Lipid Biosynthesis as an Antifungal Target
Abstract
1. Introduction
2. Classification and Function of Lipids
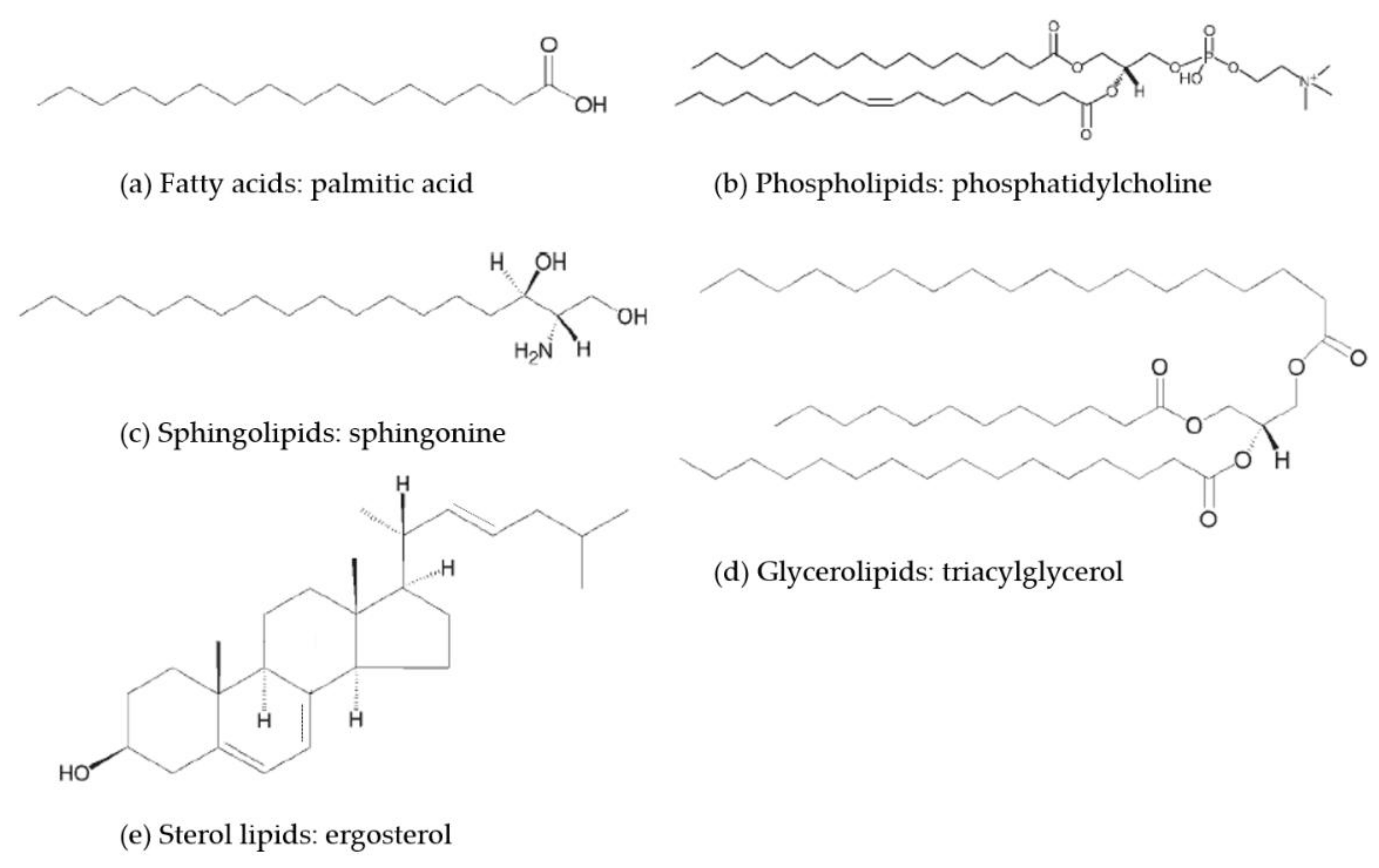
2.1. Classification of Lipids
2.2. Function of Lipids
3. Specific Lipid Metabolism in Fungi
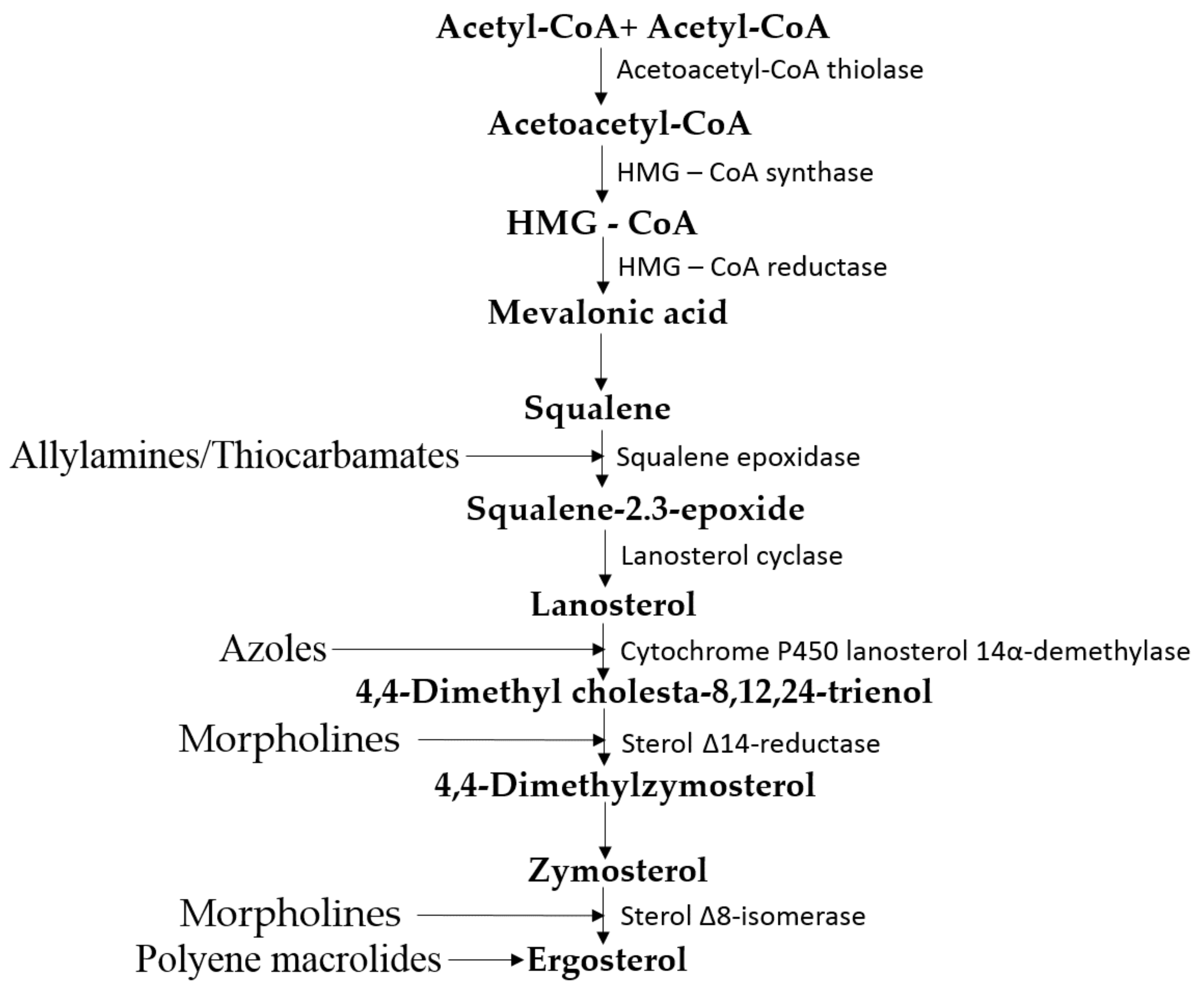
3.1. Metabolism of Ergosterol in Fungi
3.2. Metabolism of Phospholipids in Fungi
3.3. Metabolism of Sphingolipids in Fungi
3.4. Metabolism of Fatty Acids in Fungi
3.5. Metabolism of Neutral Lipids in Fungi
4. Searching for Potential Antifungal Drug Targets
5. Conclusions and Outlook
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Basha, A.; Basha, F.; Ali, S.K.; Hanson, P.R.; Mitscher, L.A.; Oakley, B.R. Recent progress in the chemotherapy of human fungal diseases. Emphasis on 1,3-β-glucan synthase and chitin synthase inhibitors. Curr. Med. Chem. 2013, 20, 4859–4887. [Google Scholar] [CrossRef] [PubMed]
- Surarit, R.; Shepherd, M.G. The effects of azole and polyene antifungals on the plasma membrane enzymes of Candida albicans. J. Med. Vet. Mycol. 1987, 25, 403–413. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhou, H.; Luo, Y.; Ouyang, H.; Hu, H.; Jin, C. Glycosylphosphatidylinositol (GPI) anchor is required in Aspergillus fumigatus for morphogenesis and virulence. Mol. Microbiol. 2007, 64, 1014–1027. [Google Scholar] [CrossRef] [PubMed]
- Fahy, E.; Subramaniam, S.; Brown, H.A.; Glass, C.K.; Merrill, A.H.; Murphy, R.C.; Raetz, C.R.; Russell, D.W.; Seyama, Y.; Shaw, W. A comprehensive classification system for lipids. Eur. J. Lipid Sci. Technol. 2005, 107, 839–861. [Google Scholar] [CrossRef]
- Huang, C.; Freter, C. Lipid metabolism, apoptosis and cancer therapy. Int. J. Mol. Sci. 2015, 16, 924–949. [Google Scholar] [CrossRef] [PubMed]
- Van, M.G.; Voelker, D.R.; Feigenson, G.W. Membrane lipids: Where they are and how they behave. Nat. Rev. Mol. Cell Biol. 2008, 9, 112. [Google Scholar]
- Garbarino, J.; Sturley, S.L. Homoeostatic systems for sterols and other lipids. Biochem. Soc. Trans. 2005, 33, 1182–1185. [Google Scholar] [CrossRef] [PubMed]
- Rajakumari, S.; Grillitsch, K.; Daum, G. Synthesis and turnover of non-polar lipids in yeast. Prog. Lipid Res. 2008, 47, 157–171. [Google Scholar] [CrossRef] [PubMed]
- Klug, L.; Daum, G. Yeast lipid metabolism at a glance. FEMS Yeast Res. 2014, 14, 369–388. [Google Scholar] [CrossRef] [PubMed]
- Bao, J.X.; Su, Y.T.; Cheng, Y.P.; Zhang, H.J.; Xie, X.P.; Chang, Y.M. Vascular sphingolipids in physiological and pathological adaptation. Front. Biosci. 2016, 21, 1168–1186. [Google Scholar]
- Farnoud, A.M.; Toledo, A.M.; Konopka, J.B.; Poeta, M.D.; London, E. Chapter seven—Raft-like membrane domains in pathogenic microorganisms. Curr. Top. Membr. 2015, 75, 233–268. [Google Scholar] [PubMed]
- Furse, S.; Shearman, G.C. Do lipids shape the eukaryotic cell cycle? Mol. Cell Biol. Lipids 2017, 1863, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Furse, S. The physical influence of inositides—A disproportionate effect? J. Chem. Biol. 2015, 8, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Shimano, H. Sterol regulatory element-binding proteins (SREBPs): Transcriptional regulators of lipid synthetic genes. Prog. Lipid Res. 2001, 40, 439–452. [Google Scholar] [CrossRef]
- Duplus, E.; Glorian, M.; Forest, C. Fatty acid regulation of gene transcription. J. Biol. Chem. 2000, 275, 30749–30752. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Zhu, P. The mechanisms of the modulation of cellular functions by diacylglycerol. Prog. Biochem. Biophys. 1996, 23, 38–42. [Google Scholar]
- Incardona, J.P.; Eaton, S. Cholesterol in signal transduction. Curr. Opin. Cell Biol. 2000, 12, 193–203. [Google Scholar] [CrossRef]
- Vance, J.E.; Vance, D.E. Phospholipid biosynthesis in mammalian cells. Biochem. Cell Biol. 2004, 82, 113–128. [Google Scholar] [CrossRef] [PubMed]
- Heinisch, J.J. Baker’s yeast as a tool for the development of antifungal kinase inhibitors—Targeting protein Kinase C and the cell integrity pathway. Biochim. Biophys. Acta 2005, 1754, 171–182. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.H. Heterotrimeric G protein signaling and RGSs in Aspergillus nidulans. J. Microbiol. 2006, 44, 145–154. [Google Scholar] [PubMed]
- Borodzicz, S.; Czarzasta, K.; Kuch, M.; Cudnochjedrzejewska, A. Sphingolipids in cardiovascular diseases and metabolic disorders. Lipids Health Dis. 2015, 14, 55. [Google Scholar] [CrossRef] [PubMed]
- Polak, A. Melanin as a virulence factor in pathogenic fungi. Mycoses 1990, 33, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Pihet, M.; Vandeputte, P.; Tronchin, G.; Renier, G.; Saulnier, P.; Georgeault, S.; Mallet, R.; Chabasse, D.; Symoens, F.; Bouchara, J.P. Melanin is an essential component for the integrity of the cell wall of Aspergillus fumigatus conidia. BMC Microbiol. 2009, 9, 177. [Google Scholar] [CrossRef] [PubMed]
- Maertens, J.A.; Boogaerts, M.A. Fungal cell wall inhibitors: Emphasis on clinical aspects. Curr. Pharm. Des. 2000, 6, 225–239. [Google Scholar] [CrossRef] [PubMed]
- Ghannoum, M.A.; Rice, L.B. Antifungal agents: Mode of action, mechanisms of resistance, and correlation of these mechanisms with bacterial resistance. Clin. Microbiol. Rev. 1999, 12, 501–517. [Google Scholar] [PubMed]
- Zotchev, S.B. Polyene macrolide antibiotics and their applications in human therapy. Curr. Med. Chem. 2003, 10, 211–223. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, S.; Takahashi, N.; Ito, M.; Teshima, K.; Yamashita, T.; Michishita, Y.; Ohyagi, H.; Shida, S.; Nagao, T.; Fujishima, M. Safety and efficacy of low-dose liposomal amphotericin b as empirical antifungal therapy for patients with prolonged neutropenia. Int. J. Clin. Oncol. 2013, 18, 983–987. [Google Scholar] [CrossRef] [PubMed]
- Maertens, J.; Glasmacher, A.; Herbrecht, R.; Thiebaut, A.; Cordonnier, C.; Segal, B.H.; Killar, J.; Taylor, A.; Kartsonis, N.; Patterson, T.F. Multicenter, noncomparative study of caspofungin in combination with other antifungals as salvage therapy in adults with invasive aspergillosis. Cancer 2006, 107, 2888–2897. [Google Scholar] [CrossRef] [PubMed]
- Campitelli, M.; Zeineddine, N.; Samaha, G.; Maslak, S. Combination antifungal therapy: A review of current data. J. Clin. Med. Res. 2017, 9, 451–456. [Google Scholar] [CrossRef] [PubMed]
- Tobias, J.S.; Wrioley, P.F.M.; Shaw, E. Combination antifungal therapy for Cryptococcal meningitis. N. Engl. J. Med. 2013, 368, 1291–1302. [Google Scholar] [CrossRef][Green Version]
- Carman, G.M.; Han, G.S. Regulation of phospholipid synthesis in the yeast Saccharomyces cerevisiae. J. Biol. Chem. 1996, 271, 13293–13296. [Google Scholar] [CrossRef] [PubMed]
- Cassilly, C.D.; Reynolds, T.B. Ps, it’s complicated: The roles of phosphatidylserine and phosphatidylethanolamine in the pathogenesis of Candida albicans and other microbial pathogens. J. Fungi 2018, 4, 28. [Google Scholar] [CrossRef] [PubMed]
- Wolf, J.M.; Espadas, J.; Luquegarcia, J.; Reynolds, T.; Casadevall, A. Lipid biosynthetic genes affect Candida albicans extracellular vesicle morphology, cargo, and immunostimulatory properties. Eukaryot. Cell 2015, 14, 745–754. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.L.; Montedonico, A.E.; Kauffman, S.; Dunlap, J.R.; Menn, F.M.; Reynolds, T.B. Phosphatidylserine synthase and phosphatidylserine decarboxylase are essential for cell wall integrity and virulence in Candida albicans. Mol. Microbiol. 2010, 75, 1112–1132. [Google Scholar] [CrossRef] [PubMed]
- Cassilly, C.D.; Maddox, M.M.; Cherian, P.T.; Bowling, J.J.; Hamann, M.T.; Lee, R.E.; Reynolds, T.B. Sb-224289 antagonizes the antifungal mechanism of the marine depsipeptide papuamide A. PLoS ONE 2016, 11, e0154932. [Google Scholar] [CrossRef] [PubMed]
- Tao, L.; Gao, N.S.; Yu, J.H. The choC gene encoding a putative phospholipid methyltransferase is essential for growth and development in Aspergillus nidulans. Curr. Genet. 2010, 56, 283–296. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.; Houweling, M. Phosphatidylcholine and cell death. Biochim. Biophys. Acta 2002, 1585, 87–96. [Google Scholar]
- Nagiec, M.M.; Nagiec, E.E.; Baltisberger, J.A.; Wells, G.B.; Lester, R.L.; Dickson, R.C. Sphingolipid synthesis as a target for antifungal drugs. Complementation of the inositol phosphorylceramide synthase defect in a mutant strain of Saccharomyces cerevisiae by the AUR1 gene. J. Biol. Chem. 1997, 272, 9809–9817. [Google Scholar] [CrossRef] [PubMed]
- Mandala, S.M.; Thornton, R.A.; Milligan, J.; Rosenbach, M.; Garciacalvo, M.; Bull, H.G.; Harris, G.; Abruzzo, G.K.; Flattery, A.M.; Gill, C.J. Rustmicin, a potent antifungal agent, inhibits sphingolipid synthesis at inositol phosphoceramide synthase. J. Biol. Chem. 1998, 273, 14942–14949. [Google Scholar] [CrossRef] [PubMed]
- Mandala, S.M.; Thornton, R.A.; Rosenbach, M.; Milligan, J.; Garciacalvo, M.; Bull, H.G.; Kurtz, M.B. Khafrefungin, a novel inhibitor of sphingolipid synthesis. J. Biol. Chem. 1997, 272, 32709–32714. [Google Scholar] [CrossRef] [PubMed]
- Chayakulkeeree, M.; Rude, T.H.; Toffaletti, D.L.; Perfect, J.R. Fatty acid synthesis is essential for survival of Cryptococcus neoformans and a potential fungicidal target. Antimicrob. Agents Chemother. 2007, 51, 3537–3545. [Google Scholar] [CrossRef] [PubMed]
- Zweytick, D.; Leitner, E.; Kohlwein, S.D.; Yu, C.; Rothblatt, J.; Daum, G. Contribution of Are1p and Are2p to steryl ester synthesis in the yeast Saccharomyces cerevisiae. FEBS J. 2000, 267, 1075–1082. [Google Scholar] [CrossRef]
- Athenstaedt, K.; Daum, G. The life cycle of neutral lipids: Synthesis, storage and degradation. Cell. Mol. Life Sci. 2006, 63, 1355–1369. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Cen, K.; Lu, Y.; Zhang, S.; Shang, Y.; Wang, C. Nitrogen-starvation triggers cellular accumulation of triacylglycerol in Metarhizium robertsii. Fungal Biol. 2017. [Google Scholar] [CrossRef]
- Cecil, A.; Ohlsen, K.; Menzel, T.; François, P.; Schrenzel, J.; Fischer, A.; Dörries, K.; Selle, M.; Lalk, M.; Hantzschmann, J. Modelling antibiotic and cytotoxic isoquinoline effects in Staphylococcus aureus, Staphylococcus epidermidis and mammalian cells. Int. J. Med. Microbiol. 2015, 305, 96–109. [Google Scholar] [CrossRef] [PubMed]
- Cecil, A.; Rikanović, C.; Ohlsen, K.; Liang, C.; Bernhardt, J.; Oelschlaeger, T.A.; Gulder, T.; Bringmann, G.; Holzgrabe, U.; Unger, M. Modeling antibiotic and cytotoxic effects of the dimeric isoquinoline iq-143 on metabolism and its regulation in Staphylococcus aureus, Staphylococcus epidermidis and human cells. Genome Biol. 2011, 12, R24. [Google Scholar] [CrossRef] [PubMed]
- Shityakov, S.; Dandekar, T.; Förster, C. Gene expression profiles and protein-protein interaction network analysis in AIDS patients with HIV-associated encephalitis and dementia. HIV 2015, 7, 265–276. [Google Scholar] [CrossRef] [PubMed]
- Schrettl, M.; Beckmann, N.; Varga, J.; Heinekamp, T.; Jacobsen, I.D.; Jöchl, C.; Moussa, T.A.; Wang, S.; Gsaller, F.; Blatzer, M. HAPX-mediated adaption to iron starvation is crucial for virulence of Aspergillus fumigatus. PLoS Pathog. 2010, 6, e1001124. [Google Scholar] [CrossRef] [PubMed]
- Willger, S.D.; Puttikamonkul, S.; Kim, K.H.; Burritt, J.B.; Grahl, N.; Metzler, L.J.; Barbuch, R.; Bard, M.; Lawrence, C.B.; Cramer, R.A. A sterol-regulatory element binding protein is required for cell polarity, hypoxia adaptation, azole drug resistance, and virulence in Aspergillus fumigatus. PLoS Pathog. 2008, 4, e1000200. [Google Scholar] [CrossRef] [PubMed]
- Andrew, M.D.; Fedorova, N.D.; Jonathan, C.; Yan, Y.; Stanley, K.; Dan, C.; Omar, L.; Timothy, C.; Gustavo, G.; Darius, A.J. Sub-telomere directed gene expression during initiation of invasive aspergillosis. PLoS Pathog. 2008, 4, e1000154. [Google Scholar]
- De Gouvêa, P.F.; Soriani, F.M.; Malavazi, I.; Savoldi, M.; Goldman, M.H.; Loss, O.; Bignell, E.; Da, S.F.M.; Goldman, G.H. Functional characterization of the Aspergillus fumigatus PHO80 homologue. Fungal Genet. Biol. 2008, 45, 1135–1146. [Google Scholar] [CrossRef] [PubMed]
- Martin, K.; Mugdha, S.; Gupta, S.K.; Liang, C.; Jasmin, B.; Anna-Maria, D.; Zohar, M.; Hubertus, H.; Nir, O.; Sven, K. Systematic identification of anti-fungal drug targets by a metabolic network approach. Front. Mol. Biosci. 2016, 3, 22. [Google Scholar]
- Boyce, K.; Kretschmer, M.; Kronstad, J. The vtc4 gene influences polyphosphate storage, morphogenesis, and virulence in the maize pathogen Ustilago maydis. Eukaryot. Cell 2006, 5, 1399–1409. [Google Scholar] [CrossRef] [PubMed]
- Ikeh, M.A.C.; Kastora, S.L.; Day, A.M.; Herrero-De-Dios, C.M.; Tarrant, E.; Waldron, K.J.; Banks, A.P.; Bain, J.M.; Lydall, D.; Veal, E.A. PHO4 mediates phosphate acquisition in Candida albicans and is vital for stress resistance and metal homeostasis. Mol. Biol. Cell 2016, 27, 2784–2801. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Li, B.; Cen, K.; Lu, Y.; Zhang, S.; Wang, C. Diverse effect of phosphatidylcholine biosynthetic genes on phospholipid homeostasis, cell autophagy and fungal developments in Metarhizium robertsii. Environ. Microbiol. 2018, 20, 293–304. [Google Scholar] [CrossRef] [PubMed]
- Long, N.N.; Trofa, D.; Nosanchuk, J.D. Fatty acid synthase impacts the pathobiology of Candida parapsilosis in vitro and during mammalian infection. PLoS ONE 2009, 4, e8421. [Google Scholar]
- Selvaraju, K.; Rajakumar, S.; Nachiappan, V. Identification of a phospholipase B encoded by the lpl1 gene in Saccharomyces cerevisiae. Mol. Cell Biol. Lipids 2014, 1841, 1383–1392. [Google Scholar] [CrossRef] [PubMed]


| Cell Fraction | Mol % of Total Phospholipids | ||||||
|---|---|---|---|---|---|---|---|
| PC | PE | PI | PS | CL | PA | Others | |
| Homogenate | 51.0 | 25.0 | 11.4 | 5.1 | 3.7 | 1.1 | 2.7 |
| Plasma membrane | 11.3 | 24.6 | 27.2 | 32.2 | nd | 3.3 | 1.4 |
| Endoplasmic reticulum | 38.9 | 18.6 | 22.4 | 6.4 | 0.3 | 6.1 | 10.0 |
| Mitochondria | 33.4 | 22.7 | 20.6 | 3.3 | 7.2 | 1.7 | 10.1 |
| Peroxisomes | 39.8 | 17.4 | 22.0 | 2.5 | 2.7 | 6.1 | 10.5 |
| Number | Enzyme | Abbreviation |
|---|---|---|
| 1 | G-3-P acyltransferase | SCT1 |
| 2 | G-3-P acyltransferase | GPT2 |
| 3 | 1-acyl-DHAP reductase | AYR1 |
| 4 | Lyso-phospholipid acyltransferase | SLC1/ALE1 |
| 5 | CDP-DAG synthase | CDS1 |
| 6 | PI synthase | PIS1 |
| 7 | PGP synthase | PGS1 |
| 8 | PGP phosphatase | GEP4 |
| 9 | CL synthase | CRD1 |
| 10 | PS synthase | CHO1 |
| 11 | PS decarboxylase 1/2 | PSD1/PSD 2 |
| 12 | PE methylase | CHO2 |
| 13 | Phospholipids methylase | OPI3 |
| 14 | PA phosphatase | PAH1 |
| 15 | DAG kinase | DGK1 |
| 16 | Etn kinase | EKI1 |
| 17 | P-Etn cytidylyltransferase | ECT1 |
| 18 | Etn phosphotransferase | EPT1 |
| 19 | Choline kinase | CKI1 |
| 20 | P-cho cytidylyltransferase | PCT1 |
| 21 | Choline phosphotransferase | CPT1 |
| 22 | Phospholipase C | PLC |
| 23 | Phospholipase D | SPO14 |
| Gene | Annotation | Fungi Tested * | Reference |
|---|---|---|---|
| AFUA_4G13680 | Phosphatidylserine synthase | C. albicans | [33] |
| AFUA_2G15970 | Phosphatidylethanolamine N-methyltransferase | S. cersvisiae et al. | [55] |
| AFUA_1G09050 | Methylene-fatty-acyl-phospholipid synthase | A. nidulans | [35] |
| AFUA_3G04210 | Fatty acid synthase α subunit FasA | C. neoformans | [41] |
| AFUA_3G04220 | Fatty acid synthase β subunit FasB | C. neoformans C. parapsilosis | [41,56] |
| AFUA_5G01960 | Phosphate transporter Pho88 | A. nidulans A. fumigatus | [51] |
| AFUA_3G12320 | Lipase/Serine esterase | S. cerevisiae | [57] |
| AFUA_2G09040 | Vacuolar transporter chaperone (Vtc4) | Ustilago maydis Candida | [53,54] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pan, J.; Hu, C.; Yu, J.-H. Lipid Biosynthesis as an Antifungal Target. J. Fungi 2018, 4, 50. https://doi.org/10.3390/jof4020050
Pan J, Hu C, Yu J-H. Lipid Biosynthesis as an Antifungal Target. Journal of Fungi. 2018; 4(2):50. https://doi.org/10.3390/jof4020050
Chicago/Turabian StylePan, Jiao, Cuiting Hu, and Jae-Hyuk Yu. 2018. "Lipid Biosynthesis as an Antifungal Target" Journal of Fungi 4, no. 2: 50. https://doi.org/10.3390/jof4020050
APA StylePan, J., Hu, C., & Yu, J.-H. (2018). Lipid Biosynthesis as an Antifungal Target. Journal of Fungi, 4(2), 50. https://doi.org/10.3390/jof4020050






