Impact of S-Wave Amplitude in Right Precordial Leads on Improvement in Mitral Regurgitation following Cardiac Resynchronization Therapy
Abstract
1. Introduction
2. Results
2.1. Patient Characteristics
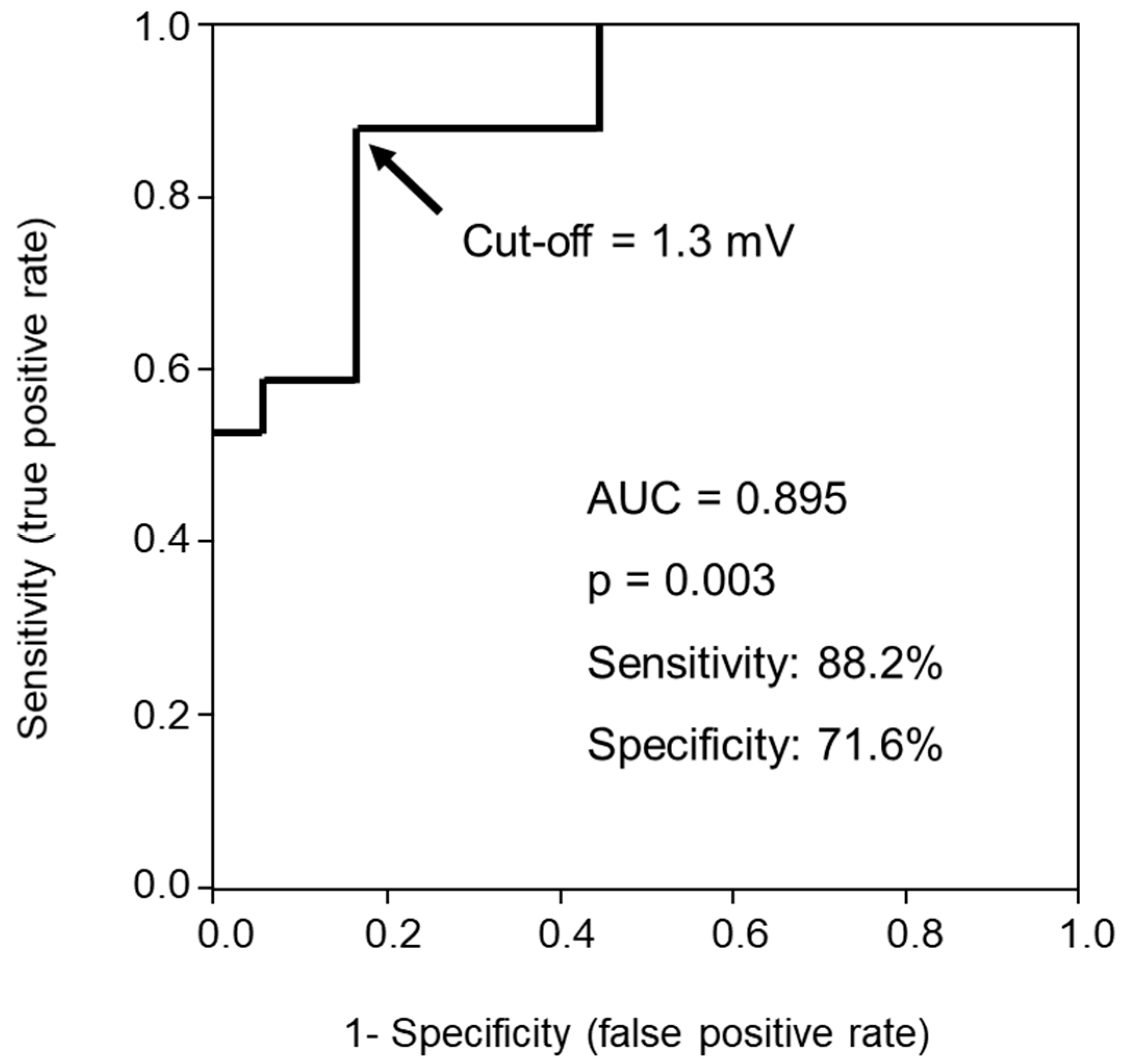
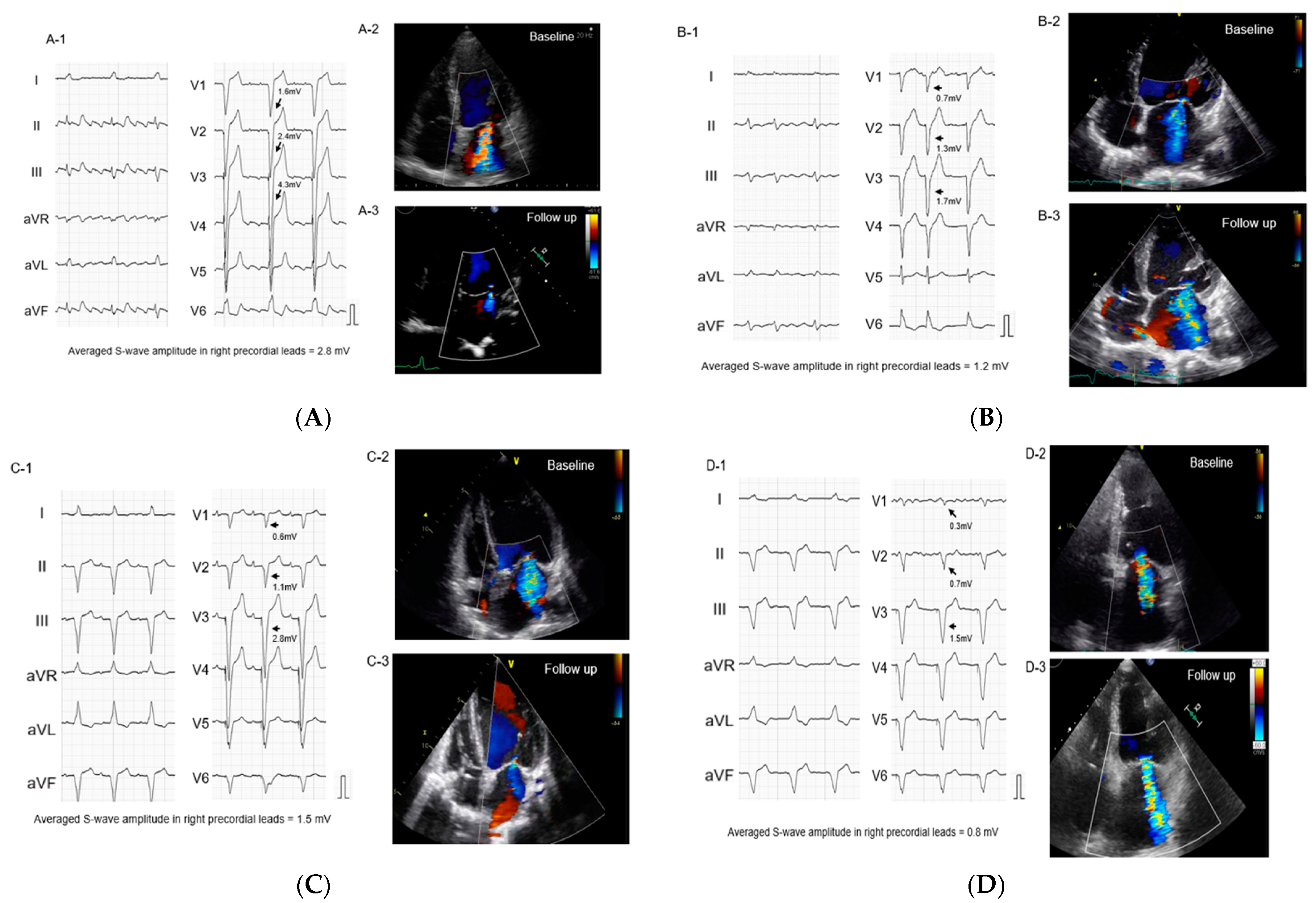
2.2. Impact of S-Wave Amplitude in Right Precordial Leads on MR Reduction
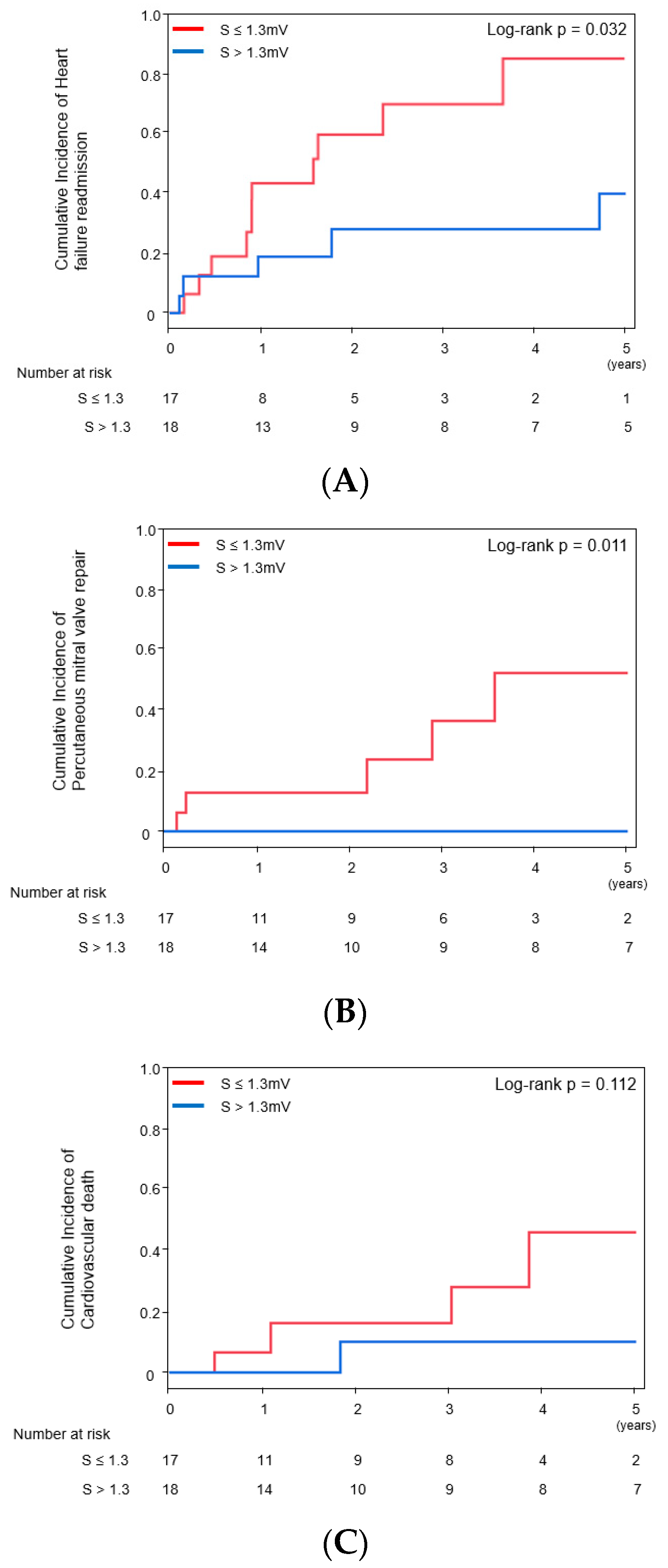
2.3. Impact of S-Wave Amplitude in Right Precordial Leads on Other Clinical Outcomes
3. Discussion
3.1. Implication of S-Wave Amplitude in Right Precordial Leads for Functional MR
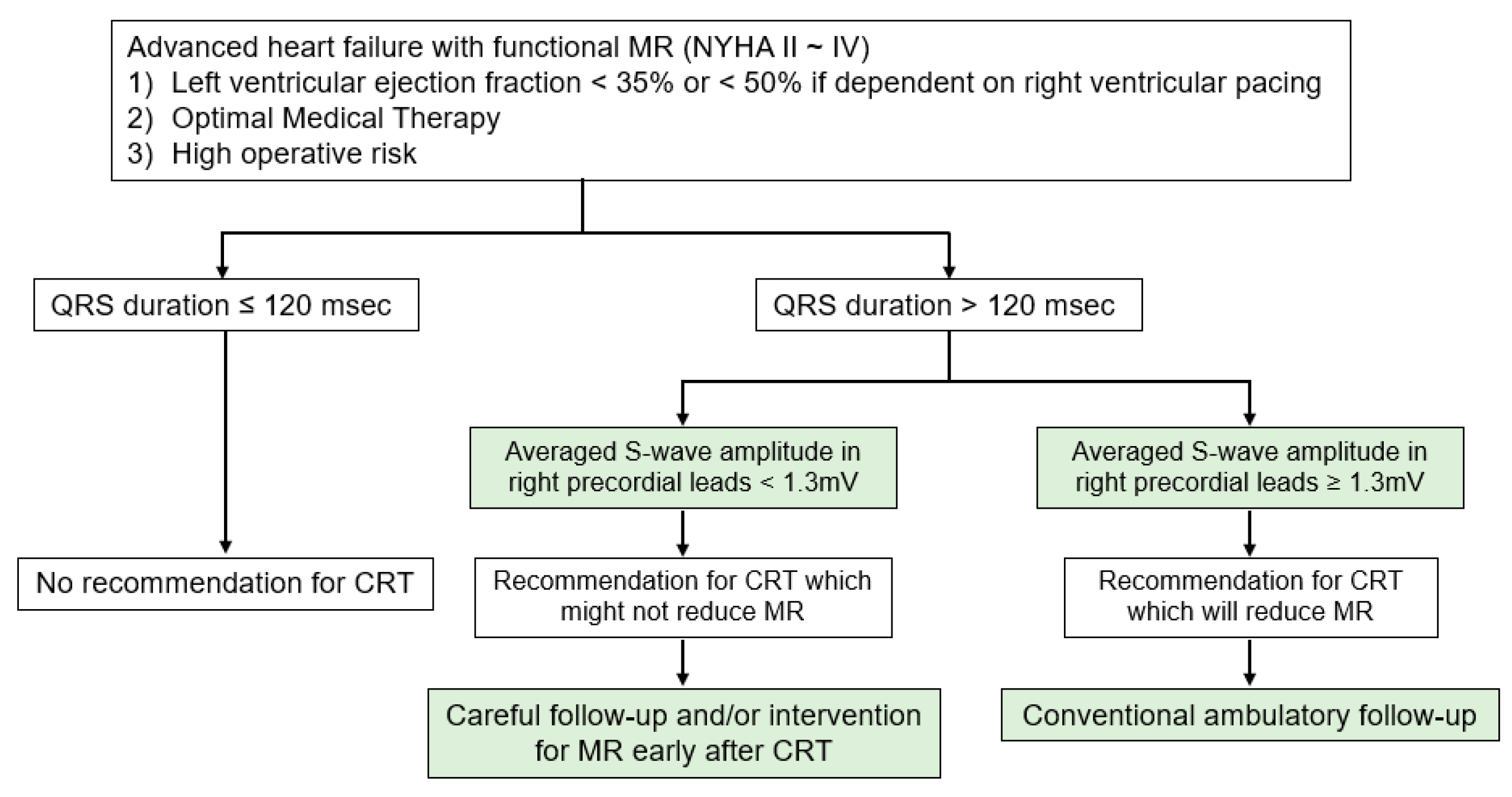
3.2. The Proposed Strategy
3.3. Study Limitations
4. Methods
4.1. Study Population
4.2. Implant Procedure
4.3. Baseline Clinical Characteristics
4.4. Standard 12-Lead Electrocardiograms
4.5. Echocardiograms and Assessment of MR
4.6. Clinical Outcomes
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CRT | cardiac resynchronization therapy |
| ECG | electrocardiogram |
| LBBB | left bundle branch block |
| LVEF | left ventricular ejection fraction |
| MR | mitral regurgitation |
| PMVR | percutaneous mitral valve repair |
| RBBB | right bundle branch block |
References
- Asgar, A.W.; Mack, M.J.; Stone, G.W. Secondary mitral regurgitation in heart failure: Pathophysiology, prognosis, and therapeutic considerations. J. Am. Coll. Cardiol. 2015, 65, 1231–1248. [Google Scholar] [CrossRef] [PubMed]
- Stone, G.W.; Lindenfeld, J.; Abraham, W.T.; Kar, S.; Lim, D.S.; Mishell, J.M.; Whisenant, B.; Grayburn, P.A.; Rinaldi, M.; Kapadia, S.R.; et al. Transcatheter Mitral-Valve Repair in Patients with Heart Failure. N. Engl. J. Med. 2018, 379, 2307–2318. [Google Scholar] [CrossRef] [PubMed]
- Van Bommel, R.J.; Marsan, N.A.; Delgado, V.; Borleffs, C.J.; van Rijnsoever, E.P.; Schalij, M.J.; Bax, J.J. Cardiac resynchronization therapy as a therapeutic option in patients with moderate-severe functional mitral regurgitation and high operative risk. Circulation 2011, 124, 912–919. [Google Scholar] [CrossRef] [PubMed]
- Yancy, C.W.; Jessup, M.; Bozkurt, B.; Butler, J.; Casey, D.E., Jr.; Drazner, M.H.; Fonarow, G.C.; Geraci, S.A.; Horwich, T.; Januzzi, J.L.; et al. American College of Cardiology Foundation. 2013 ACCF/AHA guideline for the management of heart failure: A report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines. J. Am. Coll. Cardiol. 2013, 62, e147–e239. [Google Scholar] [CrossRef]
- Ponikowski, P.; Voors, A.A.; Anker, S.D.; Bueno, H.; Cleland, J.G.F.; Coats, A.J.S.; Falk, V.; González-Juanatey, J.R.; Harjola, V.P.; Jankowska, E.A.; et al. ESC Scientific Document Group. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2016, 37, 2129–2200. [Google Scholar]
- Ezekowitz, J.A.; O’Meara, E.; McDonald, M.A.; Abrams, H.; Chan, M.; Ducharme, A.; Giannetti, N.; Grzeslo, A.; Hamilton, P.G.; Heckman, G.A.; et al. 2017 comprehensive update of the Canadian Cardiovascular Society guidelines for the management of heart failure. Can. J. Cardiol. 2017, 33, 1342–1433. [Google Scholar] [CrossRef]
- Ypenburg, C.; van Bommel, R.J.; Borleffs, C.J.; Bleeker, G.B.; Boersma, E.; Schalij, M.J.; Bax, J.J. Long-term prognosis after cardiac resynchronization therapy is related to the extent of left ventricular reverse remodeling at midterm follow-up. J. Am. Coll. Cardiol. 2009, 53, 483–490. [Google Scholar] [CrossRef]
- Onishi, T.; Onishi, T.; Marek, J.J.; Ahmed, M.; Haberman, S.C.; Oyenuga, O.; Adelstein, E.; Schwartzman, D.; Saba, S.; Gorcsan, J., 3rd. Mechanistic features associated with improvement in mitral regurgitation after cardiac resynchronization therapy and their relation to long-term patient outcome. Circ. Heart Fail. 2013, 6, 685–693. [Google Scholar] [CrossRef]
- Spartera, M.; Galderisi, M.; Mele, D.; Cameli, M.; D’Andrea, A.; Rossi, A.; Mondillo, S.; Novo, G.; Esposito, R.; D’Ascenzi, F.; et al. Echocardiographic Study Group of the Italian Society of Cardiology (SIC). Role of cardiac dyssynchrony and resynchronization therapy in functional mitral regurgitation. Eur. Heart J. Cardiovasc. Imaging 2016, 17, 471–480. [Google Scholar] [CrossRef]
- Erlebacher, J.A.; Barbarash, S. Intraventricular conduction delay and functional mitral regurgitation. Am. J. Cardiol. 2001, 88, 83–86. [Google Scholar] [CrossRef]
- Chatterjee, N.A.; Gold, M.R.; Waggoner, A.D.; Picard, M.H.; Stein, K.M.; Yu, Y.; Meyer, T.E.; Wold, N.; Ellenbogen, K.A.; Singh, J.P. Longer Left Ventricular Electric Delay Reduces Mitral Regurgitation After Cardiac Resynchronization Therapy: Mechanistic Insights From the SMART-AV Study (SmartDelay Determined AV Optimization: A Comparison to Other AV Delay Methods Used in Cardiac Resynchronization Therapy). Circ. Arrhythm. Electrophysiol. 2016, 9, e004346. [Google Scholar] [PubMed]
- Brenyo, A.; Rao, M.; Barsheshet, A.; Cannom, D.; Quesada, A.; McNitt, S.; Huang, D.T.; Moss, A.J.; Zareba, W. QRS axis and the benefit of cardiac resynchronization therapy in patients with mildly symptomatic heart failure enrolled in MADIT-CRT. J. Cardiovasc. Electrophysiol. 2013, 24, 442–448. [Google Scholar] [CrossRef] [PubMed]
- Perrotta, L.; Kandala, J.; Di Biase, L.; Valleggi, A.; Michelotti, F.; Pieragnoli, P.; Ricciardi, G.; Mascioli, G.; Lakkireddy, D.; Pillarisetti, J.; et al. Prognostic impact of QRS axis deviation in patients treated with cardiac resynchronization therapy. J. Cardiovasc. Electrophysiol. 2016, 27, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Fabiszak, T.; Łach, P.; Ratajczak, J.; Koziński, M.; Krupa, W.; Kubica, J. Influence of QRS duration and axis on response to cardiac resynchronization therapy in chronic heart failure with reduced left ventricular ejection fraction: A single center study including patients with left bundle branch block. Cardiol. J. 2020, 27, 575–582. [Google Scholar] [CrossRef] [PubMed]
- Rossi, L.; Malagoli, A.; Piepoli, M.; Franchi, F.; Malavasi, V.; Casali, E.; Rusticali, G.; Villani, G.Q. Indexed maximal left atrial volume predicts response to cardiac resynchronization therapy. Int. J. Cardiol. 2013, 168, 3629–3633. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Végh, E.M.; Kandala, J.; Januszkiewicz, L.; Ren, J.; Miller, A.; Orencole, M.; Blendea, D.; Merkely, B.; Gellér, L.; Singh, J.P.; et al. A new simplified electrocardiographic score predicts clinical outcome in patients treated with CRT. Europace 2018, 20, 492–500. [Google Scholar] [CrossRef]
- Bartko, P.E.; Arfsten, H.; Heitzinger, G.; Pavo, N.; Strunk, G.; Gwechenberger, M.; Hengstenberg, C.; Binder, T.; Hülsmann, M.; Goliasch, G. Papillary Muscle Dyssynchrony-Mediated Functional Mitral Regurgitation: Mechanistic Insights and Modulation by Cardiac Resynchronization. JACC Cardiovasc. Imaging 2019, 12, 1728–1737. [Google Scholar] [CrossRef]
- Kataoka, N.; Imamura, T.; Koi, T.; Uchida, K.; Kinugawa, K. A simple predictive marker in cardiac resynchronization therapy recipients: Prominent S-wave in right precordial leads. Medicina 2021, 57, 815. [Google Scholar] [CrossRef]
- Zoghbi, W.A.; Enriquez-Sarano, M.; Foster, E.; Grayburn, P.A.; Kraft, C.D.; Levine, R.A.; Nihoyannopoulos, P.; Otto, C.M.; Quinones, M.A.; Rakowski, H.; et al. Recommendations for evaluation of the severity of native valvular regurgitation with two-dimensional and Doppler echocardiography. J. Am. Soc. Echocardiogr. 2003, 16, 777–802. [Google Scholar] [CrossRef]
- Vahanian, A.; Alfieri, O.; Andreotti, F.; Antunes, M.J.; Barón-Esquivias, G.; Baumgartner, H.; Borger, M.A.; Carrel, T.P.; De Bonis, M.; Evangelista, A.; et al. Guidelines on the management of valvular heart disease (version 2012): The Joint Task Force on the Management of Valvular Heart Disease of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS). Eur. J. Cardiothorac. Surg. 2012, 42, S1–S44. [Google Scholar]
- Van der Bijl, P.; Vo, N.M.; Leung, M.; Ajmone Marsan, N.; Delgado, V.; Stone, G.W.; Bax, J.J. Impact of atrial fibrillation on improvement of functional mitral regurgitation in cardiac resynchronization therapy. Heart Rhythm. 2018, 15, 1816–1822. [Google Scholar] [CrossRef] [PubMed]
| Variable | Overall (N = 35) | With MR Reduction (N = 17) | Without MR Reduction (N = 18) | p-Value |
|---|---|---|---|---|
| Demographics | ||||
| Age, years | 71 [65–78] | 73 [67–81] | 70 [60–76] | 0.306 |
| Male (%) | 18 (51) | 6 (35) | 12 (67) | 0.063 |
| Body mass index, kg/m2 | 20.7 ± 3.7 | 20.0 ± 3.5 | 21.5 ± 3.7 | 0.239 |
| Ischemic etiology (%) | 4 (11) | 3 (18) | 1 (6) | 0.261 |
| Persistent atrial fibrillation/flutter (%) | 7 (20) | 3 (18) | 4 (22) | 0.735 |
| CRT-P (%) | 7 (20) | 5 (29) | 2 (11) | 0.176 |
| ICD for primary prevention (%) | 17 (61) | 9 (75) | 8 (50) | 0.180 |
| Comorbidity | ||||
| Chronic kidney disease (%) | 11 (31) | 4 (24) | 7 (39) | 0.328 |
| Diabetes mellitus (%) | 6 (17) | 4 (24) | 2 (11) | 0.330 |
| NYHA functional classification IV (%) | 4 (11) | 2 (12) | 2 (11) | 0.952 |
| Pre-implantation vital signs | ||||
| Heart rate, bpm | 73 ± 15 | 77 ± 17 | 69 ± 13 | 0.145 |
| Systolic blood pressure, mmHg | 107 ± 19 | 105 ± 19 | 108 ± 18 | 0.642 |
| Diastolic blood pressure, mmHg | 66 ± 12 | 62 ± 11 | 70 ± 12 | 0.056 |
| Medications | ||||
| ACE-I or ARB (%) | 31 (89) | 15 (88) | 16 (89) | 0.952 |
| Beta-blockers (%) | 27 (77) | 14 (82) | 13 (72) | 0.476 |
| Loops (%) | 29 (83) | 15 (88) | 14 (78) | 0.412 |
| Digitalis (%) | 2 (6) | 1 (6) | 1 (6) | 0.967 |
| Inotropes (%) | 5 (14) | 2 (12) | 3 (17) | 0.679 |
| Amiodarone (%) | 11 (31) | 4 (24) | 7 (39) | 0.328 |
| Laboratory data | ||||
| Serum albumin, g/dL | 3.7 ± 0.4 | 3.7 ± 0.4 | 3.7 ± 0.4 | 0.840 |
| Serum total bilirubin, mg/dL | 0.6 [0.5–0.9] | 0.6 [0.4–0.8] | 0.6 [0.5–1.0] | 0.414 |
| Serum creatinine, mg/dL | 0.9 [0.7–1.3] | 0.8 [0.7–1.2] | 1.1 [0.8–1.5] | 0.129 |
| Estimated GFR, mL/min/1.73 m2 | 50.4 ± 21.4 | 55.8 ± 17.5 | 45.9 ± 24.0 | 0.250 |
| Serum sodium, mEq/L | 138 [134–140] | 138 [134–140] | 139 [136–140] | 0.405 |
| Hemoglobin, g/dL | 12.5 ± 2.0 | 11.9 ± 1.8 | 13.2 ± 2.0 | 0.060 |
| Plasma B-type natriuretic peptide, pg/mL | 482 [167–909] | 488 [171–944] | 384 [162–950] | 0.918 |
| Echocardiographic parameters | ||||
| Left atrial dimension, mm | 47 ± 9 | 43 ± 8 | 50 ± 9 | 0.031 |
| Left ventricular end-diastolic dimension, mm | 63 [56–68] | 62 [57–73] | 64 [56–66] | 0.791 |
| Left ventricular end-systolic dimension, mm | 52 [48–61] | 53 [49–63] | 52 [47–58] | 0.488 |
| Left ventricular end-systolic volume, mL | 132 [106–189] | 144 [110–199] | 130 [101–168] | 0.427 |
| Left ventricular ejection fraction, % | 27 ± 8 | 25 ± 7 | 29 ± 9 | 0.164 |
| Degree of MR | 0.689 | |||
| Grade 1 | 19 (54) | 10 (59) | 9 (50) | |
| Grade 2 | 4 (11) | 1 (6) | 3 (17) | |
| Grade 3 | 3 (9) | 2 (12) | 1 (6) | |
| Grade 4 | 9 (26) | 4 (24) | 5 (28) | |
| Electrocardiographic parameters | ||||
| QRS morphology (%) | 0.001 | |||
| Left bundle branch block (%) | 10 (29) | 9 (53) | 1 (6) | 0.002 |
| Right bundle branch block (%) | 8 (23) | 1 (6) | 7 (39) | 0.020 |
| Intraventricular conduction disturbance (%) | 5 (14) | 0 (0) | 5 (28) | 0.019 |
| Right ventricular pacing (%) | 12 (34) | 7 (41) | 5 (28) | 0.404 |
| Axis, degree | 0 [–63–0] | 0 [−29–14] | −32 [−78–0] | 0.052 |
| PR interval, msec | 208 [185–287] | 200 [182–247] | 250 [186–311] | 0.157 |
| QRS duration in II, msec | 174 ± 29 | 174 ± 31 | 174 ± 27 | 0.976 |
| Averaged S-wave amplitude in V1–3, mV | 1.3 [0.7–3.0] | 3.0 [1.5–3.5] | 0.7 [0.4–1.2] | <0.001 |
| Biventricular pacing rate at six months, % | 99.0 [98.2–99.8] | 99.0 [99.0–100] | 99.0 [96.5–99.3] | 0.095 |
| Variables | Univariable | Multivariable | ||
|---|---|---|---|---|
| OR [95%CI] | p-Value | OR [95%CI] | p-Value | |
| Atrial fibrillation/flutter | 0.75 [0.14–3.98] | 0.736 | ||
| Left atrial diameter | 0.91 [0.83–1.00] | 0.046 | 0.94 [0.81–1.09] | 0.376 |
| Left bundle branch block | 19.1 [2.06–177.92] | 0.010 | 7.24 [0.40–131.71] | 0.181 |
| Right bundle branch block | 0.08 [0.01–0.72] | 0.025 | 0.36 [0.02–5.88] | 0.477 |
| Right ventricular pacing | 1.82 [0.44–7.48] | 0.406 | ||
| QRS axis > −30 degrees | 0.31 [0.07–1.31] | 0.112 | ||
| Averaged S-wave amplitude in V1–3 | 6.52 [1.88–22.62] | 0.003 | 14.00 [1.65–119.00] | 0.016 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kataoka, N.; Imamura, T.; Koi, T.; Tanaka, S.; Fukuda, N.; Ueno, H.; Kinugawa, K. Impact of S-Wave Amplitude in Right Precordial Leads on Improvement in Mitral Regurgitation following Cardiac Resynchronization Therapy. J. Cardiovasc. Dev. Dis. 2022, 9, 159. https://doi.org/10.3390/jcdd9050159
Kataoka N, Imamura T, Koi T, Tanaka S, Fukuda N, Ueno H, Kinugawa K. Impact of S-Wave Amplitude in Right Precordial Leads on Improvement in Mitral Regurgitation following Cardiac Resynchronization Therapy. Journal of Cardiovascular Development and Disease. 2022; 9(5):159. https://doi.org/10.3390/jcdd9050159
Chicago/Turabian StyleKataoka, Naoya, Teruhiko Imamura, Takahisa Koi, Shuhei Tanaka, Nobuyuki Fukuda, Hiroshi Ueno, and Koichiro Kinugawa. 2022. "Impact of S-Wave Amplitude in Right Precordial Leads on Improvement in Mitral Regurgitation following Cardiac Resynchronization Therapy" Journal of Cardiovascular Development and Disease 9, no. 5: 159. https://doi.org/10.3390/jcdd9050159
APA StyleKataoka, N., Imamura, T., Koi, T., Tanaka, S., Fukuda, N., Ueno, H., & Kinugawa, K. (2022). Impact of S-Wave Amplitude in Right Precordial Leads on Improvement in Mitral Regurgitation following Cardiac Resynchronization Therapy. Journal of Cardiovascular Development and Disease, 9(5), 159. https://doi.org/10.3390/jcdd9050159







