Transmission of Onychomycosis and Dermatophytosis between Household Members: A Scoping Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy, Study Eligibility Criteria, and Study Selection
2.2. Data Extraction and Synthesis
3. Results
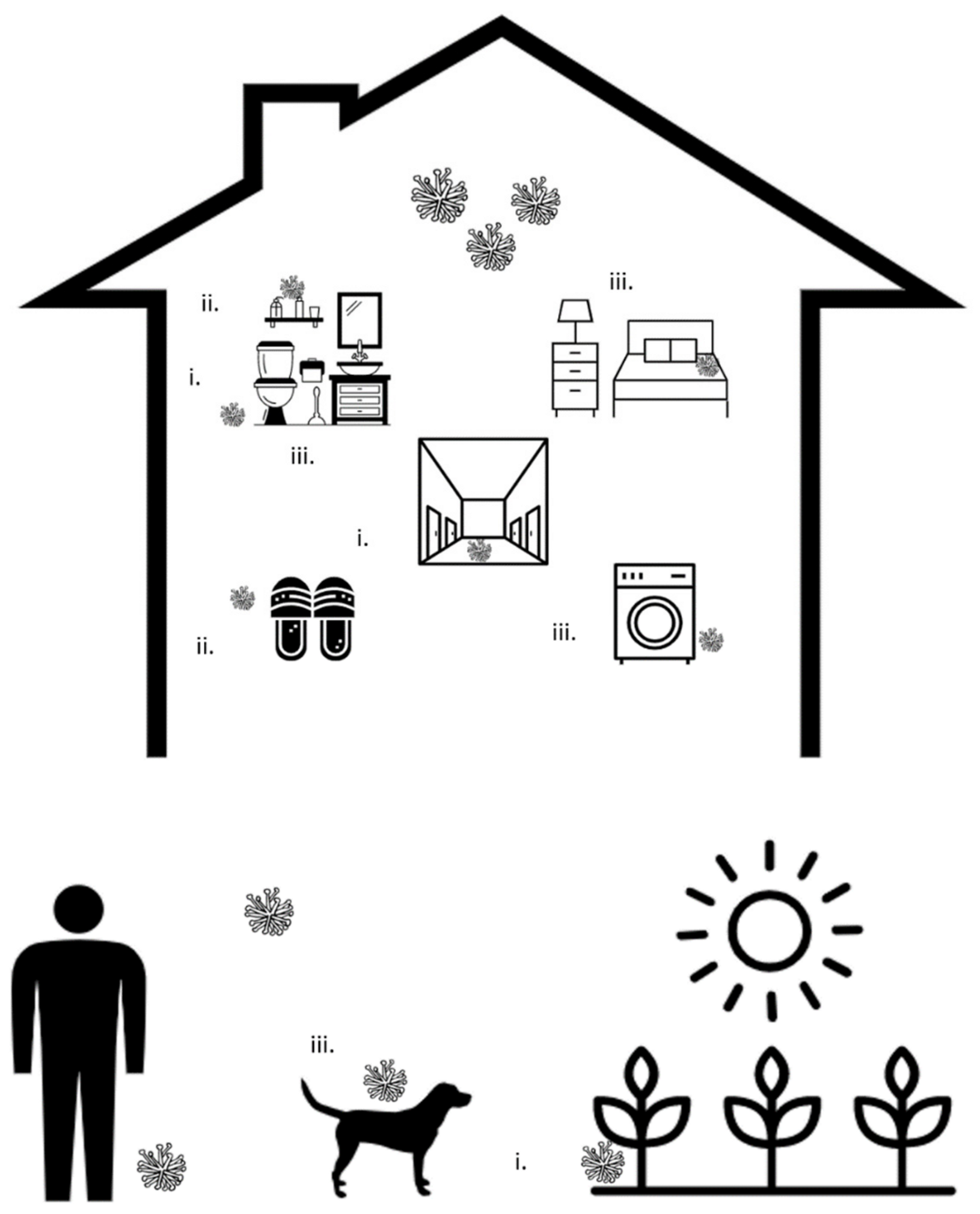
3.1. Transfer through Shared Surfaces
3.2. Transfer through Shared Equipment
3.3. Persistence of Fungi within the Household Environment
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Lipner, S.R.; Scher, R.K. Onychomycosis: Clinical Overview and Diagnosis. J. Am. Acad. Dermatol. 2019, 80, 835–851. [Google Scholar] [CrossRef] [PubMed]
- Ghannoum, M.A.; Hajjeh, R.A.; Scher, R.; Konnikov, N.; Gupta, A.K.; Summerbell, R.; Sullivan, S.; Daniel, R.; Krusinski, P.; Fleckman, P.; et al. A Large-Scale North American Study of Fungal Isolates from Nails: The Frequency of Onychomycosis, Fungal Distribution, and Antifungal Susceptibility Patterns. J. Am. Acad. Dermatol. 2000, 43, 641–648. [Google Scholar] [CrossRef] [PubMed]
- Hay, R.J.; Baran, R. Onychomycosis: A Proposed Revision of the Clinical Classification. J. Am. Acad. Dermatol. 2011, 65, 1219–1227. [Google Scholar] [CrossRef] [PubMed]
- Bolognia, J.L.; Schaffer, J.V.; Cerroni, L. Dermatology; Elsevier: Amsterdam, The Netherlands, 2017; ISBN 978-0-7020-6275-9. [Google Scholar]
- Gupta, A.K.; Mays, R.R. The Impact of Onychomycosis on Quality of Life: A Systematic Review of the Available Literature. Skin Appendage Disord. 2018, 4, 208–216. [Google Scholar] [CrossRef]
- Gupta, A.K.; Jain, H.C.; Lynde, C.W.; MacDonald, P.; Cooper, E.A.; Summerbell, R.C. Prevalence and Epidemiology of Onychomycosis in Patients Visiting Physicians’ Offices: A Multicenter Canadian Survey of 15,000 Patients. J. Am. Acad. Dermatol. 2000, 43, 244–248. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.K.; Stec, N.; Summerbell, R.C.; Shear, N.H.; Piguet, V.; Tosti, A.; Piraccini, B.M. Onychomycosis: A Review. J. Eur. Acad. Dermatol. Venereol. 2020, 34, 1972–1990. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, G.; Horwitz, B.A.; Duek, L.; Ullman, Y.; Berdicevsky, I. Infection Stages of the Dermatophyte Pathogen Trichophyton: Microscopic Characterization and Proteolytic Enzymes. Med. Mycol. 2007, 45, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Yue, X.; Li, Q.; Wang, H.; Sun, Y.; Wang, A.; Zhang, Q.; Zhang, C. An Ultrastructural Study of Trichophyton Rubrum Induced Onychomycosis. BMC Infect. Dis. 2015, 15, 532. [Google Scholar] [CrossRef]
- Esquenazi, D.; Alviano, C.S.; de Souza, W.; Rozental, S. The Influence of Surface Carbohydrates during in Vitro Infection of Mammalian Cells by the Dermatophyte Trichophyton Rubrum. Res. Microbiol. 2004, 155, 144–153. [Google Scholar] [CrossRef]
- Baldo, A.; Monod, M.; Mathy, A.; Cambier, L.; Bagut, E.T.; Defaweux, V.; Symoens, F.; Antoine, N.; Mignon, B. Mechanisms of Skin Adherence and Invasion by Dermatophytes. Mycoses 2012, 55, 218–223. [Google Scholar] [CrossRef]
- Fenstermacher, K.; Hudson, B.T. Practice Guidelines for Family Nurse Practitioners E-Book; Elsevier Health Sciences: Amsterdam, The Netherlands, 2019; ISBN 978-0-323-59481-3. [Google Scholar]
- Carrillo-Meléndrez, H.; Ortega-Hernández, E.; Granados, J.; Arroyo, S.; Barquera, R.; Arenas, R. Role of HLA-DR Alleles to Increase Genetic Susceptibility to Onychomycosis in Nail Psoriasis. Skin Appendage Disord. 2016, 2, 22–25. [Google Scholar] [CrossRef]
- Asz-Sigall, D.; López-García, L.; Vega-Memije, M.E.; Lacy-Niebla, R.M.; García-Corona, C.; Ramírez-Rentería, C.; Granados, J.; Villa, A.; Ameen, M.; Arenas, R. HLA-DR6 Association Confers Increased Resistance to T. Rubrum Onychomycosis in Mexican Mestizos. Int. J. Dermatol. 2010, 49, 1406–1409. [Google Scholar] [CrossRef]
- Faergemann, J.; Correia, O.; Nowicki, R.; Ro, B.-I. Genetic Predisposition—Understanding Underlying Mechanisms of Onychomycosis. J. Eur. Acad. Dermatol. Venereol. 2005, 19, 17–19. [Google Scholar] [CrossRef]
- Ghannoum, M.A.; Mukherjee, P.K.; Warshaw, E.M.; Evans, S.; Korman, N.J.; Tavakkol, A. Molecular Analysis of Dermatophytes Suggests Spread of Infection Among Household Members. Cutis 2013, 91, 237–245. [Google Scholar]
- English, M.P. Trichophyton Rubrum Infection in Families. Br. Med. J. 1957, 1, 744–746. [Google Scholar] [CrossRef]
- Suzuki, S.; Mano, Y.; Furuya, N.; Fujitani, K. Molecular Epidemiological Analysis of the Spreading Conditions of Trichophyton in Long-Term Care Facilities in Japan. Jpn. J. Infect. Dis. 2018, 71, 462–466. [Google Scholar] [CrossRef]
- El Fekih, N.; Belghith, I.; Trabelsi, S.; Skhiri-Aounallah, H.; Khaled, S.; Fazaa, B. Epidemiological and Etiological Study of Foot Mycosis in Tunisia. Actas Dermo-Sifiliográficas 2012, 103, 520–524. [Google Scholar] [CrossRef]
- Bonifaz, A.; Vázquez-González, D.; Hernández, M.A.; Araiza, J.; Tirado-Sánchez, A.; Ponce, R.M. Dermatophyte Isolation in the Socks of Patients with Tinea Pedis and Onychomycosis. J. Dermatol. 2013, 40, 504–505. [Google Scholar] [CrossRef]
- Klafke, G.B.; da Silva, R.A.; de Pellegrin, K.T.; Xavier, M.O. Analysis of the Role of Nail Polish in the Transmission of Onychomycosis. An. Bras. Dermatol. 2018, 93, 930–931. [Google Scholar] [CrossRef]
- Sucharew, H.; Sucharew, H. Methods for Research Evidence Synthesis: The Scoping Review Approach. J. Hosp. Med. 2019, 14, 416–418. [Google Scholar] [CrossRef]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.J.; Horsley, T.; Weeks, L.; et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and Explanation. Ann. Intern. Med. 2018, 169, 467–473. [Google Scholar] [CrossRef]
- Veritas Health Innovation. Covidence Systematic Review Software; Veritas Health Innovation: Melbourne, VIC, Australia, 2017. [Google Scholar]
- Rafiei, A.; Amirrajab, N. Fungal Contamination of Indoor Public Swimming Pools, Ahwaz, South-West of Iran. Iran J. Public Health 2010, 39, 124–128. [Google Scholar]
- Ekowati, Y.; Ferrero, G.; Kennedy, M.D.; de Roda Husman, A.M.; Schets, F.M. Potential Transmission Pathways of Clinically Relevant Fungi in Indoor Swimming Pool Facilities. Int. J. Hyg. Environ. Health 2018, 221, 1107–1115. [Google Scholar] [CrossRef]
- Tlougan, B.E.; Mancini, A.J.; Mandell, J.A.; Cohen, D.E.; Sanchez, M.R. Skin Conditions in Figure Skaters, Ice-Hockey Players and Speed Skaters: Part II—Cold-Induced, Infectious and Inflammatory Dermatoses. Sports Med. 2011, 41, 967–984. [Google Scholar] [CrossRef]
- Yenişehirli, G.; Karat, E.; Bulut, Y.; Savcı, U. Dermatophytes Isolated from the Mosques in Tokat, Turkey. Mycopathologia 2012, 174, 327–330. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, S.; Anzawa, K.; Mochizuki, T. High Prevalence of Superficial White Onychomycosis by Trichophyton Interdigitale in a Japanese Nursing Home with a Geriatric Hospital. Mycoses 2017, 60, 634–637. [Google Scholar] [CrossRef]
- Kane, J.; Leavitt, E.; Summerbell, R.C.; Krajden, S.; Kasatiya, S.S. An Outbreak of Trichophyton Tonsurans Dermatophytosis in a Chronic Care Institution for the Elderly. Eur. J. Epidemiol. 1988, 4, 144–149. [Google Scholar] [CrossRef]
- Müller, G. Occurrence of Dermatophytes in the Sands of European Beaches. Sci. Total Environ. 1973, 2, 116–118. [Google Scholar] [CrossRef]
- Shimmura, Y. Isolation of Dermatophytes from Human Cases of Dermatophytosis and from House Dust. Jpn. J. Med. Mycol. 1985, 26, 74–80. [Google Scholar] [CrossRef]
- Raboobee, N.; Aboobaker, J.; Peer, A.K.; Micro, M. Tinea Pedis et Unguium in the Muslim Community of Durban, South Africa. Int. J. Dermatol. 1998, 37, 759–765. [Google Scholar] [CrossRef]
- Ingordo, V.; Naldi, L.; Fracchiolla, S.; Colecchia, B. Prevalence and Risk Factors for Superficial Fungal Infections among Italian Navy Cadets. Dermatology 2004, 209, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Uslu, H.; Uyanik, M.; Ayyildiz, A. Mycological Examination of the Barbers’ Tools about Sources of Fungal Infections. Mycoses 2008, 51, 447–450. [Google Scholar] [CrossRef] [PubMed]
- Pomeranz, A.J.; Sabnis, S.S.; McGrath, G.J.; Esterly, N.B. Asymptomatic Dermatophyte Carriers in the Households of Children with Tinea Capitis. Arch. Pediatr. Adolesc. Med. 1999, 153, 483–486. [Google Scholar] [CrossRef][Green Version]
- Tuknayat, A.; Bhalla, M.; Kaur, A.; Garg, S. Familial Dermatophytosis in India: A Study of the Possible Contributing Risk Factors. J. Clin. Aesthet. Dermatol. 2020, 13, 58–60. [Google Scholar]
- Baumgardner, D.J. Fungal Infections From Human and Animal Contact. J. Patient-Cent. Res. Rev. 2017, 4, 78–89. [Google Scholar] [CrossRef]
- Leelavathi, M.; Noorlaily, M. Onychomycosis Nailed. Malays Fam Physician 2014, 9, 2–7. [Google Scholar]
- Jennings, M.B.; Pollak, R.; Harkless, L.B.; Kianifard, F.; Tavakkol, A. Treatment of Toenail Onychomycosis with Oral Terbinafine Plus Aggressive Debridement: IRON-CLAD, a Large, Randomized, Open-Label, Multicenter Trial. J. Am. Podiatr. Med. Assoc. 2006, 96, 465–473. [Google Scholar] [CrossRef]
- Tanaka, K.; Katoh, T.; Irimajiri, J.; Taniguchi, H.; Yokozeki, H. Preventive Effects of Various Types of Footwear and Cleaning Methods on Dermatophyte Adhesion. J. Dermatol. 2006, 33, 528–536. [Google Scholar] [CrossRef]
- Knudsen, E.A. Isolation of Dermatophytes from Footwear with Adhesive Tape Strips. J. Med. Vet. Mycol. 1987, 25, 59–61. [Google Scholar] [CrossRef]
- Ajello, L.; Getz, M.E. Recovery of Dermatophytes from Shoes and Shower Stalls1. J. Investig. Dermatol. 1954, 22, 17–24. [Google Scholar] [CrossRef]
- Gupta, A.K.; Versteeg, S.G. The Role of Shoe and Sock Sanitization in the Management of Superficial Fungal Infections of the Feet. J. Am. Podiatr. Med. Assoc. 2019, 109, 141–149. [Google Scholar] [CrossRef]
- Toukabri, N.; Dhieb, C.; El Euch, D.; Rouissi, M.; Mokni, M.; Sadfi-Zouaoui, N. Prevalence, Etiology, and Risk Factors of Tinea Pedis and Tinea Unguium in Tunisia. Can. J. Infect. Dis. Med. Microbiol. 2017, 2017, e6835725. [Google Scholar] [CrossRef]
- De Boer, D.; Moriello, K. Cuteaneous Fungal Infections; Saunders Elsevier: Amsterdam, The Netherlands, 2006. [Google Scholar]
- Oyeka, C.A.; Okoli, I. Isolation of Dermatophytes and Non-Dermatophytic Fungi from Soil in Nigeria. Mycoses 2003, 46, 318–320. [Google Scholar] [CrossRef] [PubMed]
- Alter, S.J.; McDonald, M.B.; Schloemer, J.; Simon, R.; Trevino, J. Common Child and Adolescent Cutaneous Infestations and Fungal Infections. Curr. Probl. Pediatric Adolesc. Health Care 2018, 48, 3–25. [Google Scholar] [CrossRef]
- Weese, J.S.; Fulford, M.B. Fungal Diseases. In Companion Animal Zoonoses; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2011; pp. 275–298. ISBN 978-0-470-95895-7. [Google Scholar]
- Weitzman, I.; Summerbell, R.C. The Dermatophytes. Clin. Microbiol. Rev. 1995, 8, 240–259. [Google Scholar] [CrossRef]
- Ara, K.; Hama, M.; Akiba, S.; Koike, K.; Okisaka, K.; Hagura, T.; Kamiya, T.; Tomita, F. Foot Odor Due to Microbial Metabolism and Its Control. Can. J. Microbiol. 2006, 52, 357–364. [Google Scholar] [CrossRef]
- Hammer, T.R.; Mucha, H.; Hoefer, D. Infection Risk by Dermatophytes during Storage and after Domestic Laundry and Their Temperature-Dependent Inactivation. Mycopathologia 2011, 171, 43–49. [Google Scholar] [CrossRef]
- Amichai, B.; Grunwald, M.H.; Davidovici, B.; Farhi, R.; Shemer, A. The Effect of Domestic Laundry Processes on Fungal Contamination of Socks. Int. J. Dermatol. 2013, 52, 1392–1394. [Google Scholar] [CrossRef]
- Brunton, W.A. Infection and Hospital Laundry. Lancet 1995, 345, 1574–1575. [Google Scholar] [CrossRef]
- Fijan, S.; Šostar-Turk, S.; Cencič, A. Implementing Hygiene Monitoring Systems in Hospital Laundries in Order to Reduce Microbial Contamination of Hospital Textiles. J. Hosp. Infect. 2005, 61, 30–38. [Google Scholar] [CrossRef]
- Flores, J.M.; Castillo, V.B.; Franco, F.C.; Huata, A.B. Superficial Fungal Infections: Clinical and Epidemiological Study in Adolescents from Marginal Districts of Lima and Callao, Peru. J. Infect. Dev. Ctries 2009, 3, 313–317. [Google Scholar] [CrossRef] [PubMed]
- Marchisio, V.F.; Preve, L.; Tullio, V. Fungi Responsible for Skin Mycoses in Turin (Italy). Mycoses 1996, 39, 141–150. [Google Scholar] [CrossRef]
- Tan, J.S. Human Zoonotic Infections Transmitted by Dogs and Cats. Arch. Intern. Med. 1997, 157, 1933–1943. [Google Scholar] [CrossRef] [PubMed]
- McAleer, R. Fungal Infection as a Cause of Skin Disease in Western Australia. Australas. J. Dermatol. 1981, 22, 80–84. [Google Scholar] [CrossRef]
- Nenoff, P.; Krüger, C.; Ginter-Hanselmayer, G.; Tietz, H.-J. Mycology—An Update. Part 1: Dermatomycoses: Causative Agents, Epidemiology and Pathogenesis. JDDG J. Dtsch. Dermatol. Ges. 2014, 12, 188–210. [Google Scholar] [CrossRef]
- Kraemer, A.; Hein, J.; Heusinger, A.; Mueller, R.S. Clinical Signs, Therapy and Zoonotic Risk of Pet Guinea Pigs with Dermatophytosis. Mycoses 2013, 56, 168–172. [Google Scholar] [CrossRef]
- Bartosch, T.; Frank, A.; Günther, C.; Uhrlaß, S.; Heydel, T.; Nenoff, P.; Baums, C.G.; Schrödl, W. Trichophyton Benhamiae and T. Mentagrophytes Target Guinea Pigs in a Mixed Small Animal Stock. Med. Mycol. Case Rep. 2019, 23, 37–42. [Google Scholar] [CrossRef]
- Segal, E.; Elad, D. Human and Zoonotic Dermatophytoses: Epidemiological Aspects. Front. Microbiol. 2021, 12, 713532. [Google Scholar] [CrossRef]
- Dupont, D.; Peyron, F.; Picot, S.; Wallon, M.; Bienvenu, A.-L. Dermatophytes and Transmission Risks to Family: A Retrospective Study of a Cohort of 256 Adopted Children from 1998 to 2012. Pediatr. Dermatol. 2015, 32, 426–427. [Google Scholar] [CrossRef]
- Jang, K.-A.; Chi, D.-H.; Choi, J.-H.; Sung, K.-J.; Moon, K.-C.; Koh, J.-K. Tinea Pedis in Korean Children. Int. J. Dermatol. 2000, 39, 25–27. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.S.; Ohn, J.; Mun, J.-H. Diagnosis of Onychomycosis: From Conventional Techniques and Dermoscopy to Artificial Intelligence. Front. Med. 2021, 8, 637216. [Google Scholar] [CrossRef]
| Search | Keywords |
|---|---|
| 1 | Onychomycos* |
| 2 | tinea unguium |
| 3 | Dermatophyte* |
| 4 | disease transmission, infectious |
| 5 | transmission |
| 6 | household |
| 7 | 1 or 2 or 3 |
| 8 | 4 or 5 or 6 |
| 9 | 7 and 8 |
| Minimizing Spread from Shared Surfaces |
| Properly disinfect floors and bathtubs, particularly in high traffic areas (e.g., entrances, hallways) |
| Wear socks or slippers around the house |
| Regularly wash feet and wipe them dry |
| Minimizing spread from shared equipment |
| Minimize sharing of slippers or socks, and disinfect slippers regularly |
| Minimize sharing of nail polish and nail tools (clippers, scissors, files) |
| Disinfect nail tools before and following use |
| Minimizing persistence of dermatophytes in the household environment |
| Use single-use cleaning supplies or regularly disinfect cleaning tools to prevent the spread of fungi |
| Launder textiles at temperatures > 60 °C |
| Regularly disinfect laundry machines |
| Maintain hygiene of pets |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jazdarehee, A.; Malekafzali, L.; Lee, J.; Lewis, R.; Mukovozov, I. Transmission of Onychomycosis and Dermatophytosis between Household Members: A Scoping Review. J. Fungi 2022, 8, 60. https://doi.org/10.3390/jof8010060
Jazdarehee A, Malekafzali L, Lee J, Lewis R, Mukovozov I. Transmission of Onychomycosis and Dermatophytosis between Household Members: A Scoping Review. Journal of Fungi. 2022; 8(1):60. https://doi.org/10.3390/jof8010060
Chicago/Turabian StyleJazdarehee, Aria, Leilynaz Malekafzali, Jason Lee, Richard Lewis, and Ilya Mukovozov. 2022. "Transmission of Onychomycosis and Dermatophytosis between Household Members: A Scoping Review" Journal of Fungi 8, no. 1: 60. https://doi.org/10.3390/jof8010060
APA StyleJazdarehee, A., Malekafzali, L., Lee, J., Lewis, R., & Mukovozov, I. (2022). Transmission of Onychomycosis and Dermatophytosis between Household Members: A Scoping Review. Journal of Fungi, 8(1), 60. https://doi.org/10.3390/jof8010060






