Future of Endoscopic Spine Surgery: Insights from Cutting-Edge Technology in the Industrial Field
Abstract
:1. Introduction
2. Surgical Techniques of Endoscopic Spinal Surgery
3. Advancements in Endoscopic Surgery Devices
3.1. Camera and Lighting Systems
3.1.1. Resolution
3.1.2. Color and Camera Sensor
3.1.3. Lighting System
3.1.4. Angles of Scope and Flexibility
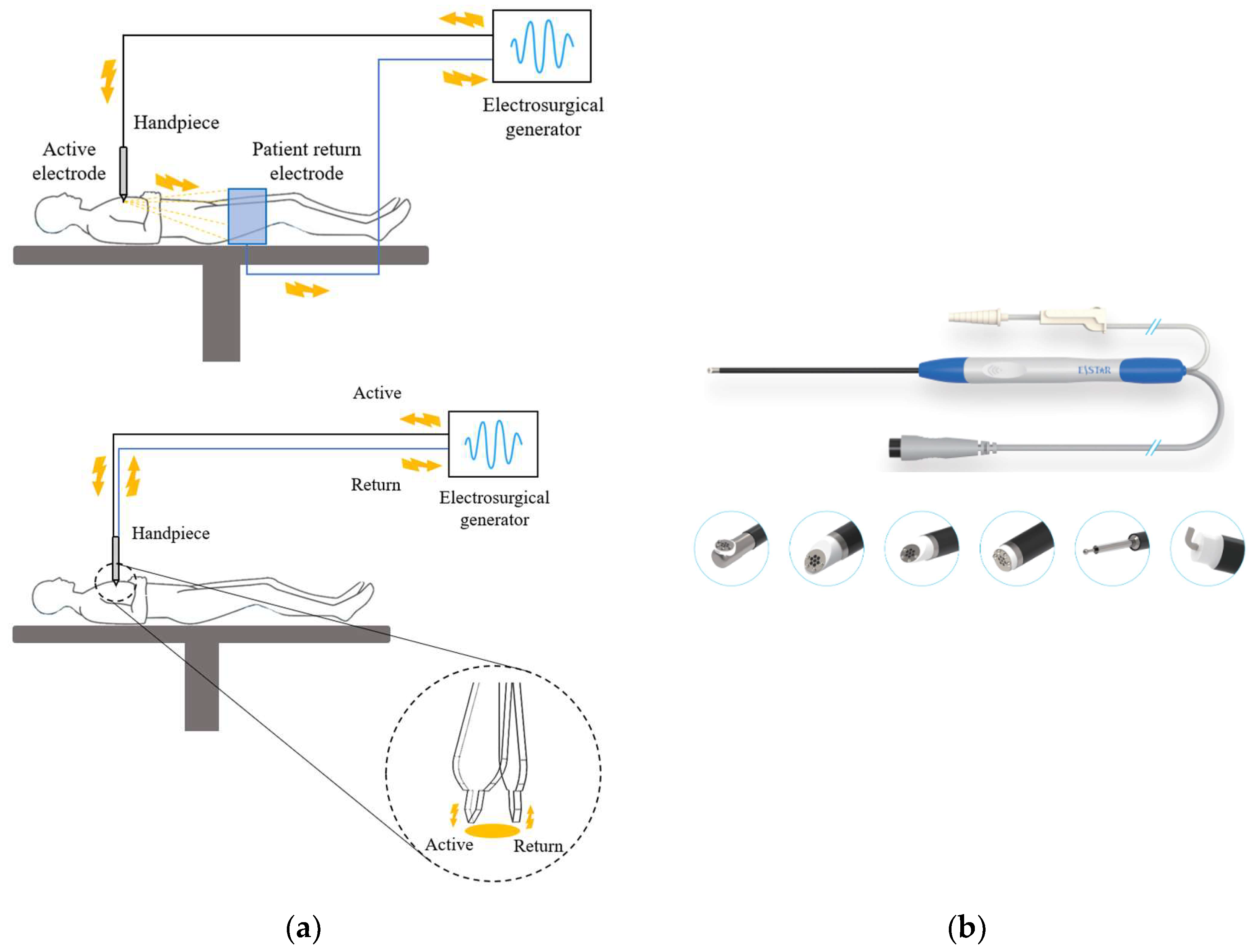
3.2. Radiofrequency Ablation Systems
3.2.1. Generator
3.2.2. Electrode
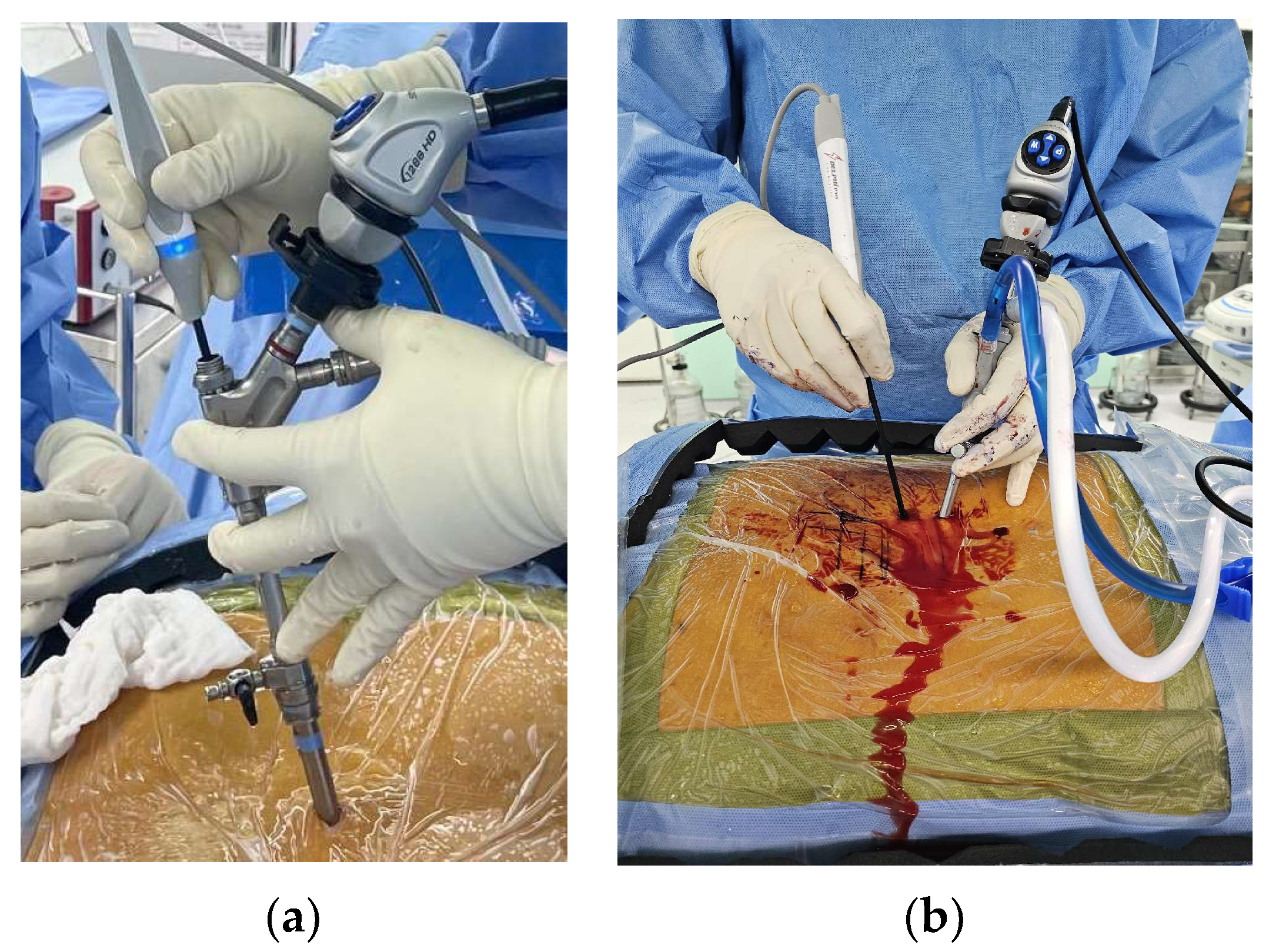
3.3. Drill Systems
4. Discussion and Future Directions
4.1. Development of ESS
4.2. Camera and Visualization
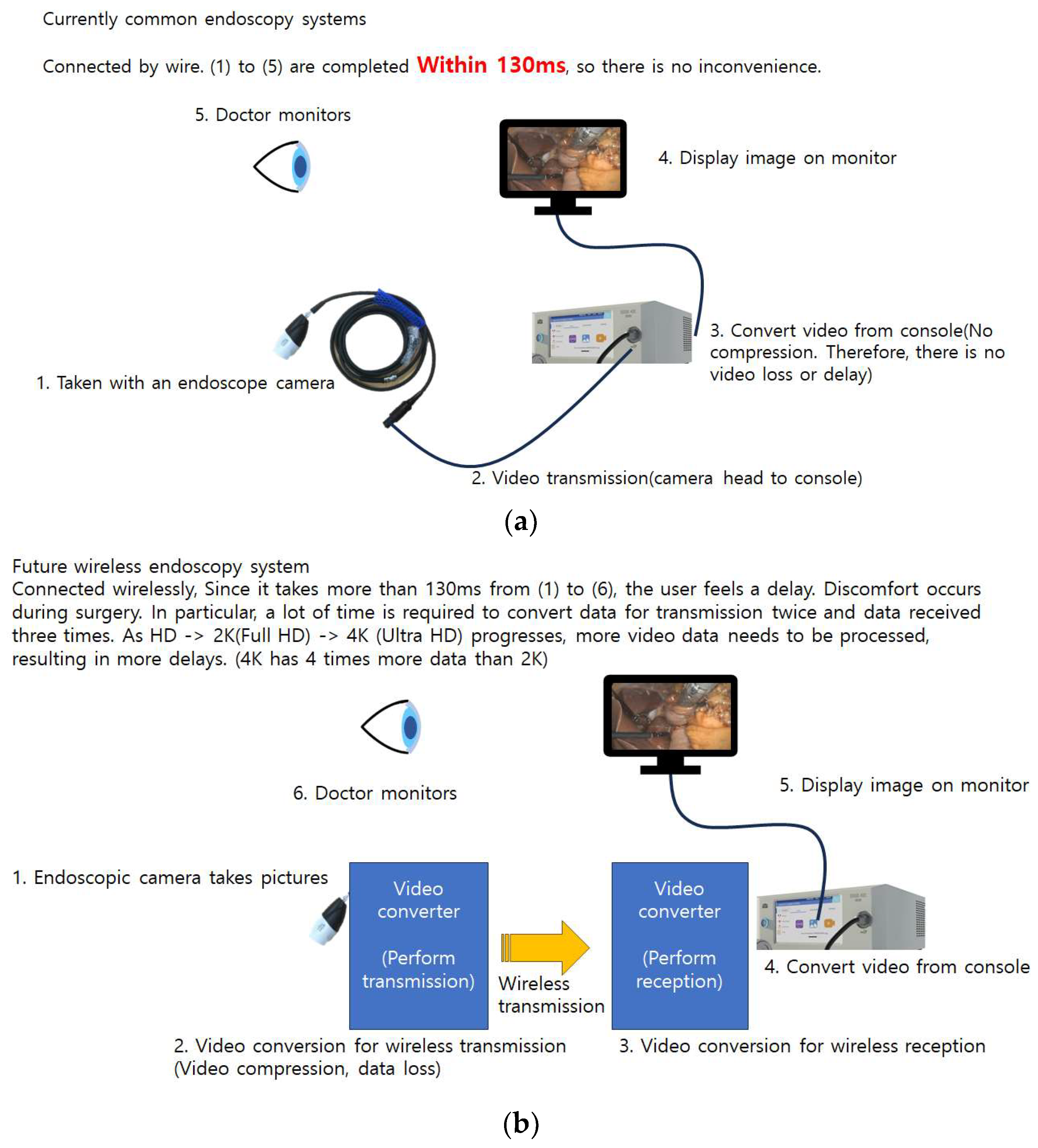
4.3. Wireless System
4.4. Coagulation and Ablation Tools
4.5. Drill System
4.5.1. Waterproofing and Durability
4.5.2. High Speed and High Torque Even with Long Thin Shaft
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ahn, Y. A Historical Review of Endoscopic Spinal Discectomy. World Neurosurg. 2021, 145, 591–596. [Google Scholar] [CrossRef]
- Chen, K.T.; Jabri, H.; Lokanath, Y.K.; Song, M.S.; Kim, J.S. The evolution of interlaminar endoscopic spine surgery. J. Spine Surg. 2020, 6, 502–512. [Google Scholar] [CrossRef] [PubMed]
- Khandge, A.V.; Sharma, S.B.; Kim, J.S. The Evolution of Transforaminal Endoscopic Spine Surgery. World Neurosurg. 2021, 145, 643–656. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.J.; Lee, S.H.; Jung, E.S.; Son, B.G.; Choi, E.S.; Shin, J.H.; Sung, J.K.; Chi, Y.C. Targeted percutaneous transforaminal endoscopic diskectomy in 295 patients: Comparison with results of microscopic diskectomy. Surg. Neurol. 2007, 68, 623–631. [Google Scholar] [CrossRef] [PubMed]
- Komp, M.; Hahn, P.; Oezdemir, S.; Giannakopoulos, A.; Heikenfeld, R.; Kasch, R.; Merk, H.; Godolias, G.; Ruetten, S. Bilateral spinal decompression of lumbar central stenosis with the full-endoscopic interlaminar versus microsurgical laminotomy technique: A prospective, randomized, controlled study. Pain Phys. 2015, 18, 61–70. [Google Scholar] [CrossRef]
- Lee, D.Y.; Shim, C.S.; Ahn, Y.; Choi, Y.G.; Kim, H.J.; Lee, S.H. Comparison of percutaneous endoscopic lumbar discectomy and open lumbar microdiscectomy for recurrent disc herniation. J. Korean Neurosurg. Soc. 2009, 46, 515–521. [Google Scholar] [CrossRef]
- Ruetten, S.; Komp, M.; Merk, H.; Godolias, G. Full-endoscopic interlaminar and transforaminal lumbar discectomy versus conventional microsurgical technique: A prospective, randomized, controlled study. Spine 2008, 33, 931–939. [Google Scholar] [CrossRef]
- Kambin, P.; Sampson, S. Posterolateral percutaneous suction-excision of herniated lumbar intervertebral discs. Report of interim results. Clin. Orthop. Relat. Res. 1986, 207, 37–43. [Google Scholar] [CrossRef]
- Hijikata, S. Percutaneous nucleotomy. A new concept technique and 12 years’ experience. Clin. Orthop. Relat. Res. 1989, 238, 9–23. [Google Scholar] [CrossRef]
- Choi, I.; Kim, J.S.; Ahn, Y. Nomenclature of Endoscopic Spine Surgery. In Advanced Techniques of Endoscopic Lumbar Spine Surgery; Kim, H.S., Mayer, M., Heo, D.H., Park, C.W., Eds.; Springer: Singapore, 2020; pp. 7–18. [Google Scholar]
- Heo, D.H.; Lee, D.C.; Kim, H.S.; Park, C.K.; Chung, H. Clinical Results and Complications of Endoscopic Lumbar Interbody Fusion for Lumbar Degenerative Disease: A Meta-Analysis. World Neurosurg. 2021, 145, 396–404. [Google Scholar] [CrossRef]
- Birkenmaier, C.; Komp, M.; Leu, H.F.; Wegener, B.; Ruetten, S. The current state of endoscopic disc surgery: Review of controlled studies comparing full-endoscopic procedures for disc herniations to standard procedures. Pain Phys. 2013, 16, 335–344. [Google Scholar] [CrossRef]
- Ruetten, S.; Komp, M.; Godolias, G. A New full-endoscopic technique for the interlaminar operation of lumbar disc herniations using 6-mm endoscopes: Prospective 2-year results of 331 patients. Minim. Invas. Neurosurg. 2006, 49, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Hofstetter, C.P.; Ahn, Y.; Choi, G.; Gibson, J.N.A.; Ruetten, S.; Zhou, Y.; Li, Z.Z.; Siepe, C.J.; Wagner, R.; Lee, J.H.; et al. AOSpine Consensus Paper on Nomenclature for Working-Channel Endoscopic Spinal Procedures. Glob. Spine J. 2020, 10, 111S–121S. [Google Scholar] [CrossRef]
- Ahn, Y. Anterior Endoscopic Cervical Discectomy: Surgical Technique and Literature Review. Neurospine 2023, 20, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Heo, D.H.; Lee, D.C.; Kim, T.H.; Park, C.K. Comparative Analysis with Modified Inclined Technique for Posterior Endoscopic Cervical Foraminotomy in Treating Cervical Osseous Foraminal Stenosis: Radiological and Midterm Clinical Outcomes. Neurospine 2022, 19, 603–615. [Google Scholar] [CrossRef]
- Kim, J.; Heo, D.H.; Lee, D.C.; Chung, H.T. Biportal endoscopic unilateral laminotomy with bilateral decompression for the treatment of cervical spondylotic myelopathy. Acta Neurochir. 2021, 163, 2537–2543. [Google Scholar] [CrossRef]
- Lee, S.H.; Musharbash, F.N. Uniportal, Transforaminal Endoscopic Thoracic Discectomy: Review and Technical Note. Neurospine 2023, 20, 19–27. [Google Scholar] [CrossRef]
- Kim, J.Y.; Ha, J.S.; Lee, C.K.; Lee, D.C.; Hong, H.J.; Choi, S.Y.; Park, C.K. Biportal Endoscopic Posterior Thoracic Laminectomy for Thoracic Spondylotic Myelopathy Caused by Ossification of the Ligamentum Flavum: Technical Developments and Outcomes. Neurospine 2023, 20, 129–140. [Google Scholar] [CrossRef]
- Lee, S.G.; Ahn, Y. Transforaminal Endoscopic Lumbar Discectomy: Basic Concepts and Technical Keys to Clinical Success. Int. J. Spine Surg. 2021, 15, S38–S46. [Google Scholar] [CrossRef]
- Won, Y.I.; Yuh, W.T.; Kwon, S.W.; Kim, C.H.; Yang, S.H.; Kim, K.T.; Chung, C.K. Interlaminar Endoscopic Lumbar Discectomy: A Narrative Review. Int. J. Spine Surg. 2021, 15, S47–S53. [Google Scholar] [CrossRef]
- Choi, G.; Lee, S.H.; Bhanot, A.; Raiturker, P.P.; Chae, Y.S. Percutaneous endoscopic discectomy for extraforaminal lumbar disc herniations: Extraforaminal targeted fragmentectomy technique using working channel endoscope. Spine 2007, 32, E93–E99. [Google Scholar] [CrossRef] [PubMed]
- Rhee, D.Y.; Ahn, Y. Full-Endoscopic Lumbar Foraminotomy for Foraminal Stenosis in Spondylolisthesis: Two-Year Follow-Up Results. Diagnostics 2022, 12, 3152. [Google Scholar] [CrossRef] [PubMed]
- Wagner, R.; Haefner, M. Uniportal Endoscopic Lumbar Interbody Fusion. Neurospine 2020, 17, S120–S128. [Google Scholar] [CrossRef] [PubMed]
- Heo, D.H.; Hong, Y.H.; Lee, D.C.; Chung, H.J.; Park, C.K. Technique of Biportal Endoscopic Transforaminal Lumbar Interbody Fusion. Neurospine 2020, 17, S129–S137. [Google Scholar] [CrossRef]
- Pao, J.L. Biportal Endoscopic Transforaminal Lumbar Interbody Fusion Using Double Cages: Surgical Techniques and Treatment Outcomes. Neurospine 2023, 20, 80–91. [Google Scholar] [CrossRef]
- Ohara, Y.; Nakajima, Y.; Kimura, T.; Kikuchi, N.; Sagiuchi, T. Full-Endoscopic Transcervical Ventral Decompression for Pathologies of Craniovertebral Junction: Case Series. Neurospine 2020, 17, S138–S144. [Google Scholar] [CrossRef] [PubMed]
- Suvithayasiri, S.; Kim, Y.J.; Liu, Y.; Trathitephun, W.; Asawasaksaku, A.; Quillo-Olvera, J.; Kotheeranurak, V.; Chagas, H.; Valencia, C.C.; Serra, M.V.; et al. The Role and Clinical Outcomes of Endoscopic Spine Surgery of Treating Spinal Metastases; Outcomes of 29 Cases From 8 Countries. Neurospine 2023, 20, 608–619. [Google Scholar] [CrossRef]
- Chang, K.S.; Sun, L.W.; Cheng, C.Y.; Chang, S.W.; Chen, C.M. Full Endoscopic Removal of Cervical Spinal Epidural Abscess: Case Report and Technical Note. Neurospine 2020, 17, S160–S165. [Google Scholar] [CrossRef]
- Heo, D.H.; Sharma, S.; Park, C.K. Endoscopic Treatment of Extraforaminal Entrapment of L5 Nerve Root (Far out Syndrome) by Unilateral Biportal Endoscopic Approach: Technical Report and Preliminary Clinical Results. Neurospine 2019, 16, 130–137. [Google Scholar] [CrossRef]
- Vargas, R.A.A.; Hagel, V.; Xifeng, Z.; Yang, H.; Ramirez Leon, J.F.; Lorio, M.; Telfeian, A.E.; Houle, P.; Gardocki, R.; Dowling, A.; et al. Durotomy- and Irrigation-Related Serious Adverse Events During Spinal Endoscopy: Illustrative Case Series and International Surgeon Survey. Int. J. Spine Surg. 2023, 17, 387–398. [Google Scholar] [CrossRef]
- Lewandrowski, K.-U. The strategies behind “inside-out” and “outside-in” endoscopy of the lumbar spine: Treating the pain generator. J. Spine Surg. 2019, 6, S35–S39. [Google Scholar] [CrossRef] [PubMed]
- Comparing and Contrasting CCD vs. CMOS Sensor Image Quality. Available online: https://www.camerasource.com/industry-news/ccd-vs-cmos-sensor-image-quality/ (accessed on 1 February 2023).
- The Difference between the Three-Chip and Single-Chip of the Endoscope Camera. Available online: https://www.xzykd.com/news/three-chip-and-single-chip-of-the-endoscope-camera.html (accessed on 24 August 2021).
- Fish, R.M.; Geddes, L.A. Conduction of electrical current to and through the human body: A review. Eplasty 2009, 9, e44. [Google Scholar]
- Aferzon, M.; Millman, B. Excision of rhinophyma with high-frequency electrosurgery. Dermatol. Surg. 2002, 28, 735–738. [Google Scholar] [CrossRef] [PubMed]
- Taheri, A.; Mansoori, P.; Sandoval, L.F.; Feldman, S.R.; Pearce, D.; Williford, P.M. Electrosurgery: Part I. Basics and principles. J. Am. Acad. Dermatol. 2014, 70, 591.e1–591.e14. [Google Scholar] [CrossRef] [PubMed]
- Cordero, I. Electrosurgical units—How they work and how to use them safely. Community Eye Health 2015, 28, 15–16. [Google Scholar]
- Vilos, G.A.; Rajakumar, C. Electrosurgical generators and monopolar and bipolar electrosurgery. J. Minim. Invas. Gynecol. 2013, 20, 279–287. [Google Scholar] [CrossRef]
- Vilos, G.A. Understanding and Practising Safe Electrosurgery in the Operating Room. J. Obstet. Gynaecol. Can. 2018, 40, 1337–1347. [Google Scholar] [CrossRef]
- Takahashi, Y.; Hakamada, K.; Morohashi, H.; Akasaka, H.; Ebihara, Y.; Oki, E.; Hirano, S.; Mori, M. Verification of delay time and image compression thresholds for telesurgery. Asian J. Endosc. Surg. 2023, 16, 255–261. [Google Scholar] [CrossRef]


| CCD | CMOS | |
|---|---|---|
| Image Quality | Excellent color representation Less light saturation Clear view in dark area | Compromised color representation More light saturation Dark areas less visible |
| Production Cost | 10 times higher than CMOS | Economical |
| Resolution | Maxes out at Full HD (2K) | Progressing from 2K to 8K |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yuh, W.-T.; Lee, Y.-S.; Jeon, J.-H.; Choi, I. Future of Endoscopic Spine Surgery: Insights from Cutting-Edge Technology in the Industrial Field. Bioengineering 2023, 10, 1363. https://doi.org/10.3390/bioengineering10121363
Yuh W-T, Lee Y-S, Jeon J-H, Choi I. Future of Endoscopic Spine Surgery: Insights from Cutting-Edge Technology in the Industrial Field. Bioengineering. 2023; 10(12):1363. https://doi.org/10.3390/bioengineering10121363
Chicago/Turabian StyleYuh, Woon-Tak, You-Sang Lee, Jong-Hyeok Jeon, and Il Choi. 2023. "Future of Endoscopic Spine Surgery: Insights from Cutting-Edge Technology in the Industrial Field" Bioengineering 10, no. 12: 1363. https://doi.org/10.3390/bioengineering10121363
APA StyleYuh, W.-T., Lee, Y.-S., Jeon, J.-H., & Choi, I. (2023). Future of Endoscopic Spine Surgery: Insights from Cutting-Edge Technology in the Industrial Field. Bioengineering, 10(12), 1363. https://doi.org/10.3390/bioengineering10121363