Development of a Real-Time PCR Assay to Identify and Distinguish between Cryptococcus neoformans and Cryptococcus gattii Species Complexes
Abstract
1. Introduction
2. Materials and Methods
2.1. Control DNA and Clinical Specimens
2.2. DNA Extraction
2.3. Primer and Probe Design
| Species Primer and Probe | Sequence (5′-3′) |
|---|---|
| Cryptococcus neoformans-gattii complex | |
| Cryptococcus cyt b F | TTCTAGCAGCTCTAGCTCTAG |
| Cryptococcus cyt b R | GCATTTGAGCTAATACCTTCAGG |
| Cryptococcus cyt b probe | 6FAM-TACATATGCTAACACTTCACACACA-BHQ1 |
| Human β globin (internal control) [23] | |
| HBG F | GAAGAGCCAAGGACAGGTAC |
| HBG R | CACCAACTTCATCCACGTTCAC |
| HBG probe | TxRd-TCAAACAGACACCATGGTGCACCTG-BHQ2 |
| Positive Control DNA | |
| C. neoformans (82 bp) | TTCACTATCTACTACCATTTATTCTAGCAGCTCTA-GCTCTAGTACAATGCTAACACTTCACACACACGGTAGCTCAAACCCTGAAGGTATTAGCTCAAATGC-TGAAAAGGCACCAATGCATCCATACTTTA |
| C. gattii (82 bp) | TTCACTATCTACTACCATTTATTCTAGCAGCTCTA-GCTCTAGTACATATGCTAACACTACACTCACAT-GGTAGTTCAAATCCTGAAGGTATTAGCTCAAAT-GCAGAAAAGGCACCAATGCATCCATACTTTA |
2.4. Multiplex Real-Time PCR and High-Resolution Melt (HRM) Curve Analysis
3. Results
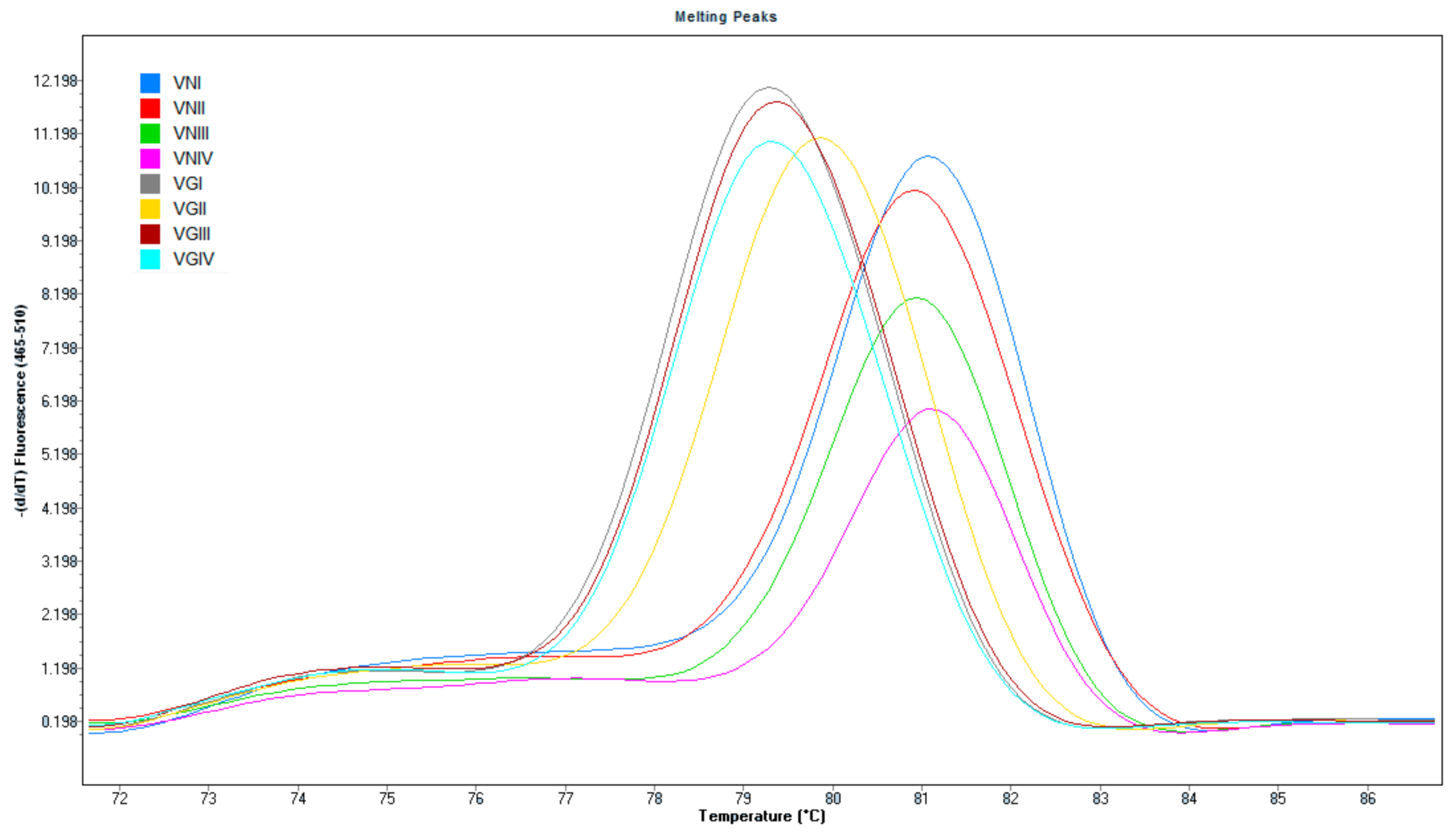
3.1. PCR Specificity and Sensitivity
3.2. PCR Performance on Clinical Specimens
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Baddley, J.W.; Chen, S.C.; Huisingh, C.; Benedict, K.; DeBess, E.E.; Galanis, E.; Jackson, B.R.; MacDougall, L.; Marsden-Haug, N.; Oltean, H.; et al. MSG07: An International Cohort Study Comparing Epidemiology and Outcomes of Patients With Cryptococcus neoformans or Cryptococcus gattii Infections. Clin. Infect. Dis. 2021, 73, 1133–1141. [Google Scholar] [CrossRef] [PubMed]
- Hagen, F.; Khayhan, K.; Theelen, B.; Kolecka, A.; Polacheck, I.; Sionov, E.; Falk, R.; Parnmen, S.; Lumbsch, T.; Boekhout, T. Recognition of seven species in the Cryptococcus gattii/Cryptococcus neoformans species complex. Fungal Genet. Biol. 2015, 78, 16–48. [Google Scholar] [CrossRef]
- Donnelly, J.P.; Chen, S.C.; Kauffman, C.A.; Steinbach, W.J.; Baddley, J.W.; Verweij, P.E.; Clancy, C.J.; Wingard, J.R.; Lockhart, S.R.; Groll, A.H.; et al. Revision and Update of the Consensus Definitions of Invasive Fungal Disease From the European Organization for Research and Treatment of Cancer and the Mycoses Study Group Education and Research Consortium. Clin. Infect. Dis. 2020, 71, 1367–1376. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.C.; Hall, V.; Cooper, C.; Grigoriadis, G.; Beardsley, J.; Sorrell, T.C.; Heath, C.H.; Slavin, M.A.; Thursky, K.A.; Roberts, J.A.; et al. Consensus guidelines for the diagnosis and management of cryptococcosis and rare yeast infections in the haematology/oncology setting, 2021. Intern. Med. J. 2021, 51 (Suppl. S7), 118–142. [Google Scholar] [CrossRef] [PubMed]
- Beardsley, J.; Sorrell, T.C.; Chen, S.C. Central Nervous System Cryptococcal Infections in Non-HIV Infected Patients. J. Fungi 2019, 5, 71. [Google Scholar] [CrossRef]
- Hsiao, P.-J.; Cheng, H.; Kao, Y.-H.; Chiu, C.-C.; Chiang, W.-F.; Kuo, C.-C.; Chuu, C.-P.; Wu, K.-A. Comparison of laboratory diagnosis, clinical manifestation, and management of pulmonary cryptococcosis: Report of the clinical scenario and literature review. Clin. Chim. Acta 2022, 524, 78–83. [Google Scholar] [CrossRef]
- Trilles, L.; Wang, B.; Firacative, C.; Lazéra, M.D.S.; Wanke, B.; Meyer, W. Identification of the Major Molecular Types of Cryptococcus neoformans and C. gattii by Hyperbranched Rolling Circle Amplification. PLoS ONE 2014, 9, e94648. [Google Scholar] [CrossRef]
- Tavares, E.R.; Azevedo, C.S.; Panagio, L.A.; Pelisson, M.; Pinge-Filho, P.; Venâncio, E.J.; Barros, T.F.; Yamada-Ogatta, S.F.; Yamauchi, L.M. Accurate and sensitive real-time PCR assays using intergenic spacer 1 region to differentiate Cryptococcus gattii sensu lato and Cryptococcus neoformans sensu lato. Med. Mycol. 2016, 54, 89–96. [Google Scholar]
- Kidd, S.E.; Chen, S.C.; Meyer, W.; Meyer, W.; Halliday, C.L. A New Age in Molecular Diagnostics for Invasive Fungal Disease: Are We Ready? Front. Microbiol. 2019, 10, 2903. [Google Scholar] [CrossRef]
- Lau, A.; Sorrell, T.C.; Chen, S.; Stanley, K.; Iredell, J.; Halliday, C. Multiplex tandem PCR: A novel platform for rapid detection and identification of fungal pathogens from blood culture specimens. J. Clin. Microbiol. 2008, 46, 3021–3027. [Google Scholar] [CrossRef]
- Tansarli, G.S.; Chapin, K.C. Diagnostic test accuracy of the BioFire® FilmArray® meningitis/encephalitis panel: A systematic review and meta-analysis. Clin. Microbiol. Infect. 2020, 26, 281–290. [Google Scholar] [CrossRef] [PubMed]
- Van, T.T.; Kim, T.H.; Butler-Wu, S.M. Evaluation of the Biofire FilmArray meningitis/encephalitis assay for the detection of Cryptococcus neoformans/gattii. Clin. Microbiol. Infect. 2020, 26, 1375–1379. [Google Scholar] [CrossRef] [PubMed]
- Lau, A.; Sorrell, T.C.; Lee, O.; Stanley, K.; Halliday, C. Colony multiplex-tandem PCR for rapid, accurate identification of fungal cultures. J. Clin. Microbiol. 2008, 46, 4058–4060. [Google Scholar] [CrossRef] [PubMed]
- Lewis, P.O.; Lanier, C.G.; Patel, P.D.; Krolikowski, W.D.; Krolikowski, M.A. False negative diagnostic errors with polymerase chain reaction for the detection of cryptococcal meningoencephalitis. Med. Mycol. 2020, 58, 408–410. [Google Scholar] [CrossRef] [PubMed]
- Chew, K.L.; Lee, C.K.; Cross, G.B.; Lum, L.H.W.; Yan, B.; Jureen, R. Culture-confirmed cryptococcal meningitis not detected by Cryptococcus PCR on the Biofire meningitis/encephalitis panel((R)). Clin. Microbiol. Infect. 2018, 24, 791–792. [Google Scholar] [CrossRef]
- Chong, B.S.W.; Kennedy, K.J. Comparison of a commercial real-time PCR panel to routine laboratory methods for the diagnosis of meningitis-encephalitis. Pathology 2021, 53, 635–638. [Google Scholar] [CrossRef]
- Yokoyama, K.; Biswas, S.K.; Miyaji, M.; Nishimura, K. Identification and phylogenetic relationship of the most common pathogenic Candida species inferred from mitochondrial cytochrome b gene sequences. J. Clin. Microbiol. 2000, 38, 4503–4510. [Google Scholar] [CrossRef]
- Biswas, S.K.; Yokoyama, K.; Wang, L.; Nishimura, K.; Miyaji, M. Identification of Candida dubliniensis based on the specific amplification of mitochondrial cytochrome b gene. Nihon Ishinkin Gakkai Zasshi 2001, 42, 95–98. [Google Scholar] [CrossRef]
- Biswas, S.K.; Wang, L.; Yokoyama, K.; Nishimura, K. Molecular analysis of Cryptococcus neoformans mitochondrial cytochrome b gene sequences. J. Clin. Microbiol. 2003, 41, 5572–5576. [Google Scholar] [CrossRef]
- Lau, A.; Chen, S.; Sorrell, T.; Carter, D.; Malik, R.; Martin, P.; Halliday, C. Development and clinical application of a panfungal PCR assay to detect and identify fungal DNA in tissue specimens. J. Clin. Microbiol. 2007, 45, 380–385. [Google Scholar] [CrossRef]
- Garnham, K.; Halliday, C.L.; Kok, J.; Jayawardena, M.; Ahuja, V.; Green, W.; Chen, S.C.-A. Knowledge at what cost? An audit of the utility of panfungal PCR performed on bronchoalveolar lavage fluid specimens at a tertiary mycology laboratory. Pathology 2020, 52, 584–588. [Google Scholar] [CrossRef] [PubMed]
- Sievers, F.; Wilm, A.; Dineen, D.; Gibson, T.J.; Karplus, K.; Li, W.; Lopez, R.; McWilliam, H.; Remmert, M.; Söding, J.; et al. Fast, scalable generation of high-quality protein multiple sequence alignments using Clustal Omega. Mol. Syst. Biol. 2011, 7, 539. [Google Scholar] [CrossRef] [PubMed]
- Zinkernagel, A.S.; Gmur, R.; Fenner, L.; Schoedon, G.; Schneemann, M. Marginal and subgingival plaque—A natural habitat of Tropheryma whipplei? Infection 2003, 31, 86–91. [Google Scholar] [CrossRef]
- Firacative, C.; Trilles, L.; Meyer, W. MALDI-TOF MS enables the rapid identification of the major molecular types within the Cryptococcus neoformans/C. gattii species complex. PLoS ONE 2012, 7, e37566. [Google Scholar] [CrossRef]
- Klein, K.R.; Hall, L.; Deml, S.M.; Rysavy, J.M.; Wohlfiel, S.L.; Wengenack, N.L. Identification of Cryptococcus gattii by use of L-canavanine glycine bromothymol blue medium and DNA sequencing. J. Clin. Microbiol. 2009, 47, 3669–3672. [Google Scholar] [CrossRef] [PubMed]
- Cogliati, M. Global Molecular Epidemiology of Cryptococcus neoformans and Cryptococcus gattii: An Atlas of the Molecular Types. Scientifica 2013, 2013, 675213. [Google Scholar] [CrossRef]
- Lockhart, S.R.; Bialek, R.; Kibbler, C.C.; Cuenca-Estrella, M.; Jensen, H.E.; Kontoyiannis, D.P. Molecular Techniques for Genus and Species Determination of Fungi From Fresh and Paraffin-Embedded Formalin-Fixed Tissue in the Revised EORTC/MSGERC Definitions of Invasive Fungal Infection. Clin. Infect. Dis. 2021, 72, S109–S113. [Google Scholar] [CrossRef]
- Katsu, M.; Kidd, S.; Ando, A.; Moretti-Branchini, M.L.; Mikami, Y.; Nishimura, K.; Meyer, W. The internal transcribed spacers and 5.8S rRNA gene show extensive diversity among isolates of the Cryptococcus neoformans species complex. FEMS Yeast Res. 2004, 4, 377–388. [Google Scholar] [CrossRef][Green Version]
- Zeller, I.; Schabereiter-Gurtner, C.; Mihalits, V.; Selitsch, B.; Barousch, W.; Hirschl, A.M.; Makristathis, A.; Willinger, B. Detection of fungal pathogens by a new broad range real-time PCR assay targeting the fungal ITS2 region. J. Med. Microbiol. 2017, 66, 1383–1392. [Google Scholar] [CrossRef]
- Trubiano, J.; Dennison, A.; Morrissey, C.; Chua, K.Y.; Halliday, C.L.; Chen, S.C.-A.; Spelman, D. Clinical utility of panfungal polymerase chain reaction for the diagnosis of invasive fungal disease: A single center experience. Med. Mycol. 2016, 54, 138–146. [Google Scholar] [CrossRef]
- Soucek, D.K.; Dumkow, L.E.; VanLangen, K.M.; Jameson, A.P. Cost Justification of the BioFire FilmArray Meningitis/Encephalitis Panel Versus Standard of Care for Diagnosing Meningitis in a Community Hospital. J. Pharm. Pract. 2019, 32, 36–40. [Google Scholar] [CrossRef] [PubMed]
| Specimen Type (n) | Culture Positive | CRAG | Histology | Panfungal PCR | Targeted PCR C. neoformans | Targeted PCR C. gattii |
|---|---|---|---|---|---|---|
| FFPE tissue (25) | N/A | N/A | 13 | 19 | 20 | 4 |
| CSF (14) | 2 | 3 | N/A | 11 | 7 | 6 |
| BALF (6) | 3 | N/A | N/A | 3 | 2 | 4 |
| FNA (5) | 2 | N/A | N/A | 5 | 4 | 1 |
| IS (3) | 0 | N/A | N/A | N/A | 3 | 0 |
| Not Specified (2) | 1 | N/A | N/A | 2 | 2 | 0 |
| TOTAL (55) | 8 | 3 | 13 | 40 | 38 | 15 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tay, E.; Chen, S.C.-A.; Green, W.; Lopez, R.; Halliday, C.L. Development of a Real-Time PCR Assay to Identify and Distinguish between Cryptococcus neoformans and Cryptococcus gattii Species Complexes. J. Fungi 2022, 8, 462. https://doi.org/10.3390/jof8050462
Tay E, Chen SC-A, Green W, Lopez R, Halliday CL. Development of a Real-Time PCR Assay to Identify and Distinguish between Cryptococcus neoformans and Cryptococcus gattii Species Complexes. Journal of Fungi. 2022; 8(5):462. https://doi.org/10.3390/jof8050462
Chicago/Turabian StyleTay, Enoch, Sharon C-A. Chen, Wendy Green, Ronald Lopez, and Catriona L. Halliday. 2022. "Development of a Real-Time PCR Assay to Identify and Distinguish between Cryptococcus neoformans and Cryptococcus gattii Species Complexes" Journal of Fungi 8, no. 5: 462. https://doi.org/10.3390/jof8050462
APA StyleTay, E., Chen, S. C.-A., Green, W., Lopez, R., & Halliday, C. L. (2022). Development of a Real-Time PCR Assay to Identify and Distinguish between Cryptococcus neoformans and Cryptococcus gattii Species Complexes. Journal of Fungi, 8(5), 462. https://doi.org/10.3390/jof8050462






