Anti-RSV Peptide-Loaded Liposomes for the Inhibition of Respiratory Syncytial Virus †
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Small Unilamellar Liposomes
2.3. Loading of Peptide
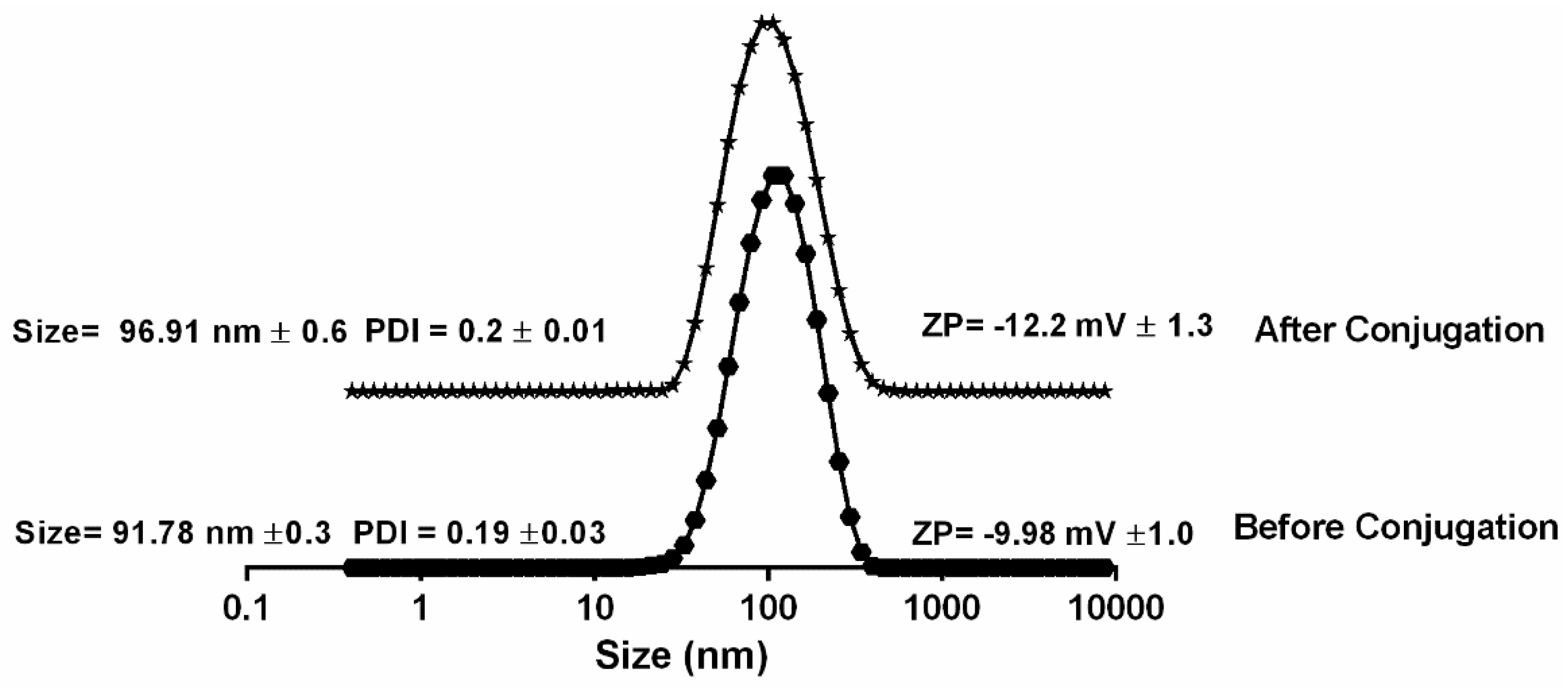
2.4. Particle Characteristics
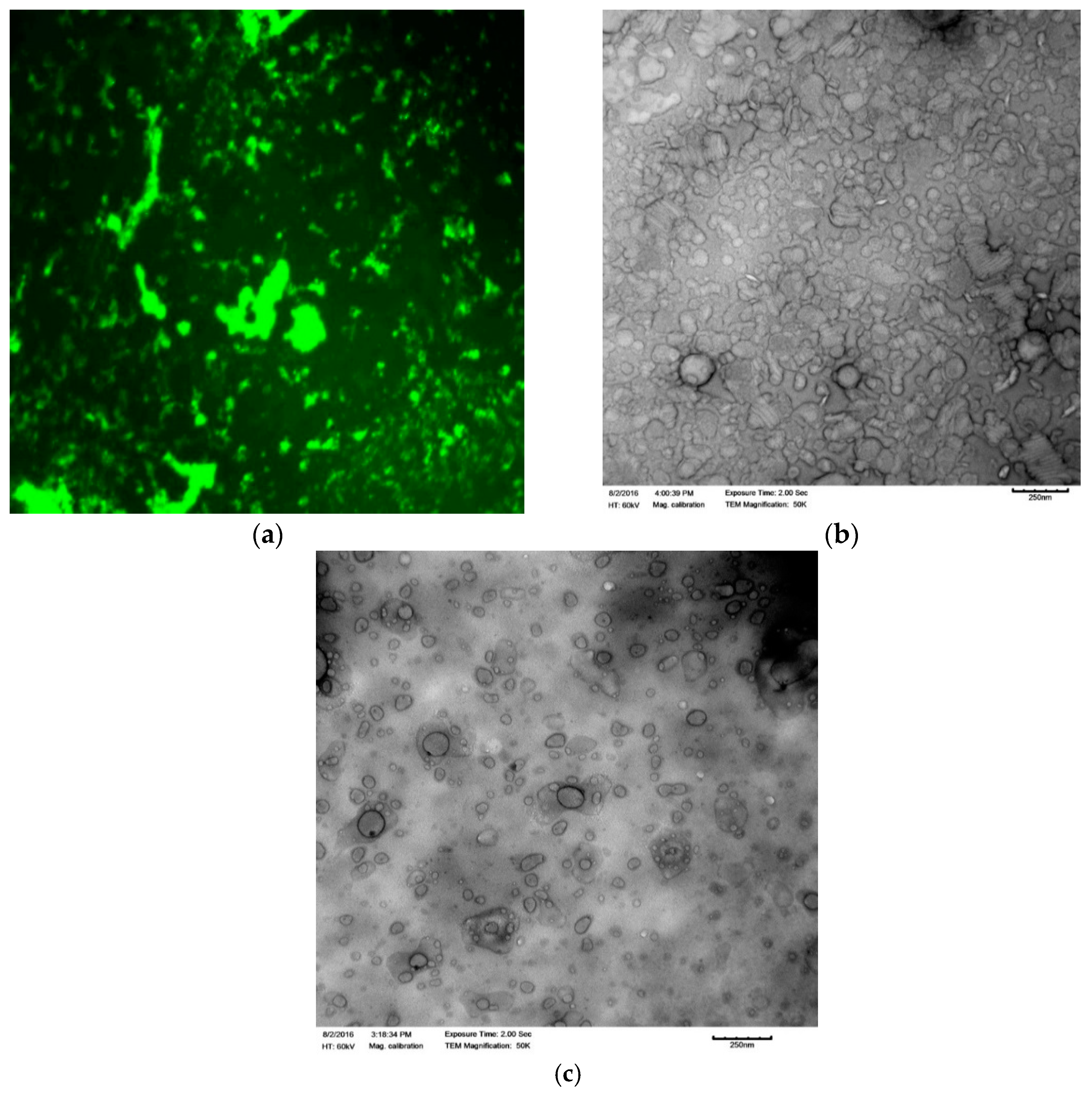
2.5. Transmission Electron Microscopy (TEM)
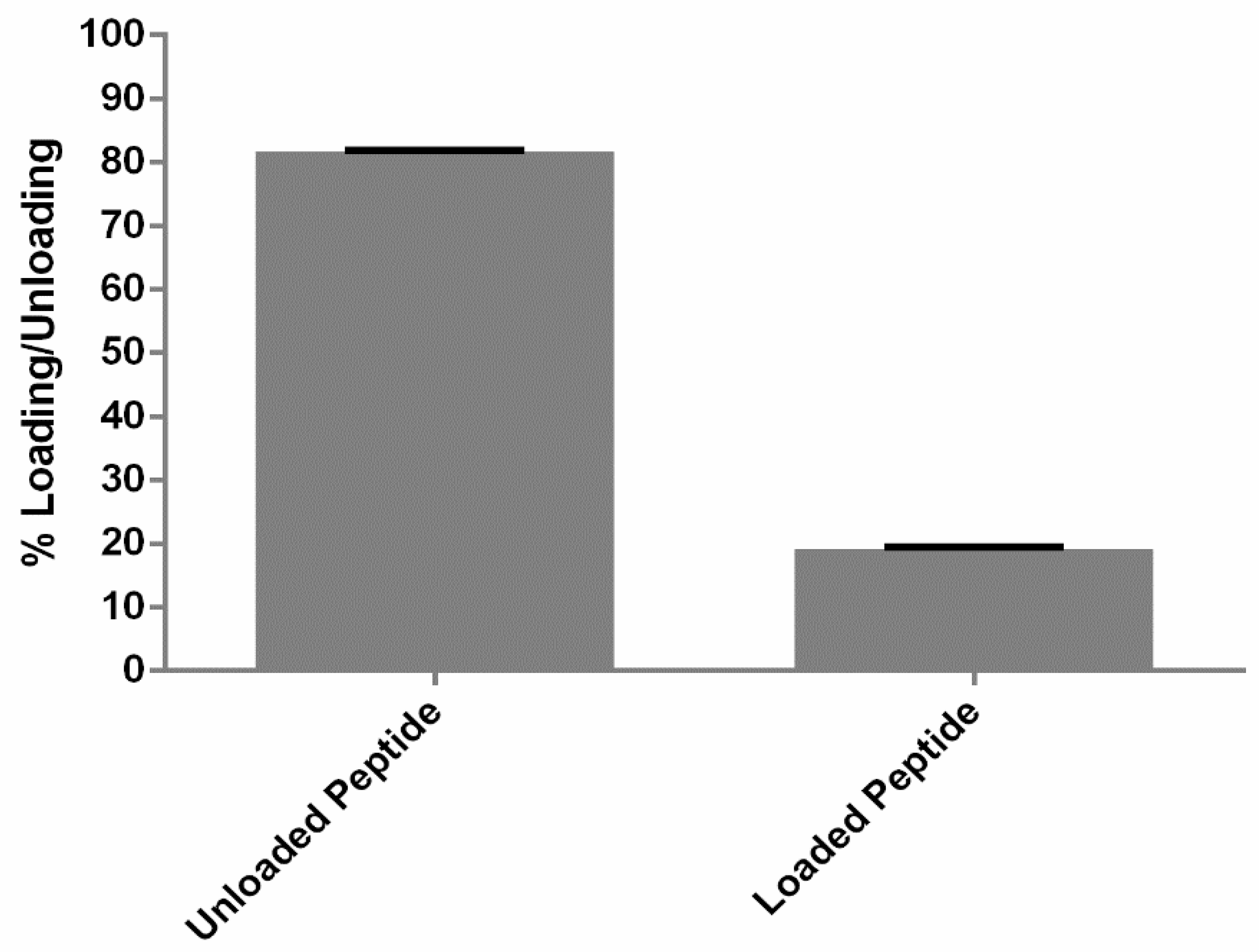
2.6. Quantification of Peptide Association
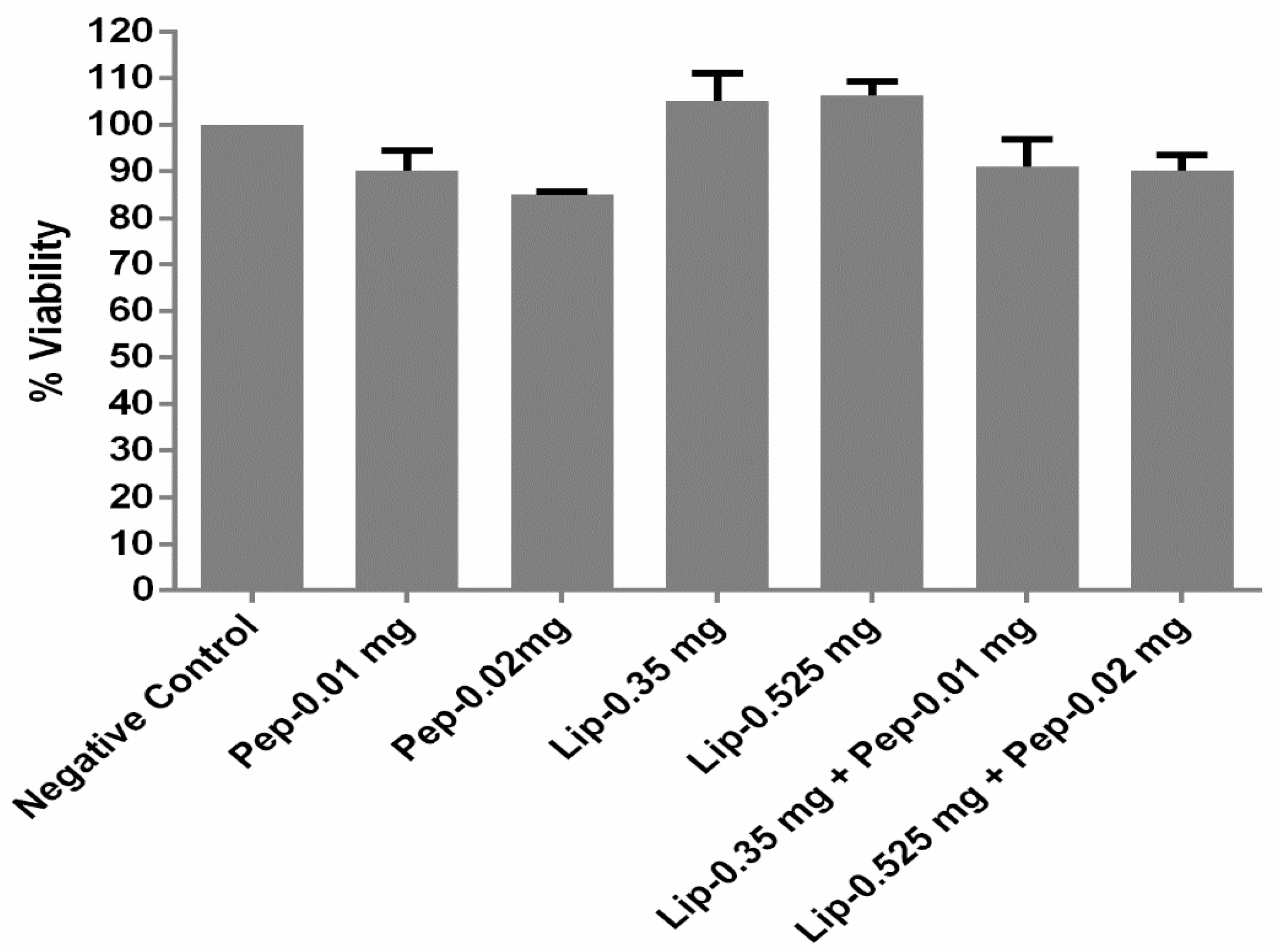
2.7. Cell Viability Assay
2.8. Fluorescence Microscopy
2.9. Plaque Assay
2.10. TaqMan qPCR Analysis
2.11. Statistical Analysis
3. Results
3.1. Confirmation of Peptide Loading
3.2. Quantification of Loading
3.3. Cell Toxicity Analysis
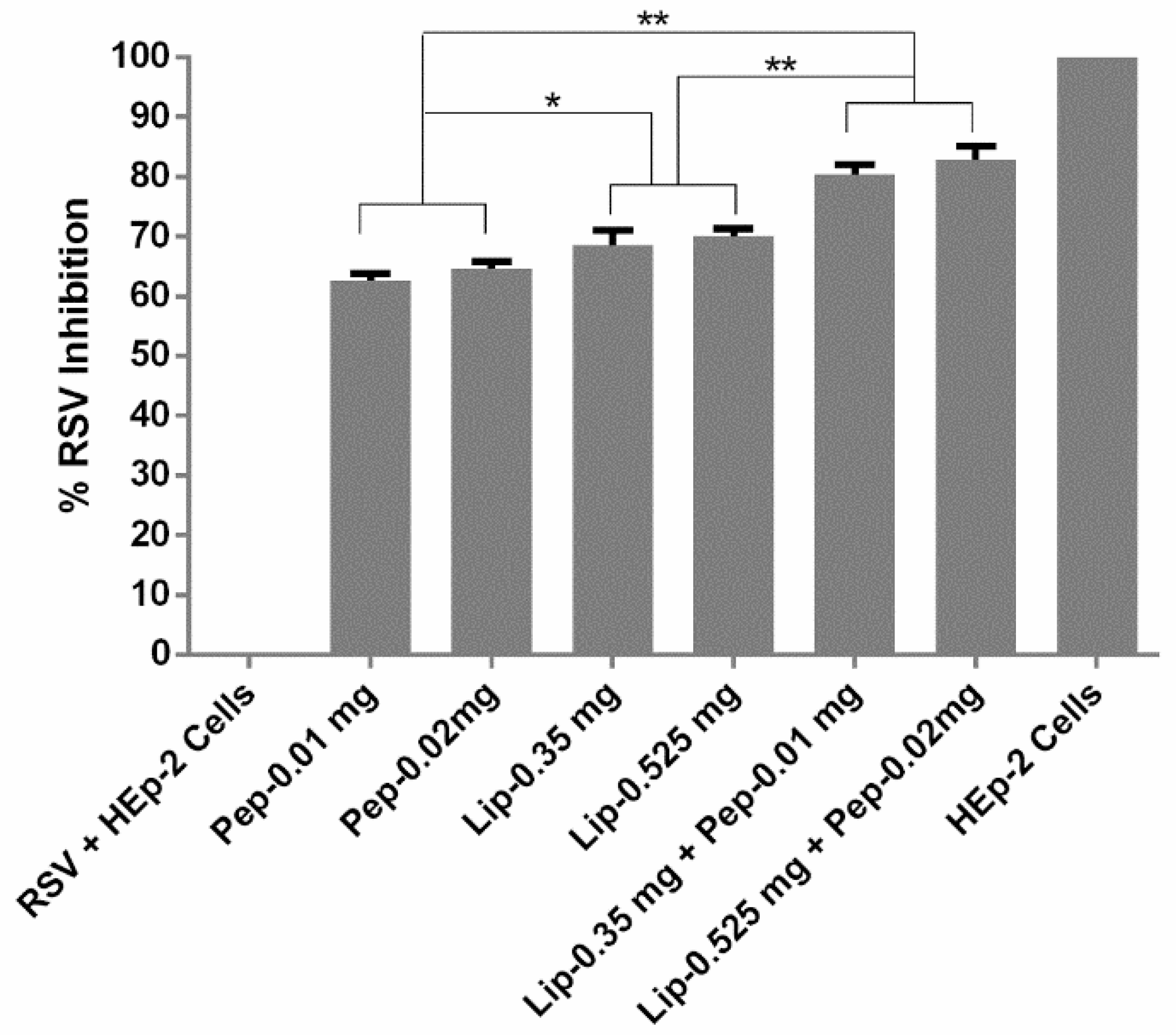
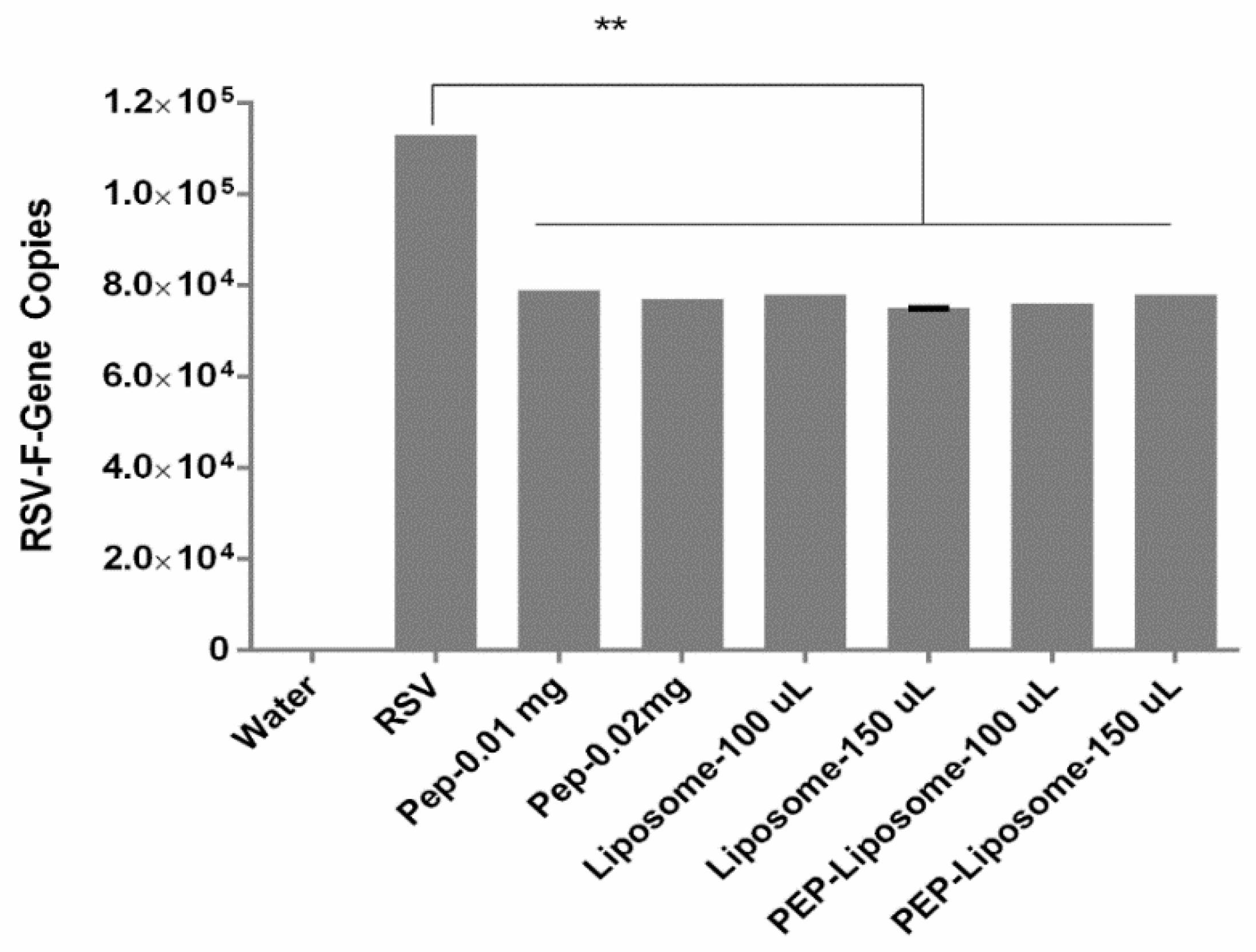
3.4. Evaluation of Viral Inhibition
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Luchsinger, V.; Ampuero, S.; Palomino, M.A.; Chnaiderman, J.; Levican, J.; Gaggero, A.; Larrañaga, C.E. Comparison of virological profiles of respiratory syncytial virus and rhinovirus in acute lower tract respiratory infections in very young Chilean infants, according to their clinical outcome. J. Clin. Virol. 2014, 61, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Tregoning, J.S.; Schwarze, J. Respiratory viral infections in infants: Causes, clinical symptoms, virology, and immunology. Clin. Microbiol. Rev. 2010, 23, 74–98. [Google Scholar] [CrossRef] [PubMed]
- Falsey, A.R.; Hennessey, P.A.; Formica, M.A.; Cox, C.; Walsh, E.E. Respiratory syncytial virus infection in elderly and high-risk adults. N. Engl. J. Med. 2005, 352, 1749–1759. [Google Scholar] [CrossRef] [PubMed]
- Rappuoli, R.; Mandl, C.W.; Black, S.; De Gregorio, E. Vaccines for the twenty-first century society. Nat. Rev. Immunol. 2011, 11, 865–872. [Google Scholar] [CrossRef] [PubMed]
- Borchers, A.T.; Chang, C.; Gershwin, M.E.; Gershwin, L.J. Respiratory syncytial virus—A comprehensive review. Clin. Rev. Allergy Immunol. 2013, 45, 331–379. [Google Scholar] [CrossRef] [PubMed]
- Kamphuis, T.; Meijerhof, T.; Stegmann, T.; Lederhofer, J.; Wilschut, J.; de Haan, A. Immunogenicity and Protective Capacity of a Virosomal Respiratory Syncytial Virus Vaccine Adjuvanted with Monophosphoryl Lipid A in Mice. PLoS ONE 2012, 7, e36812. [Google Scholar] [CrossRef] [PubMed]
- Wyde, P.R. Respiratory syncytial virus (RSV) disease and prospects for its control. Antivir. Res. 1998, 39, 63–79. [Google Scholar] [CrossRef]
- Mufson, M.A.; Belshe, R.B.; Örvell, C.; Norrby, E. Respiratory syncytial virus epidemics: Variable dominance of subgroups A and B strains among children, 1981–1986. J. Infect. Dis. 1988, 157, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Haywood, A.M. Interaction of liposomes with viruses. Ann. N. Y. Acad. Sci. 1978, 308, 275–280. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Singh, M.; Malashkevich, V.N.; Kim, P.S. Structural characterization of the human respiratory syncytial virus fusion protein core. Proc. Natl. Acad. Sci. USA 2000, 97, 14172–14177. [Google Scholar] [CrossRef] [PubMed]
- CDC. Respiratory Syncytial Virus Infection (RSV). 2014. https://www.cdc.gov/rsv/index.html (accessed on 27 April 2016).
- Piedimonte, G.; Perez, M.K. Respiratory Syncytial Virus Infection and Bronchiolitis. Pediatr. Rev. 2014, 35, 519–530. [Google Scholar] [CrossRef] [PubMed]
- Bawage, S.S.; Tiwari, P.M.; Pillai, S.; Dennis, V.; Singh, S.R. Recent advances in diagnosis, prevention, and treatment of human respiratory syncytial virus. Adv. Virol. 2013, 2013, 595768. [Google Scholar] [CrossRef] [PubMed]
- Simões, E.A.; DeVincenzo, J.P.; Boeckh, M.; Bont, L.; Crowe, J.E.; Griffiths, P.; Hayden, F.G.; Hodinka, R.L.; Smyth, R.L.; Spencer, K. Challenges and opportunities in developing respiratory syncytial virus therapeutics. J. Infect. Dis. 2015, 211, S1–S20. [Google Scholar] [CrossRef] [PubMed]
- Schwendener, R.A. Liposomes as vaccine delivery systems: A review of the recent advances. Ther. Adv. Vaccines 2014, 2, 159–182. [Google Scholar] [CrossRef] [PubMed]
- Gregoriadis, G.; Ryman, B.E. Lysosomal localization of β-fructofuranosidase-containing liposomes injected into rats. Some implications in the treatment of genetic disorders. Biochem. J. 1972, 129, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Gregoriadis, G.; Ryman, B.E. Fate of Protein-Containing Liposomes Injected into Rats. Eur. J. Biochem. 1972, 24, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Gregoriadis, G.; Leathwood, P.; Ryman, B.E. Enzyme entrapment in liposomes. FEBS Lett. 1971, 14, 95–99. [Google Scholar] [CrossRef]
- Perrie, Y.; Kastner, E.; Kaur, R.; Wilkinson, A.; Ingham, A.J. A case-study investigating the physicochemical characteristics that dictate the function of a liposomal adjuvant. Hum. Vaccines Immunother. 2013, 9, 1374–1381. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.R.; Tiwari, P.M.; Dennis, V.A. Anti Respiratory Syncytial Virus Peptide Functionalized Gold Nanoparticles. U.S. Patent 8,815,295, 26 August 2014. [Google Scholar]
- Tiwari, P.M.; Eroglu, E.; Bawage, S.S.; Vig, K.; Miller, M.E.; Pillai, S.; Dennis, V.A.; Singh, S.R. Enhanced intracellular translocation and biodistribution of gold nanoparticles functionalized with a cell-penetrating peptide (VG-21) from vesicular stomatitis virus. Biomaterials 2014, 35, 9484–9494. [Google Scholar] [CrossRef] [PubMed]
- Vabbilisetty, P.; Sun, X.-L. Liposome surface functionalization based on different anchoring lipids via Staudinger ligation. Org. Biomol. Chem. 2014, 12, 1237–1244. [Google Scholar] [CrossRef] [PubMed]
- Hendricks, G.L.; Velazquez, L.; Pham, S.; Qaisar, N.; Delaney, J.C.; Viswanathan, K.; Albers, L.; Comolli, J.C.; Shriver, Z.; Knipe, D.M. Heparin octasaccharide decoy liposomes inhibit replication of multiple viruses. Antivir. Res. 2015, 116, 34–44. [Google Scholar] [CrossRef] [PubMed]
- Joshi, S.; Kirby, D.; Perrie, Y.; Singh, S.R. Novel nano-biomaterials for inhibition of respiratory syncytial virus. Proceedings of TechConnect, Washington, DC, USA, 14 May 2017; pp. 75–78. [Google Scholar]
- Bangham, A.D.; Standish, M.M.; Watkins, J.C. Diffusion of univalent ions across the lamellae of swollen phospholipids. J. Mol. Biol. 1965, 13, 238–252. [Google Scholar] [CrossRef]
- Mentel, R.; Wegner, U.; Bruns, R.; Gürtler, L. Real-time PCR to improve the diagnosis of respiratory syncytial virus infection. J. Med. Microbiol. 2003, 52, 893–896. [Google Scholar] [CrossRef] [PubMed]
- Eroglu, E.; Tiwari, P.M.; Waffo, A.B.; Miller, M.E.; Vig, K.; Dennis, V.A.; Singh, S.R. A nonviral pHEMA+ chitosan nanosphere-mediated high-efficiency gene delivery system. Int. J. Nanomed. 2013, 8, 1403–1415. [Google Scholar]
- Nahar, K.; Absar, S.; Gupta, N.; Kotamraju, V.R.; McMurtry, I.F.; Oka, M.; Komatsu, M.; Nozik-Grayck, E.; Ahsan, F. Peptide-coated liposomal fasudil enhances site specific vasodilation in pulmonary arterial hypertension. Mol. Pharm. 2014, 11, 4374–4384. [Google Scholar] [CrossRef] [PubMed]
- Landry, M.L.; Stanat, S.; Biron, K.; Brambilla, D.; Britt, W.; Jokela, J.; Chou, S.; Drew, W.L.; Erice, A.; Gilliam, B. A standardized plaque reduction assay for determination of drug susceptibilities of cytomegalovirus clinical isolates. Antimicrob. Agents Chemother. 2000, 44, 688–692. [Google Scholar] [CrossRef] [PubMed]
- Baer, A.; Kehn-Hall, K. Viral concentration determination through plaque assays: Using traditional and novel overlay systems. J. Vis. Exp. 2014, 4, e52065. [Google Scholar] [CrossRef] [PubMed]
- Numata, M.; Chu, H.W.; Dakhama, A.; Voelker, D.R. Pulmonary surfactant phosphatidylglycerol inhibits respiratory syncytial virus–induced inflammation and infection. Proc. Natl. Acad. Sci. USA 2010, 107, 320–325. [Google Scholar] [CrossRef] [PubMed]
- Connors, M.; Kulkarni, A.; Collins, P.; Firestone, C.; Holmes, K.; Morse, H.D.; Murphy, B. Resistance to respiratory syncytial virus (RSV) challenge induced by infection with a vaccinia virus recombinant expressing the RSV M2 protein (Vac-M2) is mediated by CD8+ T cells, while that induced by Vac-F or Vac-G recombinants is mediated by antibodies. J. Virol. 1992, 66, 1277–1281. [Google Scholar] [PubMed]
- Mader, D.; Huang, Y.; Wang, C.; Fraser, R.; Issekutz, A.C.; Stadnyk, A.W.; Anderson, R. Liposome encapsulation of a soluble recombinant fragment of the respiratory syncytial virus (RSV) G protein enhances immune protection and reduces lung eosinophilia associated with virus challenge. Vaccine 2000, 18, 1110–1117. [Google Scholar] [CrossRef]
- Li, Y.; To, J.; Verdià-Baguena, C.; Dossena, S.; Surya, W.; Huang, M.; Paulmichl, M.; Liu, D.X.; Aguilella, V.M.; Torres, J. Inhibition of the human respiratory syncytial virus small hydrophobic protein and structural variations in a bicelle environment. J. Virol. 2014, 88, 11899–11914. [Google Scholar] [CrossRef] [PubMed]
- Drugbank. BTA9881. https://www.drugbank.ca/drugs/DB05226 (accessed on 22 September 2017).
- ClinicalTrials.gov. Safety Study of Oral BTA9881 to Treat RSV Infection. https://clinicaltrials.gov/ct2/show/NCT00504907 (accessed on 22 September 2017).
- Costello, H.M. The N500 Glycan of the Respiratory Syncytial Virus F Protein is Required for Fusion, but Not for Stabilization or Triggering of the Protein; The Ohio State University: Columbus, OH, USA, 2013. [Google Scholar]
- Hancock, G.E.; Speelman, D.J.; Frenchick, P.J. Adjuvants for Vaccines against Respiratory Syncytial Virus. U.S. Patents 5723130A, 3 March 1998. [Google Scholar]
- McLellan, J.S.; Ray, W.C.; Peeples, M.E. Structure and function of respiratory syncytial virus surface glycoproteins. In Challenges and Opportunities for Respiratory Syncytial Virus Vaccines; Springer: Berlin/Heidelberg, Germany, 2013; pp. 83–104. [Google Scholar]
- Bukreyev, A.; Yang, L.; Collins, P.L. The secreted G protein of human respiratory syncytial virus antagonizes antibody-mediated restriction of replication involving macrophages and complement. J. Virol. 2012, 86, 10880–10884. [Google Scholar] [CrossRef] [PubMed]
- Heminway, B.; Yu, Y.; Tanaka, Y.; Perrine, K.; Gustafson, E.; Bernstein, J.; Galinski, M. Analysis of respiratory syncytial virus F, G, and SH proteins in cell fusion. Virology 1994, 200, 801–805. [Google Scholar] [CrossRef] [PubMed]
- Techaarpornkul, S.; Barretto, N.; Peeples, M.E. Functional analysis of recombinant respiratory syncytial virus deletion mutants lacking the small hydrophobic and/or attachment glycoprotein gene. J. Virol. 2001, 75, 6825–6834. [Google Scholar] [CrossRef] [PubMed]
- Battles, M.B.; Langedijk, J.P.; Furmanova-Hollenstein, P.; Chaiwatpongsakorn, S.; Costello, H.M.; Kwanten, L.; Vranckx, L.; Vink, P.; Jaensch, S.; Jonckers, T.H. Molecular mechanism of respiratory syncytial virus fusion inhibitors. Nat. Chem. Biol. 2016, 12, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Perron, M.; Stray, K.; Kinkade, A.; Theodore, D.; Lee, G.; Eisenberg, E.; Sangi, M.; Gilbert, B.E.; Jordan, R.; Piedra, P.A. GS-5806 inhibits a broad range of respiratory syncytial virus clinical isolates by blocking the virus-cell fusion process. Antimicrob. Agents Chemother. 2016, 60, 1264–1273. [Google Scholar] [CrossRef] [PubMed]
- Samuel, D.; Xing, W.; Niedziela-Majka, A.; Wong, J.S.; Hung, M.; Brendza, K.M.; Perron, M.; Jordan, R.; Sperandio, D.; Liu, X. GS-5806 inhibits pre-to postfusion conformational changes of the respiratory syncytial virus fusion protein. Antimicrob. Agents Chemother. 2015, 59, 7109–7112. [Google Scholar] [CrossRef] [PubMed]
- Perrie, Y.; Mohammed, A.R.; Kirby, D.J.; McNeil, S.E.; Bramwell, V.W. Vaccine adjuvant systems: Enhancing the efficacy of sub-unit protein antigens. Int. J. Pharm. 2008, 364, 272–280. [Google Scholar] [CrossRef] [PubMed]
- Allison, A.; Gregoriadis, G. Liposomes as immunological adjuvants. Nature 1974, 252, 252. [Google Scholar] [CrossRef] [PubMed]
- Perrie, Y.; Frederik, P.M.; Gregoriadis, G. Liposome-mediated DNA vaccination: The effect of vesicle composition. Vaccine 2001, 19, 3301–3310. [Google Scholar] [CrossRef]
- Kim, K.S.; Kim, A.-R.; Piao, Y.; Lee, J.-H.; Quan, F.-S. A rapid, simple, and accurate plaque assay for human respiratory syncytial virus (HRSV). J. Immunol. Methods 2017, 446, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Numata, M.; Kandasamy, P.; Nagashima, Y.; Fickes, R.; Murphy, R.C.; Voelker, D.R. Phosphatidylinositol inhibits respiratory syncytial virus infection. J. Lipid Res. 2015, 56, 578–587. [Google Scholar] [CrossRef] [PubMed]
- Joshi, S.; Hussain, M.T.; Roces, C.B.; Anderluzzi, G.; Kastner, E.; Salmaso, S.; Kirby, D.J.; Perrie, Y. Microfluidics based manufacture of liposomes simultaneously entrapping hydrophilic and lipophilic drugs. Int. J. Pharm. 2016, 514, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Zurbriggen, R.; Amacker, M.; Krammer, A.R. Immunopotentiating reconstituted influenza virosomes. Liposome Technol. 2006, 1, 85–96. [Google Scholar]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Joshi, S.; Chaudhari, A.A.; Dennis, V.; Kirby, D.J.; Perrie, Y.; Singh, S.R. Anti-RSV Peptide-Loaded Liposomes for the Inhibition of Respiratory Syncytial Virus. Bioengineering 2018, 5, 37. https://doi.org/10.3390/bioengineering5020037
Joshi S, Chaudhari AA, Dennis V, Kirby DJ, Perrie Y, Singh SR. Anti-RSV Peptide-Loaded Liposomes for the Inhibition of Respiratory Syncytial Virus. Bioengineering. 2018; 5(2):37. https://doi.org/10.3390/bioengineering5020037
Chicago/Turabian StyleJoshi, Sameer, Atul A. Chaudhari, Vida Dennis, Daniel J. Kirby, Yvonne Perrie, and Shree Ram Singh. 2018. "Anti-RSV Peptide-Loaded Liposomes for the Inhibition of Respiratory Syncytial Virus" Bioengineering 5, no. 2: 37. https://doi.org/10.3390/bioengineering5020037
APA StyleJoshi, S., Chaudhari, A. A., Dennis, V., Kirby, D. J., Perrie, Y., & Singh, S. R. (2018). Anti-RSV Peptide-Loaded Liposomes for the Inhibition of Respiratory Syncytial Virus. Bioengineering, 5(2), 37. https://doi.org/10.3390/bioengineering5020037







