Optimization of Hempseed-Added Kombucha for Increasing the Antioxidant Capacity, Protein Concentration, and Total Phenolic Content
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.1.1. Plant Materials
2.1.2. Chemicals
2.2. Experimental Design
2.3. Preparation of Kombucha
2.4. Total Phenolic Content (TPC)
2.5. Antioxidant Capacity (AC)
2.6. Protein Concentration Determination (PC)
2.7. Characterization of Optimized Kombucha
2.8. Statistical Analysis
3. Results
Optimized Kombucha
4. Discussion
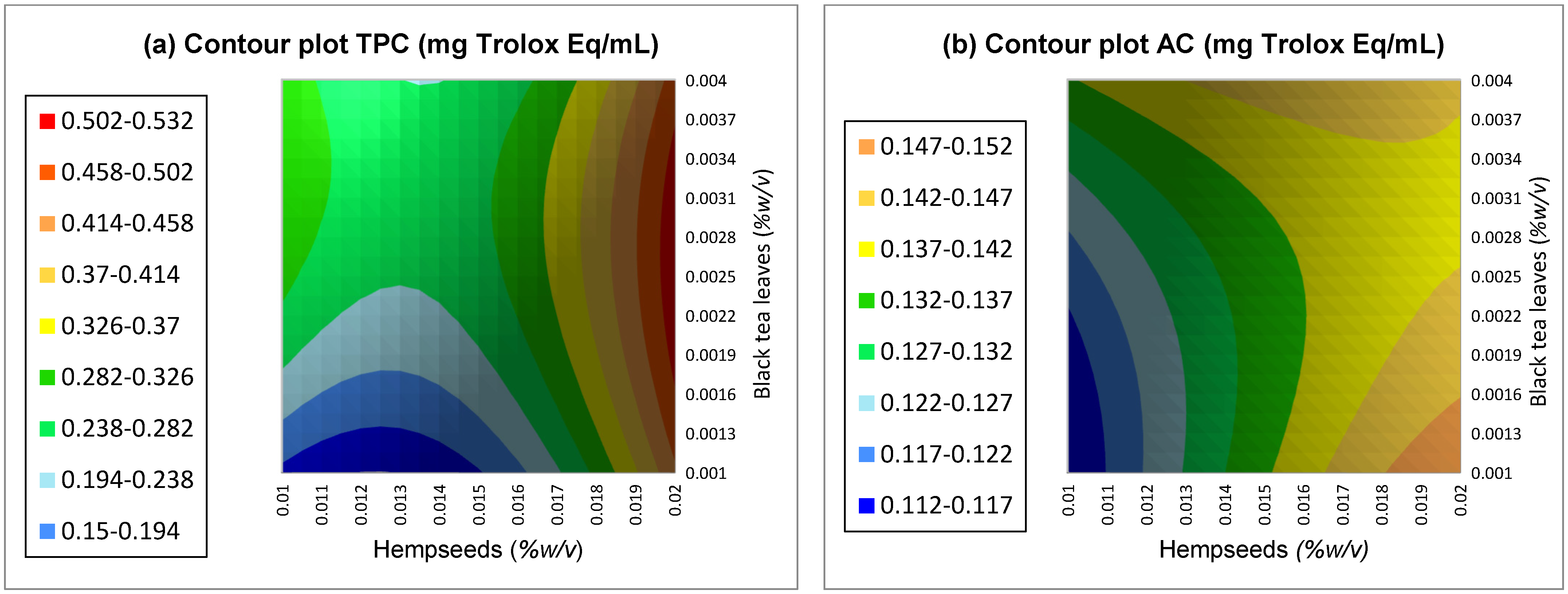
4.1. Total Phenolic Content and Antioxidant Capacity Determination
4.2. Protein Concentration Determination
4.3. Characterization of Optimized Kombucha
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dutta, H.; Paul, S.K. Kombucha Drink: Production, Quality, and Safety Aspects. In Production and Management of Beverages; Grumezescu, A.M., Holban, A.M., Eds.; Woodhead Publishing: Sawston, UK, 2019; pp. 259–288. ISBN 978-0-12-815260-7. [Google Scholar]
- Jayabalan, R.; Malbaša, R.V.; Lončar, E.S.; Vitas, J.S.; Sathishkumar, M. A Review on Kombucha Tea—Microbiology, Composition, Fermentation, Beneficial Effects, Toxicity, and Tea Fungus. Compr. Rev. Food Sci. Food Saf. 2014, 13, 538–550. [Google Scholar] [CrossRef]
- Jakubczyk, K.; Kałduńska, J.; Kochman, J.; Janda, K. Chemical Profile and Antioxidant Activity of the Kombucha Beverage Derived from White, Green, Black and Red Tea. Antioxidants 2020, 9, 447. [Google Scholar] [CrossRef] [PubMed]
- Goh, W.N.; Rosma, A.; Kaur, B.; Fazilah, A.; Karim, A.; Bhat, R. Fermentation of Black Tea Broth (Kombucha): I. Effects of Sucrose Concentration and Fermentation Time on the Yield of Microbial Cellulose. Int. Food Res. J. 2012, 19, 109–117. [Google Scholar]
- Hsieh, Y.; Chiu, M.-C.; Chou, J.-Y. Efficacy of the Kombucha Beverage Derived from Green, Black, and Pu’er Teas on Chemical Profile and Antioxidant Activity. J. Food Qual. 2021, 2021, e1735959. [Google Scholar] [CrossRef]
- Torán-Pereg, P.; del Noval, B.; Valenzuela, S.; Martinez, J.; Prado, D.; Perisé, R.; Arboleya, J.C. Microbiological and Sensory Characterization of Kombucha SCOBY for Culinary Applications. Int. J. Gastron. Food Sci. 2021, 23, 100314. [Google Scholar] [CrossRef]
- Bakowska-Barczak, A.; de Larminat, M.-A.; Kolodziejczyk, P.P. The Application of Flax and Hempseed in Food, Nutraceutical and Personal Care Products. In Handbook of Natural Fibres, 2nd ed.; Kozłowski, R.M., Mackiewicz-Talarczyk, M., Eds.; The Textile Institute Book Series; Woodhead Publishing: Sawston, UK, 2020; pp. 557–590. ISBN 978-0-12-818782-1. [Google Scholar]
- Leonard, W.; Zhang, P.; Ying, D.; Fang, Z. Hempseed in Food Industry: Nutritional Value, Health Benefits, and Industrial Applications. Compr. Rev. Food Sci. Food Saf. 2020, 19, 282–308. [Google Scholar] [CrossRef]
- Battikh, H.; Bakhrouf, A.; Ammar, E. Antimicrobial Effect of Kombucha Analogues. LWT-Food Sci. Technol. 2012, 47, 71–77. [Google Scholar] [CrossRef]
- Kim, J.; Adhikari, K. Current Trends in Kombucha: Marketing Perspectives and the Need for Improved Sensory Research. Beverages 2020, 6, 15. [Google Scholar] [CrossRef]
- Crescente, G.; Piccolella, S.; Esposito, A.; Scognamiglio, M.; Fiorentino, A.; Pacifico, S. Chemical Composition and Nutraceutical Properties of Hempseed: An Ancient Food with Actual Functional Value. Phytochem. Rev. 2018, 17, 733–749. [Google Scholar] [CrossRef]
- Martinez, J.R.; la Paz, S.M.; la Puerta, R.D.; García-Giménez, M.D.; Fernández-Arche, M.Á. Characterization of Bioactive Compounds in Defatted Hempseed (Cannabis sativa L.) by UHPLC-HRMS/MS and Anti-Inflammatory Activity in Primary Human Monocytes. Food Funct. 2020, 11, 4057–4066. [Google Scholar] [CrossRef]
- de Miranda, J.F.; Ruiz, L.F.; Silva, C.B.; Uekane, T.M.; Silva, K.A.; Gonzalez, A.G.M.; Fernandes, F.F.; Lima, A.R. Kombucha: A Review of Substrates, Regulations, Composition, and Biological Properties. J. Food Sci. 2022, 87, 503–527. [Google Scholar] [CrossRef] [PubMed]
- Jayabalan, R.; Malini, K.; Sathishkumar, M.; Swaminathan, K.; Yun, S.-E. Biochemical Characteristics of Tea Fungus Produced during Kombucha Fermentation. Food Sci. Biotechnol. 2010, 19, 843–847. [Google Scholar] [CrossRef]
- Holzapfel, W. Advances in Fermented Foods and Beverages: Improving Quality, Technologies and Health Benefits; Elsevier: Amsterdam, The Netherlands, 2014; ISBN 978-1-78242-024-8. [Google Scholar]
- Narko, T.; Wibowo, M.S.; Damayanti, S.; Wibowo, I.; Hadiyat, M.A. Optimization of Robusta Green Coffee Fermentation Using Response Surface Methodology. Coffee Sci. 2020, 15, e151671. [Google Scholar] [CrossRef]
- Bakhaidar, R.B.; Naveen, N.R.; Basim, P.; Murshid, S.S.; Kurakula, M.; Alamoudi, A.J.; Bukhary, D.M.; Jali, A.M.; Majrashi, M.A.; Alshehri, S.; et al. Response Surface Methodology (RSM) Powered Formulation Development, Optimization and Evaluation of Thiolated Based Mucoadhesive Nanocrystals for Local Delivery of Simvastatin. Polymers 2022, 14, 5184. [Google Scholar] [CrossRef]
- Ramírez-Rodrigues, M.M.; Estrada-Beristain, C.; Metri-Ojeda, J.; Pérez-Alva, A.; Baigts-Allende, D.K. Spirulina Platensis Protein as Sustainable Ingredient for Nutritional Food Products Development. Sustainability 2021, 13, 6849. [Google Scholar] [CrossRef]
- Jiménez-González, O.; Guerrero-Beltrán, J.Á. Microencapsulates by Spray of Lacticaseibacillus rhamnosus GG from Fermented Whole or Skimmed Cow’s Milk Added with Mexican Honeysuckle (Justicia spicigera) Extract Using Mesquite Gum as Carrier Agent. Heliyon 2022, 8, e10733. [Google Scholar] [CrossRef]
- Murphy, T.E.; Walia, K.; Farber, J.M. Safety Aspects and Guidance for Consumerson the Safe Preparation, Handling and Storage of Kombucha—A Fermented Tea Beverage. Food Prot. Trends 2018, 38, 329–337. [Google Scholar]
- Jayabalan, R.; Subathradevi, P.; Marimuthu, S.; Sathishkumar, M.; Swaminathan, K. Changes in Free-Radical Scavenging Ability of Kombucha Tea during Fermentation. Food Chem. 2008, 109, 227–234. [Google Scholar] [CrossRef]
- Stewart, A.J.; Mullen, W.; Crozier, A. On-Line High-Performance Liquid Chromatography Analysis of the Antioxidant Activity of Phenolic Compounds in Green and Black Tea. Mol. Nutr. Food Res. 2005, 49, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Nissen, L.; Demircan, B.; Taneyo-Saa, D.L.; Gianotti, A. Gianotti Shift of Aromatic Profile in Probiotic Hemp Drink Formulations: A Metabolomic Approach. Microorganisms 2019, 7, 509. [Google Scholar] [CrossRef] [PubMed]
- Oseyko, M.; Sova, N.; Lutsenko, M.; Kalyna, V. Chemical Aspects of the Composition of Industrial Hemp Seed Products. Ukr. Food J. 2019, 8, 544–559. [Google Scholar] [CrossRef]
- Zhu, Q.Y.; Zhang, A.; Tsang, D.; Huang, Y.; Chen, Z.Y. Stability of Green Tea Catechins. J. Agric. Food Chem. 1997, 45, 4624–4628. [Google Scholar] [CrossRef]
- Jhoo, J.-W.; Lo, C.-Y.; Li, S.; Sang, S.; Ang, C.Y.W.; Heinze, T.M.; Ho, C.-T. Stability of Black Tea Polyphenol, Theaflavin, and Identification of Theanaphthoquinone as Its Major Radical Reaction Product. J. Agric. Food Chem. 2005, 53, 6146–6150. [Google Scholar] [CrossRef] [PubMed]
- Szparaga, A.; Tabor, S.; Kocira, S.; Czerwińska, E.; Kuboń, M.; Płóciennik, B.; Findura, P. Survivability of Probiotic Bacteria in Model Systems of Non-Fermented and Fermented Coconut and Hemp Milks. Sustainability 2019, 11, 6093. [Google Scholar] [CrossRef]
- Bartkiene, E.; Zokaityte, E.; Lele, V.; Sakiene, V.; Zavistanaviciute, P.; Klupsaite, D.; Bendoraitiene, J.; Navikaite-Snipaitiene, V.; Ruzauskas, M. Technology and Characterisation of Whole Hemp Seed Beverages Prepared from Ultrasonicated and Fermented Whole Seed Paste. Int. J. Food Sci. Technol. 2020, 55, 406–419. [Google Scholar] [CrossRef]
- Nissen, L.; di Carlo, E.; Gianotti, A. Prebiotic Potential of Hemp Blended Drinks Fermented by Probiotics. Food Res. Int. 2020, 131, 109029. [Google Scholar] [CrossRef]
- Tang, C.-H.; Ten, Z.; Wang, X.-S.; Yang, X.-Q. Physicochemical and Functional Properties of Hemp (Cannabis sativa L.) Protein Isolate. J. Agric. Food Chem. 2006, 54, 8945–8950. [Google Scholar] [CrossRef]
- Malomo, S.A.; Rotimi, E.A. A Comparative Study of the Structural and Functional Properties of Isolated Hemp Seed (Cannabis sativa L.) Albumin and Globulin Fraction. Food Hydrocoll. 2015, 43, 743–752. [Google Scholar] [CrossRef]
- Chen, C.; Liu, B.Y. Changes in Major Components of Tea Fungus Metabolites during Prolonged Fermentation. J. Appl. Microbiol. 2000, 89, 834–839. [Google Scholar] [CrossRef]
- Sreeramulu, G.; Zhu, Y.; Knol, W. Kombucha Fermentation and Its Antimicrobial Activity. J. Agric. Food Chem. 2000, 48, 2589–2594. [Google Scholar] [CrossRef]
- TTBGov-Kombucha General. Available online: https://www.ttb.gov/kombucha/kombucha-general (accessed on 29 September 2022).

| Factors | Low | High |
|---|---|---|
| −1 | 1 | |
| Hempseeds concentration (% w/v) | 0.01 | 0.02 |
| Black tea leaves concentration (% w/v) | 0.001 | 0.004 |
| Independent Variables | Response Variables | ||||
|---|---|---|---|---|---|
| Samples | Hempseeds (% w/v) | Black Tea Leaves (% w/v) | AC (DPPH+ mg Trolox Eq/mL) | TPC (mg GA Eq/mL) | PC (µg/mL) |
| 1 | 0.0221 | 0.0025 | 0.142 ± 0.107 | 0.376 ± 0.001 | 6.073 ± 0.032 |
| 2 | 0.0221 | 0.0025 | 0.146 ± 0.107 | 0.260 ± 0.019 | 6.001 ± 0.019 |
| 3 | 0.02 | 0.001 | 0.118 ± 0.107 | 0.167 ± 0.001 | 6.842 ± 0.065 |
| 4 | 0.02 | 0.004 | 0.112 ± 0.077 | 0.324 ± 0.001 | 4.148 ± 0.004 |
| 5 | 0.02 | 0.004 | 0.117 ± 0.107 | 0.136 ± 0.002 | 4.080 ± 0.006 |
| 6 | 0.02 | 0.001 | 0.146 ± 0.107 | 0.907 ± 0.001 | 7.828 ± 0.040 |
| 7 | 0.015 | 0.0025 | 0.138 ± 0.323 | 0.483 ± 0.001 | 4.001 ± 0.019 |
| 8 | 0.015 | 0.0004 | 0.134 ± 0.370 | 0.290 ± 0.006 | 4.908 ± 0.002 |
| 9 | 0.015 | 0.0046 | 0.139 ± 0.107 | 0.129 ± 0.018 | 4.128 ± 0.001 |
| 10 | 0.015 | 0.0025 | 0.108 ± 0.646 | 0.126 ± 0.002 | 4.990 ± 0.003 |
| 11 | 0.015 | 0.0025 | 0.143 ± 0.431 | 0.187 ± 0.001 | 4.459 ± 0.013 |
| 12 | 0.015 | 0.0025 | 0.141 ± 0.538 | 0.394 ± 0.003 | 5.908 ± 0.006 |
| 13 | 0.015 | 0.0046 | 0.141 ± 0.909 | 0.881 ± 0.001 | 5.586 ± 0.004 |
| 14 | 0.015 | 0.0025 | 0.135 ± 0.077 | 0.181 ± 0.002 | 5.772± 0.001 |
| 15 | 0.015 | 0.0025 | 0.141 ± 0.586 | 0.107 ± 0.017 | 4.148 ± 0.004 |
| 16 | 0.015 | 0.0025 | 0.148 ± 0.538 | 0.336 ± 0.002 | 4.120 ± 0.003 |
| 17 | 0.015 | 0.0025 | 0.147 ± 0.538 | 0.382 ± 0.001 | 5.459 ± 0.013 |
| 18 | 0.015 | 0.0025 | 0.139 ± 0.215 | 0.393 ± 0.005 | 5.772 ± 0.001 |
| 19 | 0.015 | 0.0025 | 0.140 ± 0.383 | 0.277 ± 0.001 | 5.291 ± 0.009 |
| 20 | 0.015 | 0.0004 | 0.142 ± 0.771 | 0.193 ± 0.003 | 4.328 ± 0.022 |
| 21 | 0.01 | 0.004 | 0.138 ± 0.323 | 0.353 ± 0.001 | 4.041 ± 0.003 |
| 22 | 0.01 | 0.001 | 0.128 ± 0.313 | 0.266 ± 0.003 | 1.073 ± 0.032 |
| 23 | 0.01 | 0.004 | 0.103± 0.323 | 0.105 ± 0.001 | 1.842 ± 0.052 |
| 24 | 0.01 | 0.001 | 0.143 ± 0.685 | 0.287 ± 0.005 | 0.341 ± 0.002 |
| 25 | 0.0079 | 0.0025 | 0.149 ± 0.598 | 0.179 ± 0.002 | 1.337 ± 0.066 |
| 26 | 0.0079 | 0.0025 | 0.145 ± 0.969 | 0.309 ± 0.002 | 1.041 ± 0.001 |
| Control | 0 | 0.0025 | 0.134 ± 0.215 | 0.206 ± 0.005 | 0.356 ± 0.002 |
| Hempseeds (% w/v) | Black Tea Leaves (% w/v) | AC (mg Trolox Eq/mL) | TPC (mg GA Eq/mL) | PC (µg/mL) | Fit |
|---|---|---|---|---|---|
| 0.0170 | 0.0046 | 0.1560 | 0.5636 | 7.8491 | 0.8347 |
| Response | ANOVA | Regression Model | ||||
|---|---|---|---|---|---|---|
| df | Adj SS | Adj MS | p-Value | R2 | S | |
| AC (mg Trolox Eq/mL) | 5 | 0.0018 | 0.0003 | 0.0034 | 0.87 | 0.0097 |
| TPC (mg GA Eq/mL) | 5 | 0.0344 | 0.0688 | 0.0377 | 0.80 | 0.0168 |
| PC (µg/mL) | 5 | 0.0030 | 0.0060 | 0.0176 | 0.84 | 0.0080 |
| Sample | Hempseeds (% w/v) | Black Tea Leaves | pH | °Brix | Alcohol (% v/v) | AC (mg Trolox Eq/mL) | TPC (mg GA Eq/mL) | PC (µg/mL) |
|---|---|---|---|---|---|---|---|---|
| (% w/v) | ||||||||
| Optimized kombucha | 0.017 | 0.0046 | 2.68 ± 0.006 a | 2.9 ± 0.005 a | 0.047 ± 0.0901 a | 0.139 ± 0.1078 a | 0.426 ± 0.0933 a | 4.118 ± 0.1041 a |
| Control | 0 | 0.0025 | 2.62 ± 0.011 a | 3.1 ± 0.022 a | 0.044 ± 0.0003 a | 0.150 ± 0.2155 a | 0.206 ± 0.0005 b | 0.356 ± 0.0022 b |
| AC (mg Trolox Eq/mL) | ||
| Exp | RSM | Percentage of ratio (%) |
| 0.139 ± 0.1078 | 0.1560 | 89.10 |
| TPC (mg GA Eq/mL) | ||
| Exp | RSM | Percentage of ratio (%) |
| 0.426 ± 0.0933 | 0.5636 | 75.58 |
| PC (µg/mL) | ||
| Exp | RSM | Percentage of ratio (%) |
| 6.118 ± 0.1041 | 7.8491 | 77.94 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reyes-Flores, S.; Pereira, T.S.S.; Ramírez-Rodrigues, M.M. Optimization of Hempseed-Added Kombucha for Increasing the Antioxidant Capacity, Protein Concentration, and Total Phenolic Content. Beverages 2023, 9, 50. https://doi.org/10.3390/beverages9020050
Reyes-Flores S, Pereira TSS, Ramírez-Rodrigues MM. Optimization of Hempseed-Added Kombucha for Increasing the Antioxidant Capacity, Protein Concentration, and Total Phenolic Content. Beverages. 2023; 9(2):50. https://doi.org/10.3390/beverages9020050
Chicago/Turabian StyleReyes-Flores, Samanta, Taísa Sabrina Silva Pereira, and Milena Maria Ramírez-Rodrigues. 2023. "Optimization of Hempseed-Added Kombucha for Increasing the Antioxidant Capacity, Protein Concentration, and Total Phenolic Content" Beverages 9, no. 2: 50. https://doi.org/10.3390/beverages9020050
APA StyleReyes-Flores, S., Pereira, T. S. S., & Ramírez-Rodrigues, M. M. (2023). Optimization of Hempseed-Added Kombucha for Increasing the Antioxidant Capacity, Protein Concentration, and Total Phenolic Content. Beverages, 9(2), 50. https://doi.org/10.3390/beverages9020050







