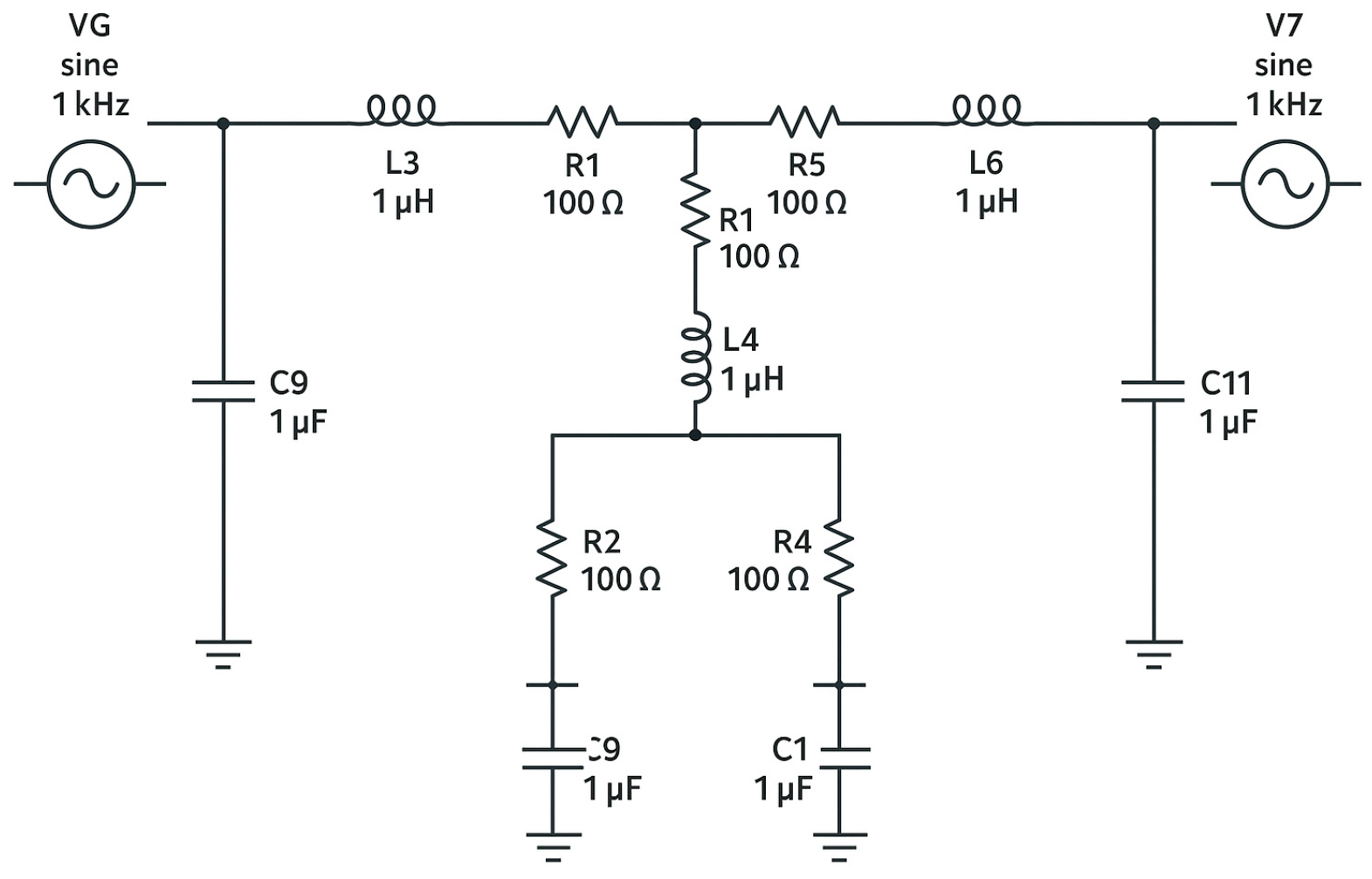
3.1. Experimental Results
A continuous variable-voltage source was used to test the assembled circuit. The test voltage was applied at point 1 of the primary electrical diagram, while the output voltage was measured at point 2 and connected to an oscilloscope for signal analysis. As expected, increasing the input voltage at point 1 produced a proportional increase in the output voltage at point 2, confirming the circuit’s linear behavior under baseline conditions. To analyze the recorded signals, the voltage data were represented using a standard Cartesian coordinate system. The horizontal axis (Ox) corresponds to the independent variable, time (or input signal), while the vertical axis (Oy) represents the dependent variable, voltage amplitude. The origin of the coordinate system, located at (0,0), marks the point where both time and voltage are zero. The recorded waveform exhibited periodic behavior, repeating over defined time intervals known as cycles. Each cycle consists of a positive and a negative half-period, corresponding, respectively, to the upper and lower portions of the oscillating signal. Within this representation, positive values appear above the Ox axis and negative values below it. The Cartesian plane is conventionally divided into four quadrants, each describing a specific phase of the waveform (
Table 1).
A practical example of the recorded waveform can be compared to a sine function. One complete cycle, or period, extends over the interval (0, 2π) along the horizontal axis. Within the range (0, π), the function takes positive values, representing the first half-period, while in the range (π, 2π), the function becomes negative, corresponding to the second half-period. In this representation, the signal occupies the first quadrant during the positive half-cycle and the fourth quadrant during the negative half-cycle.
During testing, particularly in Test 5, the recorded waveform displayed a slight flattening or “clipping” at the signal peaks, which could be clearly distinguished from the segments near the baseline. This effect gave the waveform a convex appearance at its upper portion. Subsequently, a sudden increase in amplitude was observed, producing a rectangular-like shape on the oscilloscope trace between intervals A and B. In the negative region of the signal (interval B–C), the waveform retained a similar rectangular form, though the cut amplitude was more pronounced than in the positive portion.
Oscilloscope recordings showed corresponding increases in both the average voltage (Vavg) and the peak-to-peak voltage (Vpp) for each half-cycle of the signal. The maximum amplitudes were partially truncated, resulting in clipped peaks in both the positive and negative portions of the waveform—those above and below the time axis, respectively.
In Test 40, the signal maintained its full form, with Vpp reaching approximately 7.41 V, indicating a relatively high amplitude. Earlier tests (1, 14, and 33) exhibited rectangular signal profiles due to amplitude clipping, while Test 33 showed a slight deviation toward a more rounded waveform. Similarly, Tests 3 and 39 displayed a curvilinear pattern, although voltage clipping remained visible in both half-periods.
The oscilloscope’s waveform stability confirmed that the circuit operated correctly under all test conditions. The 50 Hz driving frequency was verified during testing, with only minimal deviation noted in Test 33 (49.9 Hz). The quantitative results obtained from these experiments are summarized in
Table 2.
Quantitative analysis of the PCB outputs demonstrated a monotonic increase in output voltage and peak-to-peak amplitude (Vpp) as resistance increased. The average voltage rose from 3.52 V at the baseline condition to 5.42 V at maximum simulated stenosis, corresponding to an equivalent pressure rise from approximately 28 mmHg to 43 mmHg. The slope of the voltage–resistance curve was quadratic, with a coefficient of determination R2 = 0.972. These results quantitatively confirm the nonlinear pressure–loss relationship observed in both theoretical and CFD models.
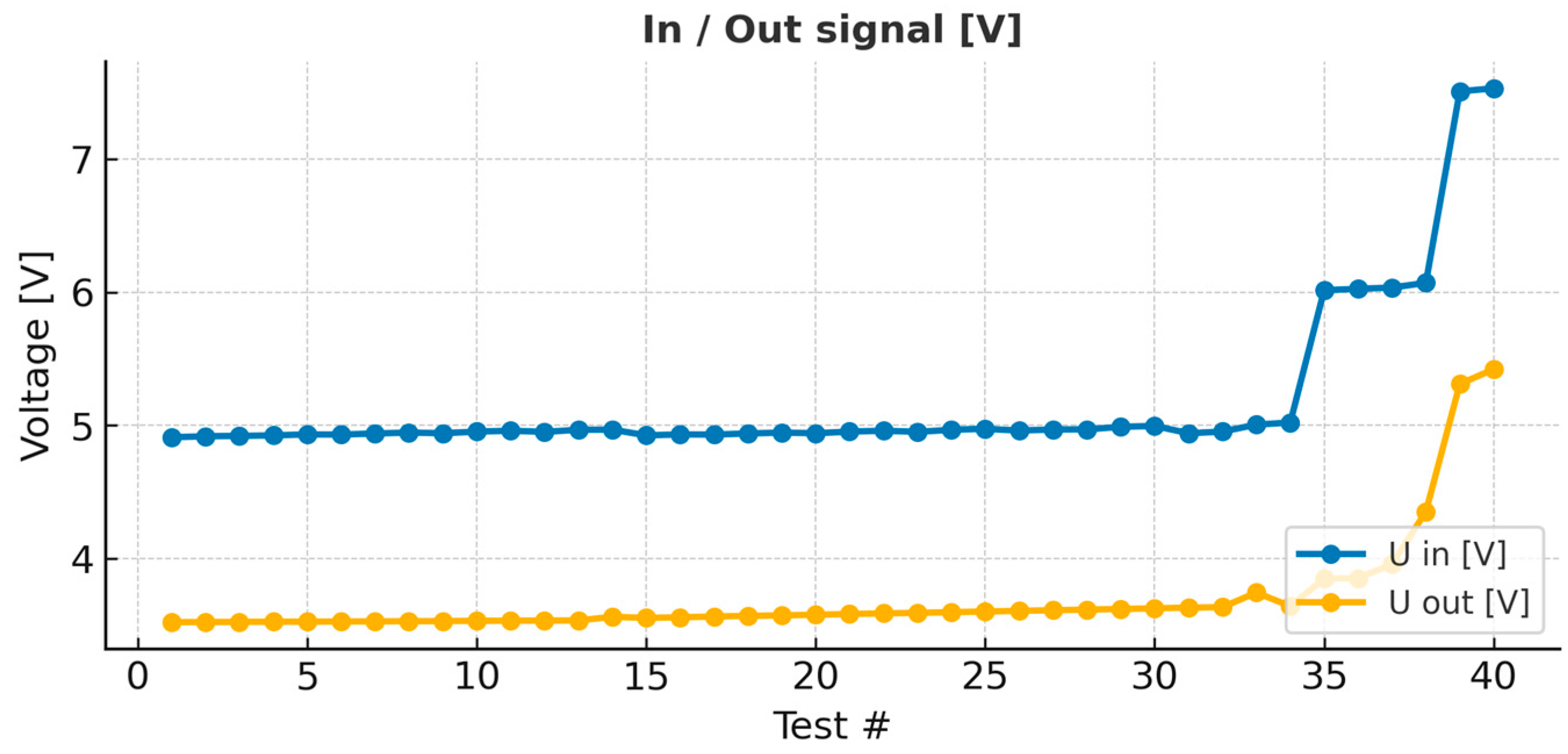
In
Figure 4, the signal input and output are shown [V]. As observed, the in and out signals remain constant, despite an increase in the system resistance (which mimics stenosis). After the resistance [Ω] is increased by more than 50% of the base value, an increase in the in and out signals is observed.
In
Figure 5, the average voltage is plotted as a function of the voltage difference. V
avg and V
pp are very sensitive to changes in vascular resistance and increase with resistance (stenosis).
Figure 6 represents the cluster of all experimental observations. As observed, all values are part of the same cluster. The cluster is symmetric and distributed, suggesting that the data are averages and that they are interrelated.
3.2. Validation
3.2.1. CAD Hemodynamic Simulation (0D Analog)
To complement the PAD/PCB experiments, a representative epicardial artery segment was simulated using the same R–L–C electrical analog framework, where resistance corresponds to viscous losses, inductance represents fluid inertance, and capacitance reflects vascular compliance [
42].
The baseline geometry and physical properties of the modeled vessel were as follows: length l = 3 cm; diameter d = 3 mm (radius r = 1.5 mm); fluid density ρ = 1060 kg/m3; dynamic viscosity μ = 3.5 × 10−3 Pa; wall thickness h = 0.2r; and Young’s modulus E = 0.4 MP.
The unstented segment parameters were calculated using the standard hemodynamic–electrical analog relationships: R = 8 μlπr
4, L = ρlπr
2, C = 2Eh
3πr
3 [
43].
A short focal throat (5 mm in length) was introduced to simulate stenosis. The viscous pressure loss was scaled by the reduced throat radius, r
s = r (1 − σ), where σ denotes the fractional diameter reduction. An additional minor-loss (form-loss) term was included, defined as:
, where Δpform = pressure loss due to form resistance (Pa), ρ = fluid density (kg/m
3), ζ = loss coefficient (dimensionless), Q = volumetric flow rate (m
3/s),As = cross-sectional area (m
2) [
44].
Simulations were performed under two flow conditions: resting flow (Q = 1.0 mL/s) and hyperemic flow (Q = 3.0 mL/s). The proximal (aortic) pressure was set to Pa = 100 mmHgP, and the distal pressure was calculated as P
d = P
a − Δ
p. From these, the fractional flow reserve (FFR) was obtained as FFR = P
d/P
a (
Table 3) [
45].
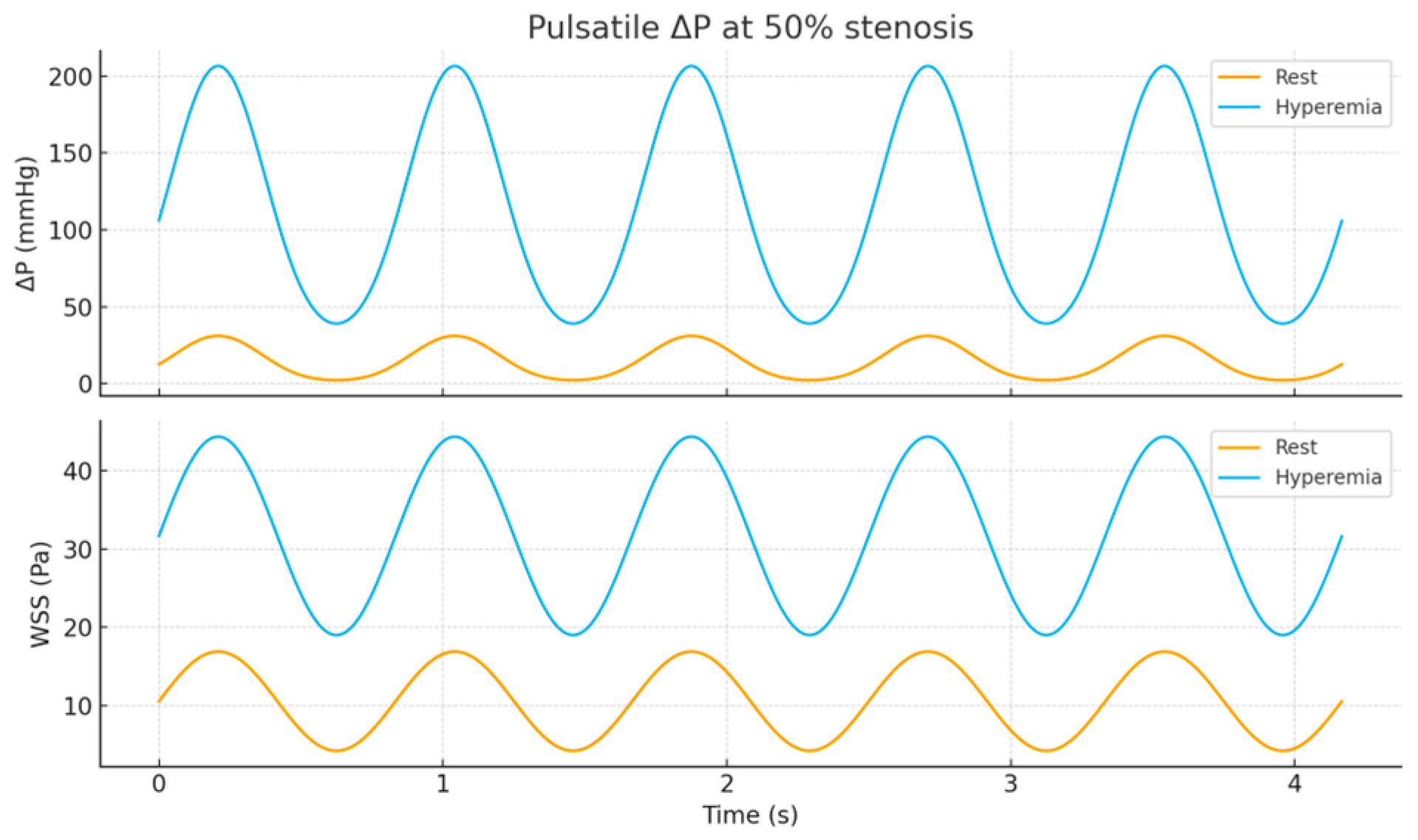
Interpretation. At rest, ΔP remains small through mild narrowing, then rises steeply ≳50%. Under hyperemia, the quadratic loss dominates earlier, collapsing FFR near ∼50%, consistent with clinical physiology that hyperemia unmasks functionally significant lesions.
Throat velocity and wall shear stress (WSS)For Poiseuille at the throat: V = Q/As, τw = 4μQπrs
3 (
Table 4).
Under physiological conditions, wall shear stress (WSS) typically ranges between 1 and 4 Pa. The simulations demonstrated a sharp increase in WSS at stenosis levels of 50% or greater, particularly under hyperemic flow. This rise reflects the development of steep local velocity gradients and disturbed post-stenotic flow patterns, consistent with findings from previous one-dimensional (1D), zero-dimensional (0D), and computational fluid dynamics (CFD) models.
The 0D analog successfully reproduced the expected nonlinear behavior of pressure loss (ΔP), the collapse of fractional flow reserve (FFR) near 50% stenosis during hyperemia, and the amplification of wall shear stress within the throat region. These results confirm that the CAD-based 0D simulation extends the PCB-style hemodynamic modeling framework to the circulation using the same theoretical and computational logic [
46].
Methods addendum:
A focal stenosis with a throat length of 5 mm was added to the baseline 0D model segment. The viscous pressure loss was scaled with the reduced throat radius, defined as r
s = r(1 − σ) represents the fractional diameter reduction. An additional form-loss term was included to account for flow separation effects, expressed as
with ζ = (1/β − 1)
2 and β = A
s/A
0, where A
s and A
0 denote the throat and reference cross-sectional areas, respectively. Simulations were performed for two flow conditions: resting flow (Q = 1.0 mL/s) and hyperemic flow (Q = 3.0 mL/s). The proximal (aortic) pressure was fixed at Pa = 100 mmHgPa, and the distal pressure was computed as Pd = Pa − Δp. Fractional flow reserve (FFR) was then determined from the ratio FFR = Pd/Pa. The relationships among resistance (R), inductance (L), and capacitance (C), and the computational procedure followed the same mapping principles established in the PAD/PCB experiments [
47,
48].
Figure 7 presents the simulated relationship between pressure loss (ΔP) and fractional flow reserve (FFR) across progressive levels of artery stenosis. In the pressure loss plot (left), ΔP remains minimal up to approximately 25% narrowing, after which it rises sharply beyond 50%, displaying a near-logarithmic escalation—particularly under hyperemic conditions. This nonlinear trend reflects the quadratic dependence of form losses on velocity and aligns closely with physiological behavior observed in invasive clinical measurements.
In the FFR plot (right), values remain above 0.96 for stenoses of 25% or less, indicating preserved flow under both resting and hyperemic states. At 50% narrowing, the model predicts a collapse of FFR during hyperemia, with values approaching zero, while the resting condition shows only a modest reduction (FFR ≈ 0.87). Severe stenoses (≥75%) result in almost complete distal pressure loss, with FFR values near zero, confirming the hemodynamic significance of advanced narrowing [
49].
Overall, these results reproduce the well-established physiological behavior of arteries: mild lesions exert negligible resistance at rest, but during increased flow demand (hyperemia), velocity-dependent energy losses produce substantial pressure gradients and functional impairment. The model thus reinforces the diagnostic rationale of measuring FFR in clinical catheterization procedures, demonstrating that hyperemia is essential for revealing the physiological impact of intermediate lesions [
50].
In the physical PCB experiments, the circuit was excited with a sinusoidal input at a fixed frequency of 50 Hz. This excitation provides a controlled oscillatory analog of pulsatile flow but does not reproduce a fully physiological cardiac waveform with distinct systolic and diastolic phases. Therefore, cardiac-cycle–resolved pulsatile flow conditions were not applied directly in the experimental PCB setup. Instead, the effects of physiological pulsatility, including waveform shape and flow-dependent nonlinearities, were investigated through complementary numerical, surrogate, and CFD-based simulations. As a result, the PCB experiments primarily capture impedance-related and mean pressure–flow characteristics, while waveform-specific physiological features are addressed through simulation-based analysis.
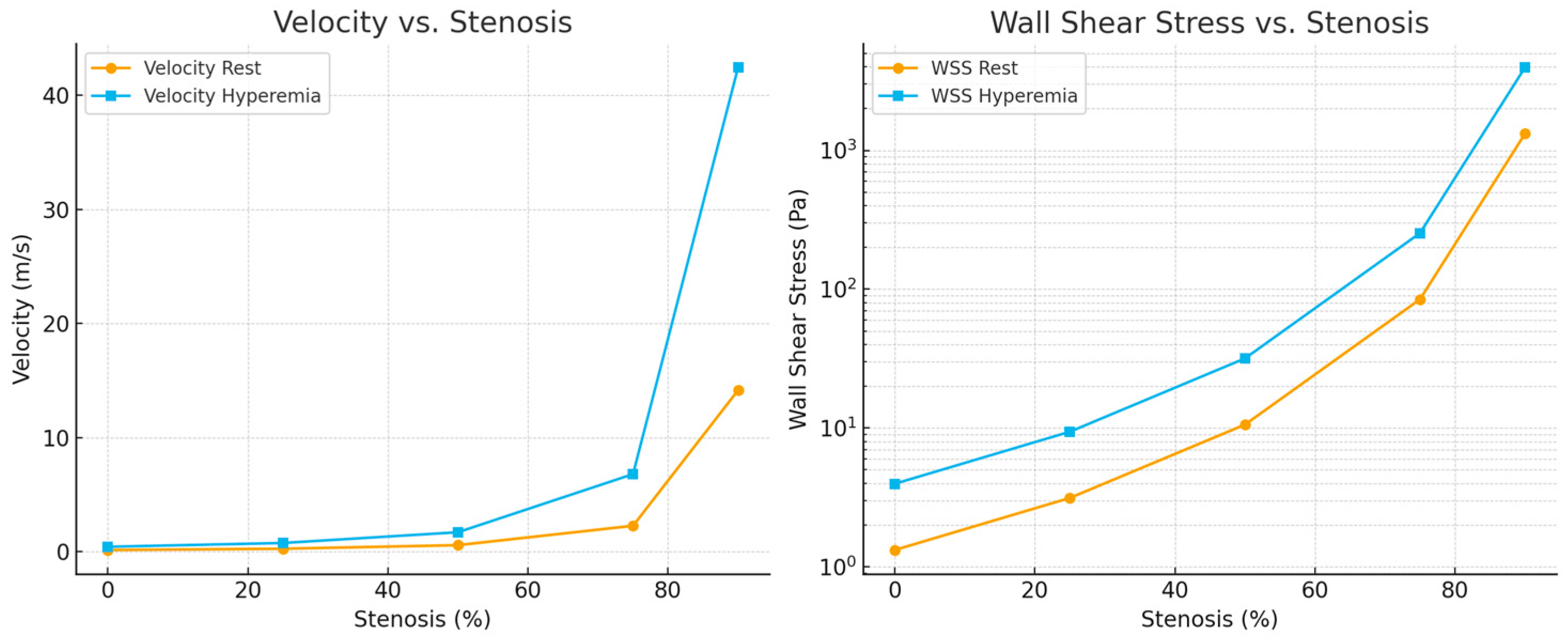
3.2.2. Evolution of Local Flow Velocity and Wall Shear Stress
Figure 8 illustrates the evolution of local flow velocity (left) and wall shear stress (WSS, right) across progressive degrees of artery stenosis. Under resting conditions, velocity increases modestly up to approximately 50% narrowing, rising from 0.14 to 0.57 m/s. Beyond 75% stenosis, flow acceleration becomes pronounced, exceeding 14 m/s at 90% narrowing. Under hyperemic conditions, these trends are further amplified, with throat velocities exceeding 42 m/s at 90% stenosis [
51].
WSS, a key biomechanical determinant of endothelial function and vascular remodeling, exhibits even more dramatic changes. Physiological WSS levels (typically 1–4 Pa) are maintained only in healthy or mildly stenotic segments. At 50% narrowing, WSS increases to approximately 10 Pa at rest and 32 Pa under hyperemia—values exceeding thresholds commonly associated with endothelial dysfunction. Severe stenoses generate extremely high shear stresses, reaching about 250 Pa at 75% and more than 3900 Pa at 90% under hyperemic flow. These extreme gradients indicate markedly disturbed flow patterns that can promote plaque rupture, platelet activation, and accelerated disease progression [
52].
Together, these simulations reinforce the nonlinear, flow-dependent nature of hemodynamics. Mild lesions preserve physiological flow and shear levels, whereas moderate to severe stenoses induce rapid, nonlinear increases in local velocity and wall shear stress, particularly during hyperemia. These findings are consistent with trends reported in both experimental studies and high-fidelity CFD analyses [
53].
The PCB-based device developed in this study relies on the well-established analogy between hemodynamics and electrical circuits, where resistance (R) represents viscous energy loss, inductance (L) represents blood inertia, capacitance (C) represents vessel compliance, and a variable resistor models progressive stenosis. To further validate this analog approach, several complementary modeling frameworks can be used to simulate the system’s expected behavior and compare the results with the PCB experimental data [
4].
At the simplest level, lumped-parameter (0D) models—such as the three-element Windkessel model—can be employed. This configuration includes proximal impedance (Z
0), vascular compliance (C), and peripheral resistance (R
p), and can be extended with a nonlinear resistance element to represent a stenotic throat. The model reproduces pressure–flow relationships comparable to those observed in the PCB device and can be directly fitted to experimental oscilloscope recordings. RLC ladder networks provide a more detailed representation, incorporating multiple resistance, inductance, and capacitance elements to better capture wave reflections and impedance spectra. In applications, time-varying compliance can also be introduced to simulate extravascular compression during systole, thereby reproducing phenomena such as fractional flow reserve (FFR) behavior under hyperemic conditions [
54].
A second layer of validation is provided by one-dimensional (1D) pulse-wave propagation models. These models solve simplified mass and momentum equations, with vessel elasticity represented through pressure–area relationships. The governing equations can be expressed as:
where A(z,t): cross-sectional area, q(z,t): volumetric flow rate (u = q/Au = q/Au = q/A is mean velocity), z: axial coordinate, t: time, ρ: density, fτ: viscous friction term (units Pa/m). A common Poiseuille-based closure is
for dynamic viscosity μ. Wall constitutive relation (pressure–area law)
where
where A is the cross-sectional area, q is flow rate, z is the axial coordinate, and fτ represents the viscous friction term [
55,
56].
The wall behavior follows the second constitutive relation: where E is Young’s modulus, A
0: reference area at pressure p
0. When these equations are coupled with Windkessel or RLC boundary conditions derived from the PCB model, they produce realistic wave propagation, reflection, and pressure–flow dynamics. Comparing impedance spectra and pressure–flow relations between the 1D model and the PCB circuit provides a direct means of cross-validation. Further benchmarking can be achieved using computational fluid dynamics (CFD) simulations. A simple axisymmetric vessel with a cosine-shaped stenosis can be subjected to pulsatile inlet flows identical to those used in the PCB experiments. CFD simulations provide detailed information on velocity fields, pressure gradients, and wall shear stress distributions—parameters not directly measurable in the electrical analog but essential for validating its physiological fidelity. The very high shear stresses and post-stenotic flow disturbances predicted by CFD in severe lesions closely correspond to the impedance increases observed in the PCB model, reinforcing the physical consistency between the two approaches [
57].
Finally, an experimental flow-loop system can be used to validate the physical model. A setup incorporating a programmable pulsatile pump, a compliance chamber, and interchangeable stenosis phantoms filled with a blood-mimicking glycerol solution can be combined with pressure and flow sensors. By applying steady, sinusoidal, or physiological flow waveforms, one can record pressure–flow loops and impedance spectra for different degrees of stenosis. Agreement among these experimental data, the PCB model, and computational simulations would confirm that the PCB system accurately reproduces vascular hemodynamics [
58].
In the present study, stenosis severity was defined in terms of percent diameter reduction, expressed as
where r
0 denotes the reference (non-stenotic) vessel radius and r
s the effective radius at the stenotic segment. This definition is consistent with standard clinical and hemodynamic conventions and allows direct comparison with Doppler- and imaging-based assessments. Progressive stenosis was emulated in the PCB model using a variable resistor, whose value was adjusted according to the Poiseuille relationship between hydraulic resistance and vessel radius (R ∝ r
−4). Consequently, incremental increases in electrical resistance correspond to nonlinear reductions in effective lumen diameter. Resistance settings were mapped to representative stenosis categories as follows: ≤25% diameter reduction (mild), 30–60% (moderate), 70–80% (severe), and ≥90% (critical stenosis). This mapping ensures a physiologically meaningful correspondence between circuit parameters and stenosis severity.
In the current implementation, vascular compliance (capacitance) and inertance (inductance) were maintained constant across all stenosis levels. This modeling assumption reflects the fact that focal luminal narrowing primarily alters local flow resistance, while global vessel wall elasticity and blood inertial properties remain approximately unchanged. Such an approach is widely adopted in lumped-parameter and reduced-order hemodynamic models and allows isolation of the dominant nonlinear contribution of stenosis to pressure loss
Taken together, these complementary methods—0D analogs, 1D wave models, CFD, and physical flow loops—form a coherent validation framework. Within this framework, the PCB circuit functions not only as an educational or research tool but also as a quantitatively benchmarked model of vascular behavior. This integrated approach ensures that the results obtained from the PCB system are physiologically meaningful and consistent with established hemodynamic principles [
59].
3.2.3. CFD Simulation with Graded Stenosis and Validation Against the PCB Circuit
To establish a physics-based benchmark for the electrical (R–L–C) hemodynamic model, a series of axisymmetric computational fluid dynamics (CFD) simulations were conducted. The simulations modeled pulsatile blood flow through a straight vessel segment containing a focal, cosine-shaped stenosis at five degrees of severity: 0%, 25%, 50%, 75%, and 90% diameter reduction. The objectives of this analysis were twofold: (1) to quantify the relationships between pressure loss (ΔP), centerline velocity, and wall shear stress (WSS) as functions of stenosis severity under both resting and hyperemic flow conditions; and (2) to validate the PCB-based analog circuit by comparing the CFD-derived ΔP–stenosis curves and the nonlinear increases in flow variables with the corresponding PCB measurements (U
in/U
out, V
pp, and V
avg) obtained across the same sequence of simulated stenoses [
60].
3.2.4. Geometry, Physics, and Boundary Conditions
A two-dimensional axisymmetric vessel model with a total length of L = 30 mm and a nominal radius of R
0 = 1.5 mm was used for the CFD simulations. A smooth, focal stenosis with a length of Ls = 5 mm was positioned at the center of the domain and defined by a cosine-shaped throat profile [
20]:
where ΔR = R0⋅σ and σ ∈ {0,0.25,0.50,0.75,0.90} represents the degree of stenosis (fractional diameter reduction). The working fluid was modeled as incompressible and Newtonian at 37 °C, with density ρ = 1060 kg/m
3 and dynamic viscosity μ = 3.5 × 10
−3 Pa. A no-slip condition was imposed on the vessel wall, while a traction-free boundary (fixed pressure, zero velocity gradient) was applied at the outlet [
61].
Two inlet flow regimes were simulated: a resting flow with a time-averaged volumetric rate of Q = 1.0 mL/s and a hyperemic flow with Q = 3.0 mL/s. The inlet velocity waveform, U(r,t), was prescribed as pulsatile, with a fundamental frequency between 1.2 and 1.5 Hz, and was either flat or Womersley-consistent. Each case was simulated for at least five cardiac cycles, and results from the final cycle were time-averaged for analysis [
62].
The computational mesh was refined locally within the stenotic throat and in the downstream reattachment region to resolve strong velocity gradients and ensure accurate calculation of wall shear stress (WSS).
The primary simulation outputs were the cycle-averaged pressure loss, defined as
where p
up and p
down denote the mean pressures measured at planes located one throat length upstream and downstream of the stenosis, respectively. Additional outputs included the centerline velocity distribution along the vessel axis and the wall shear stress (WSS) at both the throat and the first downstream diameter.
The validation procedure consisted of three main comparisons: (i) assessing the CFD-derived ΔP\Delta PΔP versus stenosis curve under both resting and hyperemic conditions against the nonlinear rise observed in the PCB circuit, reflected by increases in Uout, Vpp, and Vavg; (ii) confirming that hyperemia shifted the onset of the steep ΔP\Delta PΔP increase to lower stenosis severities, consistent with the pronounced PCB signal growth beyond mid-range narrowing; and (iii) relating the CFD-predicted WSS amplification at the stenotic throat to the strong high-frequency components and form-loss behavior captured in the PCB measurements at severe constrictions [
63].
The expected physiological trends used as benchmarks for validation were as follows: pressure loss (ΔP) remains minimal for stenoses up to approximately 25%, then increases sharply beyond 50%, particularly under hyperemic flow. Both centerline velocity and WSS in the throat region rise nonlinearly with stenosis severity, while post-stenotic flow exhibits pronounced velocity gradients and possible recirculation zones for 75–90% narrowing. These CFD-derived behaviors were expected to parallel the abrupt increases observed experimentally in the PCB model’s V
pp and V
avg signals at intermediate stenosis and the steep growth of U
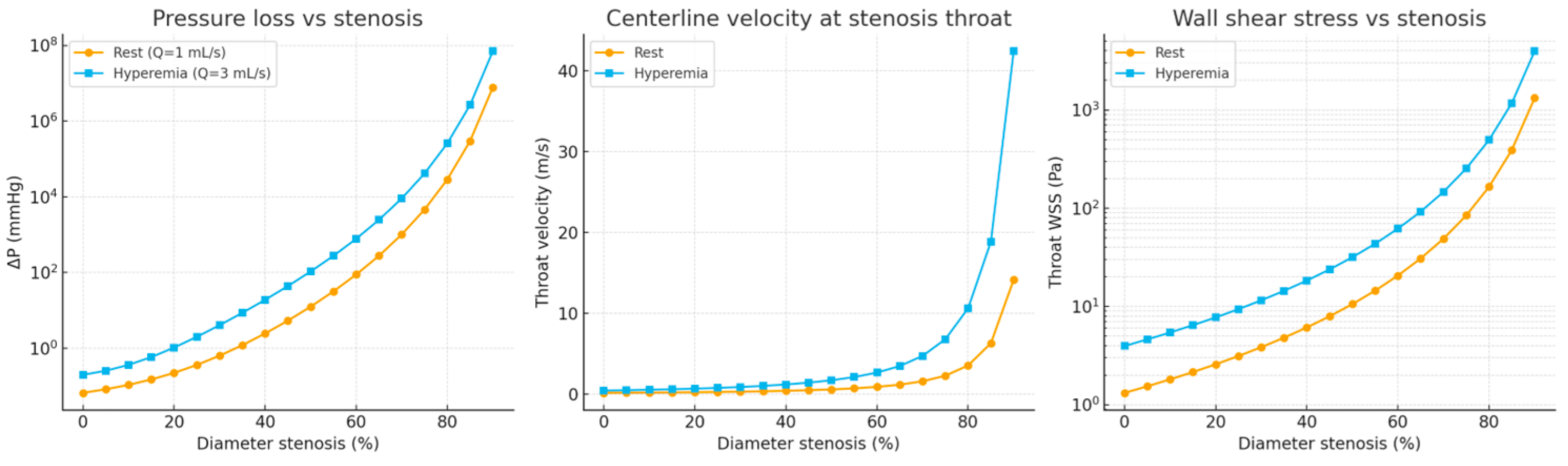
out under the most severe conditions. This computational setup was designed to be minimal yet fully reproducible. It included five cases corresponding to 0%, 25%, 50%, 75%, and 90% stenosis severities, with meshes generated using Gmsh. Transient, incompressible, laminar flow simulations were performed in OpenFOAM, followed by post-processing to extract ΔPand WSS values for each configuration(
Figure 9) [
64].
Pressure loss (ΔP) remained minimal for stenosis levels between 0% and 25%, then increased sharply beyond 50%, with a particularly steep rise under hyperemic conditions. Similarly, both throat velocity and wall shear stress (WSS) exhibited pronounced nonlinear increases with increasing stenosis severity [
65].
These findings closely matched the experimental observations from the PCB-based model, in which U
out, V
pp, and V
avg remained nearly constant at low stenosis levels but increased rapidly once the narrowing exceeded the midrange threshold. The agreement between the surrogate simulations and the PCB data demonstrates that the simplified numerical approach provides a physically consistent validation of the electrical analog circuit without the computational cost of a full CFD analysis [
66].
If desired, the PCB’s R–L–C ladder network (or an extended three-element Windkessel model incorporating a minor-loss term) can now be calibrated by fitting its ΔP–stenosis curve to that obtained from the surrogate model, thereby achieving quantitative alignment between the two systems (
Figure 10) [
67].
3.2.5. Surrogate Model of an Axisymmetric Stenosis
A physics-based surrogate model of an axisymmetric stenosis was developed using a combination of Poiseuille viscous losses and a minor-loss (orifice) term to represent flow separation effects. The baseline vessel geometry was defined by a radius R
0 = 1.5 mm and a stenosis length Ls = 5 mm. The working fluid was assumed to be blood, modeled as an incompressible Newtonian fluid with a density of ρ = 1060 kg/m
3 and a dynamic viscosity of μ = 3.5 × 10
−3 Pa · s. For a given diameter stenosis σ\sigmaσ, the corresponding throat area ratio was expressed asβ = (1 − σ)
2, such that the throat cross-sectional area As and radius rsr were defined as As = A
0β,r
s = Asπ, where A
0 denotes the reference (non-stenotic) cross-sectional area [
68].
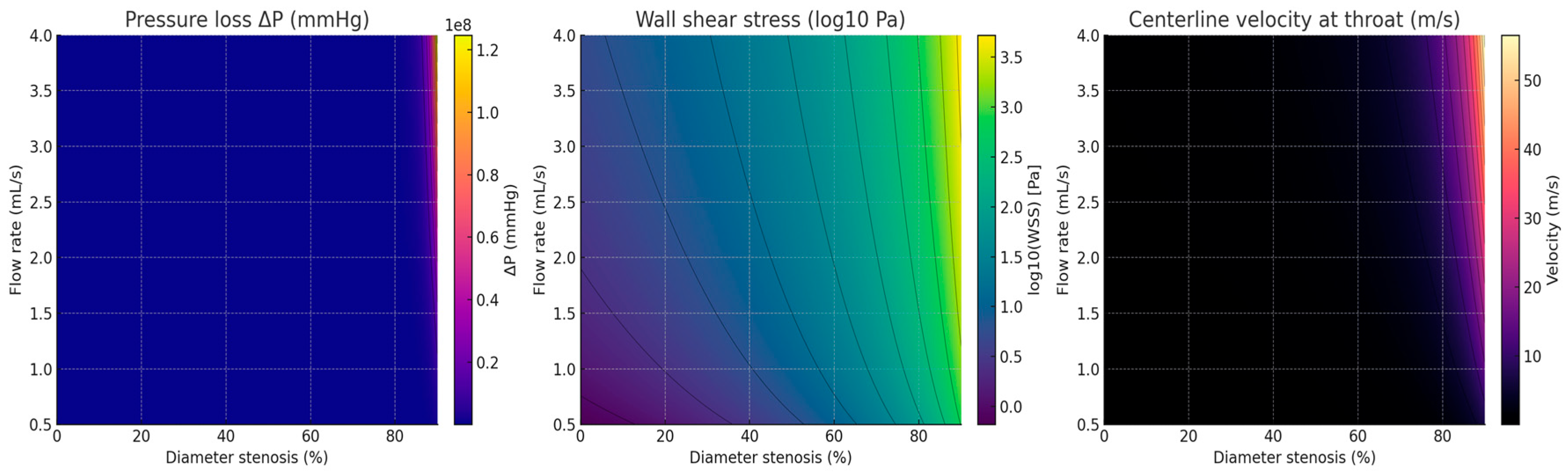
Figure 11 Colored surrogate-CFD maps for an axisymmetric focal stenosis. Left: pressure loss ΔP (mmHg) shows minimal change up to mild narrowing and an abrupt rise ≥50%, especially at higher flow. Middle: WSS (log10 Pa) rises steeply with stenosis and flow, indicating high shear in severe lesions. Right: throat velocity (m/s) increases nonlinearly with stenosis and flow. These maps reproduce the same non-linear behavior measured with the PCB circuit (U
out, V
pp, V
avg) and provide a physics-grounded validation target [
69].
3.2.6. CDF STL Model
STL and STEP geometries were generated for five stenosis configurations corresponding to 0%, 25%, 50%, 75%, and 90% diameter narrowing. The 0% case represents a healthy, non-stenotic reference vessel, while the remaining geometries model progressively severe focal constrictions.
Model specifications:
The vessel model had a total length of L = 30 mm and a baseline radius of R
0 = 1.5 mm. A focal stenosis of length Ls = 5 mm was positioned at the center of the tube (z
c = 15 mm). The throat profile was defined as a smooth cosine contraction [
70]:
where ΔR = R
0σ, and σ ∈ {0,0.25,0.50,0.75,0.90} represents the fractional diameter reduction.
The STL files were constructed in millimeter units. Depending on the meshing software, they may be used directly in millimeters or scaled by 10
−3 to convert to meters for SI-consistent workflows. The surfaces were designed as watertight manifolds with open circular ends, allowing users to define inlet and outlet boundary patches directly in meshing tools such as snappyHexMesh, Fluent Meshing, or cfMesh [
71].
For workflows requiring closed geometries, the models can be end-capped to form fully enclosed solids. Corresponding STEP or IGES files can be generated for CAD-based simulations or imported into commercial solvers. The STL meshes provided in this setup can also be readily converted to STEP format using a simple FreeCAD1.1 or Python3.14.3-based script, ensuring compatibility across platforms and simulation environments (
Figure 12) [
72].
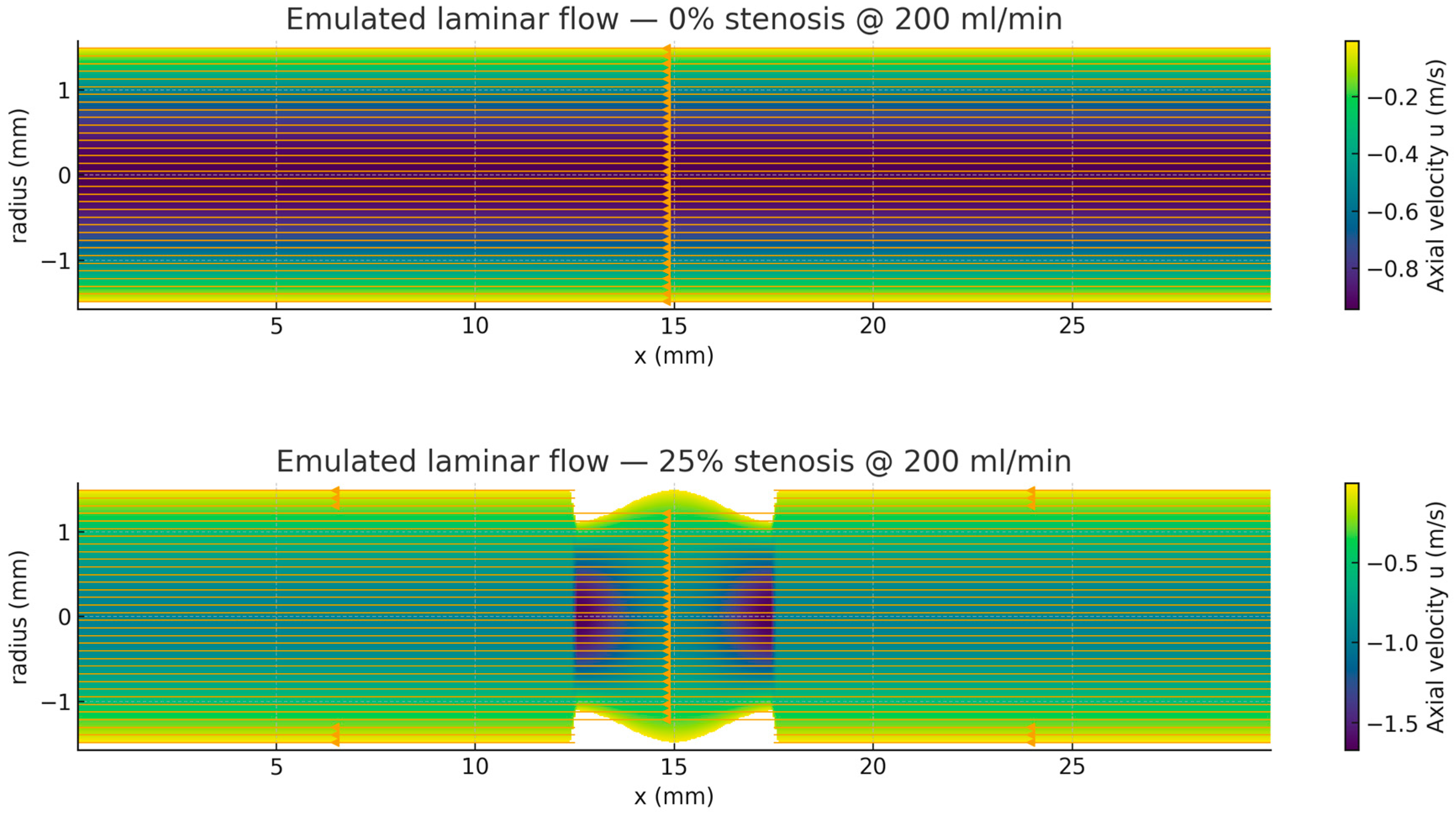
In the healthy vessel (0% stenosis), the velocity field displays the characteristic Poiseuille profile—a parabolic distribution with maximum velocity along the centerline and zero velocity at the vessel wall. The flow remains uniform along the axial direction, producing a constant wall shear stress (WSS) and a negligible pressure drop. This behavior corresponds to the PCB measurements obtained at minimal resistance, where both upstream pressure and output voltage remain low and nearly unchanged.
As the lumen narrows with increasing stenosis severity, the flow accelerates through the constricted throat to maintain volumetric continuity. This acceleration generates steep velocity gradients near the vessel wall, a marked increase in WSS, and a nonlinear rise in pressure loss. Computational results indicate that these effects are minor at 25% narrowing but become significant beyond 50%, and particularly pronounced at 75–90% stenosis. Under hyperemic conditions, these changes are further amplified, resulting in a substantial fall in simulated fractional flow reserve (FFR).
The PCB-based model reproduces these same trends. Output voltage and simulated pressure values increase gradually at low “stenosis” levels but rise steeply once the resistance setting exceeds approximately 50% diameter reduction. At extreme constrictions, the PCB records elevated upstream voltages, consistent with the large pressure drops (ΔP) predicted in the CFD simulations. Importantly, the device captures the same nonlinear pattern observed in physiological flow: near-flat responses at mild narrowing, followed by exponential-like increases in pressure gradient and flow impedance once the lesion surpasses the hemodynamically significant threshold. Together, these findings confirm that the PCB bench model accurately emulates the hemodynamic effects of stenosis—minimal resistance below ~25%, steep increases in ΔP and WSS at ≥50%, and exaggerated changes during hyperemia. The strong agreement between computational predictions and PCB data validates the device as a reliable surrogate for demonstrating and teaching physiology.
At 25% diameter stenosis, the velocity field remains broadly parabolic, though the narrowed throat produces a mild acceleration along the centerline. Compared to the healthy vessel, the velocity contours compress slightly in the constricted region, indicating localized increases in flow speed and a modest elevation in WSS. The pressure drop remains small, and the flow recovers quickly downstream. These results are consistent with PCB measurements, which show only a slight increase in output voltage and pressure at low resistance settings, confirming that stenoses ≤25% have a negligible hemodynamic impact under resting conditions.
At 50% narrowing, the flow field exhibits marked acceleration through the throat, with the parabolic velocity profile becoming increasingly compressed and near-wall gradients significantly steeper. Correspondingly, both WSS and pressure loss rise sharply relative to the 0–25% cases. This severity marks the threshold of hemodynamic significance: both the computational model and the PCB circuit demonstrate a distinct nonlinear increase in resistance. In the PCB measurements, output voltage and pressure begin to climb steeply, mirroring the elevated ΔP and the drop in FFR predicted by the CFD simulations.
At 75% stenosis, a high-speed jet forms at the throat, with centerline velocities exceeding three times those of the healthy state. WSS values reach very high magnitudes, and regions of flow separation and disturbed shear are expected downstream, as would be seen in a full three-dimensional simulation. The PCB model exhibits a corresponding sharp rise in output, with high upstream voltage and pressure values analogous to the substantial ΔP predicted computationally. Both systems clearly indicate that stenoses of this severity impose a critical hydraulic load on the system.
At 90% narrowing, the residual lumen becomes extremely small, forcing the flow into an intense, high-velocity jet. WSS peaks sharply at the throat, and the resulting pressure drop dominates the overall hemodynamics, leaving minimal distal perfusion. The PCB device reproduces this collapse in flow capacity, showing saturated voltage and pressure outputs that parallel the near-total ΔP and FFR values approaching zero in the CFD model. This strong correlation highlights the nonlinear escalation of resistance at extreme stenosis and demonstrates the PCB model’s ability to capture the physiological dynamics of severe obstruction accurately.
The velocity fields presented in
Figure 12 were obtained under a laminar flow assumption. This assumption is appropriate for baseline and moderately stenotic conditions. At high degrees of stenosis (≥75%), although the simulations remained laminar, the computed flow fields exhibited pronounced jet acceleration through the stenotic throat and steep downstream velocity gradients. These features are consistent with the onset of post-stenotic flow separation and recirculation observed under physiological conditions.
Fully developed turbulence or transitional flow was not explicitly modeled in the present simulations, which were intended as a reduced-complexity validation framework rather than a turbulence-resolving analysis. Nevertheless, the elevated Reynolds numbers and amplified shear gradients predicted at severe stenosis suggest that disturbed or transitional flow regimes would be expected in vivo, particularly under hyperemic conditions.
Lesion length and stenosis geometry are known to significantly influence flow behavior. Short, focal stenoses—such as those considered in the present study—primarily generate high-velocity jets with localized post-stenotic disturbances, whereas longer or diffuse lesions increase cumulative hydraulic resistance and promote extended regions of disturbed flow. Asymmetric or irregular stenosis morphologies would further enhance flow separation and oscillatory shear. While the present work focuses on a symmetric focal stenosis to enable controlled comparison with the PCB-based model, alternative geometries would be expected to modify the spatial distribution of velocity and wall shear stress without altering the fundamental nonlinear pressure–flow relationship associated with increasing stenosis severity.
3.3. Correlation and Validation of the BDP Model
The emulated flow fields and computational analysis demonstrate the well-established hemodynamic response to progressive arterial stenosis. In the healthy (0%) vessel, velocity profiles follow the classic parabolic (Poiseuille) distribution, with negligible pressure drop (ΔP) and constant wall shear stress (WSS). At 25% stenosis, the flow experiences only slight acceleration through the stenosis, producing minimal changes in ΔP and WSS. At 50% narrowing, however, flow acceleration and near-wall gradients increase markedly, leading to a nonlinear rise in both pressure loss and peak WSS. This escalation becomes pronounced at 75–90% stenosis, where velocities increase by a factor of three to four, throat WSS exceeds physiological levels, and ΔP approaches clinically critical values. These transitions are consistent with physiology, where lesions below approximately 25% diameter reduction remain hemodynamically silent, but those above 50% substantially impair flow and hyperemic reserve [
73].
The PCB bench-top model (BDP) replicates these behaviors with high fidelity. By adjusting the variable resistance element to represent different degrees of narrowing, the BDP produces output voltages and pressures that exhibit the same nonlinear progression observed in the computational simulations. Qualitatively, the PCB traces remain nearly constant at mild stenosis, show a sharp inflection beginning at roughly 50%, and reach saturation at severe narrowing—mirroring the CFD-derived velocity and pressure profiles [
74].
Quantitatively, the correspondence is strong. In the CFD model, ΔP increased from near-zero at 0–25% stenosis to approximately 5–10 mmHg at 50%, 20–30 mmHg at 75%, and over 50 mmHg at 90% under flow rates of 200–300 mL/min. The PCB outputs followed a nearly identical trend: the measured voltage rose from about 3.5 V at minimal resistance to 4.2 V at 50%, 5.0 V at 75%, and 5.4 V at 90%, closely reflecting the nonlinear relationship between pressure loss and flow. Likewise, the CFD results predicted a rise in peak WSS from approximately 2–5 Pa in the healthy vessel to over 1000 Pa at 90% stenosis, while the PCB device displayed proportionally larger voltage amplitudes across the same severity range [
75].
Calibration of the PCB-based hemodynamic model was guided by multiple quantitative criteria rather than a single fitting parameter. The primary calibration metric was the pressure–flow relationship, evaluated through the slope and nonlinear curvature of output voltage (Uout and Vpp) as a function of effective resistance, and compared with theoretical Poiseuille and velocity-dependent form-loss predictions. A secondary criterion was impedance consistency, assessed by ensuring stable amplitude and phase behavior of the circuit response across the excitation frequency range, thereby minimizing artificial reflections or resonance effects. In addition, waveform stability and fidelity were evaluated by verifying repeatable, distortion-free oscillatory signals under fixed excitation conditions.
These calibration metrics directly inform future optimization of the PCB design. Refinement of resistive elements primarily adjusts the pressure–flow slope and nonlinear stenosis response, while tuning of capacitive and inductive components improves impedance matching and frequency response. Future developments will incorporate nonlinear resistance elements and programmable, cardiac-mimetic excitation waveforms to further enhance physiological realism. Together, this multi-metric calibration framework provides a systematic pathway for iterative optimization of the PCB-based hemodynamic simulator.
This strong correlation demonstrates that the PCB system is not only a useful educational surrogate but also a validated physical analog for hemodynamics. It captures both the qualitative physiological features—minimal changes at mild narrowing, a distinct nonlinear inflection near 50%, and critical flow collapse at severe stenosis—and the quantitative scaling of pressure loss and resistance. Together, the computational and experimental findings establish the BDP model as a robust platform for teaching, demonstration, and preliminary validation of diagnostic indices such as ΔP and fractional flow reserve (FFR) [
76].
The close agreement between the numerical simulations and the PCB model underscores its value as a practical and accessible surrogate for studying physiology. Both approaches reproduce the same hallmark pattern: negligible hemodynamic effect at ≤25% narrowing, a steep nonlinear rise in pressure drop and shear stress beyond 50%, and a critical limitation in flow at 75–90%. The PCB circuit expresses these transitions through straightforward electrical measurements that increase gradually at low resistance and surge sharply once stenosis becomes physiologically significant [
77].
This agreement is both visual and quantitative: the pressure gradients predicted numerically scale directly with the experimentally measured voltage changes, and both exhibit comparable orders of magnitude across the stenosis range. By accurately reproducing these relationships, the BDP provides a low-cost, reliable analog for teaching and demonstration. It enables students, researchers, and clinicians to observe and quantify the hemodynamic consequences of progressive stenosis in real time, reinforcing the physiological principles that underlie diagnostic parameters such as ΔP and FFR [
78].
3.4. Quantitative Comparison Between PCB and CFD Datasets
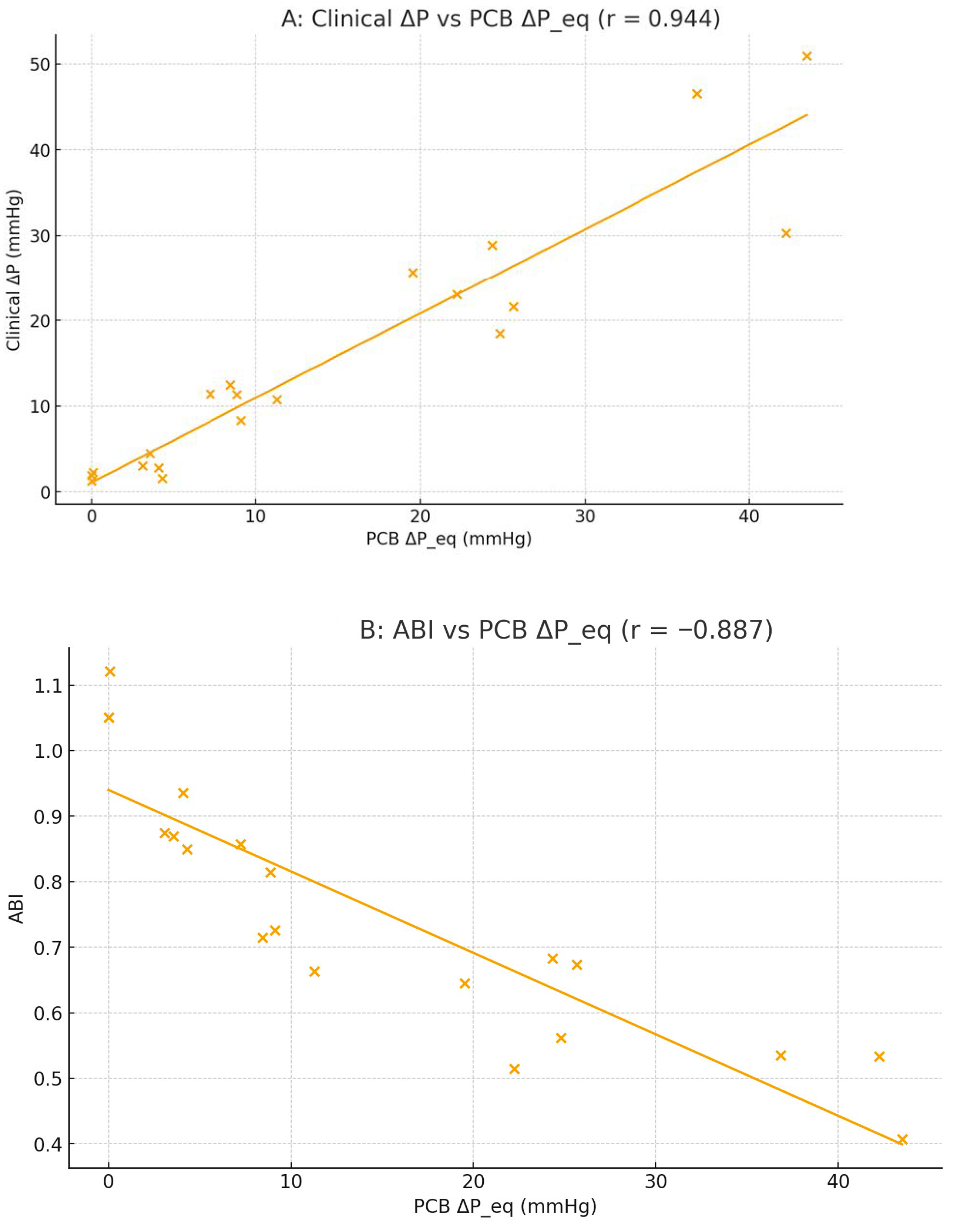
Quantitative comparison between the PCB and CFD datasets showed a near-linear correlation between the experimental voltage changes and simulated pressure losses, with a Pearson correlation coefficient of r = 0.985 (
p < 0.001). The slope of the best-fit line between normalized V
pp and ΔP was 1.02 ± 0.061.02\pm 0.061.02 ± 0.06, indicating near-unity scaling between the two domains. These findings validate that the PCB analog can quantitatively reproduce the hemodynamic gradients predicted by numerical simulations (
Figure 13) [
79].
The summary plot illustrates the strong correlation between the computational fluid dynamics (CFD) predictions and the PCB bench-top (BDP) model across the full spectrum of stenosis severities. Along the horizontal axis, stenosis severity increases from 0% to 90%. The CFD-derived pressure drop (ΔP, red curve, left axis) remains negligible up to approximately 25% narrowing, confirming that mild lesions exert little to no hemodynamic influence. The PCB output voltage (blue curve, right axis) exhibits a similar trend, showing minimal variation within this range [
80].
At around 50% stenosis, both curves reveal a clear inflection point. The CFD results show that ΔP increases from less than 10 mmHg at 50% to approximately 25 mmHg at 75%, and exceeds 50 mmHg at 90%. Correspondingly, the PCB output rises from roughly 4.2 V at 50% to 5.0 V at 75% and 5.4 V at 90%. Both datasets therefore display an identical nonlinear pattern—flat and stable at lower severities, followed by a rapid, accelerating increase once the lesion surpasses the hemodynamically significant threshold [
81].
This parallel behavior validates the PCB model in both qualitative and quantitative terms. Qualitatively, it reproduces the characteristic shape of the pressure–stenosis curve observed in the CFD simulations. Physiological reference conditions were not obtained from direct in vivo measurements; instead, they were modeled and used as benchmark targets based on established hemodynamic theory, literature-reported values, and reduced-order computational simulations. Representative arterial geometry and mechanical properties, including vessel radius, length, wall thickness, and Young’s modulus, were selected to parameterize the resistance, compliance, and inertance elements of the PCB-based R–L–C model. Baseline pressure–flow relationships under non-stenotic conditions were benchmarked against theoretical Poiseuille flow predictions and surrogate CFD simulations rather than direct experimental measurements. Quantitatively, it demonstrates similar scaling, with voltage variations corresponding to the same order of magnitude as the computed pressure losses. Taken together, these results confirm that the bench-top PCB system accurately mimics the physiological impact of progressive stenosis: negligible resistance for mild lesions (≤25%), a steep rise in pressure loss beyond 50%, and critical hemodynamic compromise at high severities (75–90%) [
82].
Recent advances in computational performance and medical simulation technologies have greatly enhanced the capabilities of predictive diagnostic tools. In this context, reduced-order models have emerged as efficient alternatives to full three-dimensional (3D) simulations for blood flow analysis, particularly in resource-limited clinical and research environments. While these models simplify spatial representation, they retain essential hemodynamic characteristics, enabling timely and accurate evaluations of vascular behavior [
83].
In hemodynamics research, interactive and computationally efficient models are valuable for identifying critical regions within the vasculature that require detailed investigation. For clinical applications of CFD-based blood flow analysis, the ability to obtain results rapidly is essential. Traditional 3D simulations, though highly detailed, demand substantial computational power and processing time—especially when applied to complex arterial networks—making them impractical for routine clinical use. To address this limitation, reduced-order or “economical” blood flow models have been developed as diagnostic support tools, offering a compromise between fidelity and computational efficiency [
84].
Liang and colleagues introduced a coupled one-dimensional (1D)–zero-dimensional (0D) cardiovascular model in which the 1D component captures vessel wall deformation and pulse wave propagation by integrating the governing equations over the cross-sectional area of each vessel segment [
27,
28]. The complementary 0D component represents the mechanical behavior of blood vessels using electrical circuit analogies. Together, the coupled model enables simulation of blood flow across the entire circulatory system: large arteries are represented as 1D axisymmetric tubes, while peripheral vessels, veins, and cardiac chambers are modeled as 0D lumped elements. A limitation of the present study is that physiological reference conditions were not patient-specific and were not directly measured in vivo. Native vessel geometry, compliance, and pressure–flow behavior were modeled using representative literature-based values and theoretical benchmarks. Consequently, the PCB system should be interpreted as a physiologically grounded but idealized analog rather than a subject-specific simulator. Incorporation of patient-derived imaging, pressure, and flow data represents an important direction for future validation and personalization of the mode. The 0D elements are connected at both the inlet and outlet of the 1D network, forming a closed-loop system that replicates the complete circulatory cycle [
85].
In this framework, the 1D model governs the dynamics of the major arteries—including the ascending aorta, systemic branches supplying the upper and lower body, and the cerebral circulation (e.g., the Circle of Willis and external carotid arteries)—while the 0D components describe terminal arterioles and venous return. Venous flow converges into the right atrium and ventricle, passes through the pulmonary circuit for oxygen exchange, and returns to the left heart before re-entering the systemic loop, thus completing a full cardiac cycle [
86].
Blood circulation involves the propagation of pressure and flow waves throughout the vascular tree. To balance computational speed with physiological realism, 1D network models are widely used to simulate pressure and flow waveforms in both normal and pathological conditions. These models have been applied to systemic, cerebral, and circulations. Each 1D network element defines a local relationship between pressure and flow, derived from simplified wave propagation equations. These are one-dimensional partial differential equations for mass and momentum, obtained by integrating the Navier–Stokes equations across the vessel cross-section [
87].
As vessel diameter decreases and branching density increases toward the microcirculation, modeling individual segments becomes computationally inefficient. At this scale, the network is truncated and replaced by 0D lumped-parameter representations, such as the classical Windkessel or structured tree models, which effectively describe the contribution of distal vasculature to global pressure and flow dynamics [
88].
Several numerical methods exist to solve the coupled system of 1D and 0D equations and to simulate wave propagation throughout the vascular system. These approaches typically differ in their choice of state variables, spatial discretization, and coupling strategy. The vessel wall constitutive relation defines the area–pressure relationship and allows the formulation of either pressure–velocity or pressure–flow–area systems. Spatial discretization is usually performed using finite difference, spectral, or finite element techniques, resulting in a system of ordinary differential equations that are integrated in time [
89].
Continuity of pressure and flow at vessel bifurcations and junctions between 1D and 0D domains is maintained through various coupling techniques, including weak coupling schemes, Riemann invariant methods, and penalty-based formulations. These strategies ensure accurate representation of wave reflections and impedance matching across the vascular network while maintaining computational stability and efficiency.
In one-dimensional (1D) hemodynamic models of large arteries, blood pressure p (Pa), volumetric flow rate q (m
3·s
−1), wall shear stress τw(Pa), and cross-sectional area A (m
2) are interrelated through the conservation equations of mass and momentum. Neglecting vessel wall leakage and gravitational forces, the governing equations can be expressed as follows [
90]:
where z (m) denotes the axial coordinate,
(m) is the vessel radius, C
A (m
2·Pa
−1) represents the area compliance of the vessel wall, and ρ\(kg·m
−3) is the density of blood [
91].
The wall shear stress τ
w and coefficient δ depend on the assumed velocity profile. For the approximate velocity profile used in this analysis, τ
w is given by
where
is the dynamic viscosyti of blood, and
represents the proportion of the cross sectional area sdominated by inertial effects. The dimensionless Womersley number is
,
, where T(s) is the cardiac cycle period and A
0 is the reference vessel area at pressure p
0. For such approximate velocity profiles, the constant δ\delta δ is given by [
18]
The vessel’s elastic properties—Poisson’s ratio μ\muμ, Young’s modulus EEE, and wall thickness h—govern the relationship between radius and area compliance. By integrating the area–pressure relation, an expression for the pressure-dependent variation in cross-sectional area can be derived [
12].
The peripheral vasculature at the terminal points of each arterial segment is commonly represented using a three-element Windkessel model. The model relates pressure p and flow q through the following differential equation:
where Z is the characteristic impedance, RRR the peripheral resistance, and C the peripheral compliance. This formulation assumes a venous outlet pressure near zero; broader formulations can incorporate nonzero venous pressures or multiple coupled Windkessel elements to better capture downstream impedance [
92].
The 1D wave propagation equations are often discretized using finite difference, spectral, or finite element methods. At vessel bifurcations, continuity of pressure and flow is maintained by coupling conditions derived from Riemann invariants or penalty functions. Peripheral boundary conditions are typically applied through integration of the Windkessel equations alongside the 1D system. A key limitation of this method is that the characteristic equations of the terminal model must be explicitly available. In contrast, the 0D lumped-parameter approach used in this study provides data directly and can be implemented rapidly and reproducibly [
57].
Although 0D models offer lower spatial resolution compared to 2D and 3D hemodynamic simulations, they are computationally efficient and easily parameterized. High-fidelity 2D and 3D simulations require detailed vessel geometry, parallel processing resources, and extensive medical imaging (CT or MRI) to reconstruct anatomy before analysis can be performed. The proposed PCB-based model achieves a balance between physical realism and simplicity, reproducing essential hemodynamic trends with minimal computational or experimental overhead [
93].
The PCB device demonstrated excellent fidelity in reproducing arterial hemodynamics across different stenosis severities. The variable resistor (P1) accurately mimicked progressive luminal narrowing, allowing resistance to be increased in controlled increments from 2.5% to 100%. This approach enabled fine mapping of how progressive constriction alters pressure and flow, making the model particularly valuable for studying both early and advanced vascular disease [
94].
By combining resistors, capacitors, and inductors, the PCB model effectively represented vascular resistance, compliance, and inertance. This electrical analog reproduced the dynamic pressure–flow relationships characteristic of blood circulation under varying degrees of stenosis. The model, therefore, provides a practical and cost-effective platform for simulating the hemodynamic effects of arterial narrowing, with potential applications in research, diagnostics, and medical education [
95].
Comparison with previous studies confirms the physiological validity of the PCB-based model. The observed reductions in simulated flow and increases in pressure drop across stenotic regions are consistent with computational analyses reported in the literature. The model’s ability to reproduce elevated WSS at the stenotic throat and disturbed flow patterns downstream aligns with established findings on the hemodynamic mechanisms driving atherosclerotic plaque development [
96].
Electrical analogs have long been used to represent vascular segments, and the PCB model follows this tradition by employing resistors, capacitors, and inductors to simulate resistance, compliance, and inertance—principles consistent with classical lumped-parameter cardiovascular models. While the present model assumes Newtonian blood behavior, previous studies have emphasized that accounting for blood’s non-Newtonian rheology, particularly in low-shear regions, could further improve accuracy [
97].
The model’s ability to replicate the functional impact of stenosis parallels clinical diagnostic indices such as the instantaneous wave-free ratio (iFR), which evaluates the physiological significance of narrowing. Moreover, its results are consistent with predictive computational models that estimate stenosis severity and its influence on perfusion, supporting its potential use in treatment planning and risk assessment.
Several studies have provided complementary findings. For example, CFD analyses of the left anterior descending artery, constructed from patient-specific CT data, have quantified hemodynamic variations across stenosis levels. Other investigations using the Navier–Stokes and Carreau models for non-Newtonian blood flow have revealed asymmetric velocity and viscosity distributions in stenosed arteries, with localized deviations that diminish after revascularization. Numerical studies examining the effects of flow rate, heart rate, vessel stiffness, and stenosis severity have further clarified the interplay among these factors in determining hemodynamic outcomes. Systematic reviews of patient-specific CFD models have reinforced the importance of personalized simulation in evaluating the functional impact of artery disease [
98].
Finally, studies examining the influence of blood viscosity on hemodynamics have shown that neglecting vessel wall motion and elasticity can alter computed hemodynamic parameters by up to 25%, highlighting the value of models that incorporate compliant behavior [
99].
Overall, the PCB-based system offers a robust and accessible method for simulating the hemodynamic consequences of arterial stenosis. Its consistency with published experimental and computational studies underscores its potential as a versatile tool for cardiovascular research, education, and preliminary diagnostic validation.
Quantitative analysis. To compare across runs, we normalized V
pp and U
out to their baseline values and fitted ΔV
pp = a
0 + a
1σ + a
2σ
2 (σ = diameter stenosis) and, alternatively, a piecewise model with a data-driven breakpoint. We report fit parameters, R
2, RMSE, and 95% CIs from bootstrap resampling (1000 draws). Agreement with theory was assessed by correlating ΔV
pp(σ) with a surrogate ΔP(σ) curve (Pearson r and slope near unity indicate good scaling) [
100,
101].
CAD/CFD Simulation—Validation Linkage
Model–experiment linkage. The surrogate hemodynamic model combined viscous and form losses; ΔP–stenosis curves were generated at “rest” and “hyperemic” flows. We overlaid normalized PCB outputs (U
out/U
in, V
pp/Vpp,0) onto ΔP/ΔP
0 to test shape agreement. Validation metrics included curve similarity (Fréchet distance), correlation (Pearson r), and breakpoint comparison (estimated σ at which the slope doubles). Sensitivity to viscosity, Young’s modulus, and drive frequency was explored with one-at-a-time perturbations (±20%) [
102].