1. Introduction
Lymphoma is a malignant tumor of lymphoid cells that proliferate within solid organs, whether in primary lymphoid structures, such as bone marrow or thymus or in secondary ones, such as lymph nodes, spleen, and mucosa-associated lymphoid tissue in the gastrointestinal tract. Lymphomas are one of dogs and cats’ most recognized and described neoplasia [
1,
2,
3,
4].
Cytological examination of fine-needle aspiration (FNA) samples or cavitary effusion fluids is an inexpensive, first-line and reliable method for diagnosing most of the lymphoma cases in dogs and cats [
3,
4]. The complete diagnostic work-up of a lymphoma case includes the clinical presentation, cell morphology features and grading, immunophenotype, and molecular biology analysis [
5]. Nowadays, phenotyping techniques comprise cell block (CB) immunolabeling, immunohistochemistry of tissues (biopsies), flow cytometry, and PCR for Antigen Receptor Rearrangements (PARR). These are the main ancillary techniques used for canine and, to a lesser extent, for feline lymphoma. Assessing the phenotype and clonality of lymphoproliferative diseases is essential to confirm or exclude the diagnosis when the morphological evaluation raises doubts (e.g., intestinal small cell lymphoma in cats) [
6]. The immunophenotype of lymphoid cells is determined with specific monoclonal and/or polyclonal antibodies. Specifically, the membranous CD3 is used as a T-cell marker, whereas CD79α, CD21, or CD20 (all membranous antigens) and, more recently, the nuclear PAX5 can be used as B-cell markers [
7,
8,
9,
10]. This latter B-cell marker was critical in diagnosing T-cell lymphomas presenting CD79α and CD20 positive cells, because PAX5 was invariably negative [
11,
12]. The B- or T-cell phenotype, as well as the degree of maturation of neoplastic lymphoid cells, has proven to be useful for the classification and prognosis of canine and feline lymphomas [
2,
7,
13]. The gold standard test for assessing the phenotype of lymphoma cells in dogs and cats is immunohistochemistry applied to biopsy tissues. However, cytology is often the first line of diagnostic tests used in most canine and feline lymphomas. Moreover, in many veterinary settings, cytology is the only affordable test for many owners [
14]. It has been reported that 70% to 90% of canine lymphomas are diagnosed by cytology [
15,
16]. Hence, maximizing the information obtained from such specimens is desirable and valuable for the prompt clinical management of lymphoma in dogs and cats.
The first report describing the use of cytological specimens for determining the phenotype of lymphoma in dogs was published more than 30 years ago using cytospin smears obtained from FNA needle rinses of canine lymph nodes [
17]. In this pioneering study, cytology slides were kept unstained until immunolabeling. However, the number of lymphoid cells and their preservation is unknown by using unstained slides prior to immunolabeling. To overcome these limitations, two recent studies showed that lymphoid markers could be detected in previous Romanowsky-stained cytology (RSC) slides [
10,
18]. Currently, the suitability of RSC for confirming or immunophenotyping feline lymphomas is largely unknown. Moreover, it has never been entirely determined if archived cytology specimens can be used for retrospective studies of canine and feline lymphoma. Every immunolabeling reaction can be influenced by several factors occurring outside the laboratory and before the immunolabeling itself. These are included in the so-called pre-analytical variables. Such variables differ, whether the immunolabeling (i.e., the test, the analytical phase) is performed in cells or tissues. For RSC, the pre-analytical variables include: the animal species, the type of specimen, the previous stain used and the exposure to air (i.e., whether the slides were coverslipped). To better evaluate the influence of pre-analytical variables, the immunostaining should be compared in the same set of cells (i.e., effusion cells on RSC compared to those on CB, obtained from the same effusion), instead of comparing different populations (e.g., immunolabeling of effusion cells on RSC compared to immunohistochemistry of lymph node/mass biopsies).
In human pathology, CB prepared from surplus effusion fluids or needle rinses are nowadays considered the most suitable specimens for prospective or retrospective studies [
19]. In canine and feline lymphomas, few studies described the utility of CB, prepared according to different methods [
8,
9,
20]. In those reports, the success of immunophenotyping canine T-cell lymphomas varied, from a low percentage using liquid-based cytology and commercial kits in lymph node samples [
9] to a high success rate using the cell tube block technique in effusion samples [
20].
In veterinary medicine, it is unknown if immunolabeling on CB has advantages over RSC. Elucidating this issue would have a major clinical relevance, since clinicians and oncologists may decide to obtain cytology samples or cytology plus a CB (from needle rinses or effusion fluids) in a canine or feline case suspected of a lymphoma. This would be particularly relevant when the owners are unwilling or cannot afford to submit the animal to a more invasive and expensive procedure, such as obtaining a surgical biopsy specimen for histopathology [
8]. Considering this, in the present study, we aimed to: (1) compare the immunolabeling results in RSC and the matched CB (obtained from surplus effusion fluids or needle rinse fluid) of canine and feline lymphomas using CD3 and PAX5 markers; (2) determine the interobserver agreement in defining the immunophenotype by RSC and by CB; (3) assess the influence of pre-analytical variables, such as species, time of archive and coverslipping on positivity, intensity, background, and non-specific staining in immunolabeling with a membranous and a nuclear lymphoid marker on RSC. All analyses were performed by consensus of two cytopathologists in a multi-headed microscope (considered as observer 1; one with more than 5 years and another with almost 20 years of experience) and by a board-certified pathologist (regarded as observer 2; with more than 30 years of experience and experience in the specific topic of canine and feline lymphomas).
4. Discussion
Lymphoma is frequent in dogs and cats with a heterogeneous clinical and morphological presentation [
28]. Cytology has a major role in diagnosing lymphoma in veterinary medicine, mostly from FNA of lymph nodes, organs, or from effusions [
2,
29,
30]. In cats, cytological diagnosis is more challenging, excluding effusion and mediastinal lymphomas, and the immunophenotype can be essential for confirming or excluding the diagnosis [
31]. The distinction of most canine lymphoma cases from reactive and inflammatory conditions is relatively straightforward to achieve by FNA of lymph nodes alone [
32]. However, their characterization and immunophenotype prediction can be puzzling, even in dogs. Recent studies reported that experienced board-certified pathologists were deceived by morphological features when predicting the immunophenotype by cytology, both in dogs and cats [
11,
30]. Therefore, complementary phenotyping methods are needed to improve the accuracy of cytology to discriminate between B- and T-lymphomas. Nowadays, B- and T-cell lymphoma can be differentiated through immunohistochemistry in tissues, flow cytometry techniques, and PARR.
To maximize the use of minimally invasive clinical samples, immunophenotyping should be attempted in FNA. On many occasions, these will probably be the only samples available to pursue with further tests. Using fresh, unstained samples is often impossible, as there are not enough slides available. It may even compromise the diagnosis if poor-quality slides (misjudged as adequate before staining) are used in the immunostaining [
10,
33]. Nowadays, there is some evidence that RSC can be used for assessing different immunomarkers [
33], including lymphoid markers, at least in dogs [
10]. In cats, limited information exists [
18]. At least in theory, RSC could be a readily available source of material to conduct retrospective studies, investigate, or address the immunophenotype in an individual clinical case. In the present study, we compared the performance of RSC and matched CB for immunophenotyping in dogs and cats.
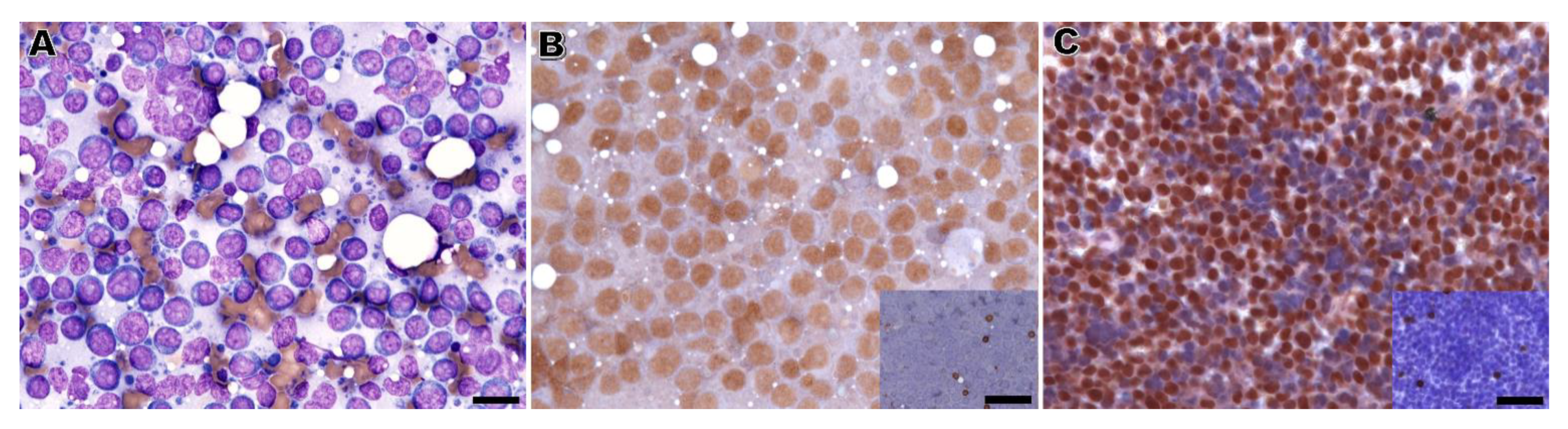
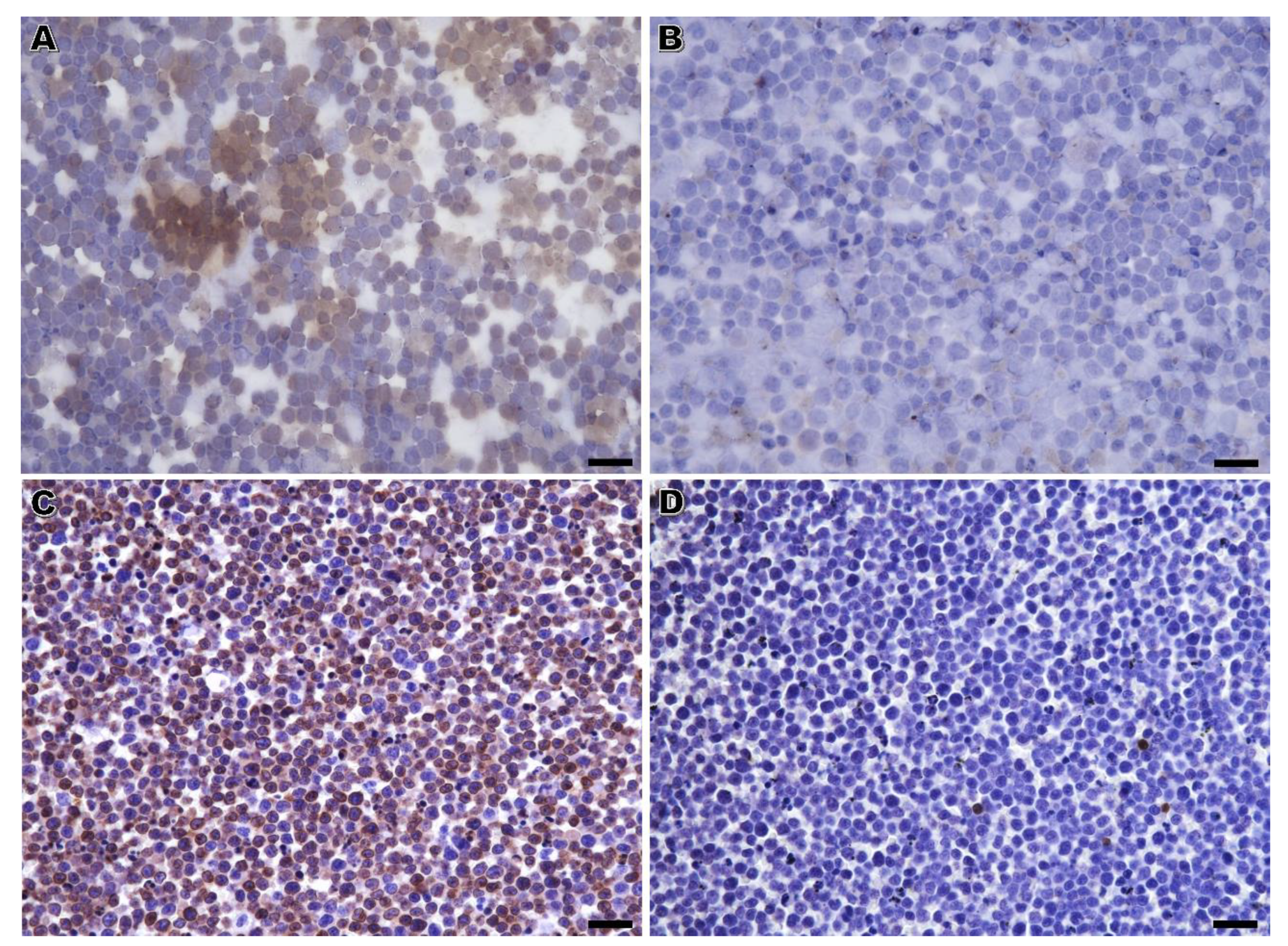
The performance of immunophenotyping on RSC was unsatisfactory, with inconclusive results in around half of the cases. This high number of inconclusive results seems unrelated to the observer, but remains an intrinsic factor of the sample resulting from a loss of antigenicity of the cells in cytology samples. A previous study has shown that the immunoreactivity for anti-CD3 and CD-20 in RSC from dogs was kept for 5 months [
10]. Surprisingly, herein non-diagnostic immunolabeling on RSC results occurred, not only in cases with a long time of archive, but also in recent ones (less than 5 months), especially in cats.
The immunophenotype of the lymphoma was established by CB in 81% of cases. In a previous study, Heinrich et al. [
9] immunophenotyped a similar percentage of canine lymphoma cases (22 out of 23 B-cell lymphomas and 1 out of 6 T-cell lymphomas) using CB and anti-CD20 and anti-CD3 antibodies. In our study, 9 of the inconclusive cases in immunolabeling on CB also rendered a non-diagnostic result in immunolabeling applied to RSC, with a single case classified as a B-cell lymphoma by RSC. This discrepancy can be related to the loss of antigens in more atypical lymphomas, tumor cell heterogeneity, or intrinsic variables of the CB. It should be recalled that using a single B-cell marker in cats is associated with a failure of B-cell lymphoma diagnosis in around 15% of the cases, even in histopathology [
34]. Other reports also showed that using of a single B-cell and T-cell marker resulted in 10 to 20% unclassified canine lymphoma immunophenotypes in histologic samples [
23].
Overall, our findings showed that in most cases with inconclusive immunophenotype in RSC, the CB allowed the evaluation of the B- or T-cell phenotype. Still, inconclusive results can be obtained in some CBs when only two lymphoid markers are used. In these cases, a large panel of T- and B-cell markers should be performed [
28]. For a large panel of markers, consecutive sections of CB are the most suitable type of platform [
19]. This larger panel could include CD3, PAX5, CD20, CD79α, and CD5, as it has been used recently in canine and feline studies [
11,
30].
The nuclear lymphoid marker PAX5 presented comparable results on RSC and CB in dogs, despite presenting a significantly less intense signal in the former samples. Apparently, this lower intensity in RSC did not jeopardize the B-cell phenotype assessment in canine samples, since all the B-cell lymphomas identified in RSC were also identified in CB. This is in accordance with a previous report of immunolabeling in RSC [
10]. Likewise, a good performance of PAX5 immunostaining on archived canine cytological smears has also been reported in human medicine [
35]. Contrasting with our findings, Cozzolino et al. [
35] suggested that the intensity could be higher in previously Papanicolaou stained smears compared to CB since the genetic material of the cells is preserved as a whole in cytology smears [
35]. In feline lymphoma cases, the performance of the PAX5 antibody on RSC was unsatisfactory. In this species, PAX5 immunostaining in RSC was not related to the CB, with only a single B-cell lymphoma (out of 3) clearly diagnosed by RSC.
The immunodetection of CD3 on RSC was highly hampered, particularly in feline and effusion cases. Notably, in cases with CD3-positive cells in CB, more than one third was completely negative in the immunolabeling applied to RSC. Considering that CD3 positivity in the majority of cells is essential for the immunophenotype assessment, it is not surprising that only 7 out of the 22 T-cell lymphomas classified by immunolabeling on CB were likewise diagnosed as such by RSC. In cytology slides, CD3 immunolabeling was also characterized by a lower intensity signal, higher background, and non-specific staining. We could not exclude that the combination of higher background and very low immunoreaction signal could have synergistically contributed to considering CD3 positive cases as being negative, thus decreasing the diagnostic yield of T-cell lymphocyte populations by RSC. This lower intensity and specificity of the CD3 marker in RSC has already been reported elsewhere [
10,
18]. The high percentage of entirely negative cases in our series including archived and very recent cases (cases diagnosed within a week before the immunolabeling on RSC) was a concerning result. The loss of CD3 antigenicity affected both coverslipped and non-coverslipped cytology smears. In the study by Dorfelt et al. [
18], the influence of destaining, coverslipping, and antigen retrieval on CD3 immunolabeling was assessed in feline samples. Our data, did not confirm that the exposure of non-covered Romanowsky-stained lymphocytes to air was a key factor in abolishing CD3 antigenicity [
18]. Actually, the present study showed that lymphoid antigen preservation in cytology specimens could be affected by different interacting pre-analytical factors. Indeed, regression analyses demonstrated that the performance of immunolabeling on RSC with CD3 and PAX5 depends on different pre-analytical variables, namely species, time of archive, and specimen type. According to our results, RSC from cats and effusion fluids are the less suitable for immunolabeling; in these cases, immunophenotyping should be performed in CB or surgical tissue biopsies. Additional caveats of using archived cytology smears for immunolabeling noticed in the present study include the possibility of cell loss during the procedure (which occurred in 14% of the cases in our study); and the higher odds for background and non-specific staining. These two features can result from disrupting the cells in cytology smears, leaking of antigens (namely those membranous or cytoplasmic), and dense areas on the smears. All these features may challenge the interpretation of the results of the immunoreaction [
3,
36,
37,
38].
The interobserver agreement in canine and feline lymphomas characterization by routine cytology, including predicting the phenotype, has been reported to be fair [
11,
30]. Herein, the interobserver reproducibility in scoring the number of immunolabeled cells for anti-CD3 and anti-PAX5 in RSC and CB, and consequently in the assignment of a phenotype, was assessed. The observers had a substantial agreement in scoring the nuclear PAX5 marker in cytology and CB, but a similar agreement for CD3 was only obtained in CB material. Indeed, interpreting the CD3 positivity on RSC was more prone to interobserver variation. On one hand, the lower immunoreactivity and higher background staining on RSC probably accounted for this lower agreement on establishing a T-lymphocyte phenotype. On the other hand, diagnosing B-cell lymphoma in dogs, supported by compatible cytological features and nuclear expression of PAX5, could be performed in archived cytology and CB, with a low interobserver variation. Few studies report the reproducibility of evaluating immunoreactions in canine and feline cells and tissues. Still, it is noteworthy that the interobserver agreement in scoring PAX5 positivity on RSC and CB was similar to that reported for scoring endocrine cytoplasmic immunomarkers in small tissue samples (tissue microarrays) of canine insulinomas [
39]. Interestingly, that study also showed that the heterogeneity of the expression of a marker in the tissue/cell of interest is the most relevant factor, contributing to a substantial difference between the scores of the raters [
39]. Considering lymphoma’s homogeneous/clonal nature, the CD3 expression heterogeneity in a T-cell population is unlikely to explain the marked variation between observers in the semi-quantitative evaluation of the CD3 positivity on RSC.