Spontaneous Left Cardiac Isomerism in Chick Embryos: Case Report, Review of the Literature, and Possible Significance for the Understanding of Ventricular Non-Compaction Cardiomyopathy in the Setting of Human Heterotaxy Syndromes
Abstract
1. Introduction
2. Materials and Methods
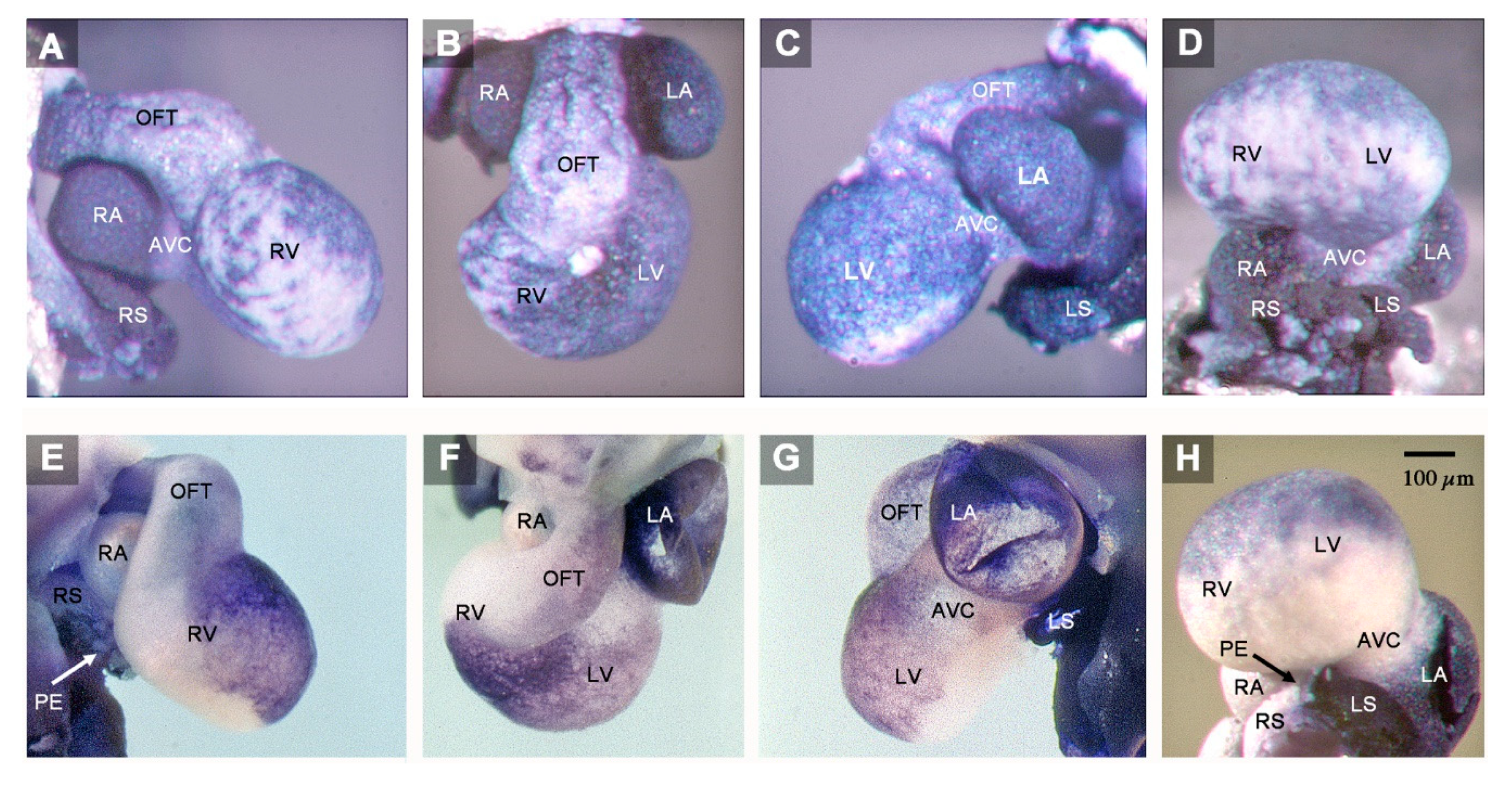
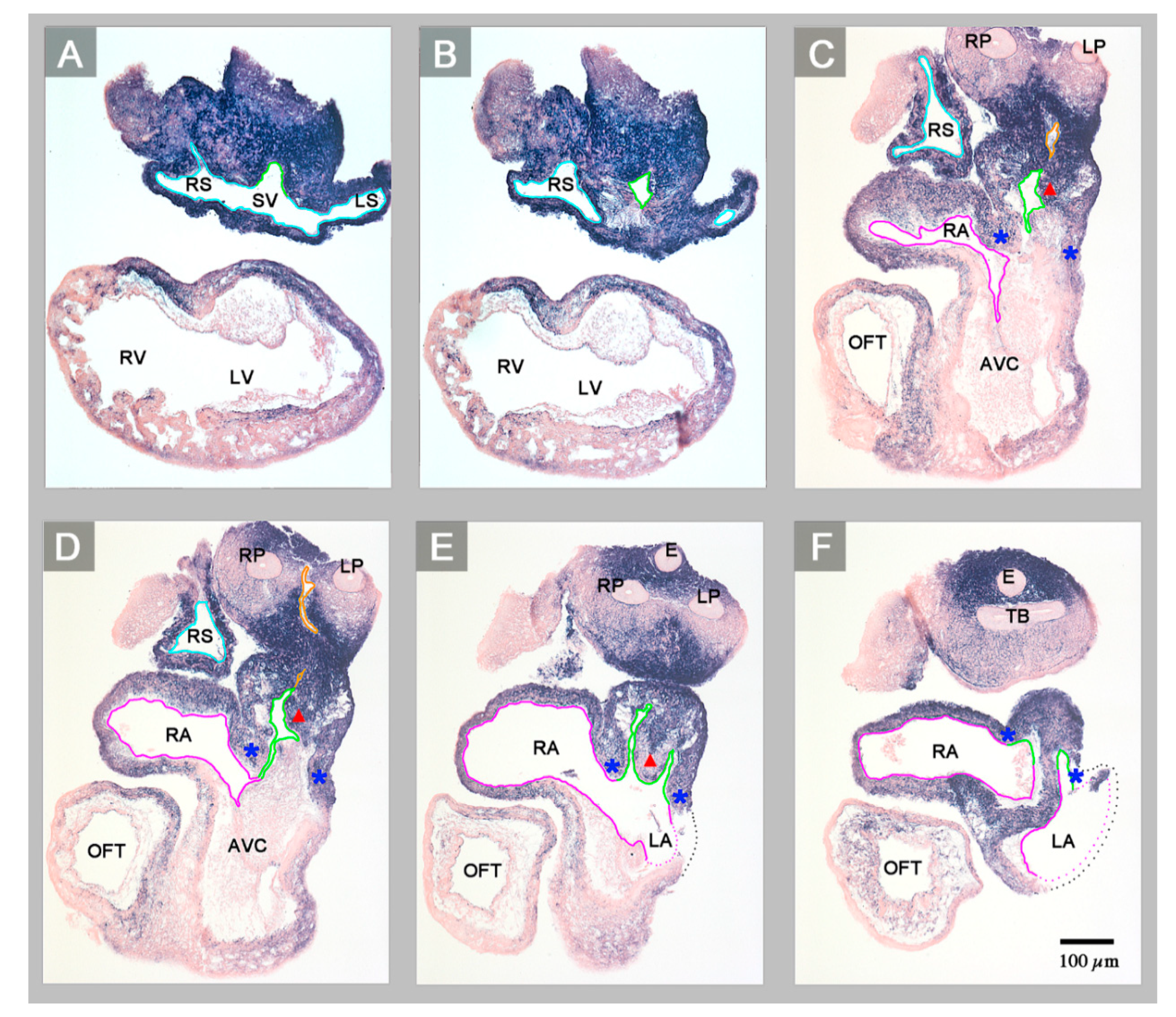
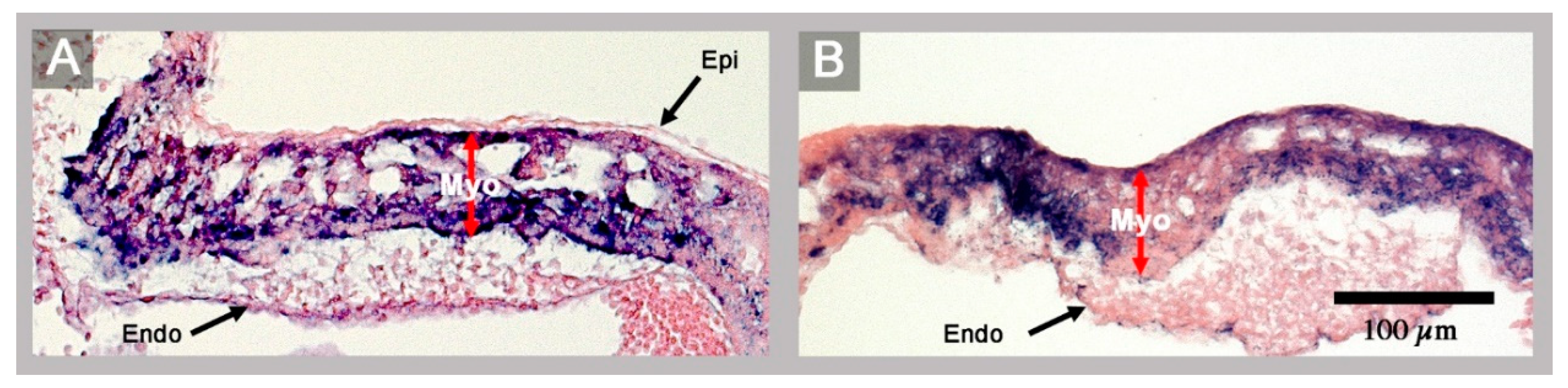
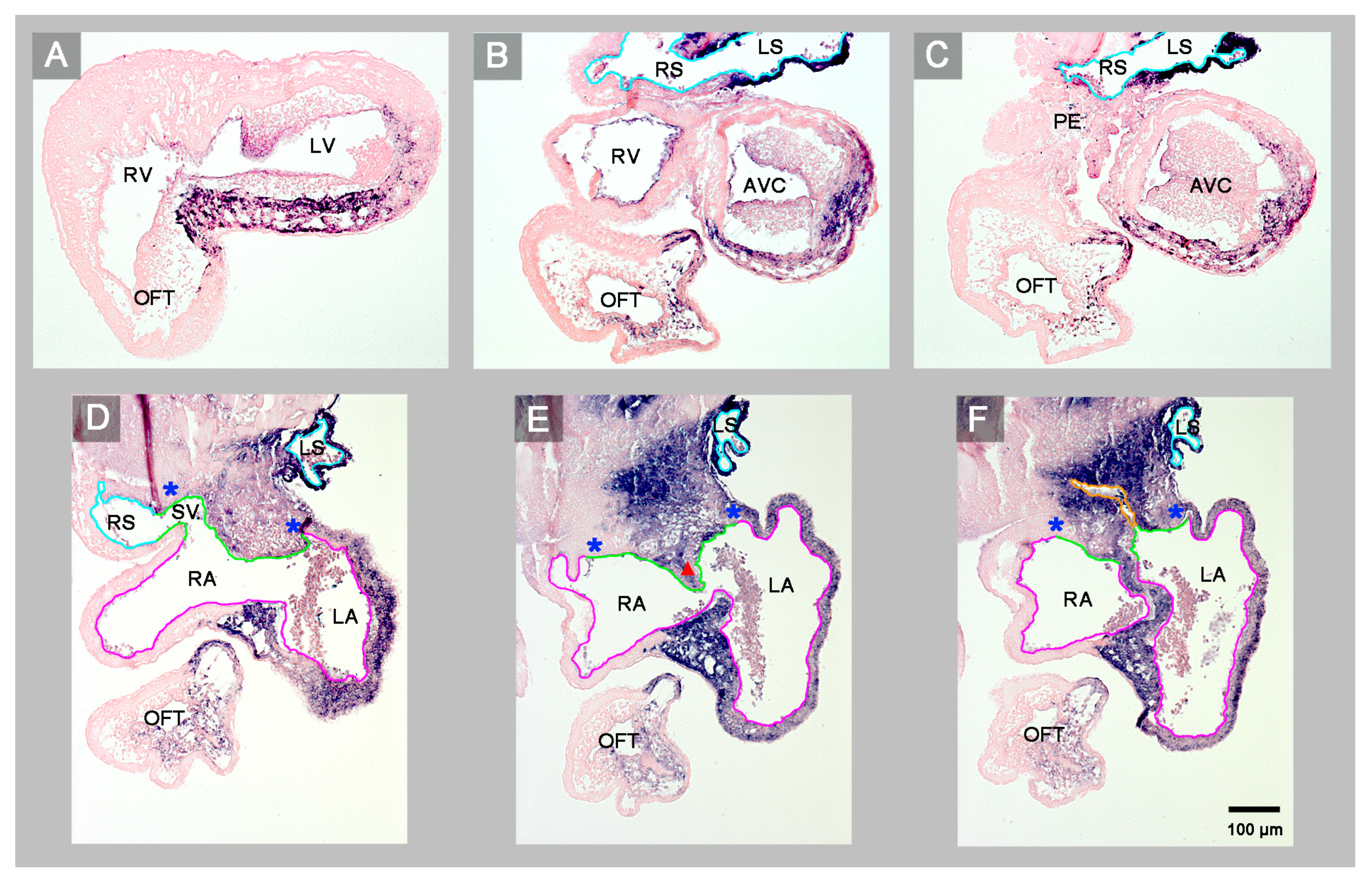
3. Results
4. Discussion
Funding
Acknowledgments
Conflicts of Interest
References
- Loomba, R.S.; Hlavacek, A.M.; Spicer, D.E.; Anderson, R.H. Isomerism or heterotaxy: Which term leads to better understanding. Cardiol. Young 2015, 25, 1037–1043. [Google Scholar] [CrossRef]
- Shiraishi, I.; Ichikawa, H. Human heterotaxy syndrome—From molecular genetics to clinical features, management, and prognosis. Circ. J. 2012, 76, 2066–2075. [Google Scholar] [CrossRef]
- Monsoro-Burq, A.H.; Levin, M. Avian models and the study of invariant asymmetry: How the chicken and the egg taught us to tell right from left. Int. J. Dev. Biol. 2018, 62, 63–77. [Google Scholar] [CrossRef]
- St Amand, T.R.; Ra, J.; Zhang, Y.; Hu, Y.; Baber, S.I.; Qiu, M.; Chen, Y. Cloning and expression pattern of chicken Pitx2: A new component of the SHH signaling pathway controlling embryonic heart looping. Biochem. Biophys. Res. Comm. 1998, 247, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Hamburger, V.; Hamilton, H.L. A series of normal stages in the development of the chick. J. Morphol. 1951, 88, 49–92. [Google Scholar] [CrossRef] [PubMed]
- Schulte, I.; Schlueter, J.; Abu-Issa, R.; Brand, T.; Männer, J. Morphological and molecular left-right asymmetries in the development of the proepicardium: A comparative analysis on mouse and chick embryos. Dev. Dyn. 2007, 236, 684–695. [Google Scholar] [CrossRef] [PubMed]
- Männer, J.; Perez-Pomares, J.M.; Macias, D.; Munoz-Chapuli, R. The origin, formation and developmental significance of the epicardium: A review. Cells Tissues Org. 2001, 196, 89–103. [Google Scholar] [CrossRef]
- Männer, J.; Merkel, N. Early morphogenesis of the sinuatrial region of the chick heart: A contribution to the understanding of the pathogenesis of direct pulmonary venous connections to the right atrium and atrial septal defects in hearts with right isomerism of the atrial appendages. Anat. Rec. 2007, 290, 168–180. [Google Scholar]
- Männer, J. The anatomy of cardiac looping: A step towards the understanding of the morphogenesis of several forms of congenital cardiac malformations. Clin. Anat. 2009, 22, 21–35. [Google Scholar] [CrossRef]
- Anderson, R.H.; Webb, S.; Brown, N. Defective lateralisation in children with congenitally malformed hearts. Cardiol. Young 1998, 8, 512–531. [Google Scholar] [CrossRef]
- Campione, M.; Ros, M.A.; Icardo, J.M.; Piedra, E.; Christoffels, V.M.; Schweickert, A.; Blum, M.; Franco, D.; Moorman, A.F. Pitx2 expression defines a left cardiac lineage of cells: Evidence for atrial and ventricular molecular isomerism in the iv/iv mice. Development 2001, 231, 252–264. [Google Scholar] [CrossRef] [PubMed]
- Schlueter, J.; Brand, T. A right sided pathway involving FGF8/Snai1 controls asymmetric development of the proepicardium in the chick embryo. Proc. Natl. Acad. Sci. USA 2009, 106, 7485–7490. [Google Scholar] [CrossRef] [PubMed]
- Moller, J.H.; Nakib, A.; Anderson, R.C.; Edwards, J.E. Congenital cardiac disease associated with polysplenia. A developmental complex of bilateral “left-sidedness”. Circulation 1967, 36, 789–799. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, D.F.; Wilkinson, J.L.; Anderson, K.R.; Smith, A.; Ho, S.Y.; Anderson, R.H. The cardiac conduction system in situs ambiguus. Circulation 1979, 59, 879–885. [Google Scholar] [CrossRef]
- Hildreth, V.; Webb, S.; Chaudhry, B.; Peat, J.D.; Phillips, H.M.; Brown, N.; Anderson, R.H.; Henderson, D.J. Left cardiac isomerism in the Sonic hedgehog null mouse. J. Anat. 2009, 214, 894–904. [Google Scholar] [CrossRef]
- De Tommasi, S.; Daliento, L.; Ho, S.Y.; Macartney, F.J.; Anderson, R.H. Analysis of atrioventricular junction, ventricular mass, and ventriculoarterial junction in 43 specimens with atrial isomerism. Br. Heart J. 1981, 45, 236–247. [Google Scholar] [CrossRef]
- Ho, S.Y.; Seo, J.W.; Brown, N.A.; Cook, A.C.; Fagg, N.L.; Anderson, R.H. Morphology of the sinus node in human and mouse hearts with isomerism of the atrial appendages. Br. Heart J. 1995, 74, 437–442. [Google Scholar] [CrossRef]
- Smith, A.; Ho, S.Y.; Anderson, R.H.; Connell, M.G.; Arnold, R.; Wilkinson, J.L.; Cook, A.C. The diverse cardiac morphology seen in hearts with isomerism of the atrial appendages with reference to the disposition of the specialised conduction system. Cardiol. Young 2006, 16, 437–454. [Google Scholar] [CrossRef]
- Yasui, H.; Morishima, M.; Nakazawa, M.; Aikawa, E. Anomalous looping, atrioventricular cushion dysplasia, and unilateral ventricular hypoplasia in the mouse embryos with right isomerism induced by retinoic acid. Anat. Rec. 1998, 250, 210–219. [Google Scholar] [CrossRef]
- Min, J.Y.; Kim, C.Y.; Oh, M.H.; Chun, Y.K.; Suh, Y.L.; Lee, H.J.; Seo, J.W. Arrangement of the systemic and pulmonary venous components of the atrial chambers in hearts with isomeric atrial appendages. Cardiol. Young 2000, 10, 396–404. [Google Scholar] [CrossRef]
- Meno, C.; Shimono, A.; Saijoh, Y.; Yashiro, K.; Mochida, K.; Ohishi, S.; Noji, S.; Kondho, H.; Hamada, H. lefty-1 is required for left-right determination as a regulator of lefty-2 and nodal. Cell 1998, 94, 287–297. [Google Scholar] [CrossRef]
- Tsukui, T.; Capdevila, J.; Tamura, K.; Ruiz-Lozano, P.; Rodriguez-Esteban, C.; Yonei-Tamura, S.; Magallón, J.; Chandraratna, R.A.S.; Chien, K.; Blumberg, B.; et al. Multiple left-right asymmetry defects in Shh (-/-) mutant mice unveil a convergence of the shh and retinoic acid pathways in the control of Lefty-1. Proc. Natl. Acad. Sci. USA 1999, 96, 11356–11381. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.P.; Li, E. The signalling pathway mediated by the type IIB activin receptor controls axial patterning and lateral asymmetry in the mouse. Genes Dev. 1997, 11, 1812–1826. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Markowitz, G.S.; Li, L.; D’Agati, V.D.; Factor, S.M.; Geng, L.; Tibara, S.; Tuchman, J.; Cai, Y.; Park, J.H.; et al. Cardiac defects and renal failure in mice with targeted mutations in Pkd2. Nat. Genet. 2000, 24, 75–78. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Liu, W.; Palie, J.; Lu, M.F.; Brown, N.A.; Martin, J.F. Pitx2c patterns anterior myocardium and aortic arch vessels and is required for local cell movement into atrioventricular cushions. Development 2002, 129, 5081–5091. [Google Scholar]
- Pennekamp, P.; Karcher, C.; Fischer, A.; Schweickert, A.; Skryabin, B.; Horst, J.; Blum, M.; Dworniczak, B. The ion channel polycystin-2 is required for left-right axis determination in mice. Curr. Biol. 2002, 12, 938–943. [Google Scholar] [CrossRef]
- Bamforth, S.D.; Braganca, J.; Farthing, C.R.; Schneider, J.E.; Broadbent, C.; Michell, A.C.; Clarke, K.; Neubauer, S.; Norris, D.; Brown, N.; et al. Cited2 controls left-right patterning and heart development through a Nodal-Pitx2c pathway. Nat. Genet. 2004, 11, 1189–1196. [Google Scholar] [CrossRef]
- Weninger, W.J.; Lopes Floro, K.; Bennett, M.B.; Withington, S.L.; Preis, J.I.; Barbera, J.P.; Mohun, T.J.; Dunwoodie, S.L. Cited2 is required both for heart morphogenesis and establishment of the left-right axis in mouse development. Development 2005, 132, 1337–1348. [Google Scholar] [CrossRef]
- Aune, C.N.; Chatterjee, B.; Zhao, X.Q.; Francis, R.; Bracero, L.; Yu, Q.; Rosenthal, J.; Leatherbury, L.; Lo, C.W. Mouse model of heterotaxy with single ventricle spectrum of cardiac anomalies. Pediatr. Res. 2008, 63, 9–14. [Google Scholar] [CrossRef]
- Seo, J.W.; Brown, N.A.; Ho, S.Y.; Anderson, R.H. Abnormal laterality and congenital cardiac anomalies. Relations of visceral and cardiac morphologies in the iv/iv mouse. Circulation 1992, 86, 642–650. [Google Scholar] [CrossRef]
- Morishima, M.; Ando, M.; Takao, A. Visceroatrial heterotaxy syndrome in the NOD mouse with special reference to atrial situs. Teratology 1991, 44, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Son, C.S.; Lee, J.W.; Tockgo, Y.C.; Chun, Y.H. Visceral heterotaxy syndrome induced by retinoids in mouse embryo. J. Korean Med. Sci. 1995, 10, 250–257. [Google Scholar] [CrossRef] [PubMed]
- Kain, K.H.; Miller, J.W.I.; Jones-Paris, C.R.; Thomason, R.T.; Lewis, J.D.; Bader, D.M.; Barnett, J.V.; Zijlstra, A. The chick embryo as an expanding experimental model for cancer and cardiovascular research. Dev. Dyn. 2014, 243, 216–228. [Google Scholar] [CrossRef] [PubMed]
- Wittig, J.G.; Münsterberg, A. The early stages of heart development: Insights from chicken embryos. J. Cardiovasc. Dev. Dis. 2016, 3, 12. [Google Scholar] [CrossRef]
- Dor, X.; Corone, P.; Jonhson, E. Origine de la veine pulmonaire commune, cloisonnement du sinus veineux primitif, situs des oreillettes et théorie du « bonhomme sinusal ». Arch. Mal. Cœur Vaiss. B 1987, 80, 438–498. [Google Scholar]
- Dor, X.; Corone, P. Embryologie cardiaque. Malformations (I). In Encyclopédie Médico-Chirurgicale; Editions Techniques: Paris, Francce, 1992; pp. 1–20. [Google Scholar]
- Jahr, M.; Schlueter, J.; Brand, T.; Männer, J. Development of the proepicardium in Xenopus laevis. Dev. Dyn. 2008, 237, 3088–3096. [Google Scholar] [CrossRef]
- Männer, J. Microsurgical procedures for studying the developmental significance of the proepicardium and epicardium in avian embryos: PE-blocking, PE-photoablation, and PE-grafting. J. Dev. Biol. 2013, 1, 47–63. [Google Scholar] [CrossRef]
- Sucov, H.M.; Gu, Y.; Thomas, S.; Li, P.; Pashmforoush, M. Epicardial control of myocardial proliferation and morphogenesis. Pediatr. Cardiol. 2009, 30, 617–625. [Google Scholar] [CrossRef]
- Barak, Y.; Hemberger, M.; Sucov, H.M. Phases and mechanisms of embryonic cardiomyocyte proliferation and ventricular wall morphogenesis. Pediatr. Cardiol. 2019. [Google Scholar] [CrossRef]
- Friedberg, M.K.; Ursell, P.C.; Silverman, N.H. Isomerism of the left atrial appendage associated with ventricular noncompaction. Am. J. Cardiol. 2005, 96, 985–990. [Google Scholar] [CrossRef]
- Wessels, M.W.; De Graaf, B.M.; Cohen-Overbeek, T.E.; Spitaels, S.E.; de Groot-de Laat, L.E.; Ten Cate, F.J.; Frohn-Mulder, I.F.; de Krijger, R.; Bartelings, M.M.; Essed, N.; et al. A new syndrome with noncompaction cardiomyopathy, bradycardia, pulmonary stenosis, atrial septal defect and heterotaxy with suggestive link to chromosome 6p. Hum. Genet. 2008, 122, 595–603. [Google Scholar] [CrossRef] [PubMed]
- Ursell, P.C. Noncompaction in the fetus and neonate: An autopsy study. Am. J. Med. Genet. C Semin. Med. Genet. 2013, 163C, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Bader, R.S.; Punn, R.; Silverman, N.H. Evaluation of risk factors for prediction of outcome in fetal spectrum of atrioventricular septal defects. Congenit. Heart Dis. 2014, 9, 286–293. [Google Scholar] [CrossRef] [PubMed]
- Loomba, R.S.; Willes, R.J.; Kovach, J.R.; Anderson, R.H. Chronic arrhythmias in the setting of heterotaxy: Differences between right and left isomerism. Congenit. Heart Dis. 2016, 11, 7–18. [Google Scholar] [CrossRef]
- Sucov, H.M.; Dyson, E.; Gumeringer, C.L.; Price, J.; Chien, K.R.; Evans, R.M. RXR mutant mice establish a genetic basis for vitamin A signaling in heart morphogenesis. Genes Dev. 1994, 8, 1007–1018. [Google Scholar] [CrossRef]
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Männer, J. Spontaneous Left Cardiac Isomerism in Chick Embryos: Case Report, Review of the Literature, and Possible Significance for the Understanding of Ventricular Non-Compaction Cardiomyopathy in the Setting of Human Heterotaxy Syndromes. J. Cardiovasc. Dev. Dis. 2019, 6, 40. https://doi.org/10.3390/jcdd6040040
Männer J. Spontaneous Left Cardiac Isomerism in Chick Embryos: Case Report, Review of the Literature, and Possible Significance for the Understanding of Ventricular Non-Compaction Cardiomyopathy in the Setting of Human Heterotaxy Syndromes. Journal of Cardiovascular Development and Disease. 2019; 6(4):40. https://doi.org/10.3390/jcdd6040040
Chicago/Turabian StyleMänner, Jörg. 2019. "Spontaneous Left Cardiac Isomerism in Chick Embryos: Case Report, Review of the Literature, and Possible Significance for the Understanding of Ventricular Non-Compaction Cardiomyopathy in the Setting of Human Heterotaxy Syndromes" Journal of Cardiovascular Development and Disease 6, no. 4: 40. https://doi.org/10.3390/jcdd6040040
APA StyleMänner, J. (2019). Spontaneous Left Cardiac Isomerism in Chick Embryos: Case Report, Review of the Literature, and Possible Significance for the Understanding of Ventricular Non-Compaction Cardiomyopathy in the Setting of Human Heterotaxy Syndromes. Journal of Cardiovascular Development and Disease, 6(4), 40. https://doi.org/10.3390/jcdd6040040



