Functional Significance of the Adcy10-Dependent Intracellular cAMP Compartments
Abstract
1. Introduction
2. Structure and Regulation of sAC Activity
2.1. Structure
2.2. Posttranslational Regulation of sAC
2.3. Pharmacological Regulation of sAC
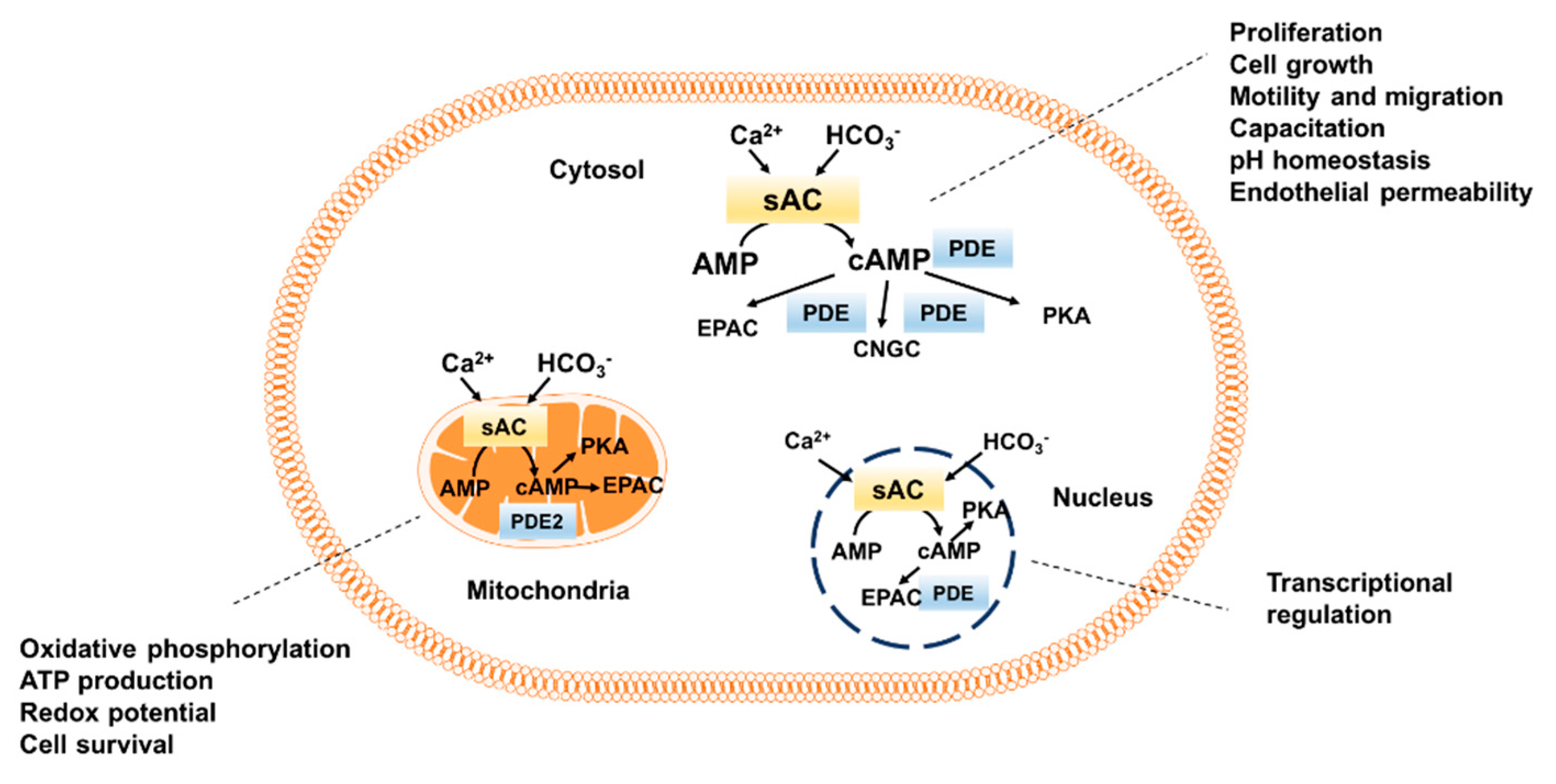
3. Functional Role of sAC in Different Cellular Compartments
3.1. Role of sAC-Dependent cAMP Signaling in Microtubules and Centrioles
3.2. Role of Cytosolic/Nuclear sAC-Dependent cAMP Signaling
3.2.1. Proliferation and Cell Growth
3.2.2. Motility
3.2.3. pH Homeostasis
3.2.4. Transcriptional Regulation
3.2.5. CFTR Regulation
3.2.6. Na+/K+-ATPas Endocytosis
3.2.7. Endothelial Permeability
3.3. Role of Mitochondrial sAC-Dependent cAMP Signaling
3.3.1. Extra-Mitochondrial sAC
3.3.2. Intra-Mitochondrial sAC
3.3.3. Intra-Mitochondrial PDE2
3.4. Importance of sAC in the Cardiovascular System
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| cAMP | 3′–5′-cyclic adenosine monophosphate |
| sAC | soluble adenylyl cyclase |
| tmAC | transmembrane adenylyl cyclase |
| PDEs | phosphodiesterases |
| PKA | protein kinase A |
| EPAC | exchange protein directly activated by cAMP |
| CREB | cAMP-response element binding protein |
| CE | Catechol estrogens |
| GC | guanylyl cyclase |
| AKAP | A-kinase anchoring proteins |
| CFTR | cystic fibrosis transmembrane conductance regulator |
| V-ATPase | vacuolar H+-ATPase |
| IMS | intra-mitochondrial space |
| OMM | outer mitochondrial membrane |
| IMM | inner mitochondrial membrane |
| NTPase | nucleoside-triphosphatase |
| OXPHOS | oxidative phosphorylation |
References
- Wu, Z.; Huang, X.; Feng, Y.; Handschin, C.; Feng, Y.; Gullicksen, P.S.; Bare, O.; Labow, M.; Spiegelman, B.; Stevenson, S.C. Transducer of regulated CREB-binding proteins (TORCs) induce PGC-1α transcription and mitochondrial biogenesis in muscle cells. Proc. Natl. Acad. Sci. USA 2006, 103, 14379–14384. [Google Scholar] [CrossRef] [PubMed]
- Chowanadisai, W.; Bauerly, K.A.; Tchaparian, E.; Wong, A.; Cortopassi, G.A.; Rucker, R.B. Pyrroloquinoline quinone stimulates mitochondrial biogenesis through cAMP response element-binding protein phosphorylation and increased PGC-1α expression. J. Biol. Chem. 2010, 285, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Catterall, W.A. Regulation of cardiac calcium channels in the fight-or-flight response. Curr. Mol. Pharmacol. 2015, 8, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Kashina, A.S.; Semenova, I.V.; Ivanov, P.A.; Potekhina, E.S.; Zaliapin, I.; Rodionov, V.I. Protein kinase A, which regulates intracellular transport, forms complexes with molecular motors on organelles. Curr. Biol. 2004, 14, 1877–1881. [Google Scholar] [CrossRef] [PubMed]
- Burdyga, A.; Conant, A.; Haynes, L.; Zhang, J.; Jalink, K.; Sutton, R.; Neoptolemos, J.; Costello, E.; Tepikin, A. cAMP inhibits migration, ruffling and paxillin accumulation in focal adhesions of pancreatic ductal adenocarcinoma cells: Effects of PKA and EPAC. Biochim. Biophys. Acta 2013, 1833, 2664–2672. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, N.P.; Roy, I.; Hauser, A.D.; Wilson, J.M.; Williams, C.L.; Dwinell, M.B. Cyclic AMP regulates the migration and invasion potential of human pancreatic cancer cells. Mol. Carcinog. 2015, 54, 203–215. [Google Scholar] [CrossRef] [PubMed]
- Acin-Perez, R.; Salazar, E.; Brosel, S.; Yang, H.; Schon, E.A.; Manfredi, G. Modulation of mitochondrial protein phosphorylation by soluble adenylyl cyclase ameliorates cytochrome oxidase defects. EMBO Mol. Med. 2009, 1, 392–406. [Google Scholar] [CrossRef] [PubMed]
- Acin-Perez, R.; Salazar, E.; Kamenetsky, M.; Buck, J.; Levin, L.R.; Manfredi, G. Cyclic AMP produced inside mitochondria regulates oxidative phosphorylation. Cell Metab. 2009, 9, 265–276. [Google Scholar] [CrossRef] [PubMed]
- Di Benedetto, G.; Scalzotto, E.; Mongillo, M.; Pozzan, T. Mitochondrial Ca2+ uptake induces cyclic AMP generation in the matrix and modulates organelle ATP levels. Cell Metab. 2013, 17, 965–975. [Google Scholar] [CrossRef] [PubMed]
- Valsecchi, F.; Ramos-Espiritu, L.S.; Buck, J.; Levin, L.R.; Manfredi, G. cAMP and mitochondria. Physiology 2013, 28, 199–209. [Google Scholar] [CrossRef] [PubMed]
- Signorile, A.; Santeramo, A.; Tamma, G.; Pellegrino, T.; D’Oria, S.; Lattanzio, P.; De Rasmo, D. Mitochondrial cAMP prevents apoptosis modulating Sirt3 protein level and OPA1 processing in cardiac myoblast cells. Biochim. Biophys. Acta 2017, 1864, 355–366. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Zhang, L.; Qi, Y.; Xu, H. Mitochondrial cAMP signaling. Cell. Mol. Life Sci. 2016, 73, 4577–4590. [Google Scholar] [CrossRef] [PubMed]
- Bacallao, K.; Monje, P.V. Opposing roles of PKA and EPAC in the cAMP-dependent regulation of schwann cell proliferation and differentiation [corrected]. PLoS ONE 2013, 8, e82354. [Google Scholar] [CrossRef] [PubMed]
- Stork, P.J.; Schmitt, J.M. Crosstalk between cAMP and MAP kinase signaling in the regulation of cell proliferation. Trends Cell Biol. 2002, 12, 258–266. [Google Scholar] [CrossRef]
- Appukuttan, A.; Kasseckert, S.A.; Micoogullari, M.; Flacke, J.P.; Kumar, S.; Woste, A.; Abdallah, Y.; Pott, L.; Reusch, H.P.; Ladilov, Y. Type 10 adenylyl cyclase mediates mitochondrial bax translocation and apoptosis of adult rat cardiomyocytes under simulated ischaemia/reperfusion. Cardiovasc. Res. 2012, 93, 340–349. [Google Scholar] [CrossRef] [PubMed]
- Ladilov, Y.; Appukuttan, A. Role of soluble adenylyl cyclase in cell death and growth. Biochim. Biophys. Acta 2014, 1842, 2646–2655. [Google Scholar] [CrossRef] [PubMed]
- Ohmori, M.; Okamoto, S. Photoresponsive cAMP signal transduction in cyanobacteria. Photochem. Photobiol. Sci. 2004, 3, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Knapp, G.S.; McDonough, K.A. Cyclic amp signaling in mycobacteria. In Molecular Genetics of Mycobacteria, 2nd ed.; American Society of Microbiology: Washington, DC, USA, 2014; pp. 281–295. [Google Scholar]
- Kamenetsky, M.; Middelhaufe, S.; Bank, E.M.; Levin, L.R.; Buck, J.; Steegborn, C. Molecular details of cAMP generation in mammalian cells: A tale of two systems. J. Mol. Biol. 2006, 362, 623–639. [Google Scholar] [CrossRef] [PubMed]
- Federman, A.D.; Conklin, B.R.; Schrader, K.A.; Reed, R.R.; Bourne, H.R. Hormonal stimulation of adenylyl cyclase through Gi-protein βγ subunits. Nature 1992, 356, 159–161. [Google Scholar] [CrossRef] [PubMed]
- Dessauer, C.W.; Watts, V.J.; Ostrom, R.S.; Conti, M.; Dove, S.; Seifert, R. International union of basic and clinical pharmacology. Ci. Structures and small molecule modulators of mammalian adenylyl cyclases. Pharmacol. Rev. 2017, 69, 93–139. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, T.A.; Dessauer, C.W. Function of adenylyl cyclase in heart: The AKAP connection. J. Cardiovasc. Dev. Dis. 2018, 5, 2. [Google Scholar] [CrossRef] [PubMed]
- Ostrom, R.S.; Naugle, J.E.; Hase, M.; Gregorian, C.; Swaney, J.S.; Insel, P.A.; Brunton, L.L.; Meszaros, J.G. Angiotensin ii enhances adenylyl cyclase signaling via Ca2+/calmodulin. Gq-Gs cross-talk regulates collagen production in cardiac fibroblasts. J. Biol. Chem. 2003, 278, 24461–24468. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Cann, M.J.; Litvin, T.N.; Iourgenko, V.; Sinclair, M.L.; Levin, L.R.; Buck, J. Soluble adenylyl cyclase as an evolutionarily conserved bicarbonate sensor. Science 2000, 289, 625–628. [Google Scholar] [CrossRef] [PubMed]
- Zippin, J.H.; Chen, Y.; Nahirney, P.; Kamenetsky, M.; Wuttke, M.S.; Fischman, D.A.; Levin, L.R.; Buck, J. Compartmentalization of bicarbonate-sensitive adenylyl cyclase in distinct signaling microdomains. FASEB J. 2003, 17, 82–84. [Google Scholar] [CrossRef] [PubMed]
- Richards, M.; Lomas, O.; Jalink, K.; Ford, K.L.; Vaughan-Jones, R.D.; Lefkimmiatis, K.; Swietach, P. Intracellular tortuosity underlies slow cAMP diffusion in adult ventricular myocytes. Cardiovasc. Res. 2016, 110, 395–407. [Google Scholar] [CrossRef] [PubMed]
- Mika, D.; Leroy, J.; Vandecasteele, G.; Fischmeister, R. Pdes create local domains of cAMP signaling. J. Mol. Cell. Cardiol. 2012, 52, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, S.R.; Clancy, C.E.; Harvey, R.D. Mechanisms restricting diffusion of intracellular cAMP. Sci. Rep. 2016, 6, 19577. [Google Scholar] [CrossRef] [PubMed]
- Fischmeister, R.; Castro, L.R.; Abi-Gerges, A.; Rochais, F.; Jurevicius, J.; Leroy, J.; Vandecasteele, G. Compartmentation of cyclic nucleotide signaling in the heart: The role of cyclic nucleotide phosphodiesterases. Circ. Res. 2006, 99, 816–828. [Google Scholar] [CrossRef] [PubMed]
- Godbole, A.; Lyga, S.; Lohse, M.J.; Calebiro, D. Internalized tsh receptors en route to the tgn induce local Gs-protein signaling and gene transcription. Nat. Commun. 2017, 8, 443. [Google Scholar] [CrossRef] [PubMed]
- Calebiro, D.; Maiellaro, I. CAMP signaling microdomains and their observation by optical methods. Front. Cell. Neurosci. 2014, 8, 350. [Google Scholar] [CrossRef] [PubMed]
- Musheshe, N.; Schmidt, M.; Zaccolo, M. cAMP: From long-range second messenger to nanodomain signalling. Trends Pharmacol. Sci. 2017, 39, 209–222. [Google Scholar] [CrossRef] [PubMed]
- Lefkimmiatis, K.; Zaccolo, M. cAMP signaling in subcellular compartments. Pharmacol. Ther. 2014, 143, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Buck, J.; Levin, L.R. Conservation of functional domain structure in bicarbonate-regulated “soluble” adenylyl cyclases in bacteria and eukaryotes. Dev. Genes Evol. 2004, 214, 503–509. [Google Scholar] [CrossRef] [PubMed]
- Kleinboelting, S.; Diaz, A.; Moniot, S.; van den Heuvel, J.; Weyand, M.; Levin, L.R.; Buck, J.; Steegborn, C. Crystal structures of human soluble adenylyl cyclase reveal mechanisms of catalysis and of its activation through bicarbonate. Proc. Natl. Acad. Sci. USA 2014, 111, 3727–3732. [Google Scholar] [CrossRef] [PubMed]
- Chaloupka, J.A.; Bullock, S.A.; Iourgenko, V.; Levin, L.R.; Buck, J. Autoinhibitory regulation of soluble adenylyl cyclase. Mol. Reprod. Dev. 2006, 73, 361–368. [Google Scholar] [CrossRef] [PubMed]
- Leipe, D.D.; Koonin, E.V.; Aravind, L. Stand, a class of p-loop ntpases including animal and plant regulators of programmed cell death: Multiple, complex domain architectures, unusual phyletic patterns, and evolution by horizontal gene transfer. J. Mol. Biol. 2004, 343, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Middelhaufe, S.; Leipelt, M.; Levin, L.R.; Buck, J.; Steegborn, C. Identification of a haem domain in human soluble adenylate cyclase. Biosci. Rep. 2012, 32, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Farrell, J.; Ramos, L.; Tresguerres, M.; Kamenetsky, M.; Levin, L.R.; Buck, J. Somatic ‘soluble’ adenylyl cyclase isoforms are unaffected in Sacytm1lex/Sacytm1lex ‘knockout’ mice. PLoS ONE 2008, 3, e3251. [Google Scholar] [CrossRef] [PubMed]
- Reed, B.Y.; Gitomer, W.L.; Heller, H.J.; Hsu, M.C.; Lemke, M.; Padalino, P.; Pak, C.Y. Identification and characterization of a gene with base substitutions associated with the absorptive hypercalciuria phenotype and low spinal bone density. J. Clin. Endocrinol. Metab. 2002, 87, 1476–1485. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Baumlin, N.; Buck, J.; Levin, L.R.; Fregien, N.; Salathe, M. A soluble adenylyl cyclase form targets to axonemes and rescues beat regulation in soluble adenylyl cyclase knockout mice. Am. J. Respir. Cell Mol. Biol. 2014, 51, 750–760. [Google Scholar] [CrossRef] [PubMed]
- Buck, J.; Sinclair, M.L.; Schapal, L.; Cann, M.J.; Levin, L.R. Cytosolic adenylyl cyclase defines a unique signaling molecule in mammals. Proc. Natl. Acad. Sci. USA 1999, 96, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Geng, W.; Wang, Z.; Zhang, J.; Reed, B.Y.; Pak, C.Y.; Moe, O.W. Cloning and characterization of the human soluble adenylyl cyclase. Am. J. Physiol. Cell Physiol. 2005, 288, C1305–C1316. [Google Scholar] [CrossRef] [PubMed]
- Steegborn, C. Structure, mechanism, and regulation of soluble adenylyl cyclases—Similarities and differences to transmembrane adenylyl cyclases. Biochim. Biophys. Acta 2014, 1842, 2535–2547. [Google Scholar] [CrossRef] [PubMed]
- Hebert-Chatelain, E.; Desprez, T.; Serrat, R.; Bellocchio, L.; Soria-Gomez, E.; Busquets-Garcia, A.; Pagano Zottola, A.C.; Delamarre, A.; Cannich, A.; Vincent, P.; et al. A cannabinoid link between mitochondria and memory. Nature 2016, 539, 555–559. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Espiritu, L.; Kleinboelting, S.; Navarrete, F.A.; Alvau, A.; Visconti, P.E.; Valsecchi, F.; Starkov, A.; Manfredi, G.; Buck, H.; Adura, C.; et al. Discovery of LRE1 as a specific and allosteric inhibitor of soluble adenylyl cyclase. Nat. Chem. Biol. 2016, 12, 838–844. [Google Scholar] [CrossRef] [PubMed]
- Tresguerres, M.; Levin, L.R.; Buck, J. Intracellular cAMP signaling by soluble adenylyl cyclase. Kidney Int. 2011, 79, 1277–1288. [Google Scholar] [CrossRef] [PubMed]
- Zippin, J.H.; Chen, Y.; Straub, S.G.; Hess, K.C.; Diaz, A.; Lee, D.; Tso, P.; Holz, G.G.; Sharp, G.W.; Levin, L.R.; et al. CO2/HCO3−- and calcium-regulated soluble adenylyl cyclase as a physiological ATP sensor. J. Biol. Chem. 2013, 288, 33283–33291. [Google Scholar] [CrossRef] [PubMed]
- Litvin, T.N.; Kamenetsky, M.; Zarifyan, A.; Buck, J.; Levin, L.R. Kinetic properties of “soluble” adenylyl cyclase. Synergism between calcium and bicarbonate. J. Biol. Chem. 2003, 278, 15922–15926. [Google Scholar] [CrossRef] [PubMed]
- Magro, C.M.; Yang, S.E.; Zippin, J.H.; Zembowicz, A. Expression of soluble adenylyl cyclase in lentigo maligna: Use of immunohistochemistry with anti-soluble adenylyl cyclase antibody (R21) in diagnosis of lentigo maligna and assessment of margins. Arch. Pathol. Lab. Med. 2012, 136, 1558–1564. [Google Scholar] [CrossRef] [PubMed]
- Magro, C.M.; Crowson, A.N.; Desman, G.; Zippin, J.H. Soluble adenylyl cyclase antibody profile as a diagnostic adjunct in the assessment of pigmented lesions. Arch. Dermatol. 2012, 148, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Ramos, L.S.; Zippin, J.H.; Kamenetsky, M.; Buck, J.; Levin, L.R. Glucose and GLP-1 stimulate cAMP production via distinct adenylyl cyclases in INS-1E insulinoma cells. J. Gen. Physiol. 2008, 132, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Flacke, J.P.; Flacke, H.; Appukuttan, A.; Palisaar, R.J.; Noldus, J.; Robinson, B.D.; Reusch, H.P.; Zippin, J.H.; Ladilov, Y. Type 10 soluble adenylyl cyclase is overexpressed in prostate carcinoma and controls proliferation of prostate cancer cells. J. Biol. Chem. 2013, 288, 3126–3135. [Google Scholar] [CrossRef] [PubMed]
- Pierre, S.; Eschenhagen, T.; Geisslinger, G.; Scholich, K. Capturing adenylyl cyclases as potential drug targets. Nat. Rev. Drug Discov. 2009, 8, 321–335. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.S.; Tresguerres, M.; Hess, K.; Marmorstein, L.Y.; Levin, L.R.; Buck, J.; Marmorstein, A.D. Regulation of anterior chamber drainage by bicarbonate-sensitive soluble adenylyl cyclase in the ciliary body. J. Biol. Chem. 2011, 286, 41353–41358. [Google Scholar] [CrossRef] [PubMed]
- Bitterman, J.L.; Ramos-Espiritu, L.; Diaz, A.; Levin, L.R.; Buck, J. Pharmacological distinction between soluble and transmembrane adenylyl cyclases. J. Pharmacol. Exp. Ther. 2013, 347, 589–598. [Google Scholar] [CrossRef] [PubMed]
- Steegborn, C.; Litvin, T.N.; Hess, K.C.; Capper, A.B.; Taussig, R.; Buck, J.; Levin, L.R.; Wu, H. A novel mechanism for adenylyl cyclase inhibition from the crystal structure of its complex with catechol estrogen. J. Biol. Chem. 2005, 280, 31754–31759. [Google Scholar] [CrossRef] [PubMed]
- Hess, K.C.; Jones, B.H.; Marquez, B.; Chen, Y.; Ord, T.S.; Kamenetsky, M.; Miyamoto, C.; Zippin, J.H.; Kopf, G.S.; Suarez, S.S.; et al. The “soluble” adenylyl cyclase in sperm mediates multiple signaling events required for fertilization. Dev. Cell 2005, 9, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Kaupp, U.B.; Seifert, R. Cyclic nucleotide-gated ion channels. Physiol. Rev. 2002, 82, 769–824. [Google Scholar] [CrossRef] [PubMed]
- Froese, A.; Breher, S.S.; Waldeyer, C.; Schindler, R.F.; Nikolaev, V.O.; Rinne, S.; Wischmeyer, E.; Schlueter, J.; Becher, J.; Simrick, S.; et al. Popeye domain containing proteins are essential for stress-mediated modulation of cardiac pacemaking in mice. J. Clin. Investig. 2012, 122, 1119–1130. [Google Scholar] [CrossRef] [PubMed]
- Simrick, S.; Schindler, R.F.; Poon, K.L.; Brand, T. Popeye domain-containing proteins and stress-mediated modulation of cardiac pacemaking. Trends Cardiovasc. Med. 2013, 23, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Dekker, F.J.; Maarsingh, H. Exchange protein directly activated by cAMP (EPAC): A multidomain cAMP mediator in the regulation of diverse biological functions. Pharmacol. Rev. 2013, 65, 670–709. [Google Scholar] [CrossRef] [PubMed]
- Lomas, O.; Zaccolo, M. Phosphodiesterases maintain signaling fidelity via compartmentalization of cyclic nucleotides. Physiology 2014, 29, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Corredor, R.G.; Trakhtenberg, E.F.; Pita-Thomas, W.; Jin, X.; Hu, Y.; Goldberg, J.L. Soluble adenylyl cyclase activity is necessary for retinal ganglion cell survival and axon growth. J. Neurosci. 2012, 32, 7734–7744. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; Stessin, A.; Roberts, J.; Hess, K.; Gautam, N.; Kamenetsky, M.; Lou, O.; Hyde, E.; Nathan, N.; Muller, W.A.; et al. Calcium-sensing soluble adenylyl cyclase mediates tnf signal transduction in human neutrophils. J. Exp. Med. 2005, 202, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Stowe, D.F.; Gadicherla, A.K.; Zhou, Y.; Aldakkak, M.; Cheng, Q.; Kwok, W.M.; Jiang, M.T.; Heisner, J.S.; Yang, M.; Camara, A.K. Protection against cardiac injury by small Ca2+-sensitive K+ channels identified in guinea pig cardiac inner mitochondrial membrane. Biochim. Biophys. Acta 2013, 1828, 427–442. [Google Scholar] [CrossRef] [PubMed]
- Monterisi, S.; Zaccolo, M. Components of the mitochondrial cAMP signalosome. Biochem. Soc. Trans. 2017, 45, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Zippin, J.H.; Farrell, J.; Huron, D.; Kamenetsky, M.; Hess, K.C.; Fischman, D.A.; Levin, L.R.; Buck, J. Bicarbonate-responsive “soluble” adenylyl cyclase defines a nuclear cAMP microdomain. J. Cell Biol. 2004, 164, 527–534. [Google Scholar] [CrossRef] [PubMed]
- Stessin, A.M.; Zippin, J.H.; Kamenetsky, M.; Hess, K.C.; Buck, J.; Levin, L.R. Soluble adenylyl cyclase mediates nerve growth factor-induced activation of Rap1. J. Biol. Chem. 2006, 281, 17253–17258. [Google Scholar] [CrossRef] [PubMed]
- Onodera, Y.; Nam, J.M.; Bissell, M.J. Increased sugar uptake promotes oncogenesis via EPAC/Rap1 and o-glcnac pathways. J. Clin. Investig. 2014, 124, 367–384. [Google Scholar] [CrossRef] [PubMed]
- Wu, K.Y.; Zippin, J.H.; Huron, D.R.; Kamenetsky, M.; Hengst, U.; Buck, J.; Levin, L.R.; Jaffrey, S.R. Soluble adenylyl cyclase is required for netrin-1 signaling in nerve growth cones. Nat. Neurosci. 2006, 9, 1257–1264. [Google Scholar] [CrossRef] [PubMed]
- Schirmer, I.; Bualeong, T.; Budde, H.; Cimiotti, D.; Appukuttan, A.; Klein, N.; Steinwascher, P.; Reusch, P.; Mugge, A.; Meyer, R.; et al. Soluble adenylyl cyclase: A novel player in cardiac hypertrophy induced by isoprenaline or pressure overload. PLoS ONE 2018, 13, e0192322. [Google Scholar] [CrossRef] [PubMed]
- Buffone, M.G.; Wertheimer, E.V.; Visconti, P.E.; Krapf, D. Central role of soluble adenylyl cyclase and cAMP in sperm physiology. Biochim. Biophys. Acta 2014, 1842, 2610–2620. [Google Scholar] [CrossRef] [PubMed]
- Esposito, G.; Jaiswal, B.S.; Xie, F.; Krajnc-Franken, M.A.; Robben, T.J.; Strik, A.M.; Kuil, C.; Philipsen, R.L.; van Duin, M.; Conti, M.; et al. Mice deficient for soluble adenylyl cyclase are infertile because of a severe sperm-motility defect. Proc. Natl. Acad. Sci. USA 2004, 101, 2993–2998. [Google Scholar] [CrossRef] [PubMed]
- Watson, R.L.; Buck, J.; Levin, L.R.; Winger, R.C.; Wang, J.; Arase, H.; Muller, W.A. Endothelial cd99 signals through soluble adenylyl cyclase and PKA to regulate leukocyte transendothelial migration. J. Exp. Med. 2015, 212, 1021–1041. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.C.; Oude-Elferink, R.P. Role of the bicarbonate-responsive soluble adenylyl cyclase in pH sensing and metabolic regulation. Front. Physiol. 2014, 5, 42. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.; Bouley, R.; Paunescu, T.G.; Breton, S.; Lu, H.A. New insights into the dynamic regulation of water and acid-base balance by renal epithelial cells. Am. J. Physiol. Cell Physiol. 2012, 302, C1421–C1433. [Google Scholar] [CrossRef] [PubMed]
- Pastor-Soler, N.M.; Hallows, K.R.; Smolak, C.; Gong, F.; Brown, D.; Breton, S. Alkaline ph- and cAMP-induced v-ATPase membrane accumulation is mediated by protein kinase a in epididymal clear cells. Am. J. Physiol. Cell Physiol. 2008, 294, C488–C494. [Google Scholar] [CrossRef] [PubMed]
- Gong, F.; Alzamora, R.; Smolak, C.; Li, H.; Naveed, S.; Neumann, D.; Hallows, K.R.; Pastor-Soler, N.M. Vacuolar h+-atpase apical accumulation in kidney intercalated cells is regulated by PKA and AMP-activated protein kinase. Am. J. Physiol.-Renal Physiol. 2010, 298, F1162–F1169. [Google Scholar] [CrossRef] [PubMed]
- Breton, S.; Brown, D. Regulation of luminal acidification by the v-atpase. Physiology 2013, 28, 318–329. [Google Scholar] [CrossRef] [PubMed]
- Rahman, N.; Ramos-Espiritu, L.; Milner, T.A.; Buck, J.; Levin, L.R. Soluble adenylyl cyclase is essential for proper lysosomal acidification. J. Gen. Physiol. 2016, 148, 325–339. [Google Scholar] [CrossRef] [PubMed]
- Inda, C.; Bonfiglio, J.J.; Dos Santos Claro, P.A.; Senin, S.A.; Armando, N.G.; Deussing, J.M.; Silberstein, S. cAMP-dependent cell differentiation triggered by activated crhr1 in hippocampal neuronal cells. Sci. Rep. 2017, 7, 1944. [Google Scholar] [CrossRef] [PubMed]
- Mewes, M.; Nedele, J.; Schelleckes, K.; Bondareva, O.; Lenders, M.; Kusche-Vihrog, K.; Schnittler, H.J.; Brand, S.M.; Schmitz, B.; Brand, E. Salt-induced Na+/K+-atpase-alpha/beta expression involves soluble adenylyl cyclase in endothelial cells. Pflugers Arch. Eur. J. Physiol. 2017, 469, 1401–1412. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, B.; Nedele, J.; Guske, K.; Maase, M.; Lenders, M.; Schelleckes, M.; Kusche-Vihrog, K.; Brand, S.M.; Brand, E. Soluble adenylyl cyclase in vascular endothelium: Gene expression control of epithelial sodium channel-alpha, Na+/K+-ATPase-α/β, and mineralocorticoid receptor. Hypertension 2014, 63, 753–761. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, R.; Tsuji, T.; Watanabe, O.; Yamaguchi, K.; Furukawa, K.; Nakamura, H.; Aoshiba, K. Hypercapnia accelerates adipogenesis: A novel role of high CO2 in exacerbating obesity. Am. J. Respir. Cell Mol. Biol. 2017, 57, 570–580. [Google Scholar] [CrossRef] [PubMed]
- Sepp, M.; Vihma, H.; Nurm, K.; Urb, M.; Page, S.C.; Roots, K.; Hark, A.; Maher, B.J.; Pruunsild, P.; Timmusk, T. The intellectual disability and schizophrenia associated transcription factor tcf4 is regulated by neuronal activity and protein kinase a. J. Neurosci. 2017, 37, 10516–10527. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.H.; Legere, E.A.; Snider, J.; Stagljar, I. Recent progress in cftr interactome mapping and its importance for cystic fibrosis. Front. Pharmacol. 2017, 8, 997. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Lam, C.S.; Wu, F.; Wang, W.; Duan, Y.; Huang, P. Regulation of cftr channels by HCO3—Sensitive soluble adenylyl cyclase in human airway epithelial cells. Am. J. Physiol. Cell Physiol. 2005, 289, C1145–C1151. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Guo, J.H.; Lu, Y.C.; Ding, G.L.; Yu, M.K.; Tsang, L.L.; Fok, K.L.; Liu, X.M.; Zhang, X.H.; Chung, Y.W.; et al. Impaired cftr-dependent amplification of fsh-stimulated estrogen production in cystic fibrosis and pcos. J. Clin. Endocrinol. Metab. 2012, 97, 923–932. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.F.; Zhou, C.X.; Shi, Q.X.; Yuan, Y.Y.; Yu, M.K.; Ajonuma, L.C.; Ho, L.S.; Lo, P.S.; Tsang, L.L.; Liu, Y.; et al. Involvement of cftr in uterine bicarbonate secretion and the fertilizing capacity of sperm. Nat. Cell Biol. 2003, 5, 902–906. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.M.; Shi, Q.X.; Chen, W.Y.; Zhou, C.X.; Ni, Y.; Rowlands, D.K.; Yi Liu, G.; Zhu, H.; Ma, Z.G.; Wang, X.F.; et al. Cystic fibrosis transmembrane conductance regulator is vital to sperm fertilizing capacity and male fertility. Proc. Natl. Acad. Sci. USA 2007, 104, 9816–9821. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.Y.; Xu, W.M.; Chen, Z.H.; Ni, Y.; Yuan, Y.Y.; Zhou, S.C.; Zhou, W.W.; Tsang, L.L.; Chung, Y.W.; Hoglund, P.; et al. Cl- is required for HCO3− entry necessary for sperm capacitation in guinea pig: Involvement of a Cl-/HCO3− exchanger (slc26a3) and cftr. Biol. Reprod. 2009, 80, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.C.; Chen, H.; Fok, K.L.; Tsang, L.L.; Yu, M.K.; Zhang, X.H.; Chen, J.; Jiang, X.; Chung, Y.W.; Ma, A.C.; et al. Cftr mediates bicarbonate-dependent activation of mir-125b in preimplantation embryo development. Cell Res. 2012, 22, 1453–1466. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Chan, H.C. Amplification of fsh signalling by cftr and nuclear soluble adenylyl cyclase in the ovary. Clin. Exp. Pharmacol. Physiol. 2017, 44 (Suppl. S1), 78–85. [Google Scholar] [CrossRef] [PubMed]
- Lecuona, E.; Sun, H.; Chen, J.; Trejo, H.E.; Baker, M.A.; Sznajder, J.I. Protein kinase a-ialpha regulates na,k-atpase endocytosis in alveolar epithelial cells exposed to high CO2 concentrations. Am. J. Respir. Cell Mol. Biol. 2013, 48, 626–634. [Google Scholar] [CrossRef] [PubMed]
- Sayner, S.L.; Frank, D.W.; King, J.; Chen, H.; VandeWaa, J.; Stevens, T. Paradoxical cAMP-induced lung endothelial hyperpermeability revealed by pseudomonas aeruginosa exoy. Circ. Res. 2004, 95, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Sayner, S.L.; Alexeyev, M.; Dessauer, C.W.; Stevens, T. Soluble adenylyl cyclase reveals the significance of cAMP compartmentation on pulmonary microvascular endothelial cell barrier. Circ. Res. 2006, 98, 675–681. [Google Scholar] [CrossRef] [PubMed]
- Das, A.; Härtel, F.; Ihle, K.; Ladilov, Y.; Noll, T. Role of soluble adenylyl cyclase on barrier function of human umbilical vein endothelial monolayers: P21–13. Acta Physiol. 2016, 216, 216. [Google Scholar]
- Di Benedetto, G.; Gerbino, A.; Lefkimmiatis, K. Shaping mitochondrial dynamics: The role of cAMP signalling. Biochem. Biophys. Res. Commun. 2017, 500, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Jayarajan, V.; Appukuttan, A.; Reusch, P.; Ladilov, Y.; Regitz-Zagrosek, V. Soluble adenylyl cyclase controls AMPK activity, mitochondrial function and biogensis and may play a role in estradiol-dependent protection against oxidative stress. In Proceedings of the 42nd FEBS Congress on Molecules to Cells and back, Jerusalim, Israel, 10–14 September 2017. [Google Scholar]
- Kumar, S.; Kostin, S.; Flacke, J.P.; Reusch, H.P.; Ladilov, Y. Soluble adenylyl cyclase controls mitochondria-dependent apoptosis in coronary endothelial cells. J. Biol. Chem. 2009, 284, 14760–14768. [Google Scholar] [CrossRef] [PubMed]
- Appukuttan, A.; Kasseckert, S.A.; Kumar, S.; Reusch, H.P.; Ladilov, Y. Oxysterol-induced apoptosis of smooth muscle cells is under the control of a soluble adenylyl cyclase. Cardiovasc. Res. 2013, 99, 734–742. [Google Scholar] [CrossRef] [PubMed]
- Acin-Perez, R.; Russwurm, M.; Gunnewig, K.; Gertz, M.; Zoidl, G.; Ramos, L.; Buck, J.; Levin, L.R.; Rassow, J.; Manfredi, G.; et al. A phosphodiesterase 2a isoform localized to mitochondria regulates respiration. J. Biol. Chem. 2011, 286, 30423–30432. [Google Scholar] [CrossRef] [PubMed]
- Hess, K.C.; Liu, J.; Manfredi, G.; Muhlschlegel, F.A.; Buck, J.; Levin, L.R.; Barrientos, A. A mitochondrial CO2-adenylyl cyclase-cAMP signalosome controls yeast normoxic cytochrome c oxidase activity. FASEB J. 2014, 28, 4369–4380. [Google Scholar] [CrossRef] [PubMed]
- DiPilato, L.M.; Cheng, X.; Zhang, J. Fluorescent indicators of cAMP and EPAC activation reveal differential dynamics of cAMP signaling within discrete subcellular compartments. Proc. Natl. Acad. Sci. USA 2004, 101, 16513–16518. [Google Scholar] [CrossRef] [PubMed]
- Pozdniakova, S.; Guitart-Mampel, M.; Garrabou, G.; Di Benedetto, G.; Ladilov, Y.; Regitz-Zagrosek, V. 17ß-estradiol reduces mitochondrial cAMP content and cytochrome oxidase activity in a phosphodiesterase 2-dependent manner. In Proceedings of the 42nd FEBS Congress on Molecules to Cells and back, Jerusalim, Israel, 10–14 September 2017. [Google Scholar]
- Wang, Z.; Liu, D.; Varin, A.; Nicolas, V.; Courilleau, D.; Mateo, P.; Caubere, C.; Rouet, P.; Gomez, A.M.; Vandecasteele, G.; et al. A cardiac mitochondrial cAMP signaling pathway regulates calcium accumulation, permeability transition and cell death. Cell Death Dis. 2016, 7, e2198. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.; Jansen, V.; Jikeli, J.F.; Hamzeh, H.; Alvarez, L.; Dombrowski, M.; Balbach, M.; Strunker, T.; Seifert, R.; Kaupp, U.B.; et al. A novel biosensor to study cAMP dynamics in cilia and flagella. eLife 2016, 5, e14052. [Google Scholar] [CrossRef] [PubMed]
- Acin-Perez, R.; Gatti, D.L.; Bai, Y.; Manfredi, G. Protein phosphorylation and prevention of cytochrome oxidase inhibition by atp: Coupled mechanisms of energy metabolism regulation. Cell Metab. 2011, 13, 712–719. [Google Scholar] [CrossRef] [PubMed]
- Valsecchi, F.; Konrad, C.; D’Aurelio, M.; Ramos-Espiritu, L.S.; Stepanova, A.; Burstein, S.R.; Galkin, A.; Magrane, J.; Starkov, A.; Buck, J.; et al. Distinct intracellular sac-cAMP domains regulate er Ca2+ signaling and oxphos function. J. Cell Sci. 2017, 130, 3713–3727. [Google Scholar] [CrossRef]
- De Rasmo, D.; Signorile, A.; Santeramo, A.; Larizza, M.; Lattanzio, P.; Capitanio, G.; Papa, S. Intramitochondrial adenylyl cyclase controls the turnover of nuclear-encoded subunits and activity of mammalian complex i of the respiratory chain. Biochim. Biophys. Acta 2015, 1853, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Sardanelli, A.M.; Signorile, A.; Nuzzi, R.; Rasmo, D.D.; Technikova-Dobrova, Z.; Drahota, Z.; Occhiello, A.; Pica, A.; Papa, S. Occurrence of a-kinase anchor protein and associated cAMP-dependent protein kinase in the inner compartment of mammalian mitochondria. FEBS Lett. 2006, 580, 5690–5696. [Google Scholar] [CrossRef] [PubMed]
- Agnes, R.S.; Jernigan, F.; Shell, J.R.; Sharma, V.; Lawrence, D.S. Suborganelle sensing of mitochondrial cAMP-dependent protein kinase activity. J. Am. Chem. Soc. 2010, 132, 6075–6080. [Google Scholar] [CrossRef] [PubMed]
- Valsecchi, F.; Konrad, C.; Manfredi, G. Role of soluble adenylyl cyclase in mitochondria. Biochim. Biophys. Acta 2014, 1842, 2555–2560. [Google Scholar] [CrossRef] [PubMed]
- Lefkimmiatis, K.; Leronni, D.; Hofer, A.M. The inner and outer compartments of mitochondria are sites of distinct cAMP/PKA signaling dynamics. J. Cell Biol. 2013, 202, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Covian, R.; French, S.; Kusnetz, H.; Balaban, R.S. Stimulation of oxidative phosphorylation by calcium in cardiac mitochondria is not influenced by cAMP and PKA activity. Biochim. Biophys. Acta 2014, 1837, 1913–1921. [Google Scholar] [CrossRef] [PubMed]
- Laudette, M.; Zuo, H.; Lezoualc’h, F.; Schmidt, M. EPAC function and cAMP scaffolds in the heart and lung. J. Cardiovasc. Dev. Dis. 2018, 5, 9. [Google Scholar] [CrossRef] [PubMed]
- De Rasmo, D.; Micelli, L.; Santeramo, A.; Signorile, A.; Lattanzio, P.; Papa, S. CAMP regulates the functional activity, coupling efficiency and structural organization of mammalian fof1 atp synthase. Biochim. Biophys. Acta 2016, 1857, 350–358. [Google Scholar] [CrossRef] [PubMed]
- Fazal, L.; Laudette, M.; Paula-Gomes, S.; Pons, S.; Conte, C.; Tortosa, F.; Sicard, P.; Sainte-Marie, Y.; Bisserier, M.; Lairez, O.; et al. Multifunctional mitochondrial EPAC1 controls myocardial cell death. Circ. Res. 2017, 120, 645–657. [Google Scholar] [CrossRef] [PubMed]
- Pavlaki, N.; Nikolaev, V.O. Imaging of pde2- and pde3-mediated cgmp-to-cAMP cross-talk in cardiomyocytes. J. Cardiovasc. Dev. Dis. 2018, 5, 4. [Google Scholar] [CrossRef] [PubMed]
- Monterisi, S.; Lobo, M.J.; Livie, C.; Castle, J.C.; Weinberger, M.; Baillie, G.; Surdo, N.C.; Musheshe, N.; Stangherlin, A.; Gottlieb, E.; et al. Pde2a2 regulates mitochondria morphology and apoptotic cell death via local modulation of cAMP/PKA signalling. eLife 2017, 6, e21374. [Google Scholar] [CrossRef] [PubMed]
- Rinaldi, L.; Pozdniakova, S.; Jayarajan, V.; Troidl, C.; Abdallah, Y.; Muhammad, A.; Ladilov, Y. Role of Soluble adenylyl cyclase in reperfusion-induced injury of cardiac cells. Clin. Res. Cardiol. 2018. [Google Scholar] [CrossRef]
- Soares, L.M.; Meyer, E.; Milani, H.; Steinbusch, H.W.; Prickaerts, J.; de Oliveira, R.M. The phosphodiesterase type 2 inhibitor bay 60-7550 reverses functional impairments induced by brain ischemia by decreasing hippocampal neurodegeneration and enhancing hippocampal neuronal plasticity. Eur. J. Neurosci. 2017, 45, 510–520. [Google Scholar] [CrossRef] [PubMed]
- Neviere, R.; Delguste, F.; Durand, A.; Inamo, J.; Boulanger, E.; Preau, S. Abnormal mitochondrial cAMP/PKA signaling is involved in sepsis-induced mitochondrial and myocardial dysfunction. Int. J. Mol. Sci. 2016, 17, 2075. [Google Scholar] [CrossRef] [PubMed]
- Lezoualc’h, F.; Fazal, L.; Laudette, M.; Conte, C. Cyclic amp sensor EPAC proteins and their role in cardiovascular function and disease. Circ. Res. 2016, 118, 881–897. [Google Scholar] [CrossRef] [PubMed]
- Khaliulin, I.; Bond, M.; James, A.F.; Dyar, Z.; Amini, R.; Johnson, J.L.; Suleiman, M.S. Functional and cardioprotective effects of simultaneous and individual activation of protein kinase a and EPAC. Br. J. Pharmacol. 2017, 174, 438–453. [Google Scholar] [CrossRef] [PubMed]
- Zoccarato, A.; Surdo, N.C.; Aronsen, J.M.; Fields, L.A.; Mancuso, L.; Dodoni, G.; Stangherlin, A.; Livie, C.; Jiang, H.; Sin, Y.Y.; et al. Cardiac hypertrophy is inhibited by a local pool of cAMP regulated by phosphodiesterase 2. Circ. Res. 2015, 117, 707–719. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pozdniakova, S.; Ladilov, Y. Functional Significance of the Adcy10-Dependent Intracellular cAMP Compartments. J. Cardiovasc. Dev. Dis. 2018, 5, 29. https://doi.org/10.3390/jcdd5020029
Pozdniakova S, Ladilov Y. Functional Significance of the Adcy10-Dependent Intracellular cAMP Compartments. Journal of Cardiovascular Development and Disease. 2018; 5(2):29. https://doi.org/10.3390/jcdd5020029
Chicago/Turabian StylePozdniakova, Sofya, and Yury Ladilov. 2018. "Functional Significance of the Adcy10-Dependent Intracellular cAMP Compartments" Journal of Cardiovascular Development and Disease 5, no. 2: 29. https://doi.org/10.3390/jcdd5020029
APA StylePozdniakova, S., & Ladilov, Y. (2018). Functional Significance of the Adcy10-Dependent Intracellular cAMP Compartments. Journal of Cardiovascular Development and Disease, 5(2), 29. https://doi.org/10.3390/jcdd5020029




