In Vitro Systems for Toxicity Evaluation of Microbial Volatile Organic Compounds on Humans: Current Status and Trends
Abstract
1. Introduction
2. Microbial Volatile Organic Compounds (mVOCs)
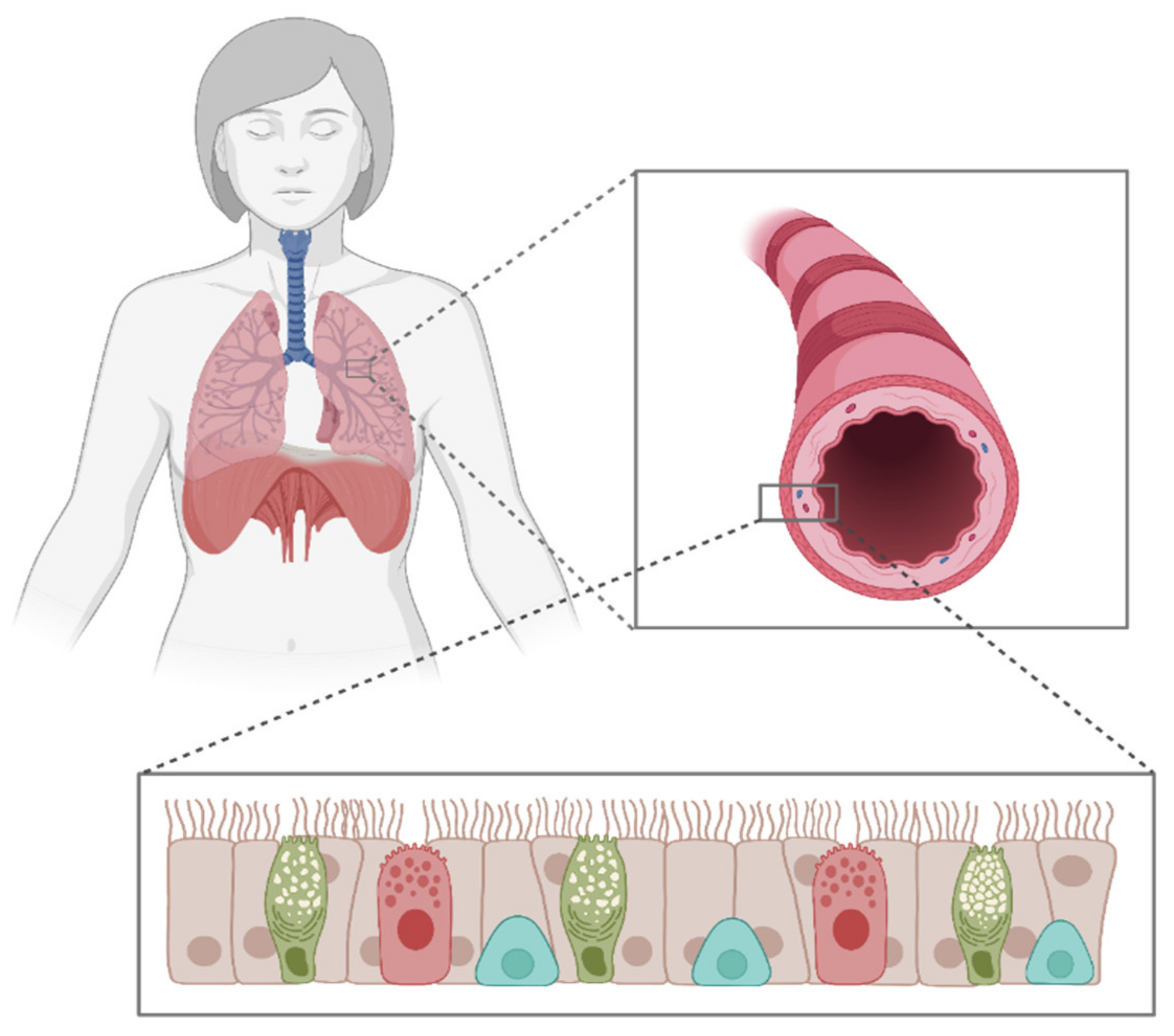
3. MVOC Toxicity: Respiratory Health-Related Symptoms and Association with mVOCS
Influence of mVOCs on Health and Detection of Potential Hazards
4. MVOC Toxicity Evaluation
4.1. Model Organisms
4.2. Cell Culture Experiments
5. Fingerprinting & Profiling
Qualification of mVOCs
6. Toxicity Evaluation of Respiratory Affecting Agents
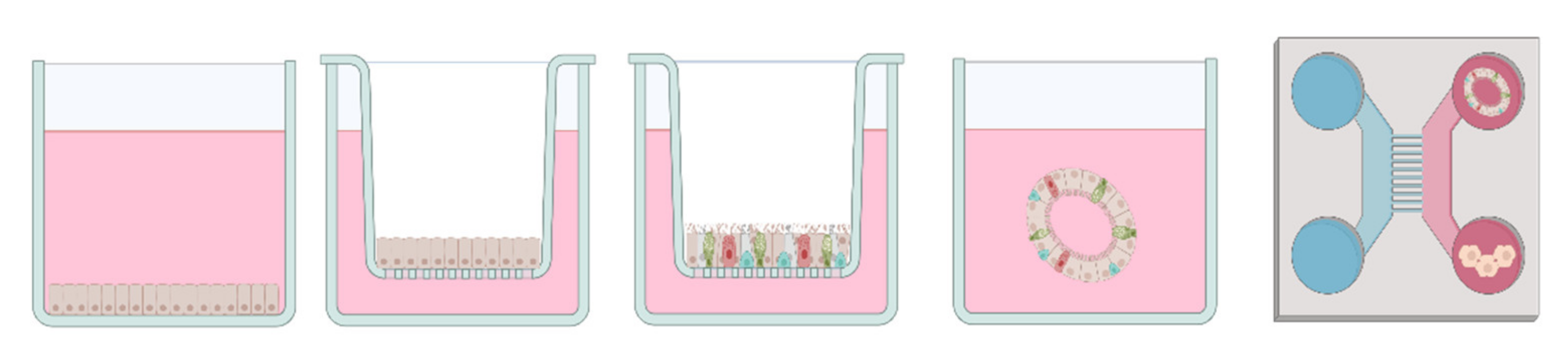
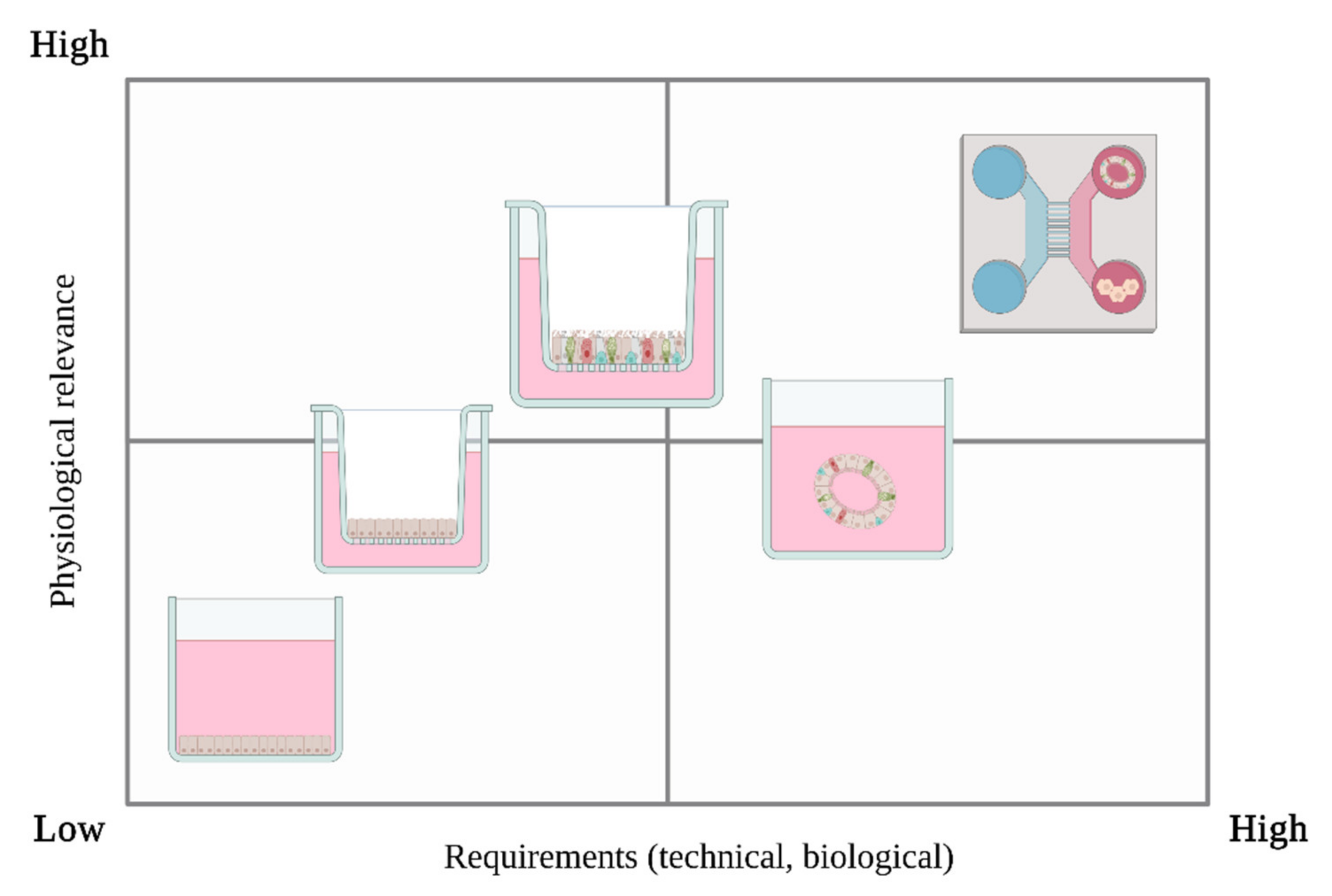
6.1. Submerged Cell Culture
6.2. Air-Liquid Interface (ALI)
6.3. 3D In Vitro Models
6.4. Microfluidics & Multi-Organ Approaches
6.5. Exposure Devices
7. Cell Sources for Modeling
7.1. Cell Lines
7.2. hPSCs, iPSCs and Primary Cells
8. Discussion
8.1. Biological Concerns in Toxicity Measurements of mVOCs
8.2. Technical Concerns in Toxicity Measurements of mVOCs
9. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Korpi, A.; Järnberg, J.; Pasanen, A.-L. Microbial Volatile Organic Compounds. Crit. Rev. Toxicol. 2009, 39, 139–193. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Crump, D. A review of the emission of VOCs from polymeric materials used in buildings. Build. Environ. 1998, 33, 357–374. [Google Scholar] [CrossRef]
- Thevenet, F.; Debono, O.; Rizk, M.; Caron, F.; Verriele, M.; Locoge, N. VOC uptakes on gypsum boards: Sorption performances and impact on indoor air quality. Build. Environ. 2018, 137, 138–146. [Google Scholar] [CrossRef]
- Veselova, M.A.; Plyuta, V.A.; Khmel, I.A. Volatile Compounds of Bacterial Origin: Structure, Biosynthesis, and Biological Activity. Microbiology 2019, 88, 261–274. [Google Scholar] [CrossRef]
- Bakand, S.; Winder, C.; Khalil, C.; Hayes, A. A novel in vitro exposure technique for toxicity testing of selected volatile organic compounds. J. Environ. Monit. 2006, 8, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Parasuraman, S. Toxicological screening. J. Pharmacol. Pharmacother. 2011, 2, 74–79. [Google Scholar] [CrossRef]
- Hayden, P.; Jackson, G.R.; Maione, A.G.; Klausner, M. Comparison of in vitro rat and human airway epithelial models for inhalation toxicity testing. Am. J. Respir. Crit. Care Med. 2018, 197, 1940. [Google Scholar]
- Morath, S.; Hung, R.; Bennett, J.W. Fungal volatile organic compounds: A review with emphasis on their biotechnological potential. Fungal Biol. Rev. 2012, 26, 73–83. [Google Scholar] [CrossRef]
- Moliszewska, E. Mushroom flavour. Folia Biol. Oecologica 2014, 10, 80–88. [Google Scholar] [CrossRef]
- Schulz, S.; Dickschat, J.S. Bacterial volatiles: The smell of small organisms. Nat. Prod. Rep. 2007, 24, 814–842. [Google Scholar] [CrossRef] [PubMed]
- Penuelas, J.; Asensio, D.; Tholl, D.; Wenke, K.; Rosenkranz, M.; Piechulla, B.; Schnitzler, J. Biogenic volatile emissions from the soil. Plant Cell Environ. 2014, 37, 1866–1891. [Google Scholar] [CrossRef]
- Effmert, U.; Kalderás, J.; Warnke, R.; Piechulla, B. Volatile Mediated Interactions between Bacteria and Fungi in the Soil. J. Chem. Ecol. 2012, 38, 665–703. [Google Scholar] [CrossRef]
- Hung, R.; Lee, S.; Bennett, J.W. Arabidopsis thaliana as a model system for testing the effect of Trichoderma volatile organic compounds. Fungal Ecol. 2013, 6, 19–26. [Google Scholar] [CrossRef]
- Kanchiswamy, C.N.; Malnoy, M.; Maffei, M. Chemical diversity of microbial volatiles and their potential for plant growth and productivity. Front. Plant Sci. 2015, 6, 151. [Google Scholar] [CrossRef]
- Hung, R.; Lee, S.; Bennett, J.W. Fungal volatile organic compounds and their role in ecosystems. Appl. Microbiol. Biotechnol. 2015, 99, 3395–3405. [Google Scholar] [CrossRef] [PubMed]
- Piechulla, B.; Degenhardt, J. The emerging importance of microbial volatile organic compounds. Plant Cell Environ. 2014, 37, 811–812. [Google Scholar] [CrossRef] [PubMed]
- Lemfack, M.C.; Gohlke, B.; Toguem, S.M.T.; Preissner, S.; Piechulla, B.; Preissner, R. mVOC 2.0: A database of microbial volatiles. Nucleic Acids Res. 2018, 46, D1261–D1265. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, R.; Cordovez, V.; de Boer, W.; Raaijmakers, J.; Garbeva, P. Volatile affairs in microbial interactions. ISME J. 2015, 9, 2329–2335. [Google Scholar] [CrossRef]
- Bennett, J.W.; Inamdar, A.A. Are Some Fungal Volatile Organic Compounds (VOCs) Mycotoxins? Toxins 2015, 7, 3785–3804. [Google Scholar] [CrossRef]
- Selim, K.A.; El Ghwas, D.E.; Selim, R.M.; Hassan, M.I.A. Microbial Volatile in Defense. In Volatiles and Food Security; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2017; pp. 135–170. [Google Scholar]
- Korn, V.L.; Padhi, S.; Bennett, J.W. An Aroma Odyssey: The Promise of Volatile Fungal Metabolites in Biotechnology. Grand Chall. Mar. Biotechnol. 2020, 11, 349–368. [Google Scholar]
- Medina-Romero, Y.M.; Roque-Flores, G.; Macías-Rubalcava, M.L. Volatile organic compounds from endophytic fungi as innovative postharvest control of Fusarium oxysporum in cherry tomato fruits. Appl. Microbiol. Biotechnol. 2017, 101, 8209–8222. [Google Scholar] [CrossRef] [PubMed]
- Salwan, R.; Rialch, N.; Sharma, V. Bioactive Volatile Metabolites of Trichoderma: An overview. In Secondary Metabolites of Plant Growth Promoting Rhizomicroorganisms; Springer: Singapore, 2019; pp. 87–111. [Google Scholar]
- Jantunen, M.; Jaakola, J.J.K.; Krzyzanowski, M. Assessment of Exposure to Indoor Air Pollutants; WHO: Geneva, Switzerland, 1997. [Google Scholar]
- Mølhave, L. Sick building syndrome. In Encyclopedia of Environmental Health; Elsevier BV: Aarhus, Denmark, 2019. [Google Scholar]
- Zhang, X.; Sahlberg, B.; Wieslander, G.; Janson, C.; Gislason, T.; Norback, D. Dampness and moulds in workplace buildings: Associations with incidence and remission of sick building syndrome (SBS) and biomarkers of inflammation in a 10year follow-up study. Sci. Total. Environ. 2012, 430, 75–81. [Google Scholar] [CrossRef]
- Wålinder, R.; Ernstgård, L.; Norbäck, D.; Wieslander, G.; Johanson, G. Acute effects of 1-octen-3-ol, a microbial volatile organic compound (MVOC)—An experimental study. Toxicol. Lett. 2008, 181, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Araki, A.; Kawai, T.; Eitaki, Y.; Kanazawa, A.; Morimoto, K.; Nakayama, K.; Shibata, E.; Tanaka, M.; Takigawa, T.; Yoshimura, T.; et al. Relationship between selected indoor volatile organic compounds, so-called microbial VOC, and the prevalence of mucous membrane symptoms in single family homes. Sci. Total. Environ. 2010, 408, 2208–2215. [Google Scholar] [CrossRef] [PubMed]
- Saijo, Y.; Kanazawa, A.; Araki, A.; Morimoto, K.; Nakayama, K.; Takigawa, T.; Tanaka, M.; Shibata, E.; Yoshimura, T.; Chikara, H.; et al. Relationships between mite allergen levels, mold concentrations, and sick building syndrome symptoms in newly built dwellings in Japan. Indoor Air 2010, 21, 253–263. [Google Scholar] [CrossRef]
- Araki, A.; Kanazawa, A.; Kawai, T.; Eitaki, Y.; Morimoto, K.; Nakayama, K.; Shibata, E.; Tanaka, M.; Takigawa, T.; Yoshimura, T.; et al. The relationship between exposure to microbial volatile organic compound and allergy prevalence in single-family homes. Sci. Total. Environ. 2012, 423, 18–26. [Google Scholar] [CrossRef][Green Version]
- Sahlberg, B.; Gunnbjörnsdottir, M.; Soon, A.; Jogi, R.; Gislason, T.; Wieslander, G.; Janson, C.; Norbäck, D. Airborne molds and bacteria, microbial volatile organic compounds (MVOC), plasticizers and formaldehyde in dwellings in three North European cities in relation to sick building syndrome (SBS). Sci. Total Environ. 2013, 444, 433–440. [Google Scholar] [CrossRef]
- Choi, H.; Schmidbauer, N.; Bornehag, C.-G. Volatile organic compounds of possible microbial origin and their risks on childhood asthma and allergies within damp homes. Environ. Int. 2017, 98, 143–151. [Google Scholar] [CrossRef]
- WHO (World Health Organization). Guidelines for indoor air quality-dampness and mould. J. Biomed. Semantics 2009, 11, 4521. [Google Scholar]
- Zheng, Y.; Chi, Z.; Lucker, B.; Chen, S. Two-stage heterotrophic and phototrophic culture strategy for algal biomass and lipid production. Bioresour. Technol. 2012, 103, 484–488. [Google Scholar] [CrossRef]
- Claeson, A.-S.; Levin, J.-O.; Blomquist, G.; Sunesson, A.-L. Volatile metabolites from microorganisms grown on humid building materials and synthetic media. J. Environ. Monit. 2002, 4, 667–672. [Google Scholar] [CrossRef] [PubMed]
- Bulian, F.; Fragassa, C.; Franco, B.; Cristiano, F. VOC emissions from wood products and furniture: A survey about legislation, standards and measures referred to different materials. FME Trans. 2016, 44, 358–364. [Google Scholar] [CrossRef]
- Choi, H.; Schmidbauer, N.; Bornehag, C.-G. Non-microbial sources of microbial volatile organic compounds. Environ. Res. 2016, 148, 127–136. [Google Scholar] [CrossRef]
- Bennett, J.W. Silver linings: A personal memoir about Hurricane Katrina and fungal volatiles. Front. Microbiol. 2015, 6, 206. [Google Scholar] [CrossRef] [PubMed]
- Schleibinger, H.; Laussmann, D.; Bornehag, C.-G.; Eis, D.; Rueden, H. Microbial volatile organic compounds in the air of moldy and mold-free indoor environments. Indoor Air 2008, 18, 113–124. [Google Scholar] [CrossRef] [PubMed]
- Schleibinger, H.; Laussmann, D.; Brattig, C.; Mangler, M.; Eis, D.; Ruden, H. Emission patterns and emission rates of MVOC and the possibility for predicting hidden mold damage? Indoor Air 2005, 15, 98–104. [Google Scholar] [CrossRef]
- Betancourt, A.; Krebs, K.; Moore, S.; Martin, S.M. Microbial volatile organic compound emissions from Stachybotrys chartarum growing on gypsum wallboard and ceiling tile. BMC Microbiol. 2013, 13, 283. [Google Scholar] [CrossRef] [PubMed]
- Schuchardt, S.; Strube, A. Microbial volatile organic compounds in moldy interiors: A long-term climate chamber study. J. Basic Microbiol. 2012, 53, 532–538. [Google Scholar] [CrossRef]
- Council Directive 1999/13 of the EU (European Union). Council Directive 1999/13/EC of 11 March 1999 on the limitation of emissions of volatile organic compounds due to the use of organic solvents in certain activities and installations. Off. J. Eur. Commut. 1999, 14, 452. [Google Scholar]
- Council Directive 2001/81 of the EU (European Union). Directive 2001/81/EC of the European Parliament and of the Council of 23 October 2001 on national emission ceilings for certain atmospheric pollutants. Off. J. Eur. Commut. 2001, 24, 412. [Google Scholar]
- Council Directive 2004/42 of the EU (European Union). Council Directive 2004/42/EC of the European Parliament and of the Council of 21 April 2004 on the limitation of emissions of volatile organic compounds due to the use of organic solvents in certain paints and varnishes and vehicle refinishing products an. Off. J. Eur. Commut. 2004, 41, 454. [Google Scholar]
- WHO (World Health Organization). Assessment of exposure to indoor air pollutants. EURO Rep. Stud. 1987, 45, 754. [Google Scholar]
- US-EPA. Technical Overview of Volatile Organic Compounds. 2016. Available online: https//www.epa.gov/indoor-air-quality-iaq/technical-overview-volatile-organic-compounds#definition (accessed on 8 June 2021).
- Inamdar, A.A.; Hossain, M.M.; Bernstein, A.; Miller, G.; Richardson, J.; Bennett, J.W. Fungal-derived semiochemical 1-octen-3-ol disrupts dopamine packaging and causes neurodegeneration. Proc. Natl. Acad. Sci. USA 2013, 110, 19561–19566. [Google Scholar] [CrossRef] [PubMed]
- Inamdar, A.A.; Masurekar, P.; Bennett, J.W. Neurotoxicity of Fungal Volatile Organic Compounds in Drosophila melanogaster. Toxicol. Sci. 2010, 117, 418–426. [Google Scholar] [CrossRef] [PubMed]
- Inamdar, A.A.; Zaman, T.; Morath, S.U.; Pu, D.C.; Bennett, J.W. Drosophila melanogasteras a model to characterize fungal volatile organic compounds. Environ. Toxicol. 2014, 29, 829–836. [Google Scholar] [CrossRef] [PubMed]
- Inamdar, A.A.; Bennett, J.W. A common fungal volatile organic compound induces a nitric oxide mediated inflammatory response in Drosophila melanogaster. Sci. Rep. 2014, 4, 3833. [Google Scholar] [CrossRef]
- Zhao, G.; Yin, G.; Inamdar, A.A.; Luo, J.; Zhang, N.; Yang, I.; Buckley, B.; Bennett, J.W. Volatile organic compounds emitted by filamentous fungi isolated from flooded homes after Hurricane Sandy show toxicity in a Drosophila bioassay. Indoor Air 2016, 27, 518–528. [Google Scholar] [CrossRef]
- Almaliki, H.S.; Angela, A.; Goraya, N.J.; Yin, G.; Bennett, J.W. Volatile Organic Compounds Produced by Human Pathogenic Fungi Are Toxic to Drosophila melanogaster. Front. Fungal Biol. 2021, 1, 629510. [Google Scholar] [CrossRef]
- Chandorkar, P.; Posch, W.; Zaderer, V.; Blatzer, M.; Steger, M.; Ammann, C.G.; Binder, U.; Hermann, M.; Hörtnagl, P.; Lass-Flörl, C.; et al. Fast-track development of an in vitro 3D lung/immune cell model to study Aspergillus infections. Sci. Rep. 2017, 7, 11644. [Google Scholar] [CrossRef]
- Keown, K.; Reid, A.; Moore, J.E.; Taggart, C.C.; Downey, D.G. Coinfection with Pseudomonas aeruginosa and Aspergillus fumigatus in cystic fibrosis. Eur. Respir. Rev. 2020, 29, 200011. [Google Scholar] [CrossRef]
- Kreja, L.; Seidel, H.-J. Evaluation of the genotoxic potential of some microbial volatile organic compounds (MVOC) with the comet assay, the micronucleus assay and the HPRT gene mutation assay. Mutat. Res. Toxicol. Environ. Mutagen. 2002, 513, 143–150. [Google Scholar] [CrossRef]
- Kreja, L.; Seidel, H.-J. On the cytotoxicity of some microbial volatile organic compounds as studied in the human lung cell line. Chemosphere 2002, 49, 105–110. [Google Scholar] [CrossRef]
- InamdarJennifer, A.A.; Moore, J.C.; Cohen, R.I.; Bennett, J.W. A Model to Evaluate the Cytotoxicity of the Fungal Volatile Organic Compound 1-octen-3-ol in Human Embryonic Stem Cells. Myopathic 2011, 173, 13–20. [Google Scholar] [CrossRef]
- Barkal, L.J.; Procknow, C.L.; Álvarez-García, Y.R.; Niu, M.; Jimenez-Torres, J.A.; Brockman-Schneider, R.A.; Gern, J.E.; Denlinger, L.C.; Theberge, A.B.; Keller, N.P.; et al. Microbial volatile communication in human organotypic lung models. Nat. Commun. 2017, 8, 1770. [Google Scholar] [CrossRef]
- Garcia-Alcega, S.; Nasir, Z.A.; Cipullo, S.; Ferguson, R.; Yan, C.; Whitby, C.; Dumbrell, A.J.; Drew, G.; Colbeck, I.; Tyrrel, S.; et al. Fingerprinting ambient air to understand bioaerosol profiles in three different environments in the south east of England. Sci. Total. Environ. 2020, 719, 137542. [Google Scholar] [CrossRef]
- Garcia-Alcega, S.; Nasar, Z.A.; Ferguson, R.; Noël, C.; Cravo-Laureau, C.; Whitby, C.; Dumbrell, A.J.; Colbeck, I.; Tyrrel, S.; Coulon, F. Can chemical and molecular biomarkers help discriminate between industrial, rural and urban environments? Sci. Total. Environ. 2018, 631–632, 1059–1069. [Google Scholar] [CrossRef]
- Garcia-Alcega, S.; Nasir, Z.A.; Ferguson, R.; Whitby, C.; Dumbrell, A.J.; Colbeck, I.; Gomes, D.; Tyrrel, S.; Coulon, F. Fingerprinting outdoor air environment using microbial volatile organic compounds (MVOCs)—A review. TrAC Trends Anal. Chem. 2017, 86, 75–83. [Google Scholar] [CrossRef]
- Fiedler, K.; Schütz, E.; Geh, S. Detection of microbial volatile organic compounds (MVOCs) produced by moulds on various materials. Int. J. Hyg. Environ. Health 2001, 204, 111–121. [Google Scholar] [CrossRef]
- Fu, X. Indoor Microbial Volatile Organic Compound (MVOC) Levels and Associations with Respiratory Health, Sick Building Syndrome (SBS), and Allergy. In Environmental Mycology in Public Health; Elsevier BV: Amsterdam, The Netherlands, 2016; pp. 387–395. [Google Scholar]
- Wilkins, K.; Larsen, K. Variation of volatile organic compound patterns of mold species from damp buildings. Chemosphere 1995, 31, 3225–3236. [Google Scholar] [CrossRef]
- Matysik, S.; Herbarth, O.; Mueller, A. Determination of volatile metabolites originating from mould growth on wall paper and synthetic media. J. Microbiol. Methods 2008, 75, 182–187. [Google Scholar] [CrossRef]
- Polizzi, V.; Adams, A.; Malysheva, S.V.; De Saeger, S.; Van Peteghem, C.; Moretti, A.; Picco, A.M.; De Kimpe, N. Identification of volatile markers for indoor fungal growth and chemotaxonomic classification of Aspergillus species. Fungal Biol. 2012, 116, 941–953. [Google Scholar] [CrossRef]
- Schuchardt, S.; Kruse, H. Quantitative volatile metabolite profiling of common indoor fungi: Relevancy for indoor air analysis. J. Basic Microbiol. 2009, 49, 350–362. [Google Scholar] [CrossRef]
- Sawoszczuk, T.; Syguła-Cholewińska, J.; del Hoyo-Meléndez, J.M. Application of solid-phase microextraction with gas chromatography and mass spectrometry for the early detection of active moulds on historical woollen objects. J. Sep. Sci. 2017, 40, 858–868. [Google Scholar] [CrossRef]
- Sawoszczuk, T.; Syguła-Cholewińska, J.; del Hoyo-Meléndez, J.M. Application of HS-SPME-GC-MS method for the detection of active moulds on historical parchment. Anal. Bioanal. Chem. 2017, 409, 2297–2307. [Google Scholar] [CrossRef]
- Sawoszczuk, T.; Syguła-Cholewińska, J.; del Hoyo-Meléndez, J.M. Optimization of headspace solid phase microextraction for the analysis of microbial volatile organic compounds emitted by fungi: Application to historical objects. J. Chromatogr. A 2015, 1409, 30–45. [Google Scholar] [CrossRef] [PubMed]
- Bingley, G.D.; Verran, J.; Munro, L.J.; Banks, C.E. Identification of microbial volatile organic compounds (MVOCs) emitted from fungal isolates found on cinematographic film. Anal. Methods 2012, 4, 1265–1271. [Google Scholar] [CrossRef]
- Norbäck, D.; Wieslander, G.; Ström, G.; Edling, C. Exposure to Volatile Organic Compounds of Microbial Origin (MVOC) during Indoor Application of Water-based Paints. Indoor Air 1995, 5, 166–170. [Google Scholar] [CrossRef]
- Lehtinen, J.; Tolvanen, O.; Nivukoski, U.; Veijanen, A.; Hänninen, K. Occupational hygiene in terms of volatile organic compounds (VOCs) and bioaerosols at two solid waste management plants in Finland. Waste Manag. 2013, 33, 964–973. [Google Scholar] [CrossRef]
- Malta-Vacas, J.; Viegas, S.; Sabino, R.; Viegas, C. Fungal and Microbial Volatile Organic Compounds Exposure Assessment in a Waste Sorting Plant. J. Toxicol. Environ. Health Part A 2012, 75, 1410–1417. [Google Scholar] [CrossRef] [PubMed]
- Korpi, A.; Pasanen, A.-L.; Pasanen, P. Volatile Compounds Originating from Mixed Microbial Cultures on Building Materials under Various Humidity Conditions. Appl. Environ. Microbiol. 1998, 64, 2914–2919. [Google Scholar] [CrossRef] [PubMed]
- Gao, P.; Korley, F.; Martin, J.; Chen, B.T. Determination of Unique Microbial Volatile Organic Compounds Produced by FiveAspergillusSpecies Commonly Found in Problem Buildings. AIHA J. 2002, 63, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Gao, P.; Martin, J. Volatile Metabolites Produced by Three Strains of Stachybotrys chartarum Cultivated on Rice and Gypsum Board. Appl. Occup. Environ. Hyg. 2002, 17, 430–436. [Google Scholar] [CrossRef]
- Ouyang, G.; Pawliszyn, J. SPME in environmental analysis. Anal. Bioanal. Chem. 2006, 386, 1059–1073. [Google Scholar] [CrossRef] [PubMed]
- Silva, E.A.S.; Risticevic, S.; Pawliszyn, J. Recent trends in SPME concerning sorbent materials, configurations and in vivo applications. TrAC Trends Anal. Chem. 2013, 43, 24–36. [Google Scholar] [CrossRef]
- Pasanen, A.-L.; Lappalainen, S.; Pasanen, P. Volatile organic metabolites associated with some toxic fungi and their mycotoxins. Analyst 1996, 121, 1949–1953. [Google Scholar] [CrossRef]
- Farag, M.A.; Ryu, C.-M.; Sumner, L.W.; Paré, P.W. GC–MS SPME profiling of rhizobacterial volatiles reveals prospective inducers of growth promotion and induced systemic resistance in plants. Phytochemistry 2006, 67, 2262–2268. [Google Scholar] [CrossRef] [PubMed]
- Merkle, S.; Kleeberg, K.K.; Fritsche, J. Recent Developments and Applications of Solid Phase Microextraction (SPME) in Food and Environmental Analysis—A Review. Chromatogram 2015, 2, 293–381. [Google Scholar] [CrossRef]
- Wady, L.; Larsson, L. Determination of microbial volatile organic compounds adsorbed on house dust particles and gypsum board using SPME/GC-MS. Indoor Air 2005, 15, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Siddiquee, S.; Cheong, B.E.; Taslima, K.; Kausar, H.; Hasan, M. Separation and Identification of Volatile Compounds from Liquid Cultures of Trichoderma harzianum by GC-MS using Three Different Capillary Columns. J. Chromatogr. Sci. 2012, 50, 358–367. [Google Scholar] [CrossRef]
- Stoppacher, N.; Kluger, B.; Zeilinger, S.; Krska, R.; Schuhmacher, R. Identification and profiling of volatile metabolites of the biocontrol fungus Trichoderma atroviride by HS-SPME-GC-MS. J. Microbiol. Methods 2010, 81, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Guo, C.; Chen, Q.; Ouyang, Q.; Shi, J.; El-Seedi, H.R.; Zou, X. Classification for Penicillium expansum Spoilage and Defect in Apples by Electronic Nose Combined with Chemometrics. Sensors 2020, 20, 2130. [Google Scholar] [CrossRef]
- Gobbi, E.; Falasconi, M.; Torelli, E.; Sberveglieri, G. Electronic nose predicts high and low fumonisin contamination in maize cultures. Food Res. Int. 2011, 44, 992–999. [Google Scholar] [CrossRef]
- Lippolis, V.; Pascale, M.; Cervellieri, S.; Damascelli, A.; Visconti, A. Screening of deoxynivalenol contamination in durum wheat by MOS-based electronic nose and identification of the relevant pattern of volatile compounds. Food Control 2014, 37, 263–271. [Google Scholar] [CrossRef]
- Mota, I.; Teixeira-Santos, R.; Rufo, J.C. Detection and identification of fungal species by electronic nose technology: A systematic review. Fungal Biol. Rev. 2021, 37, 59–70. [Google Scholar] [CrossRef]
- Van Meer, P.J.; Kooijman, M.; Wied, C.C.G.-D.; Moors, E.H.; Schellekens, H. The ability of animal studies to detect serious post marketing adverse events is limited. Regul. Toxicol. Pharmacol. 2012, 64, 345–349. [Google Scholar] [CrossRef] [PubMed]
- Van Norman, G.A. Limitations of Animal Studies for Predicting Toxicity in Clinical Trials. JACC: Basic Transl. Sci. 2019, 4, 845–854. [Google Scholar] [CrossRef] [PubMed]
- Bailey, J. Developmental Toxicity Testing: Protecting Future Generations? Altern. Lab. Anim. 2008, 36, 718–721. [Google Scholar] [CrossRef]
- European Center for the Validation of Alternative Methods (ECVAM). Validated test methods. Eur. Comm. Sci. Knowl. Serv. 2021, 14, 756. [Google Scholar]
- Jarlath, H. Advanced Non-animal Models in Biomedical Research. Eur. Comm. Sci. Knowl. Serv. 2020, 14, 657. [Google Scholar]
- Romo, M.R.; Pérez-Martínez, D.; Ferrer, C.C. Innate immunity in vertebrates: An overview. Immunology 2016, 148, 125–139. [Google Scholar] [CrossRef] [PubMed]
- Atamas, S.P.; Chapoval, S.; Keegan, A.D. Cytokines in chronic respiratory diseases. Biol. Rep. 2013, 5, 3. [Google Scholar] [CrossRef] [PubMed]
- Colebatch, H.; Ng, C. Estimating alveolar surface area during life. Respir. Physiol. 1992, 88, 163–170. [Google Scholar] [CrossRef]
- Basil, M.C.; Katzen, J.; Engler, A.E.; Guo, M.; Herriges, M.J.; Kathiriya, J.J.; Windmueller, R.; Ysasi, A.B.; Zacharias, W.J.; Chapman, H.A.; et al. The Cellular and Physiological Basis for Lung Repair and Regeneration: Past, Present, and Future. Cell Stem Cell 2020, 26, 482–502. [Google Scholar] [CrossRef] [PubMed]
- Zepp, J.A.; Morrisey, E.E. Cellular crosstalk in the development and regeneration of the respiratory system. Nat. Rev. Mol. Cell Biol. 2019, 20, 551–566. [Google Scholar] [CrossRef] [PubMed]
- Houtmeyers, E.; Gosselink, R.; Gayan-Ramirez, G.; Decramer, M. Regulation of mucociliary clearance in health and disease. Eur. Respir. J. 1999, 13, 1177–1188. [Google Scholar] [CrossRef]
- Smith, D.J.; Gaffney, E.A.; Blake, J.R. Modelling mucociliary clearance. Respir. Physiol. Neurobiol. 2008, 163, 178–188. [Google Scholar] [CrossRef] [PubMed]
- Rokicki, W.; Rokicki, M.; Wojtacha, J.; Dżeljijli, A. The role and importance of club cells (Clara cells) in the pathogenesis of some respiratory diseases. Pol. J. Cardio-Thoracic Surg. 2016, 1, 26–30. [Google Scholar] [CrossRef]
- Macklem, P.T. The Physiology of Small Airways. Am. J. Respir. Crit. Care Med. 1998, 157, S181–S183. [Google Scholar] [CrossRef] [PubMed]
- Guillot, L.; Nathan, N.; Tabary, O.; Thouvenin, G.; Le Rouzic, P.; Corvol, H.; Amselem, S.; Clement, A. Alveolar epithelial cells: Master regulators of lung homeostasis. Int. J. Biochem. Cell Biol. 2013, 45, 2568–2573. [Google Scholar] [CrossRef]
- Bakand, S.; Hayes, A. Troubleshooting methods for toxicity testing of airborne chemicals in vitro. J. Pharmacol. Toxicol. Methods 2010, 61, 76–85. [Google Scholar] [CrossRef]
- Lenz, A.-G.; Karg, E.; Brendel, E.; Hinze-Heyn, H.; Maier, K.L.; Eickelberg, O.; Stoeger, T.; Schmid, O. Inflammatory and Oxidative Stress Responses of an Alveolar Epithelial Cell Line to Airborne Zinc Oxide Nanoparticles at the Air-Liquid Interface: A Comparison with Conventional, Submerged Cell-Culture Conditions. BioMed Res. Int. 2013, 2013, 652632. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, S.; Palmberg, L. Air-Liquid Interface: Relevant In Vitro Models for Investigating Air Pollutant-Induced Pulmonary Toxicity. Toxicol. Sci. 2018, 164, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Gohlsch, K.; Mückter, H.; Steinritz, D.; Aufderheide, M.; Hoffmann, S.; Gudermann, T.; Breit, A. Exposure of 19 substances to lung A549 cells at the air liquid interface or under submerged conditions reveals high correlation between cytotoxicity in vitro and CLP classifications for acute lung toxicity. Toxicol. Lett. 2019, 316, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Loret, T.; Peyret, E.; Dubreuil, M.; Aguerre-Chariol, O.; Bressot, C.; Le Bihan, O.; Amodeo, T.; Trouiller, B.; Braun, A.; Egles, C.; et al. Air-liquid interface exposure to aerosols of poorly soluble nanomaterials induces different biological activation levels compared to exposure to suspensions. Part. Fibre Toxicol. 2016, 13, 58. [Google Scholar] [CrossRef] [PubMed]
- Lacroix, G.; Koch, W.; Ritter, D.; Gutleb, A.; Larsen, S.T.; Loret, T.; Zanetti, F.; Constant, S.; Chortarea, S.; Rothen-Rutishauser, B.; et al. Air–Liquid InterfaceIn VitroModels for Respiratory Toxicology Research: Consensus Workshop and Recommendations. Appl. Vitr. Toxicol. 2018, 4, 91–106. [Google Scholar] [CrossRef] [PubMed]
- Paur, H.-R.; Cassee, F.R.; Teeguarden, J.; Fissan, H.; Diabate, S.; Aufderheide, M.; Kreyling, W.G.; Hänninen, O.; Kasper, G.; Riediker, M.; et al. In-vitro cell exposure studies for the assessment of nanoparticle toxicity in the lung—A dialog between aerosol science and biology. J. Aerosol Sci. 2011, 42, 668–692. [Google Scholar] [CrossRef]
- Méausoone, C.; El Khawaja, R.; Tremolet, G.; Siffert, S.; Cousin, R.; Cazier, F.; Billet, S.; Courcot, D.; Landkocz, Y. In vitro toxicological evaluation of emissions from catalytic oxidation removal of industrial VOCs by air/liquid interface (ALI) exposure system in repeated mode. Toxicol. Vitr. 2019, 58, 110–117. [Google Scholar] [CrossRef]
- Mülhopt, S.; Dilger, M.; Diabaté, S.; Schlager, C.; Krebs, T.; Zimmermann, R.; Buters, J.; Oeder, S.; Wäscher, T.; Weiss, C.; et al. Toxicity testing of combustion aerosols at the air–liquid interface with a self-contained and easy-to-use exposure system. J. Aerosol Sci. 2016, 96, 38–55. [Google Scholar] [CrossRef]
- Dwivedi, A.M.; Upadhyay, S.; Johanson, G.; Ernstgård, L.; Palmberg, L. Inflammatory effects of acrolein, crotonaldehyde and hexanal vapors on human primary bronchial epithelial cells cultured at air-liquid interface. Toxicol. Vitr. 2018, 46, 219–228. [Google Scholar] [CrossRef] [PubMed]
- Pezzulo, A.A.; Starner, T.D.; Scheetz, T.E.; Traver, G.L.; Tilley, A.E.; Harvey, B.-G.; Crystal, R.G.; McCray, P.B., Jr.; Zabner, J. The air-liquid interface and use of primary cell cultures are important to recapitulate the transcriptional profile of in vivo airway epithelia. Am. J. Physiol. Lung Cell. Mol. Physiol. 2011, 300, L25–L31. [Google Scholar] [CrossRef]
- Dvorak, A.; Tilley, A.E.; Shaykhiev, R.; Wang, R.; Crystal, R.G. Do Airway Epithelium Air–Liquid Cultures Represent the In Vivo Airway Epithelium Transcriptome? Am. J. Respir. Cell Mol. Biol. 2011, 44, 465–473. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.J.; Dailey, L.A.; Brighton, L.E.; Devlin, R.B. Transcriptional Profiling of Mucociliary Differentiation in Human Airway Epithelial Cells. Am. J. Respir. Cell Mol. Biol. 2007, 37, 169–185. [Google Scholar] [CrossRef] [PubMed]
- Anderson, S.E.; Khurshid, S.S.; Meade, B.J.; Lukomska, E.; Wells, J. Toxicological analysis of limonene reaction products using an in vitro exposure system. Toxicol. Vitr. 2013, 27, 721–730. [Google Scholar] [CrossRef] [PubMed]
- Cervena, T.; Vrbova, K.; Rossnerova, A.; Topinka, J.; Rossner, P. Short-term and Long-term Exposure of the MucilAir™ Model to Polycyclic Aromatic Hydrocarbons. Altern. Lab. Anim. 2019, 47, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Ferro, M.P.; Leclerc, L.; Sleiman, M.; Marchiori, B.; Pourchez, J.; Owens, R.M.; Ramuz, M. Effect of E Cigarette Emissions on Tracheal Cells Monitored at the Air–Liquid Interface Using an Organic Electrochemical Transistor. Adv. Biosyst. 2019, 3, e1800249. [Google Scholar] [CrossRef]
- Talikka, M.; Kostadinova, R.; Xiang, Y.; Mathis, C.; Sewer, A.; Majeed, S.; Kuehn, D.; Frentzel, S.; Merg, C.; Geertz, M.; et al. The Response of Human Nasal and Bronchial Organotypic Tissue Cultures to Repeated Whole Cigarette Smoke Exposure. Int. J. Toxicol. 2014, 33, 506–517. [Google Scholar] [CrossRef]
- Wang, Q.; Khan, N.A.; Muthumalage, T.; Lawyer, G.R.; McDonough, S.R.; Chuang, T.; Gong, M.; Sundar, I.K.; Rehan, V.K.; Rahman, I. Dysregulated repair and inflammatory responses by e-cigarette-derived inhaled nicotine and humectant propylene glycol in a sex-dependent manner in mouse lung. FASEB BioAdvances 2019, 1, 609–623. [Google Scholar] [CrossRef]
- Zavala, J.; O’Brien, B.; Lichtveld, K.; Sexton, K.G.; Rusyn, I.; Jaspers, I.; Vizuete, W. Assessment of biological responses of EpiAirway 3-D cell constructs versus A549 cells for determining toxicity of ambient air pollution. Inhal. Toxicol. 2016, 28, 251–259. [Google Scholar] [CrossRef]
- Niemeyer, B.F.; Zhao, P.; Tuder, R.M.; Benam, K.H. Advanced Microengineered Lung Models for Translational Drug Discovery. SLAS Discov. Adv. Sci. Drug Discov. 2018, 23, 777–789. [Google Scholar] [CrossRef]
- Benam, K.H.; Gilchrist, S.; Kleensang, A.; Satz, A.B.; Willett, C.; Zhang, Q. Exploring new technologies in biomedical research. Drug Discov. Today 2019, 24, 1242–1247. [Google Scholar] [CrossRef]
- Sachs, N.; Papaspyropoulos, A.; Zomer-van Ommen, D.D.; Heo, I.; Böttinger, L.; Klay, D.; Weeber, F.; Huelsz-Prince, G.; Iakobachvili, N.; Amatngalim, G.D.; et al. Long-term expanding human airway organoids for disease modeling. EMBO J. 2019, 38, e100300. [Google Scholar] [CrossRef] [PubMed]
- Pomerenke, A. Organotypic Models of Lung Cancer. Curr. Top. Microbiol. Immunol. 2017, 15, 161–181. [Google Scholar]
- Van der Vaart, J.; Clevers, H. Airway organoids as models of human disease. J. Intern. Med. 2021, 289, 604–613. [Google Scholar] [CrossRef]
- Kuehn, D.; Majeed, S.; Guedj, E.; Dulize, R.; Baumer, K.; Iskandar, A.; Boue, S.; Martin, F.; Kostadinova, R.; Mathis, C.; et al. Impact Assessment of Repeated Exposure of Organotypic 3D Bronchial and Nasal Tissue Culture Models to Whole Cigarette Smoke. J. Vis. Exp. 2015, 14, e52325. [Google Scholar] [CrossRef] [PubMed]
- Kolahchi, A.R.; Mohtaram, N.K.; Modarres, H.P.; Mohammadi, M.H.; Geraili, A.; Jafari, P.; Akbari, M.; Sanati-Nezhad, A. Microfluidic-Based Multi-Organ Platforms for Drug Discovery. Micromachines 2016, 7, 162. [Google Scholar] [CrossRef]
- Gkatzis, K.; Taghizadeh, S.; Huh, D.; Stainier, D.; Bellusci, S. Use of three-dimensional organoids and lung-on-a-chip methods to study lung development, regeneration and disease. Eur. Respir. J. 2018, 52, 1800876. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Xu, C.; Jiang, L.; Qin, J. A 3D human lung-on-a-chip model for nanotoxicity testing. Toxicol. Res. 2018, 7, 1048–1060. [Google Scholar] [CrossRef]
- Doryab, A.; Amoabediny, G.; Salehi-Najafabadi, A. Advances in pulmonary therapy and drug development: Lung tissue engineering to lung-on-a-chip. Biotechnol. Adv. 2016, 34, 588–596. [Google Scholar] [CrossRef]
- Sznitman, J.; Heimsch, T.; Wildhaber, J.H.; Tsuda, A.; Rösgen, T. Respiratory Flow Phenomena and Gravitational Deposition in a Three-Dimensional Space-Filling Model of the Pulmonary Acinar Tree. J. Biomech. Eng. 2009, 131, 031010. [Google Scholar] [CrossRef]
- Stucki, A.O.; Stucki, J.D.; Hall, S.R.R.; Felder, M.; Mermoud, Y.; Schmid, R.A.; Geiser, T.; Guenat, O.T. A lung-on-a-chip array with an integrated bio-inspired respiration mechanism. Lab Chip 2015, 15, 1302–1310. [Google Scholar] [CrossRef]
- Huh, D.; Leslie, D.C.; Matthews, B.D.; Fraser, J.P.; Jurek, S.; Hamilton, G.A.; Thorneloe, K.S.; McAlexander, M.A.; Ingber, D.E. A Human Disease Model of Drug Toxicity–Induced Pulmonary Edema in a Lung-on-a-Chip Microdevice. Sci. Transl. Med. 2012, 4, 159ra147. [Google Scholar] [CrossRef]
- Baker, M. A living system on a chip. Nat. Cell Biol. 2011, 471, 661–665. [Google Scholar] [CrossRef]
- Faber, S.C.; McCullough, S.D. Through the Looking Glass: In Vitro Models for Inhalation Toxicology and Interindividual Variability in the Airway. Appl. Vitr. Toxicol. 2018, 4, 115–128. [Google Scholar] [CrossRef]
- Bovard, D.; Sandoz, A.; Luettich, K.; Frentzel, S.; Iskandar, A.; Marescotti, D.; Trivedi, K.; Guedj, E.; Dutertre, Q.; Peitsch, M.C.; et al. A lung/liver-on-a-chip platform for acute and chronic toxicity studies. Lab. Chip. 2018, 18, 3814–3829. [Google Scholar] [CrossRef]
- Schimek, K.; Frentzel, S.; Luettich, K.; Bovard, D.; Rütschle, I.; Boden, L.; Rambo, F.; Erfurth, H.; Dehne, E.-M.; Winter, A.; et al. Human multi-organ chip co-culture of bronchial lung culture and liver spheroids for substance exposure studies. Sci. Rep. 2020, 10, 642196. [Google Scholar] [CrossRef] [PubMed]
- Inamdar, A.A.; Bennett, J.W. Volatile organic compounds from fungi isolated after hurricane katrina induce developmental defects and apoptosis in aDrosophila melanogastermodel. Environ. Toxicol. 2015, 30, 614–620. [Google Scholar] [CrossRef]
- Morath, S.U.; Boland, C.E.; Bennett, J.W. Saccharomyces cerevisiae as a Model for Screening the Effects of Volatile Organic Compounds. Curr. Biotechnol. 2017, 6, 4622. [Google Scholar] [CrossRef]
- Thorne, D.; Adamson, J. A review of in vitro cigarette smoke exposure systems. Exp. Toxicol. Pathol. 2013, 65, 1183–1193. [Google Scholar] [CrossRef]
- Lucci, F.; Castro, N.D.; Rostami, A.A.; Oldham, M.J.; Hoeng, J.; Pithawalla, Y.B.; Kuczaj, A.K. Characterization and modeling of aerosol deposition in Vitrocell® exposure systems-exposure well chamber deposition efficiency. J. Aerosol Sci. 2018, 123, 141–160. [Google Scholar] [CrossRef]
- Majeed, S.; Frentzel, S.; Wagner, S.; Kuehn, D.; Leroy, P.; Guy, P.; Knorr, A.; Hoeng, J.; Peitsch, M.C. Characterization of the Vitrocell® 24/48 in vitro aerosol exposure system using mainstream cigarette smoke. Chem. Central J. 2014, 8, 62. [Google Scholar] [CrossRef][Green Version]
- Aufderheide, M.; Halter, B.; Möhle, N.; Hochrainer, D. The CULTEX RFS: A Comprehensive Technical Approach for theIn VitroExposure of Airway Epithelial Cells to the Particulate Matter at the Air-Liquid Interface. Bio. Med. Res. Int. 2013, 2013, 734137. [Google Scholar] [CrossRef] [PubMed]
- Aufderheide, M.; Mohr, U. CULTEX—An alternative technique for cultivation and exposure of cells of the respiratory tract to airborne pollutants at the air/liquid interface. Exp. Toxicol. Pathol. 2000, 52, 265–270. [Google Scholar] [CrossRef]
- Steinritz, D.; Möhle, N.; Pohl, C.; Papritz, M.; Stenger, B.; Schmidt, A.; Kirkpatrick, C.J.; Thiermann, H.; Vogel, R.; Hoffmann, S.; et al. Use of the Cultex® Radial Flow System as an in vitro exposure method to assess acute pulmonary toxicity of fine dusts and nanoparticles with special focus on the intra- and inter-laboratory reproducibility. Chem. Interact. 2013, 206, 479–490. [Google Scholar] [CrossRef]
- Vaughan, A.; Stevanovic, S.; Banks, A.; Zare, A.; Rahman, M.; Bowman, R.V.; Fong, K.; Ristovski, Z.; Yang, I. The cytotoxic, inflammatory and oxidative potential of coconut oil-substituted diesel emissions on bronchial epithelial cells at an air-liquid interface. Environ. Sci. Pollut. Res. 2019, 26, 27783–27791. [Google Scholar] [CrossRef]
- Seagrave, J.; Dunaway, S.; McDonald, J.D.; Mauderly, J.L.; Hayden, P.; Stidley, C. Responses of differentiated primary human lung epithelial cells to exposure to diesel exhaust at an air-liquid interface. Exp. Lung Res. 2007, 33, 27–51. [Google Scholar] [CrossRef] [PubMed]
- Knebel, J.; Ritter, D.; Aufderheide, M. Exposure of human lung cells to native diesel motor exhaust—Development of an optimized in vitro test strategy. Toxicol. Vitr. 2002, 16, 185–192. [Google Scholar] [CrossRef]
- Abe, S.; Takizawa, H.; Sugawara, I.; Kudoh, S. Diesel Exhaust (DE)–Induced Cytokine Expression in Human Bronchial Epithelial Cells. Am. J. Respir. Cell Mol. Biol. 2000, 22, 296–303. [Google Scholar] [CrossRef]
- PromoCell Blog. Human Primary Cells and Immortal Cell Lines: Differences and Advantages; Promocell GmbH: Heidelberg, Germany, 2019. [Google Scholar]
- Kaur, G.; Dufour, J.M. Cell lines. Spermatogenesis 2012, 2, 19885. [Google Scholar] [CrossRef]
- Hahn, W.C. Immortalization and transformation of human cells. Mol. Cells 2002, 13, 225. [Google Scholar]
- Giard, D.J.; Aaronson, S.A.; Todaro, G.J.; Arnstein, P.; Kersey, J.H.; Dosik, H.; Parks, W.P. In Vitro Cultivation of Human Tumors: Establishment of Cell Lines Derived from a Series of Solid Tumors. J. Natl. Cancer Inst. 1973, 51, 1417–1423. [Google Scholar] [CrossRef]
- Gminski, R.; Tang, T.; Mersch-Sundermann, V. Cytotoxicity and genotoxicity in human lung epithelial A549 cells caused by airborne volatile organic compounds emitted from pine wood and oriented strand boards. Toxicol. Lett. 2010, 196, 33–41. [Google Scholar] [CrossRef]
- Pariselli, F.; Sacco, M.; Ponti, J.; Rembges, D. Effects of toluene and benzene air mixtures on human lung cells (A549). Exp. Toxicol. Pathol. 2009, 61, 381–386. [Google Scholar] [CrossRef]
- Mascelloni, M.; Delgado-Saborit, J.M.; Hodges, N.J.; Harrison, R.M. Study of gaseous benzene effects upon A549 lung epithelial cells using a novel exposure system. Toxicol. Lett. 2015, 237, 38–45. [Google Scholar] [CrossRef]
- Persoz, C.; Achard, S.; Leleu, C.; Momas, I.; Seta, N. An in vitro model to evaluate the inflammatory response after gaseous formaldehyde exposure of lung epithelial cells. Toxicol. Lett. 2010, 195, 99–105. [Google Scholar] [CrossRef]
- Pariselli, F.; Sacco, M.; Rembges, D. An optimized method for in vitro exposure of human derived lung cells to volatile chemicals. Exp. Toxicol. Pathol. 2009, 61, 33–39. [Google Scholar] [CrossRef]
- Tang, T.; Gminski, R.; Könczöl, M.; Modest, C.; Armbruster, B.; Mersch-Sundermann, V. Investigations on cytotoxic and genotoxic effects of laser printer emissions in human epithelial A549 lung cells using an air/liquid exposure system. Environ. Mol. Mutagen. 2012, 53, 125–135. [Google Scholar] [CrossRef]
- Kooter, I.M.; Alblas, M.J.; Jedynska, A.D.; Steenhof, M.; Houtzager, M.M.; van Ras, M. Alveolar epithelial cells (A549) exposed at the air–liquid interface to diesel exhaust: First study in TNO’s powertrain test center. Toxicol. Vitr. 2013, 27, 2342–2349. [Google Scholar] [CrossRef] [PubMed]
- Cooney, D.J.; Hickey, A.J. Cellular response to the deposition of diesel exhaust particle aerosols onto human lung cells grown at the air–liquid interface by inertial impaction. Toxicol. Vitr. 2011, 25, 1953–1965. [Google Scholar] [CrossRef]
- Fogh, J.; Trempe, G. New human tumor cell lines. In Human Tumor Cell In Vitro; Fogh, J., Ed.; Springer: New York, NY, USA, 1975; pp. 115–159. [Google Scholar]
- Cooper, J.M. Cellular Response to Diesel Engine Exhaust Particulate Exposures: Impact of Extraction Method and Reactive Oxygen Species as a Biomarker. Ph.D. Dissertation, University of Toronto, Toronto, ON, Canada, 2016. [Google Scholar]
- Kastner, P.E.; Casset, A.; Pons, F. Formaldehyde interferes with airway epithelium integrity and functions in a dose- and time-dependent manner. Toxicol. Lett. 2011, 200, 109–116. [Google Scholar] [CrossRef]
- Delaval, M. Evaluating adverse effects of aerosols from different electronic cigarettes to airway epithelia by realistic in vitro technologies. Am. J. Respir. Crit. Care Med. 2018, 25, 424. [Google Scholar]
- Reddel, R.R. Immortalized human bronchial epitherial mesothelial cell lines. U.S. Patent 4,885,238, 5 December 1989. [Google Scholar]
- Méausoone, C.; Landkocz, Y.; Cazier, F.; Seigneur, M.; Courcot, D.; Billet, S. Toxicological responses of BEAS-2B cells to repeated exposures to benzene, toluene, m -xylene, and mesitylene using air–liquid interface method. J. Appl. Toxicol. 2020, 12, 4113. [Google Scholar] [CrossRef]
- Ito, T.; Bekki, K.; Fujitani, Y.; Hirano, S. The toxicological analysis of secondary organic aerosol in human lung epithelial cells and macrophages. Environ. Sci. Pollut. Res. 2019, 26, 22747–22755. [Google Scholar] [CrossRef]
- Holder, A.L.; Lucas, D.; Goth-Goldstein, R.; Koshland, C.P. Inflammatory response of lung cells exposed to whole, filtered, and hydrocarbon denuded diesel exhaust. Chemosphere 2007, 70, 13–19. [Google Scholar] [CrossRef]
- Holder, A.L.; Lucas, D.; Goth-Goldstein, R.; Koshland, C.P. Cellular Response to Diesel Exhaust Particles Strongly Depends on the Exposure Method. Toxicol. Sci. 2008, 103, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Lipsa, D.; Barrero-Moreno, J.; Coelhan, M. Exposure to selected limonene oxidation products: 4-OPA, IPOH, 4-AMCH induces oxidative stress and inflammation in human lung epithelial cell lines. Chemosphere 2018, 191, 937–945. [Google Scholar] [CrossRef] [PubMed]
- Lipsa, D.; Leva, P.; Barrero-Moreno, J.; Coelhan, M. Inflammatory effects induced by selected limonene oxidation products: 4-OPA, IPOH, 4-AMCH in human bronchial (16HBE14o-) and alveolar (A549) epithelial cell lines. Toxicol. Lett. 2016, 262, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Hubbard, K.; Ozer, H.L. Mechanism of immortalization. AGE 1999, 22, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Bryan, T.; Redder, R.R. SV40-lnduced Immortalization of Human Cells. Crit. Rev. Oncog. 1994, 5, 331–357. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Farris, J.; Christman, S.A.; Kong, B.-W.; Foster, L.K.; O’Grady, S.M.; Foster, D.N. Events in the immortalizing process of primary human mammary epithelial cells by the catalytic subunit of human telomerase. Biochem. J. 2002, 365, 765–772. [Google Scholar] [CrossRef][Green Version]
- Toouli, C.D.; Huschtscha, L.; Neumann, A.; Noble, J.R.; Colgin, L.M.; Hukku, B.; Reddel, R.R. Comparison of human mammary epithelial cells immortalized by simian virus 40 T-Antigen or by the telomerase catalytic subunit. Oncogene 2002, 21, 128–139. [Google Scholar] [CrossRef]
- Ganapathy, V.; Manyanga, J.; Brame, L.; McGuire, D.; Sadhasivam, B.; Floyd, E.; Rubenstein, D.; Ramachandran, I.; Wagener, T.; Queimado, L. Electronic cigarette aerosols suppress cellular antioxidant defenses and induce significant oxidative DNA damage. PLoS ONE 2017, 12, e0177780. [Google Scholar] [CrossRef] [PubMed]
- Walters, M.S.; Gomi, K.; Ashbridge, B.; Moore, M.A.S.; Arbelaez, V.; Heldrich, J.; Ding, B.-S.; Rafii, S.; Staudt, M.R.; Crystal, R.G. Generation of a human airway epithelium derived basal cell line with multipotent differentiation capacity. Respir. Res. 2013, 14, 135. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Lou, H.H.; Salit, J.; Leopold, P.L.; Driscoll, S.; Schymeinsky, J.; Quast, K.; Visvanathan, S.; Fine, J.S.; Thomas, M.J.; et al. Characterization of an immortalized human small airway basal stem/progenitor cell line with airway region-specific differentiation capacity. Respir. Res. 2019, 20, 196. [Google Scholar] [CrossRef]
- Zhang, H.; Rostami, M.R.; Leopold, P.L.; Mezey, J.G.; O’Beirne, S.L.; Strulovici-Barel, Y.; Crystal, R.G. Expression of the SARS-CoV-2 ACE2 Receptor in the Human Airway Epithelium. Am. J. Respir. Crit. Care Med. 2020, 202, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Kuehn, A. Human alveolar epithelial cells expressing tight junctions to model the air-blood barrier. ALTEX 2016, 33, 251–260. [Google Scholar] [CrossRef]
- Chang, S.-H.; Ko, P.-L.; Liao, W.-H.; Peng, C.-C.; Tung, Y.-C. Transwell Insert-Embedded Microfluidic Devices for Time-Lapse Monitoring of Alveolar Epithelium Barrier Function under Various Stimulations. Micromachines 2021, 12, 406. [Google Scholar] [CrossRef]
- Cozens, A.L.; Yezzi, M.J.; Kunzelmann, K.; Ohrui, T.; Chin, L.; Eng, K.; Finkbeiner, W.; Widdicombe, J.H.; Gruenert, D.C. CFTR expression and chloride secretion in polarized immortal human bronchial epithelial cells. Am. J. Respir. Cell Mol. Biol. 1994, 10, 38–47. [Google Scholar] [CrossRef]
- Zabner, J.; Karp, P.; Seiler, M.; Phillips, S.L.; Mitchell, C.J.; Saavedra, M.; Welsh, M.; Klingelhutz, A. Development of cystic fibrosis and noncystic fibrosis airway cell lines. Am. J. Physiol. Cell. Mol. Physiol. 2003, 284, L844–L854. [Google Scholar] [CrossRef]
- Mullins, B.; Kicic, A.; Ling, K.-M.; Mead-Hunter, R.; Larcombe, A.N. Biodiesel exhaust-induced cytotoxicity and proinflammatory mediator production in human airway epithelial cells. Environ. Toxicol. 2014, 31, 44–57. [Google Scholar] [CrossRef]
- Ramirez, R.D.; Sheridan, S.; Girard, L.; Sato, M.; Kim, Y.; Pollack, J.; Peyton, M.; Zou, Y.; Kurie, J.M.; DiMaio, J.M.; et al. Immortalization of Human Bronchial Epithelial Cells in the Absence of Viral Oncoproteins. Cancer Res. 2004, 64, 9027–9034. [Google Scholar] [CrossRef]
- Gowers, K.H.; Hynds, R.E.; Thakrar, R.M.; Carroll, B.; Birchall, M.A.; Janes, S.M. Optimized isolation and expansion of human airway epithelial basal cells from endobronchial biopsy samples. J. Tissue Eng. Regen. Med. 2017, 12, e313–e317. [Google Scholar] [CrossRef]
- Hynds, R.E.; Butler, C.R.; Janes, S.M.; Giangreco, A. Expansion of Human Airway Basal Stem Cells and Their Differentiation as 3D Tracheospheres. In Springer Protocols Handbooks; Springer: Singapore, 2016; Volume 1576, pp. 43–53. [Google Scholar]
- Hackett, N.R.; Shaykhiev, R.; Walters, M.S.; Wang, R.; Zwick, R.; Ferris, B.; Witover, B.; Salit, J.; Crystal, R.G. The Human Airway Epithelial Basal Cell Transcriptome. PLoS ONE 2011, 6, e18378. [Google Scholar] [CrossRef]
- Lechner, J.F.; Haugen, A.; McClendon, I.A.; Shamsuddin, A.M. Induction of squamous differentiation of normal human bronchial epithelial cells by small amounts of serum. Differentiation 1984, 25, 229–237. [Google Scholar] [CrossRef] [PubMed]
- Prytherch, Z.; Job, C.; Marshall, H.; Oreffo, V.; Foster, M.; Bérubé, K. Tissue-Specific Stem Cell Differentiation in an in vitro Airway Model. Macromol. Biosci. 2011, 11, 1467–1477. [Google Scholar] [CrossRef]
- Rayner, R.E.; Makena, P.; Prasad, G.L.; Cormet-Boyaka, E. Optimization of Normal Human Bronchial Epithelial (NHBE) Cell 3D Cultures for in vitro Lung Model Studies. Sci. Rep. 2019, 9, 500. [Google Scholar] [CrossRef] [PubMed]
- Li, X. In vitro toxicity testing of cigarette smoke based on the air-liquid interface exposure: A review. Toxicol. Vitr. 2016, 36, 105–113. [Google Scholar] [CrossRef]
- Aufderheide, M.; Scheffler, S.; Ito, S.; Ishikawa, S.; Emura, M. Ciliatoxicity in human primary bronchiolar epithelial cells after repeated exposure at the air–liquid interface with native mainstream smoke of K3R4F cigarettes with and without charcoal filter. Exp. Toxicol. Pathol. 2015, 67, 407–411. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Liu, W.; Song, W. Toxicity assessment of electronic cigarettes. Inhal. Toxicol. 2019, 31, 259–273. [Google Scholar] [CrossRef]
- Park, H.-R.; O’Sullivan, M.; Vallarino, J.; Shumyatcher, M.; Himes, B.E.; Park, J.-A.; Christiani, D.C.; Allen, J.; Lu, Q. Transcriptomic response of primary human airway epithelial cells to flavoring chemicals in electronic cigarettes. Sci. Rep. 2019, 9, 379139. [Google Scholar] [CrossRef]
- Pearce, K.; Gray, N.; Gaur, P.; Jeon, J.; Suarez, A.; Shannahan, J.; Pappas, R.; Watson-Wright, C. Toxicological analysis of aerosols derived from three electronic nicotine delivery systems using normal human bronchial epithelial cells. Toxicol. Vitr. 2020, 69, 104997. [Google Scholar] [CrossRef]
- Shields, P.G.; Berman, M.; Brasky, T.M.; Freudenheim, J.L.; Mathe, E.; McElroy, J.P.; Song, M.-A.; Wewers, M.D. A Review of Pulmonary Toxicity of Electronic Cigarettes in the Context of Smoking: A Focus on Inflammation. Cancer Epidemiol. Biomark. Prev. 2017, 26, 1175–1191. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Mi, Y.; Mueller, T.; Kreibich, S.; Williams, E.; Van Drogen, A.; Borel, C.; Frank, M.; Germain, P.-L.; Bludau, I.; et al. Multi-omic measurements of heterogeneity in HeLa cells across laboratories. Nat. Biotechnol. 2019, 37, 314–322. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Deng, Y.; Liu, Y.; Gong, W.; Deng, W. Stem Cell Models for Drug Discovery and Toxicology Studies. J. Biochem. Mol. Toxicol. 2013, 27, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Varma, R.; Soleas, J.P.; Waddell, T.K.; Karoubi, G.; McGuigan, A.P. Current strategies and opportunities to manufacture cells for modeling human lungs. Adv. Drug Deliv. Rev. 2020, 161–162, 90–109. [Google Scholar] [CrossRef] [PubMed]
- Faiola, F.; Yin, N.; Yao, X.; Jiang, G. The Rise of Stem Cell Toxicology. Environ. Sci. Technol. 2015, 49, 5847–5848. [Google Scholar] [CrossRef]
- Tian, L.; Gao, J.; Garcia, I.M.; Chen, H.J.; Castaldi, A.; Chen, Y. Human pluripotent stem cell-derived lung organoids: Potential applications in development and disease modeling. Wiley Interdiscip. Rev. Dev. Biol. 2020, 10, e399. [Google Scholar] [CrossRef] [PubMed]
- Loret, T.; Rogerieux, F.; Trouiller, B.; Braun, A.; Egles, C.; Lacroix, G. Predicting the in vivo pulmonary toxicity induced by acute exposure to poorly soluble nanomaterials by using advanced in vitro methods. Part. Fibre Toxicol. 2018, 15, 25. [Google Scholar] [CrossRef]
- Baxter, A.; Thain, S.; Banerjee, A.; Haswell, L.; Parmar, A.; Phillips, G.; Minet, E. Targeted omics analyses, and metabolic enzyme activity assays demonstrate maintenance of key mucociliary characteristics in long term cultures of reconstituted human airway epithelia. Toxicol. Vitr. 2015, 29, 864–875. [Google Scholar] [CrossRef]
- Cao, X.; Coyle, J.P.; Xiong, R.; Wang, Y.; Heflich, R.H.; Ren, B.; Gwinn, W.M.; Hayden, P.; Rojanasakul, L. Invited review: Human air-liquid-interface organotypic airway tissue models derived from primary tracheobronchial epithelial cells—Overview and perspectives. Cell. Dev. Biol. Anim. 2021, 57, 104–132. [Google Scholar] [CrossRef]
| Study | Methods (Summary) | References |
|---|---|---|
| Walinder et al. | Experimental study with exposition chamber | [27] |
| Araki et al. | Health outcome ascertainment coupled with GC/MS mVOC analysis | [28] |
| Saijo et al. | Health outcome ascertainment and air sampling | [29] |
| Araki et al. | Health outcome ascertainment coupled with GC/MS mVOC analysis | [30] |
| Zhang et al. | Health outcome ascertainment and air sampling | [26] |
| Sahlberg et al. | Health outcome ascertainment coupled with GC/MS mVOC analysis | [31] |
| Choi et al. | Health outcome ascertainment and air sampling | [32] |
| Airway Cell Line | Source Material | Method of Immortalization | References Material | Used in VOC or mVOC Analysis |
|---|---|---|---|---|
| A549 | Epithelial adenocarcinoma | Tumor derived | [157] | Yes [5,56,57,158,159,160,161,162,163] |
| Calu-3 | Epithelial adenocarcinoma | Tumor derived | [166] | Yes [165,167,168] |
| BEAS-2B | Bronchial epithelium | SV40 T-antigen | [170] | Yes [113,171,172] |
| 16HBE14o- | Bronchial epithelium | SV40 T-antigen | [187] | Yes [173,174,175,176] |
| NuLi-1 | Bronchial epithelium | hTERT | [188] | Yes [181,189] |
| HBEC3-KT | Bronchial epithelium | hTERT | [190] | No |
| BCi-NS1.1 | Large airways basal cell | hTERT | [182] | No |
| hAELVI | Primary alveolar epithelim | Lentivirus | [185] | Yes [186] |
| hSABCi-NS1.1 | Small airway basal cell | hTERT | [183] | No |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cerimi, K.; Jäckel, U.; Meyer, V.; Daher, U.; Reinert, J.; Klar, S. In Vitro Systems for Toxicity Evaluation of Microbial Volatile Organic Compounds on Humans: Current Status and Trends. J. Fungi 2022, 8, 75. https://doi.org/10.3390/jof8010075
Cerimi K, Jäckel U, Meyer V, Daher U, Reinert J, Klar S. In Vitro Systems for Toxicity Evaluation of Microbial Volatile Organic Compounds on Humans: Current Status and Trends. Journal of Fungi. 2022; 8(1):75. https://doi.org/10.3390/jof8010075
Chicago/Turabian StyleCerimi, Kustrim, Udo Jäckel, Vera Meyer, Ugarit Daher, Jessica Reinert, and Stefanie Klar. 2022. "In Vitro Systems for Toxicity Evaluation of Microbial Volatile Organic Compounds on Humans: Current Status and Trends" Journal of Fungi 8, no. 1: 75. https://doi.org/10.3390/jof8010075
APA StyleCerimi, K., Jäckel, U., Meyer, V., Daher, U., Reinert, J., & Klar, S. (2022). In Vitro Systems for Toxicity Evaluation of Microbial Volatile Organic Compounds on Humans: Current Status and Trends. Journal of Fungi, 8(1), 75. https://doi.org/10.3390/jof8010075






