First Analysis of Human Coccidioides Isolates from New Mexico and the Southwest Four Corners Region: Implications for the Distributions of C. posadasii and C. immitis and Human Groups at Risk
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection and Patient Information
2.2. Molecular Methods
2.3. Phylogenetic Analysis
2.4. Ethics Statement
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nguyen, C.; Barker, B.M.; Hoover, S.; Nix, D.E.; Ampel, N.M.; Frelinger, J.A.; Orbach, M.J.; Galgiani, J.N. Recent advances in our understanding of the environmental, epidemiological, immunological, and clinical dimensions of coccidioidomycosis. Clin. Microbiol. Rev. 2013, 26, 505–525. [Google Scholar] [CrossRef] [PubMed]
- Pappagianis, D. Epidemiology of coccidioidomycosis. In Coccidioidomycosis. Current Topics in Infectious Disease; Stevens, D.A., Ed.; Springer: Boston, MA, USA, 1980; pp. 63–85. [Google Scholar]
- Galgiani, J.; Ampel, N.M.; Blair, J.; Catanzaro, A. Coccidioidomycosis. Clin. Infect. Dis. 2005, 4, 1217–1223. [Google Scholar] [CrossRef] [PubMed]
- Hector, R.F.; Laniado-Laborin, R. Coccidioidomycosis: A fungal disease of the Americas. PLoS Med. 2005, 2, e2. [Google Scholar] [CrossRef] [PubMed]
- Baptista-Rosas, R.C.; Hinojosa, A.; Riquelme, M. Ecological niche modeling of Coccidioides spp. in Western North American deserts. Ann. N. Y. Acad. Sci. 2007, 1111, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Fisher, M.C.; Koenig, G.L.; White, T.J.; Taylor, J.W. Molecular and phenotypic description of Coccidioides posadasii sp. nov., previously recognized as the non-California population of Coccidioides immitis. Mycologia 2002, 94, 73–84. [Google Scholar] [CrossRef]
- Barker, B.M.; Jewell, K.A.; Kroken, S.; Orbach, M.J. The population biology of Coccidioides: Epidemiologic implications for disease outbreaks. Ann. N. Y. Acad. Sci. 2007, 1111, 147–163. [Google Scholar] [CrossRef] [PubMed]
- Edwards, P.Q.; Palmer, C.E. Prevalence of sensitivity to coccidioidin, with special reference to specific and nonspecific reactions to coccidioidin and to histoplasmin. Dis. Chest 1957, 31, 35–60. [Google Scholar] [CrossRef]
- Ampel, N.M.; Mosley, D.G.; England, B.; Vertz, P.D.; Komatsu, K.; Hajjeh, R.A. Coccidioidomycosis in Arizona: Increase in incidence from 1990 to 1995. Clin. Infect. Dis. 1998, 27, 1528–1530. [Google Scholar] [CrossRef]
- Petersen, L.R.; Marshall, S.L.; Barton–Dickson, C.; Hajjeh, R.A.; Lindsley, M.D.; Warnock, D.W.; Panackal, A.A.; Shaffer, J.B.; Haddad, M.B.; Fisher, F.S.; et al. Coccidioidomycosis among workers at an archaeological site, northeastern Utah. Emerg. Infect. Dis. 2004, 10, 637–642. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Coccidioidomycosis—United States, 1991–1992. Morb. Mortal Wkly. Rep. 2011, 62, 217–221. [Google Scholar]
- Kirkland, T.N.; Fierer, J. Coccidioidomycosis: A reemerging infectious disease. Emerg. Infect. Dis. 1996, 3, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Litvintseva, A.P.; Marsden-Haug, N.; Hurst, S.; Hill, H.; Gade, L.; Driebe, E.M.; Ralston, C.; Roe, C.; Barker, B.M.; Goldoft, M.; et al. Valley fever: Finding new places for an old disease: Coccidioides immitis found in Washington State soil associated with recent human infection. Clin. Infect. Dis. 2014, 60, e1–e3. [Google Scholar] [CrossRef] [PubMed]
- Fisher, M.C.; Rannala, B.; Chaturvedi, V.; Taylor, J.W. Disease surveillance in recombining pathogens: Multilocus genotypes identify sources of human Coccidioides infections. Proc. Natl. Acad. Sci. USA 2002, 99, 9067–9071. [Google Scholar] [CrossRef] [PubMed]
- Engelthaler, D.M.; Roe, C.C.; Hepp, C.M.; Teixeira, M.; Driebe, E.M.; Schupp, J.M.; Gade, L.; Waddell, V.; Komatsu, K.; Arathoon, E.; et al. Local population structure and patterns of western hemisphere dispersal for Coccidioides spp., the fungal cause of valley fever. mBio 2016, 7, e00550-16. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.; Benedict, K.; Park, B.J.; Thompson, G.R., III. Coccidioidomycosis: Epidemiology. Clin. Epidemiol. 2013, 5, 185–197. [Google Scholar] [PubMed]
- Louie, L.; Ng, S.; Hajjeh, R.; Johnson, R.; Vugia, D.; Werner, S.B.; Talbot, R.; Klitz, W. Influence of host genetics on the severity of coccidioidomycosis. Emerg. Infect. Dis. 1999, 5, 672–680. [Google Scholar] [CrossRef] [PubMed]
- Pappagianis, D. Epidemiology of coccidioidomycosis. Curr. Top. Med. Mycol. 1988, 2, 199–238. [Google Scholar] [PubMed]
- Koufopanou, V.; Burt, A.; Taylor, J.W. Concordance of gene genealogies reveals reproductive isolation in the pathogenic fungus Coccidioides immitis. Proc. Natl. Acad. Sci. USA 1997, 94, 5478–5482. [Google Scholar] [CrossRef] [PubMed]
- Platt, A.R.; Woodhall, R.W.; George, A.L., Jr. Improved DNA sequencing quality and efficiency using an optimized fast cycle sequencing protocol. BioTechniques 2007, 43, 58–62. [Google Scholar] [CrossRef] [PubMed]
- Sievers, F.; Wilm, A.; Dineen, D.G.; Gibson, T.J.; Karplus, K.; Li, W.; Lopez, R.; McWilliam, H.; Remmert, M.; Söding, J.; et al. Fast, scalable generation of high-quality protein multiple sequence alignments using Clustal Omega. Mol. Syst. Biol. 2011, 7, 539. [Google Scholar] [CrossRef] [PubMed]
- Felsenstein, J. PHYLIP—Phylogeny Inference Package (Version 3.2). Cladistics 1989, 5, 164–166. [Google Scholar]
- Shriber, J.; Conlon, K.C.; Benedict, K.; McCotter, O.Z.; Bell, J.E. Assessment of vulnerability to coccidioidomycosis in Arizona and California. Int. J. Environ. Res. Public Health 2017, 14, 680. [Google Scholar] [CrossRef] [PubMed]
- Gumprecht, B. Beware the spore: Fungus in New Mexico soils causes serious illness. Las Cruces Sun-News. 12 March 2019. Available online: https://www.lcsun-news.com/story/life/wellness/2019/03/11/soil-fungus-serious-illness-new-mexico/2711787002/ (accessed on 26 July 2019).
- US Census Bureau Quick Facts: New Mexico. Available online: https://www.census.gov/quickfacts/NM (accessed on 28 Dec 2018).
- Centers for Disease Control and Prevention (CDC). Summary of Notifiable Infectious Diseases and Conditions—United States, 2014. Morb. Mortal Wkly. Rep. 2016, 63, 1–152. [Google Scholar] [CrossRef] [PubMed]
- Sievers, M.L. Coccidioidomycosis among Southwestern American Indians. Am. Rev. Respir. Dis. 1964, 90, 920–926. [Google Scholar] [PubMed]
- Sievers, M.L.; Nelson, R.G.; Knowler, W.C.; Bennett, P.H. Impact of NIDDM on mortality and causes of death in Pima Indians. Diabetes Care 1992, 15, 1541–1549. [Google Scholar] [CrossRef] [PubMed]
- Park, B.J.; Sigel, K.; Vaz, V.; Komatsu, K.; McRill, C.; Phelan, M.; Colman, T.; Comrie, A.C.; Warnock, D.W.; Galgiani, J.N.; et al. An epidemic of coccidioidomycosis in Arizona associated with climatic changes, 1998–2001. J. Infect. Dis. 2005, 191, 1981–1987. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.M.; Carlson, E.L.; Fisher, F.S.; Pappagianis, D. Demonstration of Coccidioides immitis and Coccidioides posadasii DNA in soil samples collected from Dinosaur National Monument, Utah. Med. Mycol. 2014, 52, 610–617. [Google Scholar] [CrossRef]
- Neafsey, D.E.; Barker, B.M.; Sharpton, T.J.; Stajich, J.E.; Park, D.J.; Whiston, E.; Hung, C.-Y.; McMahan, C.; White, J.; Sykes, S.; et al. Population genomic sequencing of Coccidioides fungi reveals recent hybridization and transposon control. Genome Res. 2010, 20, 938–946. [Google Scholar] [CrossRef]
- Teixeira, M.M.; Barker, B.M. Use of population genetics to assess the ecology, evolution, and population structure of Coccidioides. Emerg. Infect. Dis. 2016, 22, 1022–1030. [Google Scholar] [CrossRef]
- Barker, B.M.; Tabor, J.A.; Shubitz, L.F.; Perrill, R.; Orbach, M.J. Detection and phylogenetic analysis of Coccidioides posadasii in Arizona soil samples. Fungal Ecol. 2012, 5, 163–176. [Google Scholar] [CrossRef]
- Taylor, J.W.; Barker, B.M. The endozoan, small-mammal reservoir hypothesis and the life cycle of Coccidioides species. Med. Mycol. 2019, 57, S16–S20. [Google Scholar] [CrossRef] [PubMed]
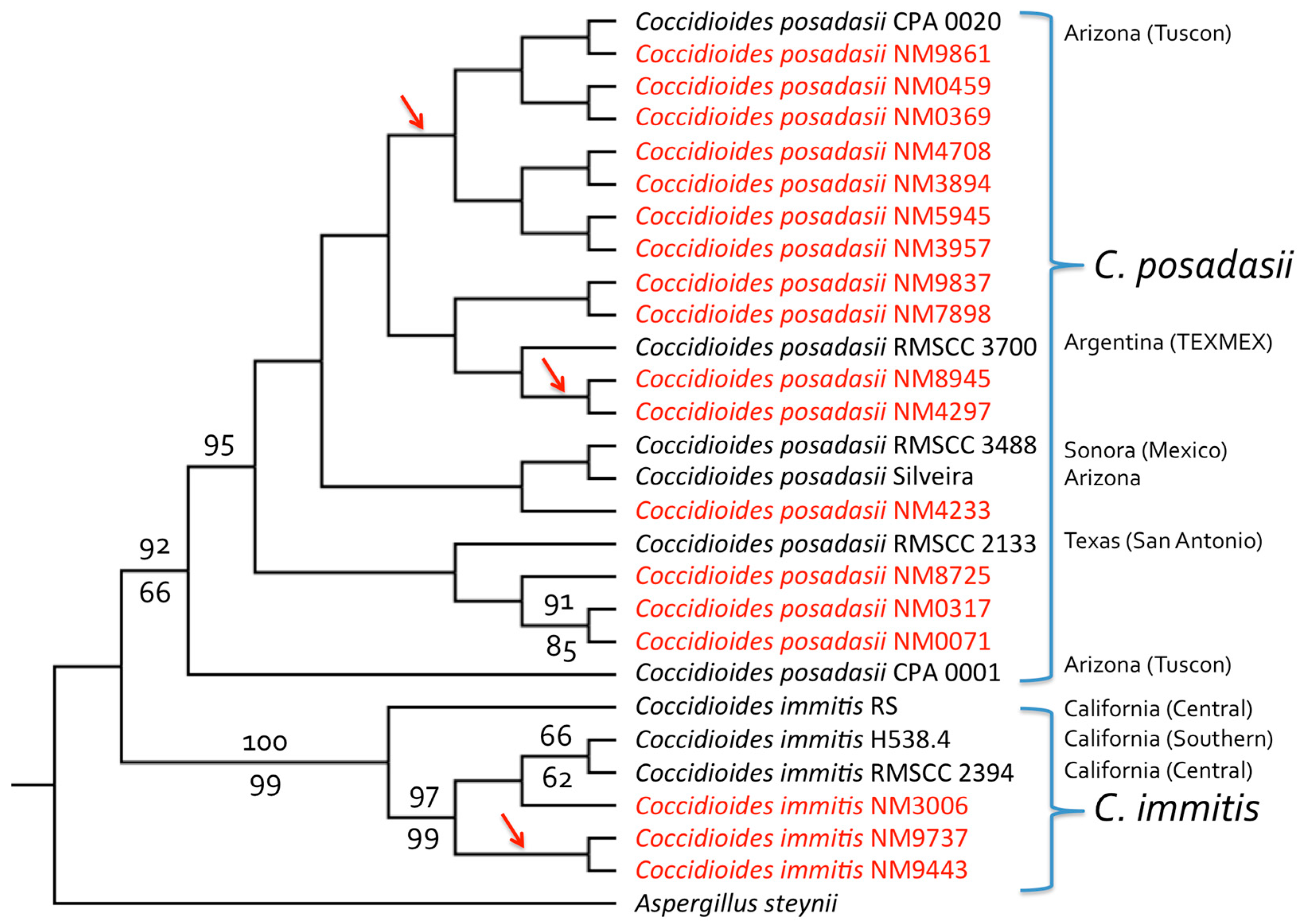
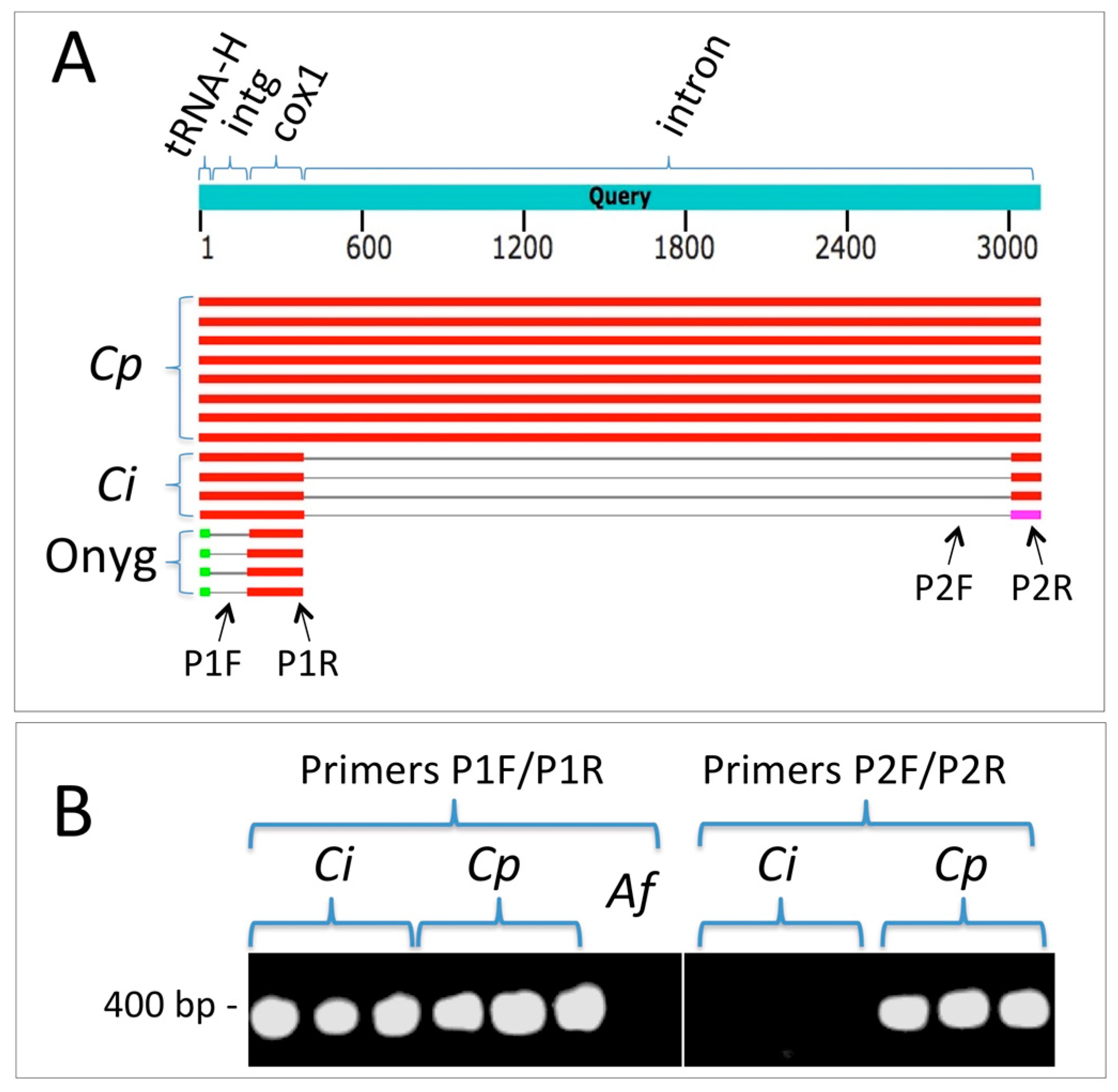
| Isolate | Isolate ID a | County or State of Residency | Source Type of Infection | Age | Sex | Race/Ethnicity | Significant Past Medical History | Occupation | Travel History |
|---|---|---|---|---|---|---|---|---|---|
| C. immitis Isolates | |||||||||
| NM3006 | Ci | San Juan, NM | Bone Vertebral osteomyelitis | 60 | M | American Indian | Yes | Railroad employee | None |
| NM9443 b | Ci | Utah | Ear | 51 | M | Unk c | Unk | Unk | Unk |
| NM9737 b | Ci | Utah | Ear | 51 | M | Unk | Unk | Unk | Unk |
| C. posadasii Isolates | |||||||||
| NM0369 | Cp | Bernalillo, NM | Bronchial wash Pulmonary infection | 66 | M | Unk | Yes | Unk | None |
| NM4233 | Cp | Bernalillo, NM | Tissue Pulmonary infection | 48 | M | Unk | Yes | Rug merchant | Tucson, AZ, 3 wks prior |
| NM9861 | Cp | Bernalillo, NM | Tissue Pulmonary infection | 30 | F | Unk | Yes | Unk | None |
| NM3894 | Cp | Bernalillo, NM | Fluid Knee infection | 77 | M | Unk | No | None | None |
| NM0317 | Cp | Chaves, NM | Ear Pulmonary infection and facial lesion | 62 | M | African American | Yes | Retired. Prior military in CA | None |
| NM4297 | Cp | Eddy, NM | Sputum Pulmonary infection | 63 | M | White | Yes | Mining engineer | Travel to Kansas |
| NM0071 | Cp | Lea, NM | Nasal sputum Pulmonary infection | 28 | M | African American | Yes | Incarcerated patient | N/A |
| NM3957 | Cp | McKinley, NM | Pleural fusion Pulmonary infection | 42 | M | Unk | Unk | Unk | Unk |
| NM9837 | Cp | McKinley, NM | Nasal sputum Pulmonary infection | 51 | M | American Indian | Yes | Welder | Recent work Phoenix, AZ |
| NM5945 | Cp | McKinley, NM | Tissue Pulmonary infection | 48 | F | American Indian | Yes | Unk | None |
| NM4708 | Cp | San Juan, NM | Bronchial wash Pulmonary infection | 40 | F | American Indian | Yes | Unk | None |
| NM0459 | Cp | Socorro, NM | Tissue Pulmonary infection | 50 | F | Unk | Unk | Unk | None |
| NM7898 | Cp | Torrance, NM | Bronchial lavage Pulmonary infection | 48 | M | Unk | Unk | Incarcerated patient | None |
| NM8725 | Cp | New Mexico | Fluid/Unk | Unk | Unk | Unk | Unk | Unk | Unk |
| NM8945 | Cp | Arizona | Bone Pulmonary infection in childhood; osteomyelitis | 62 | M | American Indian | Yes | Unk | None |
| DNA Region | Direction | Sequence | Annealing Temp. |
|---|---|---|---|
| ITS | Forward | 5′ CTTGGTCATTTAGAGGAAGTAA 3′ | 50 °C |
| Reverse | 5′ TCCTCCGCTTATTGATATGC 3′ | ||
| Serine Proteinase | Forward | 5′ ATAGAGACCACGCAGAAGGC 3′ | 55 °C |
| Reverse | 5′ AGCTGTCACGGATGGTATCG 3′ | ||
| MCM7 | Forward | 5′ TGGTTATAGCGCGATTCTCC 3′ | 50 °C |
| Reverse | 5′ CGAGTCGTTATACCTCGAACG 3′ | ||
| RPB1 | Forward | 5′ CCGCGTCATTATTTAAGCATC 3′ | 55 °C |
| Reverse | 5′ AGCGTATTCACCAACTTCTC 3′ | ||
| C. posadasii Intron a | Forward (P2F) | 5′ TCAAATCATGTGTAATATGTGG 3′ | 50 °C |
| Reverse (P2R) | 5′ GTTGACCATAAAGAAAAGTTGG 3′ | ||
| cox1 exon b | Forward (P1F) | 5′ ATAAAATAAACTACGATTTGCG 3′ | 50 °C |
| Reverse (P1R) | 5′ GATTGCATGAGCTGTAATAATAC 3′ |
| Isolate | ITS | Serine Proteinase | MCM7 | RPB1 |
|---|---|---|---|---|
| Sequences from New Mexico and Four Corners Region Coccidioides Isolates (This Study) | ||||
| NM3006 | MH725248 | MH748764 | MH748742 | MH748724 |
| NM9443 | MH725258 | MH748774 | MH748757 | MH748739 |
| NM9737 | MH725259 | MH748775 | MH748758 | MH748740 |
| NM0071 | MH725244 | MH748760 | MH748749 | MH748731 |
| NM0317 | MH725245 | MH748761 | MH748748 | MH748730 |
| NM0369 | MH725246 | MH748762 | MH748759 | MH748741 |
| NM0459 | MH725247 | MH748763 | MH748746 | MH748728 |
| NM3894 | MH725249 | MH748765 | MH748754 | MH748736 |
| NM3957 | MH725250 | MH748766 | MH748756 | MH748738 |
| NM4233 | MH725251 | MH748767 | MH748743 | MH748725 |
| NM4297 | MH725252 | MH748768 | MH748752 | MH748734 |
| NM4708 | MH725253 | MH748769 | MH748744 | MH748726 |
| NM5945 | MH725254 | MH748770 | MH748751 | MH748733 |
| NM7898 | MH725255 | MH748771 | MH748747 | MH748729 |
| NM8725 | MH725256 | MH748772 | MH748755 | MH748737 |
| NM8945 | MH725257 | MH748773 | MH748750 | MH748732 |
| NM9837 | MH725260 | MH748776 | MH748745 | MH748727 |
| NM9861 | MH725261 | MH748777 | MH748753 | MH748735 |
| Sequences from Existing GenBank Coccidioides Entries | ||||
| RMSCC 2394 | AATX01000513.1 | AATX01000326.1 | AATX01000203.1 | AATX01000264.1 |
| 224–846 | 35438–36110 | 43422–44141 | 433820–434247 | |
| H538.4 | AASO01000085.1 | AASO01002025.1 | AASO01002210.1 | AASO01003054.1 |
| 10339–10766 | 35115–35787 | 4641–5360 | 10945–11568 | |
| RS | AAEC03000009.1 | AAEC03000008.1 | AAEC03000005.1 | AAEC03000010.1 |
| 4725–5351 | 2537048–2537720 | 3687326–3688045 | 828825–829252 | |
| CPA 0020 | ABIV01003320.1 | ABIV01000896.1 | ABIV01000569.1 | ABIV01001762.1 |
| 16973–17598 | 3088–3760 | 311–1030 | 5431–5860 | |
| CPA 0001 | ABFO01003353.1 | ABFO01000797.1 | ABFO01001238.1 | ABFO01003988.1 |
| 1–346 a | ||||
| 2878–3503 | 665–1337 | ABFO01001237.1 | 6129–6558 | |
| 12376–12757 a | ||||
| RMSCC 3700 | ABFN01001891.1 | ABFN01000336.1 | ABFN01000290.1 | ABFN01001137.1 |
| 3121–3746 | 1–638 | 5242–5961 | 412–841 | |
| RMSCC 2133 | ABFM01000924.1 | ABFM01000717.1 | ABFM01000464.1 | ABFM01000297.1 |
| 22927–23552 | 3481–4153 | 1904–1262 | 6563–6992 | |
| RMSCC 3488 | ABBB01000249.1 | ABBB01000240.1 | ABBB01000156.1 | ABBB01000255.1 |
| 33179–33804 | 371029–371701 | 145000–145719 | 527482–527911 | |
| Silveira | KM588216.1 | ABAI02000152.1 | ABAI02000361.1 | ABAI02000102.1 |
| 34354–35026 | 12054–12773 | 54472–54901 | ||
| Sequences from Existing GenBank Aspergillus steynii Entries | ||||
| IBT 23096 | MSFO01000033.1 | MSFO01000001.1 | MSFO01000005.1 | MSFO01000005.1 |
| 3264–2712 | 3181742–3182796 | 1831768–1834777 | 2491035–2491482 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hamm, P.S.; Hutchison, M.I.; Leonard, P.; Melman, S.; Natvig, D.O. First Analysis of Human Coccidioides Isolates from New Mexico and the Southwest Four Corners Region: Implications for the Distributions of C. posadasii and C. immitis and Human Groups at Risk. J. Fungi 2019, 5, 74. https://doi.org/10.3390/jof5030074
Hamm PS, Hutchison MI, Leonard P, Melman S, Natvig DO. First Analysis of Human Coccidioides Isolates from New Mexico and the Southwest Four Corners Region: Implications for the Distributions of C. posadasii and C. immitis and Human Groups at Risk. Journal of Fungi. 2019; 5(3):74. https://doi.org/10.3390/jof5030074
Chicago/Turabian StyleHamm, Paris S., Miriam I. Hutchison, Pascale Leonard, Sandra Melman, and Donald O. Natvig. 2019. "First Analysis of Human Coccidioides Isolates from New Mexico and the Southwest Four Corners Region: Implications for the Distributions of C. posadasii and C. immitis and Human Groups at Risk" Journal of Fungi 5, no. 3: 74. https://doi.org/10.3390/jof5030074
APA StyleHamm, P. S., Hutchison, M. I., Leonard, P., Melman, S., & Natvig, D. O. (2019). First Analysis of Human Coccidioides Isolates from New Mexico and the Southwest Four Corners Region: Implications for the Distributions of C. posadasii and C. immitis and Human Groups at Risk. Journal of Fungi, 5(3), 74. https://doi.org/10.3390/jof5030074








