Risk of Cardiac Lesion with Chronic and Acute Use of Loperamide—An Integrative Review
Abstract
1. Introduction
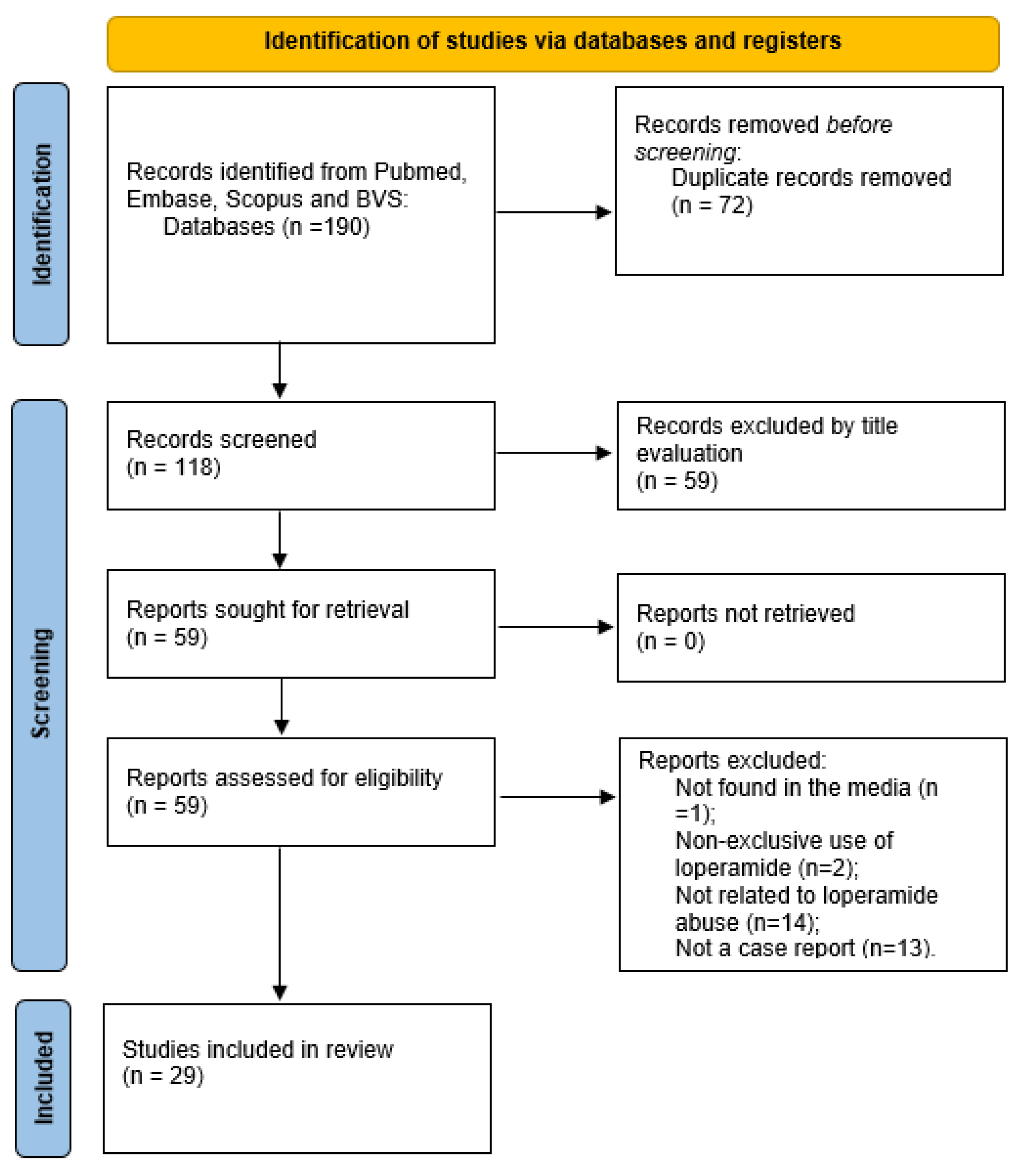
2. Materials and Methods
3. Eligibility Criteria
4. Selection Process
5. Results
6. Discussion
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ali, M.; Mujahid, A.; Bulathsinghala, C.P.; Surani, S. Cardiac Arrhythmia Secondary to Loperamide Abuse and Toxicity. Cureus 2020, 12, e6936. [Google Scholar] [CrossRef] [PubMed]
- Cicci, J.D.; Jagielski, S.M.; Clarke, M.M.; Rayson, R.A.; Cavender, M.A. Loperamide Overdose Causing Torsades de Pointes and Requiring Impella Temporary Mechanical Support: A Case Report. Eur. Heart J.-Case Rep. 2019, 3, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, A.; Bodar, V.; Malekzadegan, M.; Singh, S.; Frumkin, W.; Mangla, A.; Doshi, K. Loperamide Induced Life Threatening Ventricular Arrhythmia. Case Rep. Cardiol. 2016, 2016, 5040176. [Google Scholar] [CrossRef] [PubMed]
- Toronto, C.E.; Remington, R. A Step-By-Step Guide to Conducting an Integrative Review, 1st ed.; Springer International Publishing AG: Cham, Switzerland, 2020. [Google Scholar] [CrossRef]
- Sun, C.; Brice, J.A.; Clark, R.F. Brugada-Type Pattern on Electrocardiogram Associated with High-Dose Loperamide Abuse. J. Emerg. Med. 2018, 54, 484–486. [Google Scholar] [CrossRef] [PubMed]
- Stefek, B.; Wolfe, L.T.; Cohen, M. Brugada Syndrome Associated with Adolescent Loperamide Abuse. Pediatrics 2018, 142, e20181423. [Google Scholar] [CrossRef]
- Simon, M.; Rague, J. A Case Report of Torsade de Pointes and Brugada Pattern Associated with Loperamide Misuse and Supratherapeutic Loperamide Concentrations. J. Emerg. Med. 2021, 61, e54–e59. [Google Scholar] [CrossRef]
- Jablonski, S.; Cowgill, J.; Powers, J. Loperamide Cardiotoxicity: A Byproduct of the Opioid Epidemic; Maine Medical Center: Portland, ME, USA, 2019; Volume 1050, Available online: https://knowledgeconnection.mainehealth.org/mmc/1050 (accessed on 22 November 2022).
- Teigeler, T.; Stahura, H.; Alimohammad, R.; Kalahasty, G.; Koneru, J.N.; Ellenbogen, M.; Ellenbogen, K.A.; Padala, S.K. Electrocardiographic Changes in Loperamide Toxicity: Case Report and Review of Literature. J. Cardiovasc. Electrophysiol. 2019, 30, 2618–2626. [Google Scholar] [CrossRef]
- Kohli, U.; Altujjar, M.; Sharma, R.; Hassan, S. Wide Interindividual Variability in Cardiovascular Toxicity of Loperamide: A Case Report and Review of Literature. HeartRhythm Case Rep. 2019, 5, 221–224. [Google Scholar] [CrossRef]
- Escobedo, Y.; Wilson, G.; Costa, S.M.; Ellington, T. The Long-Lasting High: Cardiac Complications from Loperamide Toxicity’; White Medical Center: Dallas, TX, USA, 2020; Volume 2842. [Google Scholar] [CrossRef]
- Atoot, A.; Sholem, S.; Khaddash, I.; Zuberi, J. Transient Brugada Pattern Induced by Loperamide Abuse. Cureus 2020, 12, e8037. [Google Scholar] [CrossRef]
- Palkar, P.; Kothari, D. Bradycardia and Syncope in a Patient Presenting with Loperamide Abuse. Cureus 2018, 10, e2599. [Google Scholar] [CrossRef]
- Modi, V.; Krinock, M.; Desai, R.; Stevens, S.; Nanda, S. Loperamide-Induced Cardiac Events: Case Reports and Review. Cureus 2021, 13, e20744. [Google Scholar] [CrossRef] [PubMed]
- Larsen, T.R.; McMunn, J.; Ahmad, H.; AlMahameed, S.T. Ventricular Tachycardia Triggered by Loperamide and Famotidine Abuse. Drug Saf.-Case Rep. 2018, 5, 11. [Google Scholar] [CrossRef] [PubMed]
- Sheikh, S.; Baig, M.A. A heart-wrenching case of loperamide toxicity. Asian Pac. J. Med. Toxicol. 2019, 8, 141–143. Available online: http://apjmt.mumsac.ir (accessed on 22 November 2022).
- Stone, B.; Zariwala, A.; Acho, M.; Barbash, I. Managing the Cardiotoxicity of Loperamide Overdose, an Emerging Issue in the Opioid Crisis. In C41. Critical Care Case Reports: Toxicology and Poisonings; American Thoracic Society: New York, NY, USA, 2019; p. 4809. [Google Scholar]
- Parker, B.M.; Rao, T.; Matta, A.; Quitanna, M.; Reynolds, H.N.; Stein, D.M.; Haase, D. Loperamide Induced Cardiac Arrhythmia Successfully Supported with Veno-Arterial ECMO (VA-ECMO), Molecular Adsorbent Recirculating System (MARS) and Continuous Renal Replacement Therapy (CRRT). Clin. Toxicol. 2019, 57, 1118–1122. [Google Scholar] [CrossRef] [PubMed]
- Isang, E.; Shali, L.; Morris, C.B.; Mahlow, J. Loperamide-Induced Torsades de Pointes. Cureus 2021, 13, e20299. [Google Scholar] [CrossRef] [PubMed]
- Kapaganti, S.; Anwar Ansari, S.; Saba, R.; Elkhouly, A.; Hassib, M. A Rare Case of Loperamide-Induced Cardiac Arrest. Cureus 2020, 12, e9396. [Google Scholar] [CrossRef]
- Whittaker, G.; Newman, J. Loperamide: An Emerging Drug of Abuse and Cause of Prolonged QTc. Clin. Med. 2021, 21, 150–152. [Google Scholar] [CrossRef]
- Rawala, M.S.; Gulati, R.; Rizvi, S. Cardiac Dysrhythmia Associated with Opioid Toxicity. Cureus 2020, 12, e8243. [Google Scholar] [CrossRef]
- De Vera, J.; Kim, H.B.; Sakr, A.E. A Case Report of Loperamide-Induced Ventricular Storm. J. Investig. Med. High Impact Case Rep. 2021, 9, 232470962199076. [Google Scholar] [CrossRef]
- Gaines, H.; Mao, T.; Murray, C.; Sivaram, C.A. The Twisted Truth: Identifying and Treating Torsade de Pointes (TDP) in Surreptitious Drug Use; University of Oklahoma Health Sciences Center: Oklahoma City, OK, USA, 2020; Volume 2819. [Google Scholar] [CrossRef]
- Rojas, S.F.; Oglat, A.; Bonilla, H.M.G.; Jeroudi, O.; Sharp, W.; Valderrábano, M.; Schurmann, P.A. Loperamide Mimicking Brugada Pattern. Methodist DeBakey Cardiovasc. J. 2018, 14, e1–e3. [Google Scholar] [CrossRef]
- Idris, A.; Mihora, D.C.; Kaye, K. Loperamide Abuse Cardiotoxicity. Should Loperamide Still Be an over the Counter Medication? Am. J. Emerg. Med. 2018, 36, 1716.e1–1716.e3. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Chitturi, K.R.; Hatcher, C.; Clewing, M.; Nagueh, S.F. Loperamide Toxicity Revealing Apical Hypertrophic Cardiomyopathy. Methodist DeBakey Cardiovasc. J. 2021, 17, 65. [Google Scholar] [CrossRef] [PubMed]
- Zaman, M.O.; Ali, M.U.; Finkel, J. A Case of Loperamide Induced Cardiac Syncope; Crozer Chester Medical Center: Springfield, PA, USA, 2018; Volume 2550. [Google Scholar] [CrossRef]
- Wang, A.; Nguyen, M.; Diaz, J.; Smith, T. Loperamide Toxicity Mimicking Peripartum Cardiomyopathy. Am. J. Emerg. Med. 2020, 38, 693.e5–693.e6. [Google Scholar] [CrossRef] [PubMed]
- Sapra, A.; Bhandari, P.; Gupta, S.; Kraleti, S.; Bahuva, R. A Rising Concern of Loperamide Abuse: A Case Report on Resulting Cardiac Complications. Cureus 2019, 11, e6314. [Google Scholar] [CrossRef]
- Myllymaki, A.; Westerbergh, J.; Carlsson, H.; Hoje, J. Torsade de pointes following repeated massive loperamide ingestions. Lakartidningen 2020, 117, FWWW. [Google Scholar] [PubMed]
- Lee, V.R.; Vera, A.; Alexander, A.; Ruck, B.; Nelson, L.S.; Wax, P.; Campleman, S.; Brent, J.; Calello, D.P. Loperamide Misuse to Avoid Opioid Withdrawal and to Achieve a Euphoric Effect: High Doses and High Risk. Clin. Toxicol. 2019, 57, 175–180. [Google Scholar] [CrossRef]
- Akel, T.; Bekheit, S. Loperamide Cardiotoxicity: A Brief Review. Ann. Noninvasive Electrocardiol. 2018, 23, e12505. [Google Scholar] [CrossRef]
- Riaz, I.B.; Khan, M.S.; Kamal, M.U.; Sipra, Q.-A.R.; Riaz, A.; Zahid, U.; Bhattacharjee, S. Cardiac Dysrhythmias Associated with Substitutive Use of Loperamide: A Systematic Review. Am. J. Ther. 2019, 26, e170–e182. [Google Scholar] [CrossRef]
- Wolfes, J.; Ellermann, C.; Burde, S.; Leitz, P.; Bögeholz, N.; Willy, K.; Fehr, M.; Reinke, F.; Eckardt, L.; Frommeyer, G. Divergent Electrophysiological Effects of Loperamide and Naloxone in a Sensitive Whole-Heart Model. Cardiovasc. Toxicol. 2021, 21, 248–254. [Google Scholar] [CrossRef]
- Li, J.; Zhang, Q.; Zhang, M.; Egger, M. Intravenous Magnesium for Acute Myocardial Infarction. Cochrane Database Syst. Rev. 2007, 2009, CD002755. [Google Scholar] [CrossRef]
- Ho, K.M.; Sheridan, D.J.; Paterson, T. Use of Intravenous Magnesium to Treat Acute Onset Atrial Fibrillation: A Meta-Analysis. Heart 2007, 93, 1433–1440. [Google Scholar] [CrossRef] [PubMed]
| Author | Age | Dose of Ingested Loperamide | Electrocardiogram (ECG) | Symptoms/Signs | Treatment | Clinical Outcome |
|---|---|---|---|---|---|---|
| Sun C. et al., 2018 [5] | 49 | 200 to 400 mg | Corrected QT (QTc) of 644 ms and QRS of 170 ms | - | IV infusion of magnesium | 6 months before the ECG showed a QTc interval of 429 ms and normal ST segments |
| Stefek B. et al., 2018 [6] | 14 | 40 mg nightly | ECG revealing anterior precordial ST segment elevation in leads V1–V3 with a “coved” appearance consistent with a type 1 Brugada pattern QRS duration was 160 ms, and QTc interval was 568 ms | Nausea, vomiting, diaphoresis, dizziness and weakness | Magnesium sulfate, ondansetron, sodium bicarbonate infusion, pantoprazole, lorazepam, morphine, and acetaminophen | A diagnosis of Brugada syndrome was made |
| Simon M. and Rague J., 2021 [7] | 22 | 600 mg | ECG showed a prolonged QTcR interval of 667 ms, QRS of 136 ms, with ST elevations in V1–V3 with a “coved” appearance followed by a negative T wave consistent with a Type 1 Brugada pattern | Multiple witnessed syncopal episodes | IV magnesium sulfate | The patient receieved an isoproterenol infusion |
| Jablonski S. et al., 2019 [8] | 33 | >100 mg | ECG showed sinus bradycardia, first-degree atrioventricular (AV) block, QRS interval of 192 ms and QTc of 615 ms | Two witnessed cardiac arrests, both with torsades de pointes | Magnesium and cardioversion followed by temporary venous pacemaker placement | ECG and echocardiogram normalized on day 3 of admission with no further dysrhythmia |
| Teigeler T. et al., 2019 [9] | 39 | 40–60 mg twice daily | ECG revealed an idioventricular rhythm at a rate of 34 bpm, QRS duration of 154 ms, and a QTc of 444 ms | Lightheadedness and presyncope | - | Bradycardia resolved. ECG normalized after 5 days |
| Kohli U. et al., 2019 [10] | 26 | 300 mg | ECG showed markedly prolonged QTc and Brugada type 1 changes in leads V1 and V2. QTc-prolonged (>500 ms) | Syncope | A loop recorder was placed | Discharged home with a life vest |
| Escobedo Y. et al., 2020 [11] | 30 | 140 mg | EMS found refractory torsades de pointes, a normal sinus mechanism with a normal QTc that progressively increased to 800 ms | Witnessed cardiac arrest | Veno-arterial extracorporeal membrane oxygenation (VA ECMO) and inotropic support | Echocardiogram revealed normalized LVEF on discharge |
| Atoot A. et al., 2020 [12] | 28 | 280 mg | Echocardiogram demonstrated no regional wall motion abnormalities, and ST-segment elevation in V1–V3 leads | Excruciating abdominal pain | - | Transient Brugada pattern secondary to loperamide abuse |
| Ali M. et al., 2020 [1] | 31 | 400 mg | ECG revealed a widened ventricular arrhythmia, short runs (<3 s) of ventricular tachycardia, and prolonged QTc of 663 ms | Shortness of breath and weakness | IV magnesium and bicarbonate drip | 4 days after hospitalization, the ECG had improved back to normal sinus rhythm with prolonged QTc |
| Palkar P. and Kothari D., 2018 [13] | 32 | >100 mg | ECG showed right bundle branch block, bradycardia with a heart rate of 51 bpm, and normal QTc | Syncopal episode, nausea, vomiting, constipation, and abdominal cramps | Cardiac monitor with normal saline intravenous for hydration | Discharge with a Holter monitor |
| Modi V. et al., 2021 [14] | 28 | 200 mg | ECG showed widened QRS and prolonged QT interval. After treatment with a bicarbonate drip, the patient developed an episode of polymorphic ventricular tachycardia | Weakness, difficulty taking deep breaths, and mild lightheadedness | Sodium bicarbonate | Temporary pacemaker |
| Larsen T. et al., 2018 [15] | 32 | 200 mg | ECG showed sinus tachycardia at 101 bpm, first-degree AV block, nonspecific intraventricular conduction delay, and prolonged QT interval | Severe palpitations and syncope | Intravenous magnesium and supportive care | After 5 days PR interval, QRS duration, and normalized QT interval and resolved VT |
| Sheikh S. and Baig M.A., 2019 [16] | 30 | - | ECG showed prolonged QT and QTc intervals of 600 ms. Cardiac monitoring showed torsades de pointes with hemodynamic instability | Generalized tonic–clonic seizures | Intubation, IV magnesium and IV amiodarone | The patient needed cardiopulmonary resuscitation. Temporary pacemaker |
| Stone B. et al., 2021 [17] | 41 | 400 mg | ECG revealed sinus bradycardia with prolonged PR, QRS, and QTc intervals at 214, 148, and 678 ms, respectively | Syncope | Intensive care unit on norepinephrine, dopamine, and isoproterenol | Started on buprenorphine–naloxone and discharged. He stabilized on day three (QTc normal) |
| Author | Age | Dose of Ingested Loperamide | Electrocardiogram (ECG) | Symptoms/Signs | Treatment | Clinical Outcome |
|---|---|---|---|---|---|---|
| Parker M.B. et al., 2019 [18] | 36 | 400–600 mg | ECG revealed torsades de pointes, QRS 170 ms; QTc 831 ms | Bradycardia, hypothermia, bradypnea, and hypoglycemia | VA-ECMO, sodium bicarbonate infusion, N-acetylcysteine infusion, dextrose, atropine, and magnesium sulfate | ECG revealed a narrow QRS complex with normalized QTc to 410 ms |
| Isang E. et al., 2021 [19] | 34 | 96 mg | ECG showed first-degree AV block, with a heart rate of 86 bpm and a prolonged QTc of 560 ms | Gastrointestinal symptoms, lethargy, hypoxia, and bradycardia | Naloxone, intravenous magnesium, bicarbonate infusion, and isoproterenol | Torsades de pointes and was successfully defibrillated with 120 J |
| Kapaganti S. et al., 2020 [20] | 38 | Took an unknown amount of loperamide | Prolonged QTc of >600 ms | Unresponsive | Lidocaine drip, electrolyte repletion | A repeat EKG showed that QTc had shortened to less than 500 ms within 24 hhypoxic brain injury |
| Cicci J.D. et al., 2019 [2] | 23 | 160 mg | ECG showed pulseless ventricular fibrillation with a QTc of 554 ms and a cove-like ST-elevation pattern. Episodes of torsades de pointes | Arrhythmias | Multiple defibrillators and IV lidocaine. Mechanical circulatory support, IV magnesium | Torsades de pointes (TdP) (QTc 613 ms) requiring three electrical cardioversions |
| Whittaker G. and Newman J., 2021 [21] | 30 | 100 mg | ECG showed a prolonged QTc ranging between 553 and 567 ms. First-degree AV block and right bundle branch block were also present | Syncope, palpitations, light-headedness, and loss of consciousness | Lidocaine IV | Switch the loperamide to buprenorphine patches. Cardiologist and psychiatrist evaluation |
| Rawala M.S., 2020 [22] | 33 | 200 mg | ECG showed QTc of 647 ms and wide complex tachycardia | Syncope | IV amiodarone | Discharged on mexiletine with normal QTc |
| Vera De J. et al., 2021 [23] | 29 | 600 mg | ECG showed polymorphic ventricular tachycardia with prolonged QTc of 669 ms. Recurrent episodes of ventricular tachycardia that degenerated into torsades de pointes | Altered, confused, tachycardic, and hypotensive | IV magnesium sulfate and sodium bicarbonate | Multiple cardioversions, admission to the cardiac intensive care unit and Isoproterenol |
| Gaines H. et al., 2020 [24] | 28 | 400 mg | ECG showed junctional rhythm and QTc of 557 ms | Bradycardia and syncope | IV magnesium, defibrillation and isoproterenol | Isoproterenol for 10 days |
| Rojas S.F. et al., 2018 [25] | 48 | 12 a 18 mg/day | ECG mimicked type 1 Brugada pattern on leads V1–V2 and showed right axis deviation (RAD), first-degree AV block with PR interval 339 ms, and right bundle branch block with QRS duration of 270 ms and QTc of 578 ms | Syncope, palpitations, and generalized weakness | Supportive care | Monitoring by 72 h until electrocardiographic abnormalities were resolved |
| Idris A. and Kaye K., 2018 [26] | 33 | 140 mg | ECG showed sinus rhythm with first-degree AV block, normal QRS, but prolonged QT/QTc intervals of 586–724 ms | Acute onset of shortness of breath, generalized weakness, and tingling over her entire body | IV infusion of sodium bicarbonate | ECG after bicarbonate showed improved QT/QTc, up to 466/531 ms at 24 h. The patient remained asymptomatic and was discharged. Ten days after discharge, QT/QTc intervals were 426/460 ms |
| Khan N. et al., 2021 [27] | 25 | 400 mg | QT prolongation and giant inverted T waves in V4–V6 | Palpitations and arrhythmia | Metoprolol succinate | Follow-up visit with a cardiologist and genetic testing |
| Zaman M.O., et al., 2018 [28] | 25 | 30 mg daily | ECG showed non-specific ventricular rhythm with markedly increased QRS and QT interval and S3 gallop | Syncope | Amiodarone, magnesium, and sodium bicarbonate | Discontinuation of loperamide use |
| Wang A. et al., 2019 [29] | 33 | 40 mg daily | ECG showed an accelerated idioventricular rhythm, widened QRS (183 ms) and prolonged QTc (700 ms) | Hypertension and dyspnea | Betamethasone, furosemide, nitroglycerin, and nasal intermittent positive pressure ventilation (NIPPV) | Medical treatment and urgent cesarean section. After 5 days in hospital, her QTc stabilized |
| Sapra A. et al., 2019 [30] | 32 | 400 mg | ECG showed persistently prolonged QT interval, and nonspecific ST changes were noted | Atypical chest pain, palpitations, syncope, episode of polymorphic ventricular tachycardia, and shortness of breath | IV amiodarone and magnesium | Admission to the intensive care unit, the QT interval stabilized, and implantable cardiac defibrillator (ICD) therapy |
| Myllymäki L. et al., 2020 [31] | 40 | 96 to 320 mg | ECG showed irregular heartbeat, widened QRS complex of 142 ms, and QTc of 465 ms | Syncope | IV magnesium, potassium replacement | Temporary pacemaker was inserted prophylactically, and torsades de pointes |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lammoglia, B.C.; Hasselmann, G.; Pires-Oliveira, M.; Duarte Nicolau, L.A.; Rolim Medeiros, J.V.; Sabia Tallo, F.; Omar Taha, M.; Yamaguti Lima, R.; Caricati-Neto, A.; Menezes-Rodrigues, F.S. Risk of Cardiac Lesion with Chronic and Acute Use of Loperamide—An Integrative Review. J. Cardiovasc. Dev. Dis. 2022, 9, 431. https://doi.org/10.3390/jcdd9120431
Lammoglia BC, Hasselmann G, Pires-Oliveira M, Duarte Nicolau LA, Rolim Medeiros JV, Sabia Tallo F, Omar Taha M, Yamaguti Lima R, Caricati-Neto A, Menezes-Rodrigues FS. Risk of Cardiac Lesion with Chronic and Acute Use of Loperamide—An Integrative Review. Journal of Cardiovascular Development and Disease. 2022; 9(12):431. https://doi.org/10.3390/jcdd9120431
Chicago/Turabian StyleLammoglia, Bruna Cremonezi, Gabriela Hasselmann, Marcelo Pires-Oliveira, Lucas Antonio Duarte Nicolau, Jand Venes Rolim Medeiros, Fernando Sabia Tallo, Murched Omar Taha, Rildo Yamaguti Lima, Afonso Caricati-Neto, and Francisco Sandro Menezes-Rodrigues. 2022. "Risk of Cardiac Lesion with Chronic and Acute Use of Loperamide—An Integrative Review" Journal of Cardiovascular Development and Disease 9, no. 12: 431. https://doi.org/10.3390/jcdd9120431
APA StyleLammoglia, B. C., Hasselmann, G., Pires-Oliveira, M., Duarte Nicolau, L. A., Rolim Medeiros, J. V., Sabia Tallo, F., Omar Taha, M., Yamaguti Lima, R., Caricati-Neto, A., & Menezes-Rodrigues, F. S. (2022). Risk of Cardiac Lesion with Chronic and Acute Use of Loperamide—An Integrative Review. Journal of Cardiovascular Development and Disease, 9(12), 431. https://doi.org/10.3390/jcdd9120431







