1. Introduction
The undifferentiated body (or thallus) of a typical filamentous fungus is the mycelium, a complex network of branched tubular cells called hyphae. In most fungi, these are partitioned into distinct cellular compartments by internal cross-walls, the septae [
1,
2]. Hyphal compartments are continuous with one another due to the occurrence of septal pores, which allow the intercellular flow of cytoplasm, including organelles, ensuring the rapid translocation of nutrients during colony establishment and facilitating the maintenance of cellular homeostasis during mycelial growth [
3]. However, this ability of the mycelium to function as an integrated syncytium-like organism exposes it to a number of risks, e.g., excessive cytoplasmic leakage upon hyphal wounding or uncontrolled spread of selfish genetic elements (such as mycoviruses, transposable elements, and senescence plasmids) following vegetative hyphal fusion. Therefore, the intercompartmental traffic in hyphae must be highly regulated in order to protect the mycelium from such hazards, as well as to permit cellular heterogeneity and differentiation during developmental processes [
3,
4,
5,
6,
7].
Early studies in various ascomycetes implicated a specialized membrane-bound vesicle described as Woronin body (WB) in sealing of septal pores to prevent cytoplasmic loss in response to hyphal damage [
8,
9]. It was later shown that WBs are involved in the control of intercellular communication and the maintenance of hyphal heterogeneity [
10]. Woronin bodies, which are exclusively found in Pezizomycotina [
9], originate from peroxisomes, and their biogenesis requires multiple peroxins [
11]. They appear in electron microscopy images as electron-dense vesicles that are usually localized in close proximity to the septae, and they are rapidly tethered to the septal pore by interacting proteins following hyphal wounding [
12,
13]. At least 17 septal pore-associated WB-related proteins have been detected by mass spectrometry in
Neurospora crassa [
14].
The protein Hex1 was first characterized in
N. crassa as the major component of WBs [
15]. This protein spontaneously self-assembles into hexagonal crystals that comprise the dense core of WBs [
16]. Conserved homologs of the
hex1 gene have been identified and studied in several members of the Pezizomycotina [
17,
18,
19,
20,
21,
22,
23,
24,
25,
26]. The gene presumably resulted from duplication of the ancestral gene encoding the eukaryotic initiation factor 5A (eIF-5A) [
16]. After this duplication,
hex1 followed a different evolutionary trajectory that led to a new function by acquiring those amino acids that are responsible for its peroxisomal targeting and self-assembly.
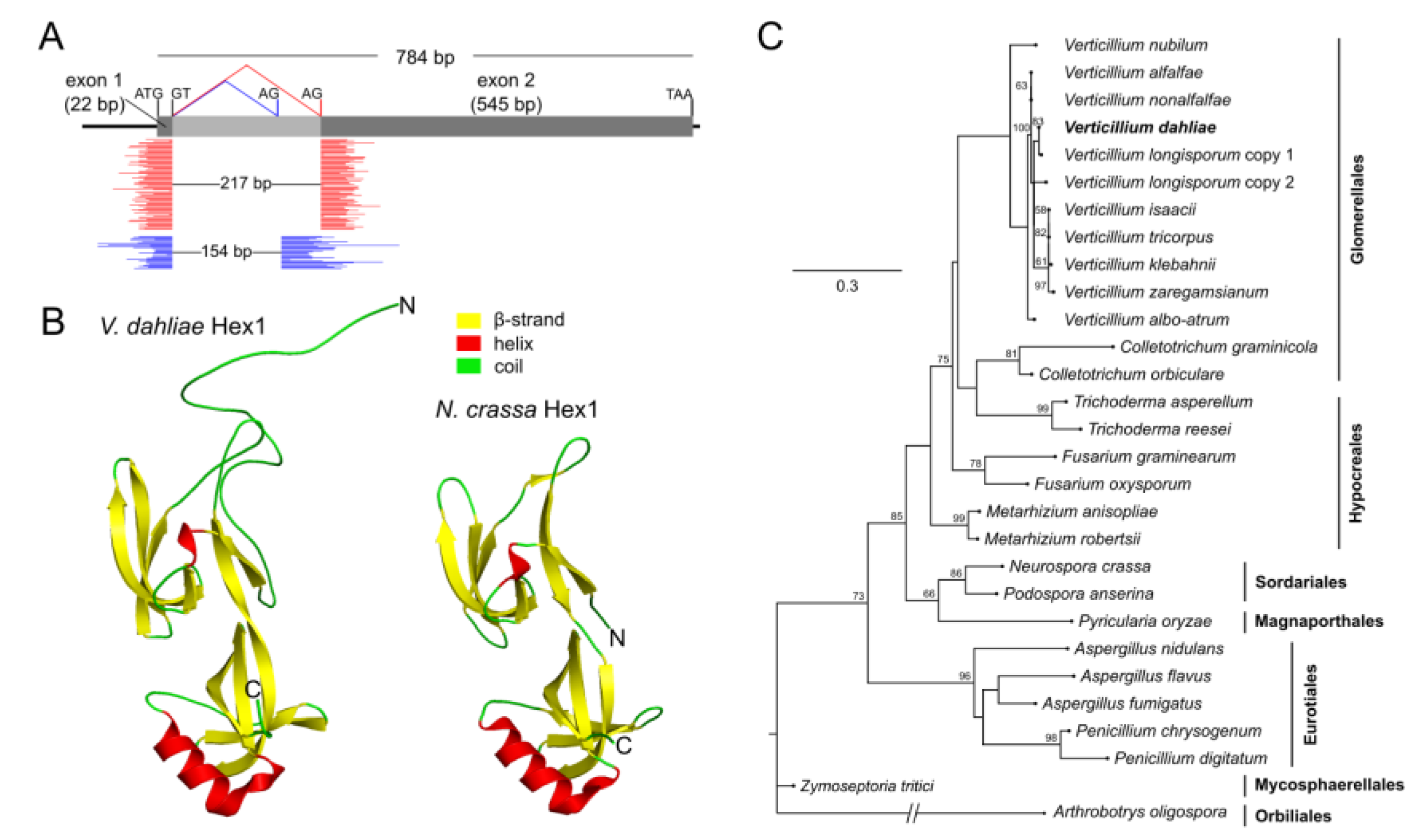
Hex1 genes retain a conserved intron close to their N-terminus, and alternative splicing of this intron has been shown to produce two isoforms of the protein [
17,
18,
19].
Deletion of
hex1 in several fungi generally led to excessive hyphal bleeding after wounding [
20,
27], as well as had pleiotropic effects on additional phenotypes associated with vegetative growth, asexual reproduction, and stress response against osmotic and cell wall-perturbating agents [
24,
25,
26]. Regarding pathogenicity and virulence, conflicting results have been gathered from studies on plant, insect, and human pathogens. These range from important roles of Hex1 in the formation of appressoria (infection structures) and pathogenicity in
Magnaporthe grisea [
18] to moderately reduced or delayed virulence in
Fusarium graminearum [
22],
Aspergillus flavus [
25], and
A. fumigates [
13], and no significant defects in pathogenicity of
Colletotrichum orbiculare [
23] and
Metarhizium robertsii [
26].
The ascomycete
Verticillium dahliae causes a wilt disease on a wide range of commercially important plants and crops, and it is responsible for enormous annual economic losses worldwide [
28]. Its resting structures (microsclerotia), which can remain viable in the soil for several years, germinate upon induction by exudates from adjacent plant roots to form hyphae. This is followed by root penetration and colonization, leading the fungus to the host vascular system, through which it can cause systemic infection of the plant [
29]. Except for the apical cells of its mycelium, hyphal compartments of
V. dahliae are regularly septated, and, upon hyphal injury, WBs rapidly plug the pores of the flanking septae to seal damaged compartments [
30,
31]. This fungus is thought to completely lack a sexual stage and, thus, propagates exclusively through the dispersion of its asexual spores (conidia). A parasexual cycle, initiated by hyphal fusion between individuals with different genotypes to the formation of heterokaryons, has been described in
Verticillium and is thought to increase genetic diversity through random chromosome assortment and frequent mitotic recombination [
28,
32]. Nevertheless, heterokaryon formation is restricted by incompatibility barriers [
28,
31], which seal and destroy incompatible fused cells, while the remaining mycelium remains unaffected [
7,
33].
This study aimed at the identification and functional characterization of hex1 in fundamental biological processes of the important plant pathogen V. dahliae. For this, we deleted its hex1 homolog to address the involvement of the gene in hyphal integrity, growth and development, pathogenicity, response to osmotic, cell wall-perturbating, and oxidative stress, and heterokaryon incompatibility. Our investigations revealed important roles of Hex1 in the biology of V. dahliae.
2. Materials and Methods
2.1. Fungal Strains, Culture Conditions, and Fungal DNA Isolation and Manipulation
All fungal strains that were constructed and used in this study are listed in
Table S1 (Supplementary Materials). Preparation and maintenance of monoconidial strains, culture media, and conditions were previously described [
34].
Total genomic DNA of fungal strains was extracted according to published methods [
34]. Standard and previously described procedures were followed for PCR amplification (all oligos are listed in
Table S2, Supplementary Materials), cloning and maintenance of plasmids in
E. coli strain DH5α (a list of plasmids is provided in
Table S3, Supplementary Materials), restriction digestion, and Sanger sequencing [
34,
35].
2.2. Agrobacterium tumefaciens-Mediated Transformation of V. dahliae
The hypervirulent
A. tumefaciens strain AGL-1 was transformed with binary plasmid vectors using a CaCl
2/heat shock-mediated freeze–thaw method [
36]. The resulting
A. tumefaciens strains were used for transforming
V. dahliae with protocols modified from [
37]. After growth of
A. tumefaciens in minimal medium (MM) supplemented with the proper selective antibiotics for 48 h (28 °C, 180 rpm), the culture was diluted to OD
600 = 0.1 and incubated for 6 h in acetosyringone-containing induction medium (IM; 25 °C, 180 rpm, without antibiotics). Aliquots of this culture (100 μL) were then mixed with 100 μL aliquots of a conidial suspension of the desired
V. dahliae strain (10
7 conidia/mL, prepared from 7 day old cultures) and plated on sterile cellophane sheets on IM agar. The mixture was incubated for 48 h at 25 °C before its transfer to selective medium (potato dextrose agar, PDA) amended with 200 μg/mL cefotaxime, 50 μg/mL 5-fluorouracil, and 100 μg/mL geneticin and/or 15 μg/mL hygromycin B).
2.3. Protoplast Transformation of V. dahliae
At least 108 conidia of the desired V. dahliae strain were incubated in Czapek-Dox complete medium (CzD-CM) for 16 h (25 °C, 175 rpm). Conidia/germlings were then pelleted, resuspended into a phosphate-KCl buffer (0.6 M KCl, 1/15 M KH2PO4, and 1/15 M Na2HPO4, pH 5.8; supplemented with 25 mM dithiothreitol (DTT) and incubated for 2 h at 30 °C (50 rpm). Protoplastation was performed by transferring the treated germlings to fresh phosphate-KCl buffer supplemented with 8 mg/mL of a lysing enzyme mix from Trichoderma harzianum (Sigma-Aldrich, St. Louis, MO, USA) and further incubating the mix at 30 °C (50 rpm) for 1–3 h. Protoplasts were separated from aggregated cellular debris by filtering through four layers of sterile gauge and resuspended into STC solution (1.2 M sorbitol, 10 mM Tris-Cl pH 7.5, and 50 mM CaCl2) at a final concentration of 108/mL. Aliquots (100 μL) of this protoplast suspension were mixed with 2–8 μg of DNA and incubated for 30 min on ice before the addition of 200 μL of PTC buffer (40% PEG-6000, 25 mM Tris-Cl pH 7.5 and 25 mM CaCl2) and further incubation for 20 min on ice. This was followed by the stepwise addition of 1 mL of fresh PTC buffer and incubation for 15 min on ice. The mixture was then washed three times with STC buffer, resuspended into fresh STC buffer, and plated on regeneration agar medium (20.54 g sucrose, 0.1 g yeast extract, and 0.14 g casamino acids per 100 mL), overlaid with a sterile cellophane sheet. Following incubation at 25 °C for 24 h, the cellophane sheet was transferred onto the appropriate selective medium (PDA amended with 100 μg/mL geneticin and/or 15 μg/mL hygromycin B).
2.4. Deletion, Complementation, and sGFP-Tagging of V. dahliae hex1
The NEBuilder HiFi DNA Assembly Master Mix (New England Biolabs, Ipswich, MA, USA) was used (according to the manufacturer’s recommendations) for the construction, in two steps, of a plasmid vector for hex1 deletion by homologous recombination. The first reaction resulted in a plasmid containing 2000 bp long homologous arms (amplified from the genomic DNA of V. dahliae isolate Ls.17 using the Herculase II Fusion DNA polymerase; Agilent, Santa Clara, CA, USA) flanking the neo cassette (conferring resistance to geneticin) from plasmid pSD1, in the backbone of plasmid pOSCAR. A second NEBuilder HiFi DNA Assembly reaction was then used for the addition to the construct of the hsv-tk thymidine kinase gene (from plasmid pGKO2) for selection against ectopic transformants. This recombinant plasmid vector (pOSCAR-hex-KO) was used for transformation of V. dahliae, using the Agrobacterium tumefaciens strain AGL-1. Monoconidial cultures were prepared from resistant V. dahliae colonies, validated as hex1 deletion mutants by spore PCR and Southern blot analysis (DIG DNA Labeling and Detection Kit, Sigma-Aldrich), and stored as 25% glycerol stocks at −80 °C.
The full-length coding sequence of hex1 plus 2000 bp long flanks were amplified from the genomic DNA of V. dahliae Ls.17 (using the KAPA HiFi DNA polymerase, Roche, Basel, Switzerland) for complementing hex1 deletion strains. The resulting 4.8 kb long amplicon was co-transformed into V. dahliae protoplasts with plasmid pUCATPH, which contains the hph cassette (conferring resistance to hygromycin B). Monoconidial transformants were confirmed by spore PCR and stored at −80 °C.
We performed C-terminal sGFP-tagging of hex1 by using the NEBuilder HiFi DNA Assembly kit to generate a construct consisting of the full-length hex1 open reading frame (ORF) together with its 2000 bp long upstream flank (which presumably includes its endogenous promoter) fused to the sGFP coding sequence from plasmid pIGPAPA, the terminal region of A. nidulans tef1, and the hph selection cassette from plasmid pFC332. The Herculase II Fusion DNA Polymerase was used for the amplification of all fragments, which were then assembled into a new recombinant plasmid (pOSCAR-hex-GFP) in the backbone of pOSCAR by a NEBuilder reaction. This was then transformed into V. dahliae Δhex1 mutants using the A. tumefaciens strain AGL-1. Resistant fungal colonies were single-cell purified, microscopically checked, and stored at −80 °C.
2.5. Cytoplasmic and Nuclear Fluorescent Labeling of V. dahliae
The sGFP gene was used for fluorescent labeling of either the cytoplasm or histone H1 (as a nuclear label) of V. dahliae strains. Construction of strains with cytoplasmic sGFP expression was performed by cloning the sGFP expression cassette from plasmid pIGPAPA and the neo cassette from plasmid pSD1 to the polylinker of plasmid pBluescript II (using the T4 DNA ligase, Takara Bio, Kusatsu, Japan). The resulting plasmid pBS-GFP-gen was used to transform V. dahliae protoplasts, and monoconidial transformants were checked microscopically for robust cytoplasmic sGFP expression.
Plasmid pMF357, which carries the hph cassette and a fusion construct of the sGFP gene to the N. crassa histone H1 gene, was used to transform V. dahliae protoplasts for nuclear labeling. Monoconidial transformants were checked microscopically for green nuclear fluorescence.
2.6. Morphological and Physiological Characterization of V. dahliae Strains
Previously described methods [
34] were used for the morphological and physiological characterization of wild-type and mutant
V. dahliae strains, with minor modifications. Colony diameters were measured for the determination of growth rates every 5 days during growth on CzD-CM, at 25 °C. Mycelial plugs (1.0 cm in diameter) from the colony periphery of 20 day old cultures were transferred into 500 μL of sterile water and vortexed extensively before determining conidial concentrations using a Neubauer improved cell counting chamber. Frequency of germination was determined by inoculating 1.0 × 10
6 conidia of each strain into CM and checking microscopically for germling formation after 14 h of growth at 25 °C. All experiments were performed in triplicate.
2.7. Plant Pathogenicity Assays
Preparation of eggplant seedlings and conidial suspensions from
V. dahliae cultures for their pathogenicity assessment was performed as previously described [
38], with minor modifications. Seedlings were transplanted at the one true-leaf stage in 100 mL pots containing soil substrate, before drenching with 20 mL of conidial suspension (5.0 × 10
6 conidia/mL) or sterile water (control plants) per pot. Plants were maintained under controlled conditions at 23 ± 3 °C with a 12 h light/dark cycle.
Typical wilt symptoms were recorded every 3–4 days for 29 days post inoculation (d.p.i.) for monitoring disease progress over time, and at 45 d.p.i. for the determination of final disease severity. Disease parameters were recorded according to previously described criteria [
38,
39]. Disease severity at each time point was defined as the percentage of symptomatic leaves, and results were plotted over time to generate disease progress curves. These were used for the determination of relative AUDPC (area under disease progress curve) values [
38]. In addition, plant growth parameters were recorded at 29 d.p.i. [
39].
The presence of the applied
V. dahliae strain in plant tissues was assessed by fungal re-isolation to verify systemic infection at 29 d.p.i., according to [
38]. In particular, nine plant stems per treatment were randomly selected and three xylem chips from different sites along the stem (base, middle, and upper part) were transferred onto acidified PDA medium after the removal of the phloem. The pathogen isolation ratio was expressed as the percentage of positive xylem chips of each plant.
2.8. Stress Response Assays and Reactive Oxygen Species (ROS) Detection
Two methods were used to analyze the sensitivity of V. dahliae wild-type and mutant strains to varying concentrations of a number of stress-inducing factors and chemicals that are commercially used as fungicides. In summary, we tested growth responses to factors that induce osmotic stress (NaCl, sorbitol), oxidative stress (H2O2, paraquat, N-acetyl cysteine, iprodione), cell wall/plasma-membrane damage or perturbation (calcofluor white M2R, Congo red, SDS, amphotericin B (Biosera, Nuaille, France), fluconazole (Pfizer, Brooklyn, NY, USA), farnesol), and other fungicides that inhibit sporulation and germination (cymoxanil, fosetyl-Al (Bayer, Leverkusen, Germany), mandipropamid, cyflufenamid); chemicals were purchased from Sigma-Aldrich, unless otherwise specified, and fresh solutions were prepared in water directly before each experiment. The first method was a spot dilution assay, in which 10 μL of 10-fold serial dilutions of conidial suspensions (5.0 × 106 to 5.0 × 103 conidia/mL) were spotted on CM agar plates containing the desired stress factor and incubated at 25 °C for 3 days. In addition, for each stress condition/concentration, relative growth inhibition was determined according to the formula: % growth inhibition = ((colony diameter on CM − colony diameter in stress condition)/(colony diameter on CM)) × 100. For each substance considered, a range of concentrations were tested in preliminary experiments for the determination of the minimum inhibitory concentration (MIC) for the wild-type strain, and an appropriate value above the MIC yielding reproducible results between replicates was chosen for further analyses. Each experiment was performed in triplicate.
The generation/accumulation of superoxide anion radicals (O2−) in the mycelium of V. dahliae strains was also examined in response to stress-inducing compounds. For this, the nitro blue tetrazolium chloride (NBT; Cayman Chemical, Ann Arbor, MI, USA) staining method was used; fungal colonies were grown on CM plates supplemented with the desired stress factor for 20 days before the addition of 10 mL of a 0.2% NBT solution on each plate. Following incubation at 25 °C (in the dark) for 45 min, the solution was discarded; then, plates were washed with ethanol, incubated again for 45 min in the dark, and briefly air-dried before recording the assessment of the staining result. All staining assays were performed in triplicate.
2.9. Generation of Nit Mutants and Heterokaryon Compatibility Tests
Nitrate non-utilizing (
nit) mutants of the
V. dahliae Δ
hex1 strain were generated by the ultraviolet irradiation method and selection on the water agar chloratemedium (WAC), as previously described [
40]. Phenotypic classification of
nit mutants and complementation tests in 96-well plates were also performed according to our previously described procedures [
40]. Each pairing was performed in three independent repetitions.
2.10. Microscopy
Fungal conidia, germlings, and hyphae were imaged using a Zeiss Axioplan epifluorescence microscope equipped with a differential interference contrast (DIC) optical system, a set of filters G 365 nm (excitation) and LP 420 nm (emission), and a Zeiss Axiocam MRc5 digital camera. Calcofluor white M2R (Sigma-Aldrich) was used for cell wall staining by its addition to the samples (to a final concentration of 10 μg/mL) and incubation at room temperature for 5 min before imaging. Methylene blue staining (final concentration 0.005% w/v) was used to differentiate live from dead cells. The samples were incubated at 25 °C for 5 min, in the dark, before imaging.
Time-lapse microscopy was performed using a Nikon Ti-E epifluorescence microscope equipped with an autofocus system (Perfect Focus System, Nikon, Minato, Japan), a light-emitting diode (LED) light engine (SpectraX, Lumencor, Beaverton, OR, USA), filter sets 390/18 and 435/48 or 469/35 and 525/50 (excitation and emission filter, respectively; all from Semrock, Rochester, NY, USA except for 525/50, which was from Chroma, Bellows Falls, VT, USA), and a scientific complementary metal–oxide–semiconductor (sCMOS) camera (Flash4, Hamamatsu, Shizuoka, Japan). Images were acquired every 10 min for 24 h (with exposure of 50 and 20 ms for the green and blue channel, respectively), and later processed using ImageJ [
41]. Images were adjusted to uniform contrast across all time points, pixel intensity plots were generated using the plot profile (for septae) and surface plot (for the whole area) functions of ImageJ, and background subtraction was performed with a rolling ball radius of 50.0 pixels and default parameters.
2.11. Bioinformatic Analyses and Statistics
Homology searches for the identification of Hex1 orthologs in
V. dahliae and other fungal species were performed using the basic local alignment search tools BlastN, tBlastN, and BlastP searches (NCBI;
https://blast.ncbi.nlm.nih.gov/Blast.cgi). All protein sequences were retrieved from GenBank (NCBI;
https://www.ncbi.nlm.nih.gov/genbank) and aligned using the MUSCLE algorithm implemented in MEGA X [
42]; the alignment was then improved using ClustalW and manually corrected. Reads from previously published RNA-seq experiments of
V. dahliae were obtained from the Sequence Read Archive (NCBI SRA;
https://www.ncbi.nlm.nih.gov/sra; datasets with accession numbers SRP020910, SRP013922, SRP041118, SRP119401, SRP198907, SRP183605, SRP244752, ERP123035, and ERP002524) using BLAST analyses and aligned to the
hex1 coding sequences that flank the predicted intron, using MEGA X.
The maximum likelihood (ML) method, implemented in the MEGA X software suite, was used for the generation of the phylogenetic tree of Hex1 sequences. Sequence alignment was performed using the MUSCLE algorithm, and then it was manually corrected. The Le Gascuel (LG) model, using a discrete Gamma distribution (parameter = 0.4452) with five rate categories, exhibited the lowest Bayesian information criterion (BIC) and corrected Akaike information criterion (AICc) values and was, therefore, chosen as the best-fit substitution model for the construction of the tree. The neighbor-join (NJ) and BioNJ algorithms were applied to generate the initial tree for the heuristic ML search, and the partial deletion option was used for handling alignment gaps. Bootstrap analysis with 1000 replications was performed for assessing tree confidence.
Prediction of the Hex1 secondary structure was performed using the PSIPRED 4.0 Workbench (UCL-CS Bioinformatics, London, UK) [
43]. For homology modeling of the Hex1 protein, the Hidden Markov Model-based tool HHPred [
44] and MODELLER 9.25 [
45] were used, based on the highly similar crystal structure of Hex1 from
N. crassa (Protein Data Bank, PDB 1KHI) (probability: 99.93%, E-value: 1.9 × 10
−23). Protein structures were visualized and compared using PyMOL 2.4 (
https://pymol.org/2).
Statistical significance of differences between compared datasets was assessed using the analysis of variance (ANOVA) test, following the evaluation of homogeneity of variance across samples (F-test p ≤ 0.05). Datasets were then subjected to means separation using Tukey’s honestly significant difference (HSD) test.
4. Discussion
Multicellularity has independently evolved from unicellular ancestors in different eukaryotic lineages, with diverse outcomes [
51]. In filamentous fungi, these transitions led to the evolution of the syncytial mycelium, which is characterized by cytoplasmic continuity [
52]. This ensures efficient translocation of nutrients and genetic resources [
53] and coordination of responses to environmental changes [
54], without the need for a dedicated vascular system. Nevertheless, uncontrolled intercompartmental traffic would lead to increased exposure to biotic and abiotic risks that could rapidly propagate throughout the mycelium [
3,
4,
5,
6,
7], which underlines the necessity for a trade-off between continuity and conditional control of exchanges. Fungal evolution has invented structures and mechanisms that address this need, including septation of hyphae in higher fungi (ascomycetes and basidiomycetes) [
2] and regulated mechanisms for the control of cytoplasmic flow through septal pores [
8]. In ascomycetes, the peroxisome-derived WBs are involved in this process of plugging septal pores as a response to mechanical or other stimuli [
9,
27,
55].
The major component of WBs was shown to be the conserved peroxisomal protein Hex1 in
N. crassa [
15,
17]. Its discovery was followed by detailed structural and functional analyses, mostly in this and to a lesser extent in other fungi [
13,
16,
18,
20,
26]. In this study, we identified the single Hex1 homolog of
V. dahliae, a plant pathogen with a particularly broad range of hosts and significant economic impact [
28]. A careful study of the deposited gene model, using automatic annotation, led us to revise the exon–intron boundaries of the gene and further revealed that it is subject to alternative splicing, a process that appears to be very common in
V. dahliae [
56] and which, specifically for
hex1, has also been reported in certain other fungi [
17,
18,
19]. The expression of two protein isoforms has also been experimentally demonstrated in other species using Hex1-specific antibodies [
17,
18,
21], which corroborates our suggestion. The inaccuracy of automatic gene annotation in this case might explain the predicted hypervariable N-terminal extensions observed in many sequences from different species [
25,
26], indicating that these data should be treated with caution.
Woronin bodies are peroxisomal vesicles with a dense core of self-assembled Hex1 into a crystal lattice [
16]. Both
V. dahliae isoforms of Hex1 were predicted to adopt essentially the same tertiary structure as their
N. crassa homolog [
16], with the exception of the N-terminal region, which is extremely variable in sequence between fungi and seems to remain in a disordered state. Typical peroxisomal targeting signal peptides [
47] were detected at their C-termini, and the conserved histidine residue that is necessary for crystallization of the protein is also present [
16]. None of the isoforms have a polyhistidine motif in the variable N-terminal region, which in
A. fumigatus is necessary for septal targeting of Hex1 [
13]. However, such motifs are also absent from other Hex1 homologs, including that of
N. crassa [
17]. Moreover, C-terminal tagging of the
V. dahliae Hex1 with sGFP demonstrated that Hex1 is still targeted to the septum, despite the lack of this motif. Overall, these findings suggest that more than one mechanism mediates septal targeting of Hex1 in fungi. On the basis of this localization pattern and the well-established role of Hex1 in septal pore plugging, we investigated the resilience of mutant Δ
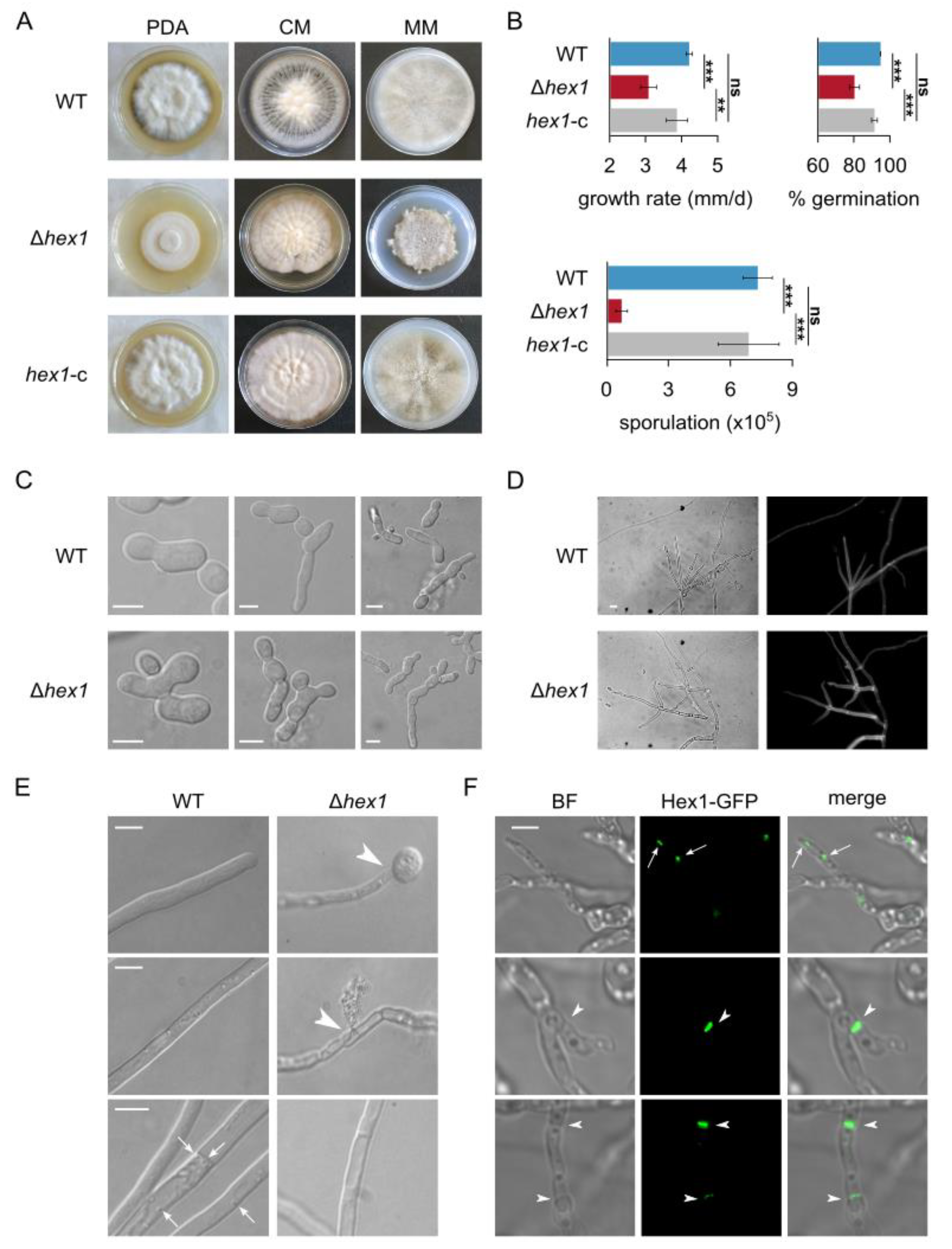
hex1 hyphae. In full concordance with its expected function, the lack of Hex1 rendered
V. dahliae hyphae prone to excessive hyphal bleeding upon hypotonic shock-induced hyphal damage. This suggests defects in hyphal compartmentalization due to dysfunctional septal pore sealing.
Growing evidence from various fungi indicates that, apart from its defining role in septal plugging upon damage, Hex1 has pleiotropic phenotypes in fungal morphology and physiology [
13,
17,
18,
22,
26]. Some of these reports present contradictory results regarding particular phenotypes in different species, including growth characteristics and pathogenicity. These considerations served as our motivation to investigate a number of hypothesized roles of Hex1 in physiology, pathogenicity, stress response, ROS metabolism, and heterokaryon incompatibility in
V. dahliae. Our results suggest the multifaceted involvement of Hex1 in several fundamental processes in this fungus. Deletion of
hex1 led to reduced growth rate, similarly to
N. crassa [
15,
17] and
Arthrobotrys oligospora [
24]
, unlike several other fungi, in which deletion of the gene did not result in growth defects [
18,
21,
22,
25]. The absence of Hex1 from
V. dahliae also resulted in drastic reduction in conidiogenesis, which has been generally observed [
17,
22,
24,
25]. Furthermore, we identified in
V. dahliae developmental phenotypes that were hitherto unknown for this gene, namely, restricted and abnormal conidial germination, increased hyphal branching, “curly” hyphal tips, and “bubble”-like cells. Collectively,
V. dahliae Hex1 appears to be involved in a number of fundamental processes that are related to asexual reproduction, hyphal development, and possibly cell wall deposition and cellular polarity. An enrichment of WBs in the apical region of germ tubes and hyphal tips has been described in
N. crassa and
A. nidulans [
11,
57,
58], and their peroxisome-unassociated apical clustering has been shown in
A. fumigatus [
21]. These resemble the localization pattern of the Spitzenkörper, the organizing center of hyphal growth that coordinates the vesicle-mediated delivery of cell wall material to the apical cell surface [
59]. These observations and the pleiotropic growth phenotypes that we report here open up the possibility that Hex1 and WBs might have multiple cellular roles in growth and morphogenesis of
V. dahliae and other fungi, some of which might be independent from their established role in septal pore occlusion upon damage, a hypothesis that welcomes future investigation.
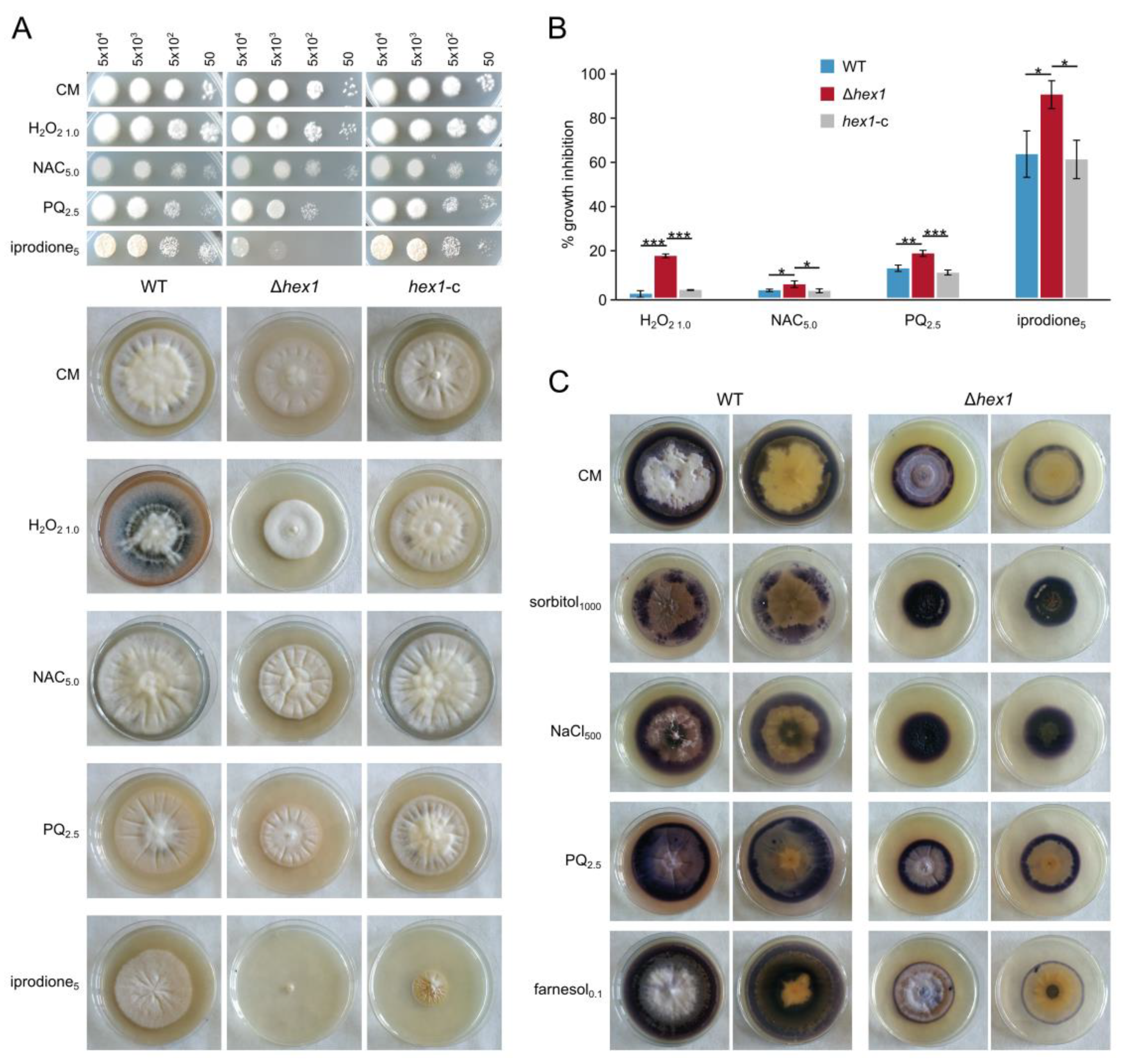
Motivated by the identification of phenotypes in Δ
hex1 cells that suggest reduced cell wall robustness, including the detection of hyphal bleeding from internal compartments upon hypotonic shock and the “curly” hyphal tips that were often observed, we further investigated the responses of the mutant strain to osmotic stress and to factors that affect cell wall and membrane integrity. We found that, apart from growth inhibition, presumably due to the reduced ability of the hyphae to isolate their damaged cellular compartments, hyperosmotic conditions also caused increased septation and frequent appearance of morphological alterations, further supporting the hypothesis that Hex1 contributes to the regulation of colony morphogenesis. Moreover, our experiments demonstrated that Hex1 in
V. dahliae mediates resistance against common fungicides that target the cell wall and the plasma membrane. In the absence of Hex1, we observed reduced germination of conidia and growth rate upon treatment with either calcofluor white or Congo red, which indicates that changes in cell wall structure and related defects in hyphal integrity possibly result in impaired germination. Contrasting results have been reported with regard to the sensitivity of Δ
hex1 mutant strains of other fungi to osmotic and cell wall/plasma-membrane stress; deletion of the gene did not significantly affect
M. robertsii and
A. flavus [
25,
26], whereas, in
A. fumigatus and
Arthrobotrys oligospora, it had a significant impact [
13,
24].
We further hypothesized and investigated whether Hex1 could be involved in the fungal response to oxidative stress and ROS metabolism. The mutant strain indeed exhibited growth defects in coping with increased oxidative damage induced by treatment with H
2O
2 and paraquat, as well as with amphotericin B, farnesol, and iprodione, but not fluconazole. This may indicate that its hyphal development might be affected not only by inhibition of ergosterol biosynthesis, but also by the oxidative damage that these factors can cause. Furthermore, NBT staining of superoxide in the mycelium indicated altered levels of ROS accumulation, both under standard conditions and in the presence of high oxidative load. Osmotic stress caused increased ROS accumulation in the Δ
hex1 mycelia and this could possibly explain the significant developmental defects in these conditions. On the other hand, in the presence of farnesol, Δ
hex1 exhibited lower ROS generation. These findings open up the possibility that Hex1 plays a role in ROS metabolism and resistance to oxidative stress in
V. dahliae. Considering the significance of intracellular ROS in signaling and regulatory functions [
60], such a connection with Hex1 might be worth investigating further. Furthermore, it possibly becomes especially relevant in pathogenic fungi, which need to cope with host responses to infection that often involve bursts of ROS generation [
48,
49]. In the pathogenic fungi
Alternaria brassicicola and
A. fumigatus, WBs were shown to be associated with the redox homeostasis-related transmembrane protein TmpL [
61], which could be consistent with a function of Hex1 in ROS metabolism. To our knowledge, we report here the first experimental data that suggest involvement of Hex1 in ROS homeostasis.
Considering the pleiotropic and significant effects that Hex1 deletion has on fungal growth and development, one could reasonably further assume a possibly important role in pathogenicity. This has been investigated in several plant and other pathogens, and results between species are contradicting, demonstrating partial attenuation of virulence to varying degrees in some cases [
13,
18,
22,
25], but no compromise in pathogenic capacity in others [
23,
26]. We report here, for
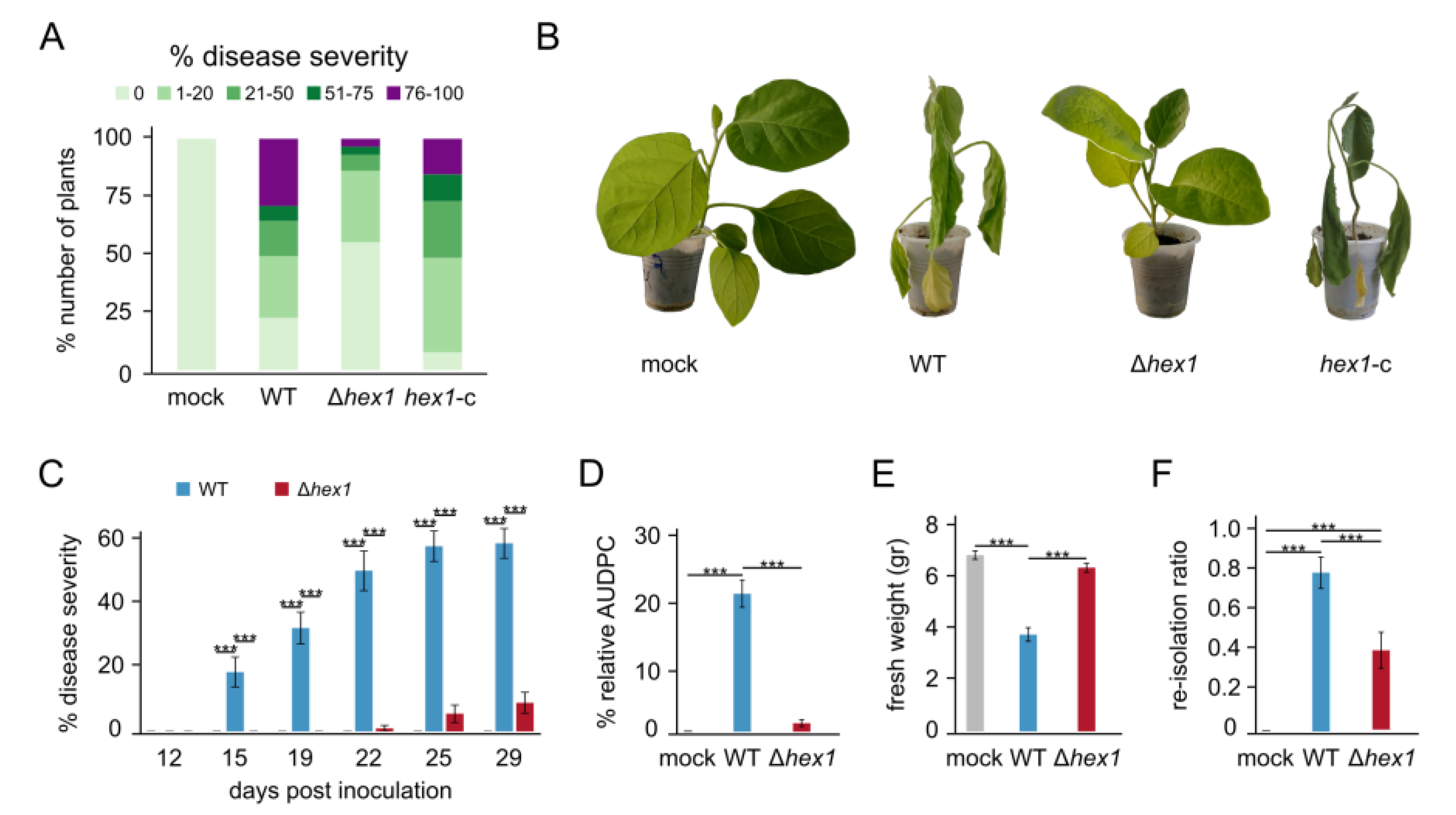
V. dahliae, one of the most pronounced roles of Hex1 in fungal pathogenicity. Deletion of
hex1 diminished virulence of
V. dahliae on eggplant, a very susceptible host of this pathogen [
38]. Consistently with this drastically reduced disease severity (by 84.7% on average), presence of the fungus in the xylem of infected plants was also significantly reduced (by 49.4%). These results suggest that Hex1 is required not only for penetration of roots by
V. dahliae, but also for its propagation in the host and its ability to cause systemic infection. It is reasonable to assume that this drastic reduction of virulence could be linked to the pleiotropic defects of Δ
hex1 in
V. dahliae growth, physiology, and stress response, rather than to solely attribute the attenuated virulence to an independent role of Hex1 in pathogenicity. The infection cycle of
V. dahliae begins with germination of microsclerotia, elongation of hyphae, and penetration of roots. In this endeavor, hyphal integrity is indispensable for efficient attack to the plant [
62]. We demonstrated that deletion of Hex1 negatively affected germination and hyphal growth, as well as cell wall integrity and resistance to stress conditions. Furthermore, propagation of the fungus in plant tissues, mostly within xylem vessels, is required in subsequent stages of the plant disease, which involves sequential conidiation, germination, and hyphal growth cycles [
63]. However, we found that the Hex1-deficient mutant has a diminished ability to produce conidia, which would probably challenge its ability to efficiently colonize the infected plant [
64]. Finally, it is possible that the lack of Hex1 and functional WBs might negatively interfere with the ability of the fungus to withstand the stressful adverse conditions induced in the xylem by the plant’s defense mechanisms. For example, one of the host responses to fungal invasion involves rapid bursts of ROS [
48]. Since Δ
hex1 cells are defective in their responses to oxidative stress, this could be an additional factor that compromises its pathogenic potential.
Establishment of viable heterokaryons in fungi, resulting from hyphal fusion between different individuals, is often prevented in natural populations by incompatibility mechanisms. One of them involves the triggering of a cell death reaction if the fused cells are genetically incompatible, resulting in intense vacuolization, nuclear degradation, cell wall shrinkage, and, finally, cell death [
7,
33]. However, this reaction is highly localized and only affects the fused cells, whereas the adjacent hyphal compartments remain unaffected [
7]. We hypothesized that the mechanism that is responsible for sealing of the incompatible fused cells to prevent the diffusion of cell death mediators to neighboring cells might involve the WBs, in a function analogous to septal pore plugging upon mechanical damage. However, our time-lapse imaging experiments of incompatible fusions clearly demonstrated that this compartmentalization function is independent of Hex1, as it normally occurs in Δ
hex1 hyphae. This is similar to what was previously observed in
N. crassa [
65]. In the course of our experiments, we invariably observed the fusion-induced thickening of septae defining the boundaries of the fused incompatible cells, presumably by intense chitin deposition. This was also detected, to a lesser extent, in the cell wall of these cells, but never affected any of the adjacent hyphal compartments. Similar observations were previously made in
Podospora anserina [
66]. We propose that the reinforcement of the septae of incompatible cells is involved in their sealing mechanism, which aims at preventing the incompatibility reaction from spreading in the hyphae, as well as protecting the neighboring cells once their fused neighbor gets disorganized as the result of the incompatibility reaction.
In conclusion, we demonstrate here important and pleiotropic roles of the protein Hex1 in the plant pathogenic fungus V. dahliae. Apart from its function in hyphal compartmentalization in response to hyphal damage, involvement of the protein was identified in fundamental biological processes related to fungal growth, physiology, asexual reproduction, stress response, and pathogenicity. Although Hex1 is highly conserved in ascomycetes, diverse functions have been attributed to its homologs in different species, which underlines the necessity of further functional investigations to fully elucidate its multifaceted role in fungal biology.