1. Introduction
Fermentation is a chemical process by which fungi or bacteria metabolize sugar to acids, gases, or alcohol in an environment with lack of oxygen. The two most common species used in this process are
Saccharomyces cerevisiae and
Zymomonas mobilis, where the first one is a fungus used more for industrial processes [
1]. Several biomasses can be used for this purpose including grasses, wood, and commodity grains. For corn, the starch is the primary glucose source for fermentation. The most commonly used method to increase ethanol production involves grinding the corn to fine grades to increase surface area during fermentation.
Fuel ethanol and beverage alcohol are two examples of products that require fermentation to be produced. In the United States of America (USA), ethanol is commonly used domestically and produced mainly from corn. Since large quantities of ethanol are much needed in the USA, production of corn and lignocellulose must be high to meet demand. For 2016, the production of fuel ethanol was 366,981 thousand barrels, and consumption was of 341,817.322 thousand barrels [
2]. According to the USDA [
3], a total of 15,345 million bushels of corn were produced in the year of 2015/2016, where 1304 million bushels were destined to ethanol production, which means that 31.5% of total produced corn in the US was used for fuel, beverage, solvent, and other uses. For instance, around 23.153 million 9-L cases of Whiskey, a corn-based alcoholic beverage, were produced in the year of 2017, leading to revenue of
$3.368 billion dollars for the American Whiskey suppliers [
4].
Heat is also a byproduct of alcoholic fermentation, which can increase the temperature of the process. During ethanol fermentation, yeast is subject to several forms of stress, such as temperature changes, nutrient deficiency, and bacteria contamination [
1]. In addition, ethanol accumulation inhibits yeast cell growth and consequently slows down ethanol production. All these forms of stress should be monitored due to its effect on yeast viability and vigor to optimize alcohol yield rate.
In ethanol and other fermentation outputs, several limiting factors can be altered to maximize and optimize by-products’ production during this process. A corn fermentation study to produce ammonium lactate (an essential component for ruminant feeds) was conducted by Mercier et al. [
5]. The ammonium lactate is produced by neutralizing the lactic acid from corn fermentation. In this study, pH was controlled to improve ammonium lactate production and found that maximum rate was reached with pH between 6.0 and 6.5. Enzyme activity is also influent to the production of ethanol; therefore, an investigative model was developed to evaluate the enzyme activity. Altintas et al. [
6] developed a kinetic model, which investigated the nonlinear enzymatic effect and how they are affected by heterologous enzymes. The authors concluded that xylose isomerase optimized ethanol production in a continuous fermentor. Moreover, Phisalaphong et al. [
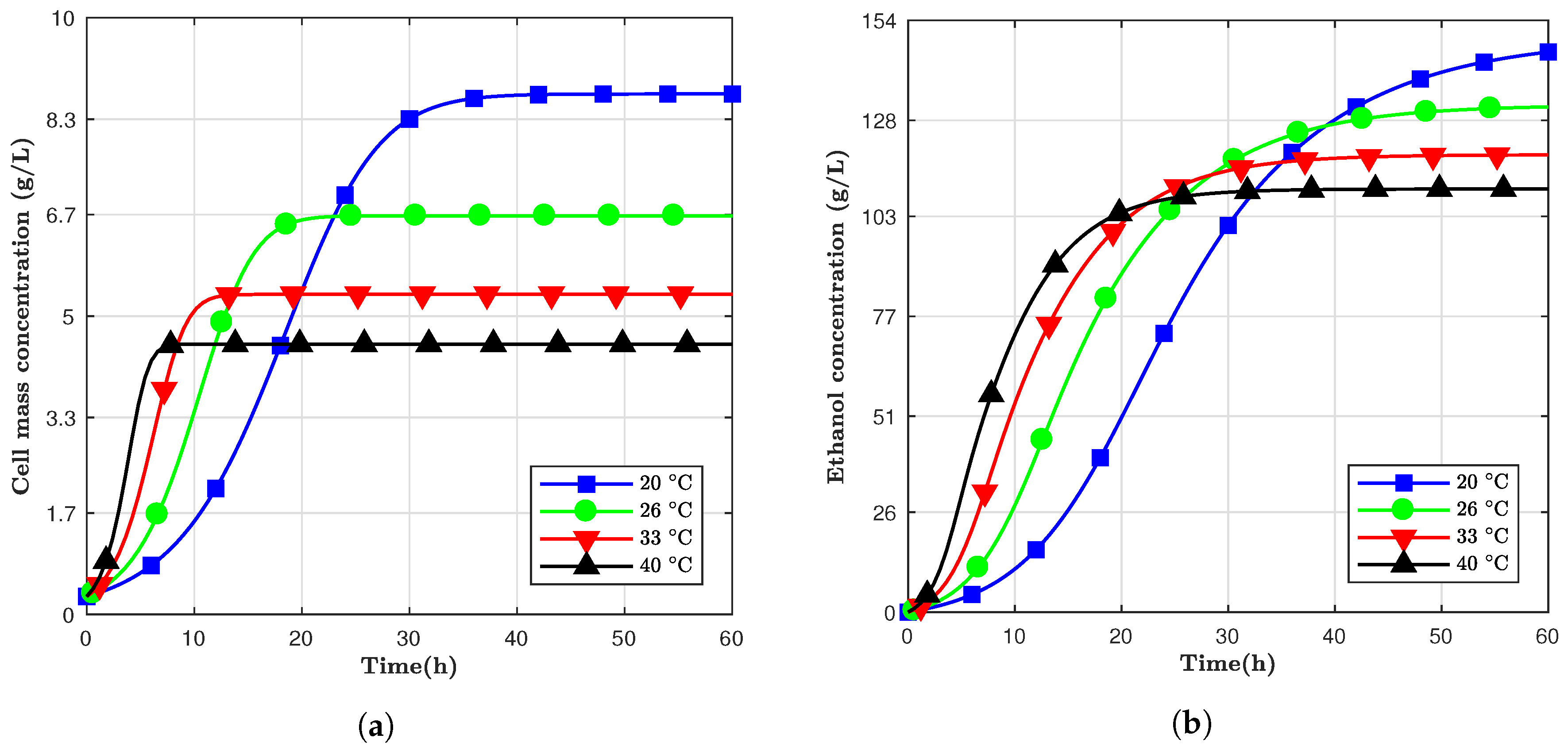
7] investigated the effect of temperature on kinetic parameters of ethanol fermentation using cane molasses as substrate. The results showed that fermentation kept at 30
C had higher ethanol concentrations than the models with the other higher temperatures studied (33, 35, 38, and 42
C).
Although the corn fermentation process is a well-established process, there is still space for improvement. Better monitoring of ethanol production can significantly improve the production of alcohol. Bialas et al. [
8] investigated the effects of mash concentration, enzyme dose, and pH on saccharification and corn starch fermentation for ethanol production. In this study, the mash temperature was kept constant at 35
C; however, there was no report about how the temperature was monitored or controlled during the process. Öhgren et al. [
9] studied two different process configurations. Simultaneous Saccharification and Fermentation (SSF) were used concurrently, with the temperature below 35
C, and Separate Hydrolysis and Fermentation (SHF), where the temperature of the fermentation was kept at 45
C for 120 h. For most cases, the results showed that SSF had higher ethanol yields compared to SHF; however, the temperature was said to be constant.
Creating models to observe the production of ethanol from biomass needs to be continually studied to investigate the numerous variables that can affect the commercialization of the final product. Indeed, mathematical software is necessary to calculate and analyze such models due to its high complexity. Based on computer modeling, the temperature could be controlled during the fermentation process to investigate the effect on ethanol yields.
The overall objective of this study was to implement a fermentation kinetic model to analyze/determine the amount of ethanol produced within a stipulated time, subjected to the catalytic microorganism (Saccharomyces cerevisiae). Additionally, this study aims to investigate the performance of the yeast at different fixed and controlled variable temperatures and determine how these affect the production of ethanol. The specific objectives were to simulate and analyze the fermentation process at different temperatures, perform a parametric sensitivity analysis as a goal to assess the effect of temperature, yeast concentration, and time on the production of ethanol and also design and implement a simulation model with changing temperature to optimize ethanol production.
2. Materials and Methods
As previously stated, in a medium rich with glucose, the yeast begins the process of fermentation under an anaerobic environment. However, the growing presence of alcohol inhibits its growth and consequently decreases the ethanol production rate in the batch.
The fermentation temperature and time should be considered when modeling the yeast behavior and ethanol production. In higher temperatures, yeast tends to grow and produce ethanol through fermentation faster compared to low temperatures. The same alcohol concentration promotes cell death, which will also be faster at higher temperatures, consequently leading to a quicker decay of living cells [
10]. Thus, the ideal process is to achieve an optimum point of temperature for high ethanol production and steady cell mass growth rate.
2.1. Fermentation Modeling
Nanba et al. [
10] proposed a model where the ethanol production and yeast growth are governed by a nonlinear system as observed in Equations (
1) and (
2). The system has two state variables: ethanol concentration in g/L (
p) and cell mass concentration in g/L
:
where
is the specific cell growth rate (h
),
is a growth-associated coefficient, and
(h
) is the specific rate of non-growth associated ethanol production consisting of maintenance and energy uncoupling terms. The ethanol production rate (h
) can be interpreted as
Both parameters
and
also change with time and ethanol concentration, whose initial values depend on some thermodynamic constants. These parameters can be described by the following equations:
where
and
are the initial values for
and
;
and
are deactivation constants;
n is the ethanol inhibition degree;
is the ethanol concentration at discrete step
i and
is the time step. The deactivation constants,
, and
can be calculated according to the following Arrhenius equation since they are constants related to chemical reactions dependent on temperature:
,
,
, and
are constants that depend on the frequency of activated complex formation from the reactants and E is the activation energy. For the reaction, these are all thermodynamics parameters that were determined empirically [
10]. R is the gas constant and equals 1.987 cal K
mol
, and
T is the temperature in Kelvin. For lower temperatures, it can be observed that both initial cell growth rate and ethanol production rate will be lower compared to high temperatures. Based on experimental data, the values for
A and
E could be determined by nonlinear modeling as shown in
Table 1.
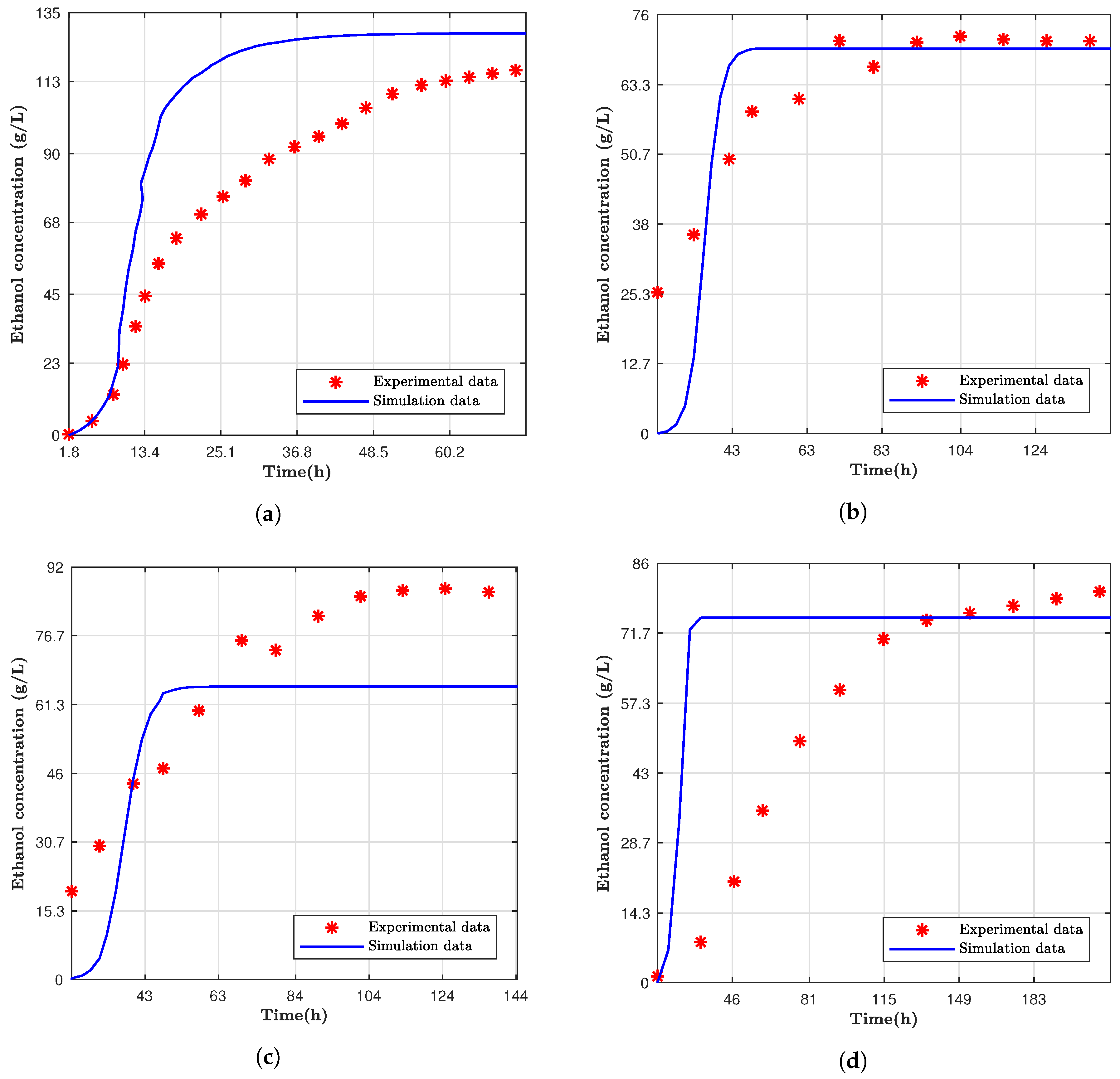
Experimental data from different authors were used to validate the data from the computational model following the same characteristics from these studies [
11,
12].
2.2. Variable Temperature Control
Temperature has a significant influence on the production of ethanol. This can be concluded by directly looking at the Arrhenius equations. Higher temperatures have higher initial values for ethanol production rate and cell growth rate; on the other hand, higher rates mean that ethanol will be produced faster, and inhibition of cell growth will be more prominent, which can lead to smaller final ethanol concentration compared to lower fermentation temperatures.
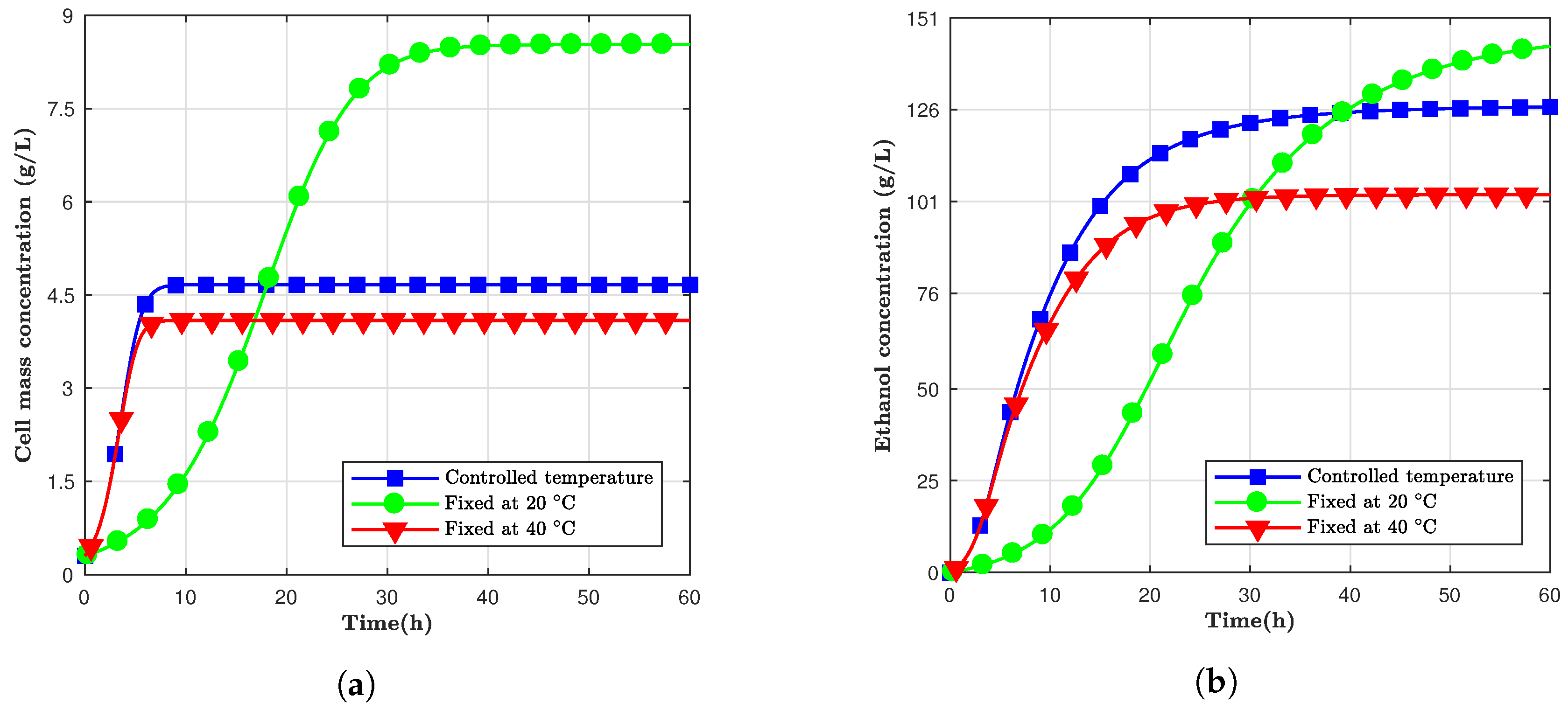
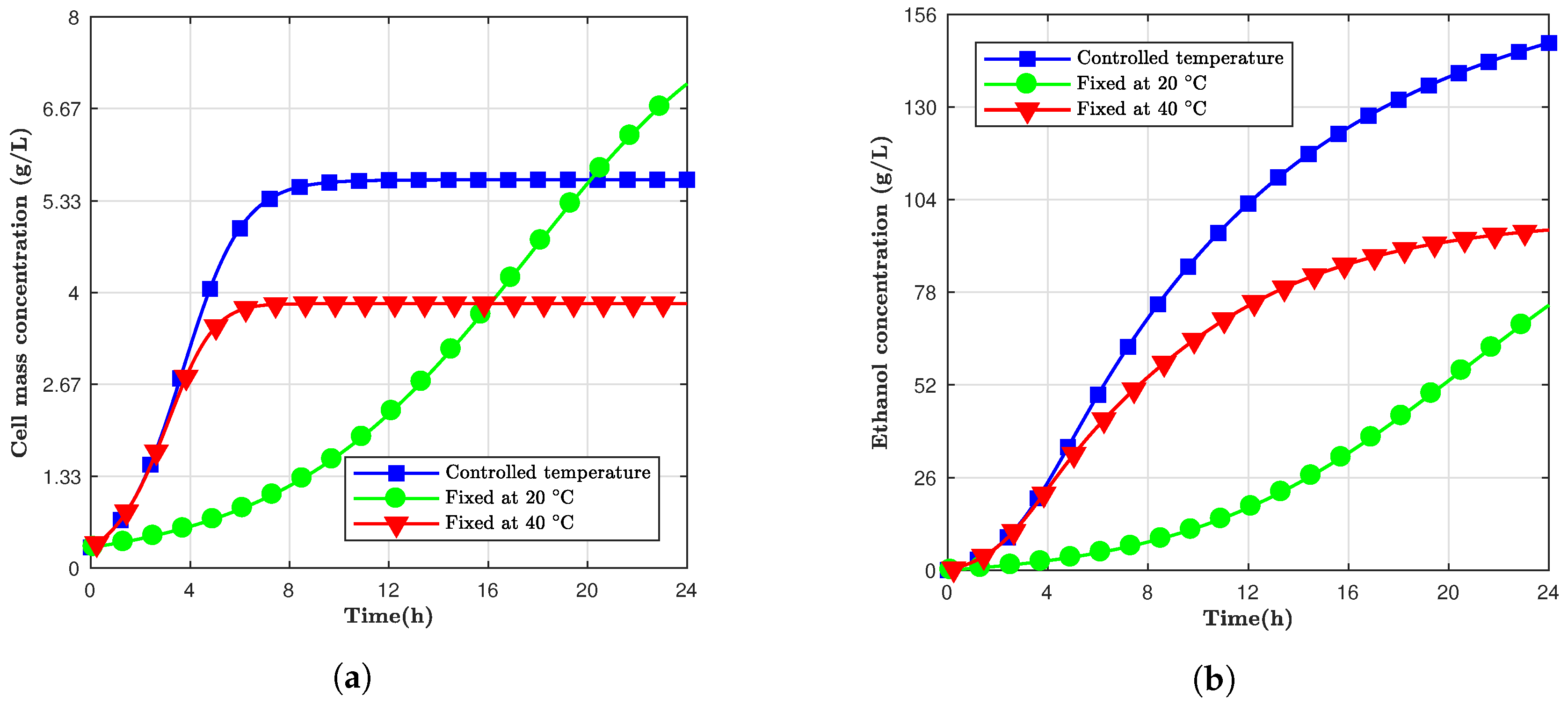
Fermentation is a process that releases energy in the form of heat. If the temperature of the batch can be controlled with time, both ethanol production and cell growth can be optimized to have a faster and more productive batch fermentation.
The
Saccharomyces cerevisiae is a mesophilic organism, that is, it has an optimum growth temperature between 28 and 35
C [
13]. Kishimoto also showed how this yeast has a higher specific growth rate in a fermentation process at 35
C than at 28
C. However, the final ethanol yield was smaller for the higher temperature compared to the lower one. Based on that, a fermentation model was designed where the temperature was controlled over time. The temperature was initiated at 40
C, to take advantage of the higher specific growth, and exponentially reduced to a final temperature of 20
C to optimize the final ethanol yield. The chosen temperature behavior was in the form of Equation (
10):
where
c is a constant, derived from Equation (
11):
Considering that the initial and final value of temperature were, respectively, 40
C and 20
C, and
t is the total time of the fermentation,
c will be equal to 0.0116 h
for a simulation of 60 h and 0.0289 for a 24 h simulation; thus, Equation (
10) will have the following form:
For this simulation, not only were the ethanol and yeast concentrations variables, but also the temperature. Thus, every time the temperature changed, a new set of kinetic parameters had to be recalculated.
This study did not take into consideration contamination of bacteria or another organism in the batch. It was assumed that the pH was constant and under control the entire time. In addition, the concentration of glucose in the batch was considered sufficient, minimizing competition for nutrients during the fermentation.
2.3. Sensitivity Analysis
In this project, a parametric sensitivity analysis was also performed to investigate the model’s performance. To execute the sensitivity analysis, a series of simulations were required. The Latin Hypercube Sampling (LHS) method was chosen to produce design points for the necessary simulations. LHS is a space-filling design method, which maximizes the minimum distance between design points and requires even spacing of the levels for each factor [
14]. This method produces designs that mimic the uniform distribution of design points over parameters’ domain. Yeast concentration, temperature, and fermentation time were the deterministic parameters of interest for the sensitivity investigation over final ethanol concentration and ethanol production rate (final production divided by fermentation time) as response parameters. The range of values for the parameters was decided based on optimal range reported in the literature with some expansion.
Table 2 shows the range of values for variable parameters.
After implementing the design points and running the model to predict response values, a Gaussian process model was used to investigate the sensitivity of input parameters on response variables. The Gaussian Process platform fits a spatial correlation model to the data. The correlation of the response between two observations decreases as the values of the independent variables become more distant. This platform was used to quantify the main and marginal effects, in addition to the total sensitivity of each factor. The JMP [
15] software package (JMP
® Pro 13.2.0, SAS Institute Inc., Cary, NC, USA) was used to produce the design points and perform sensitivity analysis.