DEMMI Scores, Length of Stay, and 30-Day Readmission of Acute Geriatric Patients in Denmark: A Cross-Sectional Observational Study with Longitudinal Follow-Up
Abstract
1. Introduction
2. Materials and Methods
Statistical Analysis
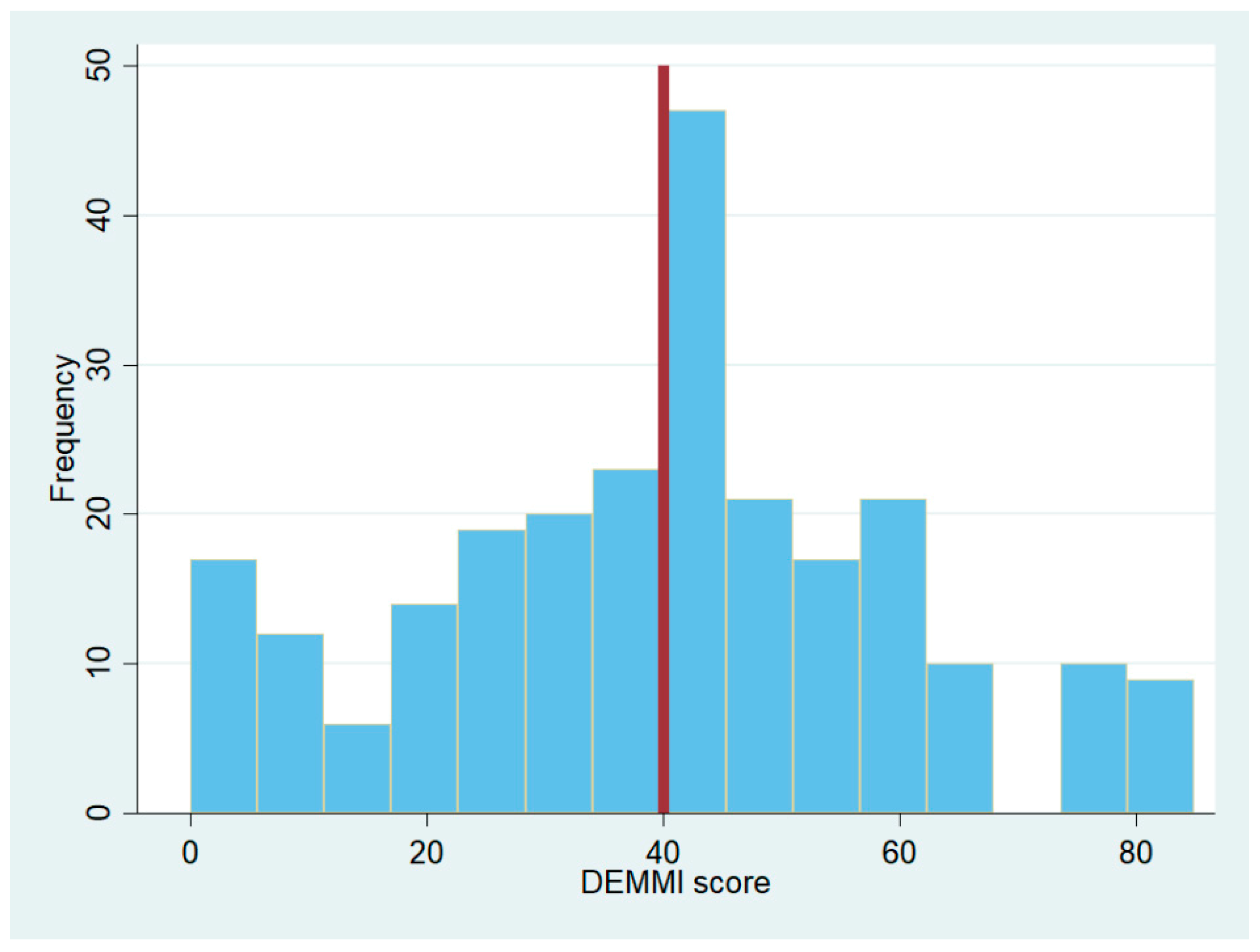
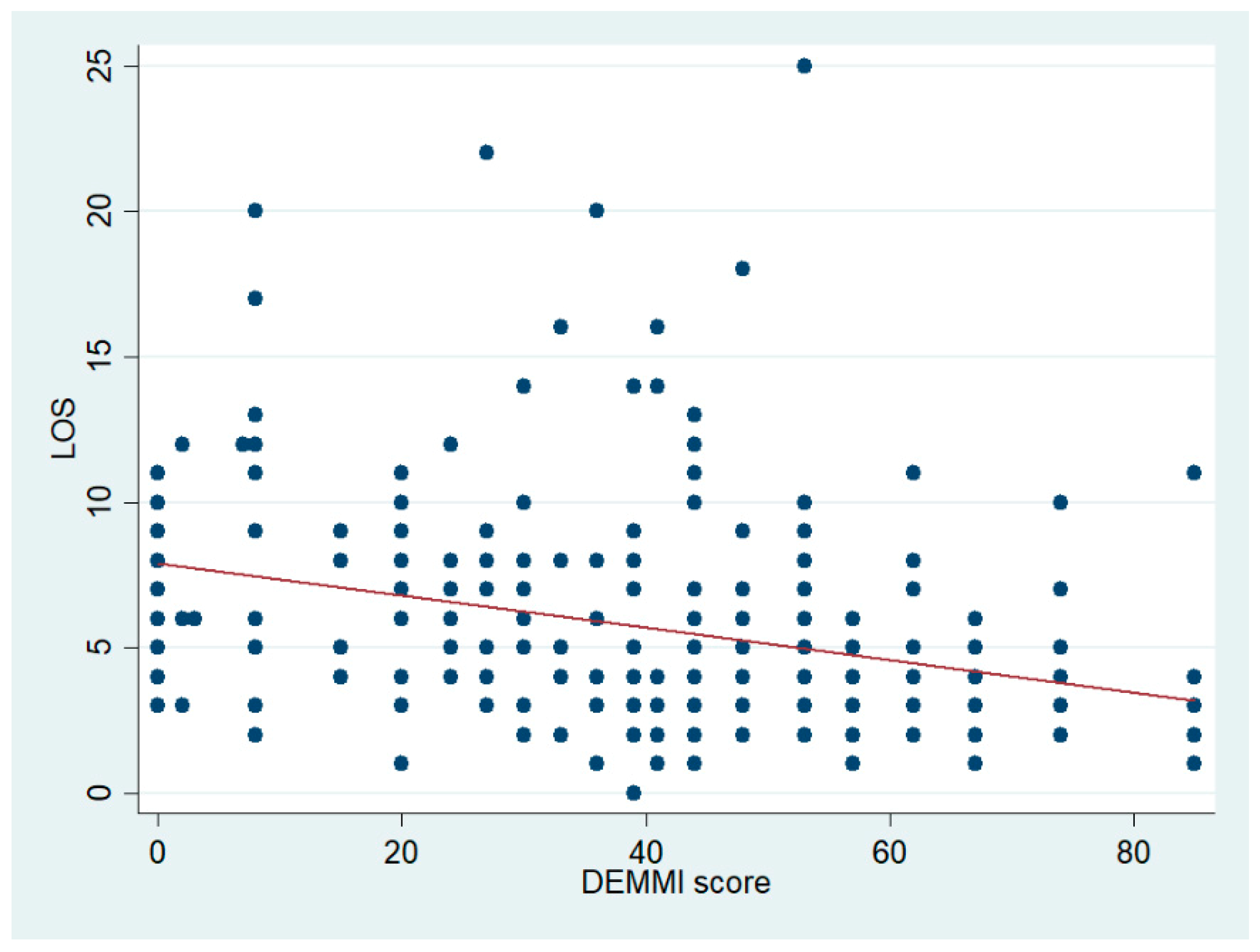
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Savino, E.; Volpato, S.; Zuliani, G.; Guralnik, J.M. Assessment of mobility status and risk of mobility disability in older persons. Curr. Pharm. Des. 2014, 20, 3099–3113. [Google Scholar] [CrossRef] [PubMed]
- De Buyser, S.L.; Petrovic, M.; Taes, Y.E.; Vetrano, D.L.; Corsonello, A.; Volpato, S.; Onder, G. Functional changes during hospital stay in older patients admitted to an acute care ward: A multicenter observational study. PLoS ONE 2014, 9, e96398. [Google Scholar] [CrossRef] [PubMed]
- Mudge, A.M.; O’Rourke, P.; Denaro, C.P. Timing and risk factors for functional changes associated with medical hospitalisation in older patients. J. Gerontol. A Biol. Sci. Med. Sci. 2010, 65, 866–872. [Google Scholar] [CrossRef] [PubMed]
- Brown, C.J.; Flood, K.L. Mobility limitation in the older patient: A clinical review. JAMA 2013, 310, 1168–1177. [Google Scholar] [CrossRef] [PubMed]
- Gibson, M.J.; Andres, R.O.; Coppard, L.C.; Kennedy, T.E. The prevention of falls in later life. A report of the Kellogg International Work Group on the Prevention of Falls by the Elderly. Dan. Med. Bull. 1987, 34 (Suppl. 4), 1–24. [Google Scholar]
- Brown, C.J.; Friedkin, R.J.; Inouye, S.K. Prevalence and outcomes of low mobility in hospitalised older patients. J. Am. Geriatr. Soc. 2004, 52, 1263–1270. [Google Scholar] [CrossRef] [PubMed]
- International Classification of Functioning, Disability and Health (ICF). Available online: http://www.who.int/classifications/icf/en/ (accessed on 1 October 2018).
- Blain, H.; Carriere, I.; Sourial, N.; Berard, C.; Favier, F.; Colvez, A.; Bergman, H. Balance and walking speed predict subsequent 8-year mortality independently of current and intermediate events in well-functioning women aged 75 years and older. J. Nutr. Health Aging 2010, 14, 595–600. [Google Scholar] [CrossRef] [PubMed]
- Gill, T.M.; Allore, H.G.; Hardy, S.E.; Guo, Z. The dynamic nature of mobility disability in older persons. J. Am. Geriatr. Soc. 2006, 54, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Studenski, S.; Perera, S.; Patel, K.; Rosano, C.; Faulkner, K.; Inzitari, M.; Brach, J.; Chandler, J.; Cawthon, P.; Connor, E.B.; et al. Gait speed and survival in older adults. JAMA 2011, 305, 50–58. [Google Scholar] [CrossRef]
- Tiedemann, A.; Shimada, H.; Sherrington, C.; Murray, S.; Lord, S. The comparative ability of eight functional mobility tests for predicting falls in community-dwelling older people. Age Ageing 2008, 37, 430–435. [Google Scholar] [CrossRef]
- Tinetti, M.E. Performance-oriented assessment of mobility problems in elderly patients. J. Am. Geriatr. Soc. 1986, 34, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Chung, J.; Demiris, G.; Thompson, H.J. Instruments to assess mobility limitation in community-dwelling older adults: A systematic review. J Aging Phys. Act. 2015, 23, 298–313. [Google Scholar] [CrossRef] [PubMed]
- Macri, E.M.; Lewis, J.A.; Khan, K.M.; Ashe, M.C.; de Morton, N.A. The de Morton mobility index: Normative data for a clinically useful mobility instrument. J. Aging Res. 2012, 2012, 353252. [Google Scholar] [CrossRef] [PubMed]
- De Morton, N.A.; Meyer, C.; Moore, K.J.; Dow, B.; Jones, C.; Hill, K. Validation of the de Morton Mobility Index (DEMMI) with older community care recipients. Aust. J. Ageing 2011, 30, 220–225. [Google Scholar] [CrossRef] [PubMed]
- De Morton, N.A.; Davidson, M.; Keating, J.L. The de Morton Mobility Index (DEMMI): An essential health index for an ageing world. Health Qual. Life Outcomes 2008, 6, 63. [Google Scholar] [CrossRef] [PubMed]
- De Morton, N.A.; Harding, K.E.; Taylor, N.F.; Harrison, G. Validity of the de Morton Mobility Index (DEMMI) for measuring the mobility of patients with hip fracture during rehabilitation. Disabil. Rehabil. 2013, 35, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Johnston, M.; de Morton, N.; Harding, K.; Taylor, N. Measuring mobility in patients living in the community with Parkinson disease. NeuroRehabilitation 2013, 32, 957–966. [Google Scholar]
- De Morton, N.A.; Lane, K. Validity and reliability of the de Morton Mobility Index in the subacute hospital setting in a geriatric evaluation and management population. J. Rehabil. Med. 2010, 42, 956–961. [Google Scholar] [CrossRef]
- New, P.W.; Scroggie, G.D.; Williams, C.M. The validity, reliability, responsiveness and minimal clinically important difference of the de Morton mobility index in rehabilitation. Disabil. Rehabil. 2017, 39, 1039–1043. [Google Scholar] [CrossRef]
- De Morton, N.A.; Davidson, M.; Keating, J.L. Validity, responsiveness and the minimal clinically important difference for the de Morton Mobility Index (DEMMI) in an older acute medical population. BMC Geriatr. 2010, 10, 72. [Google Scholar] [CrossRef]
- Carpenter, C.R.; Shelton, E.; Fowler, S.; Suffoletto, B.; Platts-Mills, T.F.; Rothman, R.E.; Hogan, T.M. Risk factors and screening instruments to predict adverse outcomes for undifferentiated older emergency department patients: A systematic review and meta-analysis. Acad. Emerg. Med. 2015, 22, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, M.K.; Nielsen, G.L.; Uhrenfeldt, L.; Lundbye-Christensen, S. Risk Assessment of Acute, All-Cause 30-Day Re-admission in Patients Aged 65+: A Nationwide, Register-Based Cohort Study. J. Gen. Intern. Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Den Landsdækkende Database for Geriatri. National Årsrapport 2016. Available online: https://www.sundhed.dk/content/cms/9/4709_geriatri_årsrapport2016_endelig_nov2017.pdf (accessed on 1 October 2018).
- Steyerberg, E.W.; Harrell, F.E., Jr.; Borsboom, G.J.; Eijkemans, M.J.; Vergouwe, Y.; Habbema, J.D. Internal validation of predictive models: Efficiency of some procedures for logistic regression analysis. J. Clin. Epidemiol. 2001, 54, 774–781. [Google Scholar] [CrossRef]
- Pepe, M.; Longton, G.; Janes, H. Estimation and Comparison of Receiver Operating Characteristic Curves. Stata J. 2009, 9, 1. [Google Scholar] [CrossRef] [PubMed]
- Melgaard, D.; Rodrigo-Domingo, M.; Mørch, M. The Prevalence of Oropharyngeal Dysphagia in Acute Geriatric Patients. Geriatrics 2018, 3, 15. [Google Scholar] [CrossRef]
- Bruun, I.H.; Maribo, T.; Norgaard, B.; Schiottz-Christensen, B.; Mogensen, C.B. A prediction model to identify hospitalised, older adults with reduced physical performance. BMC Geriatr. 2017, 17, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Bodilsen, A.C.; Klausen, H.H.; Petersen, J.; Beyer, N.; Andersen, O.; Jorgensen, L.M.; Juul-Larsen, H.G.; Bandholm, T. Prediction of Mobility Limitations after Hospitalisation in Older Medical Patients by Simple Measures of Physical Performance Obtained at Admission to the Emergency Department. PLoS ONE 2016, 11, e0154350. [Google Scholar] [CrossRef] [PubMed]
- Carroll, G.M.; Hampton, J.; Carroll, R.; Smith, S.R. Mobility scores as a predictor of length of stay in general surgery: A prospective cohort study. ANZ J. Surg. 2018, 88, 860–864. [Google Scholar] [CrossRef]
- Cohen, J. The Cost of Dichotomisation. Appl. Psychol. Meas. 1983, 7, 249–253. [Google Scholar] [CrossRef]
- Royston, P.; Altman, D.G.; Sauerbrei, W. Dichotomizing continuous predictors in multiple regression: A bad idea. Stat. Med. 2006, 25, 127–141. [Google Scholar] [CrossRef]
- Altman, D.G.; Royston, P. The cost of dichotomising continuous variables. BMJ 2006, 332, 1080. [Google Scholar] [CrossRef] [PubMed]
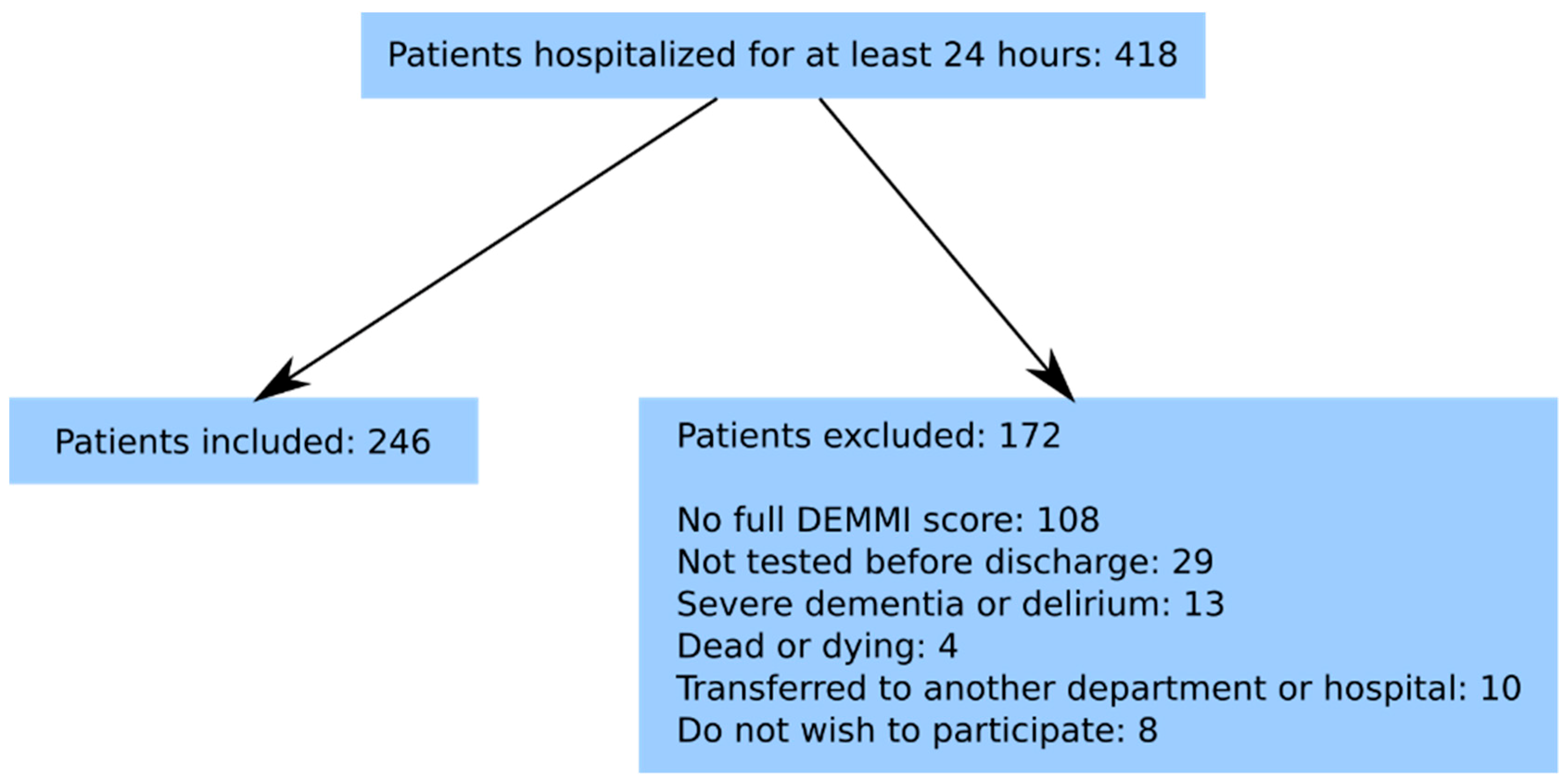
| N = 246 | DEMMI ≤ 40 N = 111 | DEMMI > 40 N = 135 | p-Value | |
|---|---|---|---|---|
| Gender (male) a | 109 (44%) | 53 (48%) | 56 (42%) | 0.325 |
| Age (years) b | 83 [70; 94] | 83 [69; 95] | 84 [70; 93] | 0.435 |
| Reason for hospitalisation a | <0.001 | |||
| Pneumonia | 16 (7%) | 7 (6%) | 9 (7%) | |
| Dyspnea | 28 (11%) | 14 (13%) | 14 (10%) | |
| Dehydration | 15 (6%) | 10 (9%) | 5 (4%) | |
| Fall | 27 (11%) | 15 (14%) | 12 (9%) | |
| Reduction in food intake | 11 (5%) | 9 (8%) | 2 (1%) | |
| Infections | 59 (24%) | 28 (25%) | 31 (23%) | |
| Diverse | 65 (26%) | 14 (13%) | 51 (38%) | |
| Pain | 25 (10%) | 14 (13%) | 11 (8%) | |
| Charlson Comorbidity Index b | 2 [0; 5] | 2 [0; 5]) | 2 [0; 9]) | 0.938 |
| Barthel 100 b | 67.5 [8; 94] | 38 [0; 77] | 79 [39; 95] | <0.001 |
| Body Mass Index b | 25.4 [17.9; 37.2] | 25.4 [19.1; 43.8] | 25.6 [17.9; 34.3] | 0.623 |
| Weight (kg) b | 69 [45; 99.3] | 67.5 [45.1; 102.4] | 69.25 [45; 97.6] | 0.847 |
| Waist line (cm) b | 100 [75; 127] | 100 [83; 136] | 100.5 [74; 120] | 0.909 |
| Lower leg circumference (cm) b | 32 [26; 42] | 32 [24; 42] | 32 [26; 43] | 0.891 |
| Upper arm circumference (cm) b | 27 [21; 34] | 28 [21; 36] | 27 [20; 34] | 0.144 |
| Handgrip—dominant hand (kg) b | 18.9 [7.6; 40.4] | 15.97 [5.6; 37] | 19.87 [8.43; 44.63] | 0.172 |
| Sit-to-stand test (repetitions) b | 0 [0; 10] | 0 [0; 0] | 0 [0; 11] | <0.001 |
| Dysphagia present a | 118 (50.0%) | 66 (59.5%) | 52 (38.5%) | 0.001 |
| Bedridden a | 22 (9.8%) | 21 (21.2%) | 1 (0.8%) | <0.001 |
| Mobility aid a,* | 146 (72.3%) | 73 (93.6%) | 73 (58.9%) | <0.001 |
| Admission time (days) b | 5 [2; 11] | 6 [2; 14] | 4 [1; 11] | <0.001 |
| Rehabilitation plans a | 186 (75.6%) | 96 (86.5%) | 90 (66.7%) | <0.001 |
| Discharged to a | ||||
| Own home | 155 (63%) | 47 (42%) | 108 (80%) | |
| Nursing home | 20 (8%) | 16 (14%) | 4 (3%) | |
| Rehabilitation | 38 (16%) | 31 (28%) | 7 (5%) | |
| Unknown | 33 (13%) | 17 (16%) | 16 (12%) | <0.001 |
| Readmitted within 30 Days after Discharge N = 52 | Not Readmitted within 30 Days after Discharge N = 180 | p-Value | |
|---|---|---|---|
| Gender (male) a | 21 (40.4%) | 82 (45.6%) | 0.509 |
| Age (years) b DEMMI score c | 85.0 [69.0; 93.0] 40.58 (19.85) | 83.0 [70.0; 94.0] 41.28 (20.66) | 0.529 0.827 |
| Reason for hospitalization a | 0.329 | ||
| Pneumonia | 2 (3.8%) | 14 (7.8%) | |
| Dyspnea | 8 (15.4%) | 17 (9.4%) | |
| Dehydration | 5 (9.6%) | 8 (4.4%) | |
| Fall | 4 (7.7%) | 22 (12.2%) | |
| Reduction in food intake | 2 (3.8%) | 7 (3.9%) | |
| Infections | 8 (15.4%) | 46 (25.6%) | |
| Diverse | 15 (28.8%) | 49 (27.2%) | |
| Pain | 8 (15.4%) | 17 (9.4%) | |
| Charlson Comorbidity Index b | 2 [0; 7] | 2 [0; 5] | 0.446 |
| Barthel 100 b | 76 [0; 100] | 68 [8; 90] | 0.736 |
| Body Mass Index b | 23.5 [18.0; 37.6] | 25.7 [17.8; 37.2] | 0.261 |
| Weight (kg) b | 67.7 [47.0; 92.1] | 70.5 [42.8; 101.0] | 0.462 |
| Waist line (cm) b | 96.5 [75.0; 135.0] | 102.0 [75.0; 126.0] | 0.127 |
| Lower leg circumference (cm) b | 32.0 [24.0; 39.0] | 33.0 [26.0; 43.0] | 0.273 |
| Upper arm circumference (cm) b | 26.0 [19.0; 34.0] | 27.0 [21.0; 36.0] | 0.897 |
| Handgrip—dominant hand (kg) b | 20.0 [5.9; 40.4] | 18.8 [8.6; 44.6] | 0.673 |
| Sit-to-stand test (repetitions) b | 0 [0; 9] | 0 [0; 10] | 0.731 |
| Dysphagia present a | 23 (44.2%) | 82 (45.6%) | 0.866 |
| Bedridden a | 3 (6.1%) | 16 (9.5%) | 0.459 |
| Mobility aid use a,* | 36 (78.3%) | 108 (71.1%) | 0.336 |
| Length of stay in hospital (days) b | 5 [1; 16] | 4 [2; 12.5] | 0.208 |
| Plans for rehabilitation a | 44 (84.6%) | 132 (73.3%) | 0.094 |
| Discharged to b | 0.303 | ||
| Own home | 36 (69.2%) | 117 (65%) | |
| Nursing home | 1 (1.9%) | 17 (94%) | |
| Rehabilitation | 8 (15.4%) | 28 (15.6%) | |
| Unknown | 7 (13.5%) | 18 (10.0%) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Melgaard, D.; Rodrigo-Domingo, M.; Mørch, M.M.; Byrgesen, S.M. DEMMI Scores, Length of Stay, and 30-Day Readmission of Acute Geriatric Patients in Denmark: A Cross-Sectional Observational Study with Longitudinal Follow-Up. Geriatrics 2019, 4, 8. https://doi.org/10.3390/geriatrics4010008
Melgaard D, Rodrigo-Domingo M, Mørch MM, Byrgesen SM. DEMMI Scores, Length of Stay, and 30-Day Readmission of Acute Geriatric Patients in Denmark: A Cross-Sectional Observational Study with Longitudinal Follow-Up. Geriatrics. 2019; 4(1):8. https://doi.org/10.3390/geriatrics4010008
Chicago/Turabian StyleMelgaard, Dorte, Maria Rodrigo-Domingo, Marianne M. Mørch, and Stephanie M. Byrgesen. 2019. "DEMMI Scores, Length of Stay, and 30-Day Readmission of Acute Geriatric Patients in Denmark: A Cross-Sectional Observational Study with Longitudinal Follow-Up" Geriatrics 4, no. 1: 8. https://doi.org/10.3390/geriatrics4010008
APA StyleMelgaard, D., Rodrigo-Domingo, M., Mørch, M. M., & Byrgesen, S. M. (2019). DEMMI Scores, Length of Stay, and 30-Day Readmission of Acute Geriatric Patients in Denmark: A Cross-Sectional Observational Study with Longitudinal Follow-Up. Geriatrics, 4(1), 8. https://doi.org/10.3390/geriatrics4010008





