Role of Platelet-Rich Plasma Injection in Anterior Cruciate Ligament Reconstruction: A Meta-Analysis of Randomized Controlled Trials
Abstract
1. Introduction
2. Methods
2.1. Literature Search
2.2. Study Eligibility
2.3. Inclusion Criteria
2.4. Exclusion Criteria
2.5. Data Extraction
2.6. Quality Assessment
2.7. Statistical Analysis
3. Results
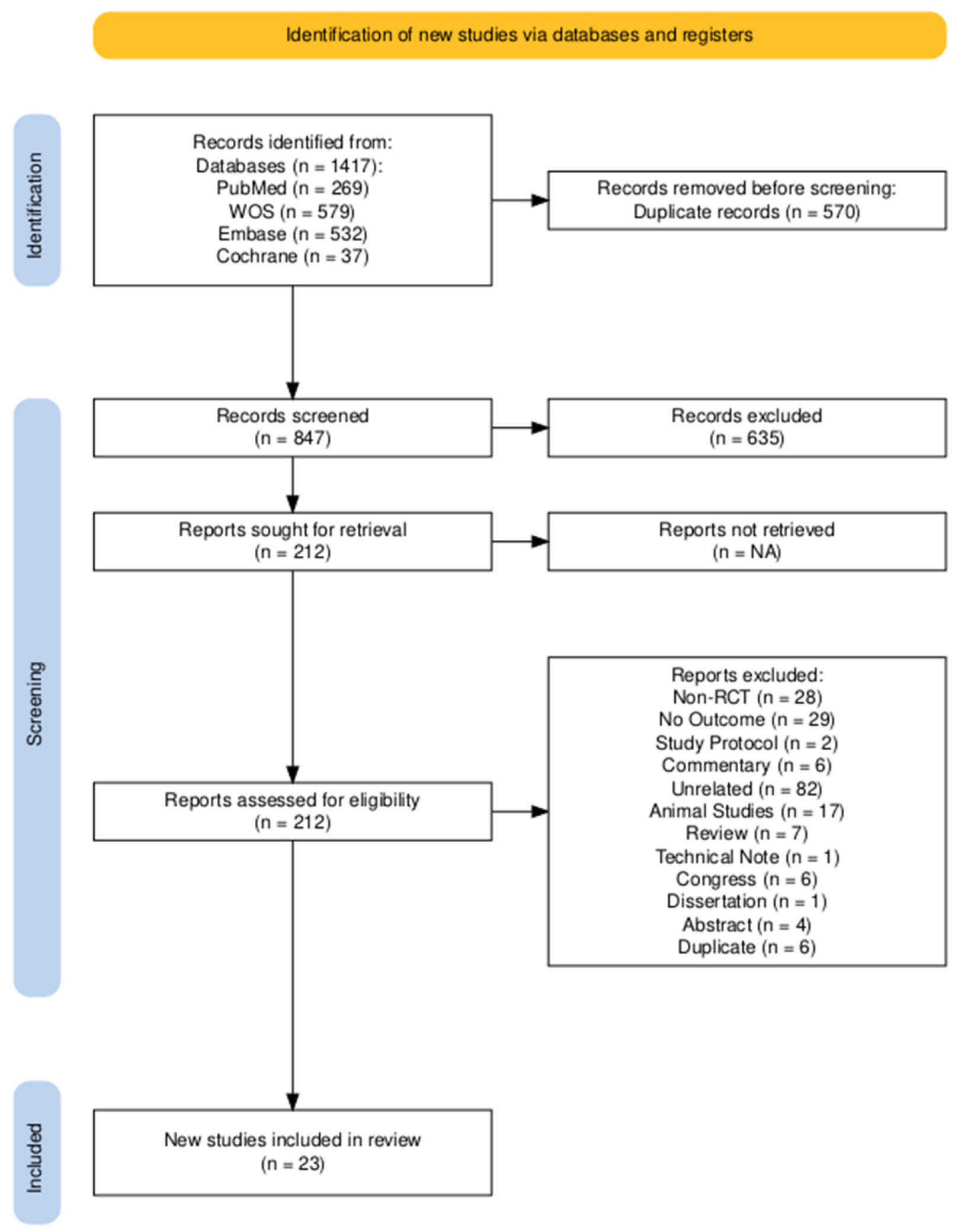
3.1. Literature Selection
3.2. Basic Characteristics of the Literature
3.3. Quality Assessment
3.4. VAS Score
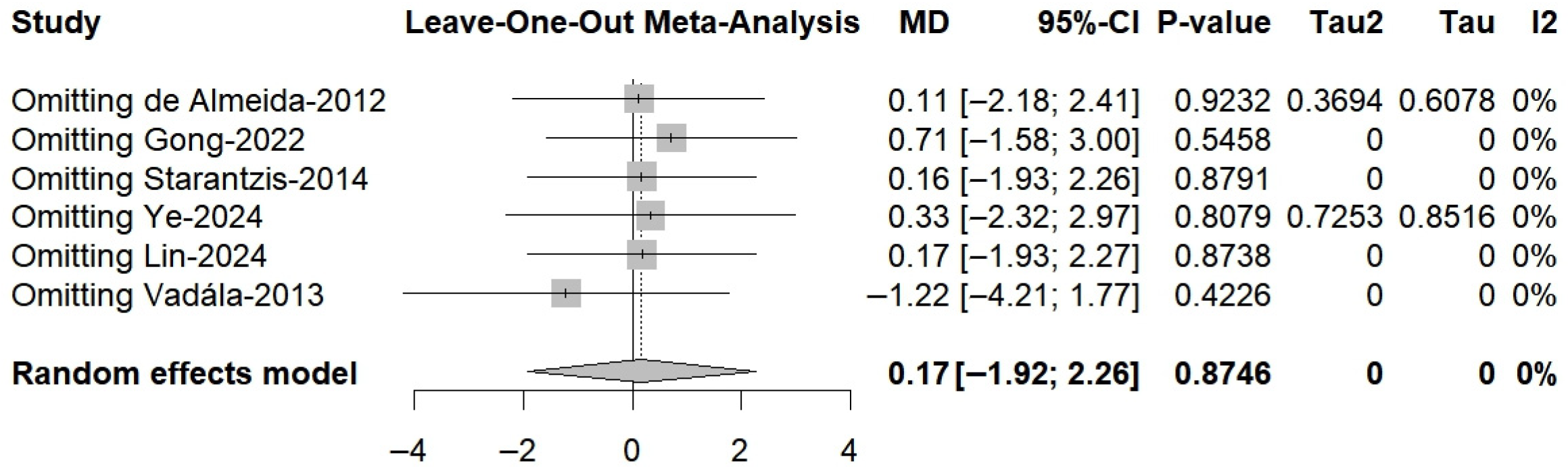
3.5. IKDC Score
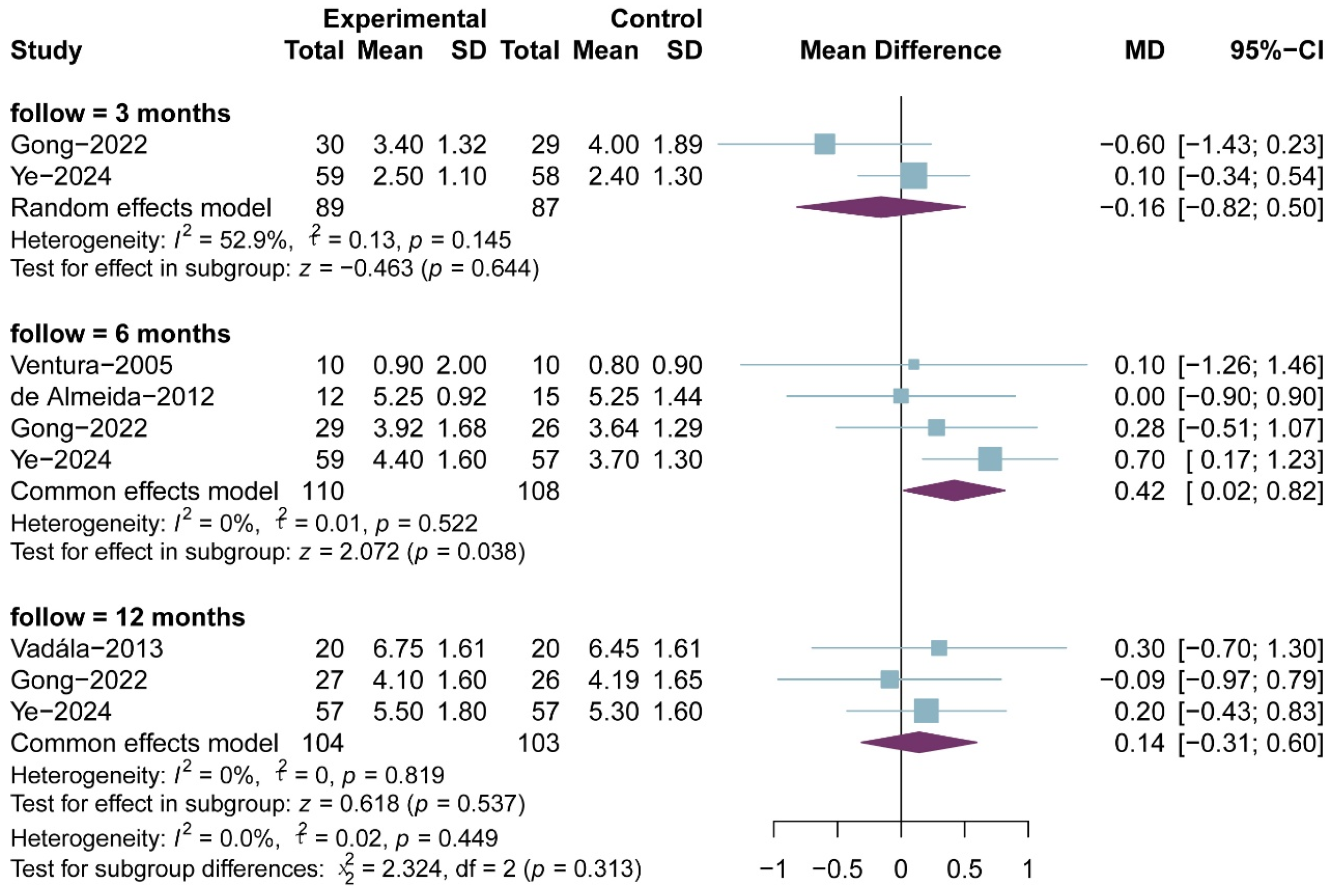
3.6. Lysholm Score
3.7. Tegner Score
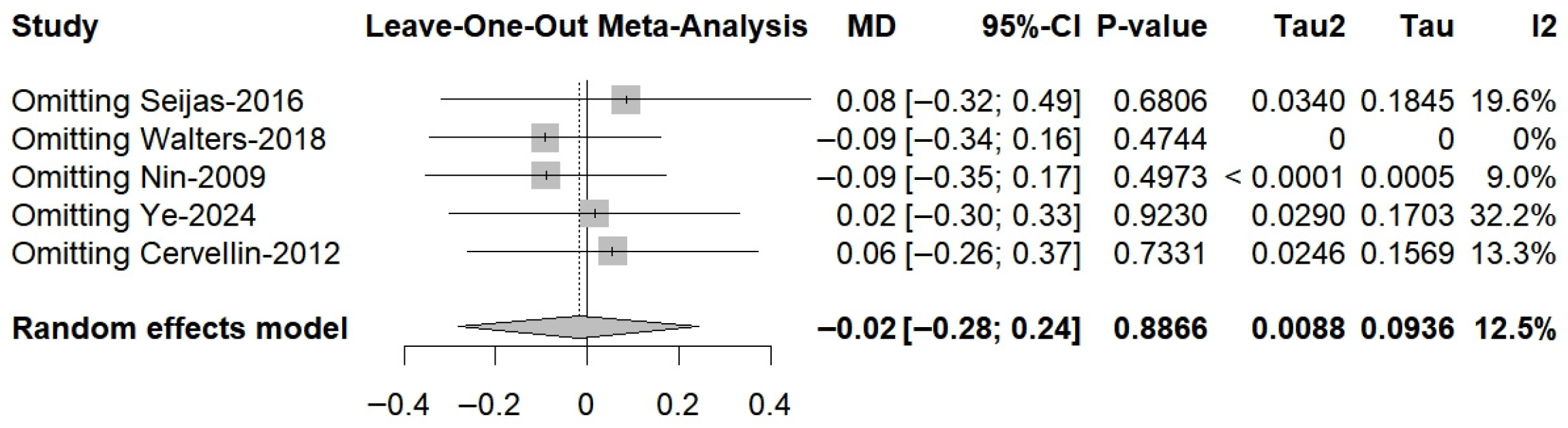
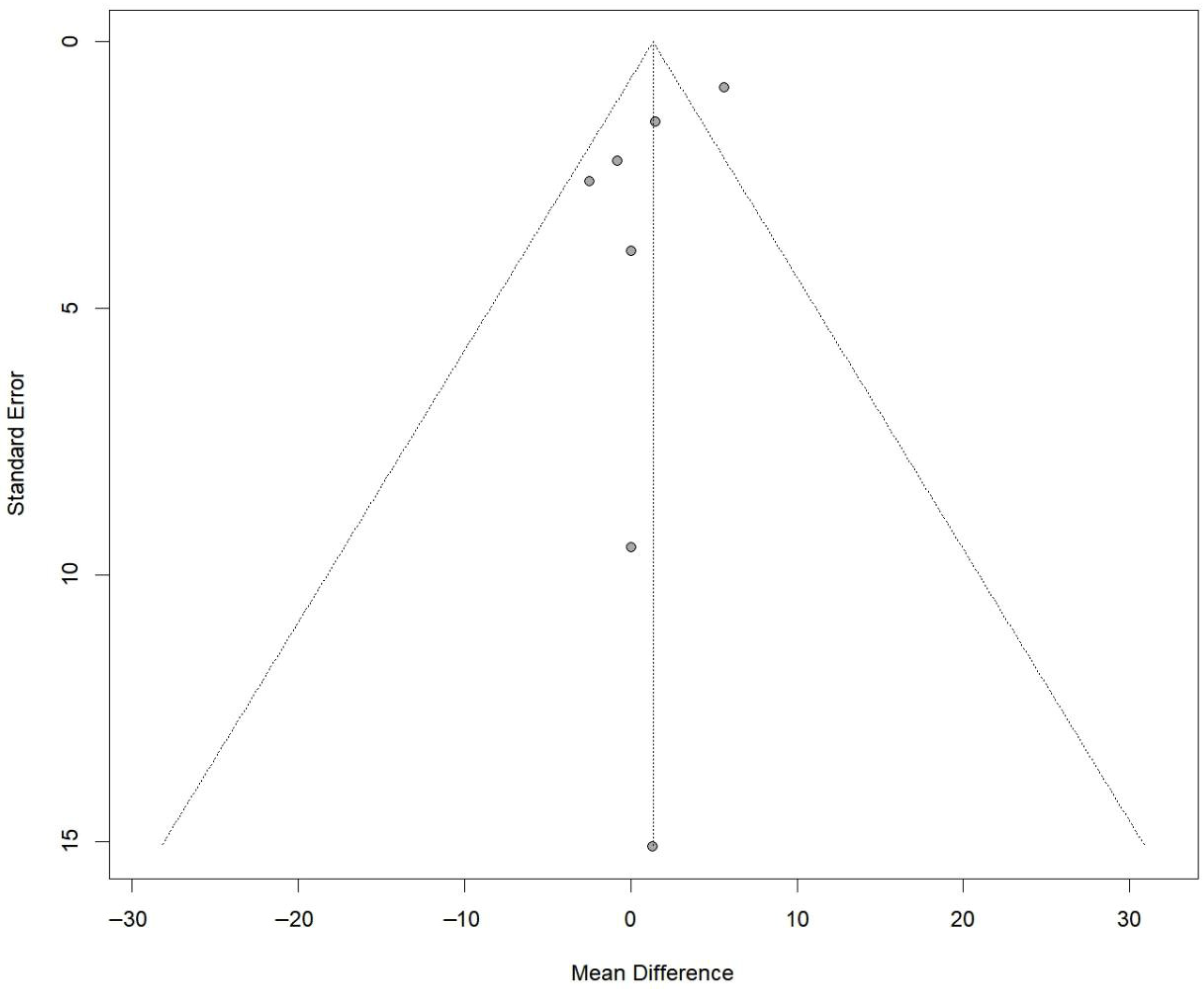
3.8. Anterior Knee Laxity
3.9. Physical Examinations and Other Outcomes
3.10. Radiological Outcomes
3.11. Data Synthesis of PRP Parameters
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACL | Anterior Cruciate Ligament |
| PRP | Platelet-Rich Plasma |
| PRISMA | Preferred Reporting Items for Systematic Reviews and Meta-Analyses |
| IKDC | International Knee Documentation Committee |
| MD | Mean Difference |
| CI | Confidence Interval |
| RCTs | Randomized Controlled Trials |
| KOOS | Knee Injury and Osteoarthritis Outcome Score |
| VAS | Visual Analogue Scale |
References
- Buoncristiani, A.M.; Tjoumakaris, F.P.; Starman, J.S.; Ferretti, M.; Fu, F.H. Anatomic double-bundle anterior cruciate ligament reconstruction. Arthroscopy 2006, 22, 1000–1006. [Google Scholar] [CrossRef]
- Shea, K.G.; Carey, J.L. Management of anterior cruciate ligament injuries: Evidence-based guideline. J. Am. Acad. Orthop. Surg. 2015, 23, e1–e5. [Google Scholar] [CrossRef]
- Griffin, L.Y.; Albohm, M.J.; Arendt, E.A.; Bahr, R.; Beynnon, B.D.; Demaio, M.; Dick, R.W.; Engebretsen, L.; Garrett, W.E.; Hannafin, J.A.; et al. Understanding and preventing noncontact anterior cruciate ligament injuries: A review of the Hunt Valley II meeting, January 2005. Am. J. Sports Med. 2006, 34, 1512–1532. [Google Scholar] [CrossRef]
- Sanders, T.L.; Maradit Kremers, H.; Bryan, A.J.; Larson, D.R.; Dahm, D.L.; Levy, B.A.; Stuart, M.J.; Krych, A.J. Incidence of Anterior Cruciate Ligament Tears and Reconstruction: A 21-Year Population-Based Study. Am. J. Sports Med. 2016, 44, 1502–1507. [Google Scholar] [CrossRef]
- Gong, H.; Huang, B.; Zheng, Z.; Fu, L.; Chen, L. Clinical Use of Platelet-Rich Plasma to Promote Tendon-Bone Healing and Graft Maturation in Anterior Cruciate Ligament Reconstruction-A Randomized Controlled Study. Indian J. Orthop. 2022, 56, 805–811. [Google Scholar] [CrossRef] [PubMed]
- Piedade, S.R.; Leite Arruda, B.P.; de Vasconcelos, R.A.; Parker, D.A.; Maffulli, N. Rehabilitation following surgical reconstruction for anterior cruciate ligament insufficiency: What has changed since the 1960s?—State of the art. J. Isakos 2023, 8, 153–162. [Google Scholar] [CrossRef]
- Giordano, L.; Maffulli, N.; Carimati, G.; Morenghi, E.; Volpi, P. Increased Time to Surgery After Anterior Cruciate Ligament Tear in Female Patients Results in Greater Risk of Medial Meniscus Tear: A Study of 489 Female Patients. Arthroscopy 2023, 39, 613–622. [Google Scholar] [CrossRef]
- Freedman, K.B.; D’Amato, M.J.; Nedeff, D.D.; Kaz, A.; Bach, B.R., Jr. Arthroscopic anterior cruciate ligament reconstruction: A metaanalysis comparing patellar tendon and hamstring tendon autografts. Am. J. Sports Med. 2003, 31, 2–11. [Google Scholar] [CrossRef] [PubMed]
- Filbay, S.R.; Ackerman, I.N.; Russell, T.G.; Macri, E.M.; Crossley, K.M. Health-related quality of life after anterior cruciate ligament reconstruction: A systematic review. Am. J. Sports Med. 2014, 42, 1247–1255. [Google Scholar] [CrossRef] [PubMed]
- Filbay, S.R.; Grindem, H. Evidence-based recommendations for the management of anterior cruciate ligament (ACL) rupture. Best Pract. Res. Clin. Rheumatol. 2019, 33, 33–47. [Google Scholar] [CrossRef]
- Du, T.; Shi, Y.; Huang, H.; Liang, W.; Miao, D. Current study on the influence of psychological factors on returning to sports after ACLR. Heliyon 2022, 8, e12434. [Google Scholar] [CrossRef] [PubMed]
- Solomon, R.; Hommen, J.P.; Travascio, F. Effects of Platelet-Rich Osteoconductive-Osteoinductive Allograft Compound on Tunnel Widening of ACL Reconstruction: A Randomized Blind Analysis Study. Pathophysiology 2022, 29, 394–404. [Google Scholar] [CrossRef] [PubMed]
- Bailey, L.; Weldon, M.; Kleihege, J.; Lauck, K.; Syed, M.; Mascarenhas, R.; Lowe, W.R. Platelet-Rich Plasma Augmentation of Meniscal Repair in the Setting of Anterior Cruciate Ligament Reconstruction. Am. J. Sports Med. 2021, 49, 3287–3292. [Google Scholar] [CrossRef]
- Magnussen, R.A.; Lawrence, J.T.; West, R.L.; Toth, A.P.; Taylor, D.C.; Garrett, W.E. Graft size and patient age are predictors of early revision after anterior cruciate ligament reconstruction with hamstring autograft. Arthroscopy 2012, 28, 526–531. [Google Scholar] [CrossRef] [PubMed]
- Ardern, C.L.; Taylor, N.F.; Feller, J.A.; Webster, K.E. Fifty-five per cent return to competitive sport following anterior cruciate ligament reconstruction surgery: An updated systematic review and meta-analysis including aspects of physical functioning and contextual factors. Br. J. Sports Med. 2014, 48, 1543–1552. [Google Scholar] [CrossRef]
- Marois, B.; Tan, X.W.; Pauyo, T.; Dodin, P.; Ballaz, L.; Nault, M.L. Can a Knee Brace Prevent ACL Reinjury: A Systematic Review. Int. J. Environ. Res. Public Health 2021, 18, 7611. [Google Scholar] [CrossRef]
- Guy, S.; Fayard, J.M.; Saithna, A.; Bahroun, S.; Ferreira, A.; Carrozzo, A.; De Jesus, S.; Bulle, S.; Vieira, T.D.; Sonnery-Cottet, B. Risk of Graft Rupture After Adding a Lateral Extra-articular Procedure at the Time of ACL Reconstruction: A Retrospective Comparative Study of Elite Alpine Skiers From the French National Team. Am. J. Sports Med. 2022, 50, 1609–1617. [Google Scholar] [CrossRef]
- Tischer, T.; Beaufilis, P.; Becker, R.; Ahmad, S.S.; Bonomo, M.; Dejour, D.; Eriksson, K.; Filardo, G.; Feucht, M.J.; Grassi, A. Management of anterior cruciate ligament revision in adults: The 2022 ESSKA consensus part I-diagnostics and preoperative planning. Knee Surg. Sports Traumatol. Arthrosc. 2023, 31, 4642–4651. [Google Scholar] [CrossRef]
- Brinlee, A.W.; Dickenson, S.B.; Hunter-Giordano, A.; Snyder-Mackler, L. ACL Reconstruction Rehabilitation: Clinical Data, Biologic Healing, and Criterion-Based Milestones to Inform a Return-to-Sport Guideline. Sports Health 2022, 14, 770–779. [Google Scholar] [CrossRef]
- Le, A.D.K.; Enweze, L.; DeBaun, M.R.; Dragoo, J.L. Current Clinical Recommendations for Use of Platelet-Rich Plasma. Curr. Rev. Musculoskelet. Med. 2018, 11, 624–634. [Google Scholar] [CrossRef]
- Lopez-Vidriero, E.; Goulding, K.A.; Simon, D.A.; Sanchez, M.; Johnson, D.H. The use of platelet-rich plasma in arthroscopy and sports medicine: Optimizing the healing environment. Arthroscopy 2010, 26, 269–278. [Google Scholar] [CrossRef]
- McCarrel, T.M.; Minas, T.; Fortier, L.A. Optimization of leukocyte concentration in platelet-rich plasma for the treatment of tendinopathy. J. Bone Jt. Surg. Am. 2012, 94, e143. [Google Scholar] [CrossRef] [PubMed]
- Southworth, T.M.; Naveen, N.B.; Nwachukwu, B.U.; Cole, B.J.; Frank, R.M. Orthobiologics for Focal Articular Cartilage Defects. Clin. Sports Med. 2019, 38, 109–122. [Google Scholar] [CrossRef] [PubMed]
- Anitua, E.; Andia, I.; Ardanza, B.; Nurden, P.; Nurden, A.T. Autologous platelets as a source of proteins for healing and tissue regeneration. Thromb. Haemost. 2004, 91, 4–15. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.H.; Jeon, S.H.; Park, J.Y.; Chung, J.H.; Choung, Y.H.; Choung, H.W.; Kim, E.-S.; Choung, P.-H. Platelet-rich fibrin is a Bioscaffold and reservoir of growth factors for tissue regeneration. Tissue Eng. Part A 2011, 17, 349–359. [Google Scholar] [CrossRef]
- Zhu, T.; Zhou, J.; Hwang, J.; Xu, X. Effects of Platelet-Rich Plasma on Clinical Outcomes After Anterior Cruciate Ligament Reconstruction: A Systematic Review and Meta-analysis. Orthop. J. Sports Med. 2022, 10, 23259671211061535. [Google Scholar] [CrossRef]
- Cao, Y.; Wan, Y.D. Effectiveness of Platelet-Rich Plasma in Anterior Cruciate Ligament Reconstruction: A Systematic Review of Randomized Controlled Trials. Orthop. Surg. 2022, 14, 2406–2417. [Google Scholar] [CrossRef]
- de Andrade, A.L.L.; Sardeli, A.V.; Garcia, T.A.; Livani, B.; Belangero, W.D. PRP does not improve the objective outcomes of anterior cruciate ligament reconstruction: A systematic review and meta-analysis. Knee Surg. Sports Traumatol. Arthrosc. 2021, 29, 3049–3058. [Google Scholar] [CrossRef]
- Davey, M.S.; Hurley, E.T.; Withers, D.; Moran, R.; Moran, C.J. Anterior Cruciate Ligament Reconstruction with Platelet-Rich Plasma: A Systematic Review of Randomized Control Trials. Arthroscopy 2020, 36, 1204–1210. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Altman, D.G.; Gotzsche, P.C.; Juni, P.; Moher, D.; Oxman, A.D.; Savović, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.C.; et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef]
- Cervellin, M.; de Girolamo, L.; Bait, C.; Denti, M.; Volpi, P. Autologous platelet-rich plasma gel to reduce donor-site morbidity after patellar tendon graft harvesting for anterior cruciate ligament reconstruction: A randomized, controlled clinical study. Knee Surg. Sports Traumatol. Arthrosc. 2012, 20, 114–120. [Google Scholar] [CrossRef]
- de Almeida, A.M.; Demange, M.K.; Sobrado, M.F.; Rodrigues, M.B.; Pedrinelli, A.; Hernandez, A.J. Patellar tendon healing with platelet-rich plasma: A prospective randomized controlled trial. Am. J. Sports Med. 2012, 40, 1282–1288. [Google Scholar] [CrossRef]
- Kumar, A.; Kushwaha, N.S.; Kumar, D.; Singh, A.; Gupta, V.; Kumar, S. Prospective Comparison of Functional and Radiological Outcomes of Arthroscopic Anterior Cruciate Ligament Reconstruction by Hamstring Graft Alone and Platelet-Rich Plasma Added to the Hamstring Graft. Cureus 2022, 14, e23017. [Google Scholar] [CrossRef]
- Lin, Y.C.; Chen, Y.J.; Fan, T.Y.; Chou, P.H.; Lu, C.C. Effect of bone marrow aspiration concentrate and platelet-rich plasma combination in anterior cruciate ligament reconstruction: A randomized, prospective, double-blinded study. J. Orthop. Surg. Res. 2024, 19, 4. [Google Scholar] [CrossRef]
- Mahdi, M.M.; Jhale, H.H. Value of platelet-rich plasma in osteointegration of tendon graft in anterior cruciate ligament reconstruction. Res. J. Med. Sci. 2019, 13, 76–81. [Google Scholar] [CrossRef]
- Mirzatolooei, F.; Alamdari, M.T.; Khalkhali, H.R. The impact of platelet-rich plasma on the prevention of tunnel widening in anterior cruciate ligament reconstruction using quadrupled autologous hamstring tendon: A randomised clinical trial. Bone Jt. J. 2013, 95, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Munde, K.; Banerjee, S.; Gahlot, N.; Elhence, A.; Mandal, S.; Yadav, T. Effect of platelet-rich plasma on healing of autologous graft after anterior cruciate ligament reconstruction: A randomized control trial. Regen. Med. 2023, 18, 601–610. [Google Scholar] [CrossRef]
- Nin, J.R.; Gasque, G.M.; Azcárate, A.V.; Beola, J.D.; Gonzalez, M.H. Has platelet-rich plasma any role in anterior cruciate ligament allograft healing? Arthroscopy 2009, 25, 1206–1213. [Google Scholar]
- Orrego, M.; Larrain, C.; Rosales, J.; Valenzuela, L.; Matas, J.; Durruty, J.; Sudy, H.; Mardones, R. Effects of Platelet Concentrate and a Bone Plug on the Healing of Hamstring Tendons in a Bone Tunnel. Arthrosc. J. Arthrosc. Relat. Surg. 2008, 24, 1373–1380. [Google Scholar] [CrossRef] [PubMed]
- Rupreht, M.; Jevtič, V.; Serša, I.; Vogrin, M.; Jevšek, M. Evaluation of the tibial tunnel after intraoperatively administered platelet-rich plasma gel during anterior cruciate ligament reconstruction using diffusion weighted and dynamic contrast-enhanced MRI. J. Magn. Reson. Imaging 2013, 37, 928–935. [Google Scholar]
- Rupreht, M.; Vogrin, M.; Hussein, M. MRI evaluation of tibial tunnel wall cortical bone formation after platelet-rich plasma applied during anterior cruciate ligament reconstruction. Radiol. Oncol. 2013, 47, 119–124. [Google Scholar] [CrossRef]
- Seijas, R.; Ares, O.; Catala, J.; Alvarez-Diaz, P.; Cusco, X.; Cugat, R. Magnetic resonance imaging evaluation of patellar tendon graft remodelling after anterior cruciate ligament reconstruction with or without platelet-rich plasma. J. Orthop. Surg. 2013, 21, 10–14. [Google Scholar] [CrossRef] [PubMed]
- Seijas, R.; Cuscó, X.; Sallent, A.; Serra, I.; Ares, O.; Cugat, R. Pain in donor site after BTB-ACL reconstruction with PRGF: A randomized trial. Arch. Orthop. Trauma Surg. 2016, 136, 829–835. [Google Scholar] [CrossRef] [PubMed]
- Seijas, R.; Rius, M.; Ares, O.; García-Balletbó, M.; Serra, I.; Cugat, R. Healing of donor site in bone-tendon-bone ACL reconstruction accelerated with plasma rich in growth factors: A randomized clinical trial. Knee Surg. Sports Traumatol. Arthrosc. 2015, 23, 991–997. [Google Scholar] [CrossRef]
- Silva, A.; Sampaio, R. Anatomic ACL reconstruction: Does the platelet-rich plasma accelerate tendon healing? Knee Surg. Sports Traumatol. Arthrosc. 2009, 17, 676–682. [Google Scholar] [CrossRef]
- Starantzis, K.A.; Mastrokalos, D.; Koulalis, D.; Papakonstantinou, O.; Soucacos, P.N.; Papagelopoulos, P.J. The Potentially Positive Role of PRPs in Preventing Femoral Tunnel Widening in ACL Reconstruction Surgery Using Hamstrings: A Clinical Study in 51 Patients. J. Sports Med. 2014, 2014, 789317. [Google Scholar] [CrossRef] [PubMed]
- Vadalà, A.; Iorio, R.; De Carli, A.; Ferretti, M.; Paravani, D.; Caperna, L.; Iorio, C.; Gatti, A.; Ferretti, A. Platelet-rich plasma: Does it help reduce tunnel widening after ACL reconstruction? Knee Surg. Sports Traumatol. Arthrosc. 2013, 21, 824–829. [Google Scholar] [CrossRef]
- Ventura, A.; Terzaghi, C.; Borgo, E.; Verdoia, C.; Gallazzi, M.; Failoni, S. Use of growth factors in ACL surgery: Preliminary study. J. Orthop. Traumatol. 2005, 6, 76–79. [Google Scholar] [CrossRef]
- Vogrin, M.; Rupreht, M.; Crnjac, A.; Dinevski, D.; Krajnc, Z.; Recnik, G. The effect of platelet-derived growth factors on knee stability after anterior cruciate ligament reconstruction: A prospective randomized clinical study. Wien. Klin. Wochenschr. 2010, 122, 91–95. [Google Scholar] [CrossRef]
- Vogrin, M.; Rupreht, M.; Dinevski, D.; Hašpl, M.; Kuhta, M.; Jevsek, M.; Knežević, M.; Rožman, P. Effects of a platelet gel on early graft revascularization after anterior cruciate ligament reconstruction: A prospective, randomized, double-blind, clinical trial. Eur. Surg. Res. 2010, 45, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Walters, B.L.; Hobart, S.; Porter, D.; Hogan, D.E.; McHugh, M.P.; Bedford, B.B.; Klein, D.A.; Harousseau, K.; Nicholas, S.J. Effect of intraoperative platelet-rich-plasma treatment on post operative donor site knee pain in patellar tendon autograft ACL reconstruction: A double-blind randomized controlled trial. Orthop. J. Sports Med. 2016, 46, 1877–1883. [Google Scholar] [CrossRef]
- Ye, Z.; Chen, H.; Qiao, Y.; Wu, C.; Cho, E.; Wu, X.; Wu, J.; Lu, S.; Xie, G.; Dong, S.; et al. Intra-Articular Platelet-Rich Plasma Injection After Anterior Cruciate Ligament Reconstruction: A Randomized Clinical Trial. JAMA Netw. Open 2024, 7, e2410134. [Google Scholar] [CrossRef]
- Pavan, D.; Morello, F.; Monachino, F.; Rovere, G.; Camarda, L.; Pitarresi, G. Similar biomechanical properties of four tripled tendon graft models for ACL reconstruction. Arch. Orthop. Trauma Surg. 2022, 142, 1155–1165. [Google Scholar] [CrossRef]
- Rovere, G.; Stramazzo, L.; Romeo, M.; D’Arienzo, A.; Maccauro, G.; Camarda, L. Hamstring Graft Preparation for ACL Reconstruction. Orthop. Rev. 2022, 14, 38408. [Google Scholar] [CrossRef]
- de Andrade, A.L.L.; Sardeli, A.V.; Garcia, T.A.; Livani, B.; Belangero, W.D. Time-Dependent Effect of Platelet-Rich Plasma in Reducing Donor-Site Pain After Anterior Cruciate Ligament Reconstruction. Am. J. Sports Med. 2021, 49, 2854–2858. [Google Scholar] [CrossRef]
- Lv, Z.T.; Zhang, J.M.; Pang, Z.Y.; Wang, Z.; Huang, J.M.; Zhu, W.T. The efficacy of platelet rich plasma on anterior cruciate ligament reconstruction: A systematic review and meta-analysis. Platelets 2022, 33, 229–241. [Google Scholar] [CrossRef]
- de Sa, D.; Shanmugaraj, A.; Weidman, M.; Peterson, D.C.; Simunovic, N.; Musahl, V.; Ayeni, O.R. All-Inside Anterior Cruciate Ligament Reconstruction-A Systematic Review of Techniques, Outcomes, and Complications. J. Knee Surg. 2018, 31, 895–904. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.J.; Kelley, B.V.; Arshi, A.; McAllister, D.R.; Fabricant, P.D. Comparative Effectiveness of Cartilage Repair with Respect to the Minimal Clinically Important Difference. Am. J. Sports Med. 2019, 47, 3284–3293. [Google Scholar] [CrossRef]
- Molloy, T.; Wang, Y.; Murrell, G. The roles of growth factors in tendon and ligament healing. Sports Med. 2003, 33, 381–394. [Google Scholar] [CrossRef]
- Xue, Y.; Su, X.; Jiang, M.; Yu, Z.; Yang, H.; Qin, L.; Giannoudis, P.V.; Guo, J.J. Pure platelet-rich plasma facilitates the repair of damaged cartilage and synovium in a rabbit hemorrhagic arthritis knee model. Arthritis Res. Ther. 2020, 22, 68. [Google Scholar] [CrossRef]
- Xu, J.; Chen, X.; Zhang, H.; Zhang, X.; Liu, R.; Li, X.; Wang, J.; Li, T. Platelet-rich plasma relieves inflammation and pain by regulating M1/M2 macrophage polarization in knee osteoarthritis rats. Sci. Rep. 2025, 15, 12805. [Google Scholar] [CrossRef] [PubMed]
- McRobb, J.; Kamil, K.H.; Ahmed, I.; Dhaif, F.; Metcalfe, A. Influence of platelet-rich plasma (PRP) analogues on healing and clinical outcomes following anterior cruciate ligament (ACL) reconstructive surgery: A systematic review. Eur. J. Orthop. Surg. Traumatol. 2023, 33, 225–253. [Google Scholar] [CrossRef]
- de Mos, M.; van der Windt, A.E.; Jahr, H.; van Schie, H.T.; Weinans, H.; Verhaar, J.A.; van Osch, G.J. Can platelet-rich plasma enhance tendon repair? A cell culture study. Am. J. Sports Med. 2008, 36, 1171–1178. [Google Scholar] [CrossRef]
- Delcogliano, M.; Sangiorgio, A.; Bensa, A.; Andriolo, L.; Boffa, A.; Filardo, G. Platelet-rich plasma augmentation in anterior cruciate ligament reconstruction: Evidence is still too scattered. A scoping review of randomised controlled trials. Knee Surg. Sports Traumatol. Arthrosc. 2024, 32, 1143–1159. [Google Scholar] [CrossRef] [PubMed]
- Rovere, G.; Bosco, F.; Miceli, A.; Ratano, S.; Freddo, G.; D’Itri, L.; Ferruzza, M.; Maccauro, G.; Farsetti, P.; Camarda, L. Adoption of blockchain as a step forward in orthopedic practice. Eur. J. Transl. Myol. 2024, 34, 12197. [Google Scholar] [CrossRef] [PubMed]





| First Author-Year | Study Design-LOE | Sample Size | Age (Years) | Sex (Male /Female) | Follow-Up (Months) | Drop Out | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| PRP | No PRP | PRP | No PRP | PRP | No PRP | PRP | No PRP | |||
| Ventura-2005 | RCT-I | 10 | 10 | 36.6 ± 9.3 | 30.2 ± 5.3 | 9/1 | 9/1 | 6 | 0 | 0 |
| Orrego-2008 | RCT-II | 29 | 29 | 30 (15–57) | 85% | 6 | 3 | 2 | ||
| Nin-2009 | RCT-I | 50 | 50 | 26.1 (14–57) | 26.6 (15–59) | 40/10 | 38/12 | 6 | 0 | 0 |
| Silva-2009 | RCT-II | 30 | 10 | 26.8 ± 5.3 | 38/2 | 3 | 0 | 0 | ||
| Vogrin-2010 | RCT-I | 25 | 25 | 37.2 ± 8.4 | 32.6 ± 12.3 | 15/10 | 16/9 | 3 | 4 | 5 |
| Vogrin-2010 | RCT-I | 25 | 25 | 35.4 ± 10.0 | 33.0 ± 12.5 | 15/10 | 16/9 | 6 | 3 | 2 |
| Cervellin-2012 | RCT-I | 20 | 20 | 22.9 ± 4.3 | 22.7 ± 3.5 | 20/0 | 20/0 | 12 | 2 | 3 |
| de Almeida-2012 | RCT-I | 12 | 15 | 25.8 (18–44) | 23.1 (15–34) | 10/2 | 14/1 | 6 | 2 | 2 |
| Mirzatolooei-2013 | RCT-I | 25 | 25 | 26.4 (18–40) | 26.9 (18–40) | 20/3 | 22/1 | 3 | 4 | 5 |
| Rupreht-2013 | RCT-I | 25 | 25 | 37.2 ± 8.4 | 32.6 ± 12.3 | 15/10 | 16/9 | 6 | 4 | 5 |
| Rupreht-2013 | RCT-I | 25 | 25 | 37.2 ± 8.4 | 32.6 ± 12.3 | 15/10 | 16/9 | 6 | 2 | 2 |
| Seijas-2013 | RCT-I | 50 | 50 | NA | NA | NA | NA | 12 | 1 | 1 |
| Vadalà-2013 | RCT-II | 20 | 20 | 34.5 (18–48) | 20/0 | 20/0 | 14.7 | 0 | 0 | |
| Starantzis-2014 | RCT-I | 30 | 30 | 29.4 ± 7.3 | 31.3 ± 8.0 | 38/13 | 12 | 5 | 4 | |
| Seijas-2015 | RCT-I | 23 | 21 | NA | NA | 20/3 | 17/3 | 12 | 0 | 1 |
| Seijas-2016 | RCT-I | 23 | 21 | NA | NA | 20/3 | 17/3 | 24 | 0 | 1 |
| Walters-2018 | RCT-II | 30 | 29 | 30 ± 12 | 10/17 | 12/11 | 24 | 7 | 8 | |
| Mahdi-2019 | RCT-I | 14 | 13 | 25.77 | 25.77 | 14/0 | 13/0 | 3 | 0 | 0 |
| Gong-2022 | RCT-I | 30 | 30 | 33.5 ± 8.97 | 34.9 ± 9.68 | 18/12 | 21/9 | 12 | 3 | 4 |
| Kumar-2022 | RCT-I | 35 | 35 | 28.34 ± 4.32 | 29.71 ± 2.99 | 22/13 | 23/12 | 3 | 0 | 0 |
| Munde-2023 | RCT-I | 44 | 43 | 28.37 ± 2.8 | 27.4 ± 7.0 | 36/4 | 38/2 | 6 | 4 | 3 |
| Lin-2024 | RCT-I | 10 | 10 | 28.4 ± 7.8 | 29.7 ± 9.9 | 6/2 | 6/4 | 12 | 2 | 0 |
| Ye-2024 | RCT-I | 60 | 60 | 28.0 ± 7.9 | 30.0 ± 8.0 | 43/17 | 41/19 | 12 | 3 | 3 |
| Author-Year | Graft Source | Graft Type | Fixation Method | Rehabilitation Protocol |
|---|---|---|---|---|
| Ventura-2005 | Autograft | Quadrupled hamstring tendon | Transcondylic fixation (femoral side) and interference screw (tibial side) | Immediate post-operative mobilization without a knee brace, protected weight bearing for 3 weeks, and return to sporting activities at 6 months. |
| Orrego-2008 | Autograft | Quadrupled hamstring tendon | Biodegradable transfixing pin (femoral side) and biodegradable interference screw (tibial side) | NA |
| Nin-2009 | Allograft | Patellar tendon | 2 biodegradable cross pins (femoral side) and biodegradable interference screw (tibial side) | Immediate post-operative immobilized with a knee brace, whole range of movement after 10 days, Cycling at 2 to 3 months, straight-line running, at 4 months, and sports at 6 months. |
| Silva-2009 | Autograft | Double-bundle hamstring tendon | EndoButton CL devices (femoral side) and bioabsorbable interference screw (tibial side) | Immobilization of the knee with a brace in full extension for a week after surgery, after which mobilization is started, below 90 of flexion until the fourth week and then, increasing the flexion of the knee 15 every week. Protection from weight bearing is used for 5 weeks after surgery. |
| Vogrin-2010 | Autograft | Double-looped hamstring tendon | 2 bioabsorbable cross pins (femoral side) and bioabsorbable interference screw (tibial side) | No rehabilitation brace was used postoperatively. Running at 12 weeks and contact sports at 6 months provided that the patient had no knee joint effusion, achieved a full range of motion and obtained muscle strength of at least 90% compared to the contralateral leg. |
| Vogrin-2010 | Autograft | Double-looped hamstring tendon | 2 bioabsorbable cross pins (femoral side) and bioabsorbable interference screw (tibial side) | No rehabilitation brace was used postoperatively. Running was allowed at 12 weeks and contact sports at 6 months in cases with no knee-joint effusion, full range of motion and obtained muscle strength of 90% compared with the contralateral leg. |
| Cervellin-2012 | Autograft | Bone-patellar tendon-bone | NA | Weight bearing with the knee extended for the first 15 days, followed by partial weight bearing for the next 15 days. walking without the aid of crutches during the second months. During the third and fourth months, closed kinetic chain exercises and gradual return to sports activity were allowed. |
| de Almeida-2012 | Autograft | Bone-patellar tendon-bone | A transverse double pin absorbable system (femoral side) and an absorbable interference screw (tibial side). | Early range of motion and progressive weight-bearing with crutches for 3 weeks. |
| Mirzatolooei-2013 | Autograft | Quadrupled hamstring tendon | A cross-pin (femoral side) and bio-absorbable interference screw (tibial side) | Wore a knee immobilizer in full extension for two weeks. |
| Rupreht-2013 | Autograft | Double-looped hamstring tendon | 2 bioabsorbable cross pins (femoral side) and one bioabsorbable interference screw (tibial side) | NA |
| Rupreht-2013 | Autograft | Double-looped hamstring tendon | 2 bioabsorbable cross pins (femoral side) and one bioabsorbable interference screw (tibial side) | NA |
| Seijas-2013 | Autograft | Bone-patellar tendon-bone | Hydroxylapatite screws (both femoral and tibia sides) | Postoperatively, the knee was immobilized with 2 plaster splints. At week 4, progressive weight-bearing ambulation was allowed as pain tolerated. Pool exercises were started at week 6, outdoor cycling at month 3, progressive running at month 4, and return to unrestricted sporting activities at month 6. |
| Vadalà-2013 | Autograft | Hamstring tendon | Swing-Bridge device (femoral side) and the Evolgate (tibial side) | Post-operatively, all patients started weight-bearing with the use of crutches the day after the operation. Within the first 6 weeks, patients started progressive isotonic and isokinetic exercises. Patients involved in sports activities were allowed to return to practice their sport 6 months after surgery, and patients involved in noncontact sports after 4 months. |
| Starantzis-2014 | Autograft | Quadrupled hamstring tendon | Crosspin or Endobutton (femoral side) and biodegradable interference screw + bone bridge suture anchoring (tibial side) | NA |
| Seijas-2015 | Autograft | Bone-patellar tendon-bone | NA | NA |
| Seijas-2016 | Autograft | Bone-patellar tendon-bone | NA | NA |
| Walters-2018 | Autograft | Bone-patellar tendon-bone | Two titanium cannulated interference screws (both femoral and tibial side) | NA |
| Mahdi-2019 | Autograft | Quadrupled-strand hamstring tendon | “Suspensory” fixation mechanism (Endobutton) in both sides. | NA |
| Gong-2022 | Autograft | Quadrupled-strand hamstring tendon | Endobutton (femoral side) and Intrafx Device (tibial side) | Partial weight-bearing was permitted with the brace locked in full extension within 4 weeks after operation. Full weight-bearing with the brace was permitted at the seventh week after operation; full weight-bearing without the brace was permitted at the third month after operation. Walking, running and contact sports were permitted after 3, 6 and 9 months, respectively. |
| Kumar-2022 | Autograft | Hamstring tendon | Endobutton (femoral side) and biodegradable interference screw (tibial side) | Standard ACL rehabilitation protocol which included gaining a range of motion, muscle strength and returning to preinjury status in phasic manner. |
| Munde-2022 | Autograft | Quadrupled hamstring tendon | Adjustable loop button (femoral side) and biodegradable interference screw (tibial side) | In the first phase (up to 2 weeks) all patients were started on full weight-bearing walking with crutches, isometric quadriceps exercises, etc. In the second phase (2–4 weeks), quadricep strengthening exercises, knee ROM exercises, and balance exercises started. In the third and fourth phase (1–6 months), resistance band quadriceps, hamstring strengthening exercises, jogging, controlled single-leg jumping, plyometric exercises, etc. were done. |
| Lin-2024 | Autograft | Fourth or fifth-strand hamstring tendon | Interference screws (femoral side) and Interference screws + cancellous post-screw (tibial side) | Every subject underwent the program at 1, 3, 5, 7, and 9 weeks post-surgery. |
| Ye-2024 | Autograft | Quadrupled-strand hamstring tendon | Cortical buttons (femoral side) and biodegradable interference screw + cortical button with an adjustable loop (tibial side) | Standardized rehabilitation education from a physical therapist through verbal instruction, printed brochures, and online videos before discharge. |
| Author-Year | Processing Machine | Whole Blood Volume (mL) | Anticoagulants | Spin Speed (rpm) | Spin Time (min) | Platelet Concentration (mm3) | Activation | Form | Leukocyte | Location | Time Point | Volume (mL) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ventura-2005 | GPS Biomet Merck technique (Biomet Inc., Warsaw, IN, USA) | 54 | Citric acid | 3200 | 12 | NA | Autologous thrombin | gel | rich | Femoral and tibial tunnels | Intraoperative | NA |
| Orrego-2008 | Biomet GPS II kit (Biomet, Warsaw, IN, USA) | 57 | NA | 200 | 15 | NA | Autologous thrombin with CaCl2 | gel + liqiud | rich | Between the strands of the graft and femoral tunnel | Intraoperative | 6 |
| Nin-2009 | Beckman J-6B, Beckman Coulter Spain, Madrid, Spain | 40 | Citric acid | 1st: 3000 2nd: 1000 | 1st: 8 2nd: 6 | 837,000 | CaCl2 | gel | poor | Graft was covered with gel and inside the tibial tunnel | Intraoperative | 4 |
| Silva-2009 | Mini GPS III Kit (Biomet) | 27 | Citric acid | 3200 | 15 | NA | Autologous thrombin | gel + liqiud | rich | Between the strands of the graft and inside the femoral tunnel | Intraoperative | 3 |
| Vogrin-2010 | Magellan (Medtronic Biologic Therapeutics and Diagnostics, Minneapolis, MN, USA) autologous platelet separator | 50 | 10% calcium citrate | NA | NA | 978 × 109/L (range 552 to 1326 × 109/L) | Autologous thrombin | gel | rich | Between the strands of the graft and inside the femoral and tibial tunnels. | Intraoperative | 6 |
| Vogrin-2010 | Magellan autologous platelet separator (Medtronic Biologic Therapeutics and Diagnostics, Minneapolis, MN, USA) | 50 | 10% calcium citrate | NA | NA | 962 (552–1326) G/L | Autologous thrombin | gel | rich | Between the strands of the graft and inside the femoral and tibial tunnels. | Intraoperative | 6 |
| Cervellin-2012 | Gravitational Platelet Separation II (GPS) system (Biomet Biologics, Inc., Warsaw, IN, USA) | 54 | ACD-A | 3200 | 15 | NA | Autologous thrombin with CaCl2 | gel | rich | Patellar and tendon bone plug harvest site | Intraoperative | NA |
| de Almeida-2012 | A Haemonetics MCS1 9000 cell separator with a specific kit for platelet apheresis 995-E (Haemonetics Corp., Braintree, MA, USA) | 450 | 10% citrate | NA | NA | 1,185,166 ± 404,472 | Autologous thrombin with CaCl2 | gel | poor | Patellar tendon defect | NA | 20–40 |
| Mirzatolooei-2013 | Double syringe system (Arthrex, Naples, FL, USA) | 10 | NA | 1500 | 5 | NA | CaCl2 | liquid | poor | Between the strands of the graft and inside the femoral and tibial tunnels. | Intraoperative | 3.5 |
| Rupreht-2013 | Magellan autologous platelet separator (Medtronic Biologic Therapeutics and Diagnostics) | 50 | NA | NA | NA | NA | Autologous thrombin | gel | rich | Between the strands of the graft and inside the femoral and tibial tunnels. | Intraoperative | 6 |
| Rupreht-2013 | Magellan autologous platelet separator (Medtronic Biologic Therapeutics and Diagnostics) | 50 | NA | NA | NA | 978 × 109/L (range 552 to 1326 × 109/L) | Autologous thrombin | gel | rich | Between the strands of the graft and inside the femoral and tibial tunnels. | Intraoperative | 6 |
| Seijas-2013 | PRGF technique (BTI Systems Vitoria, Spain) | NA | NA | NA | NA | NA | No activation | liquid | NA | Suprapatellar joint after portal suture | Intraoperative | 8 |
| Vadalà-2013 | PRP Fast Biotech kit (MyCells PPT-Platelet Preparation Tube) | 10 | NA | NA | NA | NA | Autologous thrombin with Ca-gluconate | liquid + gel | NA | Between the strands of the graft and inside the femoral and tibial tunnels. | Intraoperative | 15 |
| Starantzis-2014 | Biomet GPS III kit (Biomet, Warsaw, IN, USA) | 55 | ACD-A | 3200 | 15 | NA | CaCl2. | liquid + gel | rich | Between the strands of the graft and inside the femoral tunnel. | Intraoperative | 6 |
| Seijas-2015 | NA | 20cc | NA | NA | NA | NA | NA | NA | NA | Patellar bone gap, tibial bone gap, and the center of harvest gap. | Intraoperative | 4 |
| Seijas-2016 | NA | 34cc | Sodium citrate | 1800 | 8 | NA | CaCl2 | gel | NA | Patellar bone gap, tibial bone gap, and the center of harvest gap. | Intraoperative | 4 |
| Walters-2018 | A PRP separation kit and centrifuge system (ACP PRP; Arthrex) | 10 | ACD-A | 1500 | 5 | NA | CaCl2 | gel | poor | Patellar donor site | NA | 3–5 |
| Mahdi-2019 | Trima Accel Automated Blood Collection system | 100–150 | NA | NA | NA | 5–7 × 107 | No activation | liquid | NA | Inside femoral tunnel and intra-articularly | Intraoperative | 6 |
| Gong-2022 | Platelet Rich Plasma Preparation Kits (WEGO; Beijing, China) and a centrifuge (WEGO; Beijing, China) | 36 | ACD-A | NA | NA | NA | No activation | liquid | NA | Inside the bone tunnels and graft | Intraoperative | 4 |
| Kumar-2022 | NA | NA | NA | NA | NA | NA | No activation | liquid | NA | Between the strands of the graft and inside the femoral and tibial tunnels. | Intraoperative | NA |
| Munde-2023 | Remi R8 C fixed angle microcentrifuge | 17 | ACD-A | 1st: 1500 2nd: 2500 | 1st: 15 2nd: 10 | NA | platelet agitator | gel | NA | Inside the femoral tunnel | Intraoperative | 3–4 |
| Lin-2024 | NA | 30 | NA | 3200 | 6 | NA | NA | gel | NA | At each end of the graft (bone tunnel side) and into the knee joint. | Intraoperative | NA |
| Ye-2024 | commercially used system (Platelet-Rich Plasma Preparation Kit; WEGO Ltd.) | 45 | ACD-A | NA | 1st: 10 2nd: 15 | 678 ± 173 × 109/L | No activation | liquid | poor | Intra-articular injection | 4 weeks, 8 weeks and 3 months postoperatively | 5 |
| Author-Year | Selection Bias | Performance Bias | Detection Bias | Attrition Bias | Reporting Bias | Other Bias | |
|---|---|---|---|---|---|---|---|
| Random Sequence Generation | Allocation Concealment | Blinding of Participants and Personnel | Blinding of Outcome Assessment | Incomplete Outcome Data | Selective Reporting | ||
| Ventura-2005 | high | high | high | high | high | low | high |
| Orrego-2008 | low | unclear | low | low | low | low | low |
| Nin-2009 | low | low | low | low | low | low | unclear |
| Silva-2009 | unclear | unclear | low | unclear | low | low | unclear |
| Vogrin-2010 | low | low | low | low | low | low | unclear |
| Cervellin-2012 | low | low | low | low | low | low | low |
| de Almeida-2012 | low | low | low | low | low | low | unclear |
| Mirzatolooei-2013 | low | low | unclear | unclear | low | low | unclear |
| Rupreht-2013 | unclear | unclear | low | low | low | low | unclear |
| Rupreht-2013 | low | low | low | low | low | low | unclear |
| Seijas-2013 | unclear | unclear | unclear | low | low | low | unclear |
| Vadalà-2013 | unclear | unclear | unclear | low | low | low | unclear |
| Starantzis-2014 | low | unclear | low | low | low | low | low |
| Seijas-2015 | low | low | low | low | low | low | unclear |
| Seijas-2016 | low | low | low | low | low | low | unclear |
| Walters-2018 | low | low | low | low | low | low | low |
| Mahdi-2019 | unclear | unclear | unclear | unclear | low | low | unclear |
| Gong-2022 | low | unclear | unclear | low | low | low | low |
| Kumar-2022 | low | unclear | unclear | unclear | low | low | low |
| Munde-2023 | low | low | unclear | low | low | low | low |
| Lin-2024 | low | unclear | low | low | low | low | low |
| Ye-2024 | low | low | low | low | low | low | low |
| Outcomes | Author-Year | Grade | PRP Group | Non-PRP Group | p-Value | Follow-Up |
|---|---|---|---|---|---|---|
| Anterior drawer test | Mahdai-2019 | <5/5–10/>10 | 12/2/0 | 1/10/2 | <0.0001 | 1 month |
| Ventura-2005 | negative/positive | 9/1 | 10/0 | NS | 6 months | |
| Kumar-2022 | 1/2/3 | 33/2/0 | 31/4/0 | 0.303 | 3 months | |
| Munde-2023 | negative/positive | 39/1 | 33/7 | NS | 6 months | |
| Ye-2024 | 0/1/2/3 | 54/3/0/0 | 55/2/0/0 | 0.65 | 12 months | |
| Lachman test | Mahdai-2019 | <5/6–10 | 12/2 | 6/7 | 0.033 | 1 month |
| Ventura-2005 | negative/positive | 9/1 | 9/1 | NS | 6 months | |
| Munde-2023 | negative/positive | 39/1 | 33/7 | NS | 6 months | |
| Mirzatolooei-2013 | negative/positive | 25/0 | 25/0 | NS | 3 months | |
| Vadála-2013 | negative/positive | 20/0 | 20/0 | NS | 14.7 months | |
| Kumar-2022 | 1/2/3 | 33/2/0 | 31/4/0 | 0.808 | 3 months | |
| Ye-2024 | 0/1/2/3 | 54/0/3/0 | 55/0/0/2 | 0.68 | 12 months | |
| Pivot shift test | Starantzis-2014 | negative/positive | 30/0 | 30/0 | NS | 12 months |
| Mahdai-2019 | absent/pivot glide | 12/2 | 5/8 | 0.014 | 1 month | |
| Vadála-2013 | (neg, +1)/(neg, +1) | 16/4 | 18/2 | NS | 14.7 months | |
| Ye-2024 | 0/1/2/3 | 54/2/1/0 | 55/0/1/1 | 0.68 | 12 months | |
| Objective IKDC | Nin-2009 | A/B/C/D | 35/15/0/0 | 35/13/2/0 | NS | 24.3 months |
| Orrego-2008 | (A + B)/(C + D) | 24/2 | 26/1 | NS | 6 months | |
| Vadála-2013 | A/B/C/D | 16/4/0/0 | 16/4/0/0 | NS | 14.7 months |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Ibrahim, A.A.; Opoku, M.; Mahamat Abdramane, A.; Fang, M.; Liu, X.; Mustapha, A.; Li, Y.; Xiao, W.; Zhang, K.; Liu, S. Role of Platelet-Rich Plasma Injection in Anterior Cruciate Ligament Reconstruction: A Meta-Analysis of Randomized Controlled Trials. Bioengineering 2026, 13, 455. https://doi.org/10.3390/bioengineering13040455
Ibrahim AA, Opoku M, Mahamat Abdramane A, Fang M, Liu X, Mustapha A, Li Y, Xiao W, Zhang K, Liu S. Role of Platelet-Rich Plasma Injection in Anterior Cruciate Ligament Reconstruction: A Meta-Analysis of Randomized Controlled Trials. Bioengineering. 2026; 13(4):455. https://doi.org/10.3390/bioengineering13040455
Chicago/Turabian StyleIbrahim, Ahmed Abdirahman, Michael Opoku, Abakar Mahamat Abdramane, Mingqing Fang, Xu Liu, Abdulraheem Mustapha, Yusheng Li, Wenfeng Xiao, Kai Zhang, and Shuguang Liu. 2026. "Role of Platelet-Rich Plasma Injection in Anterior Cruciate Ligament Reconstruction: A Meta-Analysis of Randomized Controlled Trials" Bioengineering 13, no. 4: 455. https://doi.org/10.3390/bioengineering13040455
APA StyleIbrahim, A. A., Opoku, M., Mahamat Abdramane, A., Fang, M., Liu, X., Mustapha, A., Li, Y., Xiao, W., Zhang, K., & Liu, S. (2026). Role of Platelet-Rich Plasma Injection in Anterior Cruciate Ligament Reconstruction: A Meta-Analysis of Randomized Controlled Trials. Bioengineering, 13(4), 455. https://doi.org/10.3390/bioengineering13040455







