Simultaneous Determination of Glucose and Cholesterol in Milk Samples by Means of a Screen-Printed Biosensor and Artificial Neural Networks
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Instrumentation
2.2. Construction of the Bienzymatic Biosensor
2.3. Multivariate Determination
2.4. Sample Analysis
2.5. Analysis of Flavored Milk Samples with the Reference Method
3. Results
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| FTIR | Fourier transform infrared spectroscopy |
| PLS | Partial least squares regression |
| FT-Raman | Fourier transform Raman spectroscopy |
| ANN | Artificial neural network |
| SPR | Surface plasmon resonance |
| PANI | Polyaniline |
| GOx | Glucose oxidase |
| ChOx | Cholesterol oxidase |
| POD | Peroxidase |
| 4-AP | 4-aminoantipyrine |
| ChE | Cholesterol esterase |
| SPCE | Screen-printed carbon electrode |
| FAD | Flavin Adenine Dinucleotide (oxided form) |
| FADH2 | Flavin Adenine Dinucleotide (reduced form) |
| CCD | Central composite design |
| RMSE | Root mean square error |
| MSE | Mean squared error |
| RE% | Relative error percentage (), |
| REP% | Prediction relative error percentage |
References
- Wiegers, C.; Van de Burgwal, L.H.M.; Claassen, E.; Larsen, O.F.A. Trends in Nutrition, Lifestyle, and Metabolic Disease in the United States. PharmaNutrition 2023, 25, 100350. [Google Scholar] [CrossRef]
- Madhurantakam, S.; Devi, K.S.S.; Babu, J.K.; Rayappan, J.B.B.; Krishnan, U.M. Metabolic Syndrome—An Emerging Constellation of Risk Factors: Electrochemical Detection Strategies. Sensors 2020, 20, 103. [Google Scholar] [CrossRef]
- Guan, G.; Qu, L.; Zhao, Y.; Wang, P.; Kong, F.; Zhang, Y.; Lin, Z.; Ni, X.; Zhang, X.; Lu, Q.; et al. Recent Advances in Glucose Monitoring Utilizing Oxidase Electrochemical Biosensors Integrating Carbon-Based Nanomaterials and Smart Enzyme Design. Front. Chem. 2025, 13, 1591302. [Google Scholar] [CrossRef]
- World Health Organization. The Top 10 Causes of Death; World Health Organization: Geneva, Switzerland, 2024; Available online: https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death (accessed on 2 July 2025).
- Scarcelli, E.; Iacopetta, D.; Ceramella, J.; Bonofiglio, D.; Conforti, F.L.; Aiello, F.; Sinicropi, M.S. The Role of Functional Beverages in Mitigating Cardiovascular Disease Risk Factors: A Focus on Their Antidiabetic and Hypolipidemic Properties. Beverages 2025, 11, 21. [Google Scholar] [CrossRef]
- Amiri, M.; Arshia, S. An Overview on Electrochemical Determination of Cholesterol. Electroanalysis 2020, 32, 1391–1407. [Google Scholar] [CrossRef]
- Galant, A.L.; Kaufman, R.C.; Wilson, J.D. Glucose: Detection and Analysis. Food Chem. 2015, 188, 149–160. [Google Scholar] [CrossRef] [PubMed]
- Jalalvand, A.R. An Intelligent and Novel Electrochemical Biosensor For Simultaneous Enzymatic Biosensing of Cholesterol and Glucose in the Presence of Uric Acid Based on First-And Second-Order Calibration Methods. Microchem. J. 2023, 191, 108824. [Google Scholar] [CrossRef]
- Hu, T.; Zhang, M.; Dong, H.; Li, T.; Zang, X.; Li, X.; Ni, Z. Free-Standing Mxene/Chitosan/Cu2O Electrode: An Enzyme-Free and Efficient Biosensor for Simultaneous Determination of Glucose and Cholesterol. J. Zhejiang Univ.-Sci. A 2022, 23, 579–586. [Google Scholar] [CrossRef]
- Jessen, T.E.; Höskuldsson, A.T.; Bjerrum, P.J.; Verder, H.; Sørensen, L.; Bratholm, P.S.; Christensen, B.; Jensen, L.S.; Jensen, M.A.B. Simultaneous Determination of Glucose, Triglycerides, Urea, Cholesterol, Albumin and Total Protein In Human Plasma by Fourier Transform Infrared Spectroscopy: Direct Clinical Biochemistry Without Reagents. Clin. Biochem. 2014, 47, 1306–1312. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Lv, H.; Li, T.; Si, G.; Wang, Q.; Lv, J.; Hu, X. Reagent-Free Simultaneous Determination of Glucose and Cholesterol in Whole Blood By FTIR-ATR. Spectrochim. Acta A 2017, 178, 192–197. [Google Scholar] [CrossRef]
- Wang, Q.; Wu, G.; Pian, F.; Shan, P.; Li, Z.; Ma, Z. Simultaneous Detection of Glucose, Triglycerides, and Total Cholesterol in Whole Blood by Fourier-Transform Raman Spectroscopy. Spectrochim. Acta Part A 2021, 260, 119906. [Google Scholar] [CrossRef]
- Torres-Gamez, J.; Rodriguez, J.A.; Paez-Hernandez, M.E.; Galan-Vidal, C.A. Application of Multivariate Statistical Analysis to Simultaneous Spectrophotometric Enzymatic Determination of Glucose and Cholesterol in Serum Samples. Int. J. Anal. Chem. 2019, 2019, 7532687. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Zhang, Y.; Li, L.; Li, X.; Zhao, Y. A Plug-And-Play Optical Fiber SPR Sensor for Simultaneous Measurement of Glucose and Cholesterol Concentrations. Biosens. Bioelectron. 2022, 198, 113798. [Google Scholar] [CrossRef]
- Wongsing, B.; Prakobkij, A.; Anutrasakda, W.; Jarujamrus, P. Vanadium-Doped Porous Cobalt Oxide for Its Superior Peroxidase-Like Activity in Simultaneous Total Cholesterol and Glucose Determination in Whole Blood Based on A Simple Two-Dimensional Paper-Based Analytical Device. Anal. Chem. 2022, 94, 13785–13794. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Sun, X.; Hu, Y.; Zhang, L.; Zeng, L.; Liu, Q.; Duan, J. A Dual-Parameter Optical Fiber SPR Sensor for Simultaneous Measurement of Glucose and Cholesterol Concentrations. IEEE Sens. J. 2022, 22, 20413–20420. [Google Scholar] [CrossRef]
- Prakobkij, A.; Sukapanon, S.; Chunta, S.; Jarujamrus, P. Mickey Mouse-Shaped Laminated Paper-Based Analytical Device in Simultaneous Total Cholesterol and Glucose Determination in Whole Blood. Anal. Chim. Acta 2023, 1263, 341303. [Google Scholar] [CrossRef]
- Kitchawengkul, N.; Prakobkij, A.; Saenmuangchin, R.; Citterio, D.; Nacapricha, D.; Jarujamrus, P. Ratiometric Fluorometry on Microfluidic Paper-Based Analytical Device for Simultaneous Glucose and Cholesterol Detection Using MnFe-Layered Double Hydroxides as Peroxidase Mimic. Sens. Actuators B Chem. 2025, 435, 137671. [Google Scholar] [CrossRef]
- Huang, Q.; An, Y.; Tang, L.; Jiang, X.; Chen, H.; Bi, W.; Wang, Z.; Zhang, W. A dual enzymatic-biosensor for simultaneous determination of glucose and cholesterol in serum and peritoneal macrophages of diabetic mice: Evaluation of the diabetes-accelerated atherosclerosis risk. Anal. Chim. Acta 2011, 707, 135–141. [Google Scholar] [CrossRef]
- Gao, J.; Huang, W.; Chen, Z.; Yi, C.; Jiang, L. Simultaneous Detection of Glucose, Uric Acid and Cholesterol Using Flexible Microneedle Electrode Array-Based Biosensor and Multi-Channel Portable Electrochemical Analyzer. Sens. Actuators B Chem. 2019, 287, 102–110. [Google Scholar] [CrossRef]
- Jaime, J.; Rangel, G.; Muñoz-Bonilla, A.; Mayoral, A.; Herrasti, P. Magnetite as a Platform Material in the Detection of Glucose, Ethanol and Cholesterol. Sens. Actuators B Chem. 2017, 238, 693–701. [Google Scholar] [CrossRef]
- Türkarslan, Ö.; Kayahan, S.K.; Toppare, L. A New Amperometric Cholesterol Biosensor Based on Poly(3,4-Ethylenedioxypyrrole). Sens. Actuators B Chem. 2009, 136, 484–488. [Google Scholar] [CrossRef]
- Sekar, N.C.; Shaegh, S.A.M.; Ng, S.H.; Ge, L.; Tan, S.N. A Paper-Based Amperometric Glucose Biosensor Developed with Prussian Blue-Modified Screen-Printed Electrodes. Sens. Actuators B Chem. 2014, 204, 414–420. [Google Scholar] [CrossRef]
- Mamani, M.C.V.; Reyes, F.G.R.; Rath, S. Multiresidue Determination of Tetracyclines, Sulphonamides and Chloramphenicol in Bovine Milk Using HPLC-DAD. Food Chem. 2009, 117, 545–552. [Google Scholar] [CrossRef]
- Saxena, U.; Das, A.B. Nanomaterials Towards Fabrication of Cholesterol Biosensors: Key Roles and Design Approaches. Biosens. Bioelectron. 2016, 75, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Tesio, A.Y.; Robledo, S.N.; Granero, A.M.; Fernández, H.; Zon, M.A. Simultaneous Electroanalytical Determination of Luteolin and Rutin Using Artificial Neural Networks. Sens. Actuators B Chem. 2014, 203, 655–662. [Google Scholar] [CrossRef]
- Tonello, N.; Moressi, M.B.; Robledo, S.N.; D’Eramo, F.; Marioli, J.M. Square Wave Voltammetry with Multivariate Calibration Tools for Determination of Eugenol, Carvacrol and Thymol in Honey. Talanta 2016, 158, 306–314. [Google Scholar] [CrossRef] [PubMed]
- Mishra, R.K.; Alonso, G.A.; Istamboulie, G.; Bhand, S.; Mart, J.-L. Automated Flow Based Biosensor for Quantification of Binary Organophosphates Mixture in Milk Using Artificial Neural Network. Sens. Actuators B Chem. 2015, 208, 228–237. [Google Scholar] [CrossRef]
- Gutés, A.; Ibáñez, A.B.; del Valle, M.; Céspedes, F. Automated SIA e-Tongue Employing a Voltammetric Biosensor Array for the Simultaneous Determination of Glucose and Ascorbic Acid. Electroanalysis 2006, 18, 82–88. [Google Scholar] [CrossRef]




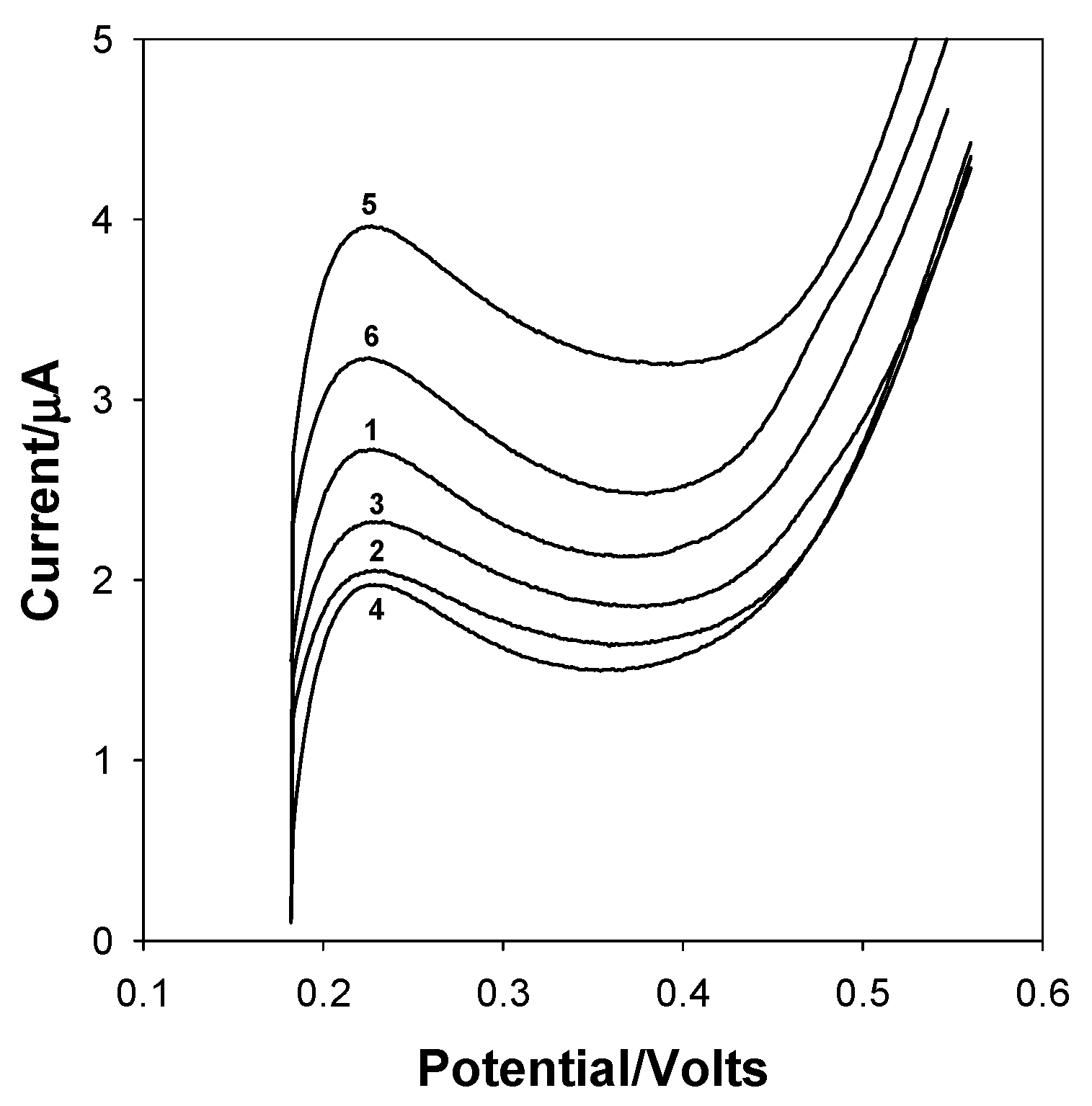
| Experiment Number | Glucose (mmol L−1) | Cholesterol (mmol L−1) |
|---|---|---|
| 1 | 0.54 | 0.30 |
| 2 | 1.00 | 0.20 |
| 3 | 1.24 | 0.23 |
| 4 | 2.00 | 0.12 |
| 5 | 2.00 | 0.33 |
| 6 | 2.00 | 0.44 |
| 7 | 2.71 | 0.23 |
| 8 | 3.00 | 0.20 |
| 9 | 3.00 | 0.40 |
| 10 | 3.41 | 0.30 |
| 11 | 1.00 | 0.40 |
| 12 | 1.50 | 0.30 |
| 13 | 1.50 | 0.15 |
| 14 | 2.00 | 0.16 |
| 15 | 2.00 | 0.30 |
| 16 | 2.00 | 0.23 |
| 17 | 2.50 | 0.15 |
| 18 | 2.50 | 0.30 |
| Parameter | Value |
|---|---|
| Architecture | 342-93-2 |
| Number of iterations | 180 |
| Hidden layer transfer function | Tansing |
| RMSETr (mmol L−1) 1 | 0.4072 |
| RMSEM (mmol L−1) 2 | 0.5364 |
| RMSET (mmol L−1) 3 | 0.3006 |
| Glucose | Cholesterol | |
|---|---|---|
| RMSEC (mmol L−1) 1 | 0.67 | 0.08 |
| REP (%) 1 | 2.88 | 3.09 |
| RMSEP (mmol L−1) 2 | 1.07 | 0.24 |
| REP (%) 2 | 5.17 | 7.19 |
| Slope | Intercept (mmol L−1) | r2 | ||
|---|---|---|---|---|
| Training | Glucose | 0.98 ± 0.02 | 0.05 ± 0.06 | 0.9949 |
| Cholesterol | 0.97 ± 0.02 | 0.01 ± 0.01 | 0.9947 | |
| Validation | Glucose | 0.97 ± 0.09 | 0.08 ± 0.17 | 0.9528 |
| Cholesterol | 1.08 ± 0.09 | −0.01 ± 0.02 | 0.9568 |
| Glucose (g/100 g) | Cholesterol (mg/100 g) | |||||
|---|---|---|---|---|---|---|
| Sample | Obtained | Reference a | REP (%) | Obtained | Reference a | REP (%) |
| 1 | 7.5 ± 0.5 | 7.7 ± 0.1 | 2.60 | 13.8 ± 1.1 | 13.7 ± 0.2 | 0.73 |
| 2 | 6.5 ± 0.9 | 6.5 ± 0.7 | 0.00 | 10.3 ± 1.1 | 9.9 ± 0.8 | 4.04 |
| 3 | 7.2 ± 1.3 | 7.3 ± 0.1 | 1.37 | 3.6 ± 0.2 | 4.0 ± 0.4 | 10.00 |
| 4 | 7.5 ± 0.5 | 8.2 ± 0.2 | 8.54 | 15.8 ± 0.4 | 15.1 ± 0.1 | 4.63 |
| 5 | 10.5 ± 0.1 | 10.6 ± 0.2 | 0.94 | 9.0 ± 0.4 | 8.5 ± 0.6 | 5.88 |
| 6 | 8.0 ± 0.3 | 8.2 ± 0.8 | 2.44 | 7.7 ± 0.5 | 8.6 ± 0.2 | 10.47 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Torres-Gámez, J.; Rodríguez, J.A.; Páez-Hernández, M.E.; Galán-Vidal, C.A. Simultaneous Determination of Glucose and Cholesterol in Milk Samples by Means of a Screen-Printed Biosensor and Artificial Neural Networks. Bioengineering 2026, 13, 274. https://doi.org/10.3390/bioengineering13030274
Torres-Gámez J, Rodríguez JA, Páez-Hernández ME, Galán-Vidal CA. Simultaneous Determination of Glucose and Cholesterol in Milk Samples by Means of a Screen-Printed Biosensor and Artificial Neural Networks. Bioengineering. 2026; 13(3):274. https://doi.org/10.3390/bioengineering13030274
Chicago/Turabian StyleTorres-Gámez, Jessica, José A. Rodríguez, María Elena Páez-Hernández, and Carlos A. Galán-Vidal. 2026. "Simultaneous Determination of Glucose and Cholesterol in Milk Samples by Means of a Screen-Printed Biosensor and Artificial Neural Networks" Bioengineering 13, no. 3: 274. https://doi.org/10.3390/bioengineering13030274
APA StyleTorres-Gámez, J., Rodríguez, J. A., Páez-Hernández, M. E., & Galán-Vidal, C. A. (2026). Simultaneous Determination of Glucose and Cholesterol in Milk Samples by Means of a Screen-Printed Biosensor and Artificial Neural Networks. Bioengineering, 13(3), 274. https://doi.org/10.3390/bioengineering13030274






