Genomic Characterization of Cryptococcus neoformans Isolated from Zebra Dove Excreta in Songkhla, Thailand
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Cryptococcus Neoformans ISOLATES
2.2. Antifungal Susceptibility Testing
2.3. DNA Extraction, PCR Confirmation, and Whole-Genome Sequencing
2.4. Genome Assembly and Genome Annotation
2.5. Searching Candidate Antifungal Resistance Genes
2.6. Comparative and Phylogenetic Tree Analysis
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ito-Kuwa, S.; Nakamura, K.; Aoki, S.; Vidotto, V. Serotype identification of Cryptococcus neoformans by multiplex PCR. Mycoses 2007, 50, 277–281. [Google Scholar] [CrossRef]
- Hatthakaroon, C.; Pharkjaksu, S.; Chongtrakool, P.; Suwannakarn, K.; Kiratisin, P.; Ngamskulrungroj, P.; Lin, B. Molecular epidemiology of cryptococcal genotype VNIc/ST5 in Siriraj Hospital, Thailand. PLoS ONE 2017, 12, e0173744. [Google Scholar] [CrossRef]
- Saidykhan, L.; Onyishi, C.U.; May, R.C.; Nosanchuk, J. The Cryptococcus gattii species complex: Unique pathogenic yeasts with understudied virulence mechanisms. PLoS Negl. Trop. Dis. 2022, 16, e0010916. [Google Scholar] [CrossRef]
- Bahn, Y.-S.; Sun, S.; Heitman, J.; Lin, X. Microbe Profile: Cryptococcus neoformans species complex. Microbiology 2020, 166, 797–799. [Google Scholar] [CrossRef] [PubMed]
- Refai, M.; Elhariri, M.; Alarousy, R. Cryptococcosis in animals and birds: A review. Eur. J. Acad. Essays 2017, 4, 202–223. [Google Scholar]
- Soogarun, S.; Wiwanitkit, V.; Palasuwan, A.; Pradniwat, P.; Suwansaksri, J.; Lertlum, T.; Maungkote, T. Detection of Cryptococcus neoformans in bird excreta. Southeast Asian J. Trop. Med. Public Health 2006, 37, 768–770. [Google Scholar]
- Khayhan, K. Prevalence of Cryptococcus neoformans in avian droppings and tree debris in the North and Northeast, Thailand. In Proceedings of the Congress of the International Society for Human and Animal Mycology, Amsterdam, The Netherlands, 30 June–4 July 2017. [Google Scholar]
- Indoung, S.; Prachantasena, S.; Wiriyaprom, R.; Laksana, G.; Ruengphodchanapruek, K.; Aleediman, W.; Yingkajorn, M.; Sasithorn, S.; Kaewrakmuk, J.; Ngasaman, R. Detection of zoonotic Cryptococcus neoformans in zebra dove (Geopelia striata) dropping by nested PCR. Southeast Asian J. Trop. Med. Public Health 2021, 54, 780–789. [Google Scholar]
- Boseret, G.; Losson, B.; Mainil, J.G.; Thiry, E.; Saegerman, C. Zoonoses in pet birds: Review and perspectives. Veter. Res. 2013, 44, 36. [Google Scholar] [CrossRef]
- Hagen, F.; Khayhan, K.; Theelen, B.; Kolecka, A.; Polacheck, I.; Sionov, E.; Falk, R.; Parnmen, S.; Lumbsch, H.T.; Boekhout, T. Recognition of seven species in the Cryptococcus gattii/Cryptococcus neoformans species complex. Fungal Genet. Biol. 2015, 78, 16–48. [Google Scholar] [CrossRef]
- Ngamskulrungroj, P.; Gilgado, F.; Faganello, J.; Litvintseva, A.P.; Leal, A.L.; Tsui, K.M.; Mitchell, T.G.; Vainstein, M.H.; Meyer, W. Correction: Genetic diversity of the Cryptococcus species complex suggests that Cryptococcus gattii deserves to have varieties. PLoS ONE 2009, 4, 10-1371. [Google Scholar] [CrossRef]
- Fessel, W.J. Cryptococcal meningitis after unusual exposures to birds. N. Engl. J. Med. 1993, 328, 1354–1355. [Google Scholar] [CrossRef]
- Lagrou, K.; VAN Eldere, J.; Keuleers, S.; Hagen, F.; Merckx, R.; Verhaegen, J.; Peetermans, W.E.; Boekhout, T. Zoonotic transmission of Cryptococcus neoformans from a magpie to an immunocompetent patient. J. Intern. Med. 2005, 257, 385–388. [Google Scholar] [CrossRef]
- Maziarz, E.K.; Perfect, J.R. Cryptococcosis. Infect. Dis. Clin. North. Am. 2016, 30, 179–206. [Google Scholar] [CrossRef]
- Levitz, S.M. The ecology of Cryptococcus neoformans and the epidemiology of cryptococcosis. Rev. Infect. Dis. 1991, 13, 1163–1169. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. About Fungal Meningitis 2025. Available online: https://www.cdc.gov/meningitis/about/fungal-meningitis.html (accessed on 28 February 2025).
- Park, B.J.; A Wannemuehler, K.; Marston, B.J.; Govender, N.; Pappas, P.G.; Chiller, T.M. Estimation of the current global burden of cryptococcal meningitis among persons living with HIV/AIDS. AIDS 2009, 23, 525–530. [Google Scholar] [CrossRef] [PubMed]
- Beale, M.A.; Sabiiti, W.; Robertson, E.J.; Fuentes-Cabrejo, K.M.; O’hAnlon, S.J.; Jarvis, J.N.; Loyse, A.; Meintjes, G.; Harrison, T.S.; May, R.C.; et al. Genotypic diversity is associated with clinical outcome and phenotype in cryptococcal meningitis across southern Africa. PLoS Negl. Trop. Dis. 2015, 9, e0003847. [Google Scholar] [CrossRef]
- Nyazika, T.K.; Hagen, F.; Machiridza, T.; Kutepa, M.; Masanganise, F.; Hendrickx, M.; Boekhout, T.; Magombei-Majinjiwa, T.; Siziba, N.; Chin’OMbe, N.; et al. Cryptococcus neoformans population diversity and clinical outcomes of HIV-associated cryptococcal meningitis patients in Zimbabwe. J. Med. Microbiol. 2016, 65, 1281–1288. [Google Scholar] [CrossRef]
- Harris, J.R.; Lindsley, M.D.; Henchaichon, S.; Poonwan, N.; Naorat, S.; Prapasiri, P.; Chantra, S.; Ruamcharoen, F.; Chang, L.S.; Chittaganpitch, M.; et al. High prevalence of cryptococcal infection among HIV-infected patients hospitalized with pneumonia in Thailand. Clin. Infect. Dis. 2012, 54, e43–e50. [Google Scholar] [CrossRef]
- Barragan, N.C.; Sorvillo, F.; Kuo, T. Cryptococcosis-related deaths and associated medical conditions in the United States, 2000–2010. Mycoses 2014, 57, 741–746. [Google Scholar] [CrossRef]
- Perfect, J.R.; Dismukes, W.E.; Dromer, F.; Goldman, D.L.; Graybill, J.R.; Hamill, R.J.; Harrison, T.S.; Larsen, R.A.; Lortholary, O.; Nguyen, M.-H.; et al. Clinical practice guidelines for the management of cryptococcal disease: 2010 update by the Infectious Diseases Society of America. Clin. Infect. Dis. 2010, 50, 291–322. [Google Scholar] [CrossRef]
- Smith, K.D.; Achan, B.; Hullsiek, K.H.; McDonald, T.R.; Okagaki, L.H.; Alhadab, A.A.; Akampurira, A.; Rhein, J.R.; Meya, D.B.; Boulware, D.R.; et al. Increased antifungal drug resistance in clinical isolates of Cryptococcus neoformans in Uganda. Antimicrob. Agents Chemother. 2015, 59, 7197–7204. [Google Scholar] [CrossRef]
- Córdoba, S.; Afeltra, J.; Vitale, R.G. Evaluation of the in vitro activity of amphotericin B by time-kill curve methodology against large and small capsulate C. neoformans isolates. Diagn. Microbiol. Infect. Dis. 2011, 71, 260–262. [Google Scholar] [CrossRef]
- Tay, S.T.; Haryanty, T.T.; Ng, K.P.; Rohani, M.; Hamimah, H. In vitro susceptibilities of Malaysian clinical isolates of Cryptococcus neoformans var. grubii and Cryptococcus gattii to five antifungal drugs. Mycoses 2006, 49, 324–330. [Google Scholar] [CrossRef]
- Alexander, B.D.; Procop, G.W.; Dufresne, P.; Fuller, J.; Ghannoum, M.A.; Hanson, K.E.; Holliday, D.; Holliday, D.M.; Kovanda, L.; Lockhart, S.R.; et al. M27 Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts, 4th ed.; Clinical and Laboratory Standards Institute: Malvern, PA, USA, 2017. [Google Scholar]
- de Vasconcelos, L.C.; Sampaio, F.C.; Albuquerque, A.d.J.d.R.; Vasconcelos, L.C.d.S. Cell viability of Candida albicans against the antifungal activity of thymol. Braz. Dent. J. 2014, 25, 277–281. [Google Scholar] [CrossRef]
- Soares, M.C.B.; Paula, C.R.; Dias, A.L.; Caseiro, M.M.; da Costa, S.O.P. Environmental strains of Cryptococcus neoformans variety grubii in the city of Santos, SP, Brazil. Rev. Inst. Med. Trop. Sao Paulo 2005, 47, 31–36. [Google Scholar] [CrossRef]
- Mitchell, T.G.; Freedman, E.Z.; White, T.J.; Taylor, J.W. Unique oligonucleotide primers in PCR for identification of Cryptococcus neoformans. J. Clin. Microbiol. 1994, 32, 253–255. [Google Scholar] [CrossRef]
- Nurk, S.; Bankevich, A.; Antipov, D.; Gurevich, A.A.; Korobeynikov, A.; Lapidus, A.; Prjibelski, A.D.; Pyshkin, A.; Sirotkin, A.; Sirotkin, Y.; et al. Assembling single-cell genomes and mini-metagenomes from chimeric MDA products. J. Comput. Biol. 2013, 20, 714–737. [Google Scholar] [CrossRef] [PubMed]
- Mikheenko, A.; Prjibelski, A.; Saveliev, V.; Antipov, D.; Gurevich, A. Versatile genome assembly evaluation with QUAST-LG. Bioinformatics 2018, 34, i142–i150. [Google Scholar] [CrossRef] [PubMed]
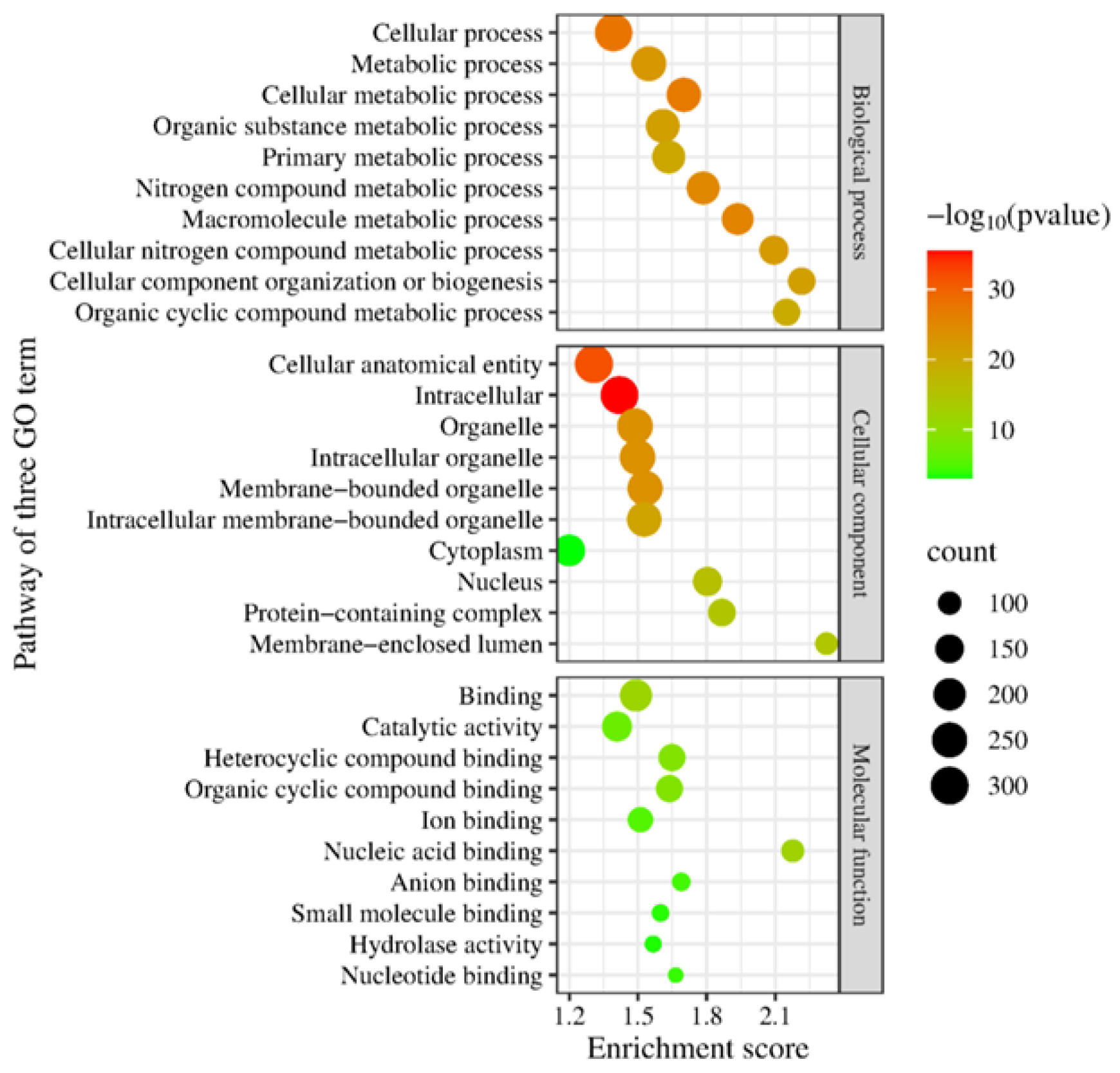
- Gene Ontology Consortium. The Gene Ontology (GO) database and informatics resource. Nucleic Acids Res. 2024, 32 (Suppl. 1), D258–D261. [Google Scholar] [CrossRef]
- Ge, S.X.; Jung, D.; Yao, R. ShinyGO: A graphical enrichment tool for animals and plants. Bioinformatics 2020, 36, 2628–2629. [Google Scholar] [CrossRef]
- Tang, D.; Chen, M.; Huang, X.; Zhang, G.; Zeng, L.; Zhang, G.; Wu, S.; Wang, Y.; Yin, Y. SRplot a free online platform for data visualization and graphing. PLoS ONE 2023, 18, e0294236. [Google Scholar] [CrossRef] [PubMed]
- Emms, D.M.; Kelly, S. OrthoFinder: Phylogenetic orthology inference for comparative genomics. Genome Biol. 2019, 20, 238. [Google Scholar] [CrossRef] [PubMed]
- Letunic, I.; Bork, P. Interactive Tree Of Life (iTOL): An online tool for phylogenetic tree display and annotation. Bioinformatics 2007, 23, 127–128. [Google Scholar] [CrossRef] [PubMed]
- Jain, C.; Rodriguez-R, L.M.; Phillippy, A.M.; Konstantinidis, K.T.; Aluru, S. High throughput ANI analysis of 90K prokaryotic genomes reveals clear species boundaries. Nat. Commun. 2018, 9, 5114. [Google Scholar] [CrossRef]
- Litvintseva, A.P.; Mitchell, T.G. Most environmental isolates of Cryptococcus neoformans var. grubii (serotype A) are not lethal for mice. Infect. Immun. 2009, 77, 3188–3195. [Google Scholar] [CrossRef]
- Litvintseva, A.P.; Thakur, R.; Vilgalys, R.; Mitchell, T.G. Multilocus sequence typing reveals three genetic subpopulations of Cryptococcus neoformans var. grubii (serotype A), including a unique population in Botswana. Genetics 2006, 172, 2223–2238. [Google Scholar] [CrossRef]
- Janbon, G.; Ormerod, K.L.; Paulet, D.; Byrnes, E.J., III; Yadav, V.; Chatterjee, G.; Mullapudi, N.; Hon, C.-C.; Billmyre, R.B.; Brunel, F.; et al. Analysis of the genome and transcriptome of Cryptococcus neoformans var. grubii reveals complex RNA expression and microevolution leading to virulence attenuation. PLoS Genet. 2014, 10, e1004261. [Google Scholar] [CrossRef]
- Khayhan, K.; Hagen, F.; Pan, W.; Simwami, S.; Fisher, M.C.; Wahyuningsih, R.; Chakrabarti, A.; Chowdhary, A.; Ikeda, R.; Taj-Aldeen, S.J.; et al. Geographically structured populations of Cryptococcus neoformans variety grubii in Asia correlate with HIV status and show a clonal population structure. PLoS ONE 2013, 8, e72222. [Google Scholar] [CrossRef]
- Simwami, S.P.; Khayhan, K.; Henk, D.A.; Aanensen, D.M.; Boekhout, T.; Hagen, F.; Brouwer, A.E.; Harrison, T.S.; Donnelly, C.A.; Fisher, M.C.; et al. Low diversity Cryptococcus neoformans variety grubii multilocus sequence types from Thailand are consistent with an ancestral African origin. PLoS Pathog. 2011, 7, e1001343. [Google Scholar] [CrossRef]
- MycoCosm. Cryptococcus. Available online: https://mycocosm.jgi.doe.gov/mycocosm/home/releases?flt=Cryptococcus. (accessed on 25 March 2025).
- Tangwattanachuleeporn, M.; Somparn, P.; Poolpol, K.; Gross, U.; Weig, M.; Bader, O. Prevalence and antifungal susceptibility of Cryptococcus neoformans isolated from pigeon excreta in Chon Buri Province, eastern Thailand. Med. Mycol. J. 2013, 54, 303–307. [Google Scholar] [CrossRef]
- Deng, H.; Song, J.; Huang, Y.; Yang, C.; Zang, X.; Zhou, Y.; Li, H.; Dai, B.; Xue, X. Combating increased antifungal drug resistance in Cryptococcus, what should we do in the future? Acta Biochim. Biophys. Sin. 2023, 55, 540–547. [Google Scholar] [CrossRef]
- Kano, R.; Okubo, M.; Hasegawa, A.; Kamata, H. Multi-azole-resistant strains of Cryptococcus neoformans var. grubii isolated from a FLZ-resistant strain by culturing in medium containing voriconazole. Med. Mycol. 2017, 55, 877–882. [Google Scholar] [CrossRef]
- Zhou, Z.; Zhu, C.; Ip, M.; Liu, M.; Zhu, Z.; Liu, R.; Li, X.; Zeng, L.; Wu, W. Molecular epidemiology and antifungal resistance of Cryptococcus neoformans from human immunodeficiency virus-negative and human immunodeficiency virus-positive patients in eastern China. Front. Microbiol. 2022, 13, 942940. [Google Scholar] [CrossRef]
- Gast, C.E.; Basso, L.R.; Bruzual, I.; Wong, B. Azole resistance in Cryptococcus gattii from the Pacific Northwest: Investigation of the role of ERG11. Antimicrob. Agents Chemother. 2013, 57, 5478–5485. [Google Scholar] [CrossRef] [PubMed]
- Delma, F.Z.; Yang, D.-H.; Cabrera-Orefice, A.; Coolen, J.; Al-Hatmi, A.M.S.; Ahmed, S.A.; Melchers, W.J.G.; Chang, Y.C.; Kwon-Chung, K.J.; de Hoog, S.; et al. Genetic mutations in Cryptococcus neoformans pyrimidine salvage pathway enzymes contribute to reduced susceptibility against 5-fluorocytosine. NPJ Antimicrob. Resist. 2024, 2, 24. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.R.; Douglas, C.M.; Li, W.; Jue, C.K.; Pramanik, B.; Yuan, X.; Rude, T.H.; Toffaletti, D.L.; Perfect, J.R.; Kurtz, M. A glucan synthase FKS1 homolog in Cryptococcus neoformans is single copy and encodes an essential function. J. Bacteriol. 1999, 181, 444–453. [Google Scholar] [CrossRef]
- Beattie, S.R.; Jezewski, A.J.; Ristow, L.C.; Wellington, M.; Krysan, D.J.; Bahn, Y.-S. FKS1 is required for Cryptococcus neoformans fitness in vivo: Application of copper-regulated gene expression to mouse models of cryptococcosis. mSphere 2022, 7, e0016322. [Google Scholar] [CrossRef]
- Kalem, M.C.; Subbiah, H.; Leipheimer, J.; Glazier, V.E.; Panepinto, J.C.; Alspaugh, J.A. Puf4 mediates post-transcriptional regulation of cell wall biosynthesis and caspofungin resistance in Cryptococcus neoformans. mBio 2021, 2. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines for the Diagnosis, Prevention and Management of Cryptococcal Disease in HIV-Infected Adults, Adolescents and Children: Supplement to the 2016 Consolidated Guidelines on the Use of Antiretroviral Drugs for Treating and Preventing HIV Infection; World Health Organization: Geneva, Switzerland, 2018. [Google Scholar]
- Fan, X.; Xiao, M.; Kong, F.; Dou, H.-T.; Wang, H.; Xiao, Y.-L.; Kang, M.; Sun, Z.-Y.; Hu, Z.-D.; Wan, Z.; et al. Predominance of Cryptococcus neoformans var. grubii multilocus sequence type 5 and emergence of isolates with non-wild-type minimum inhibitory concentrations to fluconazole: A multi-centre study in China. Clin. Microbiol. Infect. 2016, 22, 887.e1–887.e9. [Google Scholar] [CrossRef][Green Version]





| No. | Isolate | Amphotericin B (µg/mL) | Fluconazole (µg/mL) | Itraconazole (µg/mL) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| MIC | MFC | MIC | MFC | MIC | MFC | |||||
| Control | C. albicans (ATCC 90028) | 0.125 | 0.5 | 0.03 | ||||||
| 1 | Tip3 | 0.03 | S | 0.03 | 4 | S | 8 | 0.25 | SDD | 0.25 |
| 2 | Rod5 | 0.03 | S | 0.03 | 4 | S | 8 | 0.25 | SDD | 0.25 |
| 3 | DOP2 | 0.0625 | S | 0.25 | 4 | S | 8 | 0.25 | SDD | 0.25 |
| 4 | DOP3 | 0.125 | S | 0.125 | >64 | R | >64 | >8 | R | 0 |
| 5 | DOP3.1 | 0.03 | S | 0.125 | 4 | S | 16 | 0.125 | S | 0.125 |
| 6 | DOP3.2 | 0.03 | S | 0.0625 | 4 | S | 4 | 0.125 | S | 0.125 |
| 7 | DOP12.1 | 0.0625 | S | 0.125 | 4 | S | 8 | 0.125 | S | 0.25 |
| 8 | DOP12.2 | 0.03 | S | 0.125 | 4 | S | 8 | 0.125 | S | 1 |
| 9 | DOP17 | 0.03 | S | 0.125 | 4 | S | 8 | 0.125 | S | 0.25 |
| 10 | K3 | 0.125 | S | 0.25 | 4 | S | 4 | 0.25 | SDD | 0.5 |
| 11 | B3 | 0.03 | S | 0.125 | 4 | S | 8 | 0.125 | S | 0.25 |
| 12 | TL12.1 | 0.0625 | S | 0.125 | 4 | S | 8 | 0.25 | SDD | 0.25 |
| 13 | TL12.2 | 0.0625 | S | 0.125 | 4 | S | 4 | 0.25 | SDD | 0.25 |
| Genome Features | Resistant Isolate (C. neoformans DOP3) | Non-Resistant Isolate (C. neoformans DOP3.1) |
|---|---|---|
| Contigs (≥0 bp) | 716 | 433 |
| Contigs (≥1000 bp) | 304 | 178 |
| Total length (≥0 bp) | 18,495,918 | 12,457,094 |
| Total length (≥1000 bp) | 18,360,890 | 12,373,593 |
| Genome size (kb) | 18,495 | 12,373 |
| Contigs | 345 | 206 |
| Largest contigs (bp) | 478,991 | 480,110 |
| Total length (bp) | 18,388,727 | 12,391,596 |
| GC content (%) | 48.19 | 48.20 |
| N50 (bp) | 153,969 | 170,860 |
| N90 (bp) | 45,564 | 48,649 |
| auN (bp) | 172,572.5 | 184,709.8 |
| L50 | 39 | 25 |
| L90 | 128 | 78 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Indoung, S.; Chumtong, S.; Prachantasena, S.; Wiriyaprom, R.; Surachat, K.; Chusri, S.; Pomwised, R.; Ngasaman, R. Genomic Characterization of Cryptococcus neoformans Isolated from Zebra Dove Excreta in Songkhla, Thailand. Vet. Sci. 2025, 12, 827. https://doi.org/10.3390/vetsci12090827
Indoung S, Chumtong S, Prachantasena S, Wiriyaprom R, Surachat K, Chusri S, Pomwised R, Ngasaman R. Genomic Characterization of Cryptococcus neoformans Isolated from Zebra Dove Excreta in Songkhla, Thailand. Veterinary Sciences. 2025; 12(9):827. https://doi.org/10.3390/vetsci12090827
Chicago/Turabian StyleIndoung, Saowakon, Sanicha Chumtong, Sakaoporn Prachantasena, Ratchakul Wiriyaprom, Komwit Surachat, Sarunyou Chusri, Rattanaruji Pomwised, and Ruttayaporn Ngasaman. 2025. "Genomic Characterization of Cryptococcus neoformans Isolated from Zebra Dove Excreta in Songkhla, Thailand" Veterinary Sciences 12, no. 9: 827. https://doi.org/10.3390/vetsci12090827
APA StyleIndoung, S., Chumtong, S., Prachantasena, S., Wiriyaprom, R., Surachat, K., Chusri, S., Pomwised, R., & Ngasaman, R. (2025). Genomic Characterization of Cryptococcus neoformans Isolated from Zebra Dove Excreta in Songkhla, Thailand. Veterinary Sciences, 12(9), 827. https://doi.org/10.3390/vetsci12090827








