Digital Technologies in Implantology: A Narrative Review
Abstract
1. Introduction
- Three-dimensional imaging and diagnostics using CBCT and intraoral scanning;
- Virtual implant planning and surgical guide design;
- Computer-guided and dynamic navigation surgery;
- Digital impression techniques for implant-supported restorations;
- CAD/CAM fabrication of implant prosthetics;
- Artificial intelligence and machine learning applications for treatment planning and outcome prediction.
- Evaluate the current applications of digital technologies in implantology
- Assess the accuracy and efficacy of digital methods compared to conventional approaches
- Identify the benefits and limitations of digital workflows in implant dentistry
- Explore future perspectives and emerging trends in digital implantology
2. Materials and Methods
3. Results
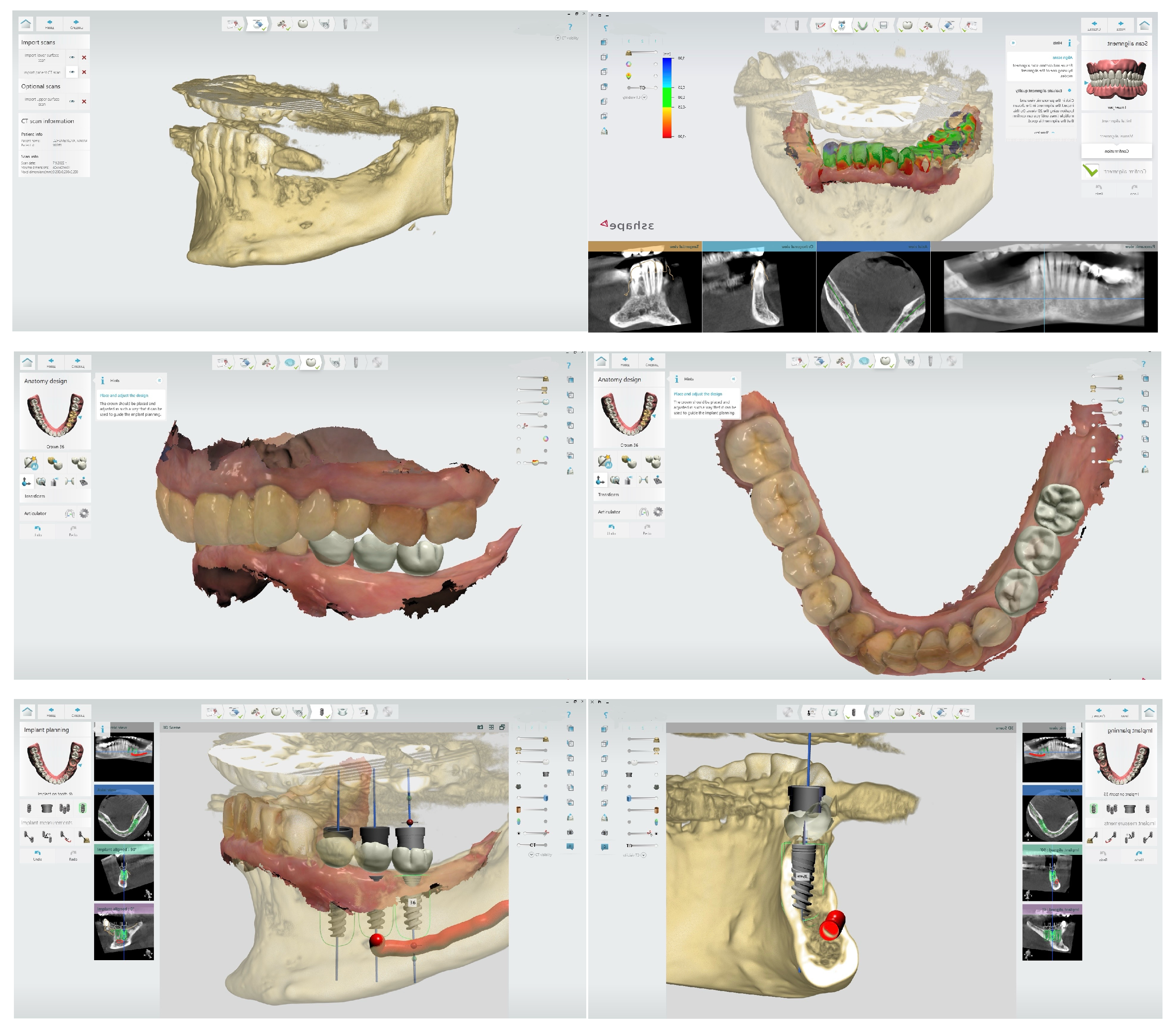
3.1. Digital Planning and Guided Surgery
3.2. Dynamic Navigation Systems
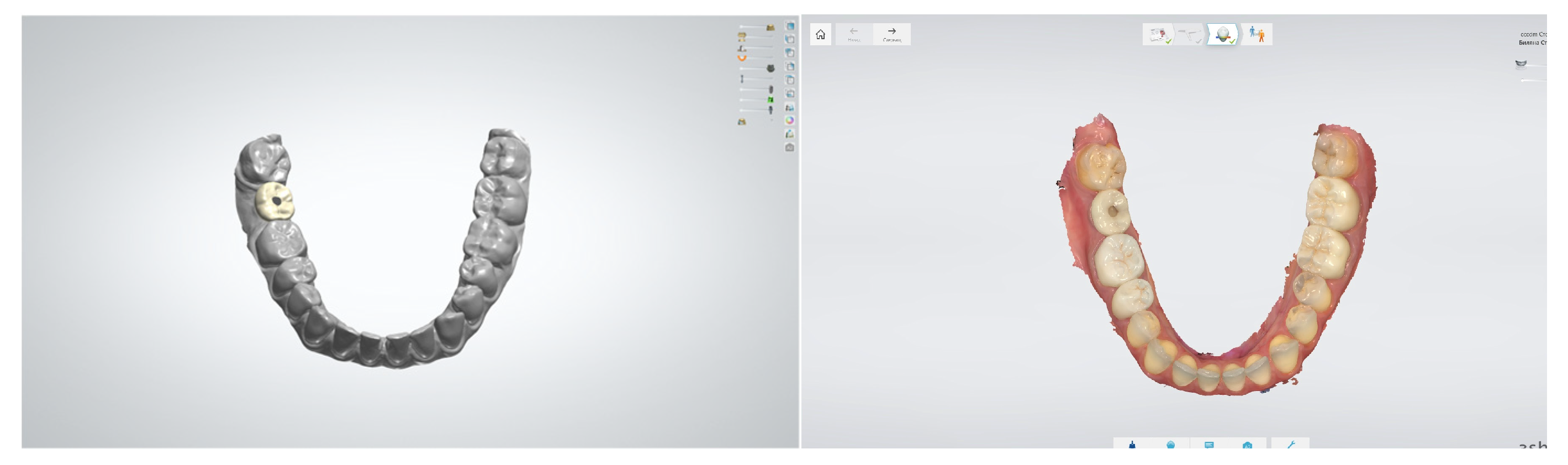
3.3. Digital Impressions
3.3.1. Accuracy and Precision
3.3.2. Workflow Efficiency and Patient Experience
3.3.3. Technological Advancements and Innovations
3.3.4. Challenges and Limitations
3.3.5. Future Directions
3.4. CAD/CAM in Implant Prosthetics
3.5. Artificial Intelligence and Machine Learning in Implantology
4. Discussion
5. Conclusions
- Long-term clinical outcomes and biological effects of fully digital workflows, especially in complex cases and full-arch rehabilitations, where robust longitudinal data are still lacking.
- The clinical accuracy, reliability, and practical implementation of emerging technologies such as photogrammetry, light-field imaging, and augmented reality for implant planning and impression-taking.
- The standardization and reproducibility of digital workflows across different software platforms, hardware systems, and operator experience levels, to ensure consistent clinical results.
- The integration and autonomous application of artificial intelligence and machine learning algorithms in routine clinical decision-making, risk assessment, and treatment outcome prediction.
- The impact of digital implantology on peri-implant tissue health, and the incidence of biological and technical complications over extended follow-up periods.
- Patient-reported outcomes, satisfaction, and psychological acceptance of digital implant treatments compared to conventional protocols, particularly in diverse populations and complex cases.
- Data security, privacy, and ethical management of digital patient information within fully digital implantology workflows.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Joda, T.; Zarone, F.; Ferrari, M. The complete digital workflow in fixed prosthodontics: A systematic review. BMC Oral Health 2017, 17, 124. [Google Scholar] [CrossRef] [PubMed]
- Buser, D.; Sennerby, L.; De Bruyn, H. Modern implant dentistry based on osseointegration: 50 years of progress, current trends and open questions. Periodontol. 2000 2017, 73, 7–21. [Google Scholar] [CrossRef]
- Mangano, F.; Shibli, J.A.; Fortin, T. Digital Dentistry: New Materials and Techniques. Int. J. Dent. 2016, 2016, 5261247. [Google Scholar] [CrossRef]
- Lee, S.J.; Jamjoom, F.Z.; Le, T.; Radics, A.; Gallucci, G.O. A clinical study comparing digital scanning and conventional impression making for implant-supported prostheses: A crossover clinical trial. J. Prosthet. Dent. 2022, 128, 42–48. [Google Scholar] [CrossRef]
- Att, W.; Witkowski, S.; Strub, J.R. Digital workflow in reconstructive dentistry. Quintessence Int. 2019, 52, 538–550. [Google Scholar]
- Pozzi, A.; Arcuri, L.; Moy, P.K. The smiling scan technique: Facially driven guided surgery and prosthetics. J. Prosthodont. Res. 2018, 62, 514–517. [Google Scholar] [CrossRef]
- Khaohoen, A.; Powcharoen, W.; Sornsuwan, T.; Chaijareenont, P.; Rungsiyakull, C.; Rungsiyakull, P. Accuracy of implant placement with computer-aided static, dynamic, and robot-assisted surgery: A systematic review and meta-analysis of clinical trials. BMC Oral Health. 2024, 24, 359. [Google Scholar] [CrossRef]
- Joda, T.; Brägger, U.; Gallucci, G. Systematic literature review of digital three-dimensional superimposition techniques to create virtual dental patients. Int. J. Oral Maxillofac. Implant. 2015, 30, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Mangano, F.; Veronesi, G. Digital versus Analog Procedures for the Prosthetic Restoration of Single Implants: A Randomized Controlled Trial with 1 Year of Follow-Up. Biomed. Res. Int. 2018, 2, 5325032. [Google Scholar] [CrossRef]
- Patzelt, S.B.M.; Spies, B.C.; Kohal, R.J. CAD/CAM-fabricated implant-supported restorations: A systematic review. Clin. Oral Implant. Res. 2015, 26, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Joda, T.; Gallucci, G.O. The virtual patient in dental medicine. Clin. Oral Implant. Res. 2015, 26, 725–726. [Google Scholar] [CrossRef] [PubMed]
- Pozzi, A.; Tallarico, M.; Moy, P.K. Three-year post-loading results of a randomised, controlled, split-mouth trial comparing implants with different prosthetic interfaces and design in partially posterior edentulous mandibles. Eur. J. Oral Implantol. 2014, 7, 47–61. [Google Scholar]
- Jacobs, R.; Salmon, B.; Codari, M.; Hassan, B.; Bornstein, M.M. Cone beam computed tomography in implant dentistry: Recommendations for clinical use. BMC Oral Health 2018, 18, 88. [Google Scholar] [CrossRef]
- Tahmaseb, A.; Wu, V.; Wismeijer, D.; Coucke, W.; Evans, C. The accuracy of static computer-aided implant surgery: A systematic review and meta-analysis. Clin. Oral Implant. Res. 2018, 29, 416–435. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, C.M.; da Fonseca, P.A.B.; Quezada, M.M.; Marques, T.; Montero, J.; Morton, D.; Correia, A. Analyzing Linear and Angular Deviations After Guided Surgery for Dental Implant Placement: A Preliminary Study. Int. J. Oral Maxillofac. Implant. 2024, 39, 738–744. [Google Scholar] [CrossRef]
- Rodrigues, J.M.M.; Santos, P.L.; Mendonça, G.; Faloni, A.P.S.; Finoti, L.S.; Margonar, R. Assessment of Deviations of Implants Installed with Prototyped Surgical Guide and Conventional Guide: In Vitro Study. Eur. J. Dent. 2023, 17, 39–45. [Google Scholar] [CrossRef]
- Joda, T.; Zitzmann, N.U. Personalized workflows in reconstructive dentistry-current possibilities and future opportunities. Clin. Oral Investig. 2022, 26, 4283–4290. [Google Scholar] [CrossRef]
- Lin, P.; Su, K. Biomechanical Design Application on the Effect of Different Occlusion Conditions on Dental Implants with Different Positions—A Finite Element Analysis. Appl. Sci. 2020, 10, 5826. [Google Scholar] [CrossRef]
- Alharbi, M.T.; Almutiq, M.M. Prediction of Dental Implants Using Machine Learning Algorithms. J. Healthc. Eng. 2022, 20, 7307675. [Google Scholar] [CrossRef]
- Mai, H.N.; Dam, V.V.; Lee, D.H. Accuracy of Augmented Reality-Assisted Navigation in Dental Implant Surgery: Systematic Review and Meta-analysis. J. Med. Internet Res. 2023, 25, e42040. [Google Scholar] [CrossRef]
- Younis, H.; Lv, C.; Xu, B.; Zhou, H.; Du, L.; Liao, L.; Zhao, N.; Long, W.; Elayah, S.A.; Chang, X.; et al. Accuracy of dynamic navigation compared to static surgical guides and the freehand approach in implant placement: A prospective clinical study. Head. Face Med. 2024, 20, 30. [Google Scholar] [CrossRef]
- Sarkar, A.; Hoda, M.M.; Malick, R.; Kumar, A. Surgical Stent Guided Versus Conventional Method of Implant Placement. J. Maxillofac. Oral Surg. 2022, 21, 580–589. [Google Scholar] [CrossRef] [PubMed]
- Naeini, E.N.; De Bruyn, H.; Bronkhorst, E.M.; D’haese, J. Long-Term Effect of Guided Implant Surgery on Clinical Outcomes and Peri-Implantitis of Maxillary Implants-An Observational Cohort Study. J. Clin. Med. 2023, 12, 4432. [Google Scholar] [CrossRef]
- Paradowska-Stolarz, A.; Mikulewicz, M.; Wieckiewicz, M.; Wezgowiec, J. The Influence of Polishing and Artificial Aging on BioMed Amber® Resin’s Mechanical Properties. J. Funct. Biomater. 2023, 14, 254. [Google Scholar] [CrossRef]
- Akdeniz, B.S.; Çarpar, Y.; Çarpar, K.A. Digital three-dimensional planning of orthodontic miniscrew anchorage: A literature review. J. Exp. Clin. Med. 2022, 39, 269–274. [Google Scholar] [CrossRef]
- Wilmes, B. The new Benefit for Aligner Technique to overcome limitations of aligners. J. Aligner Orthod. 2023, 7, 25–38. [Google Scholar]
- Mahmoud, N.R.; Kamal Eldin, M.H.; Diab, M.H.; Mahmoud, O.S.; Fekry, Y.E. Computer guided versus freehand dental implant surgery: Randomized controlled clinical trial. Saudi Dent. J. 2024, 36, 1472–1476. [Google Scholar] [CrossRef]
- Pellegrino, G.; Ferri, A.; Del Fabbro, M.; Prati, C.; Gandolfi, M.G.; Marchetti, C. Dynamic Navigation in Implant Dentistry: A Systematic Review and Meta-analysis. Int. J. Oral Maxillofac. Implant. 2021, 36, 121–140. [Google Scholar] [CrossRef]
- Zinser, M.; Neugebauer, J.; Mischkowski, R.; Karapedian, V.; Kübler, A.; Zöller, J. Comparison of static and dynamic navigation systems for insertion of dental implants. Int. Congr. Ser. 2004, 1268, 1378. [Google Scholar] [CrossRef]
- Wang, F.; Fan, S.; Huang, W.; Shen, Y.; Li, C.; Wu, Y. Dynamic navigation for prosthetically driven zygomatic implant placement in extensive maxillary defects: Results of a prospective case series. Clin. Implant. Dent. Relat. Res. 2022, 24, 435–443. [Google Scholar] [CrossRef]
- Wei, S.M.; Zhu, Y.; Wei, J.X.; Zhang, C.N.; Shi, J.Y.; Lai, H.C. Accuracy of dynamic navigation in implant surgery: A systematic review and meta-analysis. Clin. Oral Implant. Res. 2021, 32, 383–393. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Ye, M.; Wu, M.; Chen, X.; Shen, S. A retrospective study of dynamic navigation system-assisted implant placement. BMC Oral Health 2023, 23, 759. [Google Scholar] [CrossRef]
- Bhalerao, A.; Ayoub, A.; Usman, A. Application of Dynamic Navigation, Virtual Reality, and Universal Robot in Dental Implantology. Indian J. Dent. Res. 2024, 35, 361–369. [Google Scholar] [CrossRef]
- Jorba-García, A.; Bara-Casaus, J.J.; Camps-Font, O.; Figueiredo, R.; Valmaseda-Castellón, E. The influence of radiographic marker registration versus a markerless trace registration method on the implant placement accuracy achieved by dynamic computer-assisted implant surgery. An in-vitro study. J. Dent. 2024, 146, 105072. [Google Scholar] [CrossRef]
- Spille, J.; Helmstetter, E.; Kübel, P.; Weitkamp, J.T.; Wagner, J.; Wieker, H.; Naujokat, H.; Flörke, C.; Wiltfang, J.; Gülses, A. Learning Curve and Comparison of Dynamic Implant Placement Accuracy Using a Navigation System in Young Professionals. Dent. J. 2022, 10, 187. [Google Scholar] [CrossRef]
- Mangano, F.; Gandolfi, A.; Luongo, G.; Logozzo, S. Intraoral scanners in dentistry: A review of the current literature. BMC Oral Health 2017, 17, 149. [Google Scholar] [CrossRef]
- Papaspyridakos, P.; Vazouras, K.; Chen, Y.W.; Kotina, E.; Natto, Z.; Kang, K.; Chochlidakis, K. Digital vs Conventional Implant Impressions: A Systematic Review and Meta-Analysis. J. Prosthodont. 2020, 29, 660–678. [Google Scholar] [CrossRef] [PubMed]
- Gianfreda, F.; Pesce, P.; Marcano, E.; Pistilli, V.; Bollero, P.; Canullo, L. Clinical Outcome of Fully Digital Workflow for Single-Implant-Supported Crowns: A Retrospective Clinical Study. Dent. J. 2022, 10, 139. [Google Scholar] [CrossRef]
- Fu, X.J.; Liu, M.; Shi, J.Y.; Deng, K.; Lai, H.C.; Gu, W.; Zhang, X.M. Comparison of Different Intraoral Scanners with Prefabricated Aid on Accuracy and Framework Passive Fit of Digital Complete-Arch Implant Impression: An In Vitro Study. Clin. Oral Implant. Res. 2025, 36, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Arikan, H.; Muhtarogullari, M.; Uzel, S.M.; Guncu, M.B.; Aktas, G.; Marshall, L.S.; Turkyilmaz, I. Accuracy of digital impressions for implant-supported complete-arch prosthesis when using an auxiliary geometry device. J. Dent. Sci. 2023, 18, 808–813. [Google Scholar] [CrossRef]
- Emam, M.; Ghanem, L.; Abdel Sadek, H.M. Effect of different intraoral scanners and post-space depths on the trueness of digital impressions. Dent. Med. Probl. 2024, 61, 577–584. [Google Scholar] [CrossRef]
- Martins, J.N.R.; Pinto, R.; Silva, E.J.N.L.; Simões-Carvalho, M.; Marques, D.; Martins, R.F.; Versiani, M.A. 3D Surface Scanning—A Novel Protocol to Characterize Virtual Nickel–Titanium Endodontic Instruments. Materials 2023, 16, 3636. [Google Scholar] [CrossRef] [PubMed]
- Deeb, J.G.; Reddy, N.G.; Hopfensperger, L.J.; Harris, A.L.; Bencharit, S. Same-Day Digital Dentistry Restorative Workflow for Single Immediate Provisionalization of Narrow-Diameter Implants: An Exploratory Prospective Study. Prosthesis 2023, 5, 197–207. [Google Scholar] [CrossRef]
- de Oliveira, N.R.C.; Pigozzo, M.N.; Sesma, N.; Laganá, D.C. Clinical efficiency and patient preference of digital and conventional workflow for single implant crowns using immediate and regular digital impression: A meta-analysis. Clin. Oral Implant. Res. 2020, 31, 669–686. [Google Scholar] [CrossRef] [PubMed]
- Flügge, T.; Kramer, J.; Nelson, K.; Nahles, S.; Kernen, F. Digital implantology—A review of virtual planning software for guided implant surgery. Part. II: Prosthetic set-up and virtual implant planning. BMC Oral Health 2022, 22, 23. [Google Scholar]
- Schubert, O.; Schweiger, J.; Stimmelmayr, M.; Nold, E.; Güth, J.F. Digital implant planning and guided implant surgery–workflow and reliability. Br. Dent. J. 2019, 226, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Binhuraib, H.; Alreshidi, F.; Bardi, S.; Alghamdi, N.; Alhelali, S.; Althagafi, T.; Alsayegh, R.; Alsalem, F.; Aljathnan, A.; Alshahrani, N.; et al. Evaluating the efficiency of complete digital workflow in prosthodontics. J. Healthc. Sci. 2023, 3, 695–700. [Google Scholar] [CrossRef]
- Bessadet, M.; Auduc, C.; Drancourt, N.; Nicolas, E.; El Osta, N. Comparative analyses of time efficiency and cost in fabricating fixed implant-supported prostheses in digital, hybrid, and conventional workflows: A systematic review and meta-analysis. J. Prosthet. Dent. 2024, 133, 689–712. [Google Scholar] [CrossRef]
- Rutkūnas, V.; Gečiauskaitė, A.; Jegelevičius, D.; Vaitiekūnas, M. Accuracy of digital implant impressions with intraoral scanners. A systematic review. . Eur. J. Oral Implantol. 2017, 10, 101–120. [Google Scholar]
- Ahmed, S.; Hawsah, A.; Rustom, R.; Alamri, A.; Althomairy, S.; Alenezi, M.; Shaker, S.; Alrawsaa, F.; Althumairy, A.; Alteraigi, A. Digital Impressions Versus Conventional Impressions in Prosthodontics: A Systematic Review. Cureus 2024, 16, e51537. [Google Scholar] [CrossRef]
- Panahi, O. Dental Implants & the Rise of AI. On J. Dent. Oral Health 2024, 8, 1–4. [Google Scholar]
- Altalhi, A.M.; Alharbi, F.S.; Alhodaithy, M.A.; Almarshedy, B.S.; Al-Saaib, M.Y.; Al Jfshar, R.M.; Aljohani, A.S.; Alshareef, A.H.; Muhayya, M.; Al-Harbi, N.H. The Impact of Artificial Intelligence on Dental Implantology: A Narrative Review. Cureus 2023, 15, e47941. [Google Scholar] [CrossRef]
- Marques, S.; Ribeiro, P.; Falcão, C.; Lemos, B.F.; Ríos-Carrasco, B.; Ríos-Santos, J.V.; Herrero-Climent, M. Digital Impressions in Implant Dentistry: A Literature Review. Int. J. Environ. Res. Public. Health 2021, 18, 1020. [Google Scholar] [CrossRef] [PubMed]
- Mahato, M.; Hota, S.; Jain, A.; Dutta, D.; Bhushan, P.; Raut, A. Comparison of Conventional and Digital Workflows in the Fabrication of Fixed Prostheses: A Systematic Review. Cureus 2024, 16, e61764. [Google Scholar] [CrossRef] [PubMed]
- Mizumoto, R.M.; Yilmaz, B. Intraoral scan bodies in implant dentistry: A systematic review. J. Prosthet. Dent. 2018, 120, 343–352. [Google Scholar] [CrossRef]
- Wu, H.K.; Chen, G.; Huang, X.; Deng, F.; Li, Y. Accuracy of single-implant digital impression with various scanbody exposure levels at anterior and posterior regions. J. Dent. 2023, 138, 104641. [Google Scholar] [CrossRef]
- Jasim, A.G.; Abo Elezz, M.G.; Altonbary, G.Y.; Elsyad, M.A. Accuracy of digital and conventional implant-level impression techniques for maxillary full-arch screw-retained prosthesis: A crossover randomized trial. Clin. Implant. Dent. Relat. Res. 2024, 26, 714–723. [Google Scholar] [CrossRef] [PubMed]
- Róth, I.; Hermann, P.; Vitai, V.; Joós-Kovács, G.L.; Géczi, Z.; Borbély, J. Comparison of the learning curve of intraoral scanning with two different intraoral scanners based on scanning time. BMC Oral Health 2023, 23, 267. [Google Scholar] [CrossRef]
- Peñarrocha-Oltra, D.; Agustín-Panadero, R.; Bagán, L.; Giménez, B.; Peñarrocha, M. Impression of multiple implants using photogrammetry: Description of technique and case presentation. Med. Oral Patol. Oral Cir. Bucal 2014, 19, 66–71. [Google Scholar] [CrossRef]
- Azevedo, L.; Molinero-Mourelle, P.; Antonaya, J.L.; Río, J.; Correia, A.; Gomez-Polo, M. Photogrammetry Technique for the 3D Digital Impression of Multiple Dental Implants. In VipIMAGE 2019; Springer: Cham, Switzerland, 2019. [Google Scholar]
- Joda, T.; Ferrari, M.; Gallucci, G.O.; Wittneben, J.G.; Brägger, U. Digital technology in fixed implant prosthodontics. Periodontol. 2000 2017, 73, 178–192. [Google Scholar] [CrossRef]
- Smith, D.G.; Burgess, E.M. The use of CAD/CAM technology in prosthetics and orthotics--current clinical models and a view to the future. J. Rehabil. Res. Dev. 2001, 38, 27–34. [Google Scholar]
- Cai, H.; Xu, X.; Lu, X.; Zhao, M.; Jia, Q.; Jiang, H.B.; Kwon, J.S. Dental Materials Applied to 3D and 4D Printing Technologies: A Review. Polymers 2023, 15, 2405. [Google Scholar] [CrossRef] [PubMed]
- Rexhepi, I.; Santilli, M.; D’Addazio, G.; Tafuri, G.; Manciocchi, E.; Caputi, S.; Sinjari, B. Clinical Applications and Mechanical Properties of CAD-CAM Materials in Restorative and Prosthetic Dentistry: A Systematic Review. J. Funct. Biomater. 2023, 14, 431. [Google Scholar] [CrossRef]
- Hanna, E.G.; Amine, S.; Prasad, B.; Younes, K. Exploring polyetheretherketone in dental implants and abutments: A focus on biomechanics and finite element methods. Rev. Adv. Mater. Sci. 2024, 63, 20240031. [Google Scholar] [CrossRef]
- Sanz-Sánchez, I.; Molina, A.; Martin, C.; Bollain, J.; Calatrava, J.; Sanz, M. The effect of one-time abutment placement on clinical and radiographic outcomes: A 5-year randomized clinical trial. Clin. Oral Implant. Res. 2024, 35, 609–620. [Google Scholar] [CrossRef]
- Bernauer, S.A.; Zitzmann, N.U.; Joda, T. The Complete Digital Workflow in Fixed Prosthodontics Updated: A Systematic Review. Healthcare 2023, 11, 679. [Google Scholar] [CrossRef]
- Corsalini, M.; Barile, G.; Ranieri, F.; Morea, E.; Corsalini, T.; Capodiferro, S.; Palumbo, R.R. Comparison between Conventional and Digital Workflow in Implant Prosthetic Rehabilitation: A Randomized Controlled Trial. J. Funct. Biomater. 2024, 15, 149. [Google Scholar] [CrossRef] [PubMed]
- Revilla-León, M.; Gómez-Polo, M.; Vyas, S.; Barmak, B.A.; Galluci, G.O.; Att, W.; Krishnamurthy, V.R. Artificial intelligence applications in implant dentistry: A systematic review. J. Prosthet. Dent. 2023, 129, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Nazari, Y.; Lngeroodi, P.F.; Maddahi, M.; Kobravi, S.; Amin, M.R.; Bargrizaneh, A.A.; Fouladi, S. Artificial intelligence models and predicting implant success. Biomed. Res. Ther. 2025, 12, 7029–7038. [Google Scholar] [CrossRef]
- Ahn, S.; Kim, J.; Baek, S.; Kim, C.; Jang, H.; Lee, S. Toward Digital Twin Development for Implant Placement Planning Using a Parametric Reduced-Order Model. Bioengineering 2024, 11, 84. [Google Scholar] [CrossRef]
- Chen, Y.W.; Stanley, K.; Att, W. Artificial intelligence in dentistry: Current applications and future perspectives. Quintessence Int. 2020, 51, 248–257. [Google Scholar] [PubMed]
- Alqutaibi, A.Y.; Algabri, R.S.; Alamri, A.S.; Alhazmi, L.S.; Almadani, S.M.; Alturkistani, A.M.; Almutairi, A.G. Advancements of artificial intelligence algorithms in predicting dental implant prognosis from radiographic images: A systematic review. J. Prosthet. Dent. 2024. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kafedzhieva, A.; Vlahova, A.; Chuchulska, B. Digital Technologies in Implantology: A Narrative Review. Bioengineering 2025, 12, 927. https://doi.org/10.3390/bioengineering12090927
Kafedzhieva A, Vlahova A, Chuchulska B. Digital Technologies in Implantology: A Narrative Review. Bioengineering. 2025; 12(9):927. https://doi.org/10.3390/bioengineering12090927
Chicago/Turabian StyleKafedzhieva, Ani, Angelina Vlahova, and Bozhana Chuchulska. 2025. "Digital Technologies in Implantology: A Narrative Review" Bioengineering 12, no. 9: 927. https://doi.org/10.3390/bioengineering12090927
APA StyleKafedzhieva, A., Vlahova, A., & Chuchulska, B. (2025). Digital Technologies in Implantology: A Narrative Review. Bioengineering, 12(9), 927. https://doi.org/10.3390/bioengineering12090927





