1. Introduction
Shallow aquifers represent water reservoirs for public and private drinking supplies, for irrigation and livestock, and for industrial uses. Therefore, information concerning the geologic nature of such aquifers, the sources and directions of their flows, and their interactions with surface running water is of prime importance. In fact, interactions between sub-surface aquifers and surface run-off waters are often complex and depend on a variety of parameters, which include petrography of the host rocks, shape of landforms, type of climate, but also regional exploitation of the local resources [
1,
2,
3]. Hydrologic aspects and water quality of associated rivers are also strongly controlled by exchanges of water and solutes with the adjacent riverbanks, local uplands and nearby aquifers [
4,
5], not considering accidental or intentional pollution. The
18O/
16O and D/H ratios have been used routinely in hydrological studies [
6,
7,
8,
9], while Sr isotope signatures may very well complement the stable isotope analyses of aquifer and surface waters. The
87Sr/
86Sr signatures of waters may, for instance, be used to define their sources and contribution to the formation of large water reservoirs [
10]. One advantage of using the Sr isotope composition of water is that they are not altered by evaporation processes, such as the oxygen and hydrogen isotope signatures. A second advantage with the Sr isotope investigation is that Sr is present in water as a dissolved constituent and hence it is sensitive to mixing of waters of different masses. For the
87Sr/
86Sr ratio, the end members of a mixing yield most often different Sr concentrations and isotopic ratios. In fact, fluid/mineral interactions depend mainly on the type of alluvium host rocks, with limited impacts on the fluids for elements other than the dominant Ca, SO
4 and Cl of the solutes. In turn, Sr concentrations are not expected to behave conservatively in freshwater reservoirs when excessive evaporation leads to super-saturation with respect to minerals known for their Sr preference, such as celestite or strontianite [
11].
In this general context, the aim of the present study is to trace the hydraulic interconnections of lowland aquifers from northeastern Kansas (USA) with the run-off waters of a local creek and two major rivers, by combining elemental contents, oxygen, hydrogen and Sr isotopic compositions. The selected area is locally of some importance because it is located to the west of the Kansas Konza Prairie, which is a United States designated site for regular monitoring of ecological and environmental configurations on temporal scales. Our expectation is that the provided hydrological information may benefit the ongoing research efforts in the Kansas Konza Prairie. The area is framed by Summer Hill to the north and the Kansas River to the south with a lowland in between that is drained by a creek from hill to river at low-water level or is flooded by the river at high-water level, which potentially modifies the orientation of the flow paths of the local sub-surface water. As variations in the stable isotopic composition of such waters often result from potential mixing of different types of water, rather than from water-rock interactions, because of relatively short residence times of the flowing groundwater, the main goal is a comparison of potential changes of the water composition with the potential flow paths at varied depths in the host rocks. This evaluation was completed using the oxygen and hydrogen stable isotope compositions for identification of the water molecule, the 87Sr/86Sr ratio for tracing the origin(s) and potential interactions, and some dissolved chemical components of the river and aquifer waters.
2. The Study Area and the Collected Water Samples
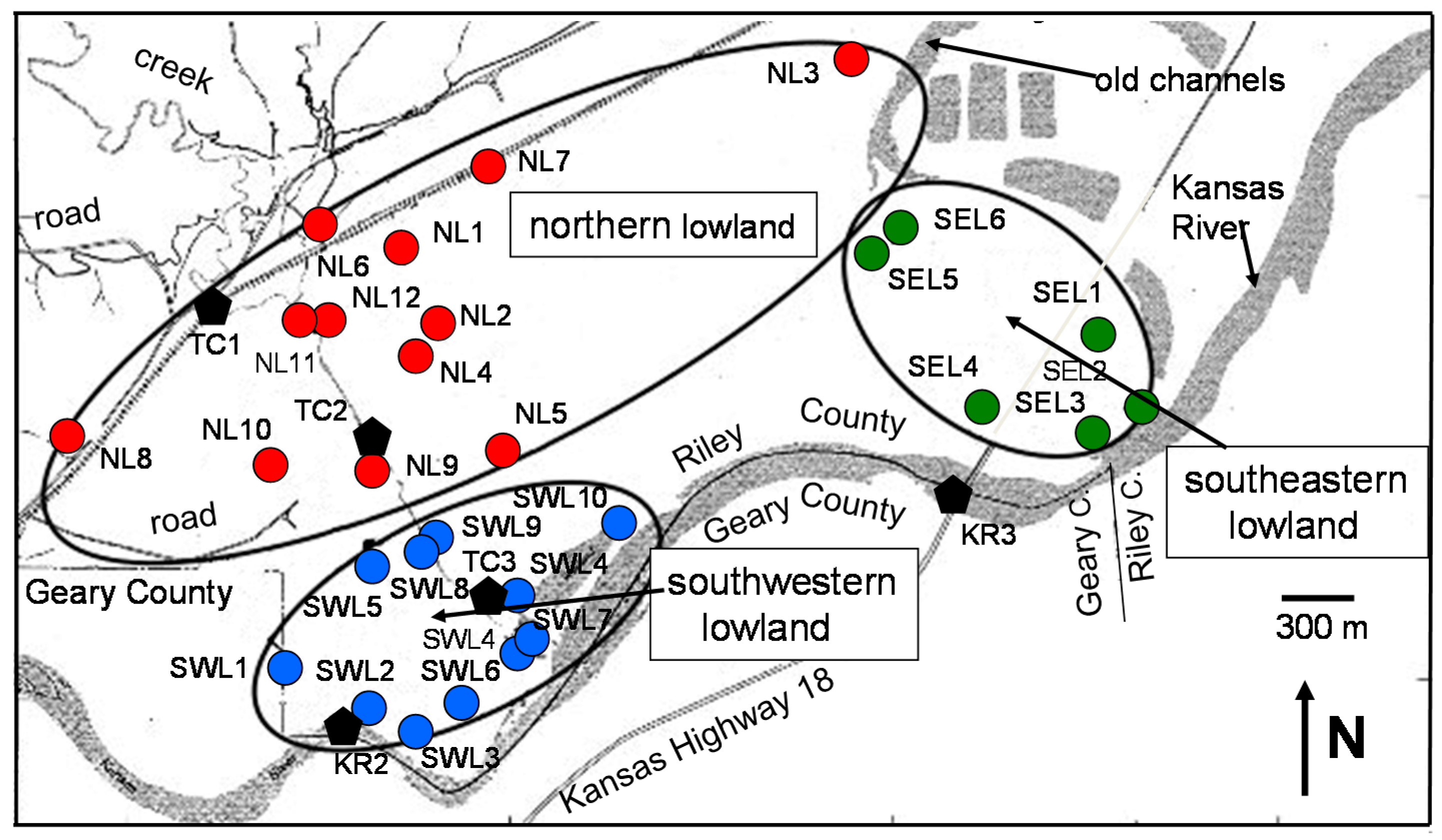
The collection site is a lowland area of about 5.7 km
2 located about 17 km to the WSW of the city of Manhattan (KS, USA) between Summer Hill and the Kansas River (39°07′24.19′′ N; 96°44′09.98′′ W;
Figure 1). It is a federal reservation at an altitude of 320 m, bordered to the north by a hill at a height of 388 m, and to the south by the river at a height of 315 m. The regional geology consists of a Permian limestone with interbedded cherts and shales [
12,
13]. The overlaying alluvium deposits derive essentially from these rocks, consisting of a 7–17 m thick sequence of gravel, coarse-to-fine sand, silt and clay layers. Lateral and vertical changes were reported in the coarser alluvial sediments away from river channels that result from deposition by ancestral river meanders in the valley, as shown by still visible lagoons and old channels. Analysis of water-level altitudes of wells and nearby water bodies has shown that groundwater moves generally ENE in the downstream direction, and towards the Smoky Hill River in the near vicinity [
14], which is compatible with the regional direction of groundwater flow in the alluvium. However, this direction reverses temporarily during high river stages. Specifically, Fader [
15] showed that the direction of the groundwater flow may be predominantly away from Kansas River during high river stages, and that the reversal flow may extend to more than 1 km away from the river.
As vegetation might have potentially a noticeable influence on the solute contents and isotope compositions of both run-off and shallow ground waters, it is needed to present the vegetal environment of the study area predominantly consisting of grassy and forested lands. Evapo-transpiration by plants, tapping groundwater via plant roots could, for instance, significantly influence the stable isotope compositions of the associated groundwater. Furthermore, decomposition of litter falls with release of Cl, sulfates from S in plant tissues oxidizing to sulfate may also influence the chemical composition of the waters, for instance their Sr and Ca contents. Chaudhuri et al. [
16] claimed that the chemistry of river water is greatly influenced by plant matter decomposition. Grigal and Ohmann [
17] and Bedel et al. [
18] showed that the deep aquifers represent a potential source for tree roots in a forest environment. Also, Clauer and Semhi [
19] modeled the interactions between plant roots and soil pore fluids, and between plant leaves and atmospheric supplies on the basis of their respective
87Sr/
86Sr ratios and Sr contents.
Forty-two groundwater samples were collected in three main areas of the lowland: in the northern area next to the Summer Hill, in the southeastern and the southwestern areas between the northern lowland and the Kansas River. Twelve of these groundwater samples were collected from wells drilled in the northern lowland (labeled NL hereafter), six were collected in the southeastern lowland (labeled SEL), and ten in the southwestern lowland (labeled SWL). The sites were installed to monitor the water quality of the shallow, at a depth of about 6.1 m (sub-labeled sw hereafter), and the deep alluvial deposits, about 18.3 m below surface (sub-labeled dw). For the other collected waters, the sampling depth is generally close to that of the shallow aquifers at about 6 m depth. To the best of our knowledge, there are no published reports describing specifically the hydrogeology or water quality of this land location. Some information is available from nearby Geary County Landfill located about 6 km to the SW [
14]. Parker [
20] and Haworth [
21] summarized data on groundwater supplies in Kansas. Latta [
22] reported on groundwater quality along the Smoky Hill Valley in the Saline, Dickinson and Geary counties, to the SW of the location studied here. Fader [
15] reported on groundwater in the Kansas River valley from Junction City to Kansas City.
Seven surface-water samples were also collected from three sites of the Kansas River (labeled KR), from one site of the Republican River to the north of Junction City (labeled RR), and three sites of the Threemile creek (labeled TC). The bridge in Fairmont Park of downtown Manhattan, about 17 km to the ENE of the site (sample KR1), was one of the Kansas River sites, the two others being to the immediate south of the selected area (samples KR2 and KR3). The water sample from the Republican River was collected at the junction with the Kansas River, about 10 km to the SW of the lowland site (sample RR1). The three sampling sites along Threemile creek were from the crossing with the local railroad (sample TC1), at half way between the railroad crossing and the Kansas River (sample TC2) and at the outlet of the wetland area next to the Kansas River (TC3;
Table 1).
3. Analytical Procedure
The water samples were filtered at the collection sites using 0.45-μm filters, stored in pre-cleaned 500 and 1000-mL polypropylene bottles and acidified to a pH of about 2.0 with vacuum-distilled, ultra-pure HNO
3 for the chemical and Sr isotopic determinations. The pH and total alkalinity were measured on site. Reported in milli-equivalents per liter (meq/L), the alkalinity was determined by titration with H
2SO
4 acid to a pH of 4.5, following a method described by Barnes [
23]. For the oxygen and hydrogen isotope analyses, the water samples were stored separately in 30 mL glass vials with no free headspace and thoroughly sealed against any significant loss of volatile organic compounds until analysis.
The Cl and SO4 contents were measured using a Dionex 2000-ion chromatograph. The uncertainty in the measurements was within ±10%. Concentrations of Sr and Ca were determined by atomic absorption spectrometry. Repeated measurements of several samples during the course of the study gave errors of ±10% for both the Sr and Ca determinations. The concentration of Cl, SO4, Sr and Ca are reported in milligrams per liter (mg/L).
Approximately 3–5 μg of Sr were separated from each water sample prior to Sr isotope analysis. Chemical separation of Sr followed a standard ion chromatography procedure, using a cleaned cation-exchange column with double-distilled 2N HCl as the eluant. Total blank Sr was less than 0.5 ng for the entire procedure including filtration, storage and chemical separation. After the column separation, about 1 μg of Sr was loaded onto a triple filament and analyzed for the 87Sr/86Sr ratio using a five-collector mass spectrometer. To compensate for any isotope fractionation during the isotope measurements, the measured 87Sr/86Sr ratios were normalized to a 86Sr/88Sr ratio of 0.11940. The internal precision of the 87Sr/86Sr ratio was close to 10 × 10−6 expressed in 2σ errors. The external reproducibility for the 87Sr/86Sr was tested by repeated analysis of the NBS 987 standard, which provided a mean ratio of 0.710227 ± 0.000017 (2σ of the mean with n = 70).
For the oxygen isotope analysis, approximately 6.0 mL of each water sample were separated with a syringe and reduced to 5.0 mL. Then, 7.0 mL of 99.9% CO
2 was introduced into the syringe through a rubber septum attached to a mercury manometer. After adjusting the CO
2 pressure in the syringe to 1 atmosphere, the syringe was removed from the manometer and the needle sealed by sticking it into a rubber stopper. The syringe was then gently shaken in a water bath maintained at 25 °C. At least 2 h later, the syringe was removed and the CO
2 used for equilibration in the syringe was injected into an evacuated part of the preparation line. The CO
2 was then introduced into the vacuum line by means of a three-way stopcock. After the CO
2 and water vapor were frozen out in the trap cooled with liquid nitrogen, the CO
2 was transferred into a sample bulb. The oxygen isotope ratio was measured on a stable gas mass spectrometer. The
18O value of the water was then derived from an equilibrium factor between the value of the CO
2 and that of the H
2O [
24].
The hydrogen isotope ratios of the samples were measured from H2 gas obtained by Zn reduction of the water. The reduction reaction took place in a bulb constructed from a high-vacuum stopcock. Approximately 100 mg of Zn turnings were placed in the reaction bulb and warmed under vacuum. The bulb was then filled with dry N under slight positive pressure, and the stopcock removed. A 1–2 μL water sample was transferred to the wall of the reaction bulb with a capillary tube. After closing the stopcock and chilling the bulb with liquid N, the bulb was re-evacuated and sealed. Reaction was allowed to occur when the bulb was inserted into an Al block maintained at 400 °C for 15 min. The H2 gas produced was measured directly on a stable isotope gas mass spectrometer.
As the hydrogen isotope ratio expressed by the ratio
2H to
1H, the oxygen isotope composition is expressed by the ratio
18O to
16O. The absolute abundance ratio of the heavy isotope to that of the light isotope being extremely small, any difference in the oxygen or hydrogen isotope composition is best described relative to a reference standard for the stable isotope compositions of water samples, which is here the Vienna Standard Mean Ocean Water (V-SMOW). Following the usual procedure, the oxygen or the hydrogen isotope composition of any water sample is expressed by its delta (δ) value given in per mil or parts per thousand as:
where R is the
18O/
16O ratio of the oxygen isotope composition or the D/H ratio of the hydrogen isotope composition. A negative δ value implies that the sample is depleted in the heavy isotope relative to the V-SMOW standard, whereas a positive δ value implies that the sample is enriched in the heavy isotope. The precision of the stable isotope compositions of the water samples analyzed here are ±0.3‰ for oxygen and ±5‰ for hydrogen at the 95% confidence limit.
5. Discussion
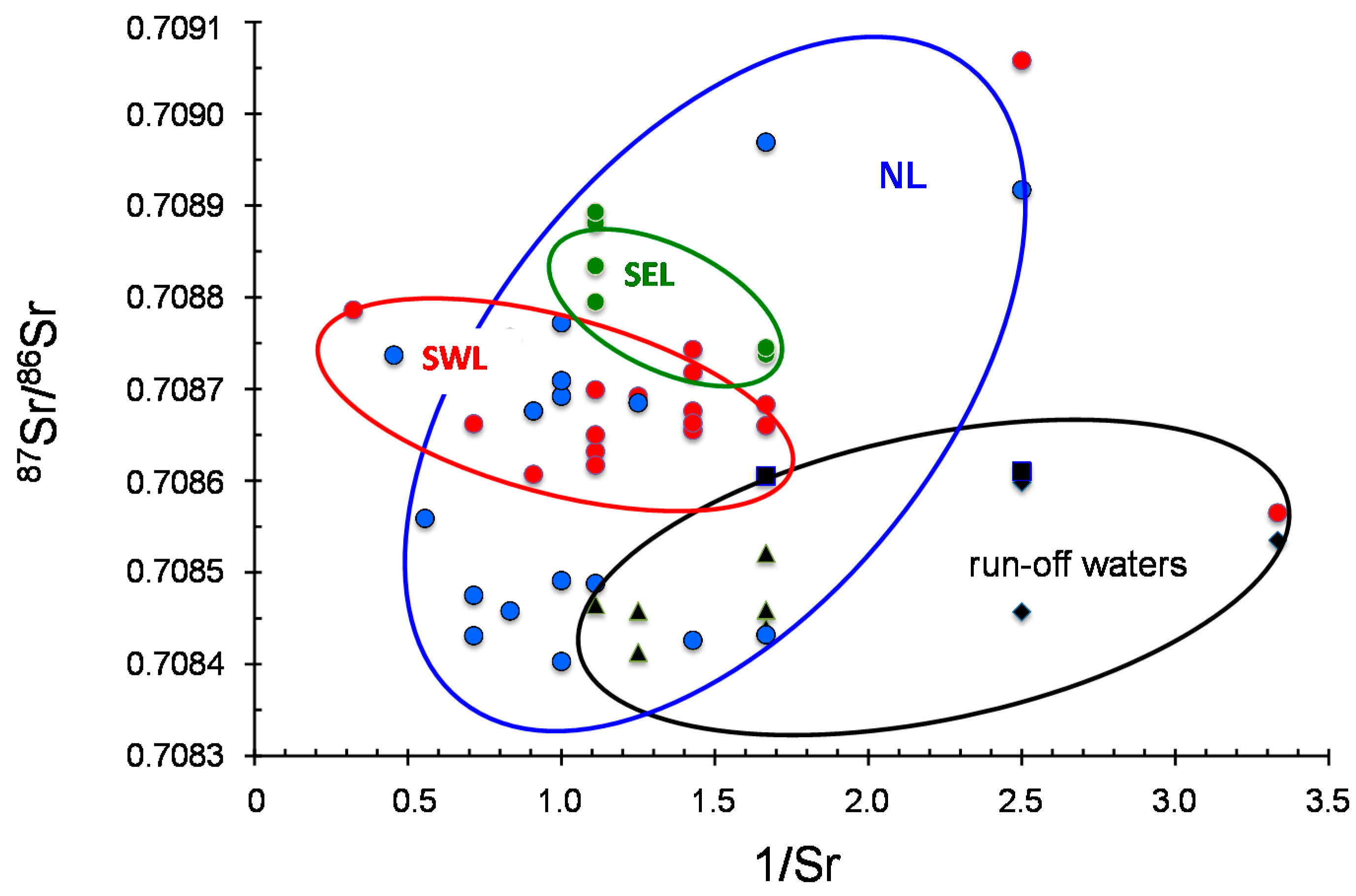
Because groundwater from alluvial deposits can potentially derive from varied surface sources, which may depend on seasonal differences, as well as from interaction with the mineral components of the soils and their vegetal cover, the Sr isotopic data was used here to seek evidence for such mixings and/or interactions. Theoretically, if two sources differing in their Sr isotopic composition and concentration are mixed in various proportion, the resultant mixtures are related linearly in their
87Sr/
86Sr and Sr contents or 1/Sr ratios. In the present case and whatever the selected lowland, no linear relation could be obtained in such a mixing diagram for the ground- and running waters (
Figure 7). In particular, the ground waters from southwestern area that is drained by two independent surface water systems of the Kansas River and the Threemile Creek, do not exhibit any discernible linear trend. This lack of correlation suggests that the Sr isotopic composition of the local ground waters cannot simply result from a mixing of local ground waters with running waters of the rivers and/or of the creek.
Also of interest is the fact that the ground waters sampled the farthest away from the run-off waters, that is to say those of the northern area at the foot of Summer Hill (NL3, NL6 and NL7) yield
87Sr/
86Sr that are exactly within the range of the run-off waters. A linear correlation for these samples in the
87Sr/
86Sr vs. 1/Sr diagram (
Figure 7) suggests that some of the NL ground waters descending from nearby hills into the lowland are running off, while others, even close to Threemile creek (NL8) are of another origin.
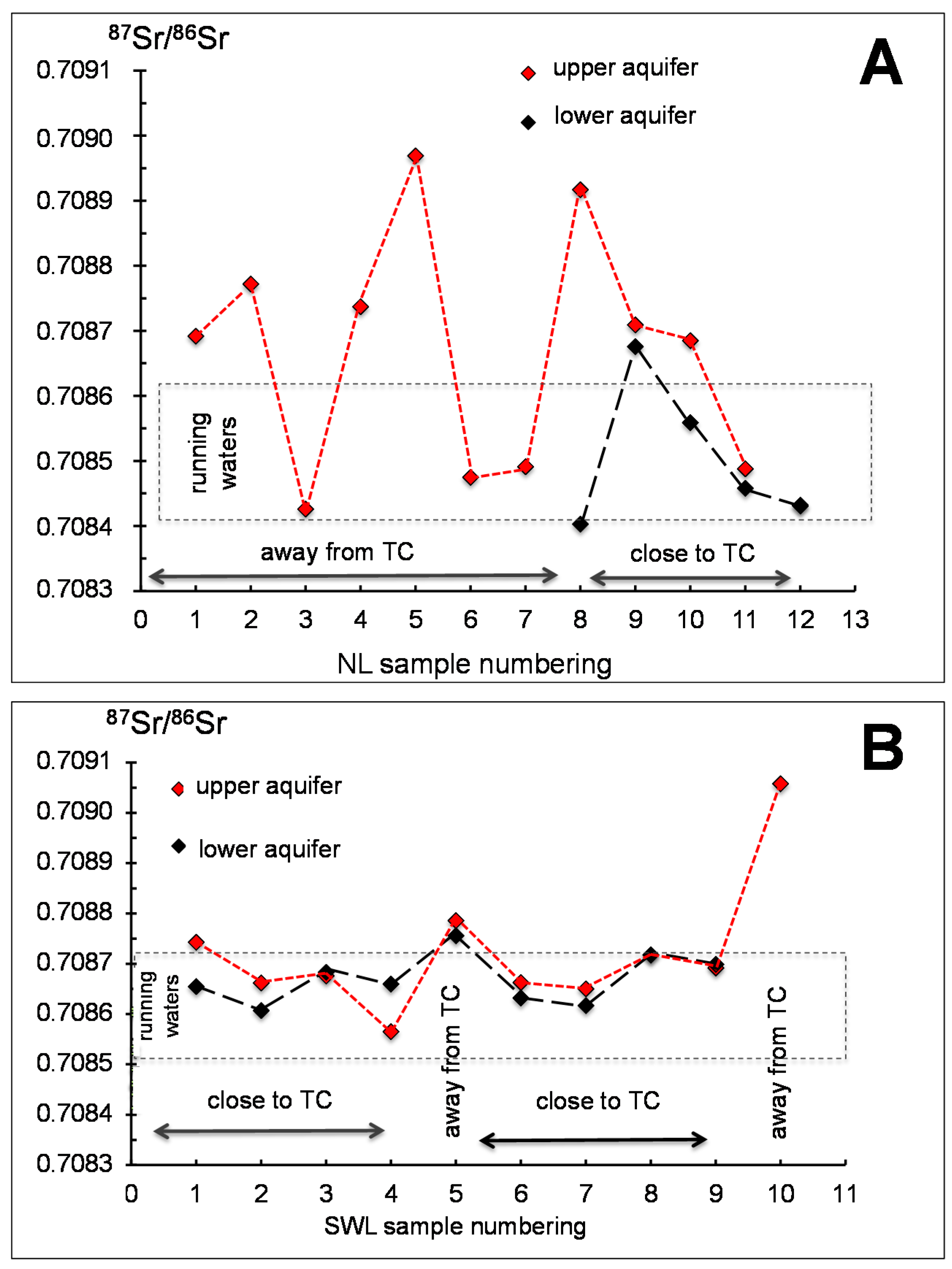
A straight comparison of the
87Sr/
86Sr ratios from ground waters collected at varied depths in potentiometric holes potentially provides some indication of a possible mixing. This aspect could be investigated here in ground waters from southwestern and northern lands by comparing the deep and shallow waters (
Figure 8A,B). In the northern land, if considering only those wells for which the
87Sr/
86Sr ratio is available for the upper and lower aquifers (samples NL8 to NL12), two different correlation trends are obtained: in wells NL9, NL11, NL12, the difference between the two Sr isotopic ratios of the waters from the superposed aquifers is low to nil. Alternatively, the difference is significant in holes NL8 and NL10, with the largest in NL8 that is to the west of the lowland away from run-off waters. In summary, only a small difference is noticeable between shallow and deep samples next to the creek, whereas it becomes significant away from running waters (
Figure 8A). Furthermore, close to the river the Sr isotopic composition of the aquifers is identical to that of the creek waters. It looks like the two aquifers evolve independently away from running waters and that they mix when collected closer to running waters.
This scenario is not exactly reproduced in the southwestern lowland, even if an analytical difference between the
87Sr/
86Sr ratios of the shallow and deep aquifers is confirmed in the wells SWL1 and SWL2 that are away from running waters (
Figure 8B). Identical ratios were also confirmed for the upper and lower aquifers at the sites close to the running waters (SWL6, SWL7, SWL8, SWL9), while it is not the case for the SWL4 well that is the sole sample in which the
87Sr/
86Sr ratio of the upper aquifer is lower than that of the deeper aquifer. In this case, it could be that the upper aquifer had some connection with nearby running waters, with no mixing occurring for the lower aquifer. Also, the highly similar
87Sr/
86Sr ratios for the two aquifers were obtained in a potentiometric well away from any running waters (SWL5). In summary, it is clear from these preliminary observations, that there is no unique interpretation for the observed variations of the
87Sr/
86Sr ratio of the shallow and deep aquifers.
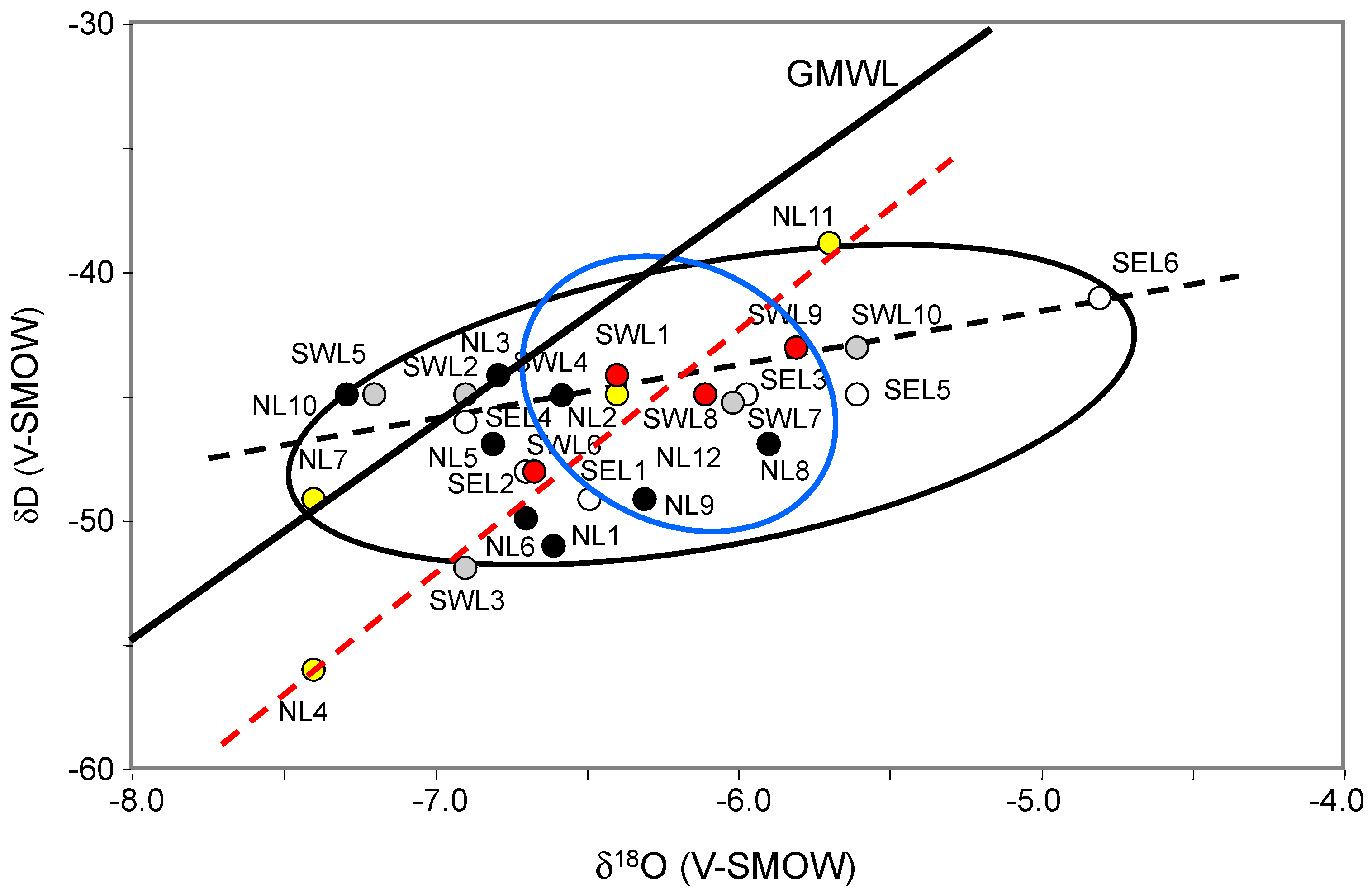
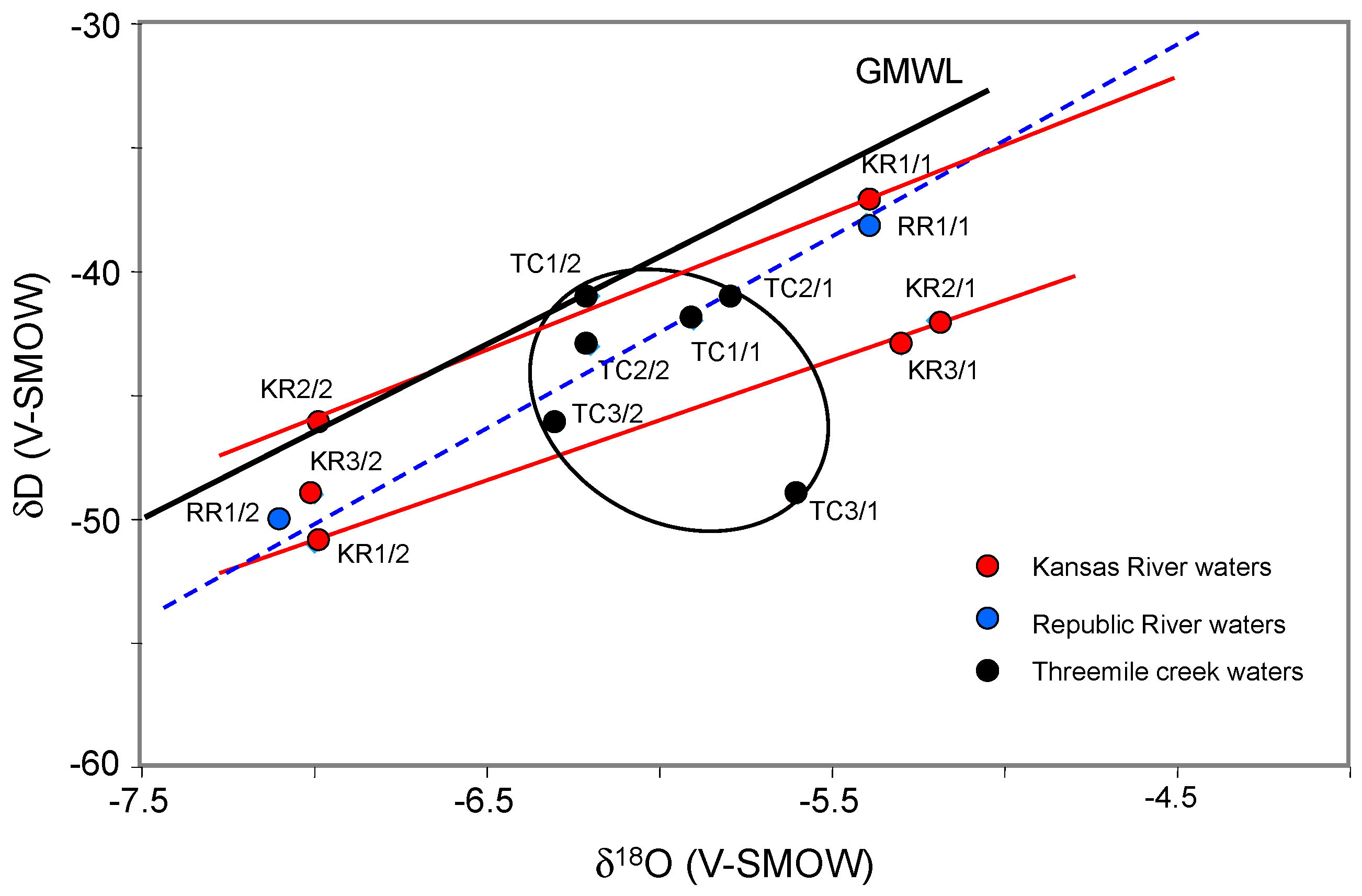
The oxygen and hydrogen isotope compositions of the analyzed waters were also compared in the search of mixing between aquifer and running waters. The fractionation coefficients between δ
18O and δD of waters from the two rivers and the creek, and of waters from local shallow aquifers range narrowly between −4.7 and −7.4‰ for δ
18O and between −39 and −52‰ for δD (
Figure 4). In fact, the poor coefficients for the δD-δ
18O correlations suggest that the isotopic values could have resulted from: (1) the combined effects of varied local interactions between waters and host alluvial deposits; (2) the varied sources of the waters; (3) different degrees of evaporation of the waters during their residence in the alluvial deposits. Interactions between the ground waters, even at a depth of about 6 m and the vegetal cover of the lowland exist, especially in forested areas [
17,
18]. Some ground waters of the southwestern area yields δ
18O values from −6.9 to −7.3‰, similar to the δ
18O values of the run-off waters from the Kansas River. The other ground waters in the southwestern area yield δ
18O values that are rather close to the values of −5.2‰ obtained for waters of the Republican River. The observed δD-δ
18O trends for both the running and aquifer waters cannot relate to an evaporation-transpiration process, as both display very similar correlation coefficients, which also precludes a significant interaction of the aquifer waters with their host rocks. Conversely, hydraulic disconnections between shallow ground waters and surficial run-off waters theoretically characterize a forested basin, like here, because vegetation has varied controls on the chemical make-up of both, due to a markedly deeper impact of tree roots on the host soils and their aquifers [
18].
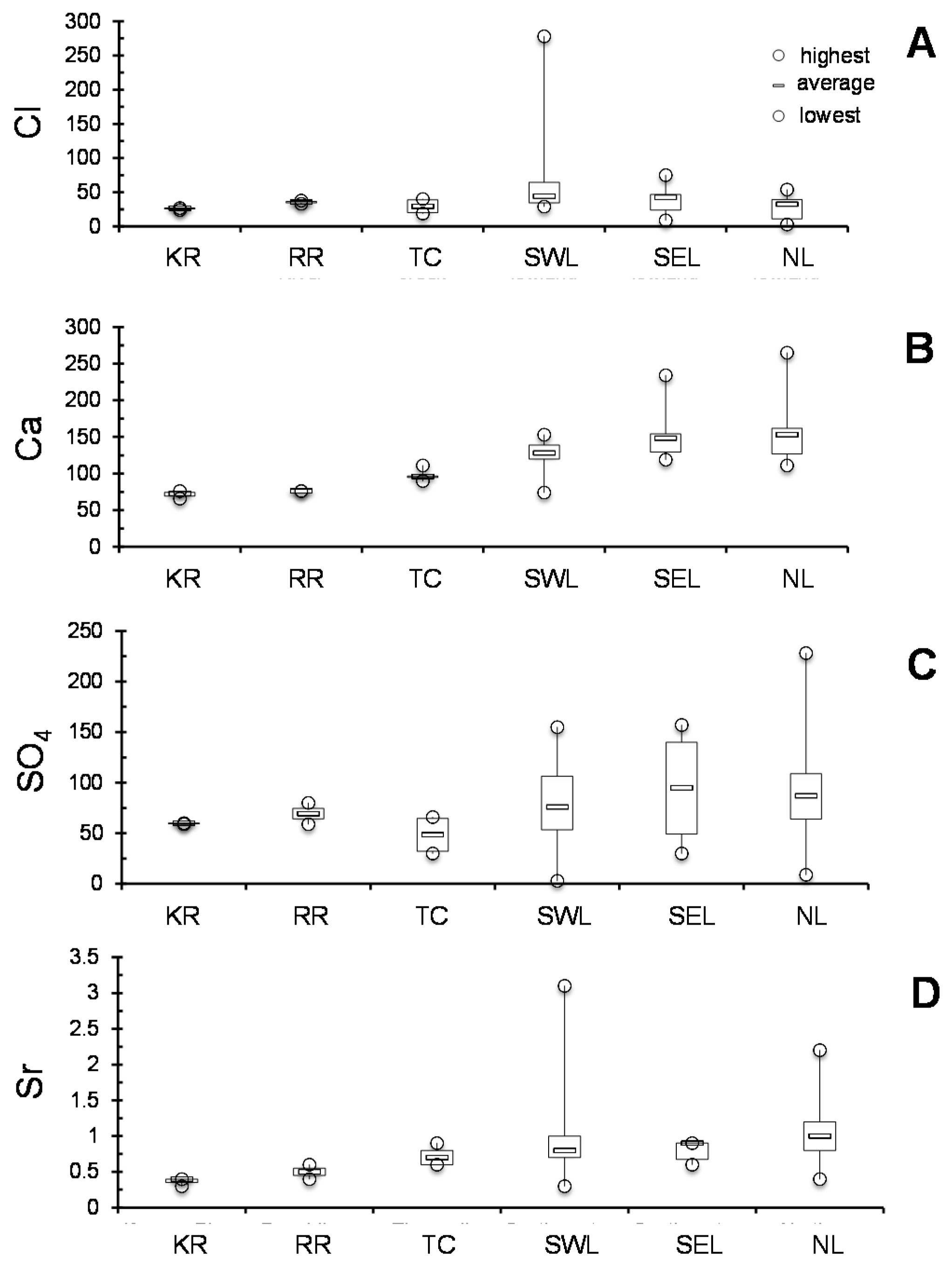
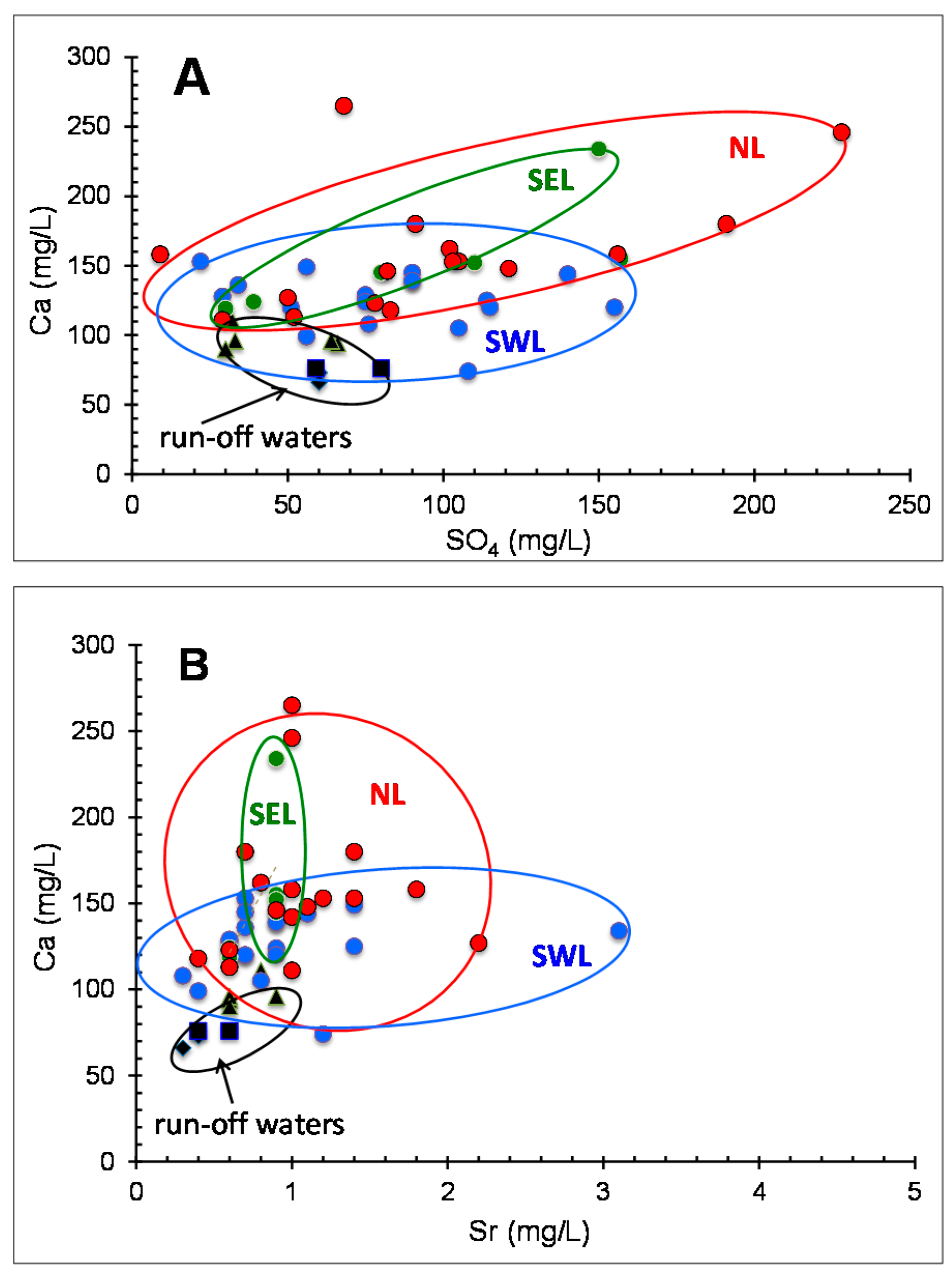
In summary, as no unique interpretation is evident, a few scenarios have been explored below, including: (1) origin, interaction and chemical loads of the aquifer and run-off waters, and (2) which hydraulic implications can be expected from chemical data. It might also be kept in mind what could not be evaluated here, that the generally poor correlations obtained between Ca and SO
4, Ca and Na, Ca and Mg and Ca and Sr in the aquifer waters of each area could also result from varied plant-microbe interactions. For instance, the scatter in the Ca/Sr ratio may arise from varied values in different plant organs, which might induce varied ratios in the percolating aquifers and in creek running-off waters. Such scatters have already been discussed on the basis of the K content of running waters as a major contributor to the solutes of a global river [
16].
5.1. Distinction between Origin and Interaction of the Aquifer Waters
Three transects based on the 87Sr/86Sr ratio relative to sample location, Sr contents and Sr/Ca ratios of the aquifers were detailed more fully to evaluate a potential distinction between the origin of the aquifer waters and their potential interactions with the host rocks.
5.1.1. Along the N-S Transect near Threemile Creek
As the shallow aquifer waters of the different wells were susceptible to interaction with the creek and river waters when the wells are located next to the banks, a transect of potentiometric wells along the Threemile creek was completed with the data of the nearby creek waters at the northern and southern end of the transect. The upper ground waters from a north-to-south transect along the Threemile creek include the data of the TC1, NL11, NL12, TC2, NL9, SWL8, SWL9, TC3, SWL4, SWL6 and SWL7 sites and the KR2 running waters (
Figure 1). The
87Sr/
86Sr values provide an informative data plot (
Figure 9). The two northernmost sites located at the edge of Summer Hill consist of ground waters with
87Sr/
86Sr ratios that are in the range of the running waters from nearby Threemile creek. Further south starting with the waters of site NL9, the
87Sr/
86Sr ratios are systematically higher than those of the nearby creek waters, and those of the nearby Kansas River at the southern end of the transect. In summary, the waters of most sites drilled along the Threemile creek are clearly disconnected from local run-off system, except at the northern end of the transect (NL11 and NL12) as discussed above, while the site SWL4 towards the south appears to be partly mixed with run-off waters, probably from the creek. The poor δD vs. δ
18O correlation does not support either a mixing with river waters, even in the SWL6 and SWL7 wells that were drilled next to it. In summary, the only interaction between aquifer waters in the wells from the N-S transect along the Threemile creek and running waters are suspected in the two northern NL11 and NL12 wells and partially in the southern SWL4 well, probably with creek waters rather than with Kansas River water.
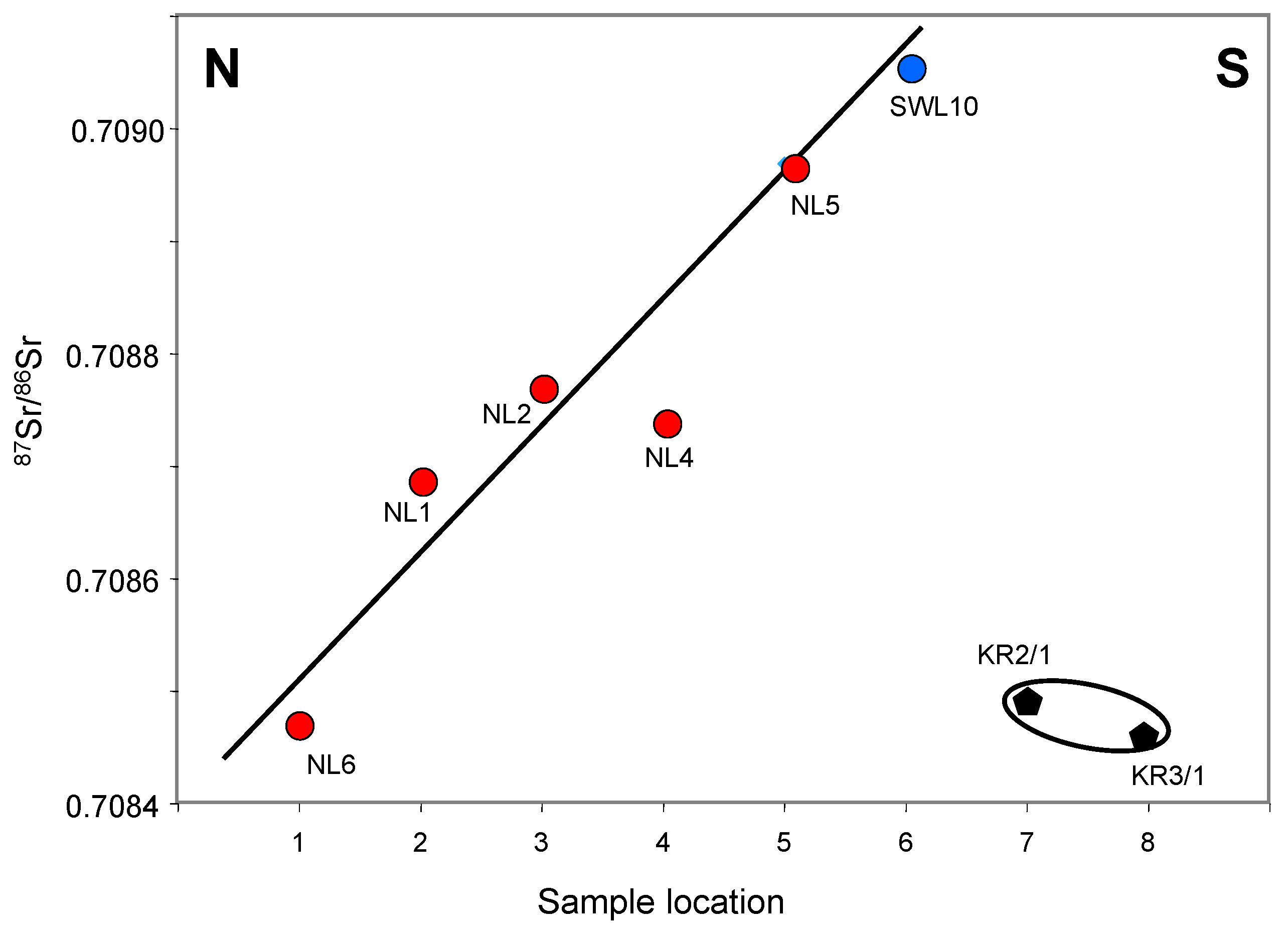
5.1.2. Along the Parallel N-S Transect
Also parallel to the Threemile creek, the second N-S transect extends about 1 km to the N-E of the previous one (
Figure 1). It starts at well NL6, the northern end at the outlet of Summer Hill and ends at well SWL10 close to an old meander channel of the Kansas River, including the NL6, NL1, NL2, NL4, NL5 and SWL10 sites. The
87Sr/
86Sr ratios of the ground waters of well SWL10 drilled the closest to the Kansas River are unexpectedly the farthest away from that of the Kansas River waters (
Figure 9), which confirms that no noticeable interaction exists between the shallow aquifer and the running waters of the creek or the river. This is supported by the relationship between the
87Sr/
86Sr ratios and the Sr contents: the waters collected the farthest away from Kansas River yield the
87Sr/
86Sr ratios and Sr contents that are the closest to those of the river waters and vice versa (
Table 1,
Figure 10). The same is apparent in the correlation between the
87Sr/
86Sr and Sr/Ca ratios, whereas the oxygen and hydrogen isotope compositions of the water components are close.
5.1.3. Along the E-W Transect in the Northern Lowland
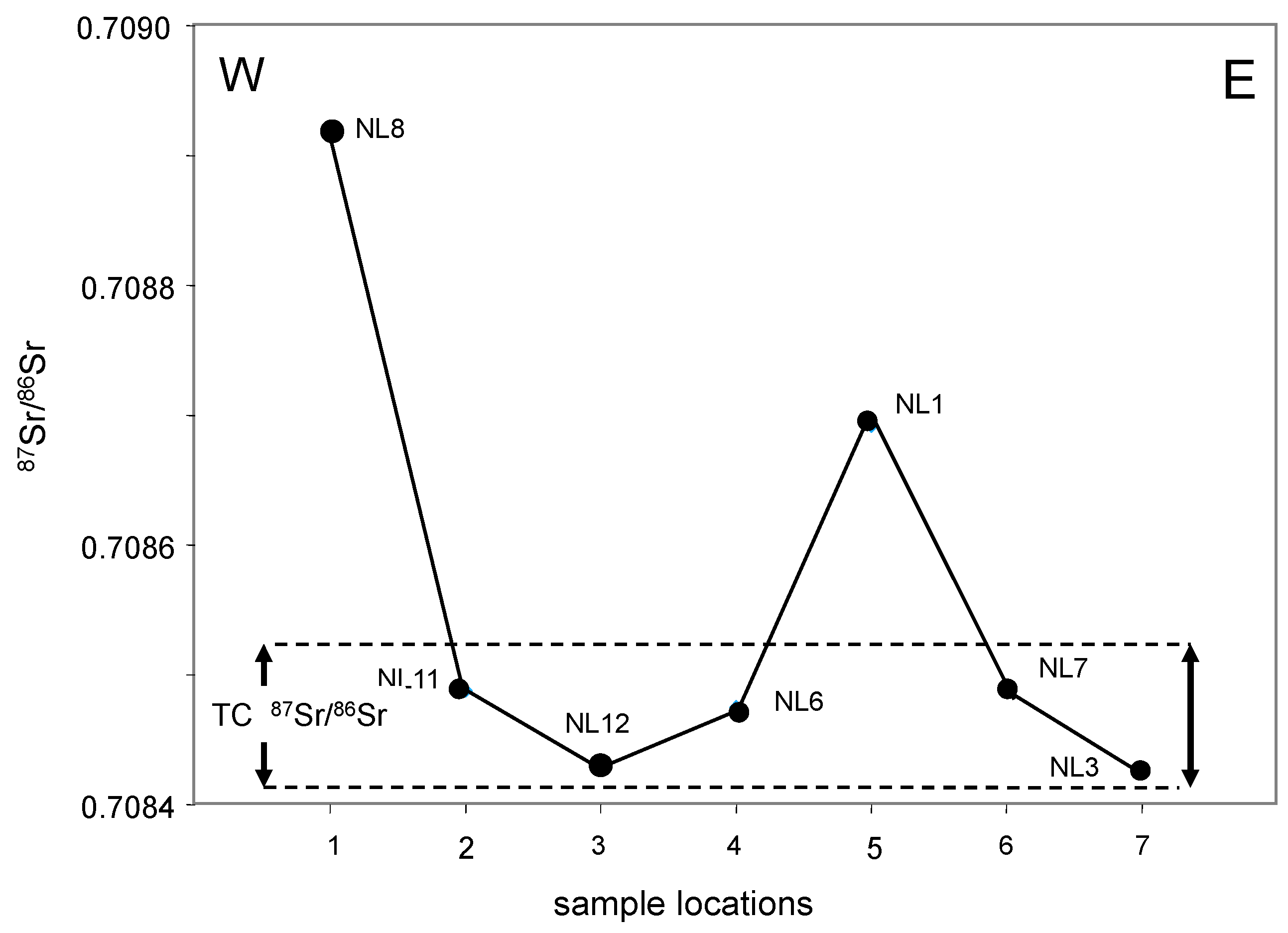
The E-W transect of the northern lowland parallels the southeastern limit along Summer Hill with a trend including the NL3, NL7, NL1, NL6, NL12, NL11 and NL8 sites. With the exception of the waters of the westernmost NL8 site and those of the intermediate NL1 site, all others contain Sr with a similar
87Sr/
86Sr ratio, suggesting a probable constant supply from nearby hills (
Figure 11). Interestingly, the
87Sr/
86Sr ratio of most of these aquifers is identical, within analytical uncertainty, to the waters of the Threemile creek. A logical interpretation is then that the E-W aquifer at the southern side of the Summer Hill is a major recharge aquifer of the nearby creek. These values are somehow in between the
87Sr/
86Sr ratio of the local rainwaters and the carbonate rocks below the alluvial level. A contribution of dissolved carbonate basement rocks by the rains seems, then, a reasonable interpretation for the chemical composition of the waters from this local reserve.
This interpretation is supported by the Sr contents and Sr/Ca ratios, while recalling that the potential supply of local plants/microbes have not been identified precisely here (
Table 1). The δD vs. δ
18O trend is close and parallel to that of the running waters, not pointing to extensive exchanges of waters from varied origins. In summary, the aquifer of the northern land seems to result from a significant supply from Summer Hill, except at its western border. No extensive interaction seems to have occurred between the shallow waters and the reservoir rocks.
5.2. Hydrologic Implications
Information to evaluate the hydrologic features of the land area between the hill and the river can be based on the chemical data of the shallow aquifer waters relative to their deeper counterparts, or on the geographic repartition of the
87Sr/
86Sr ratio of the upper aquifers. For instance, most
87Sr/
86Sr ratios of the upper and deeper ground waters of each drilling site are either identical or very narrow. Only those of the SWL1, SWL2, SWL4 and NL8 wells of the N-S transect along Threemile creek, suggest that there is either no significant chemical difference between the shallow and the deeper ground waters or a significant difference (
Figure 9).
5.2.1. In the Southwestern Area
The variations of the 87Sr/86Sr ratio of the aquifer waters from southwestern area are sufficient to identify sites where shallow and deep alluvial deposits do probably not belong to the same underground water flow-path. This seems to be the case for the SWL1, SWL2 and SWL4 sites. The idea of different flow paths in the shallow and deep alluvial deposits at each of these sites is supported further by the δ18O values, the Cl and SO4 concentrations and the Sr/Ca ratios.
Comparing the analyses of the Sr and oxygen isotope data from ground waters at different sites from southwestern area with those of waters from Kansas River and Threemile creek provides independent information about local groundwater flow paths and about potential sources for the ground waters. Different criteria also support changing flow paths for the shallow and deep alluvial deposits of site SWL1. Here, the shallow and deep ground waters differ not only in the 87Sr/86Sr ratios, but also in the δ18O values, as well as in the Cl and SO4 concentrations. Alternatively, uniform 87Sr/86Sr ratios of ground waters are not necessarily a sign of connected flow paths. Uniform flow paths need also to be identified by other geochemical evidence such as oxygen isotope data. That water flow paths in the shallow and deep alluvial deposits can still be different even though the 87Sr/86Sr ratios are nearly identical becomes apparent from oxygen and Sr isotopic compositions of the waters of site SWL5. Whereas waters from shallow and deep alluvial deposits were found to yield identical 87Sr/86Sr ratios, they differ markedly in δ18O values with the shallow waters having a value of −7.2‰ and the deep waters a value of −6.0‰. In fact, these two waters differ also in their Sr/Ca ratios, and Cl and SO4 concentrations. Like the shallow and deep waters of site SWL5, while the shallow and deep waters of site SWL6 differ from each other in their δ18O values and SO4 concentrations, but not in their Cl concentrations and 87Sr/86Sr ratios. Different groundwater flow paths in shallow and deep alluvial deposits at different sites could then reflect either different sources of water, different residence times of the water in the alluvium, or varied water/host rock interactions. A specific chemical supply of the vegetation, especially trees that occur here, to water of the upper aquifers is difficult to investigate in more detail here on the basis of the available data, but it cannot be ignored either.
In the southwestern area, the ground waters from SWL5, SWL6, SWL7, SWL8 and SWL9 sites, all located close to Threemile creek, yield markedly higher
87Sr/
86Sr ratios than the creek waters (
Figure 8B). This difference suggests that the ground waters from these sites have Sr supplies with high
87Sr/
86Sr ratios. This
87Sr-enriched source may also be the source with a δ
18O similar to the creek waters based on the fact that ground waters from the close NL9, SWL8 and SWL9 sites have oxygen isotope values similar to those of the creek waters. The SWL9 site also located near Threemile creek, just across the SWL8 site, contains groundwater with a similar
87Sr/
86Sr ratio, as well as a similar oxygen isotope value, Cl concentrations and Sr/Ca ratio. Remarkably, these two sites are separated by the creek, which waters differ considerably in their Sr isotopic signatures. Hence, the isotopic and chemical similarities among the ground waters of the two sites indicate that, either a hydrologic connection exists under the creek, or the alluvial deposits are nearly identical on each side of the creek, both receiving underground water from an identical source.
The 87Sr/86Sr ratios of the ground waters from SWL6 and SWL7 sites are lower and the δ18O values are more negative than those of the corresponding waters from SWL8 or SWL9 sites that are also located close to the creek in a similar environment. The high 87Sr/86Sr ratios for the two former sites may not result from supplies by the Kansas River or Threemile creek, because the surface waters have lower Sr isotopic ratios. Hence, the high 87Sr/86Sr ratios for the ground waters of the SWL6 and SWL7 sites necessitate Sr supplies not yet identified from a source similar to that of the ground waters of site SWL5.
5.2.2. In the Southeastern Area
The 87Sr/86Sr ratios of ground waters from SEL1 and SEL2 sites are analytically identical, which suggests that the two sites are probably connected hydraulically. Alternatively, the 87Sr/86Sr ratio of ground waters from SEL3 site being measurably lower than those from either the SEL1 or SEL2 sites suggests limited hydraulic connection between these two sites and the SEL3, SEL5 and SEL6 sites.
5.2.3. In the Northern Area
In the northern area, many groundwater samples yield 87Sr/86Sr ratios with values from 0.70841 to 0.70860 that range within those of the waters from Threemile creek and Kansas River. For instance, those from NL11 and NL12 sites closely resemble those of the Threemile creek water, suggesting that the ground waters of these sites derives largely from run-off creek waters, unless both have the same origin in the Summer Hill. The shallow and deep water samples of NL11 site also resemble Threemile creek waters in δ18O values, reinforcing the idea that waters from these sites essentially infiltrated from the creek. The slightly more negative δ18O value of waters from NL12 site implies that they mixed with infiltrated rainwater. Alternatively, the deep ground waters from NL10 site bears a close resemblance in its 87Sr/86Sr ratio and oxygen and hydrogen isotopic values with the Kansas River water, suggesting also that a connection exists between the river waters and those of NL10 site.
In general, the low Cl concentrations in most of the ground waters from intermediate northern area suggest a supply of rainwater by infiltration. Although δ18O values of rainwater in the region are not known, to the best of our knowledge, some unpublished data (S. Chaudhuri, written communication, 1997) suggest that a δ18O of −7 to −8‰ is not unlikely for the local rainwater, which may explain why the ground waters of this area are generally more negative in δ18O values than those from the southwestern area. For example, the Sr/Ca ratio in waters from shallow deposits of NL10 site is 0.005 and that from the deep alluvial deposits is two times higher at 0.011. At this site, the deep ground waters are also marked by Cl and SO4 concentrations that are much higher than those of the shallow ground waters, although both yield nearly identical oxygen isotope values.
In correlating the
87Sr/
86Sr ratio of all aquifer waters from the studied lowland, two sub-parallel areas oriented SE-NW consist of the NL3, NL6, NL7, NL11 and NL12 sites along Summer Hill and of the NL1, NL2, NL4, NL9, NL10, SEL5 and SEL6 sites (
Figure 10). The next integrating area is oriented E-W and includes the NL8, NL5, SWL10, SEL3, SEL4 sites, as well as the SEL1 and SEL2 sites, which have aquifer waters with
87Sr/
86Sr ratios slightly outside the range of those from neighboring aquifers. A fourth area can be added to those already identified, namely most sites in the SW area, which contains SWL4 site, with a different
87Sr/
86Sr ratio for the aquifer waters. The mean
87Sr/
86Sr ratio of the waters from this SW area extends towards that of the Kansas River waters and those of the second SE-NW area to the S of the area along Summer Hill.
In summary, the distribution of the 87Sr/86Sr ratios determined in the surficial aquifer waters into four areas inclines us to consider that the northernmost area reflects the supply from nearby Summer Hill that is also the reservoir of the running-off waters of Threemile creek. Farther south, the area is organized into two sub-parallel flows, also oriented roughly E-W. On top of this general E-W organization, the Threemile creek apparently represents a supplementary N-S trend towards the major Kansas River.
6. Conclusions
The present study was designed to trace hydraulic connections of local lowland aquifers with the running waters of a local creek and two major rivers in northeastern Kansas, USA, on the basis of a combined evaluation of elemental contents, and oxygen, hydrogen and Sr isotopic compositions. The area was selected on purpose because it is closely located near the Kansas Konza Prairie, which is a United States designated site for regular monitoring of ecological and environmental configurations. The δ18O and δD of the running waters from the two rivers and the creek, and of the waters from local shallow aquifers are almost identical. The observed trend for both water types cannot be related to a significant evaporation-transpiration process or to a varied vegetation supply, as both exhibit very similar correlation coefficients. It also precludes a significant interaction of the aquifer waters with the host rocks. Relative to the General Meteoric Water Line, the δ18O-δD data has a tendency to deviate towards relatively lower δ18O values, as already observed for sub-surface waters of varied intra-continental basins.
The 87Sr/86Sr ratio of the waters from wells drilled the closest to the Kansas River is the farthest away from 87Sr/86Sr ratio of the river waters, confirming that there is no noticeable interaction between aquifer and run-off waters. This is supported by the relationship between the 87Sr/86Sr ratio and Sr content: the waters collected the farthest away from Kansas River yield the 87Sr/86Sr ratios and Sr contents that are the closest to those of the Kansas River and vice versa. The same is evident in the correlation between the 87Sr/86Sr and Sr/Ca ratios. A clear interaction with the vegetation cannot be claimed either.
An underground water supply from Summer Hill has also been detected, except for its western end where no extensive interactions seem to have occurred between aquifers and reservoir rocks, or with the creek waters. The 87Sr/86Sr signatures of the ground waters suggest also the existence of roughly E-W flow systems in most of the study area. A further north-south trend occurs along Threemile creek towards the Kansas River.