Altered Hemodynamics in the Embryonic Heart Affects Outflow Valve Development
Abstract
:1. Introduction
2. Materials and Methods
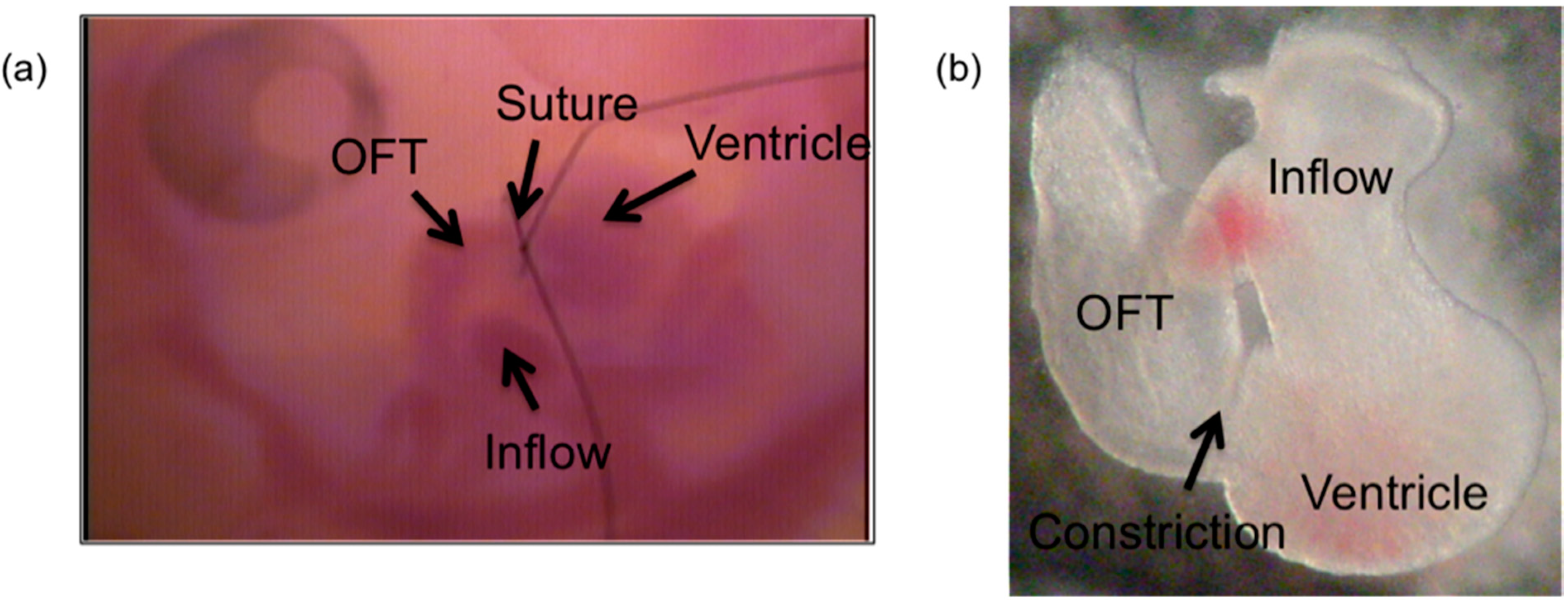
2.1. OFT Banding to alter Intracardiac Hemodynamics
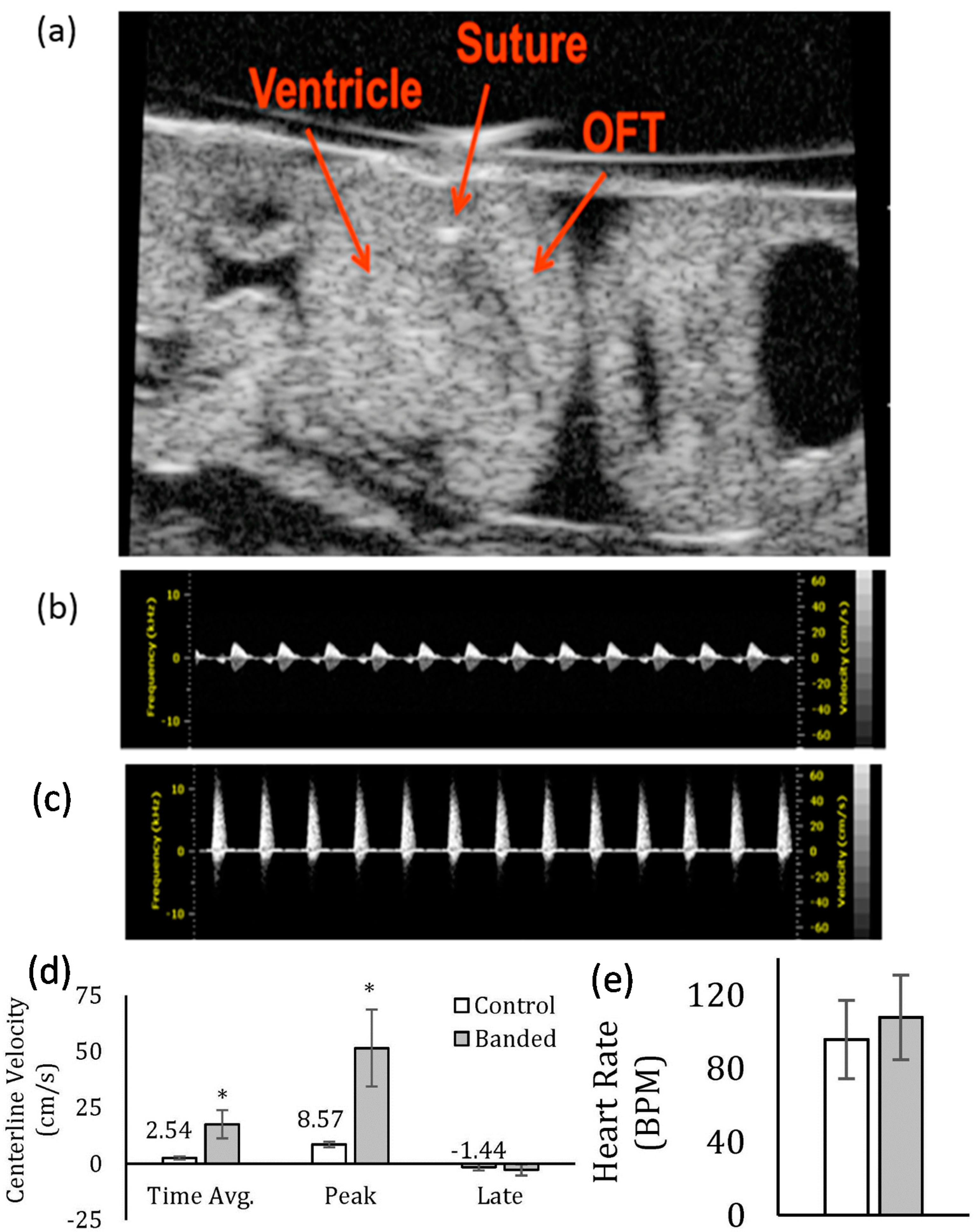
2.2. Analysis of Change in Flow Velocity
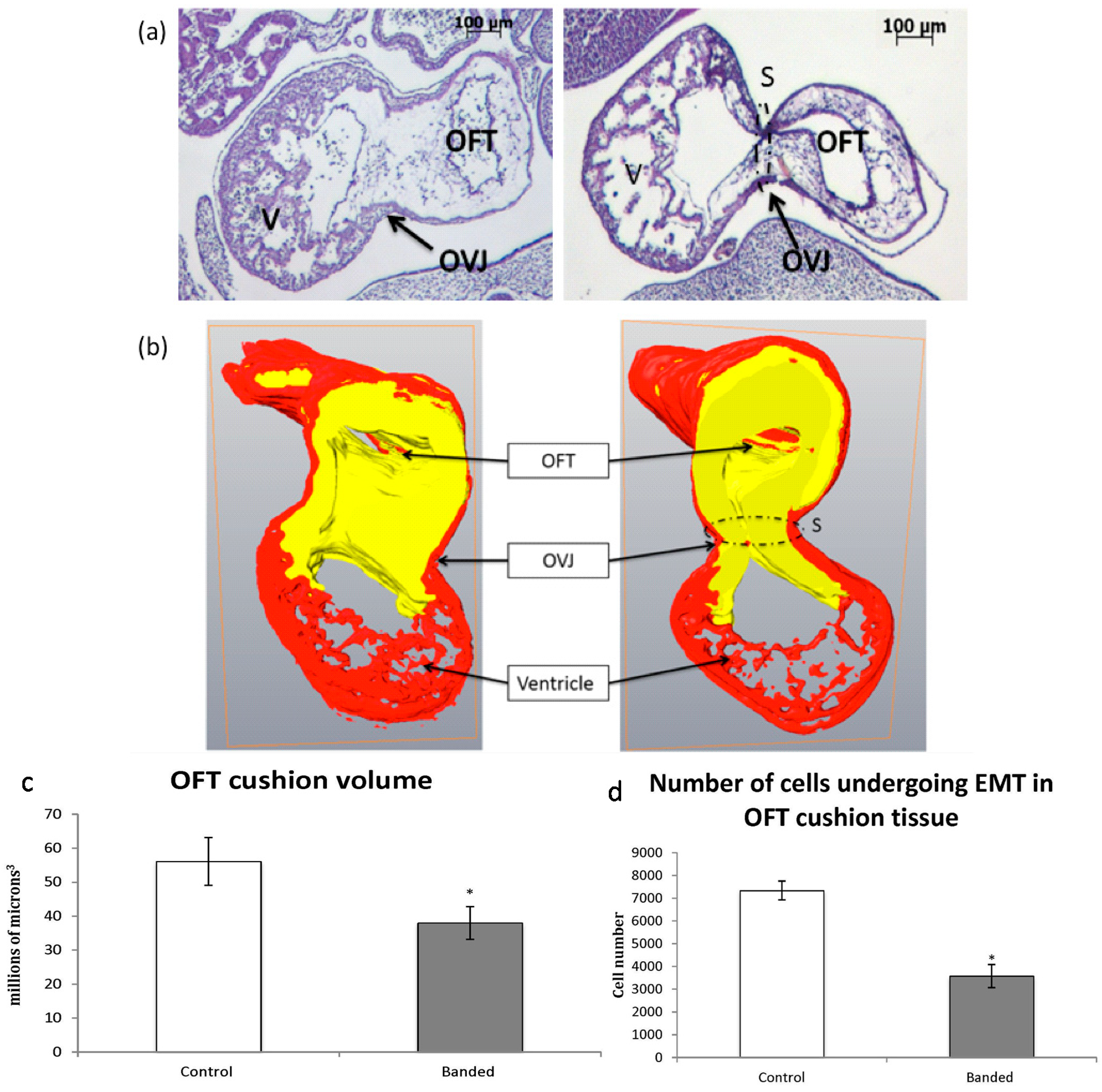
2.3. Three-Dimensional OFT Reconstruction and Determination of the Extent of EMT
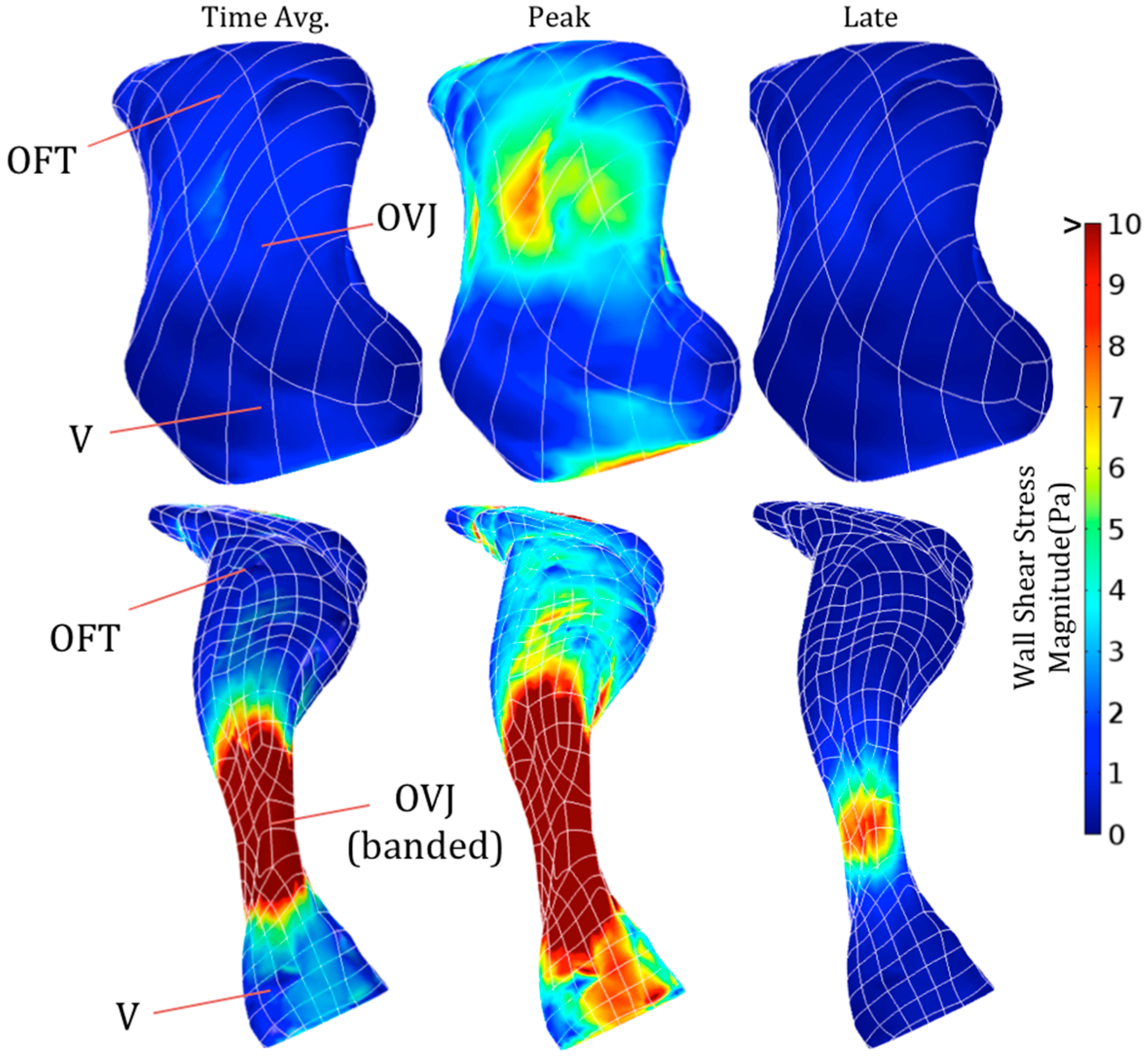
2.4. Computational Fluid Dynamics
2.5. Extraction of Total RNA from OFTs
2.6. Complementary DNA Synthesis
2.7. Quantitative Real-Time Polymerase Chain Reaction
| Gene | Forward (F) and Reverse (R) Primers |
|---|---|
| Arbp2 | F: 5′-GCTTTGCTTCGGTCTTTGAG-3′ |
| R: 5′-AACAACTTTCCGATCACCAC-3′ | |
| klf2 | F: 5′-GCTTCTACCAGACAAACCCG-3′ |
| R: 5′-CAGGACTGGCCCATAACTGT-3′ | |
| rhoA | F: 5′-CAGCACCCTGCACTTGAGTA-3′ |
| R: 5′GCATCCTGTGAGTGCAGAAA-3′ | |
| collagen 1α1 | F: 5′-TACCACTGCAAGAACAGCGT-3′ |
| R: 5′-TCGGTGACCCCATAGGTGAA-3′ | |
| vinculin | F: 5′-CAGGTAGTATCGGCTGCTCG-3′ |
| R: 5′-CCACCAGCCCTGTCATCTTT-3′ | |
| elastin | F: 5′-GTATCCCATCAAAGCTCCCA-3′ |
| R: 5′-CAGCTCCGTATTTAGCTGCC-3′ | |
| periostin | F: 5′-GGATGGTATGAGAGGATGTC-3′ |
| R: 5′GCAAAGAAAGTGAATGAACC-3′ | |
| tenascinC | F: 5′-AGGACACAGCCTCTGCAAGT-3′ |
| R: 5′-TACTGCCCCTGAGAGCTGAT-3′ | |
| CDH11 | F: 5′-AAGACACTGGACCGAGAGGA-3′ |
| R: 5′-TTCTGAGGGCGGTTCCAAAG-3′ | |
| filamin A | F: 5′-CGGCGACTACACCATCAACA-3′ |
| R: 5′-GTCACTTTGGTGGGGTCGAA-3′ | |
| TGFβRIII | F: 5′-CTCTTACCGTCGTGGGCATT-3′ |
| R: 5′-CTGCTTCCCCTGTGTGAGAG-3′ | |
| TGFβ2 | F: 5′-GAGAAAGCCAACCACAGAGC-3′ |
| R: 5′-GGTACAGCTCTATCCGCTGC-3′ | |
| TGFβ3 | F: 5′-CACAATGAGTTGGGCATTTG-3′ |
| R: 5′-GGAACTCTGCTCGAAACAGG-3′ | |
| snai2 | F: 5′-CACGCTCCTTCCTGGTCAAG-3′ |
| R: 5′-GGCTGCGGTATGATAGGGAC-3′ | |
| has2 | F: 5′-CACCGCTGCTTACATTGTGG-3′ |
| R: 5′-TGTGATGCCAGGATAGCACC-3′ | |
| mmp2 | F: 5′-TGATGATGACCGCAAGTGGG-3′ |
| R: 5′-TGTAGATCGGGGCCATGAGA-3′ |
2.8 Statistical Analysis
3. Results
3.1. Effect of OFT Banding on Blood Flow Velocity
3.2. Effect of OFT Banding on OFT Cushion Volume and Extent of EMT
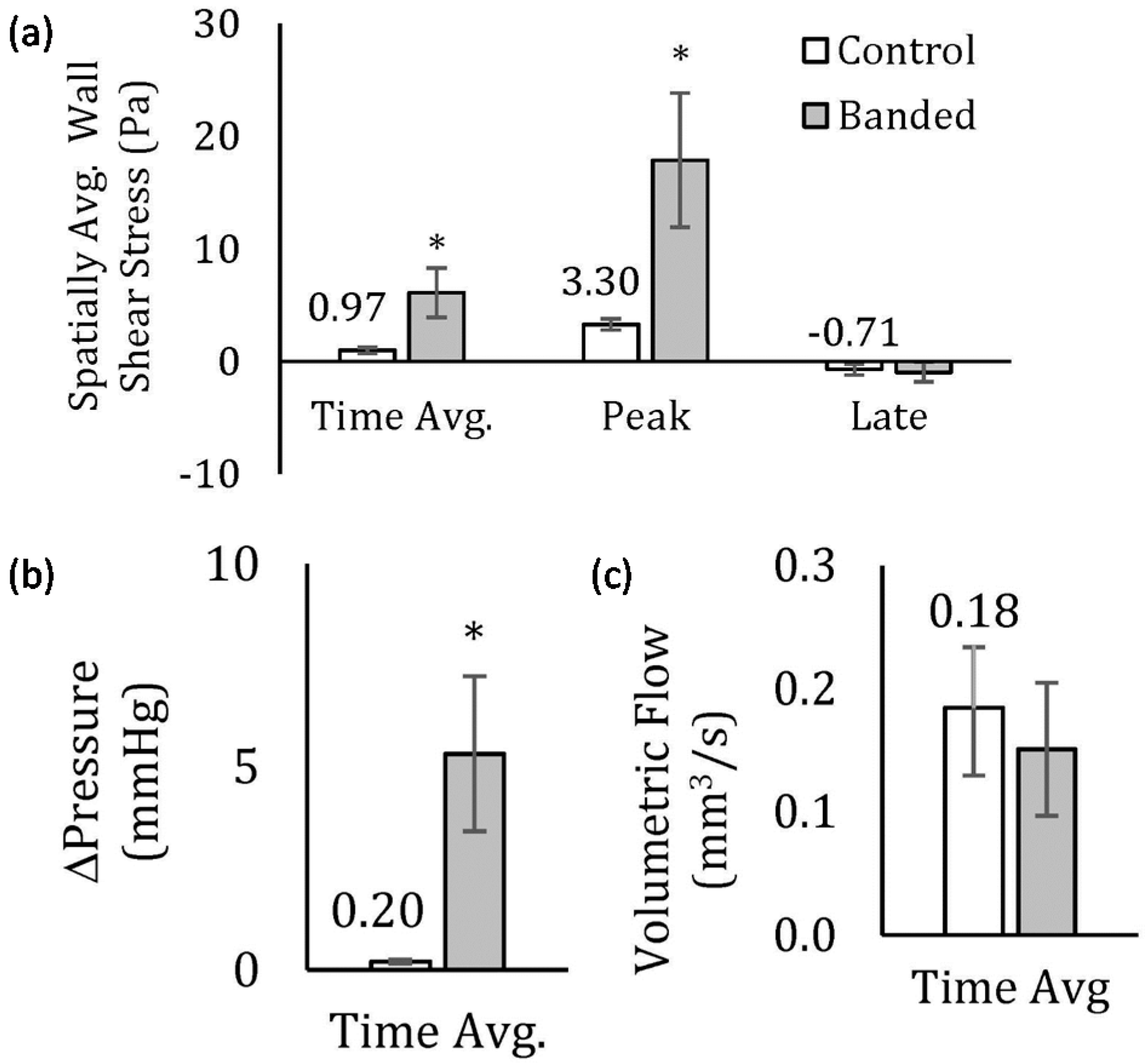
3.3. Effect of OFT Banding on Computed Hemodynamics

3.4. Effect of OFT Banding on Relative Expression of Genes Involved in Valve Development
| Gene | OFT from Control Hearts | OFT from Banded Hearts | p-Value |
|---|---|---|---|
| Shear-/Flow-Responsive | |||
| rhoA | 1.00 ± 0.08 | 0.88 ± 0.04 | 0.02 * |
| klf2 | 1.00 ± 0.24 | 0.83 ± 0.07 | 0.12 |
| ECM | |||
| collagen1a1 | 1.00 ± 0.23 | 0.60 ± 0.06 | 0.01 * |
| periostin | 1.00 ± 0.36 | 2.15 ± 0.38 | 0.01 * |
| tenascinC | 1.00 ± 0.20 | 0.80 ± 0.12 | 0.08 |
| elastin | 1.00 ± 0.32 | 0.83 ± 0.12 | 0.19 |
| vinculin | 1.00 ± 0.07 | 1.07 ± 0.06 | 0.12 |
| EMT/Cell Migration | |||
| TGFβRIII | 1.00 ± 0.12 | 1.28 ± 0.05 | 0.004 * |
| TGFβ2 | 1.00 ± 0.18 | 0.92 ± 0.10 | 0.25 |
| TGFβ3 | 1.00 ± 0.20 | 1.34 ± 0.13 | 0.02 * |
| snai2 | 1.00 ± 0.47 | 0.72 ± 0.09 | 0.14 |
| has2 | 1.00 ± 0.04 | 0.95 ± 0.05 | 0.10 |
| mmp2 | 1.00 ± 0.05 | 1.54 ± 0.27 | 0.03 * |
| CDH11 | 1.00 ± 0.12 | 0.77 ± 0.09 | 0.02 * |
| filamin A | 1.00 ± 0.23 | 0.97 ± 0.09 | 0.34 |
4. Discussion
5. Conclusion
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Neeb, Z.; Lajiness, J.; Bolanis, E.; Conway, S. Cardiac outflow tract anomalies. Wiley Interdiscip Rev. Dev. Biol. 2013, 2, 499–530. [Google Scholar] [CrossRef] [PubMed]
- Hinton, R.; Yutzey, K. Heart valve structure and function in development and disease. Annu Rev. Physiol. 2011, 73, 29–46. [Google Scholar] [CrossRef] [PubMed]
- Von Gise, A.; Pu, W. Endocardial and epicardial epithelial to mesenchymal transitions in heart development and disease. Circ. Res. 2012, 110, 1628–1645. [Google Scholar] [CrossRef] [PubMed]
- Person, A.; Klewer, S.; Runyan, R. Cell biology of cardiac cushion development. Int. Rev. Cytol. 2005, 243, 287–335. [Google Scholar] [PubMed]
- De Vlaming, A.; Sauls, K.; Hajdu, Z.; Visconti, R.; Mehesz, A.; Levine, R.; Slaugenhaupt, S.; Hagège, A.; Chester, A.; Markwald, R.; et al. Atrioventricular valve development: New perspectives on an old theme. Differentiation 2012, 84, 103–116. [Google Scholar] [CrossRef]
- Nakaya, Y.; Sheng, G. EMT in developmental morphogenesis. Cancer Lett. 2013, 341, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Bernanke, D.; Markwald, R. Migratory behavior of cardiac cushion tissue cells in a collagen-lattice culture system. Dev. Biol. 1982, 91, 235–245. [Google Scholar] [CrossRef] [PubMed]
- Runyan, R.; Markwald, R. Invasion of mesenchyme into three-dimensional collagen gels: A regional and temporal analysis of interaction in embryonic heart tissue. Dev. Biol. 1983, 95, 108–114. [Google Scholar] [CrossRef] [PubMed]
- Mjaatvedt, C.; Lepera, R.; Markwald, R. Myocardial specificity for initiating endothelial-mesenchymal cell transition in embryonic chick heart correlates with a particulate distribution of fibronectin. Dev. Biol. 1987, 119, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Culver, J.C.; Dickinson, M.E. The effects of hemodynamic force on embryonic development. Microcirculation 2010, 17, 164–178. [Google Scholar] [CrossRef] [PubMed]
- Butcher, J.; McQuinn, T.; Sedmera, D.; Turner, D.; Markwald, R. Transitions in early embryonic atrioventricular valvular function correspond with changes in cushion biomechanics that are predictable by tissue composition. Circ. Res. 2007, 100, 1503–1511. [Google Scholar] [CrossRef] [PubMed]
- Hove, J.; Köster, R.; Forouhar, A.; Acevedo-Bolton, G.; Fraser, S.; Gharib, M. Intracardiac fluid forces are an essential epigenetic factor for embryonic cardiogenesis. Nature 2003, 421, 172–177. [Google Scholar] [CrossRef]
- Tan, H.; Biechler, S.; Junor, L.; Yost, M.; Dean, D.; Li, J.; Potts, J.; Goodwin, R. Fluid flow forces and rhoa regulate fibrous development of the atrioventricular valves. Dev. Biol. 2013, 374, 345–356. [Google Scholar] [CrossRef] [PubMed]
- Biechler, S.; Junor, L.; Evans, A.; Eberth, J.; Price, R.; Potts, J.; Yost, M.; Goodwin, R. The impact of flow-induced forces on the morphogenesis of the outflow tract. Front. Physiol. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Schneider, C.; Rasband, W.; Eliceiri, K. NIH image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef] [PubMed]
- McDonald, D. Blood Flow in Arteries, 2nd ed.; Edward Arnold: London, UK, 1974. [Google Scholar]
- Pfaffl, M. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef] [PubMed]
- Combs, M.; Yutzey, K. Heart valve development: Regulatory networks in development and disease. Circ. Res. 2009, 105, 408–421. [Google Scholar] [CrossRef] [PubMed]
- Hu, N.; Clark, E. Hemodynamics of the stage 12 to stage 29 chick embryo. Circ. Res. 1989, 65, 1665–1670. [Google Scholar] [CrossRef] [PubMed]
- Hogers, B.; DeRuiter, M.; Gittenberger-de Groot, A.; Poelmann, R. Unilateral vitelline vein ligation alters intracardiac blood flow patterns and morphogenesis in the chick embryo. Circ. Res. 1997, 80, 473–481. [Google Scholar] [CrossRef] [PubMed]
- Hogers, B.; DeRuiter, M.; Gittenberger-de Groot, A.; Poelmann, R. Extraembryonic venous obstructions lead to cardiovascular malformations and can be embryolethal. Cardiovasc. Res. 1999, 41, 87–99. [Google Scholar] [CrossRef] [PubMed]
- Reckova, M.; Rosengarten, C.; de Almeida, A.; Stanley, C.; Wessels, A.; Gourdie, R.; Thompson, R.; Sedmera, D. Hemodynamics is a key epigenetic factor in development of the cardiac conduction system. Circ. Res. 2003, 93, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Stekelenburg-de Vos, S.; Ursem, N.; Hop, W.; Wladimiroff, J.; Gittenberger-de Groot, A.; Poelmann, R. Acutely altered hemodynamics following venous obstruction in the early chick embryo. J. Exp. Biol. 2003, 206, 1051–1057. [Google Scholar] [CrossRef] [PubMed]
- Lucitti, J.; Tobita, K.; Keller, B. Arterial hemodynamics and mechanical properties after circulatory intervention in the chick embryo. J. Exp. Biol. 2005, 208, 1877–1885. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Goenezen, S.; Haller, S.; Hinds, M.; Thornburg, K.; Rugonyi, S. Alterations in pulse wave propagation reflect the degree of outflow tract banding in hh18 chicken embryos. Am. J. Physiol. Heart Circ. Physiol. 2013, 305, H386–H396. [Google Scholar] [CrossRef] [PubMed]
- Bharadwaj, K.; Spitz, C.; Shekhar, A.; Yalcin, H.; Butcher, J. Computational fluid dynamics of developing avian outflow tract heart valves. Ann. Biomed. Eng. 2012, 40, 2212–2227. [Google Scholar] [CrossRef] [PubMed]
- Liu, A.; Nickerson, A.; Troyer, A.; Yin, X.; Cary, R.; Thornburg, K.; Wang, R.; Rugonyi, S. Quantifying blood flow and wall shear stresses in the outflow tract of chick embryonic hearts. Comput. Struct. 2011, 89, 855–867. [Google Scholar] [CrossRef] [PubMed]
- Midgett, M.; Goenezen, S.; Rugonyi, S. Blood flow dynamics reflect degree of outflow tract banding in hamburger-hamilton stage 18 chicken embryos. J. R. Soc. Interface 2014, 11. [Google Scholar] [CrossRef]
- Groenendijk, B.; Van der Heiden, K.; Hierck, B.; Poelmann, R. The role of shear stress on ET-1, KLF2, and NOS-3 expression in the developing cardiovascular system of chicken embryos in a venous ligation model. Physiology (Bethesda) 2007, 22, 380–389. [Google Scholar] [CrossRef]
- Huddleson, J.; Ahmad, N.; Srinivasan, S.; Lingrel, J. Induction of KLF2 by fluid shear stress requires a novel promoter element activated by a phosphatidylinositol 3-kinase-dependent chromatin-remodeling pathway. J. Biol. Chem. 2005, 280, 23371–23379. [Google Scholar] [CrossRef] [PubMed]
- Chiplunkar, A.; Lung, T.; Alhashem, Y.; Koppenhaver, B.; Salloum, F.; Kukreja, R.; Haar, J.; Lloyd, J. Krüppel-like factor 2 is required for normal mouse cardiac development. PLoS ONE 2013, 8, e54891. [Google Scholar] [CrossRef] [PubMed]
- Aikawa, E.; Whittaker, P.; Farber, M.; Mendelson, K.; Padera, R.; Aikawa, M.; Schoen, F. Human semilunar cardiac valve remodeling by activated cells from fetus to adult: Implications for postnatal adaptation, pathology, and tissue engineering. Circulation 2006, 113, 1344–1352. [Google Scholar] [CrossRef] [PubMed]
- Norris, R.; Potts, J.; Yost, M.; Junor, L.; Brooks, T.; Tan, H.; Hoffman, S.; Hart, M.; Kern, M.; Damon, B.; et al. Periostin promotes a fibroblastic lineage pathway in atrioventricular valve progenitor cells. Dev. Dyn. 2009, 238, 1052–1063. [Google Scholar] [CrossRef]
- Garside, V.; Chang, A.; Karsan, A.; Hoodless, P. Co-ordinating notch, BMP, and TGF-β signaling during heart valve development. Cell. Mol. Life Sci. 2013, 70, 2899–2917. [Google Scholar] [CrossRef] [PubMed]
- Brown, C.; Boyer, A.; Runyan, R.; Barnett, J. Requirement of type III TGF-β receptor for endocardial cell transformation in the heart. Science 1999, 283, 2080–2082. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Bowen, C.; Lu, G.; Knapp, C., III; Recknagel, A.; Norris, R.; Butcher, J. Cadherin-11 expression patterns in heart valves associate with key functions during embryonic cushion formation, valve maturation and calcification. Cells Tissues Organs 2013, 198, 300–310. [Google Scholar] [CrossRef]
- Alexander, S.; Jackson, K.; Bushnell, K.; McGuire, P. Spatial and temporal expression of the 72-KDA type IV collagenase (MMP-2) correlates with development and differentiation of valves in the embryonic avian heart. Dev. Dyn. 1997, 209, 261–268. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Menon, V.; Eberth, J.F.; Goodwin, R.L.; Potts, J.D. Altered Hemodynamics in the Embryonic Heart Affects Outflow Valve Development. J. Cardiovasc. Dev. Dis. 2015, 2, 108-124. https://doi.org/10.3390/jcdd2020108
Menon V, Eberth JF, Goodwin RL, Potts JD. Altered Hemodynamics in the Embryonic Heart Affects Outflow Valve Development. Journal of Cardiovascular Development and Disease. 2015; 2(2):108-124. https://doi.org/10.3390/jcdd2020108
Chicago/Turabian StyleMenon, Vinal, John F. Eberth, Richard L. Goodwin, and Jay D. Potts. 2015. "Altered Hemodynamics in the Embryonic Heart Affects Outflow Valve Development" Journal of Cardiovascular Development and Disease 2, no. 2: 108-124. https://doi.org/10.3390/jcdd2020108
APA StyleMenon, V., Eberth, J. F., Goodwin, R. L., & Potts, J. D. (2015). Altered Hemodynamics in the Embryonic Heart Affects Outflow Valve Development. Journal of Cardiovascular Development and Disease, 2(2), 108-124. https://doi.org/10.3390/jcdd2020108






