The Effect of Diabetes Mellitus on IGF Axis and Stem Cell Mediated Regeneration of the Periodontium
Abstract
:1. Introduction
2. Periodontitis
3. Diabetes
4. Reciprocal Interaction between Diabetes and Periodontitis
5. Current Periodontal Therapies in Diabetics
5.1. Non-Surgical Periodontal Therapy
5.2. Surgical Periodontal Therapy
5.3. Regenerative Surgical Periodontal Therapy in Diabetic Animal Models
5.4. Regenerative Surgical Periodontal Therapy in Diabetic Patients
6. Tissue Engineering and Periodontal Regeneration
6.1. Stem Cells in Regenerative Periodontal Therapy
6.1.1. Periodontal Ligament Mesenchymal Stromal Cells (PDL-MSCs)
6.1.2. Bone Marrow Mesenchymal Stromal Cells (BM-MSCs)
6.1.3. PDL-MSCs vs. BM-MSCs for Periodontal Regeneration
6.1.4. PDL-MSCs Isolated from Diabetic Patients and/or Cultured under Diabetic Conditions
6.1.5. BM-MSCs Isolated from Diabetic Patients and/or Cultured under Diabetic Conditions
6.2. Growth Factors in Regenerative Periodontal Therapy
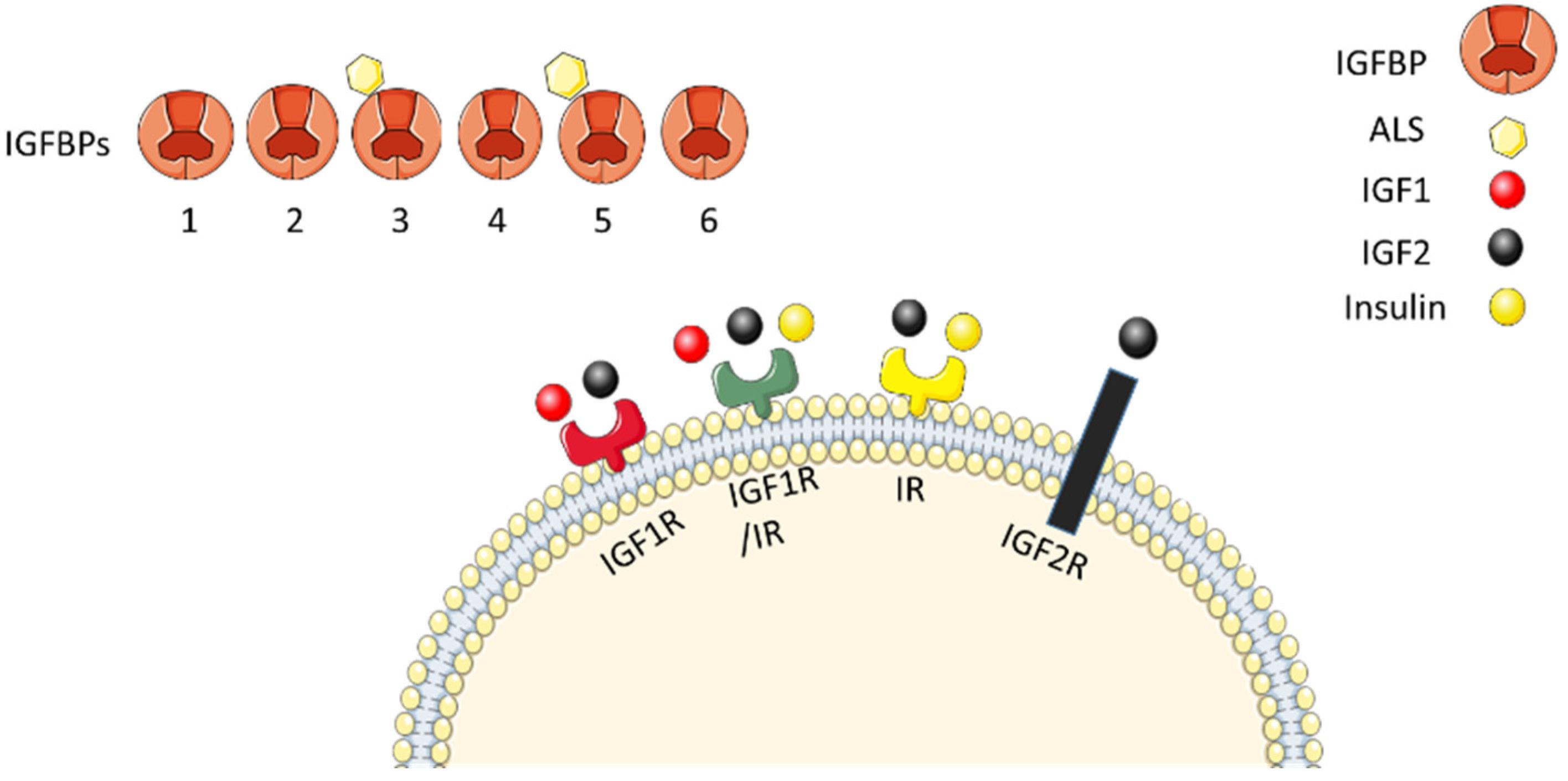
6.2.1. IGF Axis
6.2.2. Roles of Different IGF Axis Proteins in Osteogenesis
6.2.3. Expression of IGF Axis in the Periodontium
6.2.4. IGF Axis in Periodontal Regeneration
6.2.5. Effect of IGF Axis on PDL Cells
6.2.6. Effect of IGF Axis on BM-MSCs
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Carmagnola, D.; Pellegrini, G.; Dellavia, C.; Rimondini, L.; Varoni, E. Tissue engineering in periodontology: Biological mediators for periodontal regeneration. Int. J. Artif. Organs 2019, 42, 241–257. [Google Scholar] [CrossRef]
- Peng, B.; Dubey, N.K.; Mishra, V.K.; Tsai, F.; Dubey, R.; Deng, W.; Wei, H. Addressing Stem Cell Therapeutic Approaches in Pathobiology of Diabetes and Its Complications. J. Diabetes Res. 2018, 2018, 7806435. [Google Scholar] [CrossRef] [PubMed]
- Preshaw, P.M.; Alba, A.L.; Herrera, D.; Jepsen, S.; Konstantinidis, A.; Makrilakis, K.; Taylor, R. Periodontitis and diabetes: A two-way relationship. Diabetologia 2012, 55, 21–31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Villar, C.C.; Cochran, D.L. Regeneration of Periodontal Tissues: Guided Tissue Regeneration. Dent. Clin. N. Am. 2010, 54, 73–92. [Google Scholar] [CrossRef] [PubMed]
- Friedrich, N.; Thuesen, B.; Jrøgensen, T.; Juul, A.; Spielhagen, C.; Wallaschofksi, H.; Linneberg, A. The association between IGF-I and insulin resistance: A general population study in Danish adults. Diabetes Care 2012, 35, 768–773. [Google Scholar] [CrossRef] [Green Version]
- Dixit, M.; Poudel, S.B.; Yakar, S. Effects of GH/IGF axis on bone and cartilage. Mol. Cell. Endocrinol. 2021, 519, 111052. [Google Scholar] [CrossRef] [PubMed]
- Alkharobi, H.; Alhodhodi, A.; Hawsawi, Y.; Alkafaji, H.; Devine, D.; El-gendy, R.; Beattie, J. IGFBP-2 and -3 co-ordinately regulate IGF1 induced matrix mineralisation of differentiating human dental pulp cells. Stem Cell Res. 2016, 17, 517–522. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alkharobi, H.E.; Al-Khafaji, H.; Beattie, J.; Devine, D.A.; El-Gendy, R. Insulin-Like Growth Factor Axis Expression in Dental Pulp Cells Derived From Carious Teeth. Front. Bioeng. Biotechnol. 2018, 6, 36. [Google Scholar] [CrossRef] [Green Version]
- Al-Khafaji, H.; Noer, P.R.; Alkharobi, H.; Alhodhodi, A.; Meade, J.; El-Gendy, R.; Oxvig, C.; Beattie, J. A characteristic signature of insulin-like growth factor (IGF) axis expression during osteogenic differentiation of human dental pulp cells (hDPCs): Potential co-ordinated regulation of IGF action. Growth Horm. IGF Res. 2018, 42–43, 14–21. [Google Scholar] [CrossRef]
- Zheng, J.; Chen, S.; Albiero, M.L.; Vieira, G.H.A.; Wang, J.; Feng, J.Q.; Graves, D.T. Diabetes Activates Periodontal Ligament Fibroblasts via NF-κB In Vivo. J. Dent. Res. 2018, 97, 580–588. [Google Scholar] [CrossRef]
- Hajishengallis, G. Periodontitis: From microbial immune subversion to systemic inflammation. Nat. Rev. Immunol. 2015, 15, 30–44. [Google Scholar] [CrossRef]
- Kao, R.T.; Dietrich, T.; Garcia, R.; Herrera, D.; Fine, D.H.; Flemmig, T.F.; Kinane, D.F.; Kebschull, M. Periodontitis: Consensus report of workgroup 2 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant. J. Periodontal Res. 2018, 89 (Suppl. S1), S173–S182. [Google Scholar] [CrossRef] [Green Version]
- Frencken, J.E.; Sharma, P.; Stenhouse, L.; Green, D.; Laverty, D.; Dietrich, T. Global epidemiology of dental caries and severe periodontitis – a comprehensive review. J. Clin. Periodontol. 2017, 44 (Suppl. S18), S94–S105. [Google Scholar] [CrossRef] [PubMed]
- Socransky, S.S.; Haffajee, A.D.; Cugini, M.A.; Smith, C.K.J.R. Microbial complexes in subgingival plaque. J. Clin. Periodontol. 1998, 25, 134–144. [Google Scholar] [CrossRef]
- Griffen, A.L.; Beall, C.J.; Campbell, J.H.; Firestone, N.D.; Kumar, P.S.; Yang, Z.K.; Podar, M.; Leys, E.J. Distinct and complex bacterial profiles in human periodontitis and health revealed by 16S pyrosequencing. ISME J. 2012, 6, 1176–1185. [Google Scholar] [CrossRef] [Green Version]
- Hajishengallis, G. The inflammophilic character of the periodontitis-associated microbiota. Mol. Oral Microbiol. 2014, 29, 248–257. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hajishengallis, G. Immunomicrobial pathogenesis of periodontitis: Keystones, pathobionts, and host response. Trends Immunol. 2014, 35, 3–11. [Google Scholar] [CrossRef] [Green Version]
- Hajishengallis, G.; Korostoff, J.M. Revisiting the Page & Schroeder model: The good, the bad and the unknowns in the periodontal host response 40 years later. Periodontol. 2000 2017, 75, 116–151. [Google Scholar] [CrossRef]
- Sculean, A.; Stavropoulos, A.; Bosshardt, D.D. Self-regenerative capacity of intra-oral bone defects. J. Clin. Periodontol. 2019, 46 (Suppl. S21), 70–81. [Google Scholar] [CrossRef] [Green Version]
- Könönen, E.; Gursoy, M.; Gursoy, U.K. Periodontitis: A Multifaceted Disease of Tooth-Supporting Tissues. J. Clin. Med. 2019, 8, 1135. [Google Scholar] [CrossRef] [Green Version]
- Beck, J.D.; Papapanou, P.N.; Philips, K.H.; Offenbacher, S. Periodontal Medicine: 100 Years of Progress. J. Dent. Res. 2019, 98, 1053–1062. [Google Scholar] [CrossRef]
- Monsarrat, P.; Blaizot, A.; Kémoun, P.; Ravaud, P.; Nabet, C.; Sixou, M.; Vergnes, J.N. Clinical research activity in periodontal medicine: A systematic mapping of trial registers. J. Clin. Periodontol. 2016, 43, 390–400. [Google Scholar] [CrossRef]
- Retzepi, M.; Calciolari, E.; Wall, I.; Lewis, M.P.; Donos, N. The effect of experimental diabetes and glycaemic control on guided bone regeneration: Histology and gene expression analyses. Clin. Oral Implants Res. 2018, 29, 139–154. [Google Scholar] [CrossRef]
- Care, D.; Suppl, S.S. 2. Classification and diagnosis of diabetes: Standards of medical care in diabetesd—2019. Diabetes Care 2019, 42 (Suppl. S1), S13–S28. [Google Scholar] [CrossRef] [Green Version]
- WHO Classification of Diabetes Mellitus; WHO: Geneva, Switzerland, 2019.
- Donath, M.Y.; Shoelson, S.E. Type 2 diabetes as an inflammatory disease. Nat. Rev. Immunol. 2011, 11, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Lontchi-Yimagou, E.; Sobngwi, E.; Matsha, T.E.; Kengne, A.P. Diabetes mellitus and inflammation. Curr. Diab. Rep. 2013, 13, 435–444. [Google Scholar] [CrossRef] [PubMed]
- Calle, M.C.; Fernandez, M.L. Inflammation and type 2 diabetes. Diabetes Metab. 2012, 38, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Xiong, M.; Hung, B.; Chen, H. Advanced Glycation End Products Upregulate the Endoplasmic Reticulum Stress in Human Periodontal Ligament Cells. J. Periodontol. 2014, 86, 440–447. [Google Scholar] [CrossRef]
- Miranda, C.; Giner, M.; José Montoya, M.; Vázquez, M.A.; Miranda, M.J.; Pérez-Cano, R. Influence of high glucose and advanced glycation end-products (ages) levels in human osteoblast-like cells gene expression. BMC Musculoskelet. Disord. 2016, 17, 377. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, Y.Y.; Xiao, E.; Graves, D.T. Diabetes mellitus related bone metabolism and periodontal disease. Int. J. Oral Sci. 2015, 7, 63–72. [Google Scholar] [CrossRef] [Green Version]
- Fadini, G.P.; Ciciliot, S.; Albiero, M. Concise Review: Perspectives and Clinical Implications of Bone Marrow and Circulating Stem Cell Defects in Diabetes. Stem Cells 2017, 35, 106–116. [Google Scholar] [CrossRef] [PubMed]
- Loomans, C.J.M.; Van Haperen, R.; Duijs, J.M.; Verseyden, C.; De Crom, R.; Leenen, P.J.M.; Drexhage, H.A.; De Boer, H.C.; De Koning, E.J.P.; Rabelink, T.J.; et al. Differentiation of bone marrow-derived endothelial progenitor cells is shifted into a proinflammatory phenotype by hyperglycemia. Mol. Med. 2009, 15, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Negrato, C.A.; Tarzia, O.; Jovanovič, L.; Eduardo, L.; Chinellato, M. Periodontal disease and diabetes mellitus. J. App. Oral Sci. 2013, 21, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Kinane, D.F.; Stathopoulou, P.G.; Papapanou, P.N. Periodontal diseases. Nat. Rev. Dis. Prim. 2017, 3, 17038. [Google Scholar] [CrossRef] [PubMed]
- Kocher, T.; König, J.; Sylling, W.; Christiane, B.; Meisel, P.; Kocher, T. Periodontal complications of hyperglycemia/diabetes mellitus: Epidemiologic complexity and clinical challenge. Periodontol. 2000 2018, 78, 59–97. [Google Scholar] [CrossRef] [PubMed]
- Xiao, E.; Mattos, M.; Vieira, G.H.A.; Chen, S.; Corrêa, J.D.; Wu, Y.; Albiero, M.L.; Bittinger, K.; Graves, D.T. Diabetes Enhances IL-17 Expression and Alters the Oral Microbiome to Increase Its Pathogenicity. Cell Host Microbe 2017, 22, 120–128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shi, B.; Lux, R.; Klokkevold, P.; Chang, M.; Barnard, E.; Haake, S.; Li, H. The subgingival microbiome associated with periodontitis in type 2 diabetes mellitus. ISME J. 2020, 14, 519–530. [Google Scholar] [CrossRef]
- Sanz, M.; Ceriello, A.; Buysschaert, M.; Chapple, I.; Demmer, R.T.; Graziani, F.; Herrera, D.; Jepsen, S.; Lione, L.; Madianos, P.; et al. Scientific evidence on the links between periodontal diseases and diabetes: Consensus report and guidelines of the joint workshop on periodontal diseases and diabetes by the International diabetes Federation and the European Federation of Periodontology. J. Clin. Periodontol. 2018, 45, 138–149. [Google Scholar] [CrossRef]
- Patil, V.S.; Patil, V.P.; Gokhale, N.; Acharya, A.; Kangokar, P. Chronic periodontitis in type 2 diabetes mellitus: Oxidative stress as a common factor in periodontal tissue injury. J. Clin. Diagn. Res. 2016, 10, BC12–BC16. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.Y.; Wei, C.C.; Shang, T.T.; Lian, Q.; Wu, C.X.; Deng, J.Y. High glucose induces inflammatory cytokine through protein kinase C-induced toll-like receptor 2 pathway in gingival fibroblasts. Biochem. Biophys. Res. Commun. 2012, 427, 666–670. [Google Scholar] [CrossRef]
- Katz, J.; Bhattacharyya, I.; Farkhondeh-Kish, F.; Perez, F.M.; Caudle, R.M.; Heft, M.W. Expression of the receptor of advanced glycation end products in gingival tissues of type 2 diabetes patients with chronic periodontal disease: A study utilizing immunohistochemistry and RT-PCR. J. Clin. Periodontol. 2005, 32, 40–44. [Google Scholar] [CrossRef] [PubMed]
- Graves, D.T.; Ding, Z.; Yang, Y. The impact of diabetes on periodontal diseases. Periodontol. 2000 2020, 82, 214–224. [Google Scholar] [CrossRef] [PubMed]
- Graziani, F.; Gennai, S.; Solini, A.; Petrini, M. A systematic review and meta-analysis of epidemiologic observational evidence on the effect of periodontitis on diabetes An update of the EFP-AAP review. J. Clin. Periodontol. 2018, 45, 167–187. [Google Scholar] [CrossRef] [PubMed]
- Isola, G.; Matarese, G.; Ramaglia, L.; Pedullà, E.; Rapisarda, E.; Iorio-Siciliano, V. Association between periodontitis and glycosylated haemoglobin before diabetes onset: A cross-sectional study. Clin. Oral Investig. 2020, 24, 2799–2808. [Google Scholar] [CrossRef]
- Miranda, T.S.; Heluy, S.L.; Cruz, D.F.; da Silva, H.D.P.; Feres, M.; Figueiredo, L.C.; Duarte, P.M. The ratios of pro-inflammatory to anti-inflammatory cytokines in the serum of chronic periodontitis patients with and without type 2 diabetes and/or smoking habit. Clin. Oral Investig. 2019, 23, 641–650. [Google Scholar] [CrossRef] [PubMed]
- Merchant, A.T.; Georgantopoulos, P.; Howe, C.J.; Virani, S.S.; Morales, D.A.; Haddock, K.S. Effect of long-term periodontal care on hemoglobin a1c in type 2 diabetes. J. Dent. Res. 2016, 95, 408–415. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nazir, M.A. Prevalence of periodontal disease, its association with systemic diseases and prevention. Int. J. Health Sci. 2017, 1, 72–80. [Google Scholar] [CrossRef]
- Mougeot, J.L.C.; Stevens, C.B.; Paster, B.J.; Brennan, M.T.; Lockhart, P.B.; Mougeot, F.K.B. Porphyromonas gingivalis is the most abundant species detected in coronary and femoral arteries. J. Oral Microbiol. 2017, 9, 1281562. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kramer, C.D.; Simas, A.M.; He, X.; Ingalls, R.R.; Weinberg, E.O.; Attardo, C. Distinct roles for dietary lipids and Porphyromonas gingivalis infection on atherosclerosis progression and the gut microbiota. Anaerobe 2017, 45, 19–30. [Google Scholar] [CrossRef]
- Preshaw, P.M.; Bissett, S.M. Periodontitis and diabetes. Br. Dent. J. 2019, 227, 577–584. [Google Scholar] [CrossRef]
- Artese, H.P.C.; Foz, A.M.; Rabelo, M.D.S.; Gomes, G.H.; Orlandi, M.; Suvan, J.; D’Aiuto, F.; Romito, G.A. Periodontal therapy and systemic inflammation in type 2 diabetes mellitus: A meta-analysis. PLoS ONE 2015, 10, e0128344. [Google Scholar] [CrossRef] [Green Version]
- Preshaw, P.M.; Taylor, J.J.; Jaedicke, K.M.; De Jager, M.; Bikker, J.W.; Selten, W.; Bissett, S.M.; Whall, K.M.; van de Merwe, R.; Areibi, A.; et al. Treatment of periodontitis reduces systemic inflammation in type 2 diabetes. J. Clin. Periodontol. 2020, 4, 737–746. [Google Scholar] [CrossRef] [PubMed]
- Peng, C.H.; Yang, Y.S.; Chan, K.C.; Kornelius, E.; Chiou, J.Y.; Huang, C.N. Periodontal treatment and the risks of cardiovascular disease in patients with type 2 diabetes: A retrospective cohort study. Intern. Med. 2017, 56, 1015–1021. [Google Scholar] [CrossRef] [Green Version]
- Slots, J. Periodontitis: Facts, fallacies and the future. Periodontol. 2000 2017, 75, 7–23. [Google Scholar] [CrossRef] [PubMed]
- Nuñez, J.; Vignoletti, F.; Caffesse, R.G.; Sanz, M. Cellular therapy in periodontal regeneration. Periodontol. 2000 2019, 79, 107–116. [Google Scholar] [CrossRef]
- Cobb, C.M. Clinical significance of non-surgical periodontal therapy: An evidence-based perspective of scaling and root planing. J. Clin. Periodontol. 2002, 29, 22–32. [Google Scholar] [CrossRef] [Green Version]
- Lang, N.P.; Salvi, G.E.; Sculean, A. Nonsurgical therapy for teeth and implants—When and why? Periodontol. 2000 2019, 79, 15–21. [Google Scholar] [CrossRef]
- Naiff, P.; Carneiro, V.; Guimarães, M.D.C. Importance of mechanical periodontal therapy in patients with diabetes type 2 and periodontitis. Int. J. Dent. 2018, 2018, 6924631. [Google Scholar] [CrossRef]
- Rees, T.S. Periodontal management of the patient with diabetes mellitus. Periodontol. 2000 2000, 23, 63–72. [Google Scholar] [CrossRef]
- Duarte, P.M.; Feres, M.; Yassine, L.L.S.; Soares, G.M.S.; Miranda, T.S.; Faveri, M.; Retamal-Valdes, B.; Figueiredo, L.C. Clinical and microbiological effects of scaling and root planing, metronidazole and amoxicillin in the treatment of diabetic and non-diabetic subjects with periodontitis: A cohort study. J. Clin. Periodontol. 2018, 45, 1326–1335. [Google Scholar] [CrossRef]
- Christgau, M.; Palitzsch, K.D.; Schmalz, G.; Kreiner, U.; Frenzel, S. Healing response to non-surgical periodontal therapy in patients with diabetes mellitus: Clinical, microbiological, and immunologic results. J. Clin. Periodontol. 1998, 25, 112–124. [Google Scholar] [CrossRef]
- Hsu, Y.T.; Nair, M.; Angelov, N.; Lalla, E.; Lee, C.T. Impact of diabetes on clinical periodontal outcomes following non-surgical periodontal therapy. J. Clin. Periodontol. 2019, 46, 206–217. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Liao, Y.; Luo, B.; Li, L.; Zhang, Y.; Yan, F. Non-surgical Periodontal Treatment Restored the Gut Microbiota and Intestinal Barrier in Apolipoprotein E−/− Mice With Periodontitis. Front. Cell. Infect. Microbiol. 2020, 10, 498. [Google Scholar] [CrossRef]
- Sallum, E.A.; Ribeiro, F.V.; Ruiz, K.S.; Sallum, A.W. Experimental and clinical studies on regenerative periodontal therapy. Periodontol. 2000 2019, 79, 22–55. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.C.; Lin, L.K.; Song, C.J.; Su, Y.X.; Tu, Y.K. Comparisons of periodontal regenerative therapies: A meta-analysis on the long-term efficacy. J. Clin. Periodontol. 2017, 44, 511–519. [Google Scholar] [CrossRef] [PubMed]
- Moreno Rodríguez, J.A.; Ortiz Ruiz, A.J.; Caffesse, R.G. Supra-alveolar attachment gain in the treatment of combined intra-suprabony periodontal defects by non-incised papillae surgical approach. J. Clin. Periodontol. 2019, 46, 927–936. [Google Scholar] [CrossRef]
- Jayakumar, A.; Rohini, S.; Naveen, A.; Haritha, A.; Reddy, K. Horizontal alveolar bone loss: A periodontal orphan. J. Indian Soc. Periodontol. 2010, 14, 181. [Google Scholar] [CrossRef] [PubMed]
- Iorio-Siciliano, V.; Blasi, A.; Stratul, S.I.; Ramaglia, L.; Octavia, V.; Salvi, G.E.; Sculean, A. Healing of periodontal suprabony defects following treatment with open flap debridement with or without an enamel matrix derivative: A randomized controlled clinical study. Clin. Oral Investig. 2021, 25, 1019–1027. [Google Scholar] [CrossRef]
- Susin, C.; Fiorini, T.; Lee, J.; De Stefano, J.A.; Dickinson, D.P.; Wikesjö, U.M.E. Wound healing following surgical and regenerative periodontal therapy. Periodontol. 2000 2015, 68, 83–98. [Google Scholar] [CrossRef] [PubMed]
- Graziani, F.; Gennai, S.; Cei, S.; Ducci, F.; Discepoli, N.; Carmignani, A.; Tonetti, M. Does enamel matrix derivative application provide additional clinical benefits in residual periodontal pockets associated with suprabony defects? A systematic review and meta-analysis of randomized clinical trials. J. Clin. Periodontol. 2014, 41, 377–386. [Google Scholar] [CrossRef]
- Cho, Y.D.; Kim, K.H.; Lee, Y.M.; Ku, Y.; Seol, Y.J. Periodontal wound healing and tissue regeneration: A narrative review. Pharmaceuticals 2021, 14, 456. [Google Scholar] [CrossRef]
- Lee, R.S.B.; Hamlet, S.M.; Ivanovski, S. The influence of titanium surface characteristics on macrophage phenotype polarization during osseous healing in type I diabetic rats: A pilot study. Clin. Oral Implants Res. 2017, 28, e159–e168. [Google Scholar] [CrossRef]
- Retzepi, M.; Lewis, M.P. Effect of diabetes and metabolic control on de novo bone formation following guided bone regeneration. Clin. Oral Implant Res. 2010, 21, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Corrêa, M.G.; Gomes Campos, M.L.; Marques, M.R.; Casati, M.Z.; Nociti, F.H.; Sallum, E.A. Histometric Analysis of the Effect of Enamel Matrix Derivative on the Healing of Periodontal Defects in Rats With Diabetes. J. Periodontol. 2013, 84, 1309–1318. [Google Scholar] [CrossRef]
- Shirakata, Y.; Eliezer, M.; Ce, N.; Weinreb, M.; Dard, M.; Sculean, A. Periodontal healing after application of enamel matrix derivative in surgical supra/ infrabony periodontal defects in rats with streptozotocin-induced diabetes. J. Periodontal Res. 2014, 49, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Takeda, K.; Mizutani, K.; Matsuura, T.; Kido, D.; Mikami, R.; Noda, M.; Buranasin, P.; Sasaki, Y.; Izumi, Y. Periodontal regenerative effect of enamel matrix derivative in diabetes. PLoS One 2018, 13, e0207201. [Google Scholar] [CrossRef] [PubMed]
- Nemtoi, A.; Danila, V.; Dragan, E.; Pasca, S.; Nemtoi, A.; Constantin, M.; Sava, A.; Haba, D. The Effects of Insulin and Strontium Ranelate on Guided Bone Regeneration in Diabetic Rats. Rev. Chim. 2017, 68, 693–697. [Google Scholar] [CrossRef]
- Lee, S.B.; Retzepi, M.; Petrie, A.; Hakimi, A.R.; Schwarz, F.; Donos, N. The effect of diabetes on bone formation following application of the GBR principle with the use of titanium domes. Clin. Oral Implants Res. 2013, 24, 28–35. [Google Scholar] [CrossRef]
- Seshima, F.; Nishina, M.; Namba, T.; Saito, A. Periodontal Regenerative Therapy in Patient with Chronic Periodontitis and Type 2 Diabetes Mellitus: A Case Report. Bull. Tokyo Dent. Coll. 2016, 57, 97–104. [Google Scholar] [CrossRef] [Green Version]
- Mizutani, K.; Shioyama, H.; Matsuura, T.; Mikami, R.; Takeda, K.; Izumi, Y.; Aoki, A.; Iwata, T. Periodontal regenerative therapy in patients with type 2 diabetes using minimally invasive surgical technique with enamel matrix derivative under 3-year observation: A prospective cohort study. J. Periodontol. 2021, 92, 1262–1273. [Google Scholar] [CrossRef] [PubMed]
- Cimões, R.; Santiago, L.M.; de França Caldas Júnior, A.; de Carvalho Farias Vajgel, B.; Perussolo, J.; Donos, N. Treatment of intrabony periodontal defects in controlled diabetic patients with an enamel matrix derivative: A split-mouth randomized clinical trial. Clin. Oral Investig. 2021, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Langer, R.; Vacanti, J.P. Tissue Engineering. Science 1993, 260, 920–926. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, C.H. Biomaterial-based approaches for regeneration of periodontal ligament and cementum using 3D platforms. Int. J. Mol. Sci. 2019, 20, 4364. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bartold, P.M.; Gronthos, S. Standardization of Criteria Defining Periodontal Ligament Stem Cells. J. Dent. Res. 2017, 96, 487–490. [Google Scholar] [CrossRef]
- Kargozar, S.; Mozafari, M.; Hamzehlou, S.; Milan, P.B.; Hae-Won, K.; Baino, F. Bone Tissue Engineering Using Human Cells: A Comprehensive Review on Recent Trends, Current Prospects, and Recommendations. Appl. Sci. 2019, 9, 174. [Google Scholar] [CrossRef] [Green Version]
- Radwan, I.A.; Rady, D.; Abbass, M.M.S.; El Moshy, S.; Abubakr, N.; Dörfer, C.E.; Fawzy El-Sayed, K.M. Induced Pluripotent Stem Cells in Dental and Nondental Tissue Regeneration: A Review of an Unexploited Potential. Stem Cells Int. 2020, 2020, 1941629. [Google Scholar] [CrossRef] [Green Version]
- Wang, P.; Ma, T.; Guo, D.; Hu, K.; Shu, Y.; Xu, H.H.K.; Schneider, A. Metformin induces osteoblastic differentiation of human induced pluripotent stem cell-derived mesenchymal stem cells. J. Tissue Eng. Regen. Med. 2018, 12, 437–446. [Google Scholar] [CrossRef]
- Duan, X.; Tu, Q.; Zhang, J.; Ye, J.; Sommer, C.; Mostoslavsky, G.; Kaplan, D.; Yang, P.; Chen, J. Application of induced pluripotent stem (iPS) cells in periodontal tissue regeneration. J. Cell. Physiol. 2011, 226, 150–157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, M.S.; Kannan, V.; De Vries, A.E.; Czepiel, M. Characterization and comparison of osteoblasts derived from mouse embryonic stem cells and induced pluripotent stem cells. J. Bone Miner. Metab. 2017, 35, 20–29. [Google Scholar] [CrossRef]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.C.; Krause, D.S.; Deans, R.J.; Keating, A.; Prockop, D.J.; Horwitz, E.M. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Monjaraz, B.; Santiago-Osorio, E.; Monroy-García, A.; Ledesma-Martínez, E.; Mendoza-Núñez, V.M. Mesenchymal stem cells of dental origin for inducing tissue regeneration in periodontitis: A mini-review. Int. J. Mol. Sci. 2018, 19, 944. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mozaffari, M.S.; Emami, G.; Khodadadi, H.; Baban, B. Stem cells and tooth regeneration: Prospects for personalized dentistry. EPMA J. 2019, 10, 31–42. [Google Scholar] [CrossRef] [PubMed]
- Volponi, A.A.; Sharpe, P.T. The tooth—A treasure chest of stem cells. Br. Dent. J. 2013, 215, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Q.; Dong, Z.; Wang, W.; Li, B.; Jin, Y. Dental stem cell and dental tissue regeneration. Front. Med. 2019, 13, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.Y.; Li, X.; Wang, J.; He, X.T.; Sun, H.H.; Chen, F.M. Concise Review: Periodontal Tissue Regeneration Using Stem Cells: Strategies and Translational Considerations. Stem Cells Transl. Med. 2019, 8, 392–403. [Google Scholar] [CrossRef] [Green Version]
- Wang, Q.; Zhao, G.; Xing, Z.; Zhan, J.; Ma, J.I.E. Comparative evaluation of the osteogenic capacity of human mesenchymal stem cells from bone marrow and umbilical cord tissue. Exp. Ther. Med. 2019, 17, 764–772. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aurora, A.B.; Olson, E.N. Immune modulation of stem cells and regeneration. Cell Stem Cell 2014, 15, 14–25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, J.H.; Jo, C.H.; Kim, H.R.; Hwang, Y. Il Comparison of immunological characteristics of mesenchymal stem cells from the periodontal ligament, umbilical cord, and adipose tissue. Stem Cells Int. 2018, 2018, 8429042. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Liu, Z.L.; Xiao, M.; Yang, Z.Z.; Peng, M.Z.; Di Li, C.; Zhou, X.J.; Wang, J.W. Impact of bone marrow mesenchymal stem cell immunomodulation on the osteogenic effects of laponite. Stem Cell Res. Ther. 2018, 9, 100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seo, B.M.; Miura, M.; Gronthos, S.; Bartold, P.M.; Batouli, S.; Brahim, J.; Young, M.; Robey, P.G.; Wang, C.Y.; Shi, S. Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet 2004, 364, 149–155. [Google Scholar] [CrossRef]
- Tomokiyo, A.; Wada, N.; Maeda, H. Periodontal Ligament Stem Cells: Regenerative Potency in Periodontium. Stem Cells Dev. 2019, 28, 974–985. [Google Scholar] [CrossRef]
- Trubiani, O.; Pizzicannella, J.; Caputi, S.; Marchisio, M.; Mazzon, E.; Paganelli, R.; Paganelli, A.; Diomede, F. Periodontal Ligament Stem Cells: Current Knowledge and Future Perspectives. Stem Cells Dev. 2019, 28, 995–1003. [Google Scholar] [CrossRef]
- Zhu, W.; Liang, M. Periodontal ligament stem cells: Current status, concerns, and future prospects. Stem Cells Int. 2015, 2015, 972313. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, J.; An, Y.; Gao, L.N.; Zhang, Y.J.; Jin, Y.; Chen, F.M. The effect of aging on the pluripotential capacity and regenerative potential of human periodontal ligament stem cells. Biomaterials 2012, 33, 6974–6986. [Google Scholar] [CrossRef] [PubMed]
- Trubiani, O.; Zalzal, S.F.; Paganelli, R.; Marchisio, M.; Giancola, R.; Pizzicannella, J. Expression Profile of the Embryonic Markers Nanog, Frizzled-9 Receptor in Human Periodontal Ligament Mesenchymal Stem Cells. J. Cell. Physiol. 2010, 225, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Tomokiyo, A.; Yoshida, S.; Hamano, S.; Hasegawa, D.; Sugii, H.; Maeda, H. Detection, Characterization, and Clinical Application of Mesenchymal Stem Cells in Periodontal Ligament Tissue. Stem Cells Int. 2018, 2018, 5450768. [Google Scholar] [CrossRef]
- Gay, I.; Cavender, A.; Peto, D.; Sun, Z.; Speer, A.; Cao, H.; Amendt, B.A. Differentiation of human dental stem cells reveals a role for microRNA-218. J. Periodontal Res. 2014, 49, 110–120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Albiero, M.L.; Amorim, B.R.; Martins, L.; Casati, M.Z.; Sallum, E.A.; Nociti, F.H.; Silvério, K.G. Exposure of periodontal ligament progenitor cells to lipopolysaccharide from escherichia coli changes osteoblast differentiation pattern. J. Appl. Oral Sci. 2015, 23, 145–152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Volponi, A.A.; Gentleman, E.; Fatscher, R.; Pang, Y.W.Y.; Gentleman, M.M.; Sharpe, P.T. Composition of mineral produced by dental mesenchymal stem cells. J. Dent. Res. 2015, 94, 1568–1574. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mukhtar, A.H.; Alqutub, M.N. Osteogenic Potential of Periodontal Ligament Stem Cells Cultured in Osteogenic and Regular Growth Media: Confocal and Scanning Electron Microscope Study. J. Contemp. Dent. Pract. 2020, 21, 776–780. [Google Scholar] [CrossRef]
- Yamada, S.; Tauchi, T.; Awata, T.; Maeda, K.; Kajikawa, T.; Yanagita, M.; Murakami, S. Characterization of a novel periodontal ligament-specific periostin isoform. J. Dent. Res. 2014, 93, 891–897. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Komaki, M.; Iwasaki, K.; Arzate, H.; Narayanan, A.S.; Izumi, Y.; Morita, I. Cementum protein 1 (CEMP1) induces a cementoblastic phenotype and reduces osteoblastic differentiation in periodontal ligament cells. J. Cell. Physiol. 2012, 227, 649–657. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.-M.; Zhao, Y.-M.; Wu, H.; Deng, Z.-H.; Wang, Q.-T.; Zhou, W.; Liu, Q.; Dong, G.-Y.; Li, K.; Wu, Z.-F.; et al. Enhancement of periodontal tissue regeneration by locally controlled delivery of insulin-like growth factor-I from dextran-co-gelatin microspheres. J. Control. Release 2006, 114, 209–222. [Google Scholar] [CrossRef] [PubMed]
- Wada, N.; Menicanin, D.; Shi, S.; Bartold, P.M.; Gronthos, S. Immunomodulatory properties of human periodontal ligament stem cells. J. Cell. Physiol. 2009, 219, 667–676. [Google Scholar] [CrossRef] [PubMed]
- Shin, C.; Kim, M.; Han, J.A.; Choi, B.; Hwang, D.; Do, Y.; Yun, J.H. Human periodontal ligament stem cells suppress T-cell proliferation via down-regulation of non-classical major histocompatibility complex-like glycoprotein CD1b on dendritic cells. J. Periodontal Res. 2017, 52, 135–146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tang, R.; Wei, F.; Wei, L.; Wang, S.; Ding, G. Osteogenic differentiated periodontal ligament stem cells maintain their immunomodulatory capacity. J. Tissue Eng. Regen. Med. 2014, 8, 226–232. [Google Scholar] [CrossRef]
- Benatti, B.B.; Silvério, K.G.; Casati, M.Z.; Sallum, E.A.; Nociti, F.H. Inflammatory and bone-related genes are modulated by aging in human periodontal ligament cells. Cytokine 2009, 46, 176–181. [Google Scholar] [CrossRef] [PubMed]
- Itaya, T.; Kagami, H.; Okada, K.; Yamawaki, A.; Narita, Y.; Inoue, M.; Sumita, Y.; Ueda, M. Characteristic changes of periodontal ligament-derived cells during passage. J. Periodontal Res. 2009, 44, 425–433. [Google Scholar] [CrossRef]
- Tang, H.N.; Xia, Y.; Yu, Y.; Wu, R.X.; Gao, L.N.; Chen, F.M. Stem cells derived from “inflamed” and healthy periodontal ligament tissues and their sheet functionalities: A patient-matched comparison. J. Clin. Periodontol. 2016, 43, 72–84. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Tang, H.-N.; Wu, R.-X.; Yu, Y.; Gao, L.-N.; Chen, F.-M. Cell Responses to Conditioned Media Produced by Patient-Matched Stem Cells Derived From Healthy and Inflamed Periodontal Ligament Tissues. J. Periodontol. 2015, 87, e53–e63. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Xia, Y.; Xu, J.; Tian, B.; Zhang, X.; Chen, F. Assessment of cellular materials generated by co-cultured ‘inflamed’ and healthy periodontal ligament stem cells from patient-matched groups. Exp. Cell Res. 2016, 346, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Kato, H.; Taguchi, Y.; Tominaga, K.; Umeda, M.; Tanaka, A. Porphyromonas gingivalis LPS inhibits osteoblastic differentiation and promotes pro-inflammatory cytokine production in human periodontal ligament stem cells. Arch. Oral Biol. 2014, 59, 167–175. [Google Scholar] [CrossRef] [PubMed]
- Keong, J.Y.; Low, L.W.; Chong, J.M.; Ong, Y.Y.; Pulikkotil, S.J.; Singh, G.; Nagendrababu, V.; Banavar, S.R.; Khoo, S.P. Effect of lipopolysaccharide on cell proliferation and vascular endothelial growth factor secretion of periodontal ligament stem cells. Saudi Dent. J. 2020, 32, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Diomede, F.; Zingariello, M.; Cavalcanti, M.F.X.B.; Merciaro, I.; Pizzicannella, J.; de Isla, N.; Caputi, S.; Ballerini, P.; Trubiani, O. MyD88/ERK/NFkB pathways and pro-inflammatory cytokines release in Periodontal Ligament Stem Cells stimulated by Porphyromonas gingivalis. Eur. J. Histochem. 2017, 61, 122–127. [Google Scholar] [CrossRef] [Green Version]
- Iwasaki, K.; Akazawa, K.; Nagata, M.; Komaki, M.; Honda, I.; Morioka, C.; Yokoyama, N.; Ayame, H.; Yamaki, K.; Tanaka, Y.; et al. The fate of transplanted periodontal ligament stem cells in surgically created periodontal defects in rats. Int. J. Mol. Sci. 2019, 20, 192. [Google Scholar] [CrossRef] [Green Version]
- Iwata, T.; Yamato, M.; Washio, K.; Yoshida, T. Periodontal regeneration with autologous periodontal ligament- derived cell sheets e A safety and ef fi cacy study in ten patients. Regen. Ther. 2018, 9, 38–44. [Google Scholar] [CrossRef]
- Chen, F.M.; Gao, L.N.; Tian, B.M.; Zhang, X.Y.; Zhang, Y.J.; Dong, G.Y.; Lu, H.; Chu, Q.; Xu, J.; Yu, Y.; et al. Treatment of periodontal intrabony defects using autologous periodontal ligament stem cells: A randomized clinical trial. Stem Cell Res. Ther. 2016, 7, 33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sánchez, N.; Fierravanti, L.; Núñez, J.; Vignoletti, F.; González-Zamora, M.; Santamaría, S.; Suárez-Sancho, S.; Fernández-Santos, M.E.; Figuero, E.; Herrera, D.; et al. Periodontal regeneration using a xenogeneic bone substitute seeded with autologous periodontal ligament-derived mesenchymal stem cells: A 12-month quasi-randomized controlled pilot clinical trial. J. Clin. Periodontol. 2020, 47, 1391–1402. [Google Scholar] [CrossRef]
- Friedenstein, A.J.; Chailakhjan, R.K.; Lalykina, K. The Development Of Fibroblast Colonies In Monolayer Cultures Of Guinea-Pig Bone Marrow And Spleen Cells. Cell Tissue Kinet. 1970, 3, 393–403. [Google Scholar] [CrossRef] [PubMed]
- Pittenger, M.F.; Mackay, A.M.; Beck, S.C.; Jaiswal, R.K.; Douglas, R.; Mosca, J.D.; Moorman, M.A.; Simonetti, D.W.; Craig, S.; Marshak, D.R. Multilineage Potential of Adult Human Mesenchymal Stem Cells. Science 1999, 284, 143–147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chan, C.K.F.; Gulati, G.S.; Sinha, R.; Tompkins, J.V.; Lopez, M.; Carter, A.C.; Ransom, R.C.; Reinisch, A.; Wearda, T.; Murphy, M.; et al. Identification of the Human Skeletal Stem Cell. Cell 2018, 175, 43–56.e21. [Google Scholar] [CrossRef] [Green Version]
- Dawson, J.I.; Kanczler, J.; Tare, R.; Kassem, M.; Oreffo, R.O.C. Concise review: Bridging the gap: Bone regeneration using skeletal stem cell-based strategies-where are we now? Stem Cells 2014, 32, 35–44. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gómez-Barrena, E.; Rosset, P.; Gebhard, F.; Hernigou, P.; Baldini, N.; Rouard, H.; Sensebé, L.; Gonzalo-Daganzo, R.M.; Giordano, R.; Padilla-Eguiluz, N.; et al. Feasibility and safety of treating non-unions in tibia, femur and humerus with autologous, expanded, bone marrow-derived mesenchymal stromal cells associated with biphasic calcium phosphate biomaterials in a multicentric, non-comparative trial. Biomaterials 2019, 196, 100–108. [Google Scholar] [CrossRef]
- Redondo, L.M.; García, V.; Peral, B.; Verrier, A.; Becerra, J.; Sánchez, A.; García-Sancho, J. Repair of maxillary cystic bone defects with mesenchymal stem cells seeded on a cross-linked serum scaffold. J. Cranio-Maxillofac. Surg. 2018, 46, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, N.; Kawaguchi, H.; Hirachi, A.; Takeda, K.; Mizuno, N.; Nishimura, M.; Koike, C.; Tsuji, K.; Iba, H.; Kato, Y.; et al. Behavior of Transplanted Bone Marrow–Derived Mesenchymal Stem Cells in Periodontal Defects. J. Periodontol. 2006, 77, 1003–1007. [Google Scholar] [CrossRef]
- Kawaguchi, H.; Hirachi, A.; Hasegawa, N.; Iwata, T.; Hamaguchi, H.; Shiba, H.; Takata, T.; Kato, Y.; Kurihara, H. Enhancement of Periodontal Tissue Regeneration by Transplantation of Bone Marrow Mesenchymal Stem Cells. J. Periodontol. 2004, 75, 1281–1287. [Google Scholar] [CrossRef] [PubMed]
- Baba, S.; Yamada, Y.; Komuro, A.; Yotsui, Y.; Umeda, M.; Shimuzutani, K.; Nakamura, S. Phase I/II Trial of Autologous Bone Marrow Stem Cell Transplantation with a Three-Dimensional Woven-Fabric Scaffold for Periodontitis. Stem Cells Int. 2016, 2016, 6205910. [Google Scholar] [CrossRef] [Green Version]
- Gay, I.C.; Chen, S.; MacDougall, M. Isolation and characterization of multipotent human periodontal ligament stem cells. Orthod. Craniofacial Res. 2007, 10, 149–160. [Google Scholar] [CrossRef]
- Gao, L.N.; An, Y.; Lei, M.; Li, B.; Yang, H.; Lu, H.; Chen, F.M.; Jin, Y. The effect of the coumarin-like derivative osthole on the osteogenic properties of human periodontal ligament and jaw bone marrow mesenchymal stem cell sheets. Biomaterials 2013, 34, 9937–9951. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Liu, S.; Zhu, B.; Xu, Q.; Ding, Y.; Jin, Y. Composite cell sheet for periodontal regeneration: Crosstalk between different types of MSCs in cell sheet facilitates complex periodontal-like tissue regeneration. Stem Cell Res. Ther. 2016, 7, 168. [Google Scholar] [CrossRef] [Green Version]
- Tsumanuma, Y.; Iwata, T.; Washio, K.; Yoshida, T.; Yamada, A.; Takagi, R.; Ohno, T.; Lin, K.; Yamato, M.; Ishikawa, I.; et al. Comparison of different tissue-derived stem cell sheets for periodontal regeneration in a canine 1-wall defect model. Biomaterials 2011, 32, 5819–5825. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-H.; Kim, K.-H.; Seo, B.-M.; Koo, K.-T.; Kim, T.-I.; Seol, Y.-J.; Ku, Y.; Rhyu, I.-C.; Chung, C.-P.; Lee, Y.-M. Alveolar Bone Regeneration by Transplantation of Periodontal Ligament Stem Cells and Bone Marrow Stem Cells in a Canine Peri-Implant Defect Model: A Pilot Study. J. Periodontol. 2009, 80, 1815–1823. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Li, Z.G.; Si, Y.M.; Chen, B.; Meng, J. The difference on the osteogenic differentiation between periodontal ligament stem cells and bone marrow mesenchymal stem cells under inflammatory microenviroments. Differentiation 2014, 88, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Konermann, A.; Guo, T.; Jäger, A.; Zhang, L.; Jin, Y. Canonical Wnt signaling differently modulates osteogenic differentiation of mesenchymal stem cells derived from bone marrow and from periodontal ligament under inflammatory conditions. Biochim. Biophys. Acta 2014, 1840, 1125–1134. [Google Scholar] [CrossRef]
- Assem, M.; Kamal, S.; Sabry, D.; Soliman, N.; Aly, R.M. Preclinical Assessment of the Proliferation Capacity of Gingival and Periodontal Ligament Stem Cells from Diabetic Patients. Maced. J. Med. Sci. 2018, 6, 254–259. [Google Scholar] [CrossRef]
- Liu, Q.; Hu, C.; Zhou, C.; Cui, X.; Yang, K.; Deng, C. DKK1 rescues osteogenic differentiation of mesenchymal stem cells isolated from periodontal ligaments of patients with diabetes mellitus induced periodontitis. Sci. Rep. 2015, 5, 13142. [Google Scholar] [CrossRef]
- Hobbs, H.C.; Rowe, D.J.; Johnson, P.W. Periodontal Ligament Cells From Insulin- Dependent Diabetics Exhibit Altered Alkaline Phosphatase Activity in Response to Growth Factors. J. Periodontol. 1999, 70, 736–742. [Google Scholar] [CrossRef] [PubMed]
- Kato, H.; Taguchi, Y.; Tominaga, K.; Kimura, D.; Yamawaki, I. High Glucose Concentrations Suppress the Proliferation of Human Periodontal Ligament Stem Cells and Their Differentiation Into Osteoblasts. J. Periodontol. 2016, 87, e44–e51. [Google Scholar] [CrossRef]
- Guo, Z.; Chen, R.; Zhang, F.; Ding, M.; Wang, P. Exendin-4 relieves the inhibitory effects of high glucose on the proliferation and osteoblastic differentiation of periodontal ligament stem cells. Arch. Oral Biol. 2018, 91, 9–16. [Google Scholar] [CrossRef]
- Guo, Z.; Gan, S.; Cao, C.; Fu, R.; Cao, S.; Xie, C.; Chen, J.; Gibson, A.; Zheng, X.; Teng, N. Advanced glycosylated end products restrain the osteogenic differentiation of the periodontal ligament stem cell. J. Dent. Sci. 2019, 14, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.N.; Wang, X.X.; Wang, Z.; Li, K.Y.; Xu, B.H.; Zhang, J. Berberine improves advanced glycation end products-induced osteogenic differentiation responses in human periodontal ligament stem cells through the canonical Wnt/β-catenin pathway. Mol. Med. Rep. 2019, 19, 5440–5452. [Google Scholar] [CrossRef] [Green Version]
- Doxey, D.L.; Nares, S.; Park, B.; Trieu, C.; Cutler, C.W.; Iacopino, A.M. Diabetes-induced impairment of macrophage cytokine release in a rat model: Potential role of serum lipids. Life Sci. 1998, 63, 1127–1136. [Google Scholar] [CrossRef]
- Zheng, D.H.; Han, Z.Q.; Wang, X.X.; Ma, D.; Zhang, J. Erythropoietin attenuates high glucose-induced oxidative stress and inhibition of osteogenic differentiation in periodontal ligament stem cell (PDLSCs). Chem. Biol. Interact. 2019, 305, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Sundaram, M.N.; Sowmya, S.; Deepthi, S.; Bumgardener, J.D.; Jayakumar, R. Bilayered construct for simultaneous regeneration of alveolar bone and periodontal ligament. J. Biomed. Mater. Res. Part B Appl. Biomater. 2016, 104, 761–770. [Google Scholar] [CrossRef]
- Park, S.B.; An, S.Y.; Han, W.J.; Park, J.T. Three-dimensional measurement of periodontal surface area for quantifying inflammatory burden. J. Periodontal Implant Sci. 2017, 47, 154–164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koch, F.; Meyer, N.; Valdec, S.; Jung, R.E.; Mathes, S.H. Development and application of a 3D periodontal in vitro model for the evaluation of fibrillar biomaterials. BMC Oral Health 2020, 20, 148. [Google Scholar] [CrossRef] [PubMed]
- Khadre, A.; Raif, E.L.; Junaid, S.; Goudouri, O.M.; Refaat, W.; Ramadan, A.; Boccaccini, A.R.; El-Gendy, R. A bilayered tissue engineered in vitro model simulating the tooth periodontium. Eur. Cells Mater. 2021, 41, 232–245. [Google Scholar] [CrossRef]
- El-Gendy, R.; Junaid, S.; Lam, S.K.L.; Elson, K.M.; Tipper, J.L.; Hall, R.M.; Ingham, E.; Kirkham, J. Developing a Tooth in situ Organ Culture Model for Dental and Periodontal Regeneration Research. Front. Bioeng. Biotechnol. 2021, 8, 581413. [Google Scholar] [CrossRef]
- Zhen, L.; Jiang, X.; Chen, Y.; Fan, D. MiR-31 is involved in the high glucose-suppressed osteogenic differentiation of human periodontal ligament stem cells by targeting Satb2. Am. J. Transl. Res. 2017, 9, 2384–2393. [Google Scholar]
- Liu, Z.; Chen, T.; Sun, W.; Yuan, Z.; Yu, M.; Chen, G.; Guo, W.; Xiao, J.; Tian, W. DNA Demethylation Rescues the Impaired Osteogenic Differentiation Ability of Human Periodontal Ligament Stem Cells in High Glucose. Sci. Rep. 2016, 6, 27447. [Google Scholar] [CrossRef]
- Zhan, D.; Guo, L.; Zheng, L. Inhibition of the receptor for advanced glycation promotes proliferation and repair of human periodontal ligament fibroblasts in response to high glucose via the NF-κB signaling pathway. Arch. Oral Biol. 2018, 87, 86–93. [Google Scholar] [CrossRef]
- Kim, S.Y.; Lee, J.Y.; Park, Y.D.; Kang, K.L.; Lee, J.C.; Heo, J.S. Hesperetin Alleviates the Inhibitory Effects of High Glucose on the Osteoblastic Differentiation of Periodontal Ligament Stem Cells. PLoS ONE 2013, 8, e67504. [Google Scholar] [CrossRef]
- Yan, Y.; Zhang, H.; Liu, L.; Chu, Z.; Ge, Y.; Wu, J.; Liu, Y.; Tang, C. Periostin reverses high glucose-inhibited osteogenesis of periodontal ligament stem cells via AKT pathway. Life Sci. 2020, 242, 117184. [Google Scholar] [CrossRef] [PubMed]
- Deng, C.; Sun, Y.; Liu, H.; Wang, W.; Wang, J.; Zhang, F. Selective adipogenic differentiation of human periodontal ligament stem cells stimulated with high doses of glucose. PLoS ONE 2018, 13, e0199603. [Google Scholar] [CrossRef]
- Bhattarai, G.; Min, C.K.; Jeon, Y.M.; Bashyal, R.; Poudel, S.B.; Kook, S.H.; Lee, J.C. Oral supplementation with p-coumaric acid protects mice against diabetes-associated spontaneous destruction of periodontal tissue. J. Periodontal Res. 2019, 54, 690–701. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Park, J.W.; Yeo, S.I.; Choi, B.J.; Suh, J.Y. Effects of high glucose on cellular activity of periodontal ligament cells in vitro. Diabetes Res. Clin. Pract. 2006, 74, 41–47. [Google Scholar] [CrossRef]
- Liu, J.; Jiang, Y.; Mao, J.; Gu, B.; Liu, H.; Fang, B. High levels of glucose induces a dose-dependent apoptosis in human periodontal ligament fibroblasts by activating caspase-3 signaling pathway. Appl. Biochem. Biotechnol. 2013, 170, 1458–1471. [Google Scholar] [CrossRef]
- Luo, H.; Zhu, W.; Mo, W.; Liang, M. High-glucose concentration aggravates TNF-alpha-induced cell viability reduction in human CD146-positive periodontal ligament cells via TNFR-1 gene demethylation. Cell Biol. Int. 2020, 44, 2383–2394. [Google Scholar] [CrossRef]
- Zhang, L.; Ding, Y.; Rao, G.Z.; Miao, D. Effects of IL-10 and glucose on expression of OPG and RANKL in human periodontal ligament fibroblasts. Brazilian J. Med. Biol. Res. 2016, 49, 1–6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seubbuk, S.; Sritanaudomchai, H.; Kasetsuwan, J.; Surarit, R. High glucose promotes the osteogenic differentiation capability of human periodontal ligament fbroblasts. Mol. Med. Rep. 2017, 15, 2788–2794. [Google Scholar] [CrossRef] [PubMed]
- Mei, Y.M.; Li, L.; Wang, X.Q.; Zhang, M.; Zhu, L.F.; Fu, Y.W.; Xu, Y. AGEs induces apoptosis and autophagy via reactive oxygen species in human periodontal ligament cells. J. Cell. Biochem. 2019, 121, 3764–3779. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Wang, X.; Zhang, L.; Wang, B.; Xu, B.; Zhang, J. GLP-1 inhibits PKCβ2 phosphorylation to improve the osteogenic differentiation potential of hPDLSCs in the AGE microenvironment. J. Diabetes Complicat. 2020, 34, 107495. [Google Scholar] [CrossRef]
- Fang, H.; Yang, K.; Tang, P.; Zhao, N.; Ma, R.; Luo, X.; Liu, Q. Glycosylation end products mediate damage and apoptosis of periodontal ligament stem cells induced by the JNK-mitochondrial pathway. Aging 2020, 12, 12850–12868. [Google Scholar] [CrossRef]
- Zhu, W.; Qiu, Q.; Luo, H.; Zhang, F.; Wu, J.; Zhu, X.; Liang, M. High Glucose Exacerbates TNF- α -Induced Proliferative Inhibition in Human Periodontal Ligament Stem Cells through Upregulation and Activation of TNF Receptor 1. Stem Cells Int. 2020, 2020, 4910767. [Google Scholar] [CrossRef] [Green Version]
- Mahmoud, M.; Abu-Shahba, N.; Azmy, O.; El-Badri, N. Impact of Diabetes Mellitus on Human Mesenchymal Stromal Cell Biology and Functionality: Implications for Autologous Transplantation. Stem Cell Rev. Rep. 2019, 15, 194–217. [Google Scholar] [CrossRef]
- Phadnis, S.M.; Ghaskadbi, S.M.; Hardikar, A.A.; Bhonde, R.R. Mesenchymal stem cells derived from bone marrow of diabetic patients portrait unique markers influenced by the diabetic microenvironment. Rev. Diabet. Stud. 2009, 6, 260–270. [Google Scholar] [CrossRef] [Green Version]
- Gabr, M.M.; Zakaria, M.M.; Refaie, A.F.; Ismail, A.M.; Abou-El-Mahasen, M.A.; Ashamallah, S.A.; Khater, S.M.; El-Halawani, S.M.; Ibrahim, R.Y.; Uin, G.S.; et al. Insulin-producing cells from adult human bone marrow mesenchymal stem cells control streptozotocin-induced diabetes in nude mice. Cell Transplant. 2013, 22, 133–145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brewster, L.; Robinson, S.; Wang, R.; Griffiths, S.; Li, H.; Peister, A.; Copland, I.; McDevitt, T. Expansion and angiogenic potential of mesenchymal stem cells from patients with critical limb ischemia. J. Vasc. Surg. 2017, 65, 826–838.e1. [Google Scholar] [CrossRef] [Green Version]
- Andrzejewska, A.; Catar, R.; Schoon, J.; Qazi, T.H.; Sass, F.A.; Jacobi, D.; Blankenstein, A.; Reinke, S.; Krüger, D.; Streitz, M.; et al. Multi-Parameter Analysis of Biobanked Human Bone Marrow Stromal Cells Shows Little Influence for Donor Age and Mild Comorbidities on Phenotypic and Functional Properties. Front. Immunol. 2019, 10, 2474. [Google Scholar] [CrossRef] [PubMed]
- Cassidy, F.C.; Shortiss, C.; Murphy, C.G.; Kearns, S.R.; Curtin, W.; De Buitl, C.; Brien, T.O.; Coleman, C.M. Impact of Type 2 Diabetes Mellitus on Human Bone Marrow Stromal Cell Number and Phenotypic Characteristics. Int. J. Mol. Sci. 2020, 21, 2476. [Google Scholar] [CrossRef] [Green Version]
- Flouzat-Lachaniette, C.H.; Heyberger, C.; Bouthors, C.; Roubineau, F.; Chevallier, N.; Rouard, H.; Hernigou, P. Osteogenic progenitors in bone marrow aspirates have clinical potential for tibial non-unions healing in diabetic patients. Int. Orthop. 2016, 40, 1375–1379. [Google Scholar] [CrossRef] [PubMed]
- Ying, X.; Chen, X.; Liu, H.; Nie, P.; Shui, X.; Shen, Y.; Yu, K.; Cheng, S. Silibinin alleviates high glucose-suppressed osteogenic differentiation of human bone marrow stromal cells via antioxidant effect and PI3K/Akt signaling. Eur. J. Pharmacol. 2015, 765, 394–401. [Google Scholar] [CrossRef] [PubMed]
- Chang, T.C.; Hsu, M.F.; Wu, K.K. High glucose induces bone marrow-derived mesenchymal stem cell senescence by upregulating autophagy. PLoS ONE 2015, 10, e0126537. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.M.; Schilling, T.; Benisch, P.; Zeck, S.; Meissner-Weigl, J.; Schneider, D.; Limbert, C.; Seufert, J.; Kassem, M.; Schütze, N.; et al. Effects of high glucose on mesenchymal stem cell proliferation and differentiation. Biochem. Biophys. Res. Commun. 2007, 363, 209–215. [Google Scholar] [CrossRef]
- Dhanasekaran, M.; Indumathi, S.; Rajkumar, J.S.; Sudarsanam, D. Effect of high glucose on extensive culturing of mesenchymal stem cells derived from subcutaneous fat, omentum fat and bone marrow. Cell Biochem. Funct. 2013, 31, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Shiomi, K.; Yamawaki, I.; Taguchi, Y.; Kimura, D.; Umeda, M. Osteogenic effects of glucose concentration for human bone marrow stromal cells after stimulation with Porphyromonas gingivalis lipopolysaccharide. J. Hard Tissue Biol. 2020, 29, 17–24. [Google Scholar] [CrossRef]
- Qu, B.; Gong, K.; Yang, H.S.; Li, Y.G.; Jiang, T.; Zeng, Z.M.; Cao, Z.R.; Pan, X.M. MiR-449 overexpression inhibits osteogenic differentiation of bone marrow mesenchymal stem cells via suppressing Sirt1/Fra-1 pathway in high glucose and free fatty acids microenvironment. Biochem. Biophys. Res. Commun. 2018, 496, 120–126. [Google Scholar] [CrossRef]
- Bian, Y.; Ma, X.; Wang, R.; Yuan, H.; Chen, N.; Du, Y. Human amnion-derived mesenchymal stem cells promote osteogenesis of human bone marrow mesenchymal stem cells against glucolipotoxicity. FEBS Open Bio. 2019, 9, 74–81. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, Y.Q.; Lu, Y.; Li, H.J.; Cheng, X.B. Effect of advanced glycosylation end products (AGEs) on proliferation of human bone marrow mesenchymal stem cells (MSCs) in vitro. Vitr. Cell. Dev. Biol. Anim. 2012, 48, 599–602. [Google Scholar] [CrossRef]
- Deng, X.; Xu, M.; Shen, M.; Cheng, J. Effects of Type 2 Diabetic Serum on Proliferation and Osteogenic Differentiation of Mesenchymal Stem Cells. J. Diabetes 2018, 2018, 5765478. [Google Scholar] [CrossRef]
- Rezabakhsh, A.; Cheraghi, O.; Nourazarian, A.; Hassanpour, M.; Kazemi, M.; Ghaderi, S.; Faraji, E.; Rahbarghazi, R.; Avci, Ç.B.; Bagca, B.G.; et al. Type 2 Diabetes Inhibited Human Mesenchymal Stem Cells Angiogenic Response by Over-Activity of the Autophagic Pathway. J. Cell. Biochem. 2017, 118, 1518–1530. [Google Scholar] [CrossRef]
- Raja, S.; Byakod, G.; Pudakalkatti, P. Growth factors in periodontal regeneration. Int. J. Dent. Hyg. 2009, 7, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.L.; Li, L.L.; Qiu, X.R.; An, Q.; Li, M.H. Effects of Combining Insulin-like Growth Factor 1 and Platelet-derived Growth Factor on Osteogenesis around Dental Implants. Chin. J. Dent. Res. 2018, 20, 105–109. [Google Scholar] [CrossRef]
- Ausenda, F.; Rasperini, G.; Acunzo, R.; Gorbunkova, A.; Pagni, G. New perspectives in the use of biomaterials for periodontal regeneration. Materials 2019, 12, 2197. [Google Scholar] [CrossRef] [Green Version]
- Deifenderfer, D.L.; Osyczka, A.M.; Reilly, G.C.; Leboy, P.S. BMP responsiveness in human mesenchymal stem cells. Connect. Tissue Res. 2003, 44, 305–311. [Google Scholar] [CrossRef]
- Hjortebjerg, R.; Laugesen, E.; Hoyem, P.; Oxvig, C.; Stausbøl-Gron, B.; Knudsen, S.T.; Kim, W.Y.; Poulsen, P.L.; Hansen, T.K.; Bjerre, M.; et al. The IGF system in patients with type 2 diabetes: Associations with markers of cardiovascular target organ damage. Eur. J. Endocrinol. 2017, 176, 521–531. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sesti, G.; Sciacqua, A.; Cardellini, M.; Marini, M.A.; Maio, R.; Vatrano, M.; Succurro, E.; Lauro, R.; Federici, M.; Perticone, F. Plasma concentration of IGF-I is independently associated with insulin sensitivity in subjects with different degrees of glucose tolerance. Diabetes Care 2005, 28, 120–125. [Google Scholar] [CrossRef] [Green Version]
- Clemmons, D.R.; Sleevi, M.; Allan, G.; Sommer, A. Effects of combined recombinant insulin-like growth factor (IGF)-I and IGF binding protein-3 in type 2 diabetic patients on glycemic control and distribution of IGF-I and IGF-II among serum binding protein complexes. J. Clin. Endocrinol. Metab. 2007, 92, 2652–2658. [Google Scholar] [CrossRef]
- Rajpathak, S.N.; Gunter, M.J.; Wylie-rosett, J.; Ho, G.Y.F.; Kaplan, R.C.; Rohan, T.E.; Strickler, H.D. The role of insulin-like growth factor-I and its binding proteins in glucose homeostasis and type 2 diabetes. Diabetes Metab. Res. Rev. 2009, 25, 3–12. [Google Scholar] [CrossRef] [Green Version]
- Haywood, N.J.; Cordell, P.A.; Tang, K.Y.; Makova, N.; Yuldasheva, N.Y.; Imrie, H.; Viswambharan, H.; Bruns, A.F.; Cubbon, R.M.; Kearney, M.T.; et al. Insulin-like growth factor binding protein 1 could improve glucose regulation and insulin sensitivity through its RGD domain. Diabetes 2017, 66, 287–299. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haywood, N.J.; Slater, T.A.; Matthews, C.J.; Wheatcroft, S.B. The insulin like growth factor and binding protein family: Novel therapeutic targets in obesity & diabetes. Mol. Metab. 2019, 19, 86–96. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, I.; Dörpholz, G.; Ott, C.E.; Kragesteen, B.; Schanze, N.; Lee, C.T.; Köhrle, J.; Mundlos, S.; Ruschke, K.; Knaus, P. BMPs as new insulin sensitizers: Enhanced glucose uptake in mature 3T3-L1 adipocytes via PPARγ and GLUT4 upregulation. Sci. Rep. 2017, 7, 17192. [Google Scholar] [CrossRef]
- Slaaby, R.; Schäffer, L.; Lautrup-Larsen, I.; Andersen, A.S.; Shaw, A.C.; Mathiasen, I.S.; Brandt, J. Hybrid receptors formed by insulin receptor (IR) and insulin-like growth factor i receptor (IGF-IR) have low insulin and high IGF-1 affinity irrespective of the IR splice variant. J. Biol. Chem. 2006, 281, 25869–25874. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lawrence, M.C.; McKern, N.M.; Ward, C.W. Insulin receptor structure and its implications for the IGF-1 receptor. Curr. Opin. Struct. Biol. 2007, 17, 699–705. [Google Scholar] [CrossRef] [PubMed]
- Ranke, M.B.; Edin, F.; Emeritus, P. Insulin-like growth factor binding-protein-3 (IGFBP-3). Best Pract. Res. Clin. Endocrinol. Metab. 2015, 29, 701–711. [Google Scholar] [CrossRef] [PubMed]
- Bach, L. 40 YEARS OF IGF1: IGF-binding proteins. J. Mol. Endocrinol. 2018, 61, T11–T28. [Google Scholar] [CrossRef] [Green Version]
- Al-Kharobi, H.; El-gendy, R.O.; Devine, D.A.D.A.; Beattie, J. The role of the insulin-like growth factor (IGF) axis in osteogenic and odontogenic differentiation. Cell. Mol. Life Sci. 2014, 71, 1469–1476. [Google Scholar] [CrossRef] [PubMed]
- Lerner, U.H.; Kindstedt, E.; Lundberg, P. The critical interplay between bone resorbing and bone forming cells. J. Clin. Periodontol. 2019, 46, 33–51. [Google Scholar] [CrossRef] [PubMed]
- Saeed, H.; Qiu, W.; Li, C.; Flyvbjerg, A.; Abdallah, B.M.; Kassem, M. Telomerase activity promotes osteoblast differentiation by modulating IGF-signaling pathway. Biogerontology 2015, 16, 733–745. [Google Scholar] [CrossRef] [Green Version]
- Bahamonde, M.; Misra, M. Potential applications for rhIGF-I: Bone disease and IGF-I. Growth Horm. IGF Res. 2020, 52, 101317. [Google Scholar] [CrossRef]
- Xu, Y.; Kong, G.K.W.; Menting, J.G.; Margetts, M.B.; Delaine, C.A.; Jenkin, L.M.; Kiselyov, V.V.; De Meyts, P.; Forbes, B.E.; Lawrence, M.C. How ligand binds to the type 1 insulin-like growth factor receptor. Nat. Commun. 2018, 9, 821. [Google Scholar] [CrossRef] [Green Version]
- Torrente, Y.; Bella, P.; Tripodi, L.; Villa, C.; Farini, A. Role of Insulin-Like Growth Factor Receptor 2 across Muscle Homeostasis: Implications for Treating Muscular Dystrophy. Cells 2020, 9, 441. [Google Scholar] [CrossRef] [Green Version]
- Heald, A.H.; Cruickshank, J.K.; Riste, L.K.; Cade, J.E.; Anderson, S.; Greenhalgh, A.; Sampayo, J.; Taylor, W. Close relation of fasting insulin-like growth factor binding protein-1 (IGFBP-1) with glucose tolerance and cardiovascular risk in two populations. Diabetologia 2001, 44, 333–339. [Google Scholar] [CrossRef] [Green Version]
- Zheng, W.; Lai, Y.; Jin, P.; Gu, W.; Zhou, Q.; Wu, X. Association of circulating IGFBP1 level with the severity of coronary artery lesions in patients with unstable angina. Dis. Markers 2017, 2017, 1917291. [Google Scholar] [CrossRef] [Green Version]
- Hou, L.; He, Q. Inhibition of IGFBP1 induction enhances the radiosensitivity of colon cancer cells in vitro. Int. J. Clin. Exp. Pathol. 2016, 9, 8759–8766. [Google Scholar]
- Amin, S.; Riggs, B.L.; Atkinson, E.J.; Oberg, A.L.; Melton, L.J.; Khosla, S. A potentially deleterious role of IGFBP-2 on bone density in aging men and women. J. Bone Miner. Res. 2004, 19, 1075–1083. [Google Scholar] [CrossRef] [PubMed]
- Amin, S.; Riggs, B.L.; Melton, L.J.; Achenbach, S.J.; Atkinson, E.J.; Khosla, S. High serum IGFBP-2 is predictive of increased bone turnover in aging men and women. J. Bone Miner. Res. 2007, 22, 799–807. [Google Scholar] [CrossRef] [PubMed]
- Takenouchi, Y.; Ohshima, M.; Yamaguchi, Y.; Nishida, T.; Senda, N.; Idesawa, M.; Otsuka, K.; Ito, K. Insulin-like growth factor-binding protein-2 and -3 in gingival crevicular fluid. J. Periodontal Res. 2010, 45, 803–808. [Google Scholar] [CrossRef] [PubMed]
- Xi, G.; Shen, X.; Rosen, C.J.; Clemmons, D.R. IRS-1 Functions as a Molecular Scaffold to Coordinate IGF-I/ IGFBP-2 Signaling During Osteoblast Differentiation. J Bone Min. Res 2016, 31, 1300–1314. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Allard, J.B.; Duan, C. IGF-Binding Proteins: Why Do They Exist and Why Are There So Many? Front. Endocrinol. 2018, 9, 117. [Google Scholar] [CrossRef] [Green Version]
- Gillberg, P.; Olofsson, H.; Mallmin, H.; Blum, W.F.; Ljunghall, S.; Nilsson, A.G. Bone mineral density in femoral neck is positively correlated to circulating insulin-like growth factor (IGF)-I and IGF-binding protein (IGFBP)-3 in Swedish men. Calcif. Tissue Int. 2002, 70, 22–29. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Kanatani, M.; Yamauchi, M.; Kaji, H.; Sugishita, T.; Baylink, D.J.; Mohan, S.; Chihara, K.; Sugimoto, T. Serum levels of insulin-like growth factor (IGF); IGF-binding proteins-3, -4, and -5; and their relationships to bone mineral density and the risk of vertebral fractures in postmenopausal women. Calcif. Tissue Int. 2006, 78, 18–24. [Google Scholar] [CrossRef] [Green Version]
- Harb, A.N.; Holtfreter, B.; Friedrich, N.; Wallaschofski, H.; Nauck, M.; Albers, M.; Meisel, P.; Biffar, R.; Kocher, T. Association between the insulin-like growth factor axis in serum and periodontitis in the Study of Health in Pomerania: An exploratory study. J. Clin. Periodontol. 2012, 39, 931–939. [Google Scholar] [CrossRef]
- Beattie, J.; Al-Khafaji, H.; Noer, P.R.; Alkharobi, H.E.; Alhodhodi, A.; Meade, J.; El-Gendy, R.; Oxvig, C. Insulin-like growth factor-binding protein action in bone tissue: A key role for pregnancy- associated plasma protein-A. Front. Endocrinol. 2018, 9, 31. [Google Scholar] [CrossRef] [Green Version]
- Duan, C.; Allard, J.B. Insulin-Like Growth Factor Binding Protein-5 in Physiology and Disease. Front. Endocrinol. 2020, 11, 100. [Google Scholar] [CrossRef] [Green Version]
- Bach, L.A. Current ideas on the biology of IGFBP-6: More than an IGF-II inhibitor? Growth Horm. IGF Res. 2016, 30–31, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Bach, L.A. Recent insights into the actions of IGFBP-6. J. Cell Commun. Signal. 2015, 9, 189–200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liso, A.; Capitanio, N.; Gerli, R.; Conese, M. From fever to immunity: A new role for IGFBP-6? J. Cell. Mol. Med. 2018, 22, 4588–4596. [Google Scholar] [CrossRef] [Green Version]
- Götz, W.; Heinen, M.; Lossdörfer, S.; Jäger, A. Immunohistochemical localization of components of the insulin-like growth factor system in human permanent teeth. Arch. Oral Biol. 2006, 51, 387–395. [Google Scholar] [CrossRef] [PubMed]
- Reckenbeil, J.; Kraus, D.; Stark, H.; Rath-Deschner, B.; Jäger, A.; Wenghoefer, M.; Winter, J.; Götz, W. Insulin-like growth factor 1 (IGF1) affects proliferation and differentiation and wound healing processes in an inflammatory environment with p38 controlling early osteoblast differentiation in periodontal ligament cells. Arch. Oral Biol. 2017, 73, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Amar, S. Role of Insulin-Like Growth Factor-1 Signaling in Dental Fibroblast Apoptosis. J. Periodontol. 2003, 74, 1176–1182. [Google Scholar] [CrossRef] [PubMed]
- Konermann, A.; Lossdörfer, S.; Jäger, A.; Chen, Y.; Götz, W. Autoregulation of insulin-like growth factor 2 and insulin-like growth factor-binding protein 6 in periodontal ligament cells in vitro. Ann. Anat. 2013, 195, 527–532. [Google Scholar] [CrossRef]
- Lynch, S.E.; Williams, R.C.; Poison, A.M.; Howell, T.H.; Reddy, M.S.; Zappa, U.E.; Antoniades, H.N. A combination of platelet-derived and insulin-like growth factors enhances periodontal regeneration. J. Clin. Periodontol. 1989, 16, 545–548. [Google Scholar] [CrossRef]
- Rutherford, R.B.; Niekrash, C.E.; Kennedy, J.E.; Charette, M.F. Platelet-derived and insulin-like growth factors stimulate regeneration of periodontal attachment in monkeys. J. Periodontal Res. 1992, 27, 285–290. [Google Scholar] [CrossRef]
- Giannobile, W.V.; Hernandez, R.A.; Finkelman, R.D.; Ryan, S.; Kiritsy, C.P.; D’Andrea, M.; Lynch, S.E. Comparative effects of platelet-derived growth factor-BB and insulin-like growth factor-I, individually and in combination, on periodontal regeneration in Macaca fascicularis. J. Periodontal Res. 1996, 31, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Gamal, A.Y.; Mailhot, J.M.; Garnick, J.J.; Newhouse, R.; Sharawy, M.M. Human periodontal ligament fibroblast response to PDGF-BB and IGF-1 application on tetracycline HCl conditioned root surfaces. J. Clin. Periodontol. 1998, 25, 404–412. [Google Scholar] [CrossRef] [PubMed]
- Selvig, K.A.; Wikesjö, U.M.E.; Bogle, G.C.; Finkelman, R.D. Impaired early bone formation in periodontal fenestration defects in dogs following application of insulin-like growth factor (II). Basic fibroblast growth factor and transforming growth factor beta 1. J. Clin. Periodontol. 1994, 21, 380–385. [Google Scholar] [CrossRef]
- Fang, Y.; Wang, L.P.; Du, F.L.; Liu, W.J.; Ren, G.L. Effects of insulin-like growth factor I on alveolar bone remodeling in diabetic rats. J. Periodontal Res. 2013, 48, 144–150. [Google Scholar] [CrossRef]
- Han, N.; Zhang, F.; Li, G.; Zhang, X.; Lin, X.; Yang, H.; Wang, L.; Cao, Y.; Du, J.; Fan, Z. Local application of IGFBP5 protein enhanced periodontal tissue regeneration via increasing the migration, cell proliferation and osteo/dentinogenic differentiation of mesenchymal stem cells in an inflammatory niche. Stem Cell Res. Ther. 2017, 8, 210. [Google Scholar] [CrossRef] [PubMed]
- Howell, T.H.; Fiorellini, J.P.; Paquette, D.W.; Offenbacher, S.; Giannobile, W.V.; Lynch, S.E. A Phase I/II Clinical Trial to Evaluate a Combination of Recombinant Human Platelet-Derived Growth Factor-BB and Recombinant Human Insulin-Like Growth Factor-I in Patients with Periodontal Disease. J. Periodontol. 1997, 68, 1186–1193. [Google Scholar] [CrossRef]
- Haase, H.R.; Clarkson, R.W.; Waters, M.J.; Bartold, P.M. Growth factor modulation of mitogenic responses and proteoglycan synthesis by human periodontal fibroblasts. J. Cell. Physiol. 1998, 174, 353–361. [Google Scholar] [CrossRef]
- Palioto, D.B.; Coletta, R.D.; Graner, E.; Joly, J.C.; de Lima, A.F.M. The Influence of Enamel Matrix Derivative Associated With Insulin-Like Growth Factor-I on Periodontal Ligament Fibroblasts. J. Periodontol. 2004, 75, 498–504. [Google Scholar] [CrossRef] [PubMed]
- Okubo, K.; Kobayashi, M.; Takiguchi, T.; Takada, T.; Ohazama, A.; Okamatsu, Y.; Hasegawa, K. Participation of endogenous IGF-I and TGF-β1 with enamel matrix derivative-stimulated cell growth in human periodontal ligament cells. J. Periodontal Res. 2003, 38, 1–9. [Google Scholar] [CrossRef]
- Parkar, M.H.; Tonetti, M. Gene Expression Profiles of Periodontal Ligament Cells Treated With Enamel Matrix Proteins In Vitro: Analysis Using cDNA Arrays. J. Periodontol. 2004, 75, 1539–1546. [Google Scholar] [CrossRef]
- Lossdörfer, S.; Abuduwali, N.; Jäger, A. Bone Morphogenetic Protein-7 Modifies the Effects of Insulin-Like Growth Factors and Intermittent Parathyroid Hormone (1-34) on Human Periodontal Ligament Cell Physiology In Vitro. J. Periodontol. 2011, 82, 900–908. [Google Scholar] [CrossRef]
- Nemoto, E.; Shimonishi, M.; Nitta, Y.; Shimauchi, H. The involvement of platelet-derived growth factor receptors and insulin-like growth factor-1 receptors signaling during mineralized nodule formation by human periodontal ligament cells. J. Periodontal Res. 2004, 39, 388–397. [Google Scholar] [CrossRef]
- Yu, Y.; Mu, J.; Fan, Z.; Lei, G. Insulin-like growth factor 1 enhances the proliferation and osteogenic differentiation of human periodontal ligament stem cells via ERK and JNK MAPK pathways. Histochem. Cell Biol. 2012, 137, 513–525. [Google Scholar] [CrossRef]
- Li, X.; Yao, J.; Wu, J.; Du, X.; Jing, W.; Liu, L. Roles of PRF and IGF-1 in promoting alveolar osteoblast growth and proliferation and molecular mechanism. Int. J. Clin. Exp. Pathol. 2018, 11, 3294–3301. [Google Scholar]
- Han, X.; Amar, S. IGF-1 Signaling Enhances Cell Survival in Periodontal Ligament. J. Dent. Res. 2003, 82, 454–459. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zhou, J.; Cui, B.; Yu, T. Evaluation of hypoxia on the expression of miR-646/IGF-1 signaling in human periodontal ligament cells (hPDLCs). Med. Sci. Monit. 2018, 24, 5282–5291. [Google Scholar] [CrossRef]
- Pumklin, J.; Manokawinchoke, J.; Bhalang, K.; Pavasant, P. Intermittent Compressive Stress Enhanced Insulin-Like Growth Factor-1 Expression in Human Periodontal Ligament Cells. Int. J. Cell Biol. 2015, 2015, 369874. [Google Scholar] [CrossRef] [PubMed]
- Ochiai, H.; Okada, S.; Saito, A.; Hoshi, K.; Yamashita, H.; Takato, T.; Azuma, T. Inhibition of insulin-like growth factor-1 (IGF-1) expression by prolonged transforming growth factor-β1 (TGF-β1) administration suppresses osteoblast differentiation. J. Biol. Chem. 2012, 287, 22654–22661. [Google Scholar] [CrossRef] [Green Version]
- Liu, L.; Ling, J.; Wei, X.; Wu, L.; Xiao, Y. Stem Cell Regulatory Gene Expression in Human Adult Dental Pulp and Periodontal Ligament Cells Undergoing Odontogenic/Osteogenic Differentiation. J. Endod. 2009, 35, 1368–1376. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Wang, Y.; Jia, Z.; Wang, L.; Wang, J.; Yang, D.; Song, J.; Wang, S.; Fan, Z. Demethylation of IGFBP5 by Histone Demethylase KDM6B Promotes Mesenchymal Stem Cell-Mediated Periodontal Tissue Regeneration by Enhancing Osteogenic Differentiation and Anti-Inflammation Potentials. Stem Cells 2015, 33, 2523–2536. [Google Scholar] [CrossRef]
- Wu, L.; Zhang, G.; Guo, C.; Pan, Y. Intracellular Ca2+ signaling mediates IGF-1-induced osteogenic differentiation in bone marrow mesenchymal stem cells. Biochem. Biophys. Res. Commun. 2020, 527, 200–206. [Google Scholar] [CrossRef]
- Reible, B.; Schmidmaier, G.; Moghaddam, A.; Westhauser, F.; Induce, P.; Differentiation, O.; Stem, M.; Vitro, C.; Reible, B.; Schmidmaier, G.; et al. Insulin-Like Growth Factor-1 as a Possible Alternative to Bone Morphogenetic Protein-7 to Induce Osteogenic Differentiation of Human Mesenchymal Stem Cells in Vitro. Int. J. Mol. Sci. 2018, 19, 1674. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reible, B.; Schmidmaier, G.; Prokscha, M.; Moghaddam, A.; Westhauser, F. Continuous stimulation with differentiation factors is necessary to enhance osteogenic differentiation of human mesenchymal stem cells in-vitro. Growth Factors 2017, 35, 179–188. [Google Scholar] [CrossRef]
- Celil, A.B.; Campbell, P.G. BMP-2 and insulin-like growth factor-I mediate osterix (Osx) expression in human mesenchymal stem cells via the MAPK and protein kinase D signaling pathways. J. Biol. Chem. 2005, 280, 31353–31359. [Google Scholar] [CrossRef] [Green Version]
- Chen, C.Y.; Tseng, K.Y.; Lai, Y.L.; Chen, Y.S.; Lin, F.H.; Lin, S. Overexpression of insulin-like growth factor 1 enhanced the osteogenic capability of aging bone marrow mesenchymal stem cells. Theranostics 2017, 7, 1598–1611. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, C.O.; Chen, B.K.; Bale, L.K.; Overgaard, M.T.; Oxvig, C.; Conover, C.A. Transforming Growth Factor-β Regulation of the Insulin-Like Growth Factor Binding Protein-4 Protease System in Cultured Human Osteoblasts. J. Bone Miner. Res. 2003, 18, 1066–1072. [Google Scholar] [CrossRef] [PubMed]
- Severino, V.; Alessio, N.; Farina, A.; Sandomenico, A.; Cipollaro, M.; Peluso, G.; Galderisi, U.; Chambery, A. Insulin-like growth factor binding proteins 4 and 7 released by senescent cells promote premature senescence in mesenchymal stem cells. Cell Death Dis. 2013, 4, e911. [Google Scholar] [CrossRef] [PubMed]
- Youssef, A.; Aboalola, D.; Han, V.K.M. The Roles of Insulin-Like Growth Factors in Mesenchymal Stem Cell Niche. Stem Cells Int. 2017, 2017, 9453108. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.; Akhtar, S.; Mohsin, S.; Khan, S.N.; Riazuddin, S. Growth factor preconditioning increases the function of diabetes-impaired mesenchymal stem cells. Stem Cells Dev. 2011, 20, 67–75. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| # | Study | Diabetic Cond | Ca Nodules (AR Staining) | ALP Expression | ALP Activity | Osteogenic Transcription Factors | Osteogenic Markers | NF-ĸβ | Others | ||
|---|---|---|---|---|---|---|---|---|---|---|---|
| RUNX2 | OSX | OCN | OPN | Expression | |||||||
| 1 | Zhen et al. [160] | HG | ↓ | NR | NR | ↓ | ↓ | ↓ | NR | NR | ↑miR-31 |
| 2 | Liu et al. [161] | HG | NR | NR | ↓ | NR | ↓ | ↓ | ↓ | NR | ↑DNA methylation |
| 3 | Kato et al. [149] | HG | ↓ | NR | ↓ | ↑ | NR | ↓ | NR | ↑ | ↓ Proliferation |
| ↓ Viability | |||||||||||
| ↑ IL-6 and IL-8 expression | |||||||||||
| Morphology NC | |||||||||||
| 4 | Guo et al. [150] | HG | ↓ | ↓ | NR | ↓ | ↓ | NR | NR | NR | ↓ Proliferation |
| ↓ Cells in S and G2/M phases | |||||||||||
| 5 | Zheng et al. [154] | HG | NR | NR | ↓ | ↓ | ↓ | NR | NR | NR | ↓ Proliferation |
| 6 | Zhan et al. [162] * | HG | NR | NR | NR | NR | NR | NR | NR | NR | ↑ RAGEs expression |
| ↓Proliferation | |||||||||||
| 7 | Kim et al. [163] | HG | NR | ↓ | ↓ | ↓ | ↓ | NR | ↓ ** | NR | ↓COL1A1 ** |
| 8 | Yan et al. [164] | HG | ↓ | NR | ↓ | NR | NR | NR | NR | NR | ↓ Migration |
| ↑ ROS | |||||||||||
| 9 | Deng et al. [165] | HG | ↓ | ↓ | NR | ↓ | NR | NR | ↓ | NR | ↑ Adipogenic differentiation |
| 10 | Bhattarai et al. [166] * | HG | ↓ | NR | NR | NR | NR | NR | NR | NR | ↓ Proliferation |
| ↑ ROS | |||||||||||
| 11 | Kim et al. [167] * | HG | ↓ | NR | NR | NR | NR | NR | NR | NR | ↓ Proliferation |
| ↓ Viability | |||||||||||
| 12 | Liu et al. [168] * | HG | NR | NR | NR | NR | NR | NR | NR | NR | ↑ Apoptotic cells |
| ↑ Caspase 3 activity | |||||||||||
| 13 | Luo et al. [169] | HG | NR | NR | NR | NR | NR | NR | NR | NR | ↓ Proliferation |
| ↓ DNMT activity | |||||||||||
| ↑ TNFR-1 expression | |||||||||||
| 14 | Zhang et al. [170] | HG | NR | NR | NR | NR | NR | NR | NR | NR | ↑ RANKL |
| ↓ OPG | |||||||||||
| 15 | Seubbuck et al. [171] * | HG | ↑ | NR | ↑ | NR | NR | NR | NR | NR | ↑ Proliferation |
| ↑ Expression of Nanog, Oct4, Sox2, CD166, Periostin and β-Catenin | |||||||||||
| 16 | Xu et al. [29] * | AGEs | NR | NR | NR | NR | NR | NR | NR | ↑ | ↓ Viability |
| ↑ IL-6 and IL-8 expression | |||||||||||
| ↑ ERS | |||||||||||
| 17 | Guo et al. [151] | AGEs | ↓ | NR | NR | NR | NR | NR | NR | NR | ↑ RAGEs |
| ↑ ROS | |||||||||||
| 18 | Mei et al. [172] * | AGEs | NR | NR | NR | NR | NR | NR | NR | NR | ↓ Viability |
| ↑ Apoptosis | |||||||||||
| ↑ Autophagy | |||||||||||
| ↑ ROS | |||||||||||
| 19 | Wang et al. [173] | AGEs | ↓ | NR | ↓ | ↓ | NR | NR | ↓ | NR | ↓ Proliferation |
| 20 | Zhang et al. [152] | AGEs | ↓ | ↓ | ↓ | ↓ | ↓ | ↓ | ↓ | NR | ↓ COL1 expression |
| ↓ BSP expression | |||||||||||
| 21 | Fang et al. [174] | AGEs | ↓ | ↓ | ↓ | ↓ | NR | ↓ | NR | NR | ↓ Proliferation |
| ↑ ROS | |||||||||||
| ↑ Apoptosis | |||||||||||
| ↑ Mitochondrial damage | |||||||||||
| 22 | Zheng et al. [10] * | HG + TNF-α | NR | NR | NR | NR | NR | NR | NR | ↑ | ↑ RANKL expression |
| 23 | Zhu et al. [175] | HG + TNF-α | NR | NR | NR | NR | NR | NR | NR | NR | ↑ TNFR-1 expression |
| ↓ Cell viability | |||||||||||
| # | Study | Diabetic Cond | Ca Nodules (AR Staining) | ALP Expression | ALP Activity | Osteogenic Transcription Factors | Osteogenic Markers | Others | ||
|---|---|---|---|---|---|---|---|---|---|---|
| RUNX2 | OSX | OCN | OPN | |||||||
| 1 | Ying et al. [183] | HG | ↓ | ↓ | ↓ | ↓ | ↓ | ↓ | NR | ↓COL1 and BMP2 expression |
| ↓PI3k and Akt expression | ||||||||||
| ↑ ROS | ||||||||||
| 2 | Chang et al. [184] | HG | NR | NR | NR | NR | NR | NR | NR | ↓ PD time |
| ↑ Senescence | ||||||||||
| ↑ Autophagy | ||||||||||
| 3 | Li et al. [185] | HG | ↑ | NR | NR | NR | NR | NR | NR | ↓Proliferation (25 mM, long term) |
| ↓ Apoptosis (40 mM, short term) | ||||||||||
| 4 | Dhanasekaran et al. [186] | HG | NC | NR | NR | NR | NR | NR | NR | ↓ Proliferation |
| (late vs early P) | Unchanged morphology, karyotyping and surface markers | |||||||||
| 5 | Shiomi et al. [187] | HG + LPS | ↓ (24 mM, 3 wks.) | NR | ↓ (12 and 24 mM, 2 wks.) | ↑ (8 and 12 mM, 3 wks) | NR | ↓ (12 and 24 mM, 3 wks.) | NR | ↑ Proliferation |
| (purchased BM-MSCs) | ↓ (24 mM, 3 wks.) | ↓ IL-1β, IL-6, IL-8 | ||||||||
| (8 and 12 mM, 1 and 2 wks.) | ||||||||||
| ↑ IL-1β, IL-6, IL-8 | ||||||||||
| (24 mM, 1 and 2 wks.) | ||||||||||
| 6 | Qu et al. [188] | HG + free fatty acid | ↓ | NR | ↓ | NR | NR | NR | NR | ↑miR-449 |
| 7 | Bian et al. [189] | HG + palmitic acid | ↓ | NR | ↓ | ↓ | NR | ↓ | NR | ↓ Proliferation |
| (BM-MSCs cell line) | ↓p38 expression | |||||||||
| ↑ ROS | ||||||||||
| 8 | Miranda et al. [30] (primary osteoblast like cells from T2DM patients) | HG + AGEs | NR | NR | NR | ↓ | ↓ | NR | NR | ↑ OPG and RANKL expression |
| ↓ OPG/RANKL ratio | ||||||||||
| ↑ RAGEs expression | ||||||||||
| 9 | Lu et al. [190] | AGEs | NR | NR | NR | NR | NR | NR | NR | ↓ Proliferation |
| 10 | Deng et al. [191] | T2DM serum | NR | NR | NR | ↓ | NR | ↓ | ↓ | ↑ Proliferation |
| 11 | Rezabakhsh et al. [192] | T2DM serum | NR | NR | NR | NR | NR | NR | NR | ↑ Apoptosis |
| ↑ Autophagy | ||||||||||
| ↓ Chemotaxis | ||||||||||
| ↓ Angiogenesis | ||||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hussein, N.M.S.; Meade, J.L.; Pandit, H.; Jones, E.; El-Gendy, R. The Effect of Diabetes Mellitus on IGF Axis and Stem Cell Mediated Regeneration of the Periodontium. Bioengineering 2021, 8, 202. https://doi.org/10.3390/bioengineering8120202
Hussein NMS, Meade JL, Pandit H, Jones E, El-Gendy R. The Effect of Diabetes Mellitus on IGF Axis and Stem Cell Mediated Regeneration of the Periodontium. Bioengineering. 2021; 8(12):202. https://doi.org/10.3390/bioengineering8120202
Chicago/Turabian StyleHussein, Nancy M. S., Josie L. Meade, Hemant Pandit, Elena Jones, and Reem El-Gendy. 2021. "The Effect of Diabetes Mellitus on IGF Axis and Stem Cell Mediated Regeneration of the Periodontium" Bioengineering 8, no. 12: 202. https://doi.org/10.3390/bioengineering8120202
APA StyleHussein, N. M. S., Meade, J. L., Pandit, H., Jones, E., & El-Gendy, R. (2021). The Effect of Diabetes Mellitus on IGF Axis and Stem Cell Mediated Regeneration of the Periodontium. Bioengineering, 8(12), 202. https://doi.org/10.3390/bioengineering8120202







