Triple Encapsulation and Controlled Release of Vancomycin, Rifampicin and Silver from Poly (Methyl Methacrylate) or Poly (Lactic-Co-Glycolic Acid) Nanofibers
Abstract
1. Introduction
2. Materials and Methods
2.1. Drug Release
2.2. Drug Analysis
3. Results
3.1. Nanofiber Characteristics
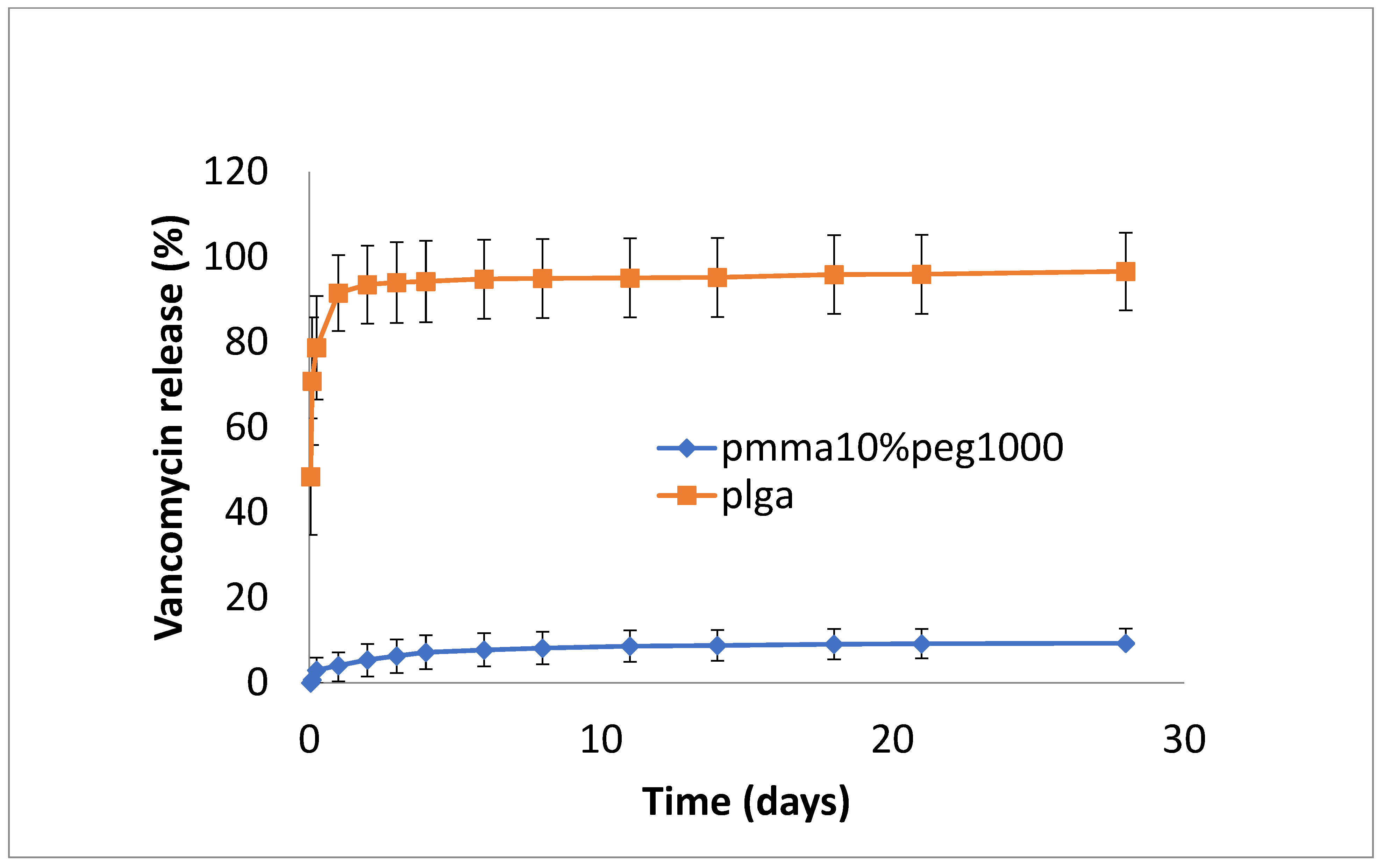
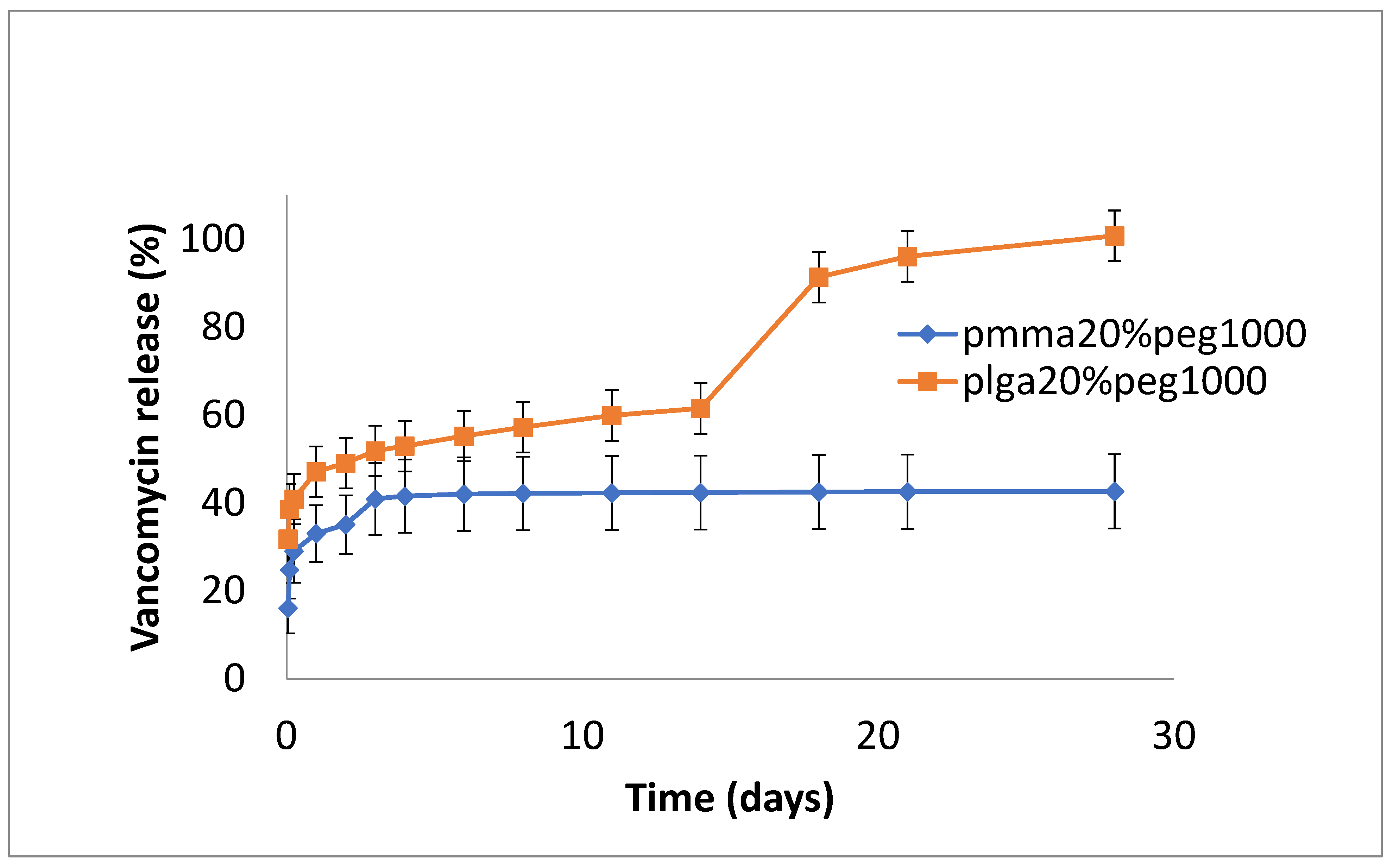
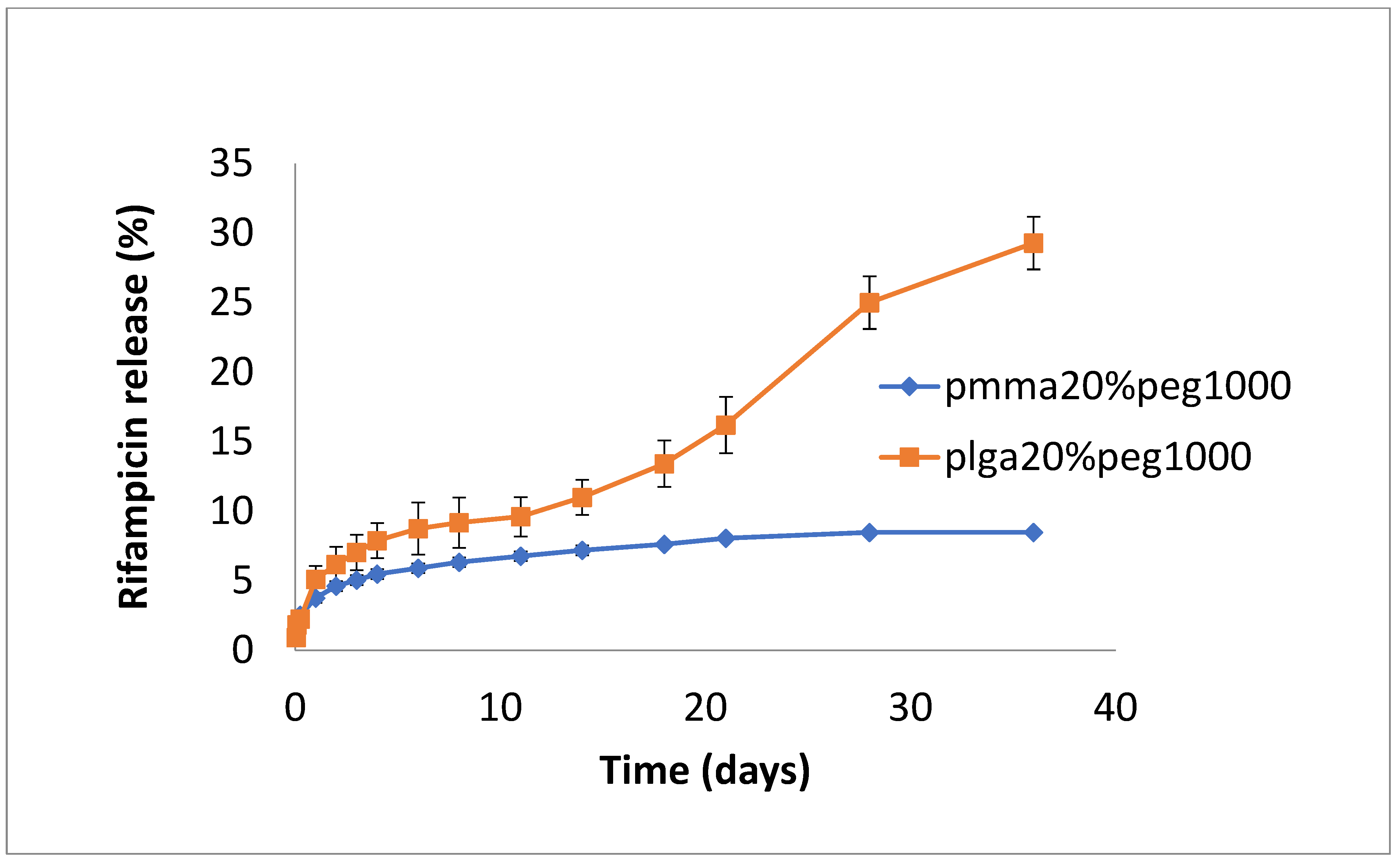
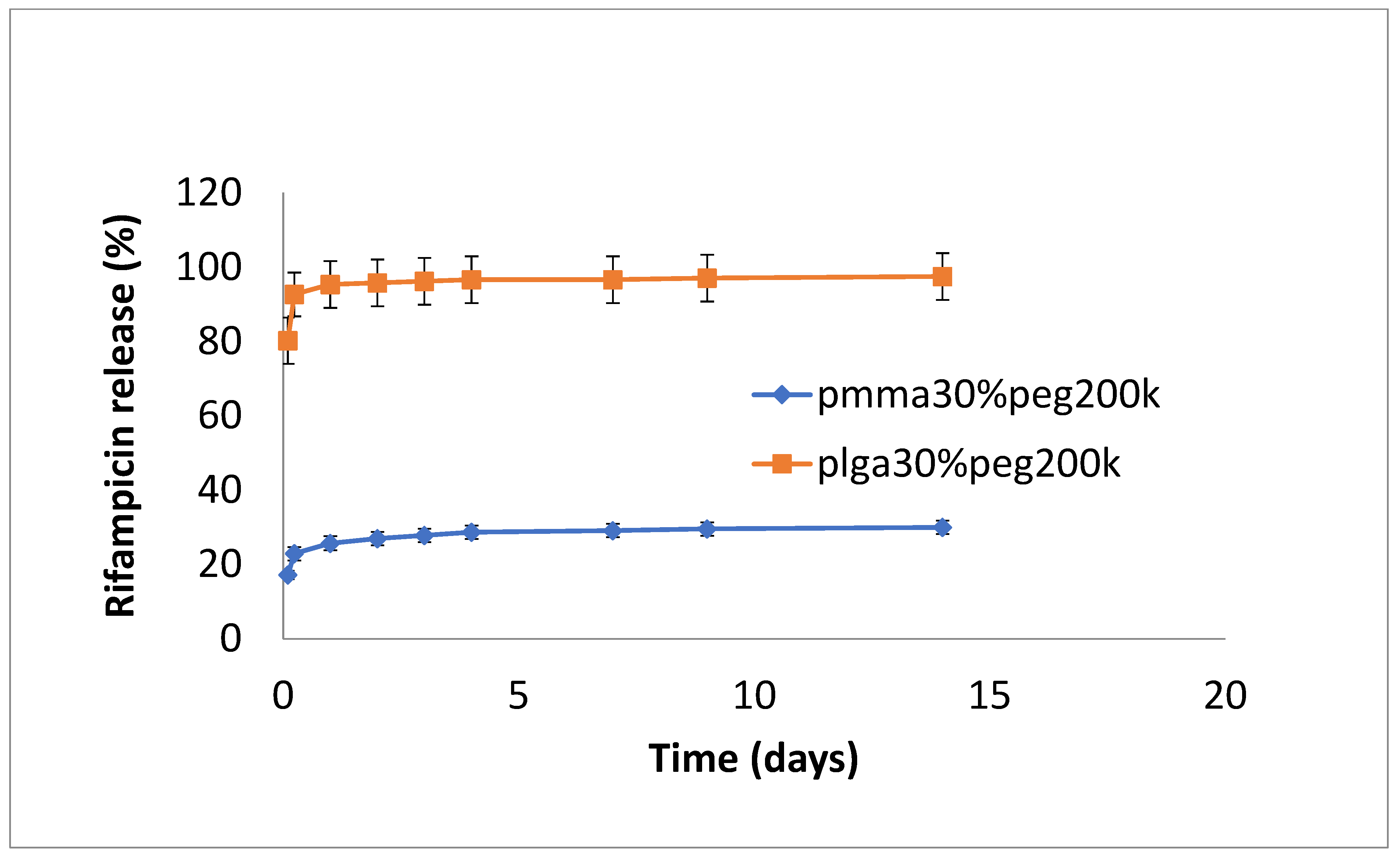
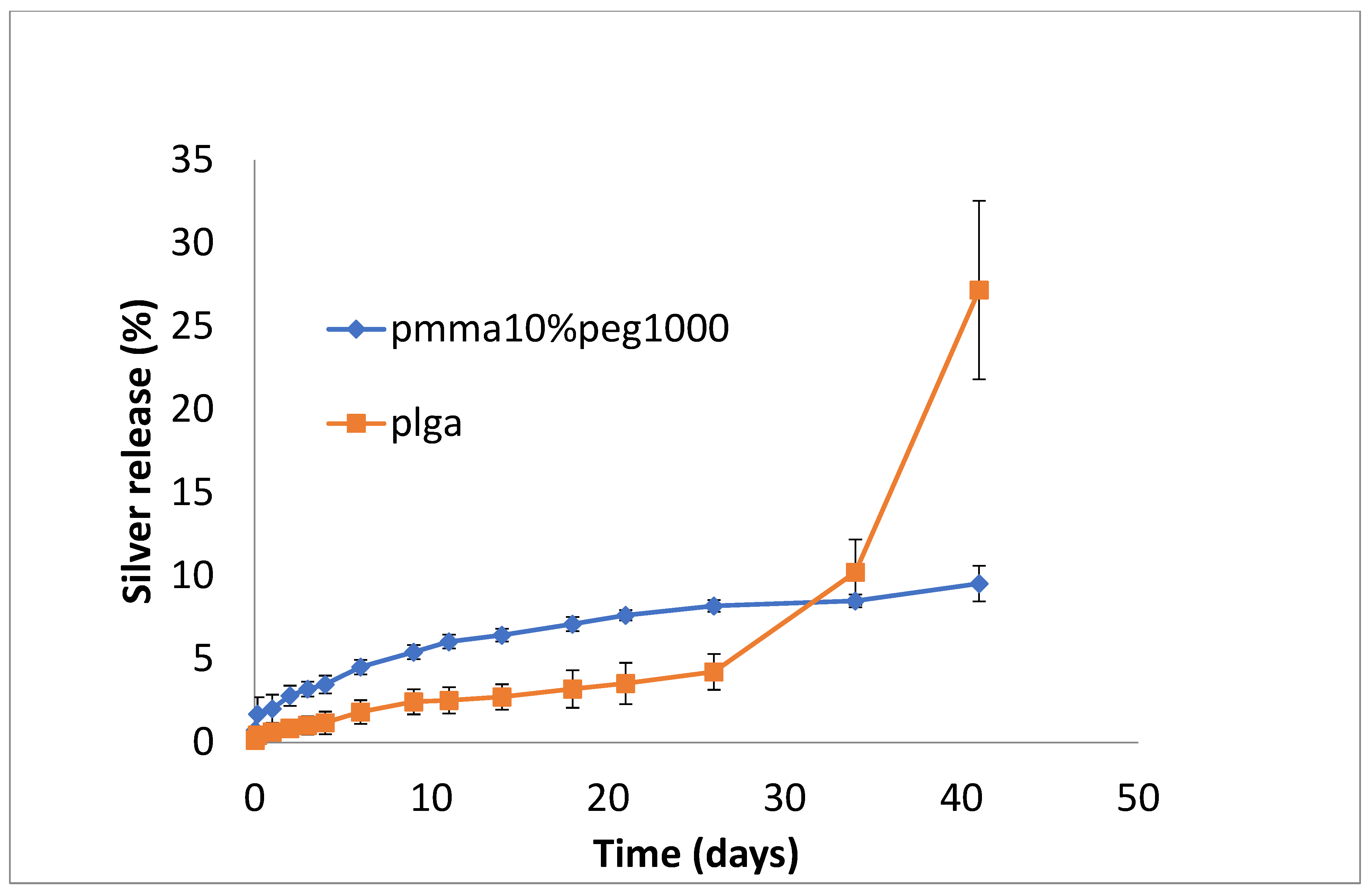
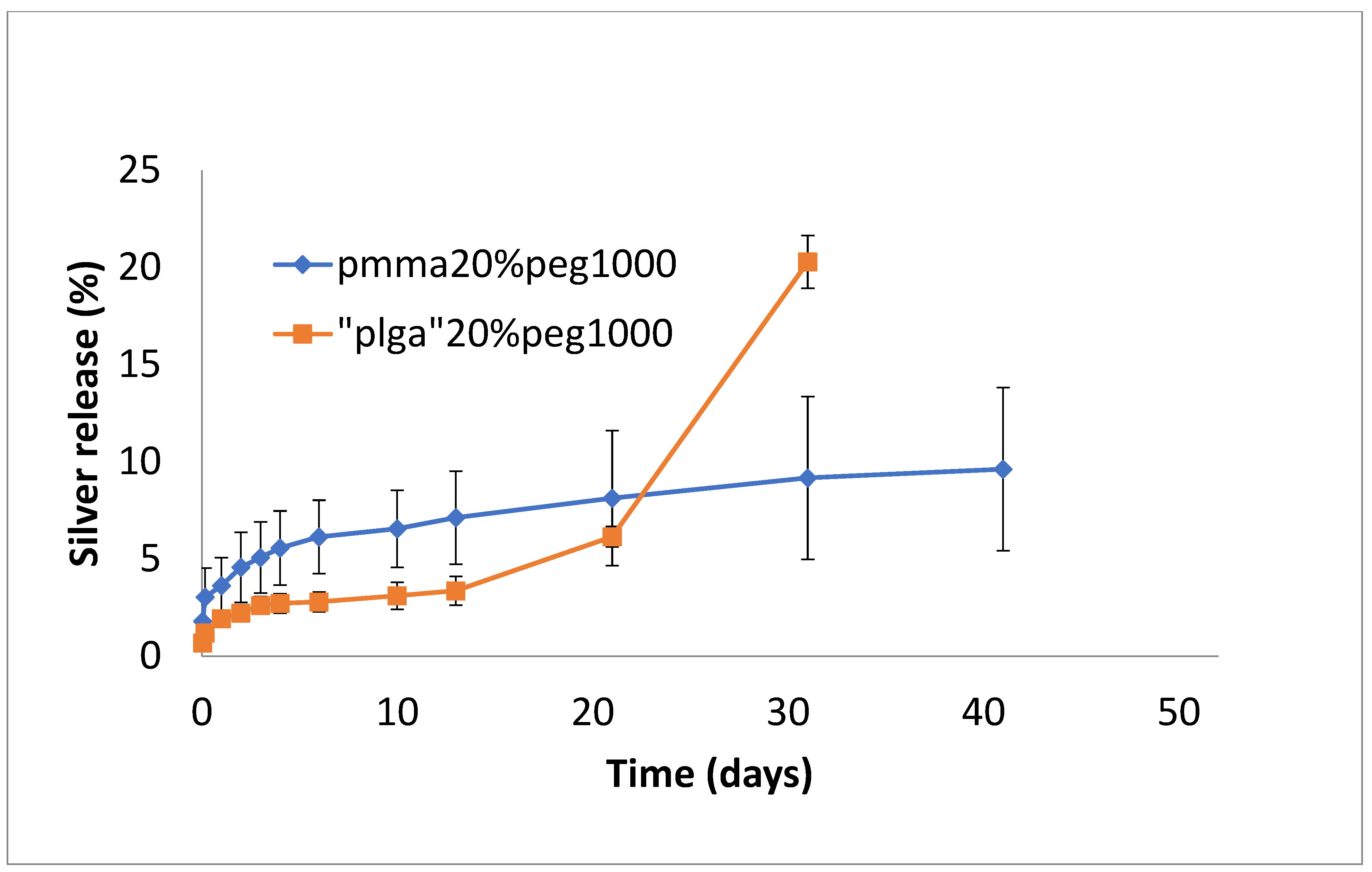
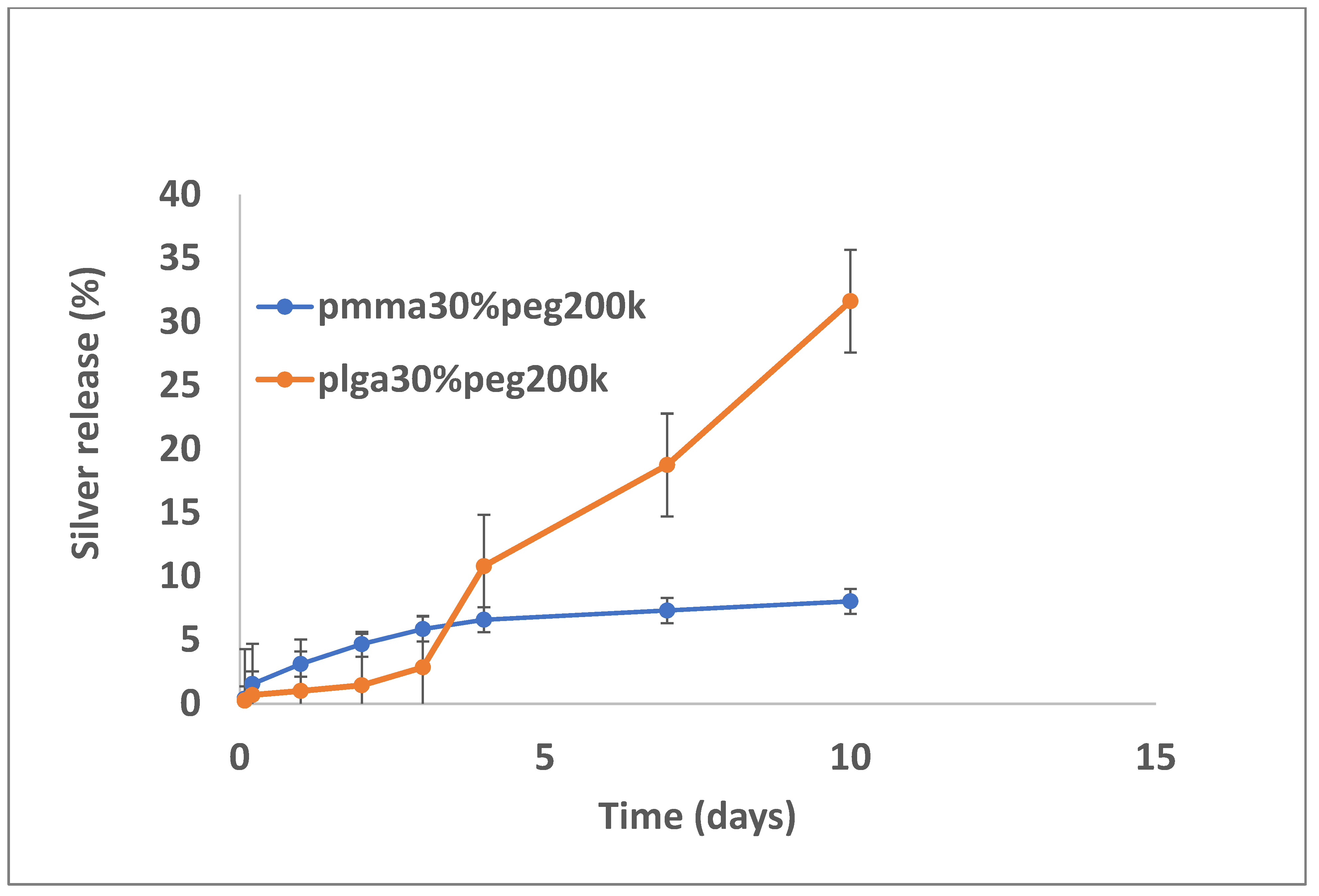
3.2. Drug Release
4. Discussion
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mancino, F.; Gant, V.; Meek, D.R.M.; Haddad, F.S. Vancomycin powder in total joint replacement. Bone Jt. J. 2023, 105, 833–836. [Google Scholar] [CrossRef] [PubMed]
- Hipfl, C.; Leopold, V.; Becker, L.; Pumberger, M.; Perka, C.; Hardt, S. Two-stage revision for periprosthetic joint infection in cemented total hip arthroplasty: An increased risk for failure? Arch. Orthop. Trauma Surg. 2023, 143, 4481–4490. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Dedeogullari, E.S.; Caglar, O.; Danisman, M.; Tokgozoglu, A.M.; Kamaci, S.; Atilla, B. Low dose vancomycin-loaded spacers for two-stage revision knee arthroplasty: High success, low toxicity. Knee 2023, 40, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Corro, S.; Vicente, M.; Rodriguez-Pardo, D.; Pigrau, C.; Lung, M.; Corona, P.S. Vancomycin-Gentamicin Prefabricated Spacers in 2-Stage Revision Arthroplasty for Chronic Hip and Knee Periprosthetic Joint Infection: Insights into Reimplantation Microbiology and Outcomes. J. Arthroplast. 2020, 35, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Zhu, J.; Yang, M.; Yang, C.; Luo, S.; Xie, Y.; Pu, D. Effect of Intra-wound Vancomycin for Spinal Surgery: A Systematic Review and Meta-analysis. Orthop. Surg. 2017, 9, 350–358. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Samuel, J.R.; Gould, F.K. Prosthetic joint infections: Single versus combination therapy. J. Antimicrob. Chemother. 2010, 65, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Rose, W.E.; Poppens, P.T. Impact of biofilm on the in vitro activity of vancomycin alone and in combination with tigecycline and rifampicin against Staphylococcus aureus. J. Antimicrob. Chemother. 2009, 63, 485–488. [Google Scholar] [CrossRef] [PubMed]
- Jackson, J.; Lo, J.; Hsu, E.; Burt, H.M.; Shademani, A.; Lange, D. The Combined Use of Gentamicin and Silver Nitrate in Bone Cement for a Synergistic and Extended Antibiotic Action against Gram-Positive and Gram-Negative Bacteria. Materials 2021, 14, 3413. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Jackson, J.; Leung, F.; Duncan, C.; Mugabe, C.; Burt, H. The use of bone cement for the localized, controlled release of the antibiotics vancomycin, linezolid, or fusidic acid: Effect of additives on drug release rates and mechanical strength. Drug Deliv. Transl. Res. 2011, 1, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Morones-Ramirez, J.R.; Winkler, J.A.; Spina, C.S.; Collins, J.J. Silver enhances antibiotic activity against gram-negative bacteria. Sci. Transl. Med. 2013, 5, 190ra81. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ni, C.; Zhong, Y.; Wu, W.; Song, Y.; Makvandi, P.; Yu, C.; Song, H. Co-Delivery of Nano-Silver and Vancomycin via Silica Nanopollens for Enhanced Antibacterial Functions. Antibiotics 2022, 11, 685. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hashimoto, A.; Miyamoto, H.; Kobatake, T.; Nakashima, T.; Shobuike, T.; Ueno, M.; Murakami, T.; Noda, I.; Sonohata, M.; Mawatari, M. The combination of silver-containing hydroxyapatite coating and vancomycin has a synergistic antibacterial effect on methicillin-resistant Staphylococcus aureus biofilm formation. Bone Jt. Res. 2020, 9, 211–218. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Varisco, M.; Khanna, N.; Brunetto, P.S.; Fromm, K.M. New antimicrobial and biocompatible implant coating with synergic silver-vancomycin conjugate action. ChemMedChem 2014, 9, 1221–1230. [Google Scholar] [CrossRef] [PubMed]
- Farooq, U.; Ahmad, T.; Khan, A.; Sarwar, R.; Shafiq, J.; Raza, Y.; Ahmed, A.; Ullah, S.; Rehman, N.U.; Al-Harrasi, A. Rifampicin conjugated silver nanoparticles: A new arena for development of antibiofilm potential against methicillin resistant Staphylococcus aureus and Klebsiella pneumoniae. Int. J. Nanomed. 2019, 14, 3983–3993. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ivashchenko, O.; Coy, E.; Peplinska, B.; Jarek, M.; Lewandowski, M.; Załęski, K.; Warowicka, A.; Wozniak, A.; Babutina, T.; Jurga-Stopa, J.; et al. Influence of silver content on rifampicin adsorptivity for magnetite/Ag/rifampicin nanoparticles. Nanotechnology 2017, 28, 055603. [Google Scholar] [CrossRef] [PubMed]
- Aragon, J.; Feoli, S.; Irusta, S.; Mendoza, G. Composite scaffold obtained by electro-hydrodynamic technique for infection prevention and treatment in bone repair. Int. J. Pharm. 2019, 557, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Poormasjedi-Meibod, M.; Pakyari, M.; Jackson, J.K.; Elizei, S.S.; Ghahary, A. Development of a nanofibrous wound dressing with an antifibrogenic properties in vitro and in vivo model. J. Biomed. Mater. Res. Part A 2016, 104, 2334–2344. [Google Scholar] [CrossRef] [PubMed]
- Krizsan, G.; Sallai, I.; Veres, D.S.; Prinz, G.; Szeker, D.; Skaliczki, G. Rifampicin resistance and risk factors associated with significantly lower recovery rates after two-stage revision in patients with prosthetic joint infection. J. Glob. Antimicrob. Resist. 2022, 30, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Hsu, Y.H.; Chen, D.W.; Tai, C.D.; Chou, Y.C.; Liu, S.J.; Ueng, S.W.; Chan, E.C. Biodegradable drug-eluting nanofiber-enveloped implants for sustained release of high bactericidal concentrations of vancomycin and ceftazidime: In vitro and in vivo studies. Int. J. Nanomed. 2014, 9, 4347–4355. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chen, D.W.; Liao, J.Y.; Liu, S.J.; Chan, E.C. Novel biodegradable sandwich-structured nanofibrous drug-eluting membranes for repair of infected wounds: An in vitro and in vivo study. Int. J. Nanomed. 2012, 7, 763–771. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zaszczyńska, A.; Kołbuk, D.; Gradys, A.; Sajkiewicz, P. Development of Poly(methyl methacrylate)/nano-hydroxyapatite (PMMA/nHA) Nanofibers for Tissue Engineering Regeneration Using an Electrospinning Technique. Polymers 2024, 16, 531. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wang, Y.; Xu, Y.; Zhai, W.; Zhang, Z.; Liu, Y.; Cheng, S.; Zhang, H. In-situ growth of robust superlubricated nano-skin on electrospun nanofibers for post-operative adhesion prevention. Nat. Commun. 2022, 13, 5056. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jackson, J. Triple Encapsulation and Controlled Release of Vancomycin, Rifampicin and Silver from Poly (Methyl Methacrylate) or Poly (Lactic-Co-Glycolic Acid) Nanofibers. Bioengineering 2024, 11, 529. https://doi.org/10.3390/bioengineering11060529
Jackson J. Triple Encapsulation and Controlled Release of Vancomycin, Rifampicin and Silver from Poly (Methyl Methacrylate) or Poly (Lactic-Co-Glycolic Acid) Nanofibers. Bioengineering. 2024; 11(6):529. https://doi.org/10.3390/bioengineering11060529
Chicago/Turabian StyleJackson, John. 2024. "Triple Encapsulation and Controlled Release of Vancomycin, Rifampicin and Silver from Poly (Methyl Methacrylate) or Poly (Lactic-Co-Glycolic Acid) Nanofibers" Bioengineering 11, no. 6: 529. https://doi.org/10.3390/bioengineering11060529
APA StyleJackson, J. (2024). Triple Encapsulation and Controlled Release of Vancomycin, Rifampicin and Silver from Poly (Methyl Methacrylate) or Poly (Lactic-Co-Glycolic Acid) Nanofibers. Bioengineering, 11(6), 529. https://doi.org/10.3390/bioengineering11060529




