A Novel Protocol for Integrated Assessment of Upper Limbs Using the Optoelectronic Motion Analysis System: Validation and Usability in Healthy People
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Data Collections
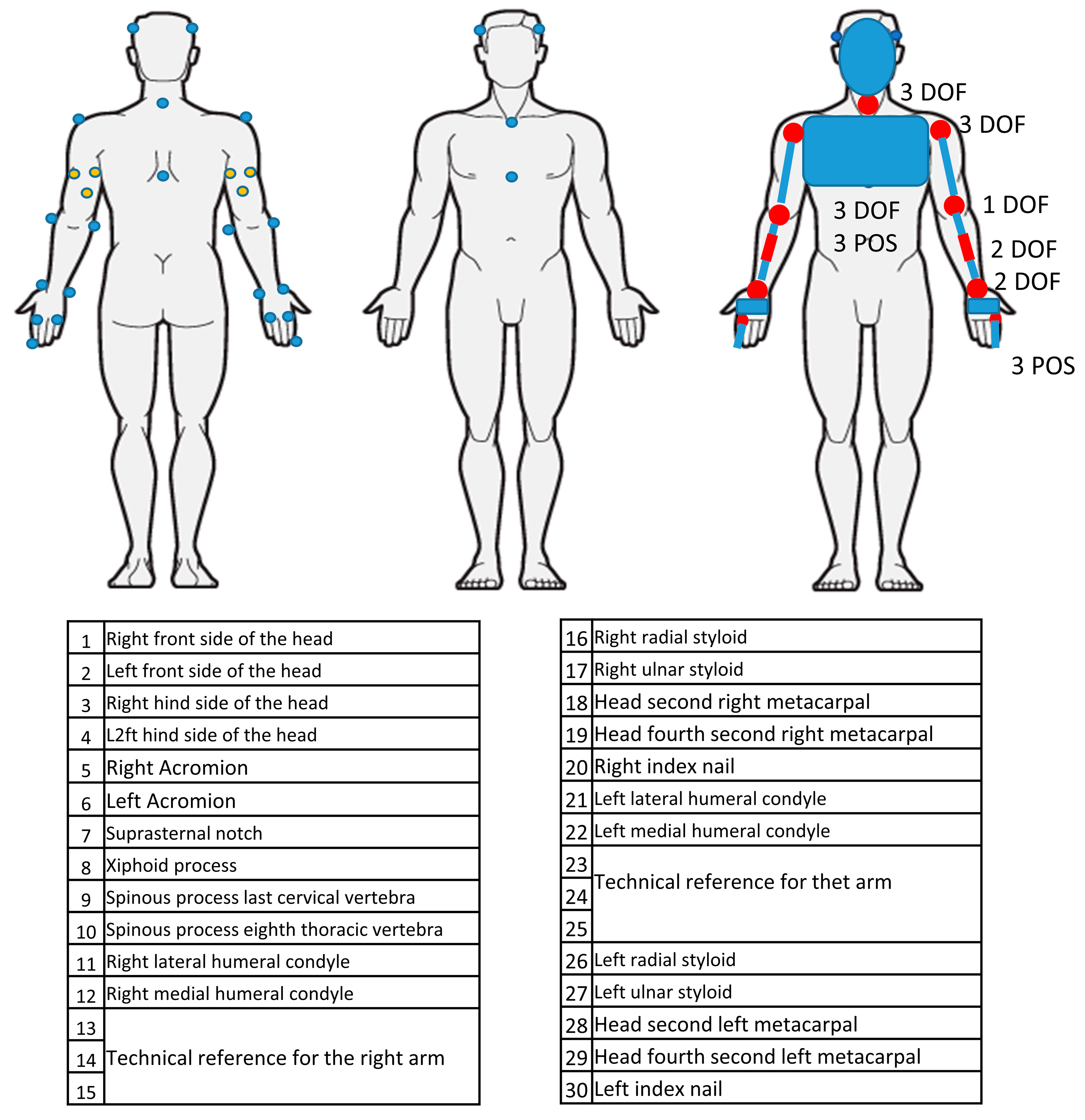
- Preparation of the subject, positioning the markers (30) on the selected body landmarks.
- The acquisition of 5 HtT trials with each arm;
- Detachment of the markers from the subject.
- Phase 1: instrument preparation and marker preparation (for the test subjects, the part of instrument preparation was excluded from the questionnaire because the subjects were not involved in this operation);
- Phase 2: marker positioning;
- Phase 3: recording the movements with the optoelectronic system;
- Phase 4: removing the markers.
2.3. Data Processing
- Going Phase: Begins when the velocity of the finger marker exceeds 50 mm/s and ends at the onset of the Adjusting Phase.
- Adjusting Phase: This phase was not defined by velocity thresholds, due to the fluctuating nature of speed during fine adjustments. Instead, it was identified based on the distance between the finger and the target, using a threshold set as the average distance plus three standard deviations across five movements. The end of this phase and the start of the Returning Phase were defined using the same criteria.
- Returning Phase: Starts at the conclusion of the Adjusting Phase and ends when the finger marker speed drops below 50 mm/s.
- End-point (finger) metrics: Derived from finger kinematic data, these metrics assess movement speed, accuracy, efficiency, and smoothness.
- Trunk compensation metrics: Calculated from the marker placed on the sternum, these metrics quantified compensatory trunk movements during reaching tasks.
2.4. Statistical Analysis
3. Results
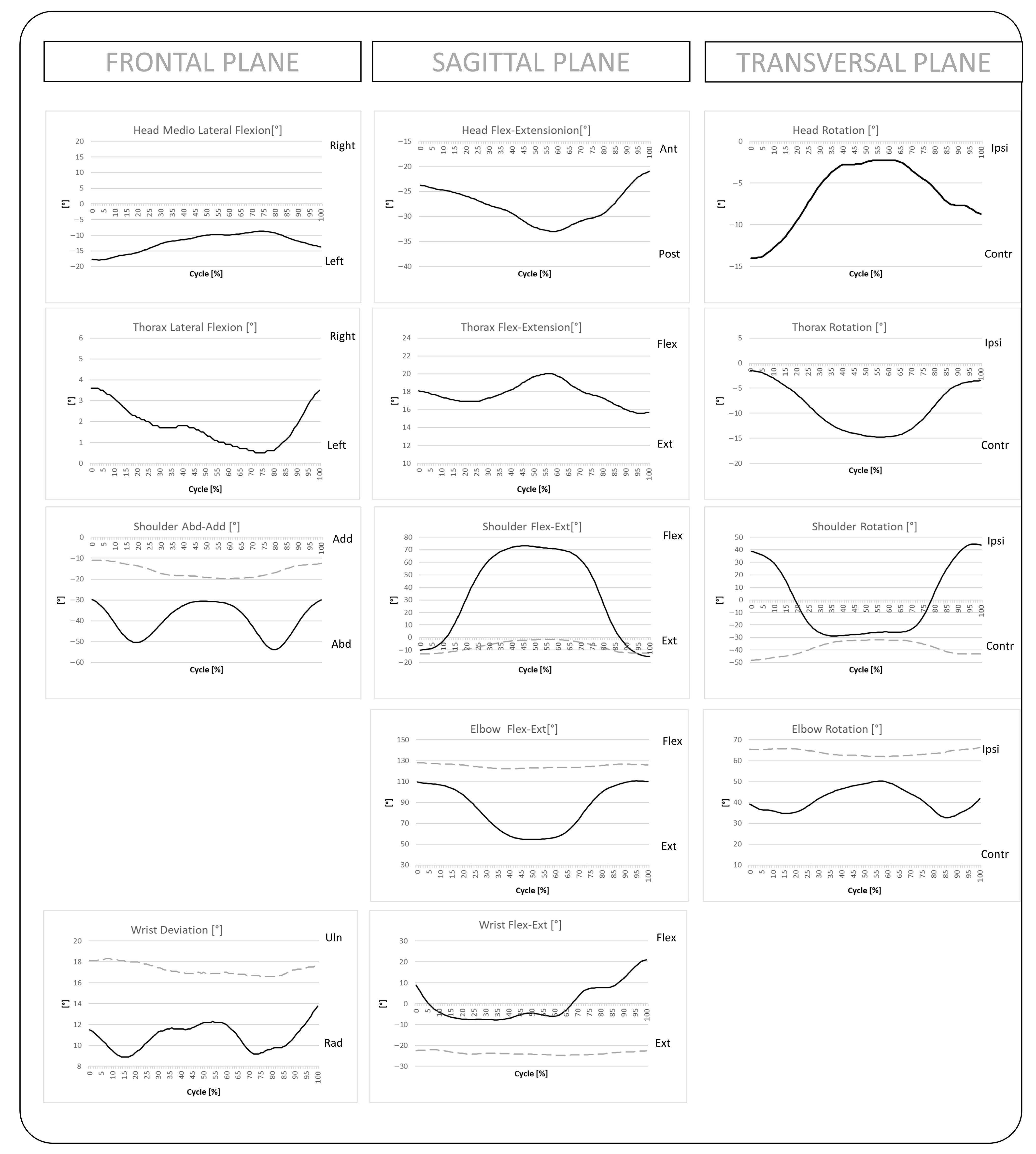
3.1. Time Phases and End-Point Metrics
3.2. Trunk Compensation Metrics
3.3. Range of Motion
3.4. Usability Assessment
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| UL | Upper Limb |
| CP | Cerebral Palsy |
| MUUL | The Melbourne Assessment of Unilateral Upper Limb Function |
| QUEST | Quality of Upper Extremity Skills Test |
| SHUEE | Shriners Hospital Upper Extremity Evaluation |
| CIMT | Constraint-Induced Movement Therapy |
| TRT | Trunk Restraint Therapy |
| BAT | Bilateral Arm Therapy |
| ICF | International Classification of Functioning |
| ISB | International Society of Biomechanics |
| HtT | Hit-to-Target |
| SUS | System Usability Scale |
| ROM | Range of motion |
| ICC | intraclass correlation coefficient |
| MMV | Mean Movement Velocity |
| IC | Index of curvature |
| NVP | Number of Velocity Peaks |
| MA | motion analysis |
References
- Langhorne, P.; Bernhardt, J.; Kwakkel, G. Stroke rehabilitation. Lancet 2011, 377, 1693–1702. [Google Scholar] [CrossRef] [PubMed]
- Francisco-Martínez, C.; Prado-Olivarez, J.; Padilla-Medina, J.A.; Díaz-Carmona, J.; Pérez-Pinal, F.J.; Barranco-Gutiérrez, A.I.; Martínez-Nolasco, J.J. Upper limb movement measurement systems for cerebral palsy: A systematic literature review. Sensors 2021, 21, 7884. [Google Scholar] [CrossRef]
- Santisteban, L.; Térémetz, M.; Bleton, J.P.; Baron, J.C.; Maier, M.A.; Lindberg, P.G. Upper limb outcome measures used in stroke rehabilitation studies: A systematic literature review. PLoS ONE 2016, 11, e0154792. [Google Scholar] [CrossRef]
- Lemmens, R.J.M.; Timmermans, A.A.A.; Janssen-Potten, Y.J.M.; Smeets, R.J.E.M.; Seelen, H.A.M. Valid and reliable instruments for arm-hand assessment at ICF activity level in persons with hemiplegia: A systematic review. BMC Neurol. 2012, 12, 21. [Google Scholar] [CrossRef]
- Demers, M.; Levin, M.F. Do Activity Level Outcome Measures Commonly Used in Neurological Practice Assess Upper-Limb Movement Quality? Neurorehabil. Neural Repair 2017, 31, 623–637. [Google Scholar] [CrossRef]
- Ashford, S.; Slade, M.; Malaprade, F.; Turner-Stokes, L. Evaluation of functional outcome measures for the hemiparetic upper limb: A systematic review. J. Rehabil. Med. 2008, 40, 787–795. [Google Scholar] [CrossRef]
- Murphy, M.A.; Resteghini, C.; Feys, P.; Lamers, I. An overview of systematic reviews on upper extremity outcome measures after stroke. BMC Neurol. 2015, 15, 29. [Google Scholar] [CrossRef]
- Koman, L.A.; Smith, B.P.; Shilt, J.S. Cerebral palsy. Lancet 2004, 363, 1619–1631. [Google Scholar] [CrossRef]
- Bourke-Taylor, H. Melbourne Assessment of Unilateral Upper Limb Function: Construct validity and correlation with the Pediatric Evaluation of Disability Inventory. Dev. Med. Child Neurol. 2003, 45, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Haga, N.; van der Heijden-Maessen, H.C.; van Hoorn, J.F.; Boonstra, A.M.; Hadders-Algra, M. Test-Retest and Inter- and Intrareliability of the Quality of the Upper-Extremity Skills Test in Preschool-Age Children With Cerebral Palsy. Arch. Phys. Med. Rehabil. 2007, 88, 1686–1689. [Google Scholar] [CrossRef]
- Davids, J.R.; Peace, L.C.; Wagner, L.V.; Gidewall, M.A.; Blackhurst, D.W.; Roberson, W.M. Validation of the Shriners Hospital for Children Upper Extremity Evaluation (SHUEE) for children with hemiplegic cerebral palsy. J. Bone Jt. Surg. 2006, 88, 326–333. [Google Scholar] [CrossRef]
- Bard, R.; Chaléat-Valayer, E.; Combey, A.; Bleu, P.E.; Perretant, I.; Bernard, J.C. Upper limb assessment in children with cerebral palsy: Translation and reliability of the French version for the Melbourne unilateral upper limb assessment (test de Melbourne). Ann. Phys. Rehabil. Med. 2009, 52, 297–310. [Google Scholar] [CrossRef]
- Rohafza M, Fluet GG, Qiu Q, Adamovich S. Correlation of reaching and grasping kinematics and clinical measures of upper extremity function in persons with stroke related hemiplegi. Physiol. Behav. 2017, 176, 139–148. [Google Scholar] [CrossRef]
- Kwakkel, G.; Lannin, N.A.; Borschmann, K.; English, C.; Ali, M.; Churilov, L.; Saposnik, G.; Winstein, C.; Van Wegen, E.E.H.; Wolf, S.L.; et al. Standardized Measurement of Sensorimotor Recovery in Stroke Trials: Consensus-Based Core Recommendations from the Stroke Recovery and Rehabilitation Roundtable. Neurorehabil. Neural Repair 2017, 31, 784–792. [Google Scholar] [CrossRef] [PubMed]
- Mesquita, I.A.; Fonseca, P.F.P.d.; Borgonovo-Santos, M.; Ribeiro, E.; Pinheiro, A.R.V.; Correia, M.V.; Silva, C. Comparison of upper limb kinematics in two activities of daily living with different handling requirements. Hum. Mov. Sci. 2020, 72, 102632. [Google Scholar] [CrossRef] [PubMed]
- Karakostas, T.; Watters, K.; King, E.C. Assessment of the Spastic Upper Limb with Computational Motion Analysis. Hand Clin. 2018, 34, 445–454. [Google Scholar] [CrossRef]
- Jaspers, E.; Desloovere, K.; Bruyninckx, H.; Molenaers, G.; Klingels, K.; Feys, H. Review of quantitative measurements of upper limb movements in hemiplegic cerebral palsy. Gait Posture 2009, 30, 395–404. [Google Scholar] [CrossRef]
- De Los Reyes-Guzmán, A.; Dimbwadyo-Terrer, I.; Trincado-Alonso, F.; Monasterio-Huelin, F.; Torricelli, D.; Gil-Agudo, A. Quantitative assessment based on kinematic measures of functional impairments during upper extremity movements: A review. Clin. Biomech. 2014, 29, 719–727. [Google Scholar] [CrossRef]
- Alt Murphy, M.; Häger, C.K. Kinematic analysis of the upper extremity after stroke–how far have we reached and what have we grasped? Phys. Ther. Rev. 2015, 20, 137–155. [Google Scholar] [CrossRef]
- Salter, K.; Jutai, J.W.; Teasell, R.; Foley, N.C.; Bitensky, J.; Bayley, M. Issues for selection of outcome measures in stroke rehabilitation: ICF activity. Disabil. Rehabil. 2005, 27, 315–340. [Google Scholar] [CrossRef]
- Butler, E.E.; Ladd, A.L.; Louie, S.A.; LaMont, L.E.; Wong, W.; Rose, J. Three-dimensional kinematics of the upper limb during a Reach and Grasp Cycle for children. Gait Posture 2010, 32, 72–77. [Google Scholar] [CrossRef]
- Artilheiro, M.C.; Corrêa, J.C.F.; Cimolin, V.; Lima, M.O.; Galli, M.; de Godoy, W.; Lucareli, P.R.G. Three-dimensional analysis of performance of an upper limb functional task among adults with dyskinetic cerebral palsy. Gait Posture 2014, 39, 875–881. [Google Scholar] [CrossRef]
- Wu, G.; Van Der Helm, F.C.T.; Veeger, H.E.J.; Makhsous, M.; Van Roy, P.; Anglin, C.; Nagels, J.; Karduna, A.R.; McQuade, K.; Wang, X.; et al. ISB recommendation on definitions of joint coordinate systems of various joints for the reporting of human joint motion—Part II: Shoulder, elbow, wrist and hand. J. Biomech. 2005, 38, 981–992. [Google Scholar] [CrossRef]
- Milanese, C.; Cavedon, V.; Corte, S.; Agostini, T. The effects of two different correction strategies on the snatch technique in weightlifting. J. Sports Sci. 2017, 35, 476–483. [Google Scholar] [CrossRef]
- Kontaxis, A.; Cutti, A.G.; Johnson, G.R.; Veeger, H.E.J. A framework for the definition of standardized protocols for measuring upper-extremity kinematics. Clin. Biomech. 2009, 24, 246–253. [Google Scholar] [CrossRef] [PubMed]
- Andreoni, G.; Negrini, S.; Ciavarro, G.L.; Santambrogio, G.C. ZooMS: A non invasive analysis of global and metameric movement of the lumbar spine. Eura Medicophys 2005, 41, 7–16. [Google Scholar]
- Zapata, B.C.; Fernández-Alemán, J.L.; Idri, A.; Toval, A. Empirical Studies on Usability of mHealth Apps: A Systematic Literature Review. J. Med. Syst. 2015, 39, 1. [Google Scholar] [CrossRef] [PubMed]
- ISO 9241-11; 2018 Ergonomics of Human-System Interactionpart 11: Usability: Definitions and Concepts. ISO/IEC: Geneva, Switzerland, 2018.
- Friesen, E.L. Measuring at Usability with the Modified System Usability Scale (SUS). Stud. Health Technol. Inform. 2017, 242, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Menegoni, F.; Milano, E.; Trotti, C.; Galli, M.; Bigoni, M.; Baudo, S.; Mauro, A. Quantitative evaluation of functional limitation of upper limb movements in subjects affected by ataxia. Eur. J. Neurol. 2009, 16, 232–239. [Google Scholar] [CrossRef]
- Cimolin, V.; Beretta, E.; Piccinini, L.; Turconi, A.C.; Locatelli, F.; Galli, M.; Strazzer, S. Constraint-induced movement therapy for children with hemiplegia after traumatic brain injury: A quantitative study. J. Head Trauma Rehabil. 2012, 27, 177–187. [Google Scholar] [CrossRef]
- Butler, E.E.; Rose, J. The Pediatric Upper Limb Motion Index and a Temporal-Spatial Logistic Regression: Quantitative Analysis of Upper Limb Movement Disorders during the Reach & Grasp Cycle. Physiol. Behav. 2018, 176, 139–148. [Google Scholar] [CrossRef]
- Beretta, E.; Cesareo, A.; Biffi, E.; Schafer, C.; Galbiati, S.; Strazzer, S. Rehabilitation of upper limb in children with acquired brain injury: A preliminary comparative study. J. Healthc. Eng. 2018, 2018, 4208492. [Google Scholar] [CrossRef] [PubMed]
- Ciavarro, G.L.; Andreoni, G.; Negrini, S.; Santambrogio, G.C. Functional assessment of the lumbar spine through the optoelectronic ZooMS system. Clin. Appl. 2006, 42, 135–143. [Google Scholar]
- Piccinini, L.; Molteni, L.E.; Panzeri, D.; Micheletti, E.; Pintabona, G.; Andreoni, G. Integrated Assessment of Gait and Spinal Kinematics Using Optoelectronic Motion Analysis Systems: Validation and Usability Assessment of a Novel Protocol. Biomechanics 2025, 5, 24. [Google Scholar] [CrossRef]
- Brooke, J. SUS: A “Quick and Dirty” Usability Scale. Usability Eval. Ind. 1995, 189, 207–212. [Google Scholar] [CrossRef]
- Koo, T.K.; Li, M.Y. A Guideline of Selecting and Reporting Intraclass Correlation Coefficients for Reliability Research. J. Chiropr. Med. 2016, 15, 155–163. [Google Scholar] [CrossRef]
- Bangor, A.; Kortum, P.; Miller, J. Determining what individual SUS scores mean: Adding an adjective rating scale. J. Usability Stud. 2009, 4, 114–123. [Google Scholar]
- Ciavarro, G.L.; Tramonte, A.; Fusca, M.; Santambrogio, G.C.; Andreoni, G. Evaluation of 3D kinematic model of the spine for ergonomic analysis. SAE Tech. Pap. 2004, 113, 2004. [Google Scholar] [CrossRef]

| Baseline Features | |
|---|---|
| Age [y] (mean—SD; range) | 18.69—SD 12.45; 8.0–41.4 |
| Weight [Kg] (mean—SD; range) | 44.5—SD 22.55; 24.00–90.00 |
| Height [cm] (mean—SD; range) | 144.5—SD 18.28; 124.0–167.0 |
| BMI (mean—SD; range) | 20.03—SD 5.65; 15.06–33.06 |
| Thorax coordinate system [23] | Origin: | The origin coincided with suprasternal notch (IJ) |
| Y axis: | The line connecting the midpoint between xiphoid process (PX) and T8 and the midpoint between IJ and C7, pointing upward | |
| Z axis: | The line perpendicular to the plane formed by IJ, C7, and the midpoint between PX and T8, pointing to the right. | |
| X axis: | The common line perpendicular to the Z and Y axis, pointing forwards. | |
| Proximal Humerus (note: This reference frame was used to calculate shoulder movement) [23] | Origin: | The origin coincident with GlenoHomeros (GH) |
| Y axis: | The line connecting GH and the midpoint of lateral elbow (EL) and medial elbow (EM)), pointing to GH. | |
| Z axis: | The common line perpendicular to the Y and Z axis, pointing to the right | |
| X axis: | The line perpendicular to the plane formed by EL, EM, and GH, pointing forward. | |
| Distal Humerus (note: This reference frame was used to calculate elbow movement) [25] | Origin: | coincides with midpoint of the elbow |
| Y axis: | Forearm axis of rotation. | |
| Z axis: | The line perpendicular to the plane formed by Y and the Y axis of the proximal humerus reference frame | |
| X axis: | perpendicular to the plane formed by X and Y, POINTing forward | |
| Forearm coordinate system [23] | Origin: | The origin coincided with ulnar styloid (US). |
| Y axis: | the line connecting US and the midpoint between EL and EM, pointing proximally | |
| Z axis: | The common line perpendicular to the X and Y axis, pointing to the right. | |
| X axis: | The line perpendicular to the plane through US, radial styloid (RS), and the midpoint between EL and EM, pointing forward | |
| Wrist coordinate system [23] | Origin: | The origin of the coordinate systems is located midway between the base and head of second metacarpal. In the transverse plane, it will be at the approximate center of the tubular bone. |
| Y axis: | The line parallel to a line from the center of the distal head of the metacarpal to the midpoint of the base of the metacarpal. | |
| Z axis: | The common line perpendicular to the X and Y axis. | |
| X axis: | The X and Y axis will form a sagittal plane that splits the metacarpal into mirror images |
| Metrics | ||
|---|---|---|
| End-point (finger) | Time | This is calculated as the total time required for completing each task. In addition, the durations of the previous three phases were computed: the going phase, the adjusting phase, and the returning phase [31] |
| Adjusting sway Index (ASI) | It is defined as the length of the 3D path described by the fingernail during the adjusting phase, which is a measure of the adjustments made to reach the final position. This represents an expression of the degree of precision [31]. | |
| Mean Movement Velocity (MMV) | It is computed during the going phase and represents the mean velocity of the fingernail marker [31]. | |
| Index of curvature (IC) | It is calculated as the ratio of the fingernail 3D path length to the linear distance between the initial and the final pointing position and is a representative of movement smoothness during the going phase [32]. | |
| Number of Velocity Peaks (NVP) | It is a quality measure of movement smoothness computed from the speed profile of the finger during the entire movement [32]. | |
| Trunk compensation | Trunk compensation | It is defined as the length 3D path described by the marker placed on the sternum [33] |
| Displacement along z-axis | Displacement of the marker placed on the sternum along the z-axis (towards the target) during the going phase. It quantifies trunk flexion [33]. | |
| Displacement along x-axis | Displacement of the marker placed on the sternum along the x-axis during the going phase. It quantifies trunk lateral bending [33]. |
| Intra Operator | Inter Operator | |||
|---|---|---|---|---|
| ICC | ICC Level | ICC | ICC Level | |
| T tot | 0.785 | ++ | 0.715 | + |
| T Going Phase | 0.738 | + | 0.610 | + |
| T Return Phase | 0.725 | + | 0.552 | + |
| T Adj Phase | 0.839 | ++ | 0.816 | ++ |
| Mean movement velocity (MMV) | 0.896 | ++ | 0.953 | +++ |
| Curvature Index (CI) | 0.898 | ++ | 0.898 | ++ |
| Adjusting sway Index (ASI) | 0.867 | ++ | 0.793 | ++ |
| Number of Velocity Peaks (NVP) | 0.739 | + | 0.721 | + |
| Intra Operator | Inter Operator | |||
|---|---|---|---|---|
| ICC | ICC Level | ICC | ICC Level | |
| Thorax Length Going Phase | 0.815 | ++ | 0.834 | ++ |
| Antero-posterior Going Phase Displacement | 0.818 | ++ | 0.890 | ++ |
| Medio-lateral Going Phase Displacement | 0.768 | ++ | 0.696 | + |
| Thorax Length Adjusting Phase | 0.926 | +++ | 0.834 | ++ |
| Antero-posterior Adjusting Phase Displacement | 0.882 | ++ | 0.707 | + |
| Medio-lateral Adjusting Phase Displacement | 0.956 | +++ | 0.854 | ++ |
| Thorax Length Return Phase | 0.870 | ++ | 0.870 | ++ |
| Antero-posterior Return Phase Displacement | 0.891 | ++ | 0.888 | ++ |
| Medio-lateral Return Phase Displacement | 0.815 | ++ | 0.825 | ++ |
| Intra Operator | Inter Operator | |||
|---|---|---|---|---|
| ICC | ICC Level | ICC | ICC Level | |
| ROM Right Shoulder FE | 0.983 | +++ | 0.992 | +++ |
| ROM Left Shoulder FE | 0.991 | +++ | 0.992 | +++ |
| ROM Right Shoulder AA | 0.906 | +++ | 0.992 | +++ |
| ROM Left Shoulder AA | 0.966 | +++ | 0.958 | +++ |
| ROM Right Shoulder IE | 0.917 | +++ | 0.965 | +++ |
| ROM Left Shoulder IE | 0.949 | +++ | 0.954 | +++ |
| ROM Right Elbow FE | 0.959 | +++ | 0.950 | +++ |
| ROM Left Elbow FE | 0.978 | +++ | 0.953 | +++ |
| ROM Right Elbow PS | 0.755 | ++ | 0.848 | ++ |
| ROM Left Elbow PS | 0.838 | ++ | 0.762 | ++ |
| ROM Right Wrist FE | 0.967 | +++ | 0.826 | ++ |
| ROM Left Wrist FE | 0.949 | +++ | 0.941 | +++ |
| ROM Right Wrist UR | 0.934 | +++ | 0.952 | +++ |
| ROM Left Wrist UR | 0.949 | +++ | 0.940 | +++ |
| ROM Right Thorax FE | 0.869 | ++ | 0.952 | +++ |
| ROM Right Thorax ML | 0.640 | + | 0.872 | ++ |
| ROM Right Thorax IE | 0.768 | ++ | 0.722 | + |
| ROM Right Head FE | 0.705 | + | 0.867 | ++ |
| ROM Right Head ML | 0.813 | ++ | 0.857 | ++ |
| ROM Right Head IE | 0.832 | ++ | 0.798 | ++ |
| Operators | Patient | ||||||
|---|---|---|---|---|---|---|---|
| Phase | 1 | 2 | 3 | 4 | 1 | 2 | 3 |
| Mean | 4.83 | 4.68 | 4.95 | 4.98 | 4.88 | 4.90 | 4.70 |
| Standard deviation | 0.38 | 0.47 | 0.22 | 0.16 | 0.33 | 0.30 | 0.56 |
| Max | 5.00 | 5.00 | 5.00 | 5.00 | 5.00 | 5.00 | 5.00 |
| Min | 4.00 | 4.00 | 4.00 | 4.00 | 4.00 | 4.00 | 3.00 |
| T [min] | |||||
|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | TOT | |
| Mean | 2.00 | 6.05 | 10.13 | 1.80 | 19.97 |
| Standar deviation | 0.16 | 0.62 | 0.54 | 0.16 | 0.69 |
| Max | 2.32 | 7.22 | 11.73 | 2.10 | 21.50 |
| Min | 1.77 | 5.17 | 8.85 | 1.20 | 18.73 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Molteni, L.E.; Piccinini, L.; Panzeri, D.; Micheletti, E.; Andreoni, G. A Novel Protocol for Integrated Assessment of Upper Limbs Using the Optoelectronic Motion Analysis System: Validation and Usability in Healthy People. Bioengineering 2025, 12, 905. https://doi.org/10.3390/bioengineering12090905
Molteni LE, Piccinini L, Panzeri D, Micheletti E, Andreoni G. A Novel Protocol for Integrated Assessment of Upper Limbs Using the Optoelectronic Motion Analysis System: Validation and Usability in Healthy People. Bioengineering. 2025; 12(9):905. https://doi.org/10.3390/bioengineering12090905
Chicago/Turabian StyleMolteni, Luca Emanuele, Luigi Piccinini, Daniele Panzeri, Ettore Micheletti, and Giuseppe Andreoni. 2025. "A Novel Protocol for Integrated Assessment of Upper Limbs Using the Optoelectronic Motion Analysis System: Validation and Usability in Healthy People" Bioengineering 12, no. 9: 905. https://doi.org/10.3390/bioengineering12090905
APA StyleMolteni, L. E., Piccinini, L., Panzeri, D., Micheletti, E., & Andreoni, G. (2025). A Novel Protocol for Integrated Assessment of Upper Limbs Using the Optoelectronic Motion Analysis System: Validation and Usability in Healthy People. Bioengineering, 12(9), 905. https://doi.org/10.3390/bioengineering12090905








