Real-Time Monitoring Using Multiplexed Multi-Electrode Bioelectrical Impedance Spectroscopy for the Stratification of Vascularized Composite Allografts: A Perspective on Predictive Analytics
Abstract
1. Introduction
2. Bioimpedance for Non-Invasive Tissue Evaluation
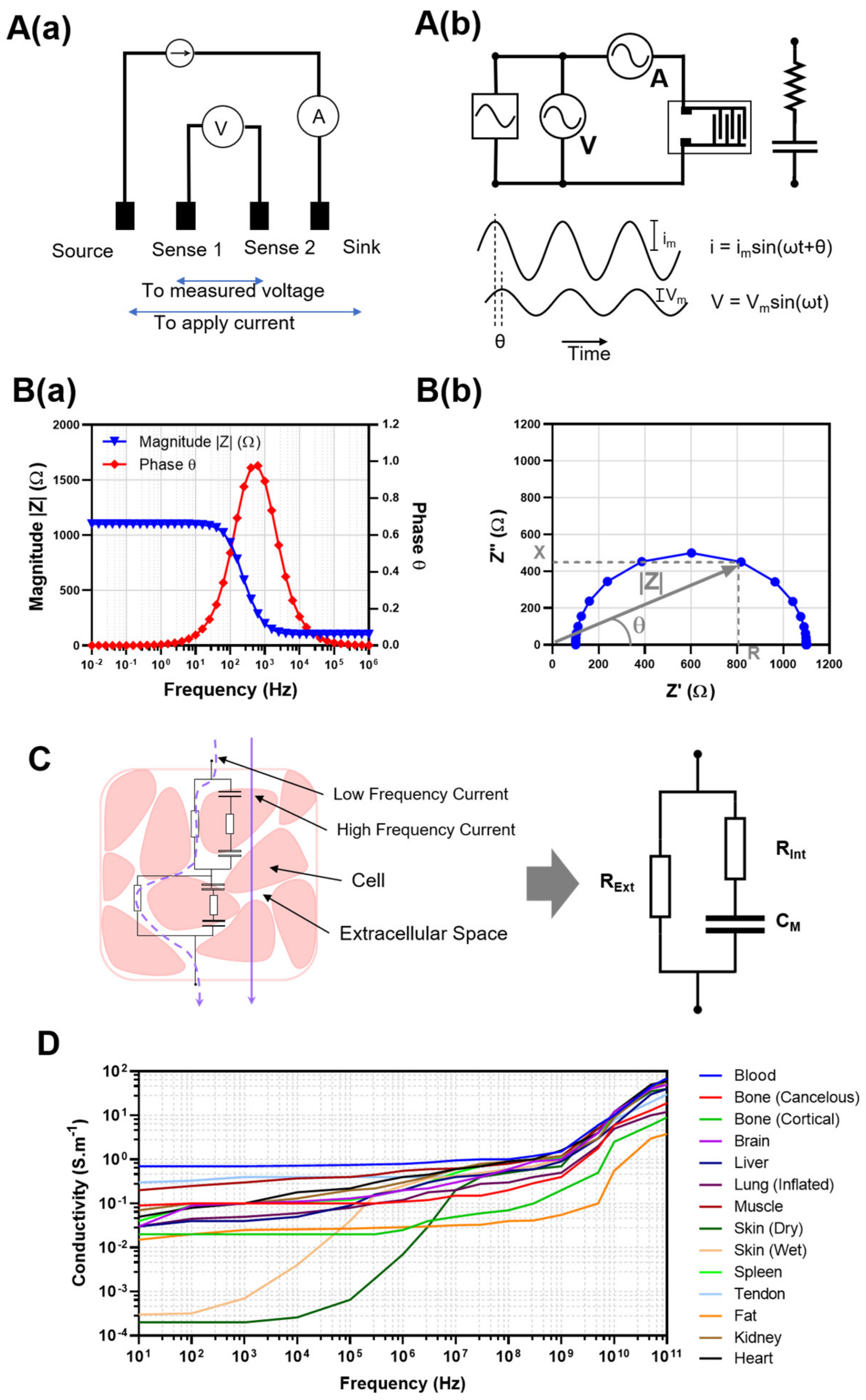
2.1. Application of BI to the Measurement of Clinical Edema
2.2. Application of Electrical Impedance Tomography (EIT) to Edema Measurement
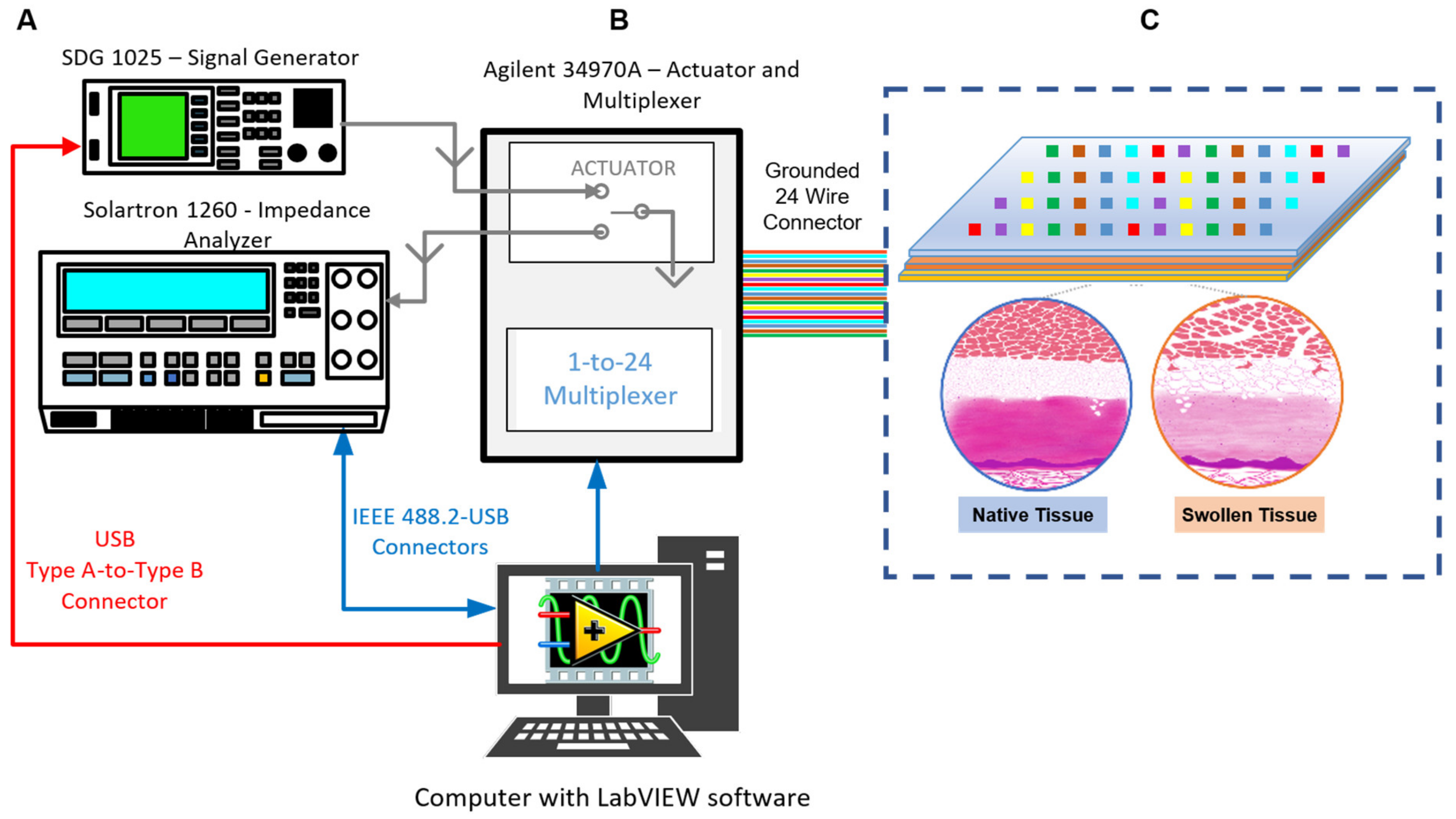
2.3. Potential for Applying BI/BIS to Ex Vivo VCA Preservation
2.4. Potential for ML/AI in Allograft Stratification
2.5. Challenges of BI/BIS Interrogation
3. Summary and Perspective
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Siemionow, M.Z.; Zor, F. Plastic Surgery—Principles and Practice; Farhadieh, R.D., Bulstrode, N.W., Mehrara, B.J., Cugno, S., Eds.; Elsevier: Amsterdam, The Netherlands, 2022; pp. 131–145. [Google Scholar]
- Kollar, B.; Pomahac, B.; Riella, L.V. Novel immunological and clinical insights in vascularized composite allotransplantation. Curr. Opin. Organ Transplant. 2019, 24, 42–48. [Google Scholar] [CrossRef]
- Shores, J.T.; Malek, V.; Lee, W.P.A.; Brandacher, G. Outcomes after hand and upper extremity transplantation. J. Mater. Sci. Mater. Med. 2017, 28, 72. [Google Scholar] [CrossRef]
- Haug, V.; Kollar, B.; Obed, D.; Kiwanuka, H.; Turk, M.; Wo, L.; Tasigiorgos, S.; Kueckelhaus, M.; Riella, L.V.; Pomahac, B. The Evolving Clinical Presentation of Acute Rejection in Facial Transplantation. JAMA Facial Plast. Surg. 2019, 21, 278–285. [Google Scholar] [CrossRef]
- Wainright, J.L.; Booker, S.E.M.; Cherikh, W.S.; Klassen, D.K. Vascularized Composite Allograft Transplantation in the United States: Current State of the Field and Future Challenges. Transplantation 2022, 106, 2093–2096. [Google Scholar] [CrossRef] [PubMed]
- Kueckelhaus, M.; Dermietzel, A.; Alhefzi, M.; Aycart, M.A.; Fischer, S.; Krezdorn, N.; Wo, L.; Maarouf, O.H.; Riella, L.V.; Abdi, R.; et al. Acellular Hypothermic Extracorporeal Perfusion Extends Allowable Ischemia Time in a Porcine Whole Limb Replantation Model. Plast. Reconstr. Surg. 2017, 139, 922e–932e. [Google Scholar] [CrossRef] [PubMed]
- McDiarmid, S.V.; Azari, K.K. Donor-Related Issues in Hand Transplantation. Hand Clin. 2011, 27, 545–552. [Google Scholar] [CrossRef]
- Burlage, L.C.; Tessier, S.N.; Etra, J.W.; Uygun, K.; Brandacher, G. Advances in Machine Perfusion, Organ Preservation, and Cryobiology: Potential Impact on VCA. Curr. Opin. Organ Transplant. 2018, 23, 561–567. [Google Scholar] [CrossRef]
- Salehi, S.; Grayson, W.; Brandacher, G.; Furtmuller, G.; Lopez, J. Establishing a rat abdominal wall perfusion model for vca preservation. Cryobiology 2018, 81, 232. [Google Scholar] [CrossRef]
- Somers, S.M.; Grayson, W.L. Protocol for the Use of a Novel Bioreactor System for Hydrated Mechanical Testing, Strained Sterile Culture, and Force of Contraction Measurement of Tissue Engineered Muscle Constructs. Front. Cell Dev. Biol. 2021, 9, 661036. [Google Scholar] [CrossRef] [PubMed]
- Somers, S.M.; Zhang, N.Y.; Morrissette-McAlmon, J.B.; Tran, K.; Mao, H.-Q.; Grayson, W.L. Myoblast maturity on aligned microfiber bundles at the onset of strain application impacts myogenic outcomes. Acta Biomater. 2019, 94, 232–242. [Google Scholar] [CrossRef]
- Tran, K. Design and Testing of a Multi-Parametric Perfusion Bioreactor for Rat Abdominal Wall Vascularized Composite Allograft Preservation. Master’s Thesis, Johns Hopkins University, Baltimore, MD, USA, 2018. [Google Scholar]
- Gilbert-Honick, J.; Grayson, W. Vascularized and Innervated Skeletal Muscle Tissue Engineering. Adv. Healthc. Mater. 2020, 9, e1900626. [Google Scholar] [CrossRef]
- Jun, I.; Li, N.; Shin, J.; Park, J.; Kim, Y.J.; Jeon, H.; Choi, H.; Cho, J.-G.; Choi, B.C.; Han, H.-S.; et al. Synergistic stimulation of surface topography and biphasic electric current promotes muscle regeneration. Bioact. Mater. 2022, 11, 118–129. [Google Scholar] [CrossRef]
- Salehi, S.; Tran, K.; Grayson, W.L. Advances in Perfusion Systems for Solid Organ Preservation. Yale J. Biol. Med. 2018, 91, 301–312. [Google Scholar]
- Lee, W.P.A.; Shores, J.T.; Brandacher, G. From Auto- to Allotransplantation: Immunomodulatory Protocol for Hand and Arm Transplantation. J. Reconstr. Microsurg. 2018, 34, 683–684. [Google Scholar] [CrossRef]
- Lei, B.; Sleiman, M.M.; Cheng, Q.; Tu, Z.; Zhu, P.; Goddard, M.; Martins, P.N.; Langerude, L.; Nadig, S.; Tomlinson, S.; et al. In Situ Pre-Treatment of Vascularized Composite Allografts with a Targeted Complement Inhibitor Protects against Brain Death and Ischemia Reperfusion Induced Injuries. Front. Immunol. 2021, 12, 630581. [Google Scholar] [CrossRef]
- Ikeguchi, R.; Kakinoki, R.; Ohta, S.; Oda, H.; Yurie, H.; Kaizawa, Y.; Mitsui, H.; Aoyama, T.; Toguchida, J.; Matsuda, S. Recipient bone marrow-derived stromal cells prolong graft survival in a rat hind limb allotransplantation model. Microsurgery 2017, 37, 632–640. [Google Scholar] [CrossRef]
- Constantinescu, M.A.; Knall, E.; Xu, X.; Kiermeir, D.M.; Jenni, H.; Gygax, E.; Rieben, R.; Banič, A.; Vögelin, E. Preservation of Amputated Extremities by Extracorporeal Blood Perfusion; a Feasibility Study in a Porcine Model. J. Surg. Res. 2011, 171, 291–299. [Google Scholar] [CrossRef]
- Werner, N.L.; Alghanem, F.; Rakestraw, S.L.; Sarver, D.C.; Nicely, B.; Pietroski, R.E.; Lange, P.; Rudich, S.M.; Mendias, C.L.; Rojas-Pena, A.; et al. Ex Situ Perfusion of Human Limb Allografts for 24 Hours. Transplantation 2017, 101, e68–e74. [Google Scholar] [CrossRef] [PubMed]
- Amin, K.R.; Stone, J.P.; Kerr, J.; Geraghty, A.; Joseph, L.; Montero-Fernandez, A.; Wong, J.K.; Fildes, J.E. Randomized preclinical study of machine perfusion in vascularized composite allografts. Br. J. Surg. 2020, 108, 574–582. [Google Scholar] [CrossRef]
- Guiseppi-Elie, A. An implantable biochip to influence patient outcomes following trauma-induced hemorrhage. Anal. Bioanal. Chem. 2011, 399, 403–419. [Google Scholar] [CrossRef] [PubMed]
- Bhat, A.; Podstawczyk, D.; Walther, B.K.; Aggas, J.R.; Machado-Aranda, D.; Ward, K.R.; Guiseppi-Elie, A. Toward a hemorrhagic trauma severity score: Fusing five physiological biomarkers. J. Transl. Med. 2020, 18, 348. [Google Scholar] [CrossRef] [PubMed]
- Abasi, S.; Aggas, J.R.; Garayar-Leyva, G.G.; Walther, B.K.; Guiseppi-Elie, A. Bioelectrical Impedance Spectroscopy for Monitoring Mammalian Cells and Tissues under Different Frequency Domains: A Review. ACS Meas. Sci. Au 2022, 2, 495–516. [Google Scholar] [CrossRef] [PubMed]
- Etra, J.W.; Grzelak, M.J.B.; Fidder, S.A.; Kolegraff, K.M.; Bonawitz, S.; Shores, J.; Oh, B.D.; Cooney, D.S.M.; Beck, S.E.D.; Brandacher, G. A Skin Rejection Grading System for Vascularized Composite Allotransplantation in a Preclinical Large Animal Model. Transplantation 2019, 103, 1385–1391. [Google Scholar] [CrossRef] [PubMed]
- Cendales, L.C.; Kanitakis, J.; Schneeberger, S.; Burns, C.; Ruiz, P.; Landin, L.; Remmelink, M.; Hewitt, C.W.; Landgren, T.; Lyons, B.; et al. The Banff 2007 Working Classification of Skin-Containing Composite Tissue Allograft Pathology. Am. J. Transplant. 2008, 8, 1396–1400. [Google Scholar] [CrossRef]
- Kueckelhaus, M.; Turk, M.; Kumamaru, K.K.; Wo, L.; Bueno, E.M.; Lian, C.G.; Alhefzi, M.; Aycart, M.A.; Fischer, S.; De Girolami, U.; et al. Transformation of Face Transplants: Volumetric and Morphologic Graft Changes Resemble Aging after Facial Allotransplantation. Am. J. Transplant. 2016, 16, 968–978. [Google Scholar] [CrossRef]
- Tsai, S.-L.; Wang, M.-H.; Chen, M.-K.; Jang, L.-S. Analytical and Numerical Modeling Methods for Electrochemical Impedance Analysis of Single Cells on Coplanar Electrodes. Electroanalysis 2014, 26, 389–398. [Google Scholar] [CrossRef]
- Heileman, K.; Daoud, J.; Tabrizian, M. Dielectric spectroscopy as a viable biosensing tool for cell and tissue characterization and analysis. Biosens. Bioelectron. 2013, 49, 348–359. [Google Scholar] [CrossRef]
- Hou, J.; Strand-Amundsen, R.; Hødnebø, S.; Tønnessen, T.I.; Høgetveit, J.O. Assessing ischemic injury in human intestine ex vivo with electrical impedance spectroscopy. J. Electr. Bioimpedance 2021, 12, 82–88. [Google Scholar] [CrossRef]
- Martinsen, O.G.; Grimnes, S. Bioimpedance and Bioelectricity Basics; Academic Press: Cambridge, MA, USA, 2011. [Google Scholar]
- Gabriel, S.; Lau, R.W.; Gabriel, C. The dielectric properties of biological tissues: III. Parametric models for the dielectric spectrum of tissues. Phys. Med. Biol. 1996, 41, 2271–2293. [Google Scholar] [CrossRef]
- Genescà, M.; Ivorra, A.; Sola, A.; Palacios, L.; Goujon, J.-M.; Hauet, T.; Villa, R.; Aguiló, J.; Hotter, G. Electrical bioimpedance measurement during hypothermic rat kidney preservation for assessing ischemic injury. Biosens. Bioelectron. 2005, 20, 1866–1871. [Google Scholar] [CrossRef]
- Yerworth, R.J.; Bayford, R.H.; Brown, B.; Milnes, P.; Conway, M.; Holder, D. Electrical impedance tomography spectroscopy (EITS) for human head imaging. Physiol. Meas. 2003, 24, 477–489. [Google Scholar] [CrossRef] [PubMed]
- Oh, T.I.; Kang, M.J.; Jeong, Y.J.; Zhang, T.; Yeo, S.G.; Park, D.C. Tissue Characterization Using an Electrical Bioimpedance Spectroscopy-Based Multi-Electrode Probe to Screen for Cervical Intraepithelial Neoplasia. Diagnostics 2021, 11, 2354. [Google Scholar] [CrossRef] [PubMed]
- González-Correa, C.-A. Bioimpedance in Biomedical Applications and Research; Simini, F., Bertemes-Filho, P., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 187–218. [Google Scholar]
- Zink, M.D.; König, F.; Weyer, S.; Willmes, K.; Leonhardt, S.; Marx, N.; Napp, A. Segmental Bioelectrical Impedance Spectroscopy to Monitor Fluid Status in Heart Failure. Sci. Rep. 2020, 10, 3577. [Google Scholar] [CrossRef] [PubMed]
- Edwick, D.O.; Hince, D.A.; Rawlins, J.M.; Wood, F.M.; Edgar, D.W. Bioimpedance Spectroscopy Is a Valid and Reliable Measure of Edema Following Hand Burn Injury (Part 1—Method Validation). J. Burn. Care Res. 2020, 41, 780–787. [Google Scholar] [CrossRef]
- Crescenzi, R.; Donahue, P.M.; Weakley, S.; Garza, M.; Donahue, M.J.; Herbst, K.L. Lipedema and Dercum’s Disease: A New Application of Bioimpedance. Lymphat. Res. Biol. 2019, 17, 671–679. [Google Scholar] [CrossRef] [PubMed]
- Crescenzi, R.; Marton, A.; Donahue, P.M.; Mahany, H.B.; Lants, S.K.; Wang, P.; Beckman, J.A.; Donahue, M.J.; Titze, J. Tissue Sodium Content is Elevated in the Skin and Subcutaneous Adipose Tissue in Women with Lipedema. Obesity 2018, 26, 310–317. [Google Scholar] [CrossRef]
- Ramírez-Chavarría, R.; Sánchez-Pérez, C.; Matatagui, D.; Qureshi, N.; Pérez-García, A.; Hernández-Ruíz, J. Ex-vivo biological tissue differentiation by the Distribution of Relaxation Times method applied to Electrical Impedance Spectroscopy. Electrochim. Acta 2018, 276, 214–222. [Google Scholar] [CrossRef]
- York, S.L.; Ward, L.C.; Czerniec, S.; Lee, M.J.; Refshauge, K.M.; Kilbreath, S.L. Single frequency versus bioimpedance spectroscopy for the assessment of lymphedema. Breast Cancer Res. Treat. 2008, 117, 177–182. [Google Scholar] [CrossRef]
- Yang, Y.; Jia, J. A multi-frequency electrical impedance tomography system for real-time 2D and 3D imaging. Rev. Sci. Instrum. 2017, 88, 085110. [Google Scholar] [CrossRef]
- Mabrouk, S.; Hersek, S.; Jeong, H.K.; Whittingslow, D.; Ganti, V.G.; Wolkoff, P.; Inan, O.T. Robust Longitudinal Ankle Edema Assessment Using Wearable Bioimpedance Spectroscopy. IEEE Trans. Biomed. Eng. 2020, 67, 1019–1029. [Google Scholar] [CrossRef]
- Lyons, K.J.; Bischoff, M.K.; Fonarow, G.; Horwich, T.B. Noninvasive Bioelectrical Impedance for Predicting Clinical Outcomes in Outpatients With Heart Failure. Crit. Pathw. Cardiol. J. Evid.-Based Med. 2017, 16, 32–36. [Google Scholar] [CrossRef]
- Strand-Amundsen, R.J.; Reims, H.M.; Tronstad, C.; Kalvøy, H.; Martinsen, G.; Høgetveit, J.O.; Ruud, T.E.; Tønnessen, T.I. Ischemic small intestine—In vivo versus ex vivo bioimpedance measurements. Physiol. Meas. 2017, 38, 715–728. [Google Scholar] [CrossRef]
- Peterson, D.M.; Beal, E.W.; Reader, B.F.; Dumond, C.; Black, S.M.; Whitson, B.A. Electrical Impedance as a Noninvasive Metric of Quality in Allografts Undergoing Normothermic Ex Vivo Lung Perfusion. ASAIO J. 2022, 68, 964–971. [Google Scholar] [CrossRef] [PubMed]
- Ta, H.Q.; Teman, N.R.; Kron, I.L.; Roeser, M.E.; Laubach, V.E. Steen solution protects pulmonary microvascular endothelial cells and preserves endothelial barrier after lipopolysaccharide-induced injury. J. Thorac. Cardiovasc. Surg. 2023, 165, e5–e20. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.; Strand-Amundsen, R.; Tronstad, C.; Tønnessen, T.I.; Høgetveit, J.O.; Martinsen, G. Small intestinal viability assessment using dielectric relaxation spectroscopy and deep learning. Sci. Rep. 2022, 12, 3279. [Google Scholar] [CrossRef]
- Nyboer, J.; Kreider, M.M.; Hannapel, L. Electrical Impedance Plethysmography. Circulation 1950, 2, 811–821. [Google Scholar] [CrossRef] [PubMed]
- Putensen, C.; Hentze, B.; Muenster, S.; Muders, T. Electrical Impedance Tomography for Cardio-Pulmonary Monitoring. J. Clin. Med. 2019, 8, 1176. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Chi, Y.; Long, Y.; Yuan, S.; Zhang, R.; Frerichs, I.; Möller, K.; Fu, F.; Zhao, Z. Bedside Evaluation of Pulmonary Embolism by Saline Contrast Electrical Impedance Tomography Method: A Prospective Observational Study. Am. J. Respir. Crit. Care Med. 2020, 202, 1464–1468. [Google Scholar] [CrossRef]
- Aguiar Santos, S.; Czaplik, M.; Orschulik, J.; Hochhausen, N.; Leonhardt, S. Lung pathologies analyzed with multi-frequency electrical impedance tomography: Pilot animal study. Respir. Physiol. Neurobiol. 2018, 254, 1–9. [Google Scholar] [CrossRef]
- Zhao, Z.; He, H.; Luo, J.; Adler, A.; Zhang, X.; Liu, R.; Lan, Y.; Lu, S.; Luo, X.; Lei, Y.; et al. Detection of pulmonary oedema by electrical impedance tomography: Validation of previously proposed approaches in a clinical setting. Physiol. Meas. 2019, 40, 054008. [Google Scholar] [CrossRef]
- Jang, J.; Seo, J.K. Detection of admittivity anomaly on high-contrast heterogeneous backgrounds using frequency difference EIT. Physiol. Meas. 2015, 36, 1179–1192. [Google Scholar] [CrossRef] [PubMed]
- Abasi, S.; Aggas, J.R.; Venkatesh, N.; Vallavanatt, I.G.; Guiseppi-Elie, A. Design, fabrication and testing of an electrical cell stimulation and recording apparatus (ECSARA) for cells in electroculture. Biosens. Bioelectron. 2020, 147, 111793. [Google Scholar] [CrossRef]
- Åström, M.; Lemaire, J.-J.; Wårdell, K. Influence of heterogeneous and anisotropic tissue conductivity on electric field distribution in deep brain stimulation. Med. Biol. Eng. Comput. 2012, 50, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Rabbani, K.S.; Sarker, M.; Akond, M.H.R.; Akter, T. Focused Impedance Measurement (FIM): A New Technique with Improved Zone Localization. Ann. N. Y. Acad. Sci. 1999, 873, 408–420. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Vargas, A.; Ivorra, A.; Arkwright, J.W. Design, Construction and Validation of an Electrical Impedance Probe with Contact Force and Temperature Sensors Suitable for in-vivo Measurements. Sci. Rep. 2018, 8, 14818. [Google Scholar] [CrossRef] [PubMed]
- Shickel, B.J.B.; Aggas, J.R.; Loftus, T.J.; Kotanen, C.N.; Rashidi, P.; Guiseppi-Elie, A. Scoring for Hemorrhage Severity in Traumatic Injury. In Biomarkers in Trauma, Injury and Critical Care; Rajkumar Rajendram, V.R.P., Vinood, B.P., Eds.; Springer-Nature: Berlin/Heidelberg, Germany, 2023; Available online: https://link.springer.com/book/9783031073946 (accessed on 24 March 2023).
- Ozer, K.; Rojas-Pena, A.; Mendias, C.L.; Bryner, B.S.; Toomasian, C.; Bartlett, R.H. The Effect of Ex Situ Perfusion in a Swine Limb Vascularized Composite Tissue Allograft on Survival up to 24 Hours. J. Hand Surg. 2016, 41, 3–12. [Google Scholar] [CrossRef]
- Kauke, M.; Safi, A.-F.; Panayi, A.C.; Palmer, W.J.; Haug, V.; Kollar, B.; Nelms, L.; Tchiloemba, B.; Pomahac, B. A systematic review of immunomodulatory strategies used in skin-containing preclinical vascularized composite allotransplant models. J. Plast. Reconstr. Aesthetic Surg. 2022, 75, 586–604. [Google Scholar] [CrossRef]
- Ziegler-Graham, K.; MacKenzie, E.J.; Ephraim, P.L.; Travison, T.G.; Brookmeyer, R. Estimating the Prevalence of Limb Loss in the United States: 2005 to 2050. Arch. Phys. Med. Rehabil. 2008, 89, 422–429. [Google Scholar] [CrossRef]
- Wolfram, D.; Starzl, R.; Hackl, H.; Barclay, D.; Hautz, T.; Zelger, B.; Brandacher, G.; Lee, W.P.A.; Eberhart, N.; Vodovotz, Y.; et al. Insights from Computational Modeling in Inflammation and Acute Rejection in Limb Transplantation. PLoS ONE 2014, 9, e99926. [Google Scholar] [CrossRef]
- Ha, N.; Xu, K.; Ren, G.; Mitchell, A.; Ou, J.Z. Machine Learning-Enabled Smart Sensor Systems. Adv. Intell. Syst. 2020, 2, 2000063. [Google Scholar] [CrossRef]
- De Faria, A.P.; Ritter, A.M.V.; Gasparetti, C.S.; Corrêa, N.B.; Brunelli, V.; Almeida, A.; Pires, N.F.; Modolo, R.; Junior, H.M. A Proposed Inflammatory Score of Circulating Cytokines/Adipokines Associated with Resistant Hypertension, but Dependent on Obesity Parameters. Arq. Bras. Cardiol. 2019, 112, 383–389. [Google Scholar] [CrossRef] [PubMed]
- Gołąbek-Dropiewska, K.; Pawłowska, J.; Witkowski, J.M.; Lasek, J.; Marks, W.; Stasiak, M.; Jaskólski, D.; Kawecka, A.; Łuczkiewicz, P.; Baczkowski, B. Analysis of selected pro- and anti-inflammatory cytokines in patients with multiple injuries in the early period after trauma. Central Eur. J. Immunol. 2018, 43, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Jastrow, K.M.; Gonzalez, E.A.; McGuire, M.F.; Suliburk, J.W.; Kozar, R.A.; Iyengar, S.; Motschall, D.A.; McKinley, B.A.; Moore, F.A.; Mercer, D.W. Early Cytokine Production Risk Stratifies Trauma Patients for Multiple Organ Failure. J. Am. Coll. Surg. 2009, 209, 320–331. [Google Scholar] [CrossRef] [PubMed]
- Koelman, L.; Pivovarova-Ramich, O.; Pfeiffer, A.F.H.; Grune, T.; Aleksandrova, K. Cytokines for evaluation of chronic inflammatory status in ageing research: Reliability and phenotypic characterisation. Immun. Ageing 2019, 16, 11. [Google Scholar] [CrossRef]
- Amin, K.; Stone, J.P.; Edge, R.J.; Parkes, J.; Kerr, J.; Joseph, L.; Wong, J.; Fildes, J.E. Optimization of an Ex-Vivo Limb Perfusion Protocol for Vascularized Composite Allograft Transplantation. Transplantation 2018, 102, S436–S437. [Google Scholar] [CrossRef]
- Honeyman, C.; Stark, H.; Wang, H.C.; Hester, J.; Issa, F.; Giele, H. Biomarker and surrogate development in vascularised composite allograft transplantation: Current progress and future challenges. J. Plast. Reconstr. Aesthetic Surg. 2021, 74, 711–717. [Google Scholar] [CrossRef]
- Dutta, N.; Lillehoj, P.; Estrela, P.; Dutta, G. Electrochemical Biosensors for Cytokine Profiling: Recent Advancements and Possibilities in the Near Future. Biosensors 2021, 11, 94. [Google Scholar] [CrossRef]
- Charlton, P.H.; Pimentel, M.; Lokhandwala, S. Secondary Analysis of Electronic Health Records; Springer International Publishing: Cham, Switzerland, 2016; pp. 325–338. [Google Scholar]
- Sexton, R.S.; Gupta, J.N. Comparative evaluation of genetic algorithm and backpropagation for training neural networks. Inf. Sci. 2000, 129, 45–59. [Google Scholar] [CrossRef]
- Nazmi, S.; Homaifar, A. 2018 IEEE International Conference on Systems, Man, and Cybernetics; IEEE Xplore: Miyazaki, Japan, 2018; pp. 668–674. [Google Scholar]
- Petrone, G.; Axerio-Cilies, J.; Quagliarella, D.; Iaccarino, G. A probabilistic non-dominated sorting GA for optimization under uncertainty. Eng. Comput. 2013, 30, 1054–1085. [Google Scholar] [CrossRef]
- Fawcett, T. An Introduction to ROC analysis. Pattern Recogn. Lett. 2006, 27, 861–874. [Google Scholar] [CrossRef]
- Muh, H.C.; Tong, J.C.; Tammi, M.T. AllerHunter: A SVM-Pairwise System for Assessment of Allergenicity and Allergic Cross-Reactivity in Proteins. PLoS ONE 2009, 4, e5861. [Google Scholar] [CrossRef] [PubMed]
- Tibshirani, R.; Walther, G. Cluster Validation by Prediction Strength. J. Comput. Graph. Stat. 2005, 14, 511–528. [Google Scholar] [CrossRef]
- Nasrabadi, N.M. Pattern recognition and machine learning. J. Electron. Imaging 2007, 16, 049901. [Google Scholar]
- Arlot, S.; Celisse, A. A survey of cross-validation procedures for model selection. Stat. Surv. 2010, 4, 40–79. [Google Scholar] [CrossRef]
- Brown, B.H.; Seagar, A.D. The Sheffield data collection system. Clin. Phys. Physiol. Meas. 1987, 8, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Birgersson, U.; Åberg, P.; Nicander, I.; Ollmar, S.; Birgersson, E. Non-invasive bioimpedance of intact skin: Mathematical modeling and experiments. Physiol. Meas. 2010, 32, 1. [Google Scholar] [CrossRef]
- Franco, S. The Howland Current Pump. All About Circuits (AAC) Technical Article; All About Circuits (AAC): Boise, ID, USA, 2019; p. 1. Available online: https://www.allaboutcircuits.com/technical-articles/the-howland-current-pump/ (accessed on 23 March 2023).
- Park, B.; Biswas, S.; Park, H. Electrical Characterization of the Tongue and the Soft Palate Using Lumped-Element Model for Intraoral Neuromodulation. IEEE Trans. Biomed. Eng. 2021, 68, 3151–3160. [Google Scholar] [CrossRef]
- Menden, T.; Matuszczyk, J.; Leonhardt, S.; Walter, M. Bandwidth and Common Mode Optimization for Current and Voltage Sources in Bioimpedance Spectroscopy. J. Electr. Bioimpedance 2021, 12, 135–146. [Google Scholar] [CrossRef]
- Zhang, T.; Jeong, Y.; Park, D.; Oh, T. Performance Evaluation of Multiple Electrodes Based Electrical Impedance Spectroscopic Probe for Screening of Cervical Intraepithelial Neoplasia. Electronics 2021, 10, 1933. [Google Scholar] [CrossRef]
- Epstein, B.R.; Foster, K.R. Anisotropy in the dielectric properties of skeletal muscle. Med. Biol. Eng. Comput. 1983, 21, 51–55. [Google Scholar] [CrossRef]
- Naranjo-Hernández, D.; Reina-Tosina, J.; Min, M. Fundamentals, Recent Advances, and Future Challenges in Bioimpedance Devices for Healthcare Applications. J. Sens. 2019, 2019, 9210258. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aggas, J.R.; Abasi, S.; Ton, C.; Salehi, S.; Liu, R.; Brandacher, G.; Grayson, W.L.; Guiseppi-Elie, A. Real-Time Monitoring Using Multiplexed Multi-Electrode Bioelectrical Impedance Spectroscopy for the Stratification of Vascularized Composite Allografts: A Perspective on Predictive Analytics. Bioengineering 2023, 10, 434. https://doi.org/10.3390/bioengineering10040434
Aggas JR, Abasi S, Ton C, Salehi S, Liu R, Brandacher G, Grayson WL, Guiseppi-Elie A. Real-Time Monitoring Using Multiplexed Multi-Electrode Bioelectrical Impedance Spectroscopy for the Stratification of Vascularized Composite Allografts: A Perspective on Predictive Analytics. Bioengineering. 2023; 10(4):434. https://doi.org/10.3390/bioengineering10040434
Chicago/Turabian StyleAggas, John R., Sara Abasi, Carolyn Ton, Sara Salehi, Renee Liu, Gerald Brandacher, Warren L. Grayson, and Anthony Guiseppi-Elie. 2023. "Real-Time Monitoring Using Multiplexed Multi-Electrode Bioelectrical Impedance Spectroscopy for the Stratification of Vascularized Composite Allografts: A Perspective on Predictive Analytics" Bioengineering 10, no. 4: 434. https://doi.org/10.3390/bioengineering10040434
APA StyleAggas, J. R., Abasi, S., Ton, C., Salehi, S., Liu, R., Brandacher, G., Grayson, W. L., & Guiseppi-Elie, A. (2023). Real-Time Monitoring Using Multiplexed Multi-Electrode Bioelectrical Impedance Spectroscopy for the Stratification of Vascularized Composite Allografts: A Perspective on Predictive Analytics. Bioengineering, 10(4), 434. https://doi.org/10.3390/bioengineering10040434








