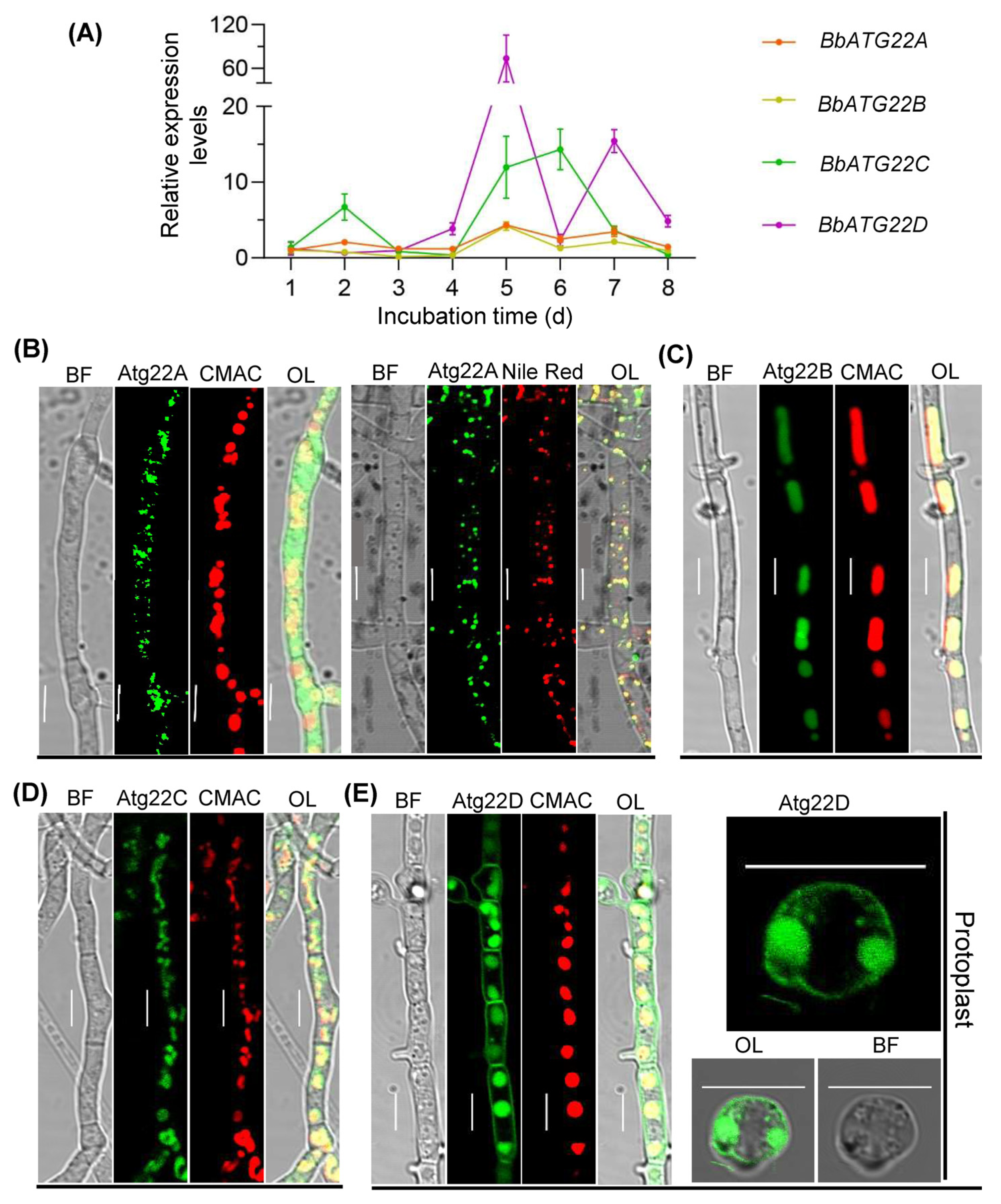
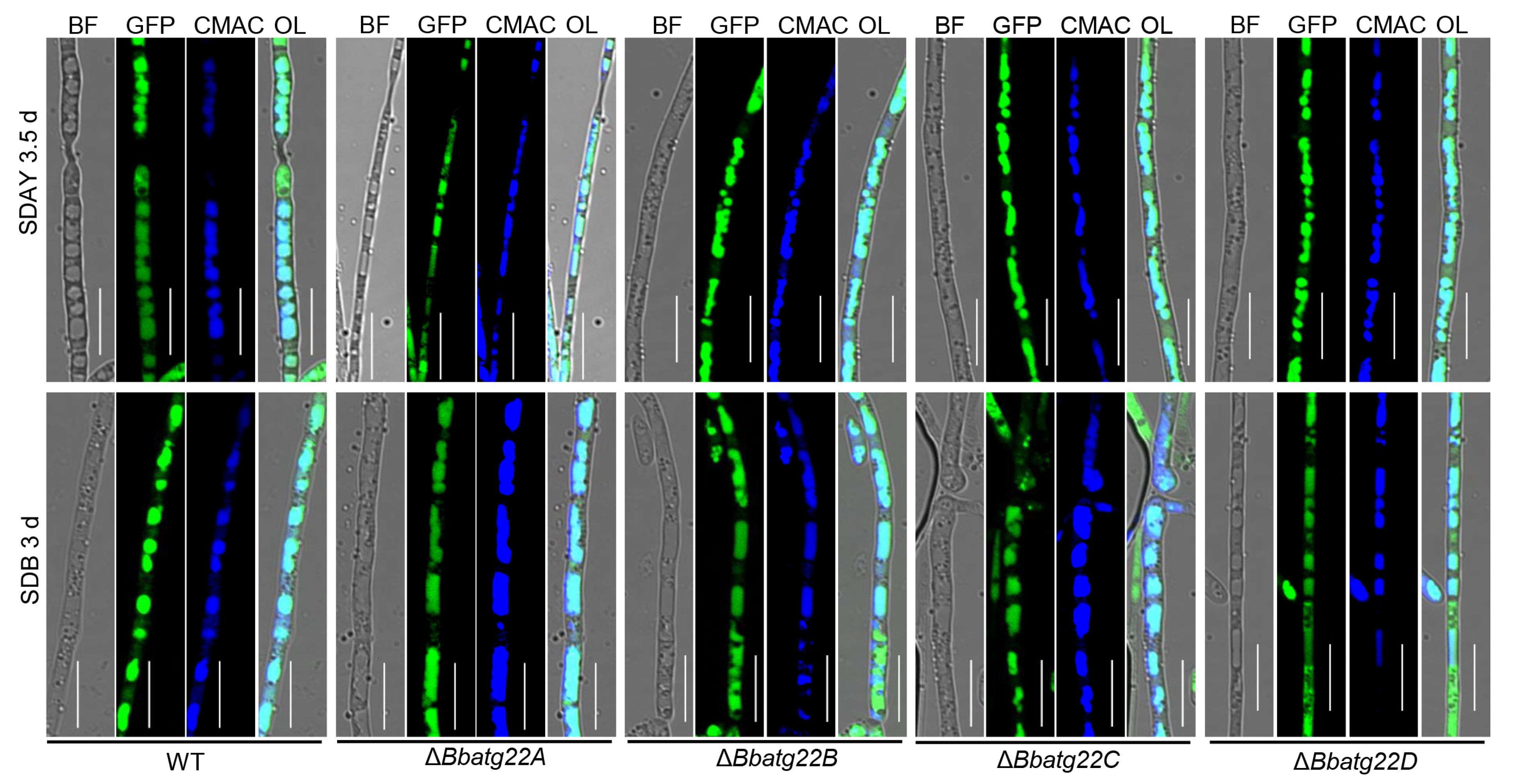
2.4. Visualizing Autophagic Flux in Fungal Development
2.5. Assays for Conidial Germination, Fungal Growth, and Development
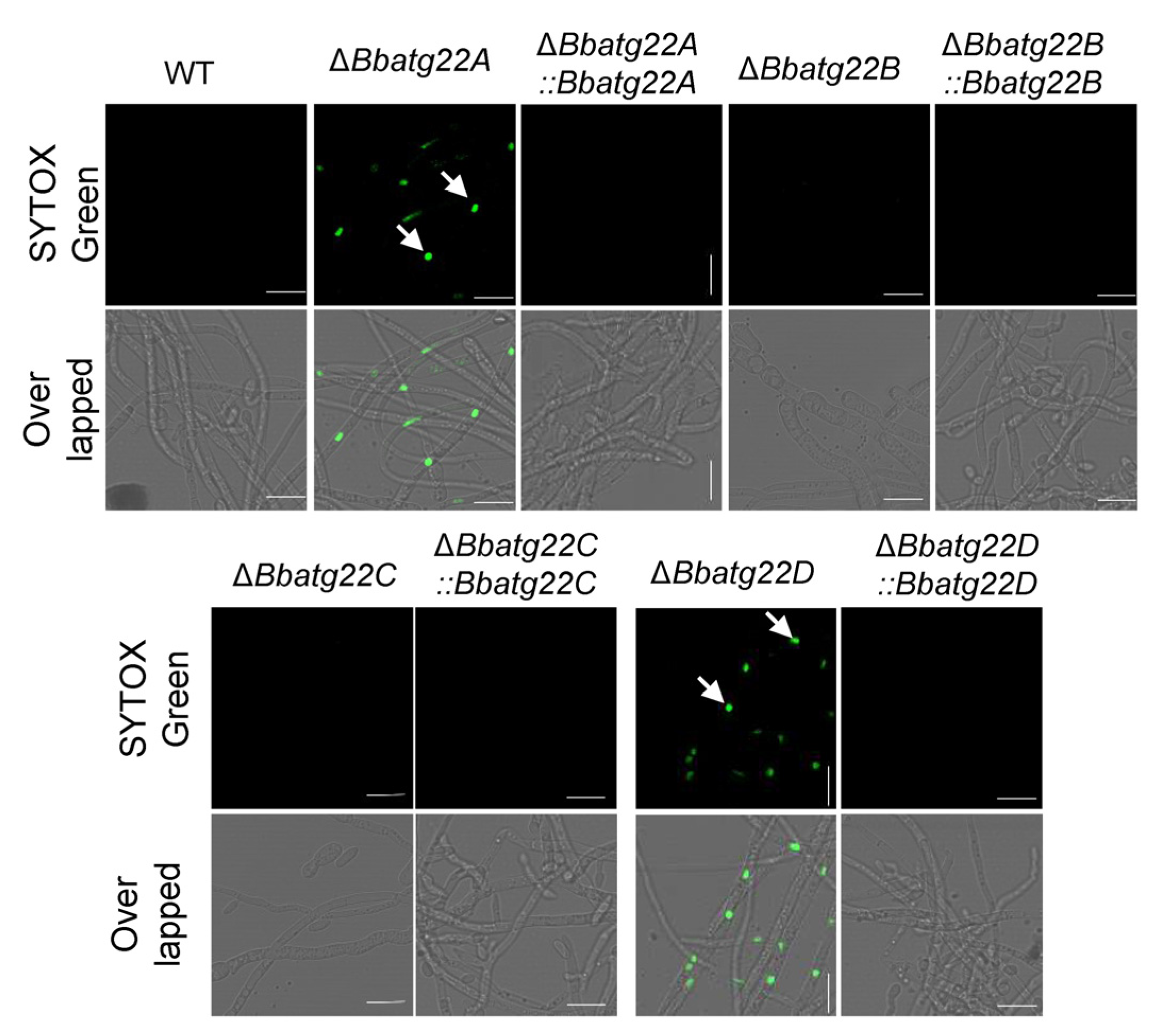
2.6. Assays for Membrane Integrity
2.7. Pathogenicity Assays
2.8. qRT-PCR Assays
2.9. Statistical Analyses
3. Results
3.1. Bioinformatic Analyses of Yeast Atg22 Orthologs in B. bassiana
3.2. Gene Expression and Cell Biology of Atg22-like Proteins
3.3. Atg22-like Proteins Had Different Contributions to Cell Membrane Integrity
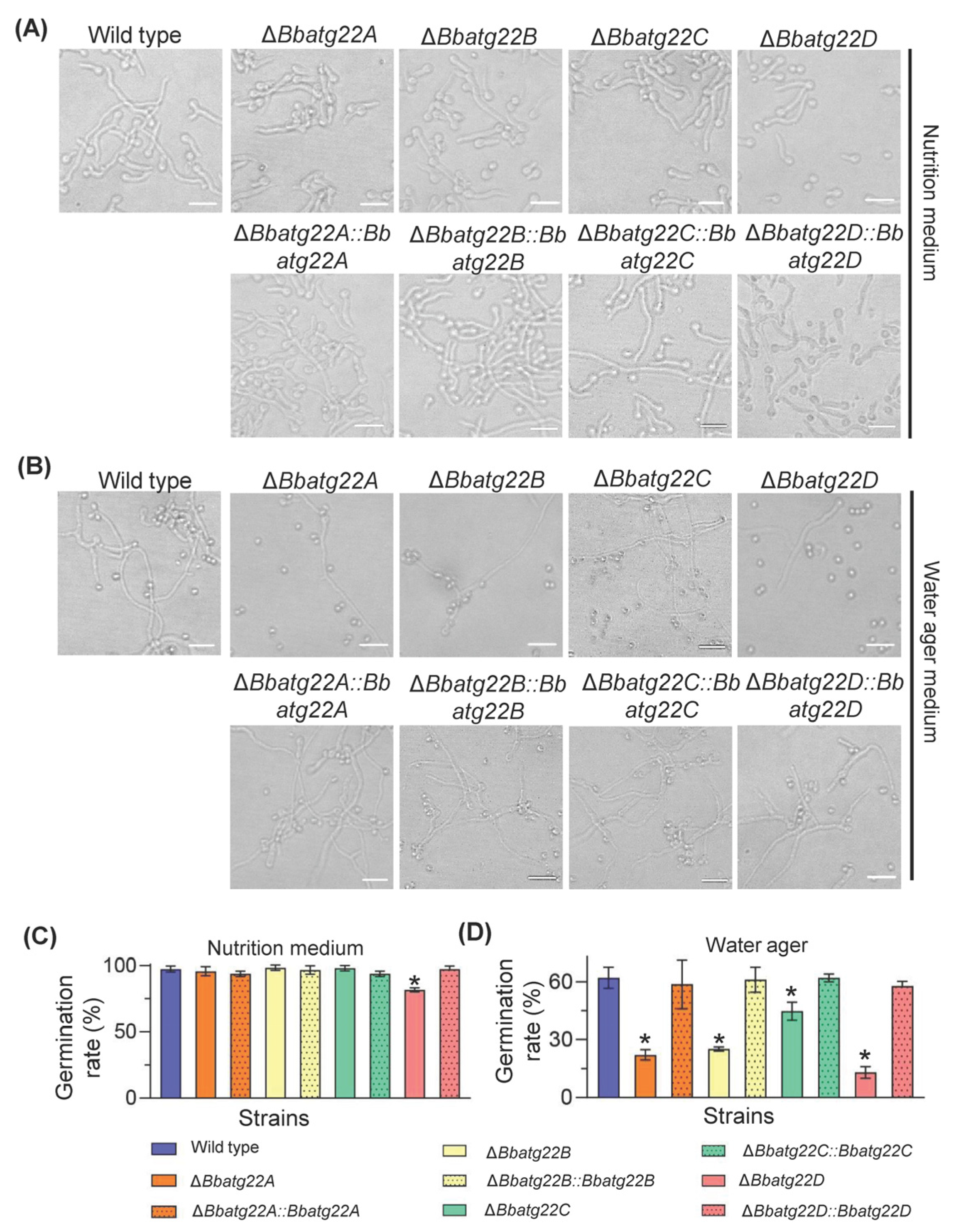
3.4. Atg22-like Proteins Is Required for Conidial Germination under Nutrient-Limitation Condition
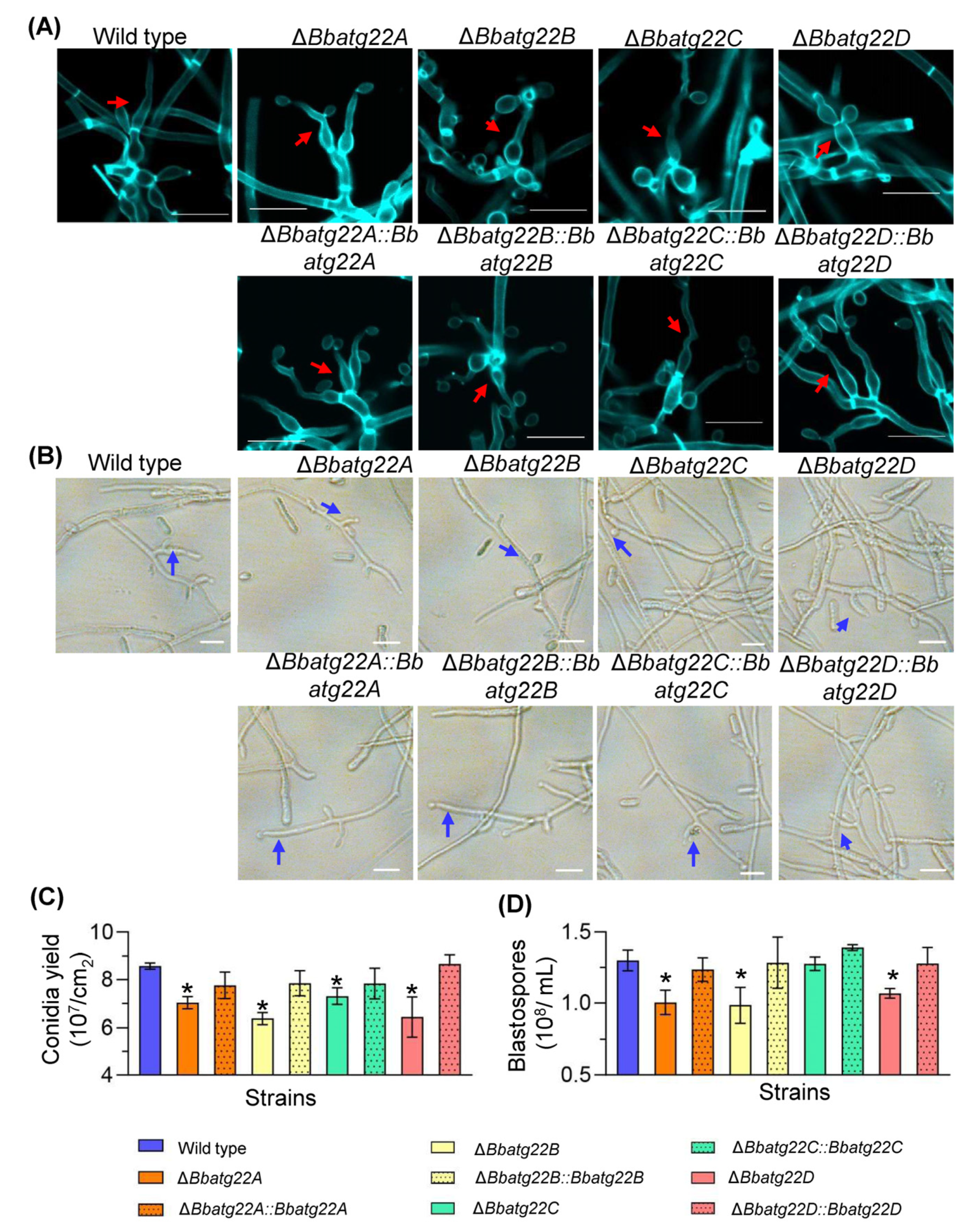
3.5. Atg22-like Proteins Are Involved in Fungal Development
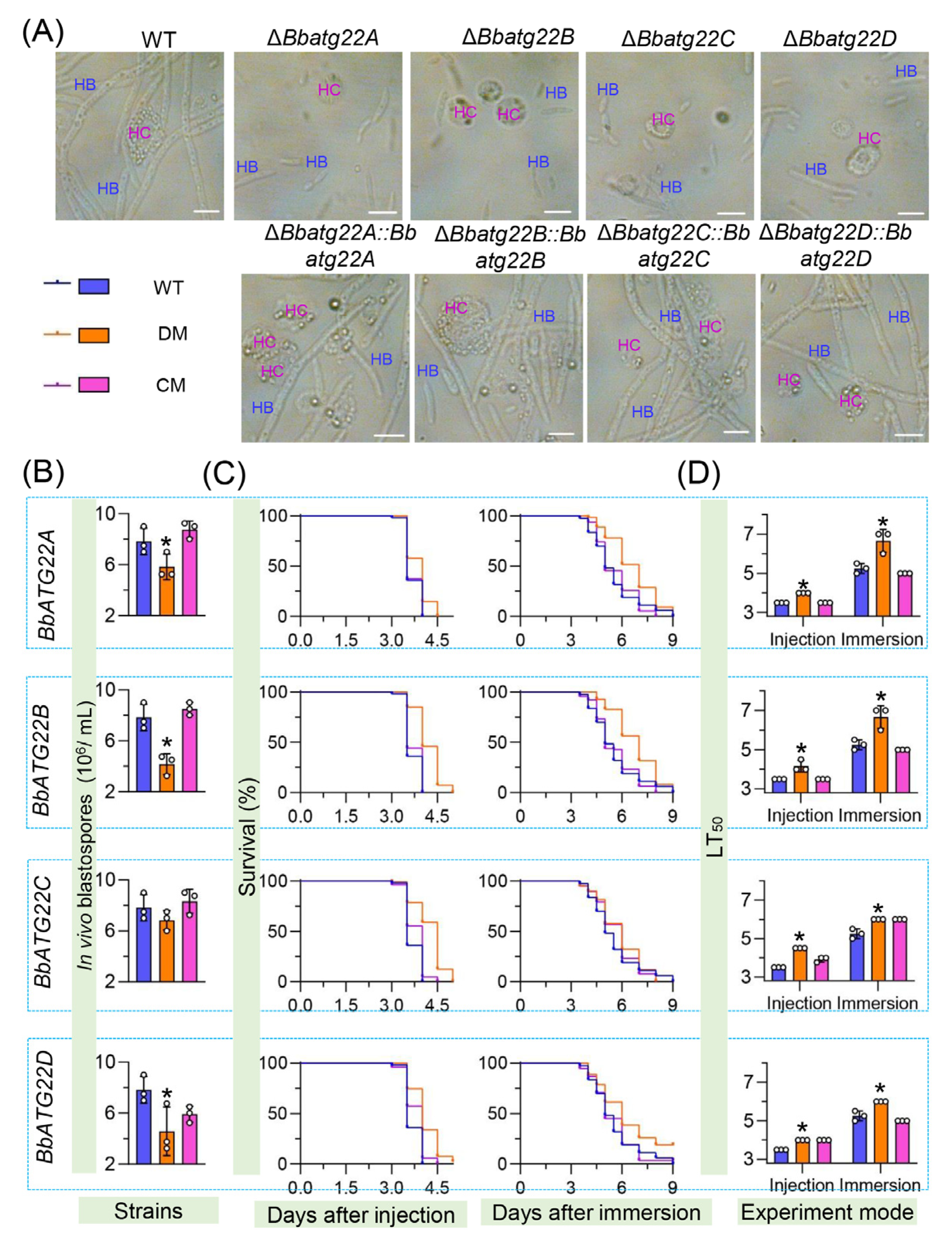
3.6. Atg22-like Proteins Significantly Contributes to Fungal Virulence
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, Z.Z.; Alves, S.B.; Roberts, D.W.; Fan, M.; Delalibera, I.; Tang, J.; Lopes, R.B.; Faria, M.; Rangel, D.E.N. Biological control of insects in Brazil and China: History, current programs and reasons for their successes using entomopathogenic fungi. Biocontrol. Sci. Technol. 2010, 20, 117–136. [Google Scholar] [CrossRef]
- De la Cruz, Q.R.; Roussos, S.; Hernández, D.; Rodríguez, R.; Castillo, F.; Aguilar, C.N. Challenges and opportunities of the biopesticides production by solid-state fermentation: Filamentous fungi as a model. Crit. Rev. Biotechnol. 2015, 35, 326–333. [Google Scholar] [CrossRef]
- Reggiori, F.; Klionsky, D.J. Autophagic processes in yeast: Mechanism, machinery and regulation. Genetics 2013, 194, 314–361. [Google Scholar] [CrossRef]
- Ying, S.H.; Liu, J.; Chu, X.L.; Xie, X.Q.; Feng, M.G. The autophagy-related genes BbATG1 and BbATG8 have different functions in differentiation, stress resistance and virulence of mycopathogen Beauveria bassiana. Sci. Rep. 2016, 6, 26376. [Google Scholar] [CrossRef]
- Lin, H.Y.; Lei, J.H.; Ding, J.L.; Peng, Y.J.; Zhang, H.; Feng, M.G.; Ying, S.H. Peroxin 14 tags peroxisomes and interacts with Nbr1 for pexophagy in the filamentous insect pathogenic fungus Beauveria bassiana. Autophagy Rep. 2023, 2, 2168337. [Google Scholar] [CrossRef]
- Ying, S.H.; Feng, M.G. Insight into vital role of autophagy in sustaining biological control potential of fungal pathogens against pest insects and nematodes. Virulence 2019, 10, 429–437. [Google Scholar] [CrossRef]
- Fukuda, T.; Kanki, T. Atg43, a novel autophagy-related protein, serves as a mitophagy receptor to bridge mitochondria with phagophores in fission yeast. Autophagy 2021, 17, 826–827. [Google Scholar] [CrossRef]
- Meijer, W.H.; van der Klei, I.J.; Veenhuis, M.; Kiel, J.A.K.W. ATG genes involved in non-selective autophagy are conserved from yeast to man, but the selective Cvt and pexophagy pathways also require organism-specific genes. Autophagy 2007, 3, 106–116. [Google Scholar] [CrossRef]
- Lin, H.Y.; Ding, J.L.; Peng, Y.J.; Feng, M.G.; Ying, S.H. Proteomic and phosphoryproteomic investigations reveal that autophagy-related protein 1, a protein kinase for autophagy initiation, synchronously deploys phospho-regulation on ubiquitin-like conjugation system in mycopathogen Beauveria bassiana. mSystems 2022, 7, e01463-21. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, J.; Xie, X.Q.; Keyhani, N.O.; Feng, M.G.; Ying, S.H. The autophagy gene BbATG5, involved in the formation of the autophagosome, contributes to cell differentiation and growth but is dispensable for pathogenesis in the entomopathogenic fungus Beauveria bassiana. Microbiol. SGM 2013, 159, 243–252. [Google Scholar] [CrossRef]
- Lin, H.Y.; Wang, J.J.; Feng, M.G.; Ying, S.H. Autophagy-related gene ATG7 participates in the asexual development, stress response and virulence of filamentous insect pathogenic fungus Beauveria bassiana. Curr. Genet. 2019, 65, 1015–1024. [Google Scholar] [CrossRef]
- Hou, J.; Wang, J.J.; Lin, H.Y.; Feng, M.G.; Ying, S.H. Roles of autophagy related genes in conidiogenesis and blastospore formation, virulence, and stress response of Beauveria bassiana. Fungal Biol. 2020, 124, 1052–1057. [Google Scholar] [CrossRef]
- Ding, J.L.; Peng, Y.J.; Chu, X.L.; Feng, M.G.; Ying, S.H. Autophagy-related gene BbATG11 is indispensable for pexophagy and mitophagy, and contributes to stress response, conidiation and virulence in the insect mycopathogen Beauveria bassiana. Environ. Microbiol. 2018, 20, 3309–3324. [Google Scholar] [CrossRef]
- Yang, Z.; Huang, J.; Geng, J.; Nair, U.; Klionsky, D.J. Atg22 recycles amino acids to link the degradative and recycling functions of autophagy. Mol. Biol. Cell. 2006, 17, 5094–5104. [Google Scholar] [CrossRef]
- Letunic, I.; Khedkar, S.; Bork, P. SMART: Recent updates, new developments and status in 2020. Nucleic Acids Res. 2021, 49, D458–D460. [Google Scholar] [CrossRef]
- Tamura, K.; Peterson, D.; Peterson, N.; Stecher, G.; Nei, M.; Kumar, S. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 2011, 28, 2731–2739. [Google Scholar] [CrossRef]
- Wang, J.J.; Peng, Y.J.; Feng, M.G.; Ying, S.H. Functional analysis of the mitochondrial gene mitofilin in the filamentous entomopathogenic fungus Beauveria bassiana. Fungal Genet. Biol. 2019, 132, 103250. [Google Scholar] [CrossRef]
- Wang, J.J.; Peng, Y.J.; Ding, J.L.; Feng, M.G.; Ying, S.H. Mitochondrial fission is necessary for mitophagy, development and virulence of the insect pathogenic fungus Beauveria bassiana. J. Appl. Microbiol. 2020, 129, 411–421. [Google Scholar] [CrossRef]
- Guo, N.; Qian, Y.; Zhang, Q.; Chen, X.; Zeng, G.; Zhang, X.; Mi, W.; Xu, C.; St Leger, R.J.; Fang, W. Alternative transcription start site selection in Mr-OPY2 controls lifestyle transitions in the fungus Metarhizium robertsii. Nat. Commun. 2017, 8, 1565. [Google Scholar] [CrossRef]
- Peng, Y.J.; Wang, J.J.; Lin, H.Y.; Ding, J.L.; Feng, M.G.; Ying, S.H. HapX, an indispensable bZIP transcription factor for iron acquisition, regulates infection initiation by orchestrating conidial oleic acid homeostasis and cytomembrane functionality in mycopathogenBeauveria bassiana. mSystems 2020, 5, e00695-20. [Google Scholar] [CrossRef]
- Peng, Y.J.; Zhang, H.; Feng, M.G.; Ying, S.H. Steryl acetyl hydrolase 1 (BbSay1) links lipid homeostasis to conidiogenesis and virulence in the entomopathogenic fungus Beauveria bassiana. J. Fungi 2022, 8, 292. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Urquiza, A.; Keyhani, N.O. Molecular genetics of Beauveria bassiana infection of insects. Adv. Genet. 2016, 94, 165–249. [Google Scholar]
- Kim, S.; Lee, S.J.; Nai, Y.S.; Yu, J.S.; Lee, M.R.; Yang, Y.T.; Kim, J.S. Characterization of T-DNA insertion mutants with decreased virulence in the entomopathogenic fungus Beauveria bassiana JEF-007. Appl. Microbiol. Biotechnol. 2016, 100, 8889–8900. [Google Scholar] [CrossRef] [PubMed]
- Bidochka, M.J.; Low, N.H.; Khachatourians, G.G. Carbohydrate storage in the entomopathogenic fungus Beauveria bassiana. Appl. Environ. Microbiol. 1990, 56, 3186–3190. [Google Scholar] [CrossRef]
- Ding, J.L.; Hou, J.; Feng, M.G.; Ying, S.H. Transcriptomic analyses reveal comprehensive responses of insect hemocytes to mycopathogen Beauveria bassiana, and fungal virulence-related cell wall protein assists pathogen to evade host cellular defense. Virulence 2020, 11, 1352–1365. [Google Scholar] [CrossRef] [PubMed]
- Gauthier, G.M. Dimorphism in fungal pathogens of mammals, plants, and insects. PLoS Pathog. 2015, 11, e1004608. [Google Scholar] [CrossRef]
- Lei, J.H.; Lin, H.Y.; Ding, J.L.; Feng, M.G.; Ying, S.H. Functional characterization of two homologs of yeast acetyl-coenzyme A synthetase in the entomopathogenic fungus Beauveria bassiana. Arch. Microbiol. 2022, 204, 653. [Google Scholar] [CrossRef]
- Lin, H.Y.; Pang, M.Y.; Feng, M.G.; Ying, S.H. A peroxisomal sterol carrier protein 2 Scp2 contributes to lipid trafficking in differentiation and virulence of the insect pathogenic fungus Beauveria bassiana. Fungal Genet. Biol. 2022, 158, 103651. [Google Scholar] [CrossRef]
- Wang, X.X.; He, P.H.; Feng, M.G.; Ying, S.H. BbSNF1 contributes to cell differentiation, extracellular acidification, and virulence in Beauveria bassiana, a filamentous entomopathogenic fungus. Appl. Microbiol. Biotechnol. 2014, 98, 8657–8673. [Google Scholar] [CrossRef]
- Kikuma, T.; Arioka, M.; Kitamoto, K. Autophagy during conidiation and conidial germination in filamentous fungi. Autophagy 2007, 3, 128–129. [Google Scholar] [CrossRef] [PubMed]
- Khalid, A.R.; Zhang, S.; Luo, X.; Mehmood, K.; Rahim, J.; Shaheen, H.; Dong, P.; Qiu, D.; Ren, M. Role of Autophagy-related gene atg22 in developmental process and virulence of Fusarium oxysporum. Genes 2019, 10, 365. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ding, J.-L.; Zhang, H.; Feng, M.-G.; Ying, S.-H. Divergent Physiological Functions of Four Atg22-like Proteins in Conidial Germination, Development, and Virulence of the Entomopathogenic Fungus Beauveria bassiana. J. Fungi 2023, 9, 262. https://doi.org/10.3390/jof9020262
Ding J-L, Zhang H, Feng M-G, Ying S-H. Divergent Physiological Functions of Four Atg22-like Proteins in Conidial Germination, Development, and Virulence of the Entomopathogenic Fungus Beauveria bassiana. Journal of Fungi. 2023; 9(2):262. https://doi.org/10.3390/jof9020262
Chicago/Turabian StyleDing, Jin-Li, Hao Zhang, Ming-Guang Feng, and Sheng-Hua Ying. 2023. "Divergent Physiological Functions of Four Atg22-like Proteins in Conidial Germination, Development, and Virulence of the Entomopathogenic Fungus Beauveria bassiana" Journal of Fungi 9, no. 2: 262. https://doi.org/10.3390/jof9020262
APA StyleDing, J.-L., Zhang, H., Feng, M.-G., & Ying, S.-H. (2023). Divergent Physiological Functions of Four Atg22-like Proteins in Conidial Germination, Development, and Virulence of the Entomopathogenic Fungus Beauveria bassiana. Journal of Fungi, 9(2), 262. https://doi.org/10.3390/jof9020262







